Design and Evaluation of pH-Dependent Nanosystems Based on Cellulose Acetate Phthalate, Nanoparticles Loaded with Chlorhexidine for Periodontal Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Nanospheres and Nanocapsules by the Emulsification Diffusion Technique
2.3. Physicochemical Characterization of the Cellulose Acetate Phthalate (CAP)-Nanospheres (NSphs)/CAP-Nanocapsules (NCs)
2.3.1. Particle Size Measurement and Surface Charge
2.3.2. Scanning Electron Microscopy
2.3.3. Stabilizer Quantification
2.3.4. Determination of Encapsulation Efficiency and Chlorhexidine Loading Capacity
2.3.5. Quantitative Determination of the Chlorhexidine Base
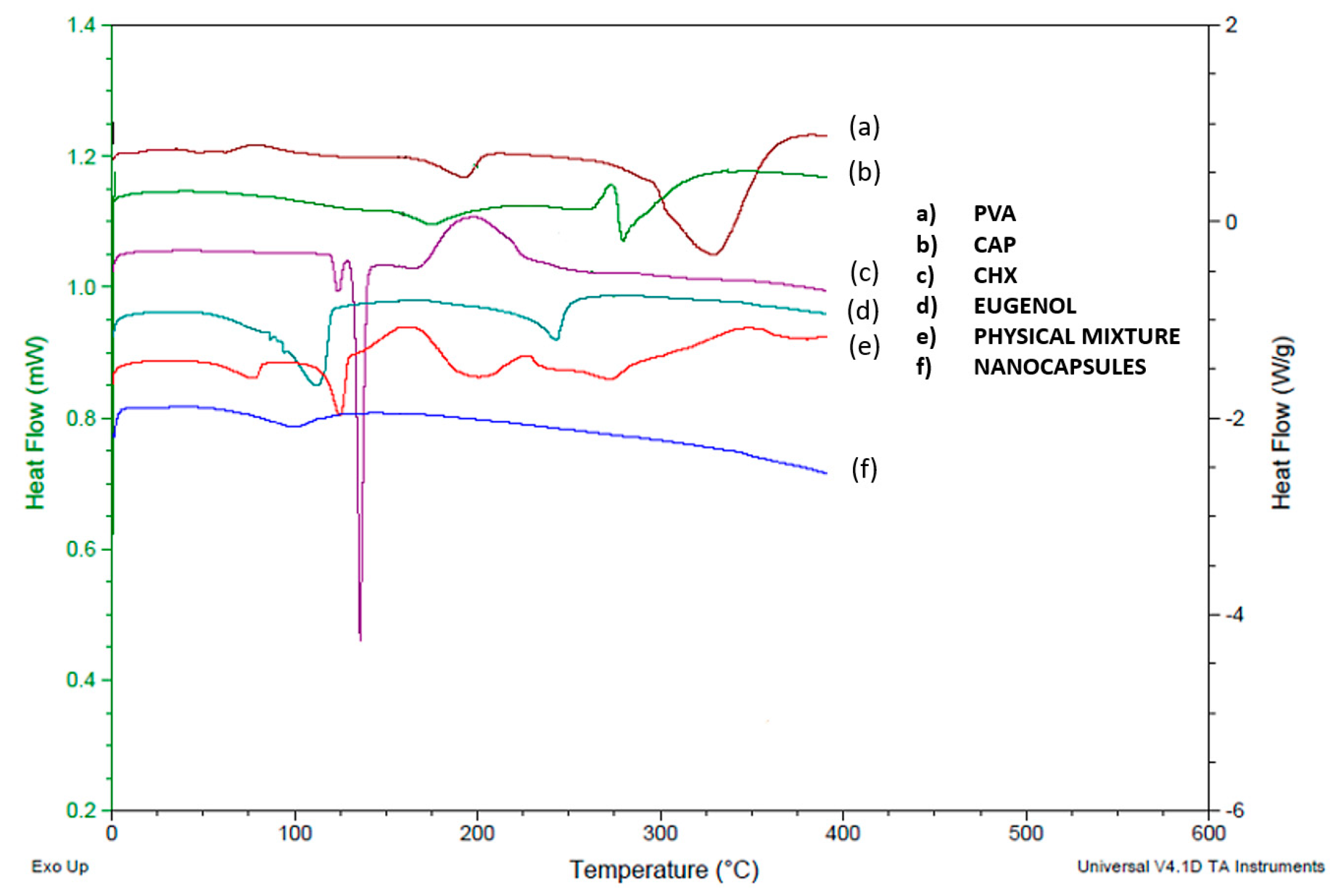
2.3.6. Differential Scanning Calorimetry (DSC)
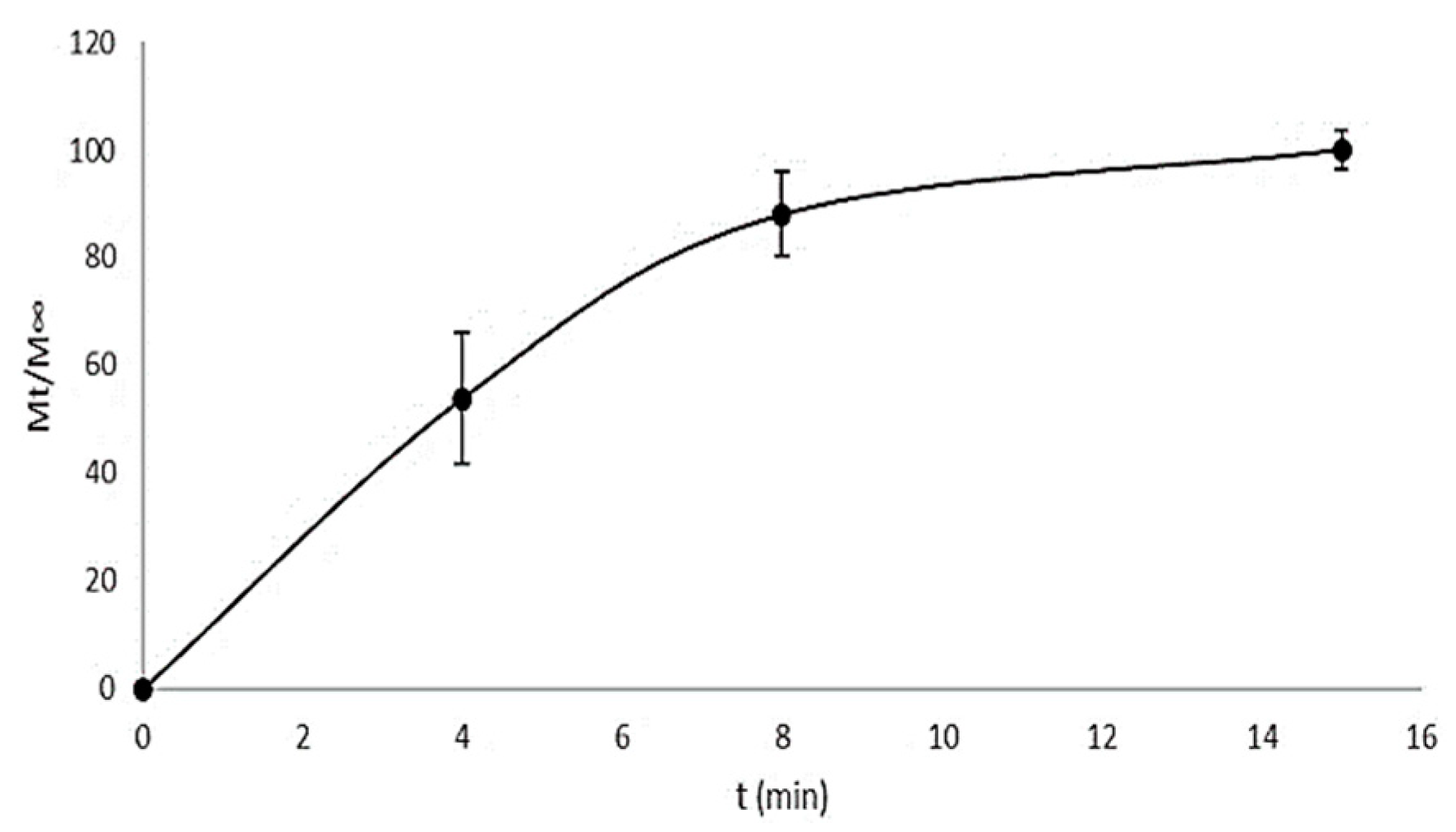
2.4. In Vitro Drug Release Study
2.5. Statistical Analyses
2.6. Panel Test
3. Results and Discussion
3.1. Physicochemical Characterization of the CAP-NSphs/CAP-NCs
3.2. In Vitro Drug Release Study
3.3. Panel Test
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Petersen, P.E.; Ogawa, H. The global burden of periodontal disease: Towards integration with chronic disease prevention and control. Periodontology 2000 2012, 60, 15–39. [Google Scholar] [CrossRef] [PubMed]
- Hatipoglu, H.; Yaylak, F.; Gungor, Y. A brief review on the periodontal health in metabolic syndrome patients. Diabetes Metab. Syndr. Clin. Res. Rev. 2015, 9, 124–126. [Google Scholar] [CrossRef] [PubMed]
- Ouderaa, F.J.G. Anti-plaque agents. Rationale and prospects for prevention of gingivitis and periodontal disease. J. Clin. Periodontol. 1991, 18, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Harvey, J.D. Periodontal Microbiology. Dent. Clin. N. Am. 2017, 61, 253–269. [Google Scholar] [CrossRef]
- Yue, I.C.; Poff, J.; Cortés, M.E.; Sinisterra, R.D.; Faris, C.B.; Hildgen, P.; Langer, R.; Shastri, V.P. A novel polymeric chlorhexidine delivery device for the treatment of periodontal disease. Biomaterials 2004, 25, 3743–3750. [Google Scholar] [CrossRef]
- Kopytynska-Kasperczyk, A.; Dobrzynski, P.; Pastusiak, M.; Jarzabek, B.; Prochwicz, W. Local delivery system of doxycycline hyclate based on -caprolactone copolymers for periodontitis treatment. Int. J. Pharm. 2015, 491, 335–344. [Google Scholar] [CrossRef]
- Jain, N.; Jain, G.K.; Javed, S.; Iqbal, Z.; Talegaonkar, S.; Ahmad, F.J.; Khar, R.K. Recent approaches for the treatment of periodontitis. Drug Discov. Today 2008, 13, 932–943. [Google Scholar] [CrossRef]
- Huynh, T.T.N.; Padois, K.; Sonvico, F.; Rossi, A.; Zani, F.; Pirot, F.; Doury, J.; Falson, F. Characterization of a polyurethane-based controlled release system for local delivery of chlorhexidine diacetate. Eur. J. Pharm. Biopharm. 2010, 74, 255–264. [Google Scholar] [CrossRef]
- Zeng, P.; Zhang, G.; Rao, A.; Bowles, W.; Wiedmann, T.S. Concentration dependent aggregation properties of chlorhexidine salts. Int. J. Pharm. 2009, 367, 73–78. [Google Scholar] [CrossRef]
- Lee, D.Y.; Spångberg, L.S.W.; Bok, Y.B.; Lee, C.Y.; Kum, K.Y. The sustaining effect of three polymers on the release of chlorhexidine from a controlled release drug device for root canal disinfection. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2005, 100, 105–111. [Google Scholar] [CrossRef]
- Lboutounne, H.; Chaulet, J.F.; Ploton, C.; Falson, F.; Pirot, F. Sustained ex vivo skin antiseptic activity of chlorhexidine in poly(ε-caprolactone) nanocapsule encapsulated form and as a digluconate. J. Control. Release 2002, 82, 319–334. [Google Scholar] [CrossRef]
- Matthijs, S.; Adriaens, P.A. Chlorhexidine varnishes: A review. J. Clin. Periodontol. 2002, 29, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Cavallari, C.; Brigidi, P.; Fini, A. Ex-vivoand in-vitro assessment of mucoadhesive patches containing the gel-forming polysaccharide psyllium for buccal delivery of chlorhexidine base. Int. J. Pharm. 2015, 496, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Onnainty, R.; Onida, B.; Páez, P.; Longhi, M.; Barresi, A.; Granero, G. Targeted chitosan-based bionanocomposites for controlled oral mucosal delivery of chlorhexidine. Int. J. Pharm. 2016, 509, 408–418. [Google Scholar] [CrossRef]
- Withers, N.J.; Senthil, A.; Osinski, M.; Nguyen, J.Q.; Alas, G.; Minetos, C.; Jaiswal, N.; Ivanov, S.A.; Smolyakov, G.A.; Huber, D.L. Effects of Iron-Oxide Nanoparticles on Compound Biofilms of Streptococcus Gordonii and Fusobacterium Nucleatum. In Proceedings of the Colloidal Nanoparticles for Biomedical Applications XIII, San Francisco, CA, USA, 27–29 January 2018; Liang, X.J., Parak, W.J., Osiński, M., Eds.; SPIE: Bellingham, WA, USA, 2018; Volume 10507, p. 17. [Google Scholar]
- Zupancic, S.; Kocbek, P.; Baumgartner, S.; Kristl, J. Contribution of Nanotechnology to Improved Treatment of Periodontal Disease. Curr. Pharm. Des. 2015, 21, 3257–3271. [Google Scholar] [CrossRef]
- Heinämäki, J.T.; Iraizoz Colarte, A.; Nordström, A.J.; Yliruusi, J.K. Comparative evaluation of ammoniated aqueous and organic-solvent-based cellulose ester enteric coating systems: A study on free films. Int. J. Pharm. 1994, 109, 9–16. [Google Scholar] [CrossRef]
- Edgar, K.J. Cellulose esters in drug delivery. Cellulose 2006, 14, 49–64. [Google Scholar] [CrossRef]
- Rowe, R.C.; Sheskey, P.J.; Owen, S.C.; American Pharmacists Association. Handbook of Pharmaceutical Excipients; APhA/Pharmaceutical Press: London, UK, 2009; ISBN 9781582121352. [Google Scholar]
- Shokri, J.; Adibki, K. Application of Cellulose and Cellulose Derivatives in Pharmaceutical Industries. In Cellulose—Medical, Pharmaceutical and Electronic Applications; van de Ven, T., Godbout, L., Eds.; InTechOpen: Rijeka, Croatia, 2013. [Google Scholar] [CrossRef]
- Vázquez, G.; Castro, T.; Hernández, A.; Castro, J.; De Lara, R. Comparación del efecto anestésico del aceite de clavo, solución salina y solución coloidal en juveniles de Chirostoma jordani (Woolman, 1894). Arch. Med. Vet. 2013, 45, 59–66. [Google Scholar] [CrossRef]
- Shahabadi, N.; Akbari, A.; Karampour, F.; Falsafi, M. Cytotoxicity and antibacterial activities of new chemically synthesized magnetic nanoparticles containing eugenol. J. Drug Deliv. Sci. Technol. 2019, 49, 113–122. [Google Scholar] [CrossRef]
- Shao, Y.; Wu, C.; Wu, T.; Li, Y.; Chen, S.; Yuan, C.; Hu, Y. Eugenol-chitosan nanoemulsions by ultrasound-mediated emulsification: Formulation, characterization and antimicrobial activity. Carbohydr. Polym. 2018, 193, 144–152. [Google Scholar] [CrossRef]
- Park, C.K.; Kim, K.; Jung, S.J.; Kim, M.J.; Ahn, D.K.; Hong, S.D.; Kim, J.S.; Oh, S.B. Molecular mechanism for local anesthetic action of eugenol in the rat trigeminal system. Pain 2009, 144, 84–94. [Google Scholar] [CrossRef] [PubMed]
- Parodi, T.V.; Cunha, M.A.; Heldwein, C.G.; De Souza, D.M.; Martins, Á.C.; Garcia, L.D.O.; Junior, W.W.; Monserrat, J.M.; Schmidt, D.; Caron, B.O.; et al. The anesthetic efficacy of eugenol and the essential oils of Lippia alba and Aloysia triphylla in post-larvae and sub-adults of Litopenaeus vannamei (Crustacea, Penaeidae). Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2012, 155, 462–468. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, C.P.B.; da Paixão Lemos, C.H.; Felix e Silva, A.; de Souza, S.A.; Albinati, A.C.L.; Lima, A.O.; Copatti, C.E. Use of eugenol for the anaesthesia and transportation of freshwater angelfish (Pterophyllum scalare). Aquaculture 2019, 513, 734409. [Google Scholar] [CrossRef]
- Moon, S.E.; Kim, H.Y.; Cha, J.D. Synergistic effect between clove oil and its major compounds and antibiotics against oral bacteria. Arch. Oral Biol. 2011, 56, 907–916. [Google Scholar] [CrossRef]
- Hemaiswarya, S.; Doble, M. Synergistic interaction of eugenol with antibiotics against Gram negative bacteria. Phytomedicine 2009, 16, 997–1005. [Google Scholar] [CrossRef]
- Hashieh, I.A.; Pommel, L.; Camps, J. Concentration of eugenol apically released from zinc oxide-eugenol-based sealers. J. Endod. 1999, 25, 713–715. [Google Scholar] [CrossRef]
- Scholz, O.A.; Wolff, A.; Schumacher, A.; Giannola, L.I.; Campisi, G.; Ciach, T.; Velten, T. Drug delivery from the oral cavity: Focus on a novel mechatronic delivery device. Drug Discov. Today 2008, 13, 247–253. [Google Scholar] [CrossRef]
- Phaechamud, T.; Setthajindalert, O. Cholesterol in situ forming gel loaded with doxycycline hyclate for intra-periodontal pocket delivery. Eur. J. Pharm. Sci. 2017, 99, 258–265. [Google Scholar] [CrossRef]
- Piñón-Segundo, E.; Ganem-Quintanar, A.; Alonso-Pérez, V.; Quintanar-Guerrero, D. Preparation and characterization of triclosan nanoparticles for periodontal treatment. Int. J. Pharm. 2005, 294, 217–232. [Google Scholar] [CrossRef]
- Alvarez Echazú, M.I.; Olivetti, C.E.; Anesini, C.; Perez, C.J.; Alvarez, G.S.; Desimone, M.F. Development and evaluation of thymol-chitosan hydrogels with antimicrobial-antioxidant activity for oral local delivery. Mater. Sci. Eng. C 2017, 81, 588–596. [Google Scholar] [CrossRef]
- Quintanar-Guerrero, D.; Fessi, H.; Allémann, E.; Doelker, E. Influence of stabilizing agents and preparative variables on the formation of poly(D,L-lactic acid) nanoparticles by an emulsification-diffusion technique. Int. J. Pharm. 1996, 143, 133–141. [Google Scholar] [CrossRef]
- Allémann, E.E.; Doelker, R.G. Drug loaded poly(lactic acid)nanoparticles produced by a reversible salting-out process: Purification of an injectable dosage form. Eur. J. Pharm. Biopharm. 1993, 39, 13–18. [Google Scholar]
- Gjoseva, S.; Geskovski, N.; Sazdovska, S.D.; Popeski-Dimovski, R.; Petruševski, G.; Mladenovska, K.; Goracinova, K. Design and biological response of doxycycline loaded chitosan microparticles for periodontal disease treatment. Carbohydr. Polym. 2018, 186, 260–272. [Google Scholar] [CrossRef] [PubMed]
- Agullo, M.J.A.; Loscos, F.G. Periodoncia para el higienista dental Importancia del uso de índices en la práctica periodontal diaria del higienista dental. Periodoncia Y Osteointegración 2003, 13, 233–244. [Google Scholar]
- Michel, B.; Maribel, A. Plaque Control with O’Leary index, instructing the Bass brushing technique in pediatric patients postgraduate Pediatric Dentistry of the UAN. Educateconciencia 2015, 5, 106–118. [Google Scholar]
- Taleghani, F.; Rezvani, G.; Birjandi, M.; Valizadeh, M. Impact of green tea intake on clinical improvement in chronic periodontitis: A randomized clinical trial. J. Stomatol. Oral Maxillofac. Surg. 2018, 119, 365–368. [Google Scholar] [CrossRef] [PubMed]
- De Araújo Lopes, A.; Da Fonseca, F.N.; Rocha, T.M.; De Freitas, L.B.; Araújo, E.V.O.; Wong, D.V.T.; Lima Júnior, R.C.P.; Leal, L.K. Eugenol as a Promising Molecule for the Treatment of Dermatitis: Antioxidant and Anti-inflammatory Activities and Its Nanoformulation. Oxid. Med. Cell. Longev. 2018, 2018, 8194849. [Google Scholar] [CrossRef]
- Seneviratne, C.J.; Leung, K.C.F.; Wong, C.H.; Lee, S.F.; Li, X.; Leung, P.C.; Lau, C.B.S.; Wat, E.; Jin, L. Nanoparticle-Encapsulated Chlorhexidine against Oral Bacterial Biofilms. PLoS ONE 2014, 9, e103234. [Google Scholar] [CrossRef]
- Liakos, I.L.; Iordache, F.; Carzino, R.; Scarpellini, A.; Oneto, M.; Bianchini, P.; Grumezescu, A.M.; Holban, A.M. Cellulose acetate—Essential oil nanocapsules with antimicrobial activity for biomedical applications. Colloids Surf. B Biointerfaces 2018, 172, 471–479. [Google Scholar] [CrossRef]
- ASTM International. Standart test methods for zeta potential of colloids in water and waste water. In The Annual Book of ASTM Standarts; ASTM International: West Conshohocken, PA, USA, 1985; pp. 4182–4187. [Google Scholar]
- Quintanar-Guerrero, D.; Allémann, E.; Doelker, E.; Fessi, H. A mechanistic study of the formation of polymer nanoparticles by the emulsification-diffusion technique. Colloid Polym. Sci. 1997, 275, 640–647. [Google Scholar] [CrossRef]
- Li, M.; Rouaud, O.; Poncelet, D. Microencapsulation by solvent evaporation: State of the art for process engineering approaches. Int. J. Pharm. 2008, 363, 26–39. [Google Scholar] [CrossRef] [PubMed]
- Jesudasan, J.S.; Wahab, P.U.A.; Sekhar, M.R.M. Effectiveness of 0.2% chlorhexidine gel and a eugenol-based paste on postoperative alveolar osteitis in patients having third molars extracted: A randomised controlled clinical trial. Br. J. Oral Maxillofac. Surg. 2015, 53, 826–830. [Google Scholar] [CrossRef] [PubMed]
- Albano, M.; Alves, F.C.B.; Andrade, B.F.M.; Barbosa, L.N.; Pereira, A.F.M.; de Souza, M.D.L.R.; Rall, V.L.M.; Júnior, A.F. Antibacterial and anti-staphylococcal enterotoxin activities of phenolic compounds. Innov. Food Sci. Emerg. Technol. 2016, 38, 83–90. [Google Scholar] [CrossRef]
- Miladi, H.; Zmantar, T.; Kouidhi, B.; Al Qurashi, Y.M.A.; Bakhrouf, A.; Chaabouni, Y.; Mahdouani, K.; Chaieb, K. Synergistic effect of eugenol, carvacrol, thymol, p-cymene and γ-terpinene on inhibition of drug resistance and biofilm formation of oral bacteria. Microb. Pathog. 2017, 112, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Amidon, G.L.; Lennernäs, H.; Shah, V.P.; Crison, J.R. A Theoretical Basis for a Biopharmaceutic Drug Classification: The Correlation of in Vitro Drug Product Dissolution and in Vivo Bioavailability. Pharm. Res. 1995, 12, 413–420. [Google Scholar] [CrossRef]
- Campos, J.C.; Ferreira, D.C.; Lima, S.; Reis, S.; Costa, P.J. Swellable polymeric particles for the local delivery of budesonide in oral mucositis. Int. J. Pharm. 2019, 566, 126–140. [Google Scholar] [CrossRef]
- Russo, E.; Selmin, F.; Baldassari, S.; Gennari, C.G.M.; Caviglioli, G.; Cilurzo, F.; Minghetti, P.; Parodi, B. A focus on mucoadhesive polymers and their application in buccal dosage forms. J. Drug Deliv. Sci. Technol. 2016, 32, 113–125. [Google Scholar] [CrossRef]
- Vieira Colombo, A.P.; Magalhães, C.B.; Hartenbach, F.A.R.; Martins do Souto, R.; Maciel da Silva-Boghossian, C. Periodontal-disease-associated biofilm: A reservoir for pathogens of medical importance. Microb. Pathog. 2016, 94, 27–34. [Google Scholar] [CrossRef]
- Costa, P.; Sousa Lobo, J.M. Modeling and comparison of dissolution profiles. Eur. J. Pharm. Sci. 2001, 13, 123–133. [Google Scholar] [CrossRef]
- Ritger, P.L.; Peppas, N.A. A simple equation for description of solute release II. Fickian and anomalous release from swellable devices. J. Control. Release 1987, 5, 37–42. [Google Scholar] [CrossRef]
- Ganem-Quintanar, A. Ètude de la Permeàbilitè de la Muqueuse Orale: Èvaluation de Diffèrentes Approaches Pour Augmenter le Passage des Principes Actifs. Ph.D. Thesis, Universitè de Genève/Universitè Claude Bernard, Lyon, Genève, 1997. [Google Scholar]
- Rodrigues, I.F.; Machion, L.; Casati, M.Z.; Nociti, F.H.; Toledo, S.; Sallum, A.W.; Sallum, E.A. Clinical evaluation of the use of locally delivered chlorhexidine in periodontal maintenance therapy. J. Periodontol. 2007, 78, 624–628. [Google Scholar] [CrossRef] [PubMed]
- Kida, D.; Junka, A.; Sender-Janeczek, A.; Dus, I.; Dominik, M. Metronidazole-Loaded Porous Matrices for Local Periodontitis Treatment: In Vitro Evaluation and In Vivo Pilot Study. Appl. Sci. 2019, 9, 4545. [Google Scholar] [CrossRef]
- Beatriz, N.; Hernández, B.; Gabriela, M.; García, N.; Eric, M.; Rivera, G.; Ancona, M.B.; Lira, G.H.; Castellanos, G.L.; Belinda, L.; et al. Eficacia de la clorhexidina en el tratamiento de gingivitis: Tabletas o colutorio. Rev. Medi. UV 2012, 2, 6–12. [Google Scholar]
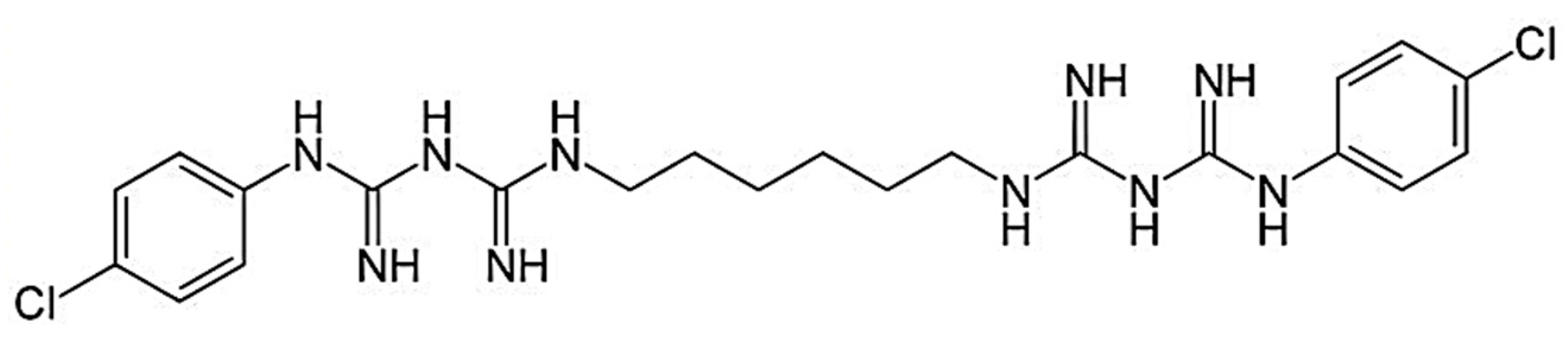



| Material | NCs | NSphs | ||
|---|---|---|---|---|
| Formulation A | Formulation B | Formulation C | Formulation | |
| Polymer (CAP) (mg) | 200 | 200 | 200 | 200 |
| Oil (eugenol) (mg) | 365 | 365 | 365 | – * |
| Drug (CHX) (mg) | 10 | 20 | 30 | 10 |
| External phase solvent (PVA 5%) (mL) | 40 | 40 | 40 | 40 |
| Inners phase solvent (MEC **) (mL) | 20 | 20 | 20 | 20 |
| Formulation | Amount of CHX (mg) | Size ± SD (nm) | PI * ± SD | Z-Potential ± SD (mV) | Process Efficiency ± SD (%) | Entrapment Efficiency ± SD (%) |
|---|---|---|---|---|---|---|
| Control | – | 180.6 ± 0.70 | – | −10.83 ± 0.77 | – | – |
| NCs | 10 | 290.65 ± 15.70 | 0.14 ± 0.02 | −20.16 ± 2.64 | 84.30 ± 0.74 | 61.93 ± 4.28 |
| 20 | 324.46 ± 55.46 | 0.238 ± 0.02 | −18.77 ± 3.46 | 72.00 ± 1.66 | 64.49 ± 0.80 | |
| 30 | 296.35 ± 39.00 | 0.291 ± 0.05 | −18.71 ± 5.21 | 69.25 ± 0.43 | 59.88 ± 2.77 | |
| NSphs | 10 | 247.60 ± 9.61 | 0.242 ± 0.01 | −20.35 ± 1.91 | 57.17 ± 1.23 | 77.36 ± 0.62 |
| Nanosystem | Korsmeyer-Peppas Model | Higuchi Model | ||
|---|---|---|---|---|
| r2 | n | r2 | KH (mg/min1/2) | |
| NSphs | 0.913 | 0.471 | 0.890 | 0.2402 |
| NCs | 0.9464 | 0.6083 | 0.9343 | 0.1313 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vidal-Romero, G.; Zambrano-Zaragoza, M.L.; Martínez-Acevedo, L.; Leyva-Gómez, G.; Mendoza-Elvira, S.E.; Quintanar-Guerrero, D. Design and Evaluation of pH-Dependent Nanosystems Based on Cellulose Acetate Phthalate, Nanoparticles Loaded with Chlorhexidine for Periodontal Treatment. Pharmaceutics 2019, 11, 604. https://doi.org/10.3390/pharmaceutics11110604
Vidal-Romero G, Zambrano-Zaragoza ML, Martínez-Acevedo L, Leyva-Gómez G, Mendoza-Elvira SE, Quintanar-Guerrero D. Design and Evaluation of pH-Dependent Nanosystems Based on Cellulose Acetate Phthalate, Nanoparticles Loaded with Chlorhexidine for Periodontal Treatment. Pharmaceutics. 2019; 11(11):604. https://doi.org/10.3390/pharmaceutics11110604
Chicago/Turabian StyleVidal-Romero, Gustavo, María L. Zambrano-Zaragoza, Lizbeth Martínez-Acevedo, Gerardo Leyva-Gómez, Susana E. Mendoza-Elvira, and David Quintanar-Guerrero. 2019. "Design and Evaluation of pH-Dependent Nanosystems Based on Cellulose Acetate Phthalate, Nanoparticles Loaded with Chlorhexidine for Periodontal Treatment" Pharmaceutics 11, no. 11: 604. https://doi.org/10.3390/pharmaceutics11110604
APA StyleVidal-Romero, G., Zambrano-Zaragoza, M. L., Martínez-Acevedo, L., Leyva-Gómez, G., Mendoza-Elvira, S. E., & Quintanar-Guerrero, D. (2019). Design and Evaluation of pH-Dependent Nanosystems Based on Cellulose Acetate Phthalate, Nanoparticles Loaded with Chlorhexidine for Periodontal Treatment. Pharmaceutics, 11(11), 604. https://doi.org/10.3390/pharmaceutics11110604







