Folate Receptor-Targeting and Reactive Oxygen Species-Responsive Liposomal Formulation of Methotrexate for Treatment of Rheumatoid Arthritis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Cultures
2.3. Animals
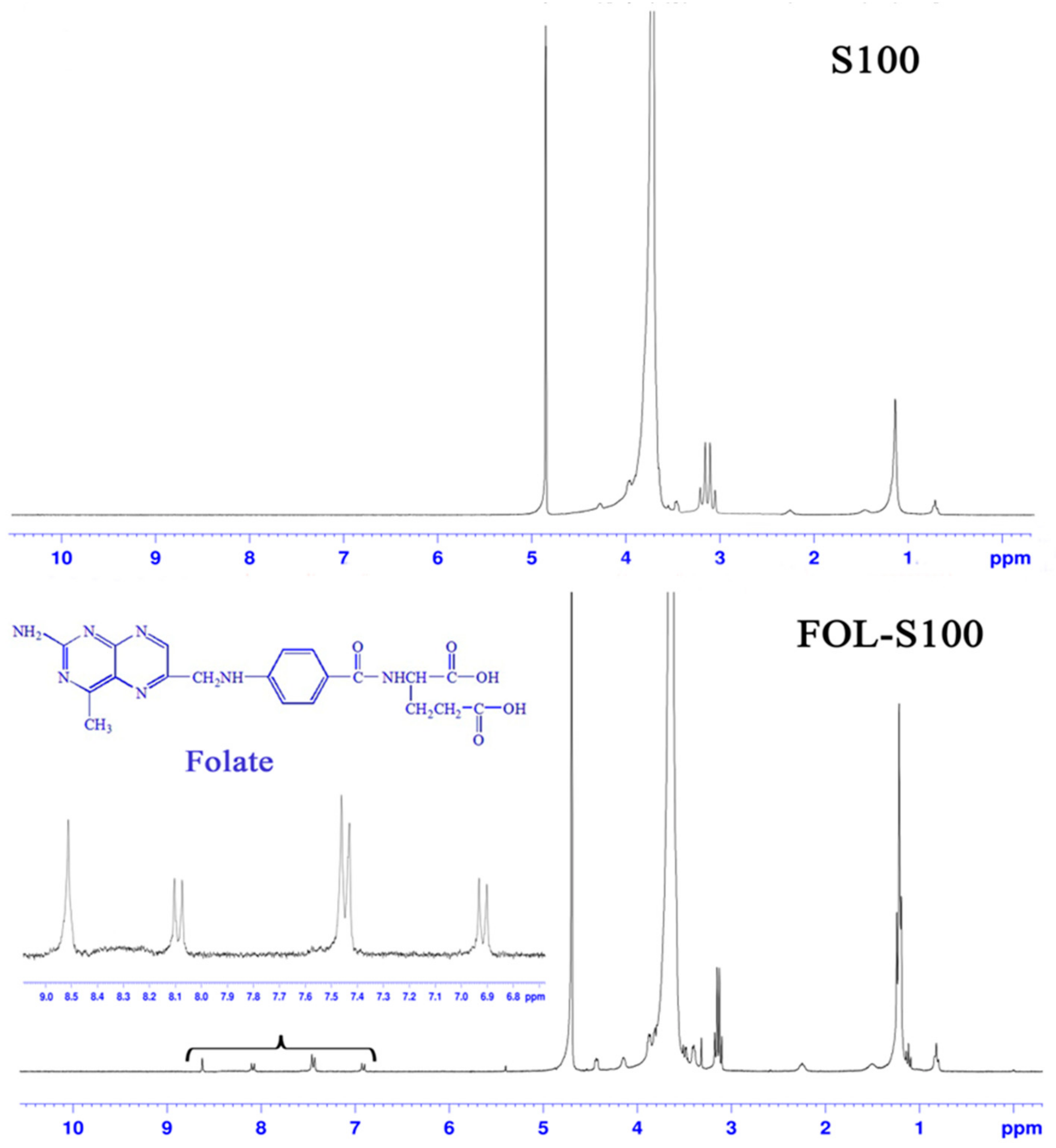
2.4. Synthesis of FOL-S100
2.5. Preparation of FOL-MTX&CAT Liposomes
2.6. Characterization of Liposomes
2.7. In Vitro Drug Release
2.8. Validation of LPS Activated RAW264.7 Cell Model
2.9. Intracellular Uptake of Liposomes
2.10. In Vitro Cytotoxicity Study
2.11. Pharmacokinetic and In Vivo Biodistribution Studies
2.12. Therapeutic Efficacy of Liposomes in CIA Mice
2.13. Statistical Analysis
3. Results and Discussion
3.1. Characterization of FOL-S100
3.2. Preparation and Characterization of Liposomes
3.3. In Vitro Drug Release
3.4. Intracellular Uptake of Liposomes in LPS Activated RAW 264.7 Cells
3.5. In Vitro Cytotoxicity Study
3.6. Pharmacokinetics and In Vivo Biodistribution
3.7. Therapeutic Efficacy and Safety
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- McInnes, I.B.; Schett, G. Pathogenetic insights from the treatment of rheumatoid arthritis. Lancet 2017, 389, 2328–2337. [Google Scholar] [CrossRef] [Green Version]
- Koender, M.I.; van den Berg, W.B. Novel therapeutic targets in rheumatoid arthritis. Trends Pharmacol. Sci. 2015, 36, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Yuan, F.; Quan, L.D.; Cui, L.; Goldring, S.R.; Wang, D. Development of macromolecular prodrug for rheumatoid arthritis. Adv. Drug Deliv. Rev. 2012, 64, 1205–1219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwoh, C.K.; Anderson, L.G.; Greene, J.M.; Johnson, D.A.; O’Dell, J.R.; Robbins, M.L. American College of Rheumatology Subcommittee on Rheumatoid Arthritis Guidelines. Guidelines for the management of rheumatoid arthritis: 2002 Update. Arthritis Rheumatol. 2002, 46, 328–346. [Google Scholar]
- Kremer, J.M.; Russell, A.S.; Emery, P.; Abud-Mendoza, C.; Szechinski, J.; Westhovens, R.; Li, T.; Zhou, X.; Becker, J.C.; Aranda, R.; et al. Long-term safety, efficacy and inhibition of radiographic progression with abatacept treatment in patients with rheumatoid arthritis and an inadequate response to methotrexate: 3-year results from the AIM trial. Ann. Rheumatol. Dis. 2011, 70, 1826–1830. [Google Scholar] [CrossRef] [PubMed]
- Bologna, C.; Viu, P.; Picot, M.C.; Jorgensen, C.; Sany, J. Long-term follow-up of 453 rheumatoid arthritis patients treated with methotrexate: an open, retrospective, observational study. Br. J. Rheumatol. 1997, 36, 535–540. [Google Scholar] [CrossRef] [Green Version]
- Alarcón, G.S.; Tracy, I.C.; Blackburn, W.D., Jr. Methotrexate in rheumatoid arthritis. Toxic effects as the major factor in limiting long-term treatment. Arthritis Rheumatol. 1989, 32, 671–676. [Google Scholar] [CrossRef]
- Fries, J.F.; Williams, C.A.; Ramey, D.; Bloch, D.A. The relative toxicity of disease-modifying antirheumatic drugs. Arthritis. Rheumatol. 1993, 36, 297–306. [Google Scholar] [CrossRef]
- Wang, W.; Zhou, H.; Liu, L. Side effects of methotrexate therapy for rheumatoid arthritis: A systematic review. Eur. J. Med. Chem. 2018, 158, 502–516. [Google Scholar] [CrossRef]
- Dolati, S.; Sadreddini, S.; Rostamzadeh, D.; Ahmadi, M.; Jadidi-Niaragh, F.; Yousefi, M. Utilization of nanoparticle technology in rheumatoid arthritis treatment. Biomed. Pharmacother. 2016, 80, 30–41. [Google Scholar] [CrossRef]
- Yang, M.; Feng, X.; Ding, J.; Chang, F.; Chen, X. Nanotherapeutics relieve rheumatoid arthritis. J. Control Release 2017, 252, 108–124. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, H.Y.; Song, S.Y.; Go, S.H.; Sohn, H.S.; Baik, S.; Soh, M.; Kim, K.; Kim, D.; Kim, H.C.; et al. Synergistic Oxygen Generation and Reactive Oxygen Species Scavenging by Manganese Ferrite/Ceria Co-decorated Nanoparticles for Rheumatoid Arthritis Treatment. ACS Nano 2019, 13, 3206–3217. [Google Scholar] [CrossRef] [PubMed]
- Kinne, R.W.; Stuhlmüller, B.; Burmester, G.R. Cells of the synovium in rheumatoid arthritis Macrophages. Arthritis Res. Ther. 2007, 9, 224. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, A.K.; Pandey, A. Inflammation and rheumatoid arthritis. J. Physiol. Biochem. 2013, 69, 335–347. [Google Scholar] [CrossRef]
- Baschant, U.; Lane, N.E.; Tuckermann, J. The multiple facets of glucocorticoid action in rheumatoid arthritis. Nat. Rev. Rheumatol. 2012, 8, 645–655. [Google Scholar] [CrossRef]
- Xia, W.; Hilgenbrink, A.R.; Matteson, E.L.; Lockwood, M.B.; Cheng, J.X.; Low, P.S. A functional folate receptor is induced during macrophage activation and can be used to target drugs to activated macrophages. Blood 2009, 113, 438–446. [Google Scholar] [CrossRef] [Green Version]
- Chandrupatla, D.M.S.H.; Molthoff, C.F.M.; Lammertsma, A.A.; van der Laken, C.J.; Jansen, G. The folate receptor β as a macrophage-mediated imaging and therapeutic target in rheumatoid arthritis. Drug Deliv. Transl. Res. 2019, 9, 366–378. [Google Scholar] [CrossRef]
- Naor, D.; Nedvetzki, S. CD44 in rheumatoid arthritis. Arthritis Res. Ther. 2003, 5, 105–115. [Google Scholar] [CrossRef]
- Akinrinmade, O.A.; Chetty, S.; Daramola, A.K.; Islam, M.U.; Thepen, T.; Barth, S. CD64: An Attractive Immunotherapeutic Target for M1-type Macrophage Mediated Chronic Inflammatory Diseases. Biomedicines 2017, 5, 56. [Google Scholar] [CrossRef]
- Nakashima-Matsushita, N.; Homma, T.; Yu, S.; Matsuda, T.; Sunahara, N.; Nakamura, T.; Tsukano, M.; Ratnam, M.; Matsuyama, T. Selective expression of folate receptor β and its possible role in methotrexate transport in synovial macrophages from patients with rheumatoid arthritis. Arthritis Rheumatol. 1999, 42, 1609–1616. [Google Scholar] [CrossRef]
- Gonzalez-Rey, E.; Delgado, M. Vasoactive intestinal peptide inhibits cyclooxygenase-2 expression in activated macrophages, microglia, and dendritic cells. Brain Behav. Immun. 2008, 22, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Kodama, T.; Freeman, M.; Rohrer, L.; Zabrecky, J.; Matsudaira, P.; Krieger, M. Type I macrophage scavenger receptor contains alpha-helical and collagen-like coiled coils. Nature 1990, 343, 531–535. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Ma, Y.; Adebayo, A.; Pope, R.M. Increased macrophage activation mediated through toll-like receptors in rheumatoid arthritis. Arthritis Rheumatol. 2007, 56, 2192–2201. [Google Scholar] [CrossRef] [PubMed]
- Heo, R.; Park, J.S.; Jang, H.J.; Kim, S.H.; Shin, J.M.; Suh, Y.D.; Jeong, J.H.; Jo, D.G.; Park, J.H. Hyaluronan nanoparticles bearing γ-secretase inhibitor: in vivo therapeutic effects on rheumatoid arthritis. J. Control Release 2014, 192, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Moura, C.C.; Sequndo, M.A.; Neves, J.D.; Reis, S.; Sarmento, B. Co-association of methotrexate and SPIONs into anti-CD64 antibody-conjugated PLGA nanoparticles for theranostic application. Int. J. Nanomed. 2014, 9, 4911–4922. [Google Scholar] [Green Version]
- Sethi, V.; Rubinstein, I.; Kuzmis, A.; Kastrissios, H.; Artwohl, J.; Onyuksel, H. Novel, biocompatible, and disease modifying VIP nanomedicine for rheumatoid arthritis. Mol. Pharm. 2013, 10, 728–738. [Google Scholar] [CrossRef]
- Bilthariya, U.; Jain, N.; Rajoriya, V.; Jain, A.K. Folate-conjugated albumin nanoparticles for rheumatoid arthritis-targeted delivery of etoricoxib. Drug Dev. Ind. Pharm. 2015, 41, 95–104. [Google Scholar] [CrossRef]
- Nogueira, E.; Lager, F.; Le Roux, D.; Nogueira, P.; Freitas, J.; Charvet, C.; Renault, G.; Loureiro, A.; Almeida, C.R.; Ohradanova-Repic, A.; et al. Enhancing methotrexate tolerance with folate tagged liposomes in arthritic mice. J. Biomed. Nanotechnol. 2015, 11, 2243–2252. [Google Scholar] [CrossRef]
- Kim, S.H.; Kim, J.H.; You, D.G.; Saravanakumar, G.; Yoon, H.Y.; Choi, K.Y.; Thambi, T.; Deepagan, V.G.; Jo, D.G.; Park, J.H. Self-assembled dextran sulphate nanoparticles for targeting rheumatoid arthritis. Chem. Commun. (Camb). 2013, 49, 10349–10351. [Google Scholar] [CrossRef]
- Nogueira, E.; Gomes, A.C.; Preto, A.; Cavaco-Paulo, A. Folate-targeted nanoparticles for rheumatoid arthritis therapy. Nanomedicine 2016, 12, 1113–1126. [Google Scholar] [CrossRef] [Green Version]
- Paulos, C.M.; Turk, M.J.; Breur, G.J.; Low, P.S. Folate receptor-mediated targeting of therapeutic and imaging agents to activated macrophages in rheumatoid arthritis. Adv. Drug Deliv. Rev. 2004, 56, 1205–1217. [Google Scholar] [CrossRef] [PubMed]
- Thomas, T.P.; Goonewardena, S.N.; Majoros, I.J.; Kotlyar, A.; Cao, Z.; Leroueil, P.R.; Baker, J.R., Jr. Folate-targeted nanoparticles show efficacy in the treatment of inflammatory arthritis. Arthritis Rheumatol. 2011, 63, 2671–2680. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeon, J.; Rao, N.V.; Byun, J.H.; Heo, R.; Han, H.S.; Park, J.H. pH-responsive hyaluronic acid-based nanocarrier for treatment of rheumatoid arthritis. J. Nanosci. Nanotechnol. 2016, 16, 11849–11856. [Google Scholar] [CrossRef]
- Costa Lima, S.A.; Reis, S. Temperature-responsive polymeric nanospheres containing methotrexate and gold nanoparticles: a multi-drug system for theranostic in rheumatoid arthritis. Colloids Surf. B Biointerfaces 2015, 133, 378–387. [Google Scholar] [CrossRef]
- Gabriel, D.; Lange, N.; Chobaz-Peclat, V.; Zuluaga, M.F.; Gurny, R.; van den Bergh, H.; Busso, N. Thrombin-sensitive dual fluorescence imaging and therapeutic agent for detection and treatment of synovial inflammation in murine rheumatoid arthritis. J. Control Release 2012, 163, 178–186. [Google Scholar] [CrossRef]
- Khojah, H.M.; Ahmed, S.; Abdel-Rahman, M.S.; Hamza, A.B. Reactive oxygen and nitrogen species in patients with rheumatoid arthritis as potential biomarkers for disease activity and the role of antioxidants. Free Radic. Biol. Med. 2016, 97, 285–291. [Google Scholar] [CrossRef]
- Mateen, S.; Moin, S.; Khan, A.Q.; Zafar, A.; Fatima, N. Increased Reactive Oxygen Species Formation and Oxidative Stress in Rheumatoid Arthritis. PLoS ONE 2016, 11, e0152925. [Google Scholar] [CrossRef]
- Mirshafiey, A.; Mohsenzadegan, M. The role of reactive oxygen species in immunopathogenesis of rheumatoid arthritis. Iran. J. Allergy Asthma Immunol. 2008, 7, 195–202. [Google Scholar]
- Afonso, V.; Champy, R.; Mitrovic, D.; Collin, P.; Lomri, A. Reactive oxygen species and superoxide dismutases: Role in joint diseases. Joint Bone Spine 2007, 74, 324–329. [Google Scholar] [CrossRef]
- Hitchon, C.A.; El-Gabalawy, H.S. Oxidation in rheumatoid arthritis. Arthritis Res. Ther. 2004, 6, 265–278. [Google Scholar] [CrossRef] [Green Version]
- Quiñonez-Flores, C.M.; González-Chávez, S.A.; Pacheco-Tena, C. Hypoxia and its implications in rheumatoid arthritis. J. Biomed. Sci. 2016, 23, 62. [Google Scholar] [CrossRef] [PubMed]
- Nash, K.M.; Ahmed, S. Nanomedicine in the ROS-mediated pathophysiology: applications and clinical advances. Nanomedicine 2015, 11, 2033–2040. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; He, C.; Xiao, C.; Chen, X. Reactive oxygen species (ROS) sesponsive polymers for biomedical applications. Macromol. Biosci. 2016, 16, 635–646. [Google Scholar] [CrossRef] [PubMed]
- Andersen, N.S.; Peiró Cadahía, J.; Previtali, V.; Bondebjerg, J.; Hansen, C.A.; Hansen, A.E.; Andresen, T.L.; Clausen, M.H. Methotrexate prodrugs sensitive to reactive oxygen species for the improved treatment of rheumatoid arthritis. Eur. J. Med. Chem. 2018, 156, 738–746. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, Z.; Li, Z.; Ju, E.; Gao, N.; Zhou, L.; Ren, J.; Qu, X. Upconversion nanoprobes for efficiently in vitro imaging reactive oxygen species and in vivo diagnosing rheumatoid arthritis. Biomaterials 2015, 39, 15–22. [Google Scholar] [CrossRef]
- Wang, X.; Chen, H.; Zheng, Y.; Ma, M.; Chen, Y.; Zhang, K.; Zeng, D.; Shi, J. Au-nanoparticle coated mesoporous silica nanocapsule-based multifunctional platform for ultrasound mediated imaging, cytoclasis and tumor ablation. Biomaterials 2013, 34, 2057–2068. [Google Scholar] [CrossRef]
- Han, H.D.; Jeon, Y.W.; Kwon, H.J.; Jeon, H.N.; Byeon, Y.; Lee, C.O.; Cho, S.H.; Shin, B.C. Therapeutic efficacy of doxorubicin delivery by a CO2 generating liposomal platform in breast carcinoma. Acta Biomater. 2015, 24, 279–285. [Google Scholar] [CrossRef]
- Baffou, G.; Polleux, J.; Rigneault, H.; Monneret, S. Super-heating and micro-bubble generation around plasmonic nanoparticles under cw illumination. J. Phys. Chem. C 2014, 118, 4890–4898. [Google Scholar] [CrossRef]
- Patra, S.; Roy, E.; Madhuri, R.; Sharma, P.K. Creation of ultrasound and temperature-triggered bubble liposomes from economical precursors to enhance the therapeutic efficacy of curcumin in cancer cells. RSC Adv. 2016, 6, 85473–85485. [Google Scholar] [CrossRef]
- Yang, C.; Li, Y.; Du, M.; Chen, Z. Recent Advances in ultrasound-triggered therapy. J. Drug Target 2019, 27, 33–50. [Google Scholar] [CrossRef]
- Chen, H.; He, W.; Guo, Z. An H2O2-responsive nanocarrier for dual-release of platinum anticancer drugs and O2: controlled release and enhanced cytotoxicity against cisplatin resistant cancer cells. Chem. Commun. (Camb) 2014, 50, 9714–9717. [Google Scholar] [CrossRef] [PubMed]
- Alfonso-Prieto, M.; Biarnés, X.; Vidossich, P.; Rovira, C. The molecular mechanism of the catalase reaction. J. Am. Chem. Soc. 2009, 131, 11751–11761. [Google Scholar] [CrossRef] [PubMed]
- Yoshimoto, M.; Sato, M.; Yoshimoto, N.; Nakao, K. Liposomal encapsulation of yeast alcohol dehydrogenase with cofactor for stabilization of the enzyme structure and activity. Biotechnol. Prog. 2008, 24, 576–582. [Google Scholar] [CrossRef] [PubMed]
- Yoshimoto, M.; Sakamoto, H.; Yoshimoto, N.; Kuboi, R.; Nakao, K. Stabilization of quaternary structure and activity of bovine liver catalase through encapsulation in liposomes. Enzyme Microb. Tech. 2007, 41, 849–858. [Google Scholar] [CrossRef]
- Yoshimoto, M.; Miyazaki, Y.; Sato, M.; Fukunaga, K.; Kuboi, R.; Nakao, K. Mechanism for high stability of liposomal glucose oxidase to inhibitor hydrogen peroxide produced in prolonged glucose oxidation. Bioconjug. Chem. 2004, 15, 1055–1061. [Google Scholar] [CrossRef]
- Shi, Y.; Su, Z.; Li, S.; Chen, Y.; Chen, X.; Xiao, Y.; Sun, M.; Ping, Q.; Zong, L. Multistep targeted nano drug delivery system aiming at leukemic stem cells and minimal residual disease. Mol. Pharm. 2013, 10, 2479–2489. [Google Scholar] [CrossRef]
- Gabizon, A.; Horowitz, A.T.; Goren, D.; Tzemach, D.; Mandelbaum-Shavit, F.; Qazen, M.M.; Zalipsky, S. Targeting folate receptor with folate linked to extremities of poly(ethylene glycol)-grafted liposomes: in vitro studies. Bioconjug. Chem. 1999, 10, 289–298. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Y.; Li, T.; Tian, W.; Zhang, Q.; Cheng, Y. Generation 9 polyamidoamine dendrimer encapsulated platinum nanoparticle mimics catalase size, shape, and catalytic activity. Langmuir 2013, 29, 5262–5270. [Google Scholar] [CrossRef]
- Gay, C.A.; Gebicki, J.M. Perchloric acid enhances sensitivity and reproducibility of the ferric-xylenol orange peroxide assay. Anal. Biochem. 2002, 304, 42–46. [Google Scholar] [CrossRef]
- Pandey, P.K.; Maheshwari, R.; Raval, N.; Gondaliya, P.; Kalia, K.; Tekade, R.K. Nanogold-core multifunctional dendrimer for pulsatile chemo-, photothermal- and photodynamic- therapy of rheumatoid arthritis. J. Colloid Interface Sci. 2019, 544, 61–77. [Google Scholar] [CrossRef]
- Shang, C.H.; Zhang, Q.Q.; Zhou, J.H. Oridonin inhibits cell proliferation and induces apoptosis in rheumatoid arthritis fibroblast-like synovicytes. Inflammation 2016, 39, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Jin, X.; Ping, Q.; Cheng, J.; Sun, M.; Cao, F.; You, W.; Yuan, D. A novel lipoprotein-mimic nanocarriers composed of the modified protein and lipid for tumor cell targeting delivery. J. Control Release 2010, 146, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Guissi, N.E.; Li, H.; Xu, Y.; Semcheddine, F.; Chen, M.; Su, Z.; Ping, Q. Mitoxantrone- and folate-TPGS2k conjugate hybrid micellar aggregates to circumvent toxicity and enhance efficiency for breast cancer therapy. Mol. Pharm. 2017, 14, 1082–1094. [Google Scholar] [CrossRef] [PubMed]
- Brand, D.D.; Latham, K.A.; Rosloniec, E.F. Collagen-induced arthritis. Nat. Protoc. 2007, 2, 1269–1275. [Google Scholar] [CrossRef] [PubMed]
- Pietrosimone, K.M.; Jin, M.; Poston, B.; Liu, P. Collagen-induced arthritis: a model for murine autoimmune arthritis. Bio Protoc 2015, 5, e1626. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.Y.; Wan, Y.; Xu, J.Y.; Wu, G.H.; Li, L.; Yao, X.H. Ultrasound extraction of polysaccharides from mulberry leaves and their effect on enhancing antioxidant activity. Carbohydr. Polym. 2016, 137, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Wan, A.; Sun, Y.; Li, H. Characterization of folate-graft-chitosan as a scaffold for nitric oxide release. Int. J. Biol. Macromol. 2008, 43, 415–421. [Google Scholar] [CrossRef]
- Othman, S.H.; Ibrahim, I.A.; Hatab, M.H.; Elbarbary, A.M. Preparation, characterization and biodistribution in quails of 99mTc-folic acid/chitosan nanostructure. Int. J. Biol. Macromol. 2006, 92, 550–560. [Google Scholar] [CrossRef]
- Walde, P.; Ichikawa, S. Enzymes inside lipid vesicles: preparation, reactivity, and applications. Biomol. Eng. 2001, 18, 143–177. [Google Scholar] [CrossRef]
- Hwang, S.Y.; Kim, H.K.; Choo, J.; Seong, G.H.; Hien, T.B.; Lee, E.K. Effects of operating parameters on the efficiency of liposomal encapsulation of enzymes. Colloids Surf. B Biointerfaces 2012, 94, 296–303. [Google Scholar] [CrossRef]
- Gupta, K.; Jang, H.; Harlen, K.; Puri, A.; Nussinov, R.; Schneider, J.P.; Blumenthal, R. Mechanism of membrane permeation induced by synthetic β-hairpin peptides. Biophys. J. 2013, 105, 2093–2103. [Google Scholar] [CrossRef]
- Benedetto, A.; Bingham, R.J.; Ballone, P. Structure and dynamics of POPC bilayers in water solutions of room temperature ionic liquids. J. Chem. Phys. 2015, 142, 124706. [Google Scholar] [CrossRef] [PubMed]
- Chibowski, E.; Szcześ, A. Zeta potential and surface charge of DPPC and DOPC liposomes in the presence of PLC enzyme. Adsorption 2016, 22, 755–765. [Google Scholar] [CrossRef] [Green Version]
- Yao, Y.; Li, B.; Yin, C.; Cong, F.; Ma, G.S.; Liu, N.F.; Fan, Q.L.; Teng, G.J. A folate-conjugated dual-modal fluorescent magnetic resonance imaging contrast agent that targets activated macrophages in vitro and in vivo. J. Biomed. Nanotechnol. 2016, 12, 2161–2171. [Google Scholar] [CrossRef] [PubMed]
- Hattori, Y.; Yamashita, J.; Sakaida, C.; Kawano, K.; Yonemochi, E. Evaluation of antitumor effect of zoledronic acid entrapped in folate-linked liposome for targeting to tumor-associated macrophages. J. Liposome Res. 2015, 25, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Kintzel, P.E. Anticancer drug-induced kidney disorders. Drug Saf. 2001, 24, 19–38. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.T.; Mahmood, U.; Weissleder, R.; Tung, C.H. Arthritis imaging using a near-infrared fluorescence folate-targeted probe. Arthritis Res. Ther. 2005, 7, R310–R317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paulos, C.M.; Varghese, B.; Widmer, W.R.; Breur, G.J.; Vlashi, E.; Low, P.S. Folate-targeted immunotherapy effectively treats established adjuvant and collagen induced arthritis. Arthritis Res. Ther. 2006, 8, R77. [Google Scholar] [CrossRef] [PubMed]
- Alam, M.M.; Han, H.S.; Sung, S.; Kang, J.H.; Sa, K.H.; Al Farugue, H.; Hong, J.; Nam, E.J.; Kim, I.S.; Park, J.H.; et al. Endogenous inspired biomineral-installed hyaluronan nanoparticles as pH-responsive carrier of methotrexate for rheumatoid arthritis. J. Control Release 2017, 252, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Duan, W.; Li, H. Combination of NF-κB targeted siRNA and methotrexate in a hybrid nanocarrier towards the effective treatment in rheumatoid arthritis. J. Nanobiotechnol. 2018, 16, 58. [Google Scholar] [CrossRef]
- Zhou, M.; Hou, J.; Zhong, Z.; Hao, N.; Lin, Y.; Li, C. Targeted delivery of hyaluronic acid-coated solid lipid nanoparticles for rheumatoid arthritis therapy. Drug Deliv. 2018, 25, 716–722. [Google Scholar] [CrossRef] [PubMed]
- Hirvonen, H.; Kautiainen, H.; Moilanen, E.; Mikkelsson, M.; Leirisalo-Repo, M. The effect of cryotherapy on total antioxidative capacity in patients with active seropositive rheumatoid arthritis. Rheumatol. Int. 2017, 37, 1481–1487. [Google Scholar] [CrossRef] [PubMed]
- Sarban, S.; Kocyigit, A.; Yazar, M.; Isikan, U.E. Plasma total antioxidant capacity, lipid peroxidation, and erythrocyte antioxidant enzyme activities in patients with rheumatoid arthritis and osteoarthritis. Clin. Biochem. 2005, 38, 981–986. [Google Scholar] [CrossRef] [PubMed]










| Formulations | Particle Size (nm) | PDI a | Zeta Potential (mV) | EE (%) b | DL (%) c | ||
|---|---|---|---|---|---|---|---|
| MTX | CAT | MTX | CAT | ||||
| MTX-L | 134.9 ± 2.6 | 0.17 ± 0.06 | –8.5 ± 0.9 | 89.24 ± 3.18 | — | 7.18 ± 0.36 | — |
| MTX&CAT-L | 141.2 ± 3.2 | 0.18 ± 0.04 | –8.8 ±1.1 | 84.65 ± 3.74 | 76.52 ± 2.62 | 6.75 ± 0.52 | 2.36 ± 0.42 |
| FOL-MTX&CAT-L | 145.5 ± 4.5 | 0.17 ± 0.03 | –4.2 ± 0.8 | 86.72 ± 2.54 | 79.44 ± 1.85 | 6.94 ± 0.72 | 2.52 ± 0.26 |
| Parameters | MTX-S | MTX-L | MTX&CAT-L | FOL-MTX&CAT-L |
|---|---|---|---|---|
| AUC0-24 (h⋅μg/mL) | 4.91 ± 0.08 | 78.86 ± 2.84 ** | 80.13 ± 4.52 ** | 82.22 ± 6.74 ** |
| T1/2 (h) | 1.23 ± 0.02 | 8.52 ± 0.82 ** | 9.16 ± 1.19 ** | 9.40 ± 2.06 ** |
| MRT (h) | 1.85 ± 0.06 | 11.96 ± 2.86 ** | 12.85 ± 3.63 ** | 13.08 ± 2.65 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, M.; Daddy J.C., K.A.; Su, Z.; Guissi, N.E.I.; Xiao, Y.; Zong, L.; Ping, Q. Folate Receptor-Targeting and Reactive Oxygen Species-Responsive Liposomal Formulation of Methotrexate for Treatment of Rheumatoid Arthritis. Pharmaceutics 2019, 11, 582. https://doi.org/10.3390/pharmaceutics11110582
Chen M, Daddy J.C. KA, Su Z, Guissi NEI, Xiao Y, Zong L, Ping Q. Folate Receptor-Targeting and Reactive Oxygen Species-Responsive Liposomal Formulation of Methotrexate for Treatment of Rheumatoid Arthritis. Pharmaceutics. 2019; 11(11):582. https://doi.org/10.3390/pharmaceutics11110582
Chicago/Turabian StyleChen, Minglei, Kambere Amerigos Daddy J.C., Zhigui Su, Nida El Islem Guissi, Yanyu Xiao, Li Zong, and Qineng Ping. 2019. "Folate Receptor-Targeting and Reactive Oxygen Species-Responsive Liposomal Formulation of Methotrexate for Treatment of Rheumatoid Arthritis" Pharmaceutics 11, no. 11: 582. https://doi.org/10.3390/pharmaceutics11110582
APA StyleChen, M., Daddy J.C., K. A., Su, Z., Guissi, N. E. I., Xiao, Y., Zong, L., & Ping, Q. (2019). Folate Receptor-Targeting and Reactive Oxygen Species-Responsive Liposomal Formulation of Methotrexate for Treatment of Rheumatoid Arthritis. Pharmaceutics, 11(11), 582. https://doi.org/10.3390/pharmaceutics11110582





