Why Human Papillomavirus Acute Infections Matter
Abstract
1. Introduction
2. State of the Fight Against Human Papillomavirus
3. HPV Acute Infections
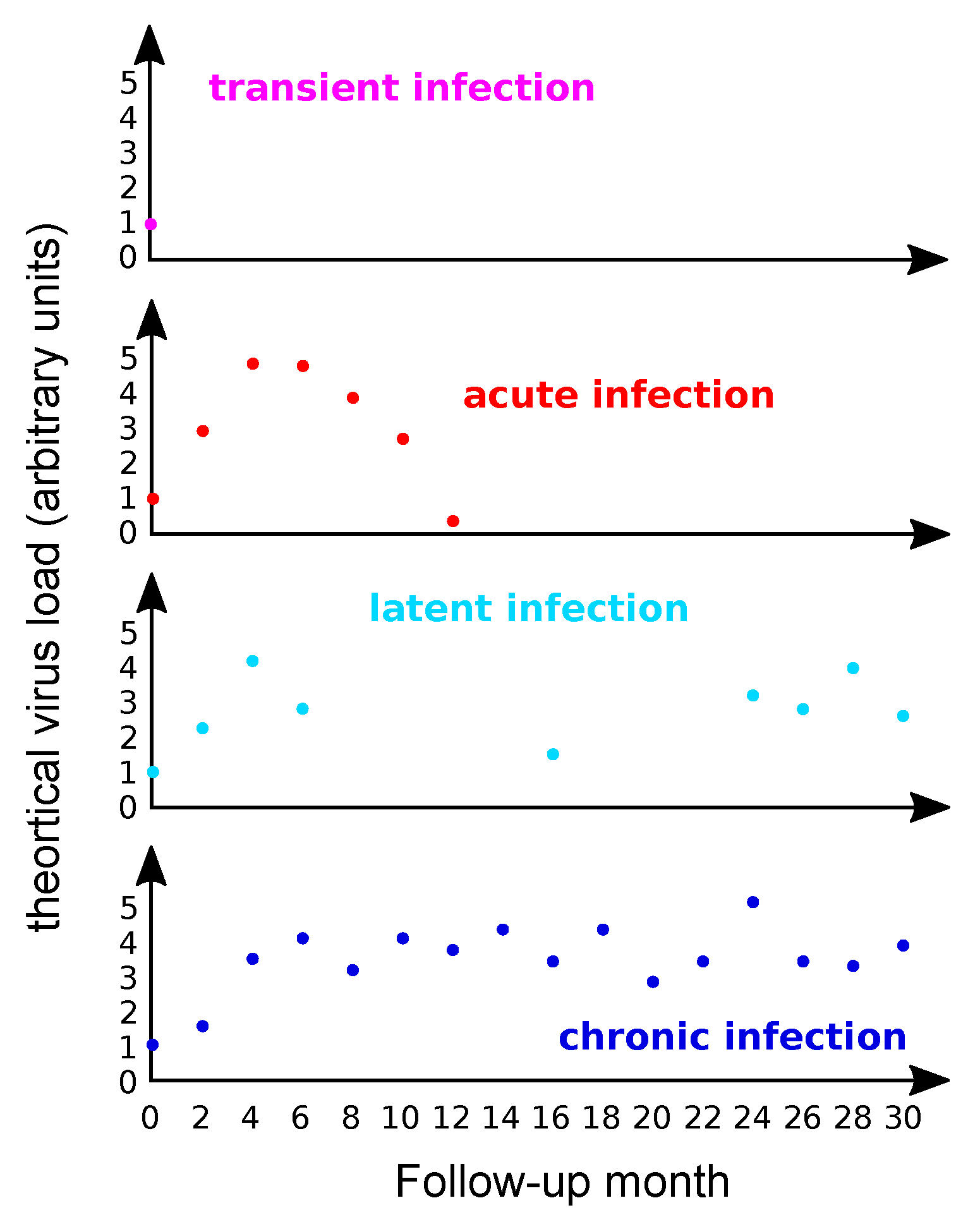
3.1. A Definition Challenge
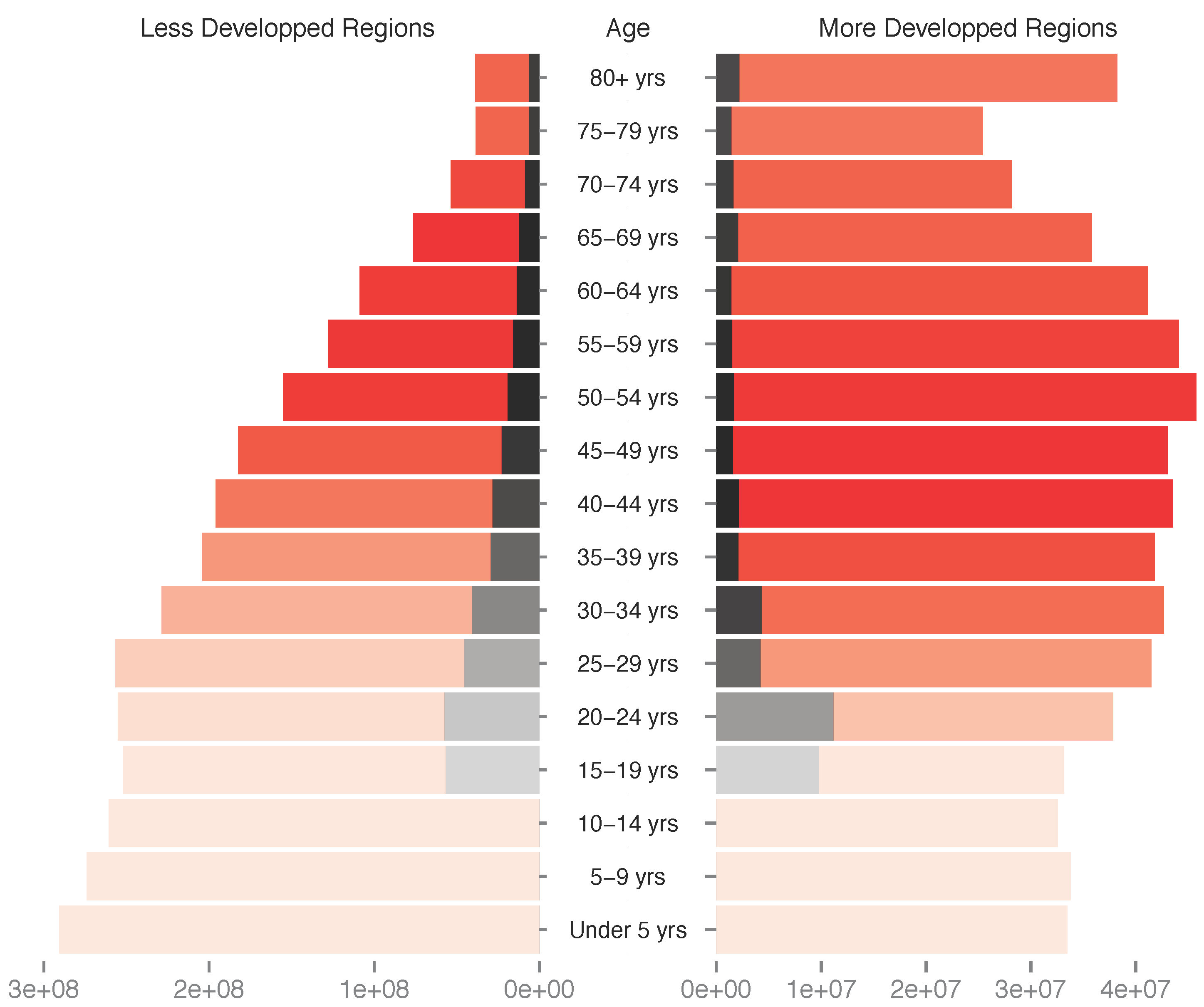
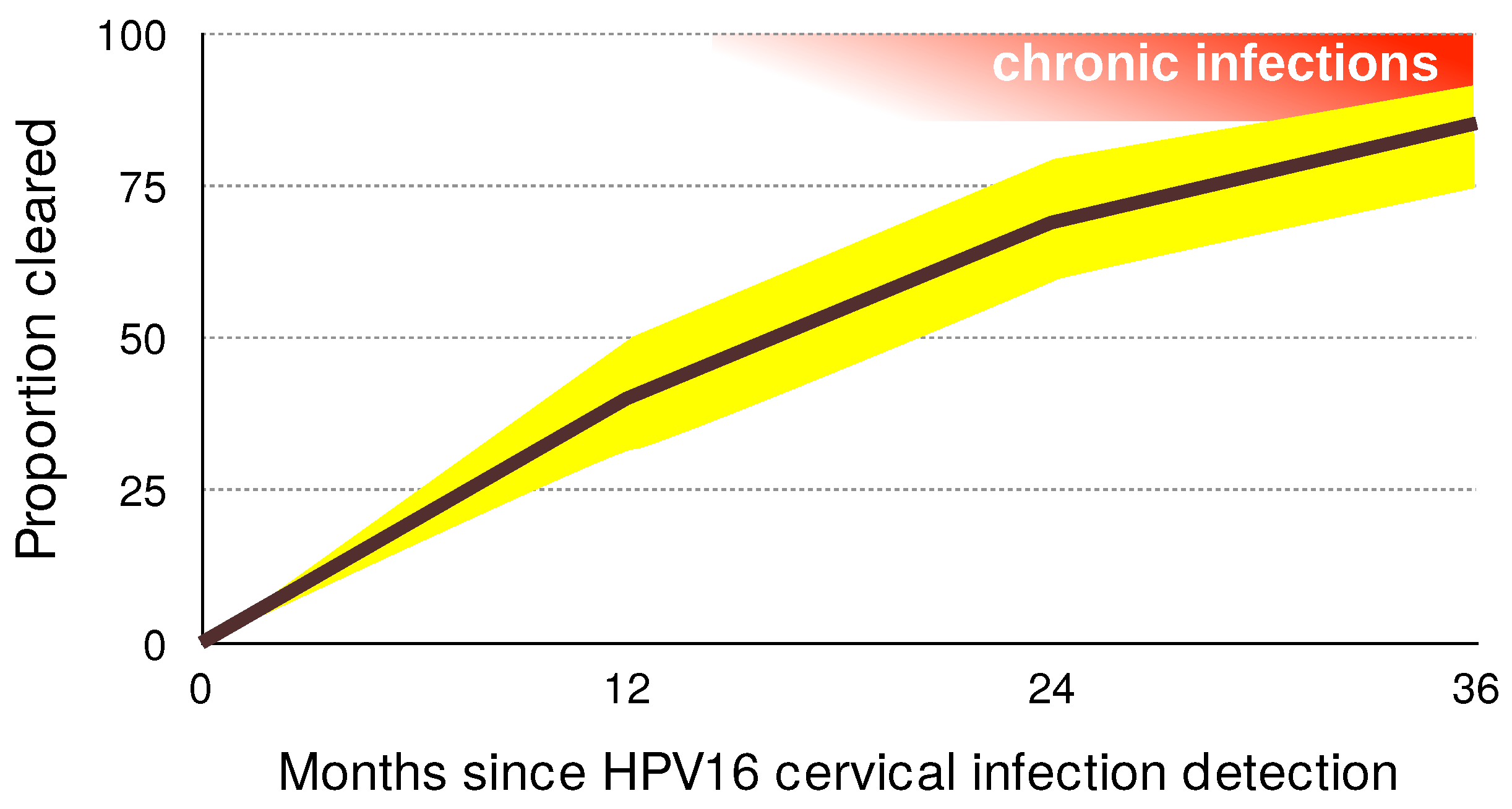
3.2. Most Infections by HPVs Are Acute
3.3. The Immunology of Clearance
4. Open Challenges
4.1. Deciphering HPV Kinetics: The Meta-Population Hypothesis
4.2. Cellular, Viral and Environmental Heterogeneity: Towards Meta-Communities
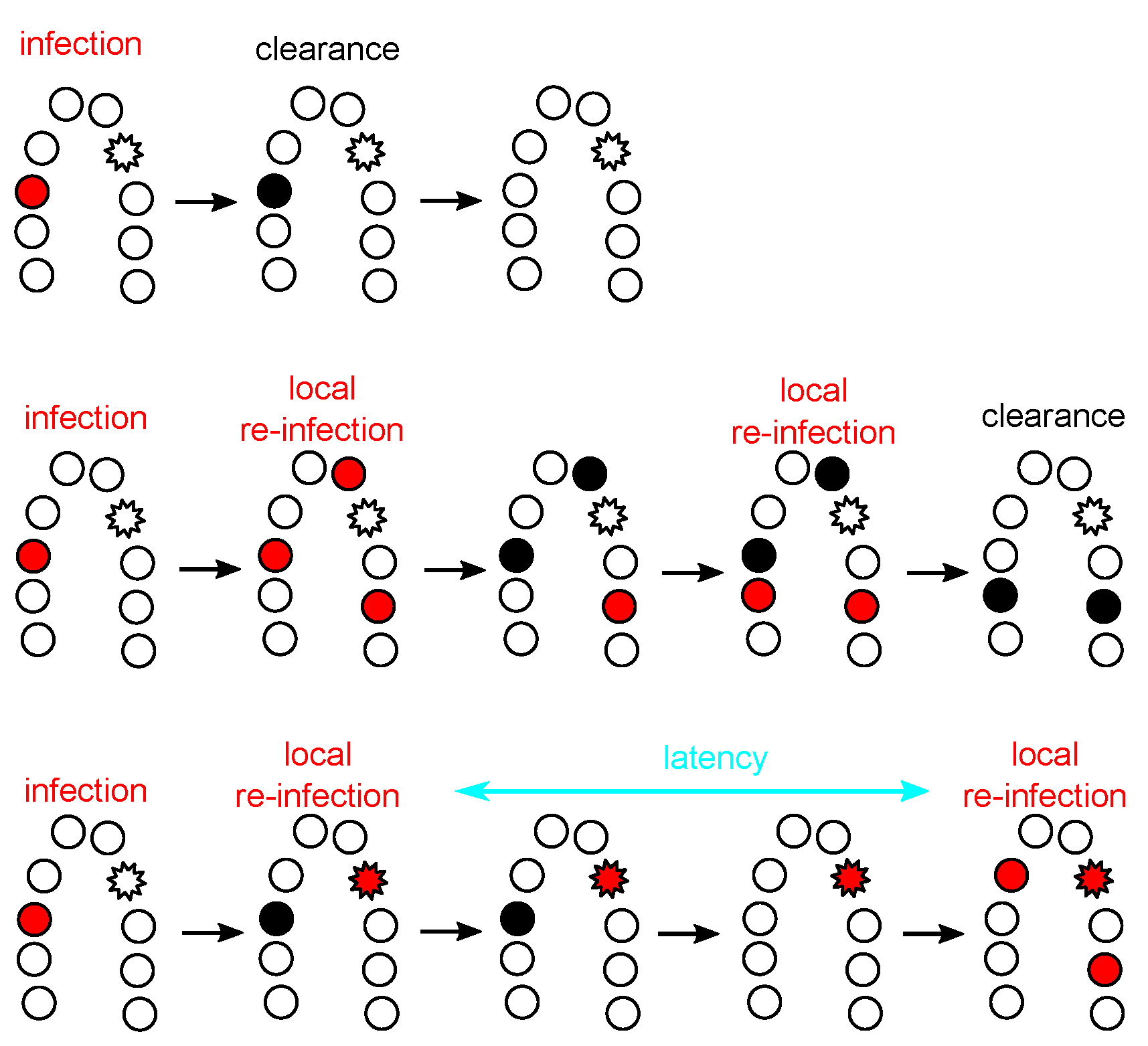
4.3. HPV Latency
4.4. Immunotherapies
4.5. Fertility
4.6. Vaccine Escape
4.7. Scars That Matter Long after Clearance?
5. Conclusions
- Do some innate immunity evasion mechanisms lead to delayed clearance? [43]
- How common is immunological tolerance to HPVs?
- What role, if any, do natural B-cell responses play in clearance?
- Are there differences in immunological clearance mechanisms for different HPVs or for different infected cell types, tissue structures or anatomical sites?
- Are there long term effects after HPV infection clearance (immunological scaring)?
- How do HPV infections affect male and female fertility?
- How do HPV infection kinetics affect transmission? [112]
- Are HPVs infections structured in host tissues (the meta-population hypothesis)?
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CIN | Cervical Intraepithelial Neoplasia |
| CTL | Cytotoxic T Lymphocytes |
| HPVs | Human Papillomaviruses |
| UN | United Nations |
References
- IARC Monographs on the Evaluation of Carcinogenic Risks to Human; v. 100B; International Agency for Research on Cancer: Lyon, France, 2012.
- Virgin, H.W.; Wherry, E.J.; Ahmed, R. Redefining chronic viral infection. Cell 2009, 138, 30–50. [Google Scholar] [CrossRef] [PubMed]
- Insinga, R.P.; Dasbach, E.J.; Elbasha, E.H.; Liaw, K.L.; Barr, E. Incidence and duration of cervical human papillomavirus 6, 11, 16, and 18 infections in young women: An evaluation from multiple analytic perspectives. Cancer Epidemiol. Biomark. Prev. 2007, 16, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Bruni, L.; Diaz, M.; Castellsagué, X.; Ferrer, E.; Bosch, F.X.; de Sanjosé, S. Cervical human papillomavirus prevalence in 5 continents: Meta-analysis of 1 million women with normal cytological findings. J. Infect. Dis. 2010, 202, 1789–1799. [Google Scholar] [CrossRef] [PubMed]
- Plummer, M.; de Martel, C.; Vignat, J.; Ferlay, J.; Bray, F.; Franceschi, S. Global burden of cancers attributable to infections in 2012: A synthetic analysis. Lancet Glob. Health 2016, 4, e609–e616. [Google Scholar] [CrossRef]
- Drolet, M.; Bénard, É.; Boily, M.C.; Ali, H.; Baandrup, L.; Bauer, H.; Beddows, S.; Brisson, J.; Brotherton, J.M.L.; Cummings, T.; et al. Population-level impact and herd effects following human papillomavirus vaccination programmes: a systematic review and meta-analysis. Lancet Infect. Dis. 2015, 15, 565–580. [Google Scholar] [CrossRef]
- Herrero, R.; González, P.; Markowitz, L.E. Present status of human papillomavirus vaccine development and implementation. Lancet Oncol. 2015, 16, e206–e216. [Google Scholar] [CrossRef]
- Schiffman, M.; Solomon, D. Cervical-cancer screening with human papillomavirus and cytologic cotesting. N. Engl. J. Med. 2013, 369, 2324–2331. [Google Scholar] [CrossRef] [PubMed]
- Gakidou, E.; Nordhagen, S.; Obermeyer, Z. Coverage of Cervical Cancer Screening in 57 Countries: Low Average Levels and Large Inequalities. PLoS Med. 2008, 5, e132. [Google Scholar] [CrossRef] [PubMed]
- Palència, L.; Espelt, A.; Rodríguez-Sanz, M.; Puigpinós, R.; Pons-Vigués, M.; Pasarín, M.I.; Spadea, T.; Kunst, A.E.; Borrell, C. Socio-economic inequalities in breast and cervical cancer screening practices in Europe: Influence of the type of screening program. Int. J. Epidemiol. 2010, 39, 757–765. [Google Scholar] [CrossRef] [PubMed]
- Bruni, L.; Diaz, M.; Barrionuevo-Rosas, L.; Herrero, R.; Bray, F.; Bosch, F.X.; de Sanjosé, S.; Castellsagué, X. Global estimates of human papillomavirus vaccination coverage by region and income level: A pooled analysis. Lancet Glob. Health 2016, 4, e453–e463. [Google Scholar] [CrossRef]
- Ronco, G.; Dillner, J.; Elfström, K.M.; Tunesi, S.; Snijders, P.J.F.; Arbyn, M.; Kitchener, H.; Segnan, N.; Gilham, C.; Giorgi-Rossi, P.; et al. Efficacy of HPV-based screening for prevention of invasive cervical cancer: Follow-up of four European randomised controlled trials. Lancet 2014, 383, 524–532. [Google Scholar] [CrossRef]
- Pettersson, B.F.; Hellman, K.; Vaziri, R.; Andersson, S.; Hellström, A.C. Cervical cancer in the screening era: Who fell victim in spite of successful screening programs? J. Gynecol. Oncol. 2011, 22, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Castanon, A.; Landy, R.; Sasieni, P.D. Is cervical screening preventing adenocarcinoma and adenosquamous carcinoma of the cervix? Int. J. Cancer 2016, 139, 1040–1045. [Google Scholar] [CrossRef] [PubMed]
- Bertisch, B.; Franceschi, S.; Lise, M.; Vernazza, P.; Keiser, O.; Schöni-Affolter, F.; Bouchardy, C.; Dehler, S.; Levi, F.; Jundt, G.; et al. Risk Factors for Anal Cancer in Persons Infected with HIV: A Nested Case-Control Study in the Swiss HIV Cohort Study. Am. J. Epidemiol. 2013, 178, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Gillison, M.L.; Chaturvedi, A.K.; Anderson, W.F.; Fakhry, C. Epidemiology of Human Papillomavirus-Positive Head and Neck Squamous Cell Carcinoma. J. Clin. Oncol. 2015, 33, 3235–3242. [Google Scholar] [CrossRef] [PubMed]
- Herse, F.; Reissell, E. The annual costs associated with human papillomavirus types 6, 11, 16, and 18 infections in Finland. Scand. J. Infect. Dis. 2011, 43, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Gravitt, P.E. The known unknowns of HPV natural history. J. Clin. Investig. 2011, 121, 4593–4599. [Google Scholar] [CrossRef] [PubMed]
- Schiffman, M.; Wentzensen, N.; Wacholder, S.; Kinney, W.; Gage, J.C.; Castle, P.E. Human Papillomavirus Testing in the Prevention of Cervical Cancer. J. Natl. Cancer Inst. 2011, 103, 368–383. [Google Scholar] [CrossRef] [PubMed]
- Hazard, K.; Karlsson, A.; Andersson, K.; Ekberg, H.; Dillner, J.; Forslund, O. Cutaneous Human Papillomaviruses Persist on Healthy Skin. J. Investig. Dermatol. 2007, 127, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Depuydt, C.E.; Criel, A.M.; Benoy, I.H.; Arbyn, M.; Vereecken, A.J.; Bogers, J.J. Changes in type-specific human papillomavirus load predict progression to cervical cancer. J. Cell. Mol. Med. 2012, 16, 3096–3104. [Google Scholar] [CrossRef] [PubMed]
- Houlihan, C.F.; Baisley, K.; Bravo, I.G.; Kapiga, S.; de Sanjosé, S.; Changalucha, J.; Ross, D.A.; Hayes, R.J.; Watson-Jones, D. Rapid acquisition of HPV around the time of sexual debut in adolescent girls in Tanzania. Int. J. Epidemiol. 2016, 45, 762–773. [Google Scholar] [CrossRef] [PubMed]
- Doorbar, J. Latent papillomavirus infections and their regulation. Curr. Opin. Virol. 2013, 3, 416–421. [Google Scholar] [CrossRef] [PubMed]
- Maglennon, G.A.; McIntosh, P.B.; Doorbar, J. Immunosuppression Facilitates the Reactivation of Latent Papillomavirus Infections. J. Virol. 2014, 88, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Doorbar, J.; Quint, W.; Banks, L.; Bravo, I.G.; Stoler, M.; Broker, T.R.; Stanley, M.A. The biology and life-cycle of human papillomaviruses. Vaccine 2012, 30 (Suppl. 5), F55–F70. [Google Scholar] [CrossRef] [PubMed]
- Antonsson, A.; Karanfilovska, S.; Lindqvist, P.G.; Hansson, B.G. General Acquisition of Human Papillomavirus Infections of Skin Occurs in Early Infancy. J. Clin. Microbiol. 2003, 41, 2509–2514. [Google Scholar] [CrossRef] [PubMed]
- Rintala, M.A.M.; Grénman, S.E.; Puranen, M.H.; Isolauri, E.; Ekblad, U.; Kero, P.O.; Syrjänen, S.M. Transmission of High-Risk Human Papillomavirus (HPV) between Parents and Infant: A Prospective Study of HPV in Families in Finland. J. Clin. Microbiol. 2005, 43, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Michael, K.M.; Waterboer, T.; Sehr, P.; Rother, A.; Reidel, U.; Boeing, H.; Bravo, I.G.; Schlehofer, J.; Gärtner, B.C.; Pawlita, M. Seroprevalence of 34 Human Papillomavirus Types in the German General Population. PLoS Pathog. 2008, 4, e1000091. [Google Scholar] [CrossRef] [PubMed]
- Depuydt, C.E.; Jonckheere, J.; Berth, M.; Salembier, G.M.; Vereecken, A.J.; Bogers, J.J. Serial type-specific human papillomavirus (HPV) load measurement allows differentiation between regressing cervical lesions and serial virion productive transient infections. Cancer Med. 2015, 4, 1294–1302. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, N.; Méndez, F.; Posso, H.; Molano, M.; van den Brule, A.J.; Ronderos, M.; Meijer, C.; Muñoz, A. Incidence, Duration, and Determinants of Cervical Human Papillomavirus Infection in a Cohort of Colombian Women with Normal Cytological Results. J. Infect. Dis. 2004, 190, 2077–2087. [Google Scholar] [CrossRef] [PubMed]
- Castle, P.E.; Schiffman, M.; Herrero, R.; Hildesheim, A.; Rodriguez, A.C.; Bratti, M.C.; Sherman, M.E.; Wacholder, S.; Tarone, R.; Burk, R.D. A Prospective Study of Age Trends in Cervical Human Papillomavirus Acquisition and Persistence in Guanacaste, Costa Rica. J. Infect. Dis. 2005, 191, 1808–1816. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, A.R.; Lee, J.H.; Fulp, W.; Villa, L.L.; Lazcano, E.; Papenfuss, M.R.; Abrahamsen, M.; Salmeron, J.; Anic, G.M.; Rollison, D.E.; et al. Incidence and clearance of genital human papillomavirus infection in men (HIM): A cohort study. Lancet 2011, 377, 932–940. [Google Scholar] [CrossRef]
- Sanjosé, S.D.; Diaz, M.; Castellsagué, X.; Clifford, G.; Bruni, L.; Muñoz, N.; Bosch, F.X. Worldwide prevalence and genotype distribution of cervical human papillomavirus DNA in women with normal cytology: a meta-analysis. Lancet Infect. Dis. 2007, 7, 453–459. [Google Scholar] [CrossRef]
- Guimerà, N.; Lloveras, B.; Lindeman, J.; Alemany, L.; Sandt, M.V.D.; Alejo, M.; Hernandez-suarez, G.; Bravo, I.G.; Molijn, A.; Jenkins, D.; et al. The Occasional Role of Low-risk Human Papillomaviruses 6, 11, 42, 44, and 70 in Anogenital Carcinoma Defined by Laser Capture Microdissection/PCR Methodology: Results From a Global Study. Am. J. Surg. Pathol. 2013, 37, 1299–1310. [Google Scholar] [CrossRef] [PubMed]
- Herrero, R.; Wacholder, S.; Rodríguez, A.C.; Solomon, D.; González, P.; Kreimer, A.R.; Porras, C.; Schussler, J.; Jiménez, S.; Sherman, M.E.; et al. Prevention of persistent human papillomavirus infection by an HPV16/18 vaccine: A community-based randomized clinical trial in Guanacaste, Costa Rica. Cancer Discov. 2011, 1, 408–419. [Google Scholar] [CrossRef] [PubMed]
- Castellsagué, X.; Naud, P.; Chow, S.N.; Wheeler, C.M.; Germar, M.J.V.; Lehtinen, M.; Paavonen, J.; Jaisamrarn, U.; Garland, S.M.; Salmerón, J.; et al. Risk of newly detected infections and cervical abnormalities in women seropositive for naturally acquired human papillomavirus type 16/18 antibodies: Analysis of the control arm of PATRICIA. J. Infect. Dis. 2014, 210, 517–534. [Google Scholar] [CrossRef] [PubMed]
- Moreira, E.D.; Giuliano, A.R.; Palefsky, J.; Flores, C.A.; Goldstone, S.; Ferris, D.; Hillman, R.J.; Moi, H.; Stoler, M.H.; Marshall, B.; et al. Incidence, Clearance, and Disease Progression of Genital Human Papillomavirus Infection in Heterosexual Men. J. Infect. Dis. 2014, 210, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.H.; Cummings, D.A.T.; Zenilman, J.M.; Gravitt, P.E.; Brotman, R.M. Characterizing the Temporal Dynamics of Human Papillomavirus DNA Detectability Using Short-Interval Sampling. Cancer Epidemiol. Biomark. Prev. 2014, 23, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Shew, M.L.; Ermel, A.C.; Tong, Y.; Tu, W.; Qadadri, B.; Brown, D.R. Episodic detection of human papillomavirus within a longitudinal cohort of young women. J. Med. Virol. 2015, 87, 2122–2129. [Google Scholar] [CrossRef] [PubMed]
- Ferenczy, A.; Franco, E. Persistent human papillomavirus infection and cervical neoplasia. Lancet Oncol. 2002, 3, 11–16. [Google Scholar] [CrossRef]
- Stanley, M. Immunology of HPV Infection. Curr. Obstet. Gynecol. Rep. 2015, 4, 195–200. [Google Scholar] [CrossRef]
- Stanley, M. Immune responses to human papillomavirus. Vaccine 2006, 24 (Suppl. 1), S16–S22. [Google Scholar] [CrossRef] [PubMed]
- Van Hede, D.; Langers, I.; Delvenne, P.; Jacobs, N. Origin and immunoescape of uterine cervical cancer. Presse Med. 2014, 43, e413–e421. [Google Scholar] [CrossRef] [PubMed]
- Levins, R. Some Demographic and Genetic Consequences of Environmental Heterogeneity for Biological Control. Bull. Entomol. Soc. Am. 1969, 15, 237–240. [Google Scholar] [CrossRef]
- Hanski, I. Metapopulation Ecology; Oxford University Press: Oxford, UK, 1999. [Google Scholar]
- Frost, S.D.W.; Dumaurier, M.J.; Wain-Hobson, S.; Brown, A.J.L. Genetic drift and within-host metapopulation dynamics of HIV-1 infection. Proc. Natl. Acad. Sci. USA 2001, 98, 6975–6980. [Google Scholar] [CrossRef] [PubMed]
- Lythgoe, K.A.; Blanquart, F.; Pellis, L.; Fraser, C. Large Variations in HIV-1 Viral Load Explained by Shifting-Mosaic Metapopulation Dynamics. PLoS Biol. 2016, 14, e1002567. [Google Scholar] [CrossRef] [PubMed]
- Murall, C.L.; Bauch, C.T.; Day, T. Could the human papillomavirus vaccines drive virulence evolution? Proc. Biol. Sci. 2015, 282, 20141069. [Google Scholar] [CrossRef] [PubMed]
- Ryser, M.D.; Myers, E.R.; Durrett, R. HPV clearance and the neglected role of stochasticity. PLoS Comput. Biol. 2015, 11, e1004113. [Google Scholar] [CrossRef] [PubMed]
- Moore, P.S.; Chang, Y. Why do viruses cause cancer? Highlights of the first century of human tumour virology. Nat. Rev. Cancer 2010, 10, 878–889. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.M.; Ritchie, J.M.; Yankowitz, J.; Wang, D.; Turek, L.P.; Haugen, T.H. HPV prevalence and concordance in the cervix and oral cavity of pregnant women. Infect. Dis. Obstet. Gynecol. 2004, 12, 45–56. [Google Scholar] [CrossRef] [PubMed]
- De Vuyst, H.; Chung, M.H.; Baussano, I.; Mugo, N.R.; Tenet, V.; van Kemenade, F.J.; Rana, F.S.; Sakr, S.R.; Meijer, C.J.; Snijders, P.J.; et al. Comparison of HPV DNA testing in cervical exfoliated cells and tissue biopsies among HIV-positive women in Kenya. Int. J. Cancer 2013, 133, 1441–1446. [Google Scholar] [CrossRef] [PubMed]
- Nunes, E.M.; López, R.V.M.; Sudenga, S.L.; Gheit, T.; Tommasino, M.; Baggio, M.L.; Ferreira, S.; Galan, L.; Silva, R.C.; Lazcano-Ponce, E.; et al. Concordance of Beta-papillomavirus across anogenital and oral anatomic sites of men: The HIM Study. Virology 2017, 510, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Hampras, S.S.; Rollison, D.E.; Giuliano, A.R.; McKay-Chopin, S.; Minoni, L.; Sereday, K.; Gheit, T.; Tommasino, M. Prevalence and Concordance of Cutaneous Beta Human Papillomavirus Infection at Mucosal and Cutaneous Sites. J. Infect. Dis. 2017, 216, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Hernandez, B.Y.; Shvetsov, Y.B.; Goodman, M.T.; Wilkens, L.R.; Thompson, P.J.; Zhu, X.; Tom, J.; Ning, L. Genital and extra-genital warts increase the risk of asymptomatic genital human papillomavirus infection in men. Sex. Transm. Infect. 2011, 87, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Kalantari, M.; Garcia-Carranca, A.; Morales-Vazquez, C.D.; Zuna, R.; Montiel, D.P.; Calleja-Macias, I.E.; Johansson, B.; Andersson, S.; Bernard, H.U. Laser capture microdissection of cervical human papillomavirus infections: Copy number of the virus in cancerous and normal tissue and heterogeneous DNA methylation. Virology 2009, 390, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Quint, W.; Jenkins, D.; Molijn, A.; Struijk, L.; van de Sandt, M.; Doorbar, J.; Mols, J.; Van Hoof, C.; Hardt, K.; Struyf, F.; et al. One virus, one lesion—Individual components of CIN lesions contain a specific HPV type. J. Pathol. 2012, 227, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Lechner, M.; Fenton, T.; West, J.; Wilson, G.; Feber, A.; Henderson, S.; Thirlwell, C.; Dibra, H.K.; Jay, A.; Butcher, L.; et al. Identification and functional validation of HPV-mediated hypermethylation in head and neck squamous cell carcinoma. Genome Med. 2013, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Candi, E.; Schmidt, R.; Melino, G. The cornified envelope: a model of cell death in the skin. Nat. Rev. Mol. Cell Biol. 2005, 6, 328–340. [Google Scholar] [CrossRef] [PubMed]
- Eckhart, L.; Valle, L.D.; Jaeger, K.; Ballaun, C.; Szabo, S.; Nardi, A.; Buchberger, M.; Hermann, M.; Alibardi, L.; Tschachler, E. Identification of reptilian genes encoding hair keratin-like proteins suggests a new scenario for the evolutionary origin of hair. Proc. Natl. Acad. Sci. USA 2008, 105, 18419–18423. [Google Scholar] [CrossRef] [PubMed]
- Henry, J.; Toulza, E.; Hsu, C.Y.; Pellerin, L.; Balica, S.; Mazereeuw-Hautier, J.; Paul, C.; Serre, G.; Jonca, N.; Simon, M. Update on the epidermal differentiation complex. Front. Biosci. 2012, 17, 1517–1532. [Google Scholar] [CrossRef]
- Dhouailly, D. A new scenario for the evolutionary origin of hair, feather, and avian scales. J. Anat. 2009, 214, 587–606. [Google Scholar] [CrossRef] [PubMed]
- Gilliam, A.C.; Kremer, I.B.; Yoshida, Y.; Stevens, S.R.; Tootell, E.; Teunissen, M.B.; Hammerberg, C.; Cooper, K.D. The human hair follicle: A reservoir of CD40+ B7-deficient Langerhans cells that repopulate epidermis after UVB exposure. J. Investig. Dermatol. 1998, 110, 422–427. [Google Scholar] [CrossRef] [PubMed]
- Malissen, B.; Tamoutounour, S.; Henri, S. The origins and functions of dendritic cells and macrophages in the skin. Nat. Rev. Immunol. 2014, 14, 417–428. [Google Scholar] [CrossRef] [PubMed]
- Wilson, D.S. Complex Interactions in Metacommunities, with Implications for Biodiversity and Higher Levels of Selection. Ecology 1992, 73, 1984–2000. [Google Scholar] [CrossRef]
- Murall, C.L.; McCann, K.S.; Bauch, C.T. Food webs in the human body: Linking ecological theory to viral dynamics. PLoS ONE 2012, 7, e48812. [Google Scholar] [CrossRef] [PubMed]
- Rynkiewicz, E.C.; Pedersen, A.B.; Fenton, A. An ecosystem approach to understanding and managing within-host parasite community dynamics. Trends Parasitol. 2015, 31, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Bravo, I.G.; Félez-Sánchez, M. Papillomaviruses: Viral evolution, cancer and evolutionary medicine. Evol. Med. Public Health 2015, 2015, 32–51. [Google Scholar] [CrossRef] [PubMed]
- Egawa, N.; Egawa, K.; Griffin, H.; Doorbar, J. Human Papillomaviruses; Epithelial Tropisms, and the Development of Neoplasia. Viruses 2015, 7, 3863–3890. [Google Scholar] [CrossRef] [PubMed]
- Murray, R.F.; Hobbs, J.; Payne, B. Possible clonal origin of common warts (Verruca vulgaris). Nature 1971, 232, 51–52. [Google Scholar] [CrossRef] [PubMed]
- Herfs, M.; Yamamoto, Y.; Laury, A.; Wang, X.; Nucci, M.R.; McLaughlin-Drubin, M.E.; Münger, K.; Feldman, S.; McKeon, F.D.; Xian, W.; et al. A discrete population of squamocolumnar junction cells implicated in the pathogenesis of cervical cancer. Proc. Natl. Acad. Sci. USA 2012, 109, 10516–10521. [Google Scholar] [CrossRef] [PubMed]
- Nicolás-Párraga, S.; Gandini, C.; Pimenoff, V.N.; Alemany, L.; de Sanjosé, S.; Xavier Bosch, F.; Bravo, I.G.; The RIS HPV TT and HPV VVAP study groups. HPV16 variants distribution in invasive cancers of the cervix, vulva, vagina, penis, and anus. Cancer Med. 2016, 5, 2909–2919. [Google Scholar]
- Nicolás-Párraga, S.; Alemany, L.; de Sanjosé, S.; Bosch, F.; Bravo, I.; RIS HPV TT and HPV VVAP study groups. Differential HPV16 variant distribution in squamous cell carcinoma, adenocarcinoma and adenosquamous cell carcinoma. Int. J. Cancer 2017, 140, 2092–2100. [Google Scholar]
- Maglennon, G.A.; McIntosh, P.; Doorbar, J. Persistence of viral DNA in the epithelial basal layer suggests a model for papillomavirus latency following immune regression. Virology 2011, 414, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Reinson, T.; Henno, L.; Toots, M., Jr.; Ustav, M. The Cell Cycle Timing of Human Papillomavirus DNA Replication. PLoS ONE 2015, 10, e0131675. [Google Scholar] [CrossRef] [PubMed]
- Van der Weele, P.; Meijer, C.J.L.M.; King, A.J. Whole-Genome Sequencing and Variant Analysis of Human Papillomavirus 16 Infections. J. Virol. 2017, 91, e00844-17. [Google Scholar] [CrossRef] [PubMed]
- Bosch, F.X.; Robles, C.; Díaz, M.; Arbyn, M.; Baussano, I.; Clavel, C.; Ronco, G.; Dillner, J.; Lehtinen, M.; Petry, K.U.; et al. HPV-FASTER: Broadening the scope for prevention of HPV-related cancer. Nat. Rev. Oncol. 2015, 13, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Skeate, J.G.; Woodham, A.W.; Einstein, M.H.; Silva, D.M.D.; Kast, W.M. Current therapeutic vaccination and immunotherapy strategies for HPV-related diseases. Hum. Vaccines Immunother. 2016, 12, 1418–1429. [Google Scholar] [CrossRef] [PubMed]
- Trimble, C.L.; Morrow, M.P.; Kraynyak, K.A.; Shen, X.; Dallas, M.; Yan, J.; Edwards, L.; Parker, R.L.; Denny, L.; Giffear, M.; et al. Safety, efficacy, and immunogenicity of VGX-3100, a therapeutic synthetic DNA vaccine targeting human papillomavirus 16 and 18 E6 and E7 proteins for cervical intraepithelial neoplasia 2/3: A randomised, double-blind, placebo-controlled phase 2b trial. Lancet 2015, 386, 2078–2088. [Google Scholar] [CrossRef]
- Amador-Molina, A.; Hernández-Valencia, J.F.; Lamoyi, E.; Contreras-Paredes, A.; Lizano, M. Role of innate immunity against human papillomavirus (HPV) infections and effect of adjuvants in promoting specific immune response. Viruses 2013, 5, 2624–2642. [Google Scholar] [CrossRef] [PubMed]
- Derkay, C.S.; Wiatrak, B. Recurrent Respiratory Papillomatosis: A Review. Laryngoscope 2008, 118, 1236–1247. [Google Scholar] [CrossRef] [PubMed]
- Martin, D.; Rödel, F.; Balermpas, P.; Rödel, C.; Fokas, E. The immune microenvironment and HPV in anal cancer: Rationale to complement chemoradiation with immunotherapy. BBA Rev. Cancer 2017, 1868, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Silasi, M.; Cardenas, I.; Kwon, J.Y.; Racicot, K.; Aldo, P.; Mor, G. Viral Infections During Pregnancy. Am. J. Reprod. Immunol. 2015, 73, 199–213. [Google Scholar] [CrossRef] [PubMed]
- Kyrgiou, M.; Athanasiou, A.; Paraskevaidi, M.; Mitra, A.; Kalliala, I.; Martin-Hirsch, P.; Arbyn, M.; Bennett, P.; Paraskevaidis, E. Adverse obstetric outcomes after local treatment for cervical preinvasive and early invasive disease according to cone depth: systematic review and meta-analysis. BMJ 2016, 354, i3633. [Google Scholar] [CrossRef] [PubMed]
- Zuo, Z.; Goel, S.; Carter, J.E. Association of Cervical Cytology and HPV DNA Status During Pregnancy With Placental Abnormalities and Preterm Birth. Am. J. Clin. Pathol. 2011, 136, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Laprise, C.; Trottier, H.; Monnier, P.; Coutlée, F.; Mayrand, M.H. Prevalence of human papillomaviruses in semen: A systematic review and meta-analysis. Hum. Reprod. 2014, 29, 640–651. [Google Scholar] [CrossRef] [PubMed]
- Luttmer, R.; Dijkstra, M.G.; Snijders, P.J.F.; Hompes, P.G.A.; Pronk, D.T.M.; Hubeek, I.; Berkhof, J.; Heideman, D.A.M.; Meijer, C.J.L.M. Presence of human papillomavirus in semen in relation to semen quality. Hum. Reprod. 2016, 31, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Foresta, C.; Noventa, M.; De Toni, L.; Gizzo, S.; Garolla, A. HPV-DNA sperm infection and infertility: From a systematic literature review to a possible clinical management proposal. Andrology 2015, 3, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Castellsagué, X.; Drudis, T.; Cañadas, M.P.; Goncé, A.; Ros, R.; Pérez, J.M.; Quintana, M.J.; Muñoz, J.; Albero, G.; de Sanjosé, S.; et al. Human Papillomavirus (HPV) infection in pregnant women and mother-to-child transmission of genital HPV genotypes: a prospective study in Spain. BMC Infect. Dis. 2009, 9, 74. [Google Scholar] [CrossRef] [PubMed]
- Racicot, K.; Cardenas, I.; Wünsche, V.; Aldo, P.; Guller, S.; Means, R.E.; Romero, R.; Mor, G. Viral Infection of the Pregnant Cervix Predisposes to Ascending Bacterial Infection. J. Immunol. 2013, 191, 934–941. [Google Scholar] [CrossRef] [PubMed]
- Slatter, T.L.; Hung, N.G.; Clow, W.M.; Royds, J.A.; Devenish, C.J.; Hung, N.A. A clinicopathological study of episomal papillomavirus infection of the human placenta and pregnancy complications. Mod. Pathol. 2015, 28, 1369–1382. [Google Scholar] [CrossRef] [PubMed]
- Gandon, S.; Day, T. Evidences of parasite evolution after vaccination. Vaccine 2008, 26S, C4–C7. [Google Scholar] [CrossRef]
- Bernard, H.U.; Burk, R.D.; Chen, Z.; van Doorslaer, K.; Hausen, H.Z.; de Villiers, E.M. Classification of papillomaviruses (PVs) based on 189 PV types and proposal of taxonomic amendments. Virology 2010, 401, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Joura, E.A.; Giuliano, A.R.; Iversen, O.E.; Bouchard, C.; Mao, C.; Mehlsen, J.; Moreira, E.D., Jr.; Ngan, Y.; Petersen, L.K.; Lazcano-Ponce, E.; et al. A 9-valent HPV vaccine against infection and intraepithelial neoplasia in women. N. Engl. J. Med. 2015, 372, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Draper, E.; Bissett, S.L.; Howell-Jones, R.; Edwards, D.; Munslow, G.; Soldan, K.; Beddows, S. Neutralization of non-vaccine human papillomavirus pseudoviruses from the A7 and A9 species groups by bivalent HPV vaccine sera. Vaccine 2011, 29, 8585–8590. [Google Scholar] [CrossRef] [PubMed]
- Kemp, T.J.; Hildesheim, A.; Safaeian, M.; Dauner, J.G.; Pan, Y.; Porras, C.; Schiller, J.T.; Lowy, D.R.; Herrero, R.; Pinto, L.A. HPV16/18 L1 VLP vaccine induces cross-neutralizing antibodies that may mediate cross-protection. Vaccine 2011, 29, 2011–2014. [Google Scholar] [CrossRef] [PubMed]
- Markowitz, L.E.; Liu, G.; Hariri, S.; Steinau, M.; Dunne, E.F.; Unger, E.R. Prevalence of HPV After Introduction of the Vaccination Program in the United States. Pediatrics 2016, 137, e20151968. [Google Scholar] [CrossRef] [PubMed]
- Mirabello, L.; Yeager, M.; Yu, K.; Clifford, G.M.; Xiao, Y.; Zhu, B.; Cullen, M.; Boland, J.F.; Wentzensen, N.; Nelson, C.W.; et al. HPV16 E7 Genetic Conservation Is Critical to Carcinogenesis. Cell 2017, 170, 1164.e6. [Google Scholar] [CrossRef] [PubMed]
- Worobey, M.; Han, G.Z.; Rambaut, A. A synchronized global sweep of the internal genes of modern avian influenza virus. Nature 2014, 508, 254–257. [Google Scholar] [CrossRef] [PubMed]
- Weinert, L.A.; Depledge, D.P.; Kundu, S.; Gershon, A.A.; Nichols, R.A.; Balloux, F.; Welch, J.J.; Breuer, J. Rates of Vaccine Evolution Show Strong Effects of Latency: Implications for Varicella Zoster Virus Epidemiology. Mol. Biol. Evol. 2015, 32, 1020–1028. [Google Scholar] [CrossRef] [PubMed]
- Da Fonseca, D.M.; Hand, T.W.; Han, S.J.; Gerner, M.Y.; Glatman Zaretsky, A.; Byrd, A.L.; Harrison, O.J.; Ortiz, A.M.; Quinones, M.; Trinchieri, G.; et al. Microbiota-Dependent Sequelae of Acute Infection Compromise Tissue-Specific Immunity. Cell 2015, 163, 354–366. [Google Scholar] [CrossRef] [PubMed]
- Paschos, K.; Allday, M.J. Epigenetic reprogramming of host genes in viral and microbial pathogenesis. Trends Microbiol. 2010, 18, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Hattori, N.; Ushijima, T. Epigenetic impact of infection on carcinogenesis: Mechanisms and applications. Genome Med. 2016, 8, 10. [Google Scholar] [CrossRef] [PubMed]
- Johannsen, E.; Lambert, P.F. Epigenetics of human papillomaviruses. Virology 2013, 445, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Quint, K.D.; Genders, R.E.; de Koning, M.N.; Borgogna, C.; Gariglio, M.; Bouwes Bavinck, J.N.; Doorbar, J.; Feltkamp, M.C. Human Beta-papillomavirus infection and keratinocyte carcinomas. J. Pathol. 2015, 235, 342–354. [Google Scholar] [CrossRef] [PubMed]
- Bzhalava, D.; Johansson, H.; Ekström, J.; Faust, H.; Möller, B.; Eklund, C.; Nordin, P.; Stenquist, B.; Paoli, J.; Persson, B.; et al. Unbiased Approach for Virus Detection in Skin Lesions. PLoS ONE 2013, 8, e65953. [Google Scholar] [CrossRef] [PubMed]
- Gillison, M.L.; Broutian, T.; Pickard, R.K.L.; Tong, Z.Y.; Xiao, W.; Kahle, L.; Graubard, B.I.; Chaturvedi, A.K. Prevalence of Oral HPV Infection in the United States, 2009–2010. JAMA 2012, 307, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, A.R.; Lazcano-Ponce, E.; Villa, L.L.; Flores, R.; Salmeron, J.; Lee, J.H.; Papenfuss, M.R.; Abrahamsen, M.; Jolles, E.; Nielson, C.M.; et al. The Human Papillomavirus Infection in Men Study: Human Papillomavirus Prevalence and Type Distribution among Men Residing in Brazil, Mexico, and the United States. Cancer Epidemiol. Biomark. Prev. 2008, 17, 2036–2043. [Google Scholar] [CrossRef] [PubMed]
- Nyitray, A.G.; Smith, D.; Villa, L.; Lazcano-Ponce, E.; Abrahamsen, M.; Papenfuss, M.; Giuliano, A.R. Prevalence of and Risk Factors for Anal Human Papillomavirus Infection in Men Who Have Sex with Women: A Cross-National Study. J. Infect. Dis. 2010, 201, 1498–1508. [Google Scholar] [CrossRef] [PubMed]
- Brisson, M.; Bénard, E.; Drolet, M.; Bogaards, J.A.; Baussano, I.; Vänskä, S.; Jit, M.; Boily, M.C.; Smith, M.A.; Berkhof, J.; et al. Population-level impact, herd immunity, and elimination after human papillomavirus vaccination: A systematic review and meta-analysis of predictions from transmission-dynamic models. Lancet Public Health 2016, 1, e8–e17. [Google Scholar] [CrossRef]
- Grabowski, M.K.; Kong, X.; Gray, R.H.; Serwadda, D.; Kigozi, G.; Gravitt, P.E.; Nalugoda, F.; Reynolds, S.J.; Wawer, M.J.; Redd, A.D.; et al. Partner Human Papillomavirus Viral Load and Incident Human Papillomavirus Detection in Heterosexual Couples. J. Infect. Dis. 2016, 213, 948–956. [Google Scholar] [CrossRef] [PubMed]
- Brotman, R.M.; Shardell, M.D.; Gajer, P.; Tracy, J.K.; Zenilman, J.M.; Ravel, J.; Gravitt, P.E. Interplay between the temporal dynamics of the vaginal microbiota and human papillomavirus detection. J. Infect. Dis. 2014, 210, 1723–1733. [Google Scholar] [CrossRef] [PubMed]
- Mitra, A.; MacIntyre, D.A.; Marchesi, J.R.; Lee, Y.S.; Bennett, P.R.; Kyrgiou, M. The vaginal microbiota, human papillomavirus infection and cervical intraepithelial neoplasia: what do we know and where are we going next? Microbiome 2016, 4, 58. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alizon, S.; Murall, C.L.; Bravo, I.G. Why Human Papillomavirus Acute Infections Matter. Viruses 2017, 9, 293. https://doi.org/10.3390/v9100293
Alizon S, Murall CL, Bravo IG. Why Human Papillomavirus Acute Infections Matter. Viruses. 2017; 9(10):293. https://doi.org/10.3390/v9100293
Chicago/Turabian StyleAlizon, Samuel, Carmen Lía Murall, and Ignacio G. Bravo. 2017. "Why Human Papillomavirus Acute Infections Matter" Viruses 9, no. 10: 293. https://doi.org/10.3390/v9100293
APA StyleAlizon, S., Murall, C. L., & Bravo, I. G. (2017). Why Human Papillomavirus Acute Infections Matter. Viruses, 9(10), 293. https://doi.org/10.3390/v9100293





