Trans-Amplifying RNA: A Journey from Alphavirus Research to Future Vaccines
Abstract
1. Introduction
2. Natural Trans-Replicating RNA
2.1. Investigation of Genome Sequence Requirements for Replication Using Defective Interfering Particles
2.2. Engineered Defective Interfering RNAs to Introduce Foreign Genes into Cells
3. Recombinant Alphavirus Production by Exploiting Defective Interfering RNA
3.1. Helper RNA
3.2. Development of Advanced Helper RNA Systems
4. Trans-Replicating RNA for Studying Alphaviral RNA Replication and Replicase Biology
4.1. Defining RNA Promoter Requirements
4.2. Investigating Replicase Subunit Functions
4.3. Investigating Spherule Formation
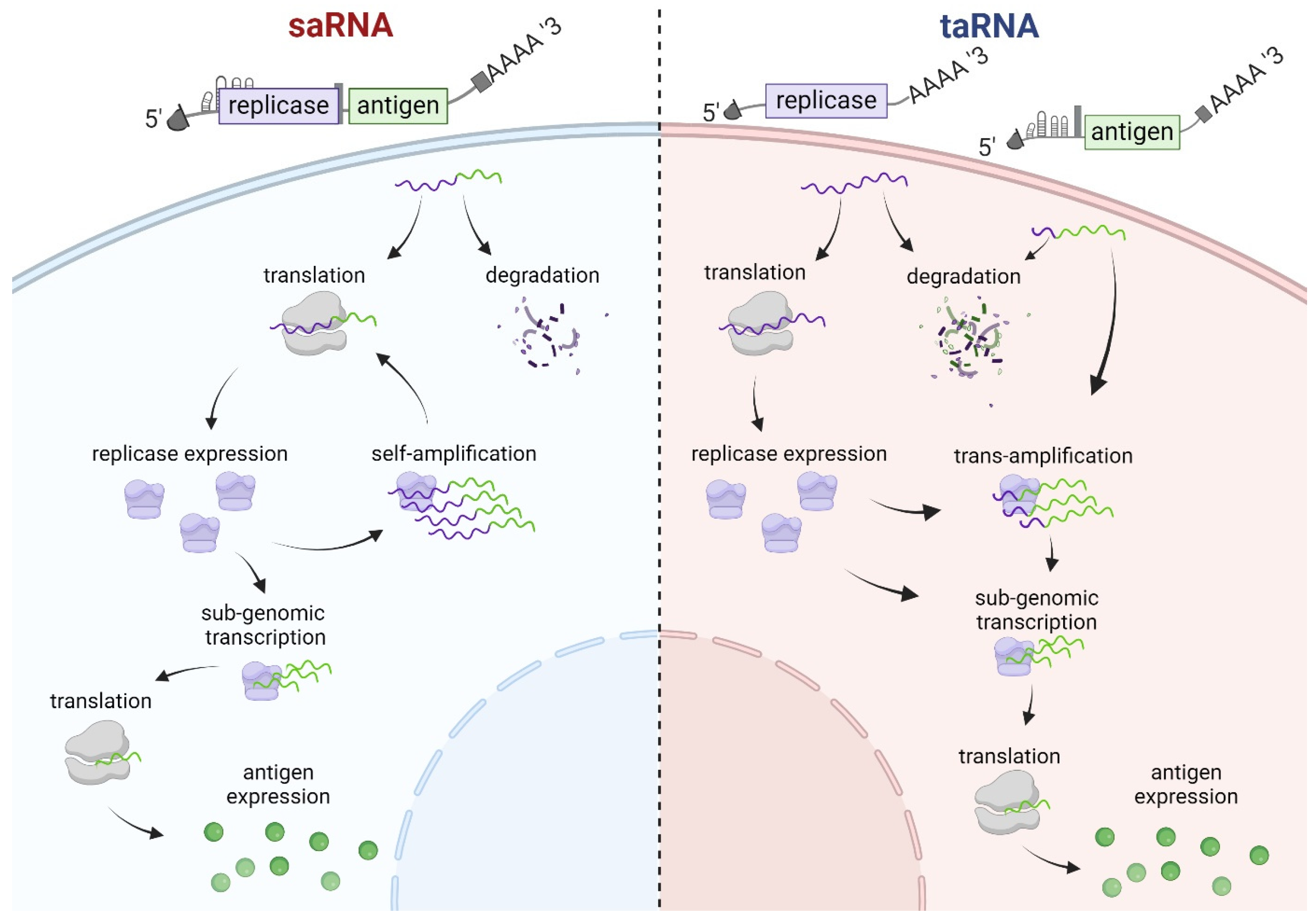
5. Trans-Amplifying RNA Vaccines
6. Future Perspective of taRNA Vaccines, Challenges and Open Questions
6.1. Towards a Non-Immunogenic taRNA Vaccine
6.2. Realizing Multivalent Vaccines Using taRNA
6.3. taRNA to Accelerate Seasonal Vaccine Production and Improve Pandemic Preparedness
6.4. taRNA Formulation
7. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Lee, J.; Woodruff, M.C.; Kim, E.H.; Nam, J.-H. Knife’s edge: Balancing immunogenicity and reactogenicity in mRNA vaccines. Exp. Mol. Med. 2023, 55, 1305–1313. [Google Scholar] [CrossRef] [PubMed]
- Dolgin, E. Self-copying RNA vaccine wins first full approval: What’s next? Nature 2023, 624, 236–237. [Google Scholar] [CrossRef] [PubMed]
- Oda, Y.; Kumagai, Y.; Kanai, M.; Iwama, Y.; Okura, I.; Minamida, T.; Yagi, Y.; Kurosawa, T.; Greener, B.; Zhang, Y.; et al. Immunogenicity and safety of a booster dose of a self-amplifying RNA COVID-19 vaccine (ARCT-154) versus BNT162b2 mRNA COVID-19 vaccine: A double-blind, multicentre, randomised, controlled, phase 3, non-inferiority trial. Lancet Infect. Dis. 2024, 24, 351–360. [Google Scholar] [CrossRef]
- Vogel, A.B.; Lambert, L.; Kinnear, E.; Busse, D.; Erbar, S.; Reuter, K.C.; Wicke, L.; Perkovic, M.; Beissert, T.; Haas, H.; et al. Self-Amplifying RNA Vaccines Give Equivalent Protection against Influenza to mRNA Vaccines but at Much Lower Doses. Mol. Ther. J. Am. Soc. Gene Ther. 2018, 26, 446–455. [Google Scholar] [CrossRef]
- Sahin, U.; Karikó, K.; Türeci, Ö. mRNA-based therapeutics—Developing a new class of drugs. Nat. Rev. Drug Discov. 2014, 13, 759–780. [Google Scholar] [CrossRef]
- Houseley, J.; Tollervey, D. The many pathways of RNA degradation. Cell 2009, 136, 763–776. [Google Scholar] [CrossRef] [PubMed]
- Pourseif, M.M.; Masoudi-Sobhanzadeh, Y.; Azari, E.; Parvizpour, S.; Barar, J.; Ansari, R.; Omidi, Y. Self-amplifying mRNA vaccines: Mode of action, design, development and optimization. Drug Discov. Today 2022, 27, 103341. [Google Scholar] [CrossRef] [PubMed]
- Beissert, T.; Perkovic, M.; Vogel, A.; Erbar, S.; Walzer, K.C.; Hempel, T.; Brill, S.; Haefner, E.; Becker, R.; Türeci, Ö.; et al. A Trans-amplifying RNA Vaccine Strategy for Induction of Potent Protective Immunity. Mol. Ther. J. Am. Soc. Gene Ther. 2020, 28, 119–128. [Google Scholar] [CrossRef]
- Hyde, J.L.; Chen, R.; Trobaugh, D.W.; Diamond, M.S.; Weaver, S.C.; Klimstra, W.B.; Wilusz, J. The 5′ and 3′ ends of alphavirus RNAs--Non-coding is not non-functional. Virus Res. 2015, 206, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Pietilä, M.K.; Hellström, K.; Ahola, T. Alphavirus polymerase and RNA replication. Virus Res. 2017, 234, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Lundstrom, K. Alphaviruses in gene therapy. Viruses 2015, 7, 2321–2333. [Google Scholar] [CrossRef] [PubMed]
- Meshram, C.D.; Phillips, A.T.; Lukash, T.; Shiliaev, N.; Frolova, E.I.; Frolov, I. Mutations in Hypervariable Domain of Venezuelan Equine Encephalitis Virus nsP3 Protein Differentially Affect Viral Replication. J. Virol. 2020, 94, e01841-19. [Google Scholar] [CrossRef] [PubMed]
- Götte, B.; Liu, L.; McInerney, G.M. The Enigmatic Alphavirus Non-Structural Protein 3 (nsP3) Revealing Its Secrets at Last. Viruses 2018, 10, 105. [Google Scholar] [CrossRef]
- Ou, J.H.; Strauss, E.G.; Strauss, J.H. The 5′-terminal sequences of the genomic RNAs of several alphaviruses. J. Mol. Biol. 1983, 168, 1–15. [Google Scholar] [CrossRef]
- Ou, J.H.; Rice, C.M.; Dalgarno, L.; Strauss, E.G.; Strauss, J.H. Sequence studies of several alphavirus genomic RNAs in the region containing the start of the subgenomic RNA. Proc. Natl. Acad. Sci. USA 1982, 79, 5235–5239. [Google Scholar] [CrossRef]
- Levis, R.; Schlesinger, S.; Huang, H.V. Promoter for Sindbis virus RNA-dependent subgenomic RNA transcription. J. Virol. 1990, 64, 1726–1733. [Google Scholar] [CrossRef]
- Ou, J.H.; Strauss, E.G.; Strauss, J.H. Comparative studies of the 3′-terminal sequences of several alpha virus RNAs. Virology 1981, 109, 281–289. [Google Scholar] [CrossRef]
- Denison, M.R.; Graham, R.L.; Donaldson, E.F.; Eckerle, L.D.; Baric, R.S. Coronaviruses: An RNA proofreading machine regulates replication fidelity and diversity. RNA Biol. 2011, 8, 270–279. [Google Scholar] [CrossRef]
- Huang, A.S.; Baltimore, D. Defective viral particles and viral disease processes. Nature 1970, 226, 325–327. [Google Scholar] [CrossRef] [PubMed]
- Schlesinger, S.; Schlesinger, M.; Burge, B.W. Defective virus particles from Sindbis virus. Virology 1972, 48, 615–617. [Google Scholar] [CrossRef]
- Stark, C.; Kennedy, S.I. The generation and propagation of defective-interfering particles of Semliki Forest virus in different cell types. Virology 1978, 89, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Lehtovaara, P.; Söderlund, H.; Keränen, S.; Pettersson, R.F.; Kääriäinen, L. 18S defective interfering RNA of Semliki Forest virus contains a triplicated linear repeat. Proc. Natl. Acad. Sci. USA 1981, 78, 5353–5357. [Google Scholar] [CrossRef] [PubMed]
- Levis, R.; Weiss, B.G.; Tsiang, M.; Huang, H.; Schlesinger, S. Deletion mapping of Sindbis virus DI RNAs derived from cDNAs defines the sequences essential for replication and packaging. Cell 1986, 44, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Weiss, B.; Schlesinger, S. Defective interfering particles of Sindbis virus do not interfere with the homologous virus obtained from persistently infected BHK cells but do interfere with Semliki Forest virus. J. Virol. 1981, 37, 840–844. [Google Scholar] [CrossRef] [PubMed]
- Xiong, C.; Levis, R.; Shen, P.; Schlesinger, S.; Rice, C.M.; Huang, H.V. Sindbis virus: An efficient, broad host range vector for gene expression in animal cells. Science 1989, 243, 1188–1191. [Google Scholar] [CrossRef] [PubMed]
- Levis, R.; Huang, H.; Schlesinger, S. Engineered defective interfering RNAs of Sindbis virus express bacterial chloramphenicol acetyltransferase in avian cells. Proc. Natl. Acad. Sci. USA 1987, 84, 4811–4815. [Google Scholar] [CrossRef] [PubMed]
- Liljeström, P.; Garoff, H. A new generation of animal cell expression vectors based on the Semliki Forest virus replicon. Bio/Technol. 1991, 9, 1356–1361. [Google Scholar] [CrossRef]
- Bredenbeek, P.J.; Frolov, I.; Rice, C.M.; Schlesinger, S. Sindbis virus expression vectors: Packaging of RNA replicons by using defective helper RNAs. J. Virol. 1993, 67, 6439–6446. [Google Scholar] [CrossRef]
- Pushko, P.; Parker, M.; Ludwig, G.V.; Davis, N.L.; Johnston, R.E.; Smith, J.F. Replicon-helper systems from attenuated Venezuelan equine encephalitis virus: Expression of heterologous genes in vitro and immunization against heterologous pathogens in vivo. Virology 1997, 239, 389–401. [Google Scholar] [CrossRef] [PubMed]
- Frolov, I.; Frolova, E.; Schlesinger, S. Sindbis virus replicons and Sindbis virus: Assembly of chimeras and of particles deficient in virus RNA. J. Virol. 1997, 71, 2819–2829. [Google Scholar] [CrossRef]
- Smerdou, C.; Liljeström, P. Two-helper RNA system for production of recombinant Semliki forest virus particles. J. Virol. 1999, 73, 1092–1098. [Google Scholar] [CrossRef] [PubMed]
- Lundstrom, K. Replicon RNA Viral Vectors as Vaccines. Vaccines 2016, 4, 39. [Google Scholar] [CrossRef] [PubMed]
- Kamrud, K.I.; Alterson, K.; Custer, M.; Dudek, J.; Goodman, C.; Owens, G.; Smith, J.F. Development and characterization of promoterless helper RNAs for the production of alphavirus replicon particle. J. Gen. Virol. 2010, 91, 1723–1727. [Google Scholar] [CrossRef] [PubMed]
- Monroe, S.S.; Schlesinger, S. Common and distinct regions of defective-interfering RNAs of Sindbis virus. J. Virol. 1984, 49, 865–872. [Google Scholar] [CrossRef] [PubMed]
- Frolov, I.; Hardy, R.; Rice, C.M. Cis-acting RNA elements at the 5′ end of Sindbis virus genome RNA regulate minus- and plus-strand RNA synthesis. RNA 2001, 7, 1638–1651. [Google Scholar] [CrossRef] [PubMed]
- Monroe, S.S.; Schlesinger, S. RNAs from two independently isolated defective interfering particles of Sindbis virus contain a cellular tRNA sequence at their 5′ ends. Proc. Natl. Acad. Sci. USA 1983, 80, 3279–3283. [Google Scholar] [CrossRef]
- Gorchakov, R.; Hardy, R.; Rice, C.M.; Frolov, I. Selection of functional 5′ cis-acting elements promoting efficient sindbis virus genome replication. J. Virol. 2004, 78, 61–75. [Google Scholar] [CrossRef]
- Kulasegaran-Shylini, R.; Atasheva, S.; Gorenstein, D.G.; Frolov, I. Structural and functional elements of the promoter encoded by the 5′ untranslated region of the Venezuelan equine encephalitis virus genome. J. Virol. 2009, 83, 8327–8339. [Google Scholar] [CrossRef]
- Kulasegaran-Shylini, R.; Thiviyanathan, V.; Gorenstein, D.G.; Frolov, I. The 5′UTR-specific mutation in VEEV TC-83 genome has a strong effect on RNA replication and subgenomic RNA synthesis, but not on translation of the encoded proteins. Virology 2009, 387, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Shirako, Y.; Strauss, E.G.; Strauss, J.H. Modification of the 5′ terminus of Sindbis virus genomic RNA allows nsP4 RNA polymerases with nonaromatic amino acids at the N terminus to function in RNA replication. J. Virol. 2003, 77, 2301–2309. [Google Scholar] [CrossRef]
- Hardy, R.W.; Rice, C.M. Requirements at the 3′ end of the sindbis virus genome for efficient synthesis of minus-strand RNA. J. Virol. 2005, 79, 4630–4639. [Google Scholar] [CrossRef] [PubMed]
- Hardy, R.W. The role of the 3′ terminus of the Sindbis virus genome in minus-strand initiation site selection. Virology 2006, 345, 520–531. [Google Scholar] [CrossRef]
- Lello, L.S.; Utt, A.; Bartholomeeusen, K.; Wang, S.; Rausalu, K.; Kendall, C.; Coppens, S.; Fragkoudis, R.; Tuplin, A.; Alphey, L.; et al. Cross-utilisation of template RNAs by alphavirus replicases. PLoS Pathog. 2020, 16, e1008825. [Google Scholar] [CrossRef]
- Götte, B.; Utt, A.; Fragkoudis, R.; Merits, A.; McInerney, G.M. Sensitivity of Alphaviruses to G3BP Deletion Correlates with Efficiency of Replicase Polyprotein Processing. J. Virol. 2020, 94, e01681-19. [Google Scholar] [CrossRef]
- Merits, A.; Vasiljeva, L.; Ahola, T.; Kääriäinen, L.; Auvinen, P. Proteolytic processing of Semliki Forest virus-specific non-structural polyprotein by nsP2 protease. J. Gen. Virol. 2001, 82, 765–773. [Google Scholar] [CrossRef] [PubMed]
- Vasiljeva, L.; Valmu, L.; Kääriäinen, L.; Merits, A. Site-specific protease activity of the carboxyl-terminal domain of Semliki Forest virus replicase protein nsP2. J. Biol. Chem. 2001, 276, 30786–30793. [Google Scholar] [CrossRef]
- Vasiljeva, L.; Merits, A.; Golubtsov, A.; Sizemskaja, V.; Kääriäinen, L.; Ahola, T. Regulation of the sequential processing of Semliki Forest virus replicase polyprotein. J. Biol. Chem. 2003, 278, 41636–41645. [Google Scholar] [CrossRef]
- Lemm, J.A.; Rice, C.M. Assembly of functional Sindbis virus RNA replication complexes: Requirement for coexpression of P123 and P34. J. Virol. 1993, 67, 1905–1915. [Google Scholar] [CrossRef]
- Lemm, J.A.; Rice, C.M. Roles of nonstructural polyproteins and cleavage products in regulating Sindbis virus RNA replication and transcription. J. Virol. 1993, 67, 1916–1926. [Google Scholar] [CrossRef]
- Lemm, J.A.; Bergqvist, A.; Read, C.M.; Rice, C.M. Template-dependent initiation of Sindbis virus RNA replication in vitro. J. Virol. 1998, 72, 6546–6553. [Google Scholar] [CrossRef]
- Li, G.P.; Prágai, B.M.; Rice, C.M. Rescue of Sindbis virus-specific RNA replication and transcription by using a vaccinia virus recombinant. J. Virol. 1991, 65, 6714–6723. [Google Scholar] [CrossRef]
- Thal, M.A.; Wasik, B.R.; Posto, J.; Hardy, R.W. Template requirements for recognition and copying by Sindbis virus RNA-dependent RNA polymerase. Virology 2007, 358, 221–232. [Google Scholar] [CrossRef]
- Kallio, K.; Hellström, K.; Balistreri, G.; Spuul, P.; Jokitalo, E.; Ahola, T. Template RNA length determines the size of replication complex spherules for Semliki Forest virus. J. Virol. 2013, 87, 9125–9134. [Google Scholar] [CrossRef]
- Spuul, P.; Balistreri, G.; Hellström, K.; Golubtsov, A.V.; Jokitalo, E.; Ahola, T. Assembly of alphavirus replication complexes from RNA and protein components in a novel trans-replication system in mammalian cells. J. Virol. 2011, 85, 4739–4751. [Google Scholar] [CrossRef]
- Lello, L.S.; Bartholomeeusen, K.; Wang, S.; Coppens, S.; Fragkoudis, R.; Alphey, L.; Ariën, K.K.; Merits, A.; Utt, A. nsP4 Is a Major Determinant of Alphavirus Replicase Activity and Template Selectivity. J. Virol. 2021, 95, e0035521. [Google Scholar] [CrossRef]
- Scholte, F.E.M.; Tas, A.; Albulescu, I.C.; Žusinaite, E.; Merits, A.; Snijder, E.J.; van Hemert, M.J. Stress granule components G3BP1 and G3BP2 play a proviral role early in Chikungunya virus replication. J. Virol. 2015, 89, 4457–4469. [Google Scholar] [CrossRef]
- Utt, A.; Quirin, T.; Saul, S.; Hellström, K.; Ahola, T.; Merits, A. Versatile Trans-Replication Systems for Chikungunya Virus Allow Functional Analysis and Tagging of Every Replicase Protein. PLoS ONE 2016, 11, e0151616. [Google Scholar] [CrossRef] [PubMed]
- Rausalu, K.; Utt, A.; Quirin, T.; Varghese, F.S.; Žusinaite, E.; Das, P.K.; Ahola, T.; Merits, A. Chikungunya virus infectivity, RNA replication and non-structural polyprotein processing depend on the nsP2 protease’s active site cysteine residue. Sci. Rep. 2016, 6, 37124. [Google Scholar] [CrossRef] [PubMed]
- Cherkashchenko, L.; Rausalu, K.; Basu, S.; Alphey, L.; Merits, A. Expression of Alphavirus Nonstructural Protein 2 (nsP2) in Mosquito Cells Inhibits Viral RNA Replication in Both a Protease Activity-Dependent and -Independent Manner. Viruses 2022, 14, 1327. [Google Scholar] [CrossRef]
- Schütz, S.; Sarnow, P. Interaction of viruses with the mammalian RNA interference pathway. Virology 2006, 344, 151–157. [Google Scholar] [CrossRef]
- Kash, J.C.; Goodman, A.G.; Korth, M.J.; Katze, M.G. Hijacking of the host-cell response and translational control during influenza virus infection. Virus Res. 2006, 119, 111–120. [Google Scholar] [CrossRef]
- Froshauer, S.; Kartenbeck, J.; Helenius, A. Alphavirus RNA replicase is located on the cytoplasmic surface of endosomes and lysosomes. J. Cell Biol. 1988, 107, 2075–2086. [Google Scholar] [CrossRef]
- Friedman, R.M.; Levin, J.G.; Grimley, P.M.; Berezesky, I.K. Membrane-associated replication complex in arbovirus infection. J. Virol. 1972, 10, 504–515. [Google Scholar] [CrossRef]
- Grimley, P.M.; Levin, J.G.; Berezesky, I.K.; Friedman, R.M. Specific membranous structures associated with the replication of group A arboviruses. J. Virol. 1972, 10, 492–503. [Google Scholar] [CrossRef]
- Gorchakov, R.; Garmashova, N.; Frolova, E.; Frolov, I. Different types of nsP3-containing protein complexes in Sindbis virus-infected cells. J. Virol. 2008, 82, 10088–10101. [Google Scholar] [CrossRef]
- Tan, Y.B.; Chmielewski, D.; Law, M.C.Y.; Zhang, K.; He, Y.; Chen, M.; Jin, J.; Luo, D. Molecular architecture of the Chikungunya virus replication complex. Sci. Adv. 2022, 8, eadd2536. [Google Scholar] [CrossRef]
- Laurent, T.; Kumar, P.; Liese, S.; Zare, F.; Jonasson, M.; Carlson, A.; Carlson, L.-A. Architecture of the chikungunya virus replication organelle. eLife 2022, 11, e83042. [Google Scholar] [CrossRef] [PubMed]
- Frolova, E.I.; Gorchakov, R.; Pereboeva, L.; Atasheva, S.; Frolov, I. Functional Sindbis virus replicative complexes are formed at the plasma membrane. J. Virol. 2010, 84, 11679–11695. [Google Scholar] [CrossRef] [PubMed]
- Hellström, K.; Kallio, K.; Utt, A.; Quirin, T.; Jokitalo, E.; Merits, A.; Ahola, T. Partially Uncleaved Alphavirus Replicase Forms Spherule Structures in the Presence and Absence of RNA Template. J. Virol. 2017, 91, 10-1128. [Google Scholar] [CrossRef] [PubMed]
- Lemm, J.A.; Rümenapf, T.; Strauss, E.G.; Strauss, J.H.; Rice, C.M. Polypeptide requirements for assembly of functional Sindbis virus replication complexes: A model for the temporal regulation of minus- and plus-strand RNA synthesis. EMBO J. 1994, 13, 2925–2934. [Google Scholar] [CrossRef] [PubMed]
- Blakney, A.K.; McKay, P.F.; Shattock, R.J. Structural Components for Amplification of Positive and Negative Strand VEEV Splitzicons. Front. Mol. Biosci. 2018, 5, 71. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.; Haefner, E.; Gerbeth, J.; Beissert, T.; Sahin, U.; Perkovic, M.; Schnierle, B.S. A taRNA vaccine candidate induces a specific immune response that protects mice against Chikungunya virus infections. Molecular therapy. Nucleic Acids 2022, 28, 743–754. [Google Scholar] [CrossRef]
- Schmidt, C.; Hastert, F.D.; Gerbeth, J.; Beissert, T.; Sahin, U.; Perkovic, M.; Schnierle, B.S. A Bivalent Trans-Amplifying RNA Vaccine Candidate Induces Potent Chikungunya and Ross River Virus Specific Immune Responses. Vaccines 2022, 10, 1374. [Google Scholar] [CrossRef] [PubMed]
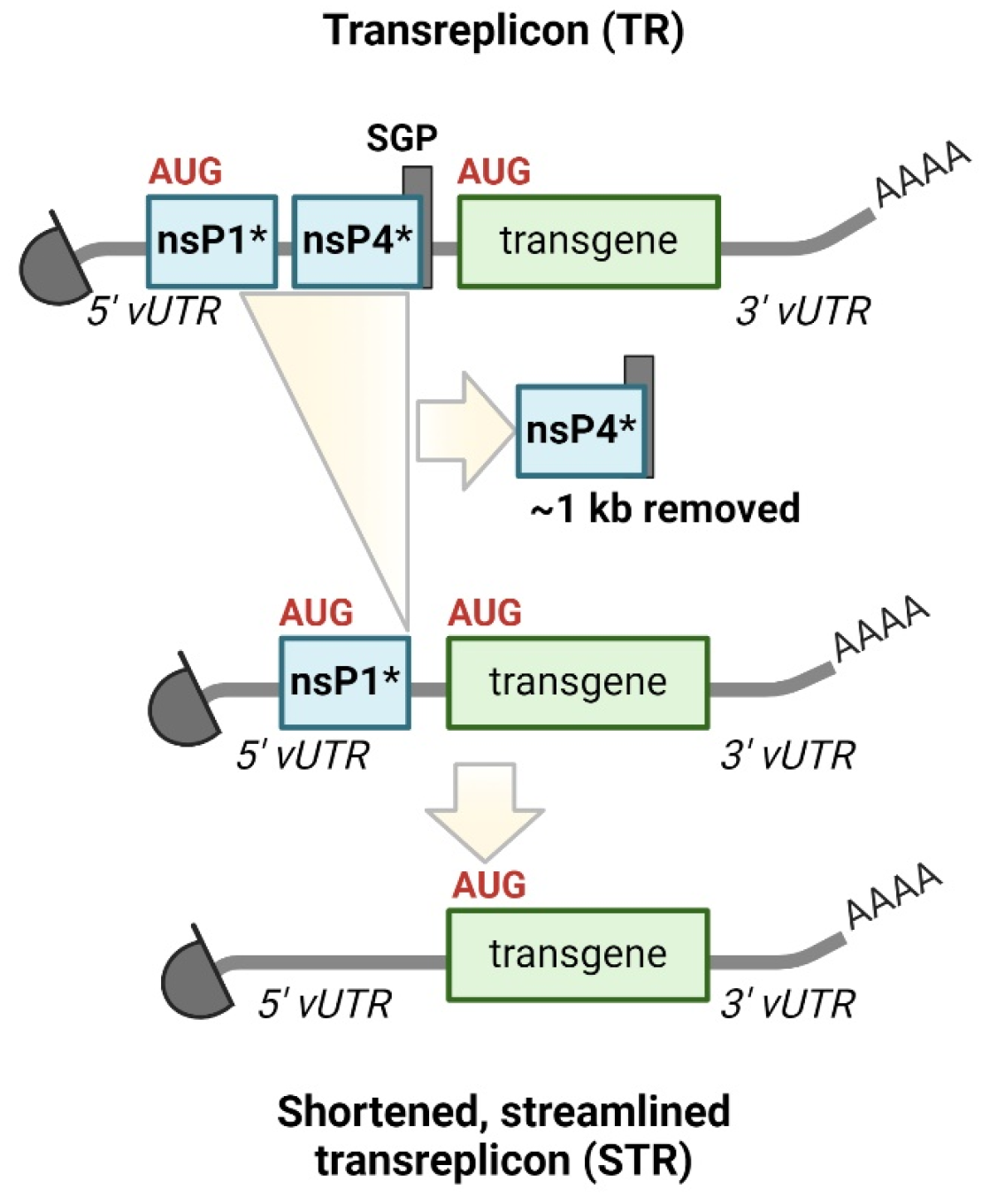
- Perkovic, M.; Gawletta, S.; Hempel, T.; Brill, S.; Nett, E.; Sahin, U.; Beissert, T. A trans-amplifying RNA simplified to essential elements is highly replicative and robustly immunogenic in mice. Mol. Ther. J. Am. Soc. Gene Ther. 2023, 31, 2297. [Google Scholar] [CrossRef] [PubMed]
- Kremsner, P.G.; Mann, P.; Kroidl, A.; Leroux-Roels, I.; Schindler, C.; Gabor, J.J.; Schunk, M.; Leroux-Roels, G.; Bosch, J.J.; Fendel, R.; et al. Safety and immunogenicity of an mRNA-lipid nanoparticle vaccine candidate against SARS-CoV-2: A phase 1 randomized clinical trial. Wien. Klin. Wochenschr. 2021, 133, 931–941. [Google Scholar] [CrossRef]
- Kremsner, P.G.; Ahuad Guerrero, R.A.; Arana-Arri, E.; Aroca Martinez, G.J.; Bonten, M.; Chandler, R.; Corral, G.; Block, E.J.L.; de Ecker, L.; Gabor, J.J.; et al. Efficacy and safety of the CVnCoV SARS-CoV-2 mRNA vaccine candidate in ten countries in Europe and Latin America (HERALD): A randomised, observer-blinded, placebo-controlled, phase 2b/3 trial. The Lancet. Infect. Dis. 2022, 22, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Fenton, C.; Lee, A. mRNA COVID-19 vaccines are well tolerated and myopericarditis is a rare adverse event following immunisation. Drugs Ther. Perspect. Ration. Drug Sel. Use 2022, 38, 532–540. [Google Scholar] [CrossRef]
- Kouhpayeh, H.; Ansari, H. Adverse events following COVID-19 vaccination: A systematic review and meta-analysis. Int. Immunopharmacol. 2022, 109, 108906. [Google Scholar] [CrossRef]
- Erasmus, J.H.; Khandhar, A.P.; O’Connor, M.A.; Walls, A.C.; Hemann, E.A.; Murapa, P.; Archer, J.; Leventhal, S.; Fuller, J.T.; Lewis, T.B.; et al. An Alphavirus-derived replicon RNA vaccine induces SARS-CoV-2 neutralizing antibody and T cell responses in mice and nonhuman primates. Sci. Transl. Med. 2020, 12, eabc9396. [Google Scholar] [CrossRef] [PubMed]
- McGee, J.E.; Kirsch, J.R.; Kenney, D.; Chavez, E.; Shih, T.-Y.; Douam, F.; Wong, W.W.; Grinstaff, M.W. Complete substitution with modified nucleotides suppresses the early interferon response and increases the potency of self-amplifying RNA. bioRxiv 2023. [Google Scholar] [CrossRef]
- Komori, M.; Morey, A.L.; Quiñones-Molina, A.A.; Fofana, J.; Romero, L.; Peters, E.; Matsuda, K.; Gummuluru, S.; Smith, J.F.; Akahata, W.; et al. Incorporation of 5 methylcytidine alleviates innate immune response to self-amplifying RNA vaccine. bioRxiv 2023. [Google Scholar] [CrossRef]
- Arevalo, C.P.; Bolton, M.J.; Le Sage, V.; Ye, N.; Furey, C.; Muramatsu, H.; Alameh, M.-G.; Pardi, N.; Drapeau, E.M.; Parkhouse, K.; et al. A multivalent nucleoside-modified mRNA vaccine against all known influenza virus subtypes. Science 2022, 378, 899–904. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yıldız, A.; Răileanu, C.; Beissert, T. Trans-Amplifying RNA: A Journey from Alphavirus Research to Future Vaccines. Viruses 2024, 16, 503. https://doi.org/10.3390/v16040503
Yıldız A, Răileanu C, Beissert T. Trans-Amplifying RNA: A Journey from Alphavirus Research to Future Vaccines. Viruses. 2024; 16(4):503. https://doi.org/10.3390/v16040503
Chicago/Turabian StyleYıldız, Ayşegül, Cristian Răileanu, and Tim Beissert. 2024. "Trans-Amplifying RNA: A Journey from Alphavirus Research to Future Vaccines" Viruses 16, no. 4: 503. https://doi.org/10.3390/v16040503
APA StyleYıldız, A., Răileanu, C., & Beissert, T. (2024). Trans-Amplifying RNA: A Journey from Alphavirus Research to Future Vaccines. Viruses, 16(4), 503. https://doi.org/10.3390/v16040503





