SARS-CoV-2-Induced TSLP Is Associated with Duration of Hospital Stay in COVID-19 Patients
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population for Collection of Plasma Samples
2.2. Inclusion and Exclusion Criteria
2.3. Collection of Blood Samples
2.4. Normal Human Bronchial Epithelial (NHBE) Cell Cultures
2.5. Human Bronchial Epithelial Cells (HBECs) from Bronchial Brushings Derived from Non-Infected Participants
2.6. Assays for Cytokines
2.7. Immunofluorescence for TSLP
2.8. Statistical Analysis
3. Results
3.1. Demographic and Clinical Profiles
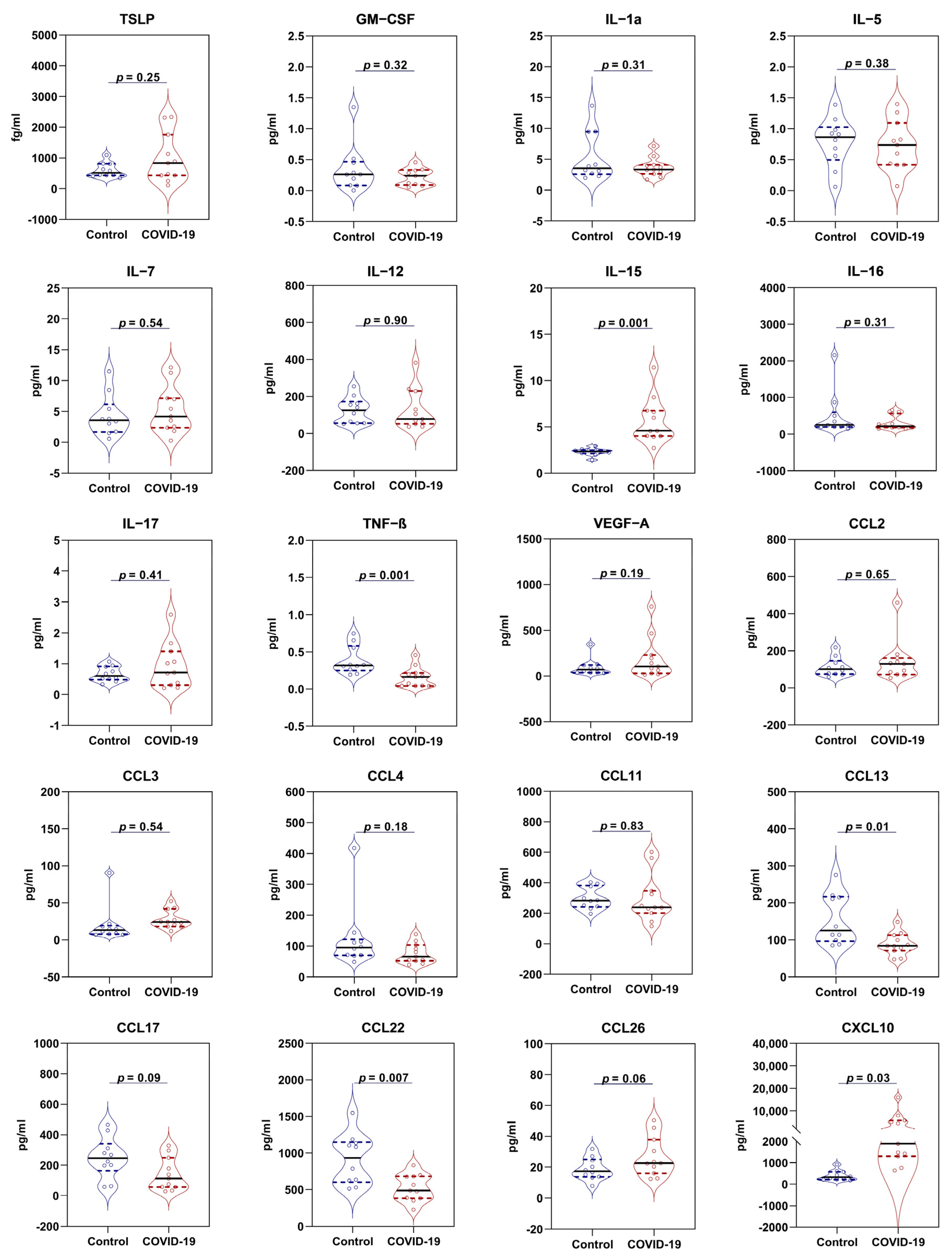
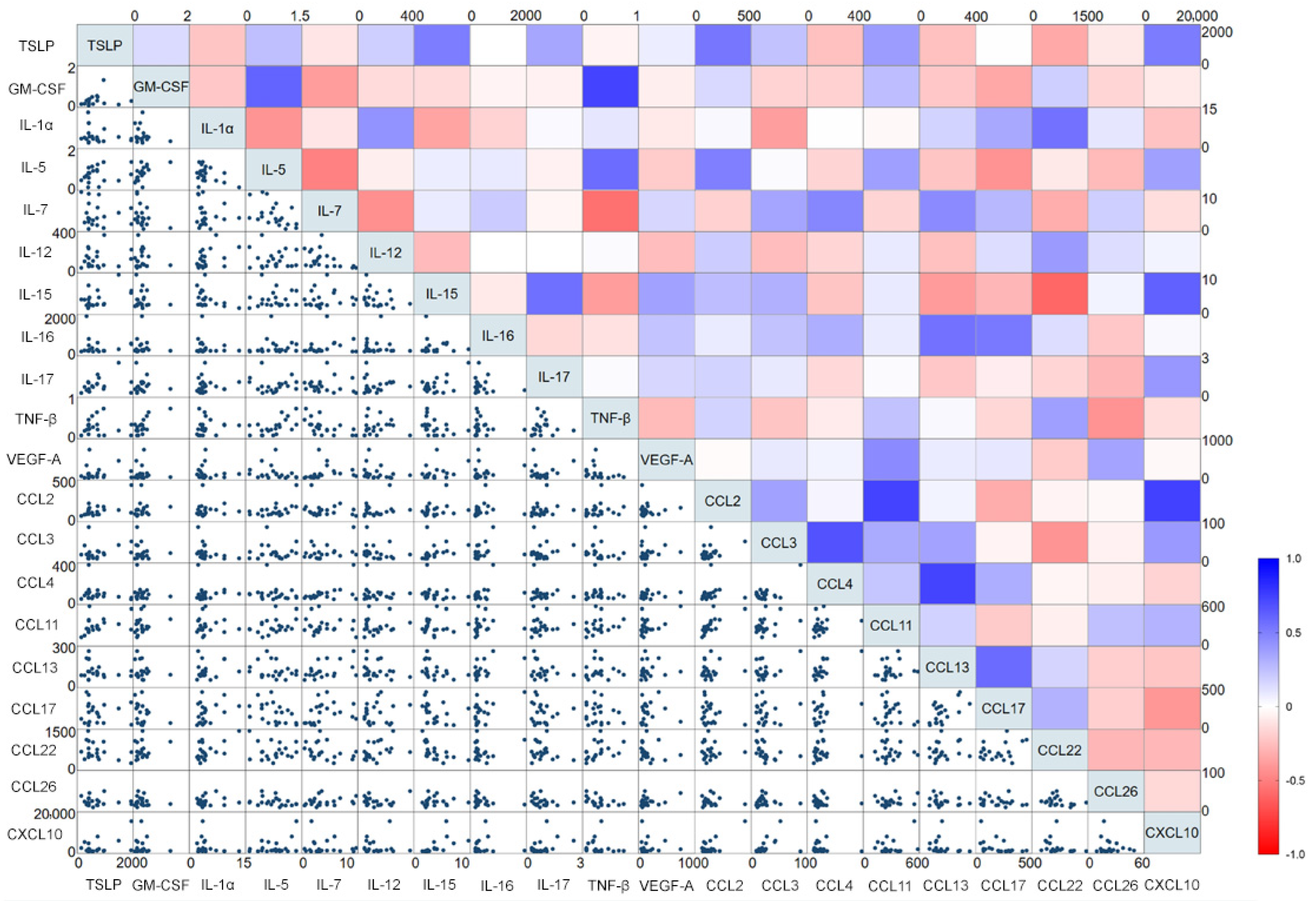
3.2. Cytokine Profile
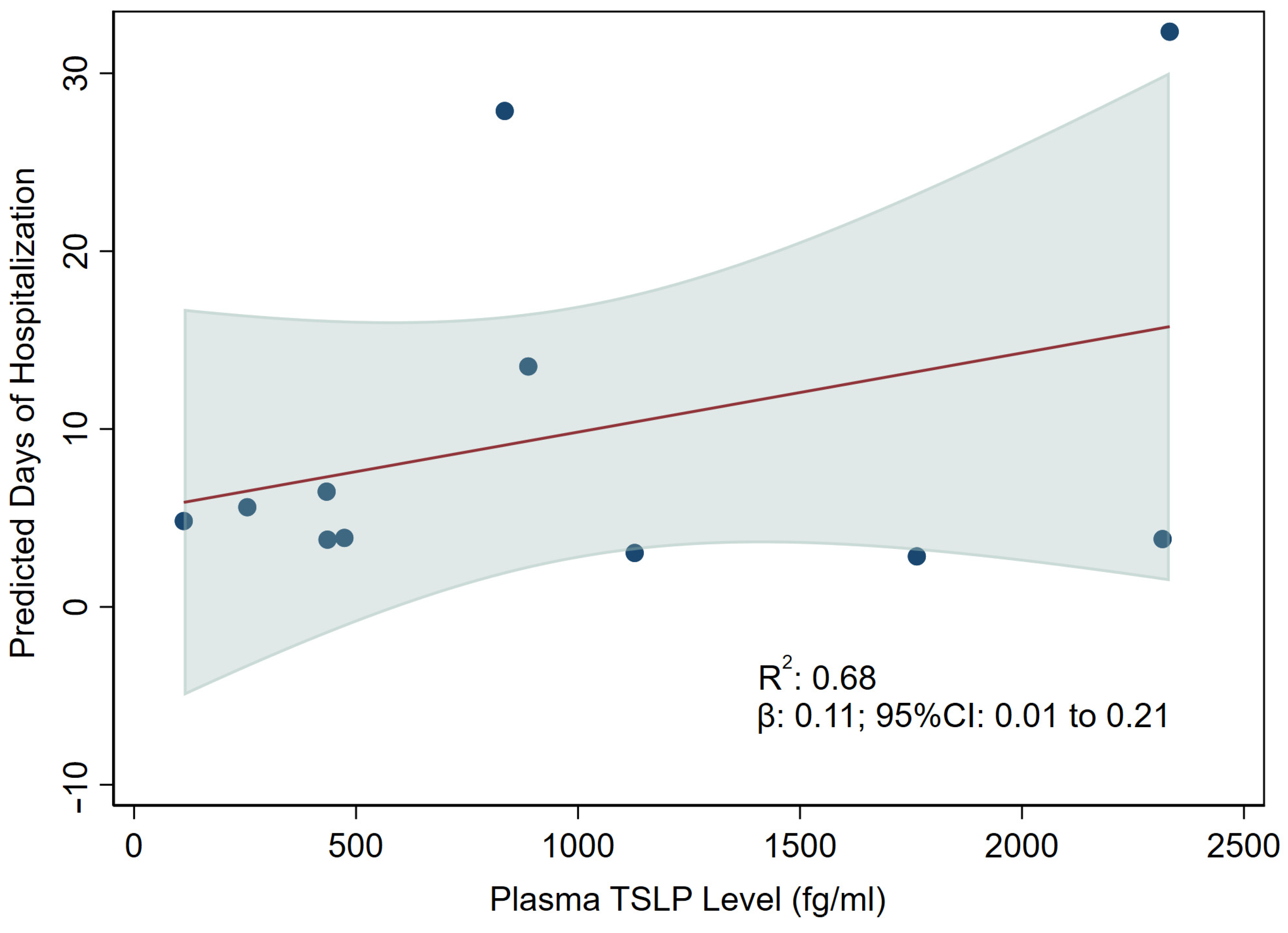
3.3. Association between TSLP and Hospitalization
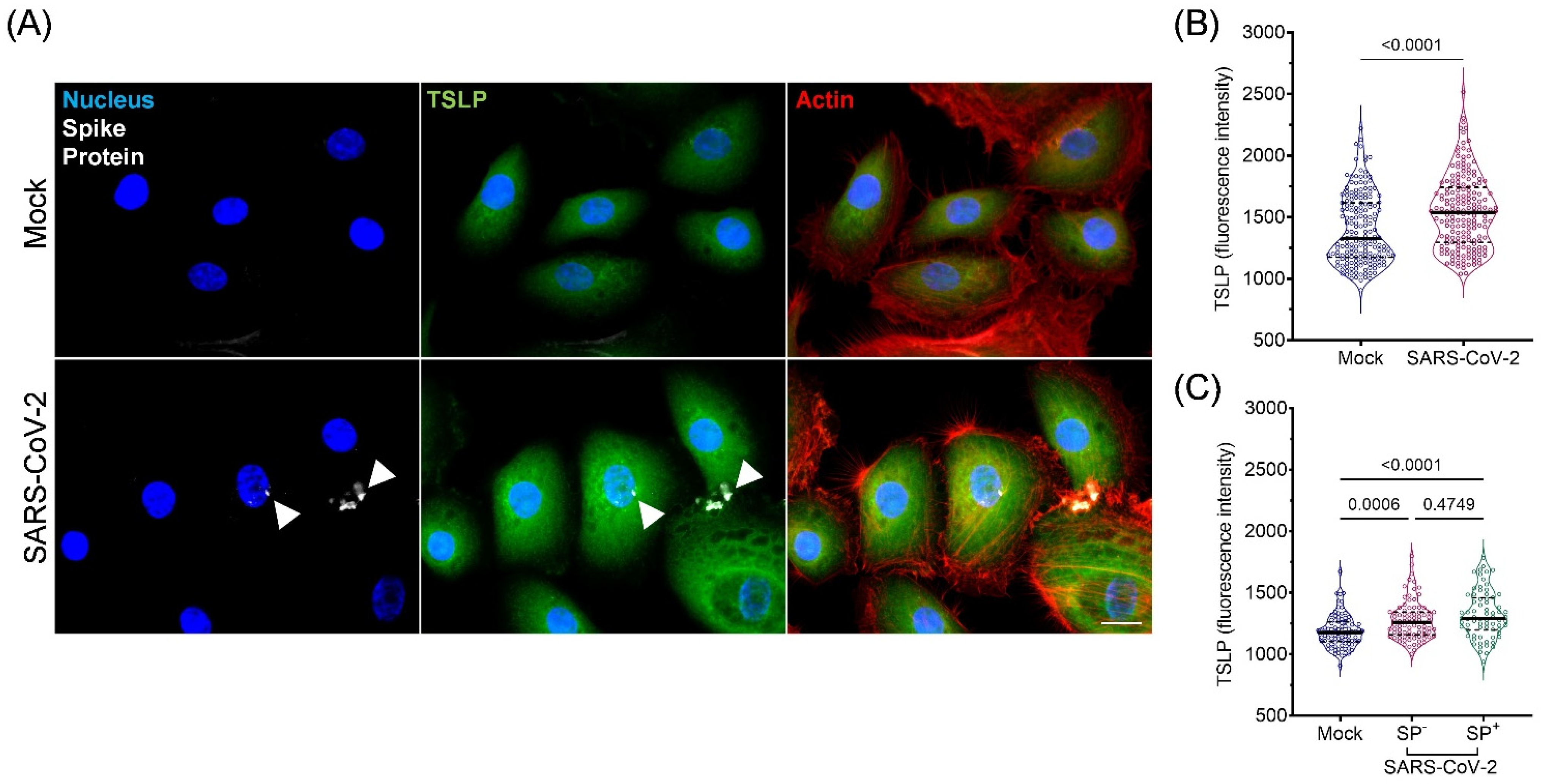
3.4. Expression and Secretion of TSLP in Cultured AIRWAY Epithelial Cells during SARS-CoV-2 Infection
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Ethics Statement
References
- Leonard, W.J. TSLP: Finally in the limelight. Nat. Immunol. 2002, 3, 605–607. [Google Scholar] [CrossRef] [PubMed]
- Rochman, Y.; Spolski, R.; Leonard, W.J. New insights into the regulation of T cells by gamma(c) family cytokines. Nat. Rev. Immunol. 2009, 9, 480–490. [Google Scholar] [CrossRef] [PubMed]
- Corren, J.; Ziegler, S.F. TSLP: From allergy to cancer. Nat. Immunol. 2019, 20, 1603–1609. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Melo, D.; Nilsson-Payant, B.E.; Liu, W.C.; Uhl, S.; Hoagland, D.; Møller, R.; Jordan, T.X.; Oishi, K.; Panis, M.; Sachs, D.; et al. Imbalanced Host Response to SARS-CoV-2 Drives Development of COVID-19. Cell 2020, 181, 1036–1045.e9. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.G.; Simpson, L.J.; Ferreira, A.-M.; Rustagi, A.; Roque, J.; Asuni, A.; Ranganath, T.; Grant, P.M.; Subramanian, A.; Rosenberg-Hasson, Y.; et al. Cytokine profile in plasma of severe COVID-19 does not differ from ARDS and sepsis. J. Clin Investig. 2020, 5, e140289. [Google Scholar] [CrossRef]
- Copaescu, A.; Smibert, O.; Gibson, A.; Phillips, E.J.; Trubiano, J.A. The role of IL-6 and other mediators in the cytokine storm associated with SARS-CoV-2 infection. J. Allergy. Clin Immunol. 2020, 146, 518–534. [Google Scholar] [CrossRef]
- Curatola, A.; Chiaretti, A.; Ferretti, S.; Bersani, G.; Lucchetti, D.; Capossela, L.; Sgambato, A.; Gatto, A. Cytokine Response to SARS-CoV-2 Infection in Children. Viruses 2021, 13, 1868. [Google Scholar] [CrossRef]
- Kato, A.; Favoreto, S., Jr.; Avila, P.C.; Schleimer, R.P. TLR3- and Th2 cytokine-dependent production of thymic stromal lymphopoietin in human airway epithelial cells. J. Immunol. 2007, 179, 1080–1087. [Google Scholar] [CrossRef]
- Yoshikawa, T.; Hill, T.; Li, K.; Peters, C.J.; Tseng, C.-T.K. Severe Acute Respiratory Syndrome (SARS) Coronavirus-Induced Lung Epithelial Cytokines Exacerbate SARS Pathogenesis by Modulating Intrinsic Functions of Monocyte-Derived Macrophages and Dendritic Cells. J. Virol. 2009, 83, 3039–3048. [Google Scholar] [CrossRef]
- Vareille, M.; Kieninger, E.; Edwards, M.R.; Regamey, N. The airway epithelium: Soldier in the fight against respiratory viruses. Clin Microbiol. Rev. 2011, 24, 210–229. [Google Scholar] [CrossRef]
- García-García, M.L.; Calvo, C.; Moreira, A.; Cañas, J.A.; Pozo, F.; Sastre, B.; Quevedo, S.; Casas, I.; Del Pozo, V. Thymic stromal lymphopoietin, IL-33, and periostin in hospitalized infants with viral bronchiolitis. Medicine 2017, 96, e6787. [Google Scholar] [CrossRef] [PubMed]
- Perez, G.F.; Pancham, K.; Huseni, S.; Preciado, D.; Freishtat, R.J.; Colberg-Poley, A.M.; Hoffman, E.P.; Rose, M.C.; Nino, G. Rhinovirus infection in young children is associated with elevated airway TSLP levels. Eur. Respir. J. 2014, 44, 1075–1078. [Google Scholar] [CrossRef] [PubMed]
- Lucas, C.; Wong, P.; Klein, J.; Castro, T.B.R.; Silva, J.; Sundaram, M.; Ellingson, M.K.; Mao, T.; Oh, J.E.; Israelow, B.; et al. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature 2020, 584, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Caterino, M.; Gelzo, M.; Sol, S.; Fedele, R.; Annunziata, A.; Calabrese, C.; Fiorentino, G.; D’Abbraccio, M.; Dell’Isola, C.; Fusco, F.M.; et al. Dysregulation of lipid metabolism and pathological inflammation in patients with COVID-19. Sci. Rep. 2021, 11, 2941. [Google Scholar] [CrossRef]
- Choreño-Parra, J.A.; Jiménez-Álvarez, L.A.; Cruz-Lagunas, A.; Rodríguez-Reyna, T.S.; Ramírez-Martínez, G.; Sandoval-Vega, M.; Hernández-García, D.L.; Choreño-Parra, E.M.; Balderas-Martínez, Y.I.; Martinez-Sánchez, M.E.; et al. Clinical and Immunological Factors That Distinguish COVID-19 From Pandemic Influenza A(H1N1). Front. Immunol. 2021, 12, 593595. [Google Scholar] [CrossRef]
- Akaike, H. Canonical Correlation Analysis of Time Series and the Use of an Information Criterion. In System Identification Advances and Case Studies; Mehra, R.K., Lainiotis, D.G., Eds.; Academic Press Inc.: New York, NY, USA, 1976; pp. 27–96. [Google Scholar] [CrossRef]
- Zeuthen, L.H.; Fink, L.N.; Frokiaer, H. Epithelial cells prime the immune response to an array of gut-derived commensals towards a tolerogenic phenotype through distinct actions of thymic stromal lymphopoietin and transforming growth factor-beta. Immunology 2008, 123, 197–208. [Google Scholar] [CrossRef]
- Xu, G.; Zhang, L.; Wang, D.Y.; Xu, R.; Liu, Z.; Han, D.M.; Wang, X.D.; Zuo, K.J.; Li, H.B. Opposing roles of IL-17A and IL-25 in the regulation of TSLP production in human nasal epithelial cells. Allergy 2010, 65, 581–589. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280.e8. [Google Scholar] [CrossRef]
- Sungnak, W.; Huang, N.; Bécavin, C.; Berg, M.; Queen, R.; Litvinukova, M.; Talavera-López, C.; Maatz, H.; Reichart, D.; Sampaziotis, F.; et al. SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nat Med. 2020, 26, 681–687. [Google Scholar] [CrossRef]
- Votto, M.; Castagnoli, R.; Marseglia, G.L.; Licari, A.; Brambilla, I. COVID-19 and autoimmune diseases: Is there a connection? Curr. Opin. Allergy Clin Immunol. 2023, 10, 1097. [Google Scholar] [CrossRef]
- Inaba, H.; Aizawa, T. Coronavirus Disease 2019 and the Thyroid—Progress and Perspectives. Front. Endocrinol. 2021, 12, 708333. [Google Scholar] [CrossRef] [PubMed]
- Vahabi, M.; Ghazanfari, T.; Sepehrnia, S. Molecular mimicry, hyperactive immune system, and SARS-COV-2 are three prerequisites of the autoimmune disease triangle following COVID-19 infection. Int. Immunopharmacol. 2022, 112, 109183. [Google Scholar] [CrossRef] [PubMed]
- Garg, Y.; Kakria, N.; Singh, S.; Jindal, A. Coronavirus disease-19 triggered systemic lupus erythematous: A novel entity. Lung India 2023, 40, 79. [Google Scholar] [CrossRef] [PubMed]
- Lazarian, G.; Quinquenel, A.; Bellal, M.; Siavellis, J.; Jacquy, C.; Re, D.; Merabet, F.; Mekinian, A.; Braun, T.; Damaj, G.; et al. Autoimmune haemolytic anaemia associated with COVID-19 infection. Br. J. Haematol. 2020, 190, 29–31. [Google Scholar] [CrossRef]
- Astrakhan, A.; Omori, M.; Nguyen, T.; Becker-Herman, S.; Iseki, M.; Aye, T.; Hudkins, K.; Dooley, J.; Farr, A.; Alpers, C.E.; et al. Local increase in thymic stromal lymphopoietin induces systemic alterations in B cell development. Nat. Immunol. 2007, 8, 522–531. [Google Scholar] [CrossRef]
- Yan, B.; Freiwald, T.; Chauss, D.; Wang, L.; West, E.; Mirabelli, C.; Zhang, C.J.; Nichols, E.M.; Malik, N.; Gregory, R.; et al. SARS-CoV-2 drives JAK1/2-dependent local complement hyperactivation. Sci. Immunol. 2021, 6, eabg0833. [Google Scholar] [CrossRef]
- Singh, A.K.; Kasarpalkar, N.; Bhowmick, S.; Paradkar, G.; Talreja, M.; Shah, K.; Tiwari, A.; Palav, H.; Kaginkar, S.; Kulkarni, R.; et al. Opposing roles for sMAdCAM and IL-15 in COVID-19 associated cellular immune pathology. J. Leukoc. Biol. 2022, 111, 1287–1295. [Google Scholar] [CrossRef]
- Tato, C.M.; Cua, D.J. SnapShot: Cytokines, I. Cell 2008, 132, 324.e1. [Google Scholar] [CrossRef]
- Ozger, H.S.; Karakus, R.; Kuscu, E.N.; Bagriacik, U.E.; Oruklu, N.; Yaman, M.; Turkoglu, M.; Erbas, G.; Atak, A.Y.; Senol, E. Serial measurement of cytokines strongly predict COVID-19 outcome. PLoS ONE 2021, 16, e0260623. [Google Scholar] [CrossRef]
- Coperchini, F.; Chiovato, L.; Rotondi, M. Interleukin-6, CXCL10 and Infiltrating Macrophages in COVID-19-Related Cytokine Storm: Not One for All But All for One! Front. Immunol. 2021, 12, 668507. [Google Scholar] [CrossRef]
- Lorè, N.I.; De Lorenzo, R.; Rancoita, P.M.V.; Cugnata, F.; Agresti, A.; Benedetti, F.; Bianchi, M.E.; Bonini, C.; Capobianco, A.; Conte, C.; et al. CXCL10 levels at hospital admission predict COVID-19 outcome: Hierarchical assessment of 53 putative inflammatory biomarkers in an observational study. Mol. Med. 2021, 27, 129. [Google Scholar] [CrossRef] [PubMed]
- Abers, M.S.; Delmonte, O.M.; Ricotta, E.E.; Fintzi, J.; Fink, D.L.; de Jesus, A.A.A.; Zarember, K.A.; Alehashemi, S.; Oikonomou, V.; Desai, J.V.; et al. An immune-based biomarker signature is associated with mortality in COVID-19 patients. J. Clin. Investig. 2021, 6, e144455. [Google Scholar] [CrossRef] [PubMed]
- Desvaux, E.; Hamon, A.; Hubert, S.; Boudjeniba, C.; Chassagnol, B.; Swindle, J.; Aussy, A.; Laigle, L.; Laplume, J.; Soret, P.; et al. Network-based repurposing identifies anti-alarmins as drug candidates to control severe lung inflammation in COVID-19. PLoS ONE 2021, 16, e0254374. [Google Scholar] [CrossRef] [PubMed]
- Fornasa, G.; Tsilingiri, K.; Caprioli, F.; Botti, F.; Mapelli, M.; Meller, S.; Kislat, A.; Homey, B.; Di Sabatino, A.; Sonzogni, A.M.; et al. Dichotomy of short and long thymic stromal lymphopoietin isoforms in inflammatory disorders of the bowel and skin. J. Allergy Clin. Immunol. 2015, 136, 413–422. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gerla, L.; Moitra, S.; Pink, D.; Govindasamy, N.; Duchesne, M.; Reklow, E.; Hillaby, A.; May, A.; Lewis, J.D.; Melenka, L.; et al. SARS-CoV-2-Induced TSLP Is Associated with Duration of Hospital Stay in COVID-19 Patients. Viruses 2023, 15, 556. https://doi.org/10.3390/v15020556
Gerla L, Moitra S, Pink D, Govindasamy N, Duchesne M, Reklow E, Hillaby A, May A, Lewis JD, Melenka L, et al. SARS-CoV-2-Induced TSLP Is Associated with Duration of Hospital Stay in COVID-19 Patients. Viruses. 2023; 15(2):556. https://doi.org/10.3390/v15020556
Chicago/Turabian StyleGerla, Luke, Subhabrata Moitra, Desmond Pink, Natasha Govindasamy, Marc Duchesne, Eileen Reklow, Angela Hillaby, Amy May, John D. Lewis, Lyle Melenka, and et al. 2023. "SARS-CoV-2-Induced TSLP Is Associated with Duration of Hospital Stay in COVID-19 Patients" Viruses 15, no. 2: 556. https://doi.org/10.3390/v15020556
APA StyleGerla, L., Moitra, S., Pink, D., Govindasamy, N., Duchesne, M., Reklow, E., Hillaby, A., May, A., Lewis, J. D., Melenka, L., Hobman, T. C., Mayers, I., & Lacy, P. (2023). SARS-CoV-2-Induced TSLP Is Associated with Duration of Hospital Stay in COVID-19 Patients. Viruses, 15(2), 556. https://doi.org/10.3390/v15020556







