Subjective and Objective Cognitive Impairments in Non-Hospitalized Persons 9 Months after SARS-CoV-2 Infection
Abstract
1. Introduction
2. Material and Methods
2.1. Design and Study Population
2.2. Survey Data
2.3. Cognitive Assessments
2.4. Data Analysis
3. Results
3.1. Demographic Characteristics
3.2. Cognitive Impairments
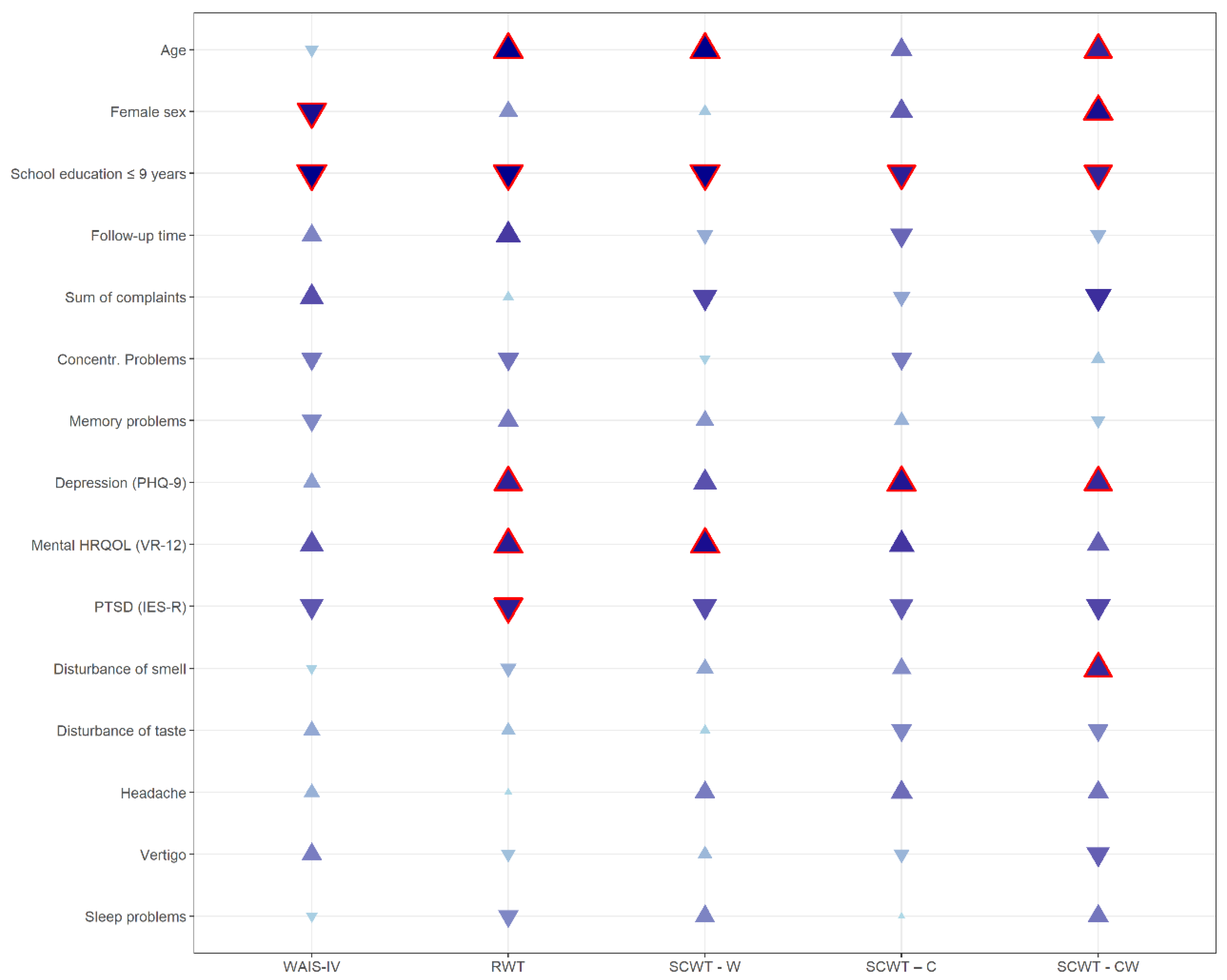
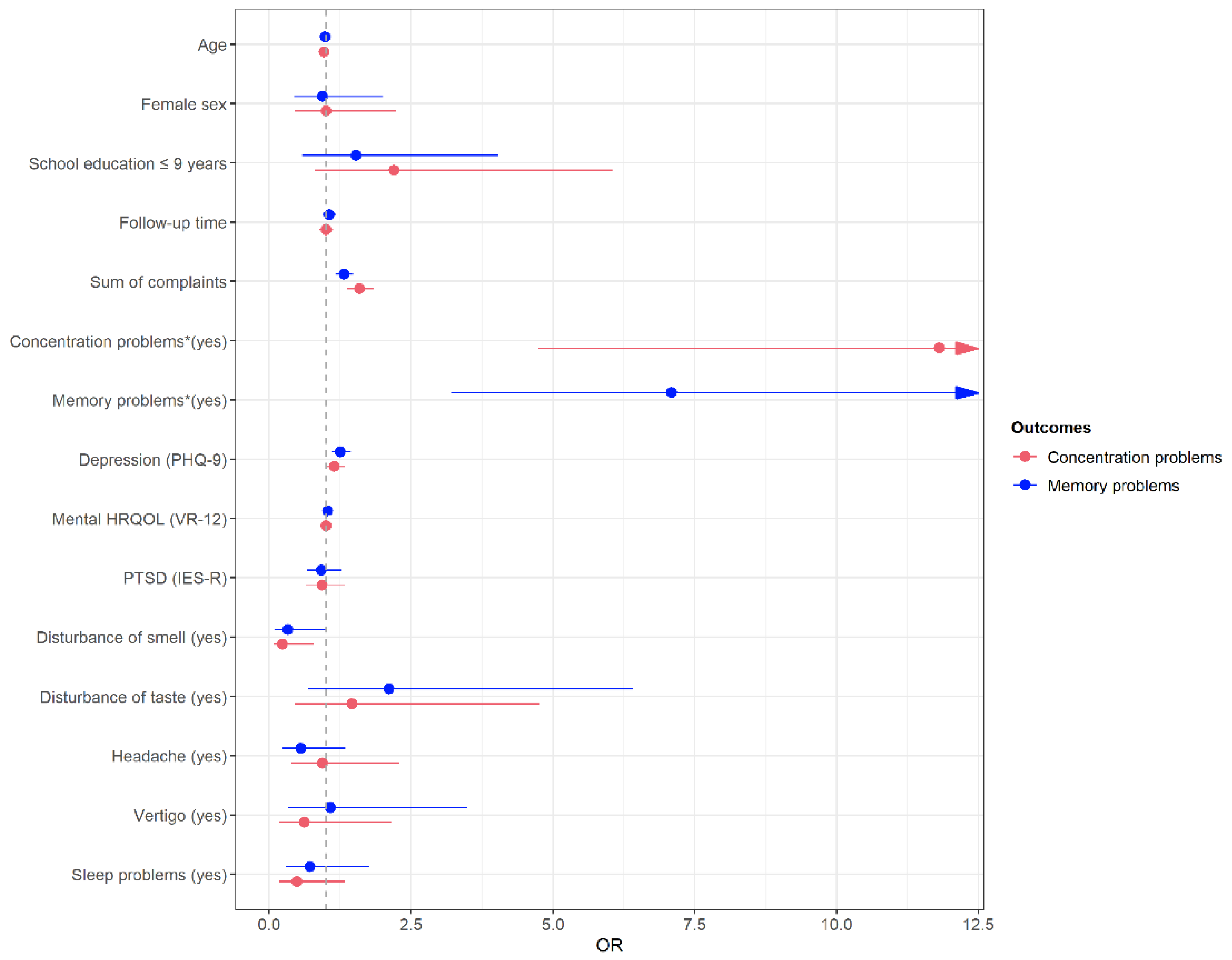
3.3. Variables Associated with Cognitive Impairments
3.4. Sensitivity Analyses
4. Discussion
5. Strengths and Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- COVID-19 Map—Johns Hopkins Coronavirus Resource Center [updated 24 May 2022]. Available online: https://coronavirus.jhu.edu/map.html (accessed on 24 May 2022).
- Nasserie, T.; Hittle, M.; Goodman, S.N. Assessment of the Frequency and Variety of Persistent Symptoms Among Patients with COVID-19: A Systematic Review. JAMA Netw. Open 2021, 4, e2111417. [Google Scholar] [CrossRef] [PubMed]
- Di Pietro, D.A.; Comini, L.; Gazzi, L.; Luisa, A.; Vitacca, M. Neuropsychological Pattern in a Series of Post-Acute COVID-19 Patients in a Rehabilitation Unit: Retrospective Analysis and Correlation with Functional Outcomes. Int. J. Environ. Res. Public Health 2021, 18, 5917. [Google Scholar] [CrossRef] [PubMed]
- Dix, E.; Roy, K. COVID-19: Brain Effects. Psychiatr. Clin. N. Am. 2022, 45, 625–637. [Google Scholar] [CrossRef] [PubMed]
- Sodagar, A.; Javed, R.; Tahir, H.; Razak, S.I.A.; Shakir, M.; Naeem, M.; Yusof, A.H.A.; Sagadevan, S.; Hazafa, A.; Uddin, J.; et al. Pathological Features and Neuroinflammatory Mechanisms of SARS-CoV-2 in the Brain and Potential Therapeutic Approaches. Biomolecules 2022, 12, 971. [Google Scholar] [CrossRef]
- Mao, L.; Jin, H.; Wang, M.; Hu, Y.; Chen, S.; He, Q.; Chang, J.; Hong, C.; Zhou, Y.; Wang, D.; et al. Neurologic Manifestations of Hospitalized Patients With Coronavirus Disease 2019 in Wuhan, China. JAMA Neurol. 2020, 77, 683–690. [Google Scholar] [CrossRef] [PubMed]
- Tavares-Júnior, J.W.; de Souza, A.C.C.; Borges, J.W.P.; Oliveira, D.N.; Siqueira-Neto, J.I.; Sobreira-Neto, M.A.; Braga-Neto, P. COVID-19 associated cognitive impairment: A systematic review. Cortex 2022, 152, 77–97. [Google Scholar] [CrossRef]
- Crivelli, L.; Palmer, K.; Calandri, I.; Guekht, A.; Beghi, E.; Carroll, W.; Frontera, J.; García-Azorín, D.; Westenberg, E.; Winkler, A.S.; et al. Changes in cognitive functioning after COVID-19: A systematic review and meta-analysis. Alzheimers Dement. 2022, 18, 1047–1066. [Google Scholar] [CrossRef]
- Ceban, F.; Ling, S.; Lui, L.M.W.; Lee, Y.; Gill, H.; Teopiz, K.M.; Rodrigues, N.B.; Subramaniapillai, M.; Di Vincenzo, J.D.; Cao, B.; et al. Fatigue and cognitive impairment in Post-COVID-19 Syndrome: A systematic review and meta-analysis. Brain Behav. Immun. 2022, 101, 93–135. [Google Scholar] [CrossRef]
- Rabinovitz, B.; Jaywant, A.; Fridman, C.B. Neuropsychological functioning in severe acute respiratory disorders caused by the coronavirus: Implications for the current COVID-19 pandemic. Clin. Neuropsychol. 2020, 34, 1453–1479. [Google Scholar] [CrossRef]
- Rogers, J.P.; Chesney, E.; Oliver, D.; Pollak, T.A.; McGuire, P.; Fusar-Poli, P.; Zandi, M.S.; Lewis, G.; David, A.S. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: A systematic review and meta-analysis with comparison to the COVID-19 pandemic. Lancet Psychiatry 2020, 7, 611–627. [Google Scholar] [CrossRef]
- van Kessel, S.A.M.; Olde Hartman, T.C.; Lucassen, P.L.B.J.; van Jaarsveld, C.H.M. Post-acute and long-COVID-19 symptoms in patients with mild diseases: A systematic review. Fam. Pract. 2021, 39, 159–167. [Google Scholar] [CrossRef]
- Becker, J.H.; Lin, J.J.; Doernberg, M.; Stone, K.; Navis, A.; Festa, J.R.; Wisnivesky, J.P. Assessment of Cognitive Function in Patients After COVID-19 Infection. JAMA Netw. Open 2021, 4, e2130645. [Google Scholar] [CrossRef]
- García-Sánchez, C.; Calabria, M.; Grunden, N.; Pons, C.; Arroyo, J.A.; Gómez-Anson, B.; Lleó, A.; Alcolea, D.; Belvís, R.; Morollón, N.; et al. Neuropsychological deficits in patients with cognitive complaints after COVID-19. Brain Behav. 2022, 12, e2508. [Google Scholar] [CrossRef]
- Garg, S.; Kim, L.; Whitaker, M.; O’Halloran, A.; Cummings, C.; Holstein, R.; Prill, M.; Chai, S.J.; Kirley, P.D.; Alden, N.B.; et al. Hospitalization Rates and Characteristics of Patients Hospitalized with Laboratory-Confirmed Coronavirus Disease 2019—COVID-NET, 14 States, March 1–30, 2020. Morb. Mortal. Wkly. Rep. 2020, 69, 458–464. [Google Scholar] [CrossRef]
- Coronavirus SARS-CoV-2—COVID-19-Fälle Nach Meldewoche und Geschlecht Sowie Anteile Mit fÜr COVID-19 Relevanten Symptomen, Anteile Hospitalisierter/Verstorbener und Altersmittelwert/-median (Tabelle Wird Jeden Donnerstag Aktualisiert). Available online: https://www.rki.de/DE/Content/InfAZ/N/Neuartiges_Coronavirus/Daten/Klinische_Aspekte.html. (accessed on 24 May 2022).
- Vanderlind, W.M.; Rabinovitz, B.B.; Miao, I.Y.; Oberlin, L.E.; Bueno-Castellano, C.; Fridman, C.; Jaywant, A.; Kanellopoulos, D. A systematic review of neuropsychological and psychiatric sequalae of COVID-19: Implications for treatment. Curr. Opin. Psychiatry 2021, 34, 420–433. [Google Scholar] [CrossRef]
- Baker, H.A.; Safavynia, S.A.; Evered, L.A. The ‘third wave’: Impending cognitive and functional decline in COVID-19 survivors. Br. J. Anaesth. 2021, 126, 44–47. [Google Scholar] [CrossRef] [PubMed]
- Hampshire, A.; Trender, W.; Chamberlain, S.R.; Jolly, A.E.; Grant, J.E.; Patrick, F.; Mazibuko, N.; Williams, S.C.; Barnby, J.M.; Hellyer, P.; et al. Cognitive deficits in people who have recovered from COVID-19. EClinicalMedicine 2021, 39, 101044. [Google Scholar] [CrossRef] [PubMed]
- Crivelli, L.; Calandri, I.; Corvalán, N.; Carello, M.A.; Keller, G.; Martínez, C.; Arruabarrena, M.; Allegri, R. Cognitive consequences of COVID-19: Results of a cohort study from South America. Arq Neuropsiquiatr. 2022, 80, 240–247. [Google Scholar] [CrossRef]
- Miskowiak, K.W.; Johnsen, S.; Sattler, S.M.; Nielsen, S.; Kunalan, K.; Rungby, J.; Lapperre, T.; Porsberg, C.M. Cognitive impairments four months after COVID-19 hospital discharge: Pattern, severity and association with illness variables. Eur. Neuropsychopharmacol. 2021, 46, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Damiano, R.F.; Guedes, B.F.; de Rocca, C.C.; de Pádua Serafim, A.; Castro, L.H.M.; Munhoz, C.D.; Nitrini, R.; Filho, G.B.; Miguel, E.C.; Lucchetti, G.; et al. Cognitive decline following acute viral infections: Literature review and projections for post-COVID-19. Eur. Arch. Psychiatry Clin. Neurosci. 2022, 272, 139–154. [Google Scholar] [CrossRef] [PubMed]
- Kroenke, K.; Spitzer, R.L.; Williams, J.B.W.; Löwe, B. The Patient Health Questionnaire Somatic, Anxiety, and Depressive Symptom Scales: A systematic review. Gen. Hosp. Psychiatry 2010, 32, 345–359. [Google Scholar] [CrossRef]
- Rosner, R.; Hagl, M. Die revidierte Impact of Event-Skala (IES-R). Psychosom. Konsiliarpsychiatr. 2008, 2, 240–243. [Google Scholar] [CrossRef]
- Hüppe, M.; Schneider, K.; Casser, H.R.; Knille, A.; Kohlmann, T.; Lindena, G.; Nagel, B.; Nelles, J.; Pfingsten, M.; Petzke, F. Kennwerte und teststatistische Güte des Veterans RAND 12-Item Health Survey (VR-12) bei Patienten mit chronischem Schmerz: Eine Auswertung auf Basis des KEDOQ-Schmerz-Datensatzes. Der Schmerz 2021, 6, 109–120. [Google Scholar] [CrossRef]
- Petermann, F. Wechsler Adult Intelligence Scale, 4th ed.; Pearson: London, UK, 2021. [Google Scholar]
- Bäumler, G. Farb-Wort-Interferenztest (FWIT) nach J. R. Stroop; Hogrefe: Göttingen, Germany, 1985. [Google Scholar]
- Aschenbrenner, S.; Tucha, O.; Lange, K.W. RWT. Regensburger Wortflüssigkeits-Test; Hogrefe: Göttingen, Germany, 2001. [Google Scholar]
- Scarpina, F.; Tagini, S. The Stroop Color and Word Test. Front. Psychol. 2017, 8, 557. [Google Scholar] [CrossRef]
- Garrigues, E.; Janvier, P.; Kherabi, Y.; Le Bot, A.; Hamon, A.; Gouze, H.; Doucet, L.; Berkani, S.; Oliosi, E.; Mallart, E.; et al. Post-discharge persistent symptoms and health-related quality of life after hospitalization for COVID-19. J. Infect. 2020, 81, e4–e6. [Google Scholar] [CrossRef]
- Mannan, A.; Mehedi, H.M.H.; Chy, N.U.H.A.; Qayum, M.O.; Akter, F.; Rob, M.A.; Biswas, P.; Hossain, S.; Ayub, M. A multi-centre, cross-sectional study on coronavirus disease 2019 in Bangladesh: Clinical epidemiology and short-term outcomes in recovered individuals. New Microbes New Infect. 2021, 40, 100838. [Google Scholar] [CrossRef]
- Mattioli, F.; Stampatori, C.; Righetti, F.; Sala, E.; Tomasi, C.; de Palma, G. Neurological and cognitive sequelae of Covid-19: A four month follow-up. J. Neurol. 2021, 268, 4422–4428. [Google Scholar] [CrossRef]
- Nogueira, J.; Gerardo, B.; Silva, A.R.; Pinto, P.; Barbosa, R.; Soares, S.; Baptista, B.; Paquete, C.; Cabral-Pinto, M.; Vilar, M.M.; et al. Effects of restraining measures due to COVID-19: Pre- and post-lockdown cognitive status and mental health. Curr. Psychol. 2021, 41, 7383–7392. [Google Scholar] [CrossRef]
- Del Brutto, O.H.; Rumbea, D.A.; Recalde, B.Y.; Mera, R.M. Cognitive sequelae of long COVID may not be permanent: A prospective study. Eur. J. Neurol. 2022, 29, 1218–1221. [Google Scholar] [CrossRef]
- Albu, S.; Zozaya, N.R.; Murillo, N.; García-Molina, A.; Chacón, C.A.F.; Kumru, H. What’s going on following acute covid-19? Clinical characteristics of patients in an out-patient rehabilitation program. NeuroRehabilitation 2021, 48, 469–480. [Google Scholar] [CrossRef]
- Jaywant, A.; Vanderlind, W.M.; Alexopoulos, G.S.; Fridman, C.B.; Perlis, R.H.; Gunning, F. Frequency and profile of objective cognitive deficits in hospitalized patients recovering from COVID-19. Neuropsychopharmacology 2021, 46, 2235–2240. [Google Scholar] [CrossRef]
- Mazza, M.G.; Palladini, M.; De Lorenzo, R.; Magnaghi, C.; Poletti, S.; Furlan, R.; Ciceri, F.; COVID-19 BioB Outpatient Clinic Study group; Rovere-Querini, P.; Benedetti, F. Persistent psychopathology and neurocognitive impairment in COVID-19 survivors: Effect of inflammatory biomarkers at three-month follow-up. Brain Behav. Immun. 2021, 94, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Rass, V.; Beer, R.; Schiefecker, A.J.; Kofler, M.; Lindner, A.; Mahlknecht, P.; Heim, B.; Limmert, V.; Sahanic, S.; Pizzini, A.; et al. Neurological outcome and quality of life 3 months after COVID-19: A prospective observational cohort study. Eur. J. Neurol. 2021, 28, 3348–3359. [Google Scholar] [CrossRef] [PubMed]
- Negrini, F.; Ferrario, I.; Mazziotti, D.; Berchicci, M.; Bonazzi, M.; de Sire, A.; Negrini, S.; Zapparoli, L. Neuropsychological Features of Severe Hospitalized Coronavirus Disease 2019 Patients at Clinical Stability and Clues for Postacute Rehabilitation. Arch. Phys. Med. Rehabil. 2021, 102, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Almeria, M.; Cejudo, J.C.; Sotoca, J.; Deus, J.; Krupinski, J. Cognitive profile following COVID-19 infection: Clinical predictors leading to neuropsychological impairment. Brain Behav. Immun. Health 2020, 9, 100163. [Google Scholar] [CrossRef] [PubMed]
- Amalakanti, S.; Arepalli, K.V.R.; Jillella, J.P. Cognitive assessment in asymptomatic COVID-19 subjects. Virusdisease 2021, 32, 146–149. [Google Scholar] [CrossRef]
- Bungenberg, J.; Humkamp, K.; Hohenfeld, C.; Rust, M.I.; Ermis, U.; Dreher, M.; Hartmann, N.K.; Marx, G.; Binkofski, F.; Finke, C.; et al. Long COVID-19: Objectifying most self-reported neurological symptoms. Ann. Clin. Transl. Neurol. 2022, 9, 141–154. [Google Scholar] [CrossRef]
| Demographic Characteristics | |
|---|---|
| Age (years) (mean (SD), MIN–MAX) | 46.8 (15.2), 18–87 |
| Sex | |
| Female | 202 (54.3%) |
| Male | 170 (45.7%) |
| School education | |
| ≤ 9 years | 63 (17.0%) |
| > 9 years | 308 (83.0%) |
| Marital status | |
| Married | 246 (66.5%) |
| Single | 98 (26.5%) |
| Divorced | 20 (5.4%) |
| Widowed | 6 (1.6%) |
| Smoking | |
| Never-smoker | 203 (54.6%) |
| Current smoker | 29 (7.8%) |
| Former smoker | 140 (37.6%) |
| Clinical characteristics | |
| Comorbidities (yes) | |
| Hypertension | 71 (19.1%) |
| Coronary heart disease or angina pectoris | 17 (4.6%) |
| Heart attack | 9 (2.4%) |
| Diabetes | 14 (3.8%) |
| Cancer | 17 (4.6%) |
| Depression | 32 (8.6%) |
| Anxiety Disease | 22 (6.0%) |
| Chronic Bronchitis | 21 (5.7%) |
| Autoimmune Disease | 30 (8.1%) |
| Sum comorbidities | |
| None (0) | 213 (57.0%) |
| At least one (≥1) | 156 (43.0%) |
| FU time (months) (median (Q1-Q3)) | 9.1 (6.0–11.3) |
| ≥ 3–≤ 6 | 95 (25.5%) |
| > 6–≤ 9 | 88 (23.7%) |
| > 9–≤ 12 | 111 (32.8%) |
| > 12–≤ 15 | 67 (18.1%) |
| Neurological symptoms | |
| Headache | |
| Acute COVID-19 phase | 244 (65.8%) |
| FU examination | 80 (21.6%) |
| Disturbance of sense of taste | |
| Acute COVID-19 phase | 233 (62.8%) |
| FU examination | 60 (16.3%) |
| Disturbance of smell | |
| Acute COVID-19 phase | 228 (61.5%) |
| FU examination | 75 (20.3%) |
| Vertigo | |
| Acute COVID-19 phase | 130 (35.1%) |
| FU examination | 34 (9.2%) |
| Sleeping problems | |
| Acute COVID-19 phase | 108 (29.2%) |
| FU examination | 87 (23.5%) |
| Coordination of movements | |
| Acute COVID-19 phase | 53 (14.3%) |
| FU examination | 16 (4.4%) |
| Tiredness or exhaustion | |
| Acute COVID-19 phase | 310 (83.8%) |
| FU examination | 126 (34.0%) |
| Depressive mood | |
| Acute COVID-19 phase | 97 (26.2%) |
| FU examination | 61 (16.5%) |
| Anxiety or panic | |
| Acute COVID-19 phase | 80 (21.6%) |
| FU examination | 28 (7.6%) |
| Sum of complaints | |
| Acute COVID-19 phase (median, (Q1–Q3)) | 13 (9–19) |
| FU examination (median, (Q1–Q3)) | 3 (1–7) |
| Depressiveness (PHQ-9) (median (Q1–Q3)) | 4 (2–7) |
| <5: | 221 (59.4%) |
| ≥ 5–<10: mild depressiveness | 103 (27.7%) |
| ≥ 10–<15: moderate depressiveness | 38 (10.2%) |
| ≥ 15–<20: moderately severe depressiveness | 8 (2.2%) |
| ≥ 20: severe depression | 2 (0.5%) |
| Post-traumatic stress disorder (IES-R total score)- | |
| 0: no suspected diagnosis | 353 (95.9%) |
| >0: suspected diagnosis | 15 (4.1%) |
| Physical HRQOL (VR-12) (median (Q1–Q3)) | 52.7 (45.6–55.8) |
| Mental HRQOL (VR-12) (median (Q1–Q3)) | 50.9 (43.8–56.3) |
| Subjective Ratings | |
|---|---|
| Difficulties Concentrating | |
| Acute COVID-19 phase (yes) | 164 (44.2%) |
| FU examination (yes) | 92 (24.9%) |
| Severity at FU (n = 91) | |
| No impairment | 3 (3.3%) |
| Slightly impaired | 38 (41.8%) |
| Moderately impaired | 20 (22.0%) |
| Fairly impaired | 17 (18.7%) |
| Severely impaired | 13 (14.3%) |
| Frequency/severity at FU compared with acute COVID-19 phase (n = 78) | |
| Much less often/weaker (−3) | 9 (11.5%) |
| −2 | 11 (14.1%) |
| −1 | 10 (12.8%) |
| Equal frequency/severity (0) | 20 (25.6%) |
| +1 | 6 (7.7%) |
| +2 | 12 (15.4%) |
| Much more often/stronger (+3) | 10 (12.8%) |
| Memory problems | |
| Acute COVID-19 phase (yes) | 90 (24.3%) |
| FU examination (yes) | 81 (21.9%) |
| Severity at FU (n = 81) | |
| No impairment | 6 (7.4%) |
| Slightly impaired | 31 (38.3%) |
| Moderately impaired | 21 (25.9%) |
| Fairly impaired | 11 (13.6%) |
| Severely impaired | 12 (14.8%) |
| Frequency/severity at FU compared to acute COVID-19 phase (n = 51) | |
| Much less often/weaker (−3) | 7 (13.7%) |
| −2 | 4 (7.8%) |
| −1 | 10 (19.6%) |
| Equal frequency/severity (0) | 10 (19.6%) |
| +1 | 6 (11.8%) |
| +2 | 9 (17.7%) |
| Much more often/stronger (+3) | 5 (9.8%) |
| Cognitive tests | |
| WAIS-IV: median, (Q1–Q3) | 8 (6–9) |
| ≥8: normal | 196 (52.7%) |
| >4–≤7: mild/borderline negative alteration | 153 (41.1%) |
| ≤4: impairment | 23 (6.2%) |
| RWT: median, (Q1–Q3) | 63 (28–87) |
| ≥17: normal | 302 (81.2%) |
| >2–≤16: mild/borderline negative alteration | 47 (12.6%) |
| ≤2: impairment | 23 (6.2%) |
| SCWT–W: median, (Q1–Q3) | 54 (50–60) |
| ≥41: normal | 358 (97.0%) |
| >30–≤ 40: mild/borderline negative alteration | 10 (2.7%) |
| ≤30: impairment | 1 (0.3%) |
| SCWT–C: median (Q1–Q3) | 55 (50–62) |
| ≥41: normal | 361 (97.1%) |
| >30–≤ 40: mild/borderline negative alteration | 6 (1.6%) |
| ≤30: impairment | 1 (0.3%) |
| SCWT–CW: median (Q1–Q3) | 56 (53–62) |
| ≥41: normal | 363 (98.6%) |
| >30–≤ 40: mild/borderline negative alteration | 4 (1.1%) |
| ≤30: impairment | 1 (0.3%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kirchberger, I.; Peilstöcker, D.; Warm, T.D.; Linseisen, J.; Hyhlik-Dürr, A.; Meisinger, C.; Goßlau, Y. Subjective and Objective Cognitive Impairments in Non-Hospitalized Persons 9 Months after SARS-CoV-2 Infection. Viruses 2023, 15, 256. https://doi.org/10.3390/v15010256
Kirchberger I, Peilstöcker D, Warm TD, Linseisen J, Hyhlik-Dürr A, Meisinger C, Goßlau Y. Subjective and Objective Cognitive Impairments in Non-Hospitalized Persons 9 Months after SARS-CoV-2 Infection. Viruses. 2023; 15(1):256. https://doi.org/10.3390/v15010256
Chicago/Turabian StyleKirchberger, Inge, Daniela Peilstöcker, Tobias D. Warm, Jakob Linseisen, Alexander Hyhlik-Dürr, Christine Meisinger, and Yvonne Goßlau. 2023. "Subjective and Objective Cognitive Impairments in Non-Hospitalized Persons 9 Months after SARS-CoV-2 Infection" Viruses 15, no. 1: 256. https://doi.org/10.3390/v15010256
APA StyleKirchberger, I., Peilstöcker, D., Warm, T. D., Linseisen, J., Hyhlik-Dürr, A., Meisinger, C., & Goßlau, Y. (2023). Subjective and Objective Cognitive Impairments in Non-Hospitalized Persons 9 Months after SARS-CoV-2 Infection. Viruses, 15(1), 256. https://doi.org/10.3390/v15010256







