Metagenomic Analysis of Ampelographic Collections of Dagestan Revealed the Presence of Two Novel Grapevine Viruses
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Preparation of mRNA Libraries
2.2. Analysis of HTS Data and Assembly of Viral Genomes
2.3. Validation of Grapevine Viruses and Viroids
2.4. Assembly of New Virus Sequences and Analysis of their Genomes
2.5. Phylogenetic Analysis and Genetic Diversity
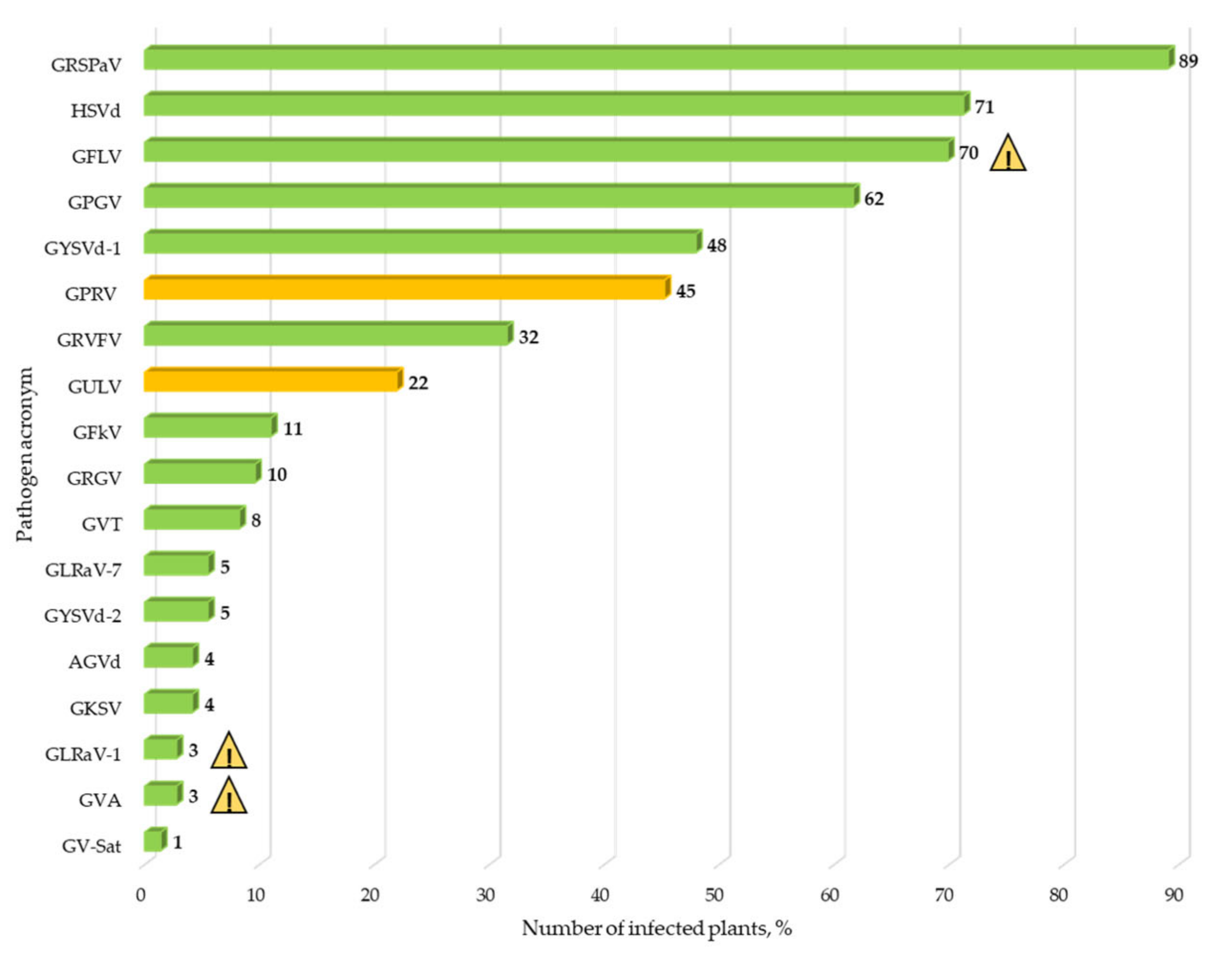
3. Results
3.1. mRNA-Seq Data Analysis
3.2. Identification of Known Grapevine Viruses and Viroids in the Germplasm Virome
3.2.1. Family: Secoviridae
Grapevine Fanleaf Virus
3.2.2. Family: Betaflexiviridae
Grapevine Rupestris Stem Pitting-Associated Virus
Grapevine Pinot Gris Virus
Grapevine Virus A
Grapevine Virus T
Grapevine Kizil Sapak Virus
3.2.3. Family: Tymoviridae
Grapevine Fleck Virus
Grapevine Rupestris Vein Feathering Virus
Grapevine Red Globe Virus
3.2.4. Family: Closteroviridae
Grapevine Leafroll-Associated Virus 1
Grapevine Leafroll-Associated Virus 7
3.2.5. Family: Unassigned
Grapevine Satellite Virus
3.2.6. Family: Pospiviroidae
Hop Stunt Viroid
Grapevine Yellow Speckle Viroid 1
Grapevine Yellow Speckle Viroid 2
Australian Grapevine Viroid
3.3. Discovery of Novel Grapevine Viruses
3.3.1. Family: Tombusviridae
Grapevine Umbra-Like Virus
3.3.2. Family: Caulimoviridae
Grapevine Pararetrovirus
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ardid-Ruiz, A.; Harazin, A.; Barna, L.; Walter, F.R.; Bladé, C.; Suárez, M.; Deli, M.A.; Aragonès, G. The effects of Vitis vinifera L. phenolic compounds on a blood-brain barrier culture model: Expression of leptin receptors and protection against cytokine-induced damage. J. Ethnopharmacol. 2020, 247, 112253. [Google Scholar] [CrossRef] [PubMed]
- Terral, J.F.; Tabard, E.; Bouby, L.; Ivorra, S.; Pastor, T.; Figueiral, I.; Picq, S.; Chevance, J.B.; Jung, C.; Fabre, L.; et al. Evolution and history of grapevine (Vitis vinifera) under domestication: New morphometric perspectives to understand seed domestication syndrome and reveal origins of ancient European cultivars. Ann. Bot. 2010, 105, 443–455. [Google Scholar] [CrossRef]
- Reynolds, A.G. The Grapevine, Viticulture, and Winemaking: A Brief Introduction. In Grapevine Viruses: Molecular Biology, Diagnostics and Management; Meng, B., Martelli, G., Golino, D., Fuchs, M., Eds.; Springer: Cham, Switzerland, 2017; pp. 3–29. [Google Scholar]
- Rad, S.Z.K.; Javadi, B.; Hayes, A.W.; Karimi, G. Potential angiotensin converting enzyme (ACE) inhibitors from Iranian traditional plants described by Avicenna’s Canon of Medicine. Avicenna J. Phytomed. 2019, 9, 291–309. [Google Scholar]
- Kandylis, P. Grapes and Their Derivatives in Functional Foods. Foods 2021, 10, 672. [Google Scholar] [CrossRef] [PubMed]
- Martelli, G.P. An overview on grapevine viruses, viroids, and the diseases they cause. In Grapevine Viruses: Molecular Biology, Diagnostics and Management; Meng, B., Martelli, G., Golino, D., Fuchs, M., Eds.; Springer: Cham, Switzerland, 2017; pp. 31–46. ISBN 9783319577067. [Google Scholar]
- Fuchs, M. Grapevine viruses: A multitude of diverse species with simple but overall poorly adopted management solutions in the vineyard. J. Plant Pathol. 2020, 102, 643–653. [Google Scholar] [CrossRef]
- Chiaki, Y.; Ito, T. Complete genome sequence of a novel putative polerovirus detected in grapevine. Arch. Virol. 2020, 165, 1007–1010. [Google Scholar] [CrossRef]
- Reynard, J.S.; Brodard, J.; Remoliff, E.; Lefebvre, M.; Schumpp, O.; Candresse, T. A novel foveavirus identified in wild grapevine (Vitis vinifera subsp. sylvestris). Arch. Virol. 2020, 165, 2999–3002. [Google Scholar] [CrossRef]
- Nabeshima, T.; Abe, J. High-Throughput Sequencing Indicates Novel Varicosavirus, Emaravirus, and Deltapartitivirus Infections in Vitis coignetiae. Viruses 2021, 13, 827. [Google Scholar] [CrossRef]
- Al Rwahnih, M.; Alabi, O.J.; Hwang, M.S.; Tian, T.; Mollov, D.; Golino, D. Characterization of a new nepovirus infecting grapevine. Plant Dis. 2021, 105, 1432–1439. [Google Scholar] [CrossRef]
- Current ICTV Taxonomy Release ICTV. Available online: https://ictv.global/taxonomy (accessed on 8 November 2022).
- Martelli, G.P. Directory of virus and virus-like diseases of the grapevine and their agents. J. Plant Pathol. 2014, 96, 1–136. [Google Scholar] [CrossRef]
- Al Rwahnih, M.; Alabi, O.J.; Hwang, M.S.; Stevens, K.; Golino, D. Identification and genomic characterization of grapevine Kizil Sapak virus, a novel grapevine-infecting member of the family Betaflexiviridae. Arch. Virol. 2019, 164, 3145–3149. [Google Scholar] [CrossRef]
- Bragard, C.; Dehnen-Schmutz, K.; Gonthier, P.; Jacques, M.A.; Jaques Miret, J.A.; Justesen, A.F.; MacLeod, A.; Magnusson, C.S.; Milonas, P.; Navas-Cortes, J.A.; et al. Pest categorisation of non-EU viruses and viroids of Vitis L. EFSA J. 2019, 17, 5669. [Google Scholar] [CrossRef]
- Zhang, Y.; Singh, K.; Kaur, R.; Qiu, W. Association of a Novel DNA Virus with the Grapevine Vein-Clearing and Vine Decline Syndrome. Virology 2011, 101, 1081–1090. [Google Scholar] [CrossRef] [PubMed]
- Beach, S.; Kovens, M.; Hubbert, L.A.; Honesty, S.; Guo, Q.; Pap, D.; Dai, R.; Kovacs, L.; Qiu, W. Genetic and phenotypic characterization of Grapevine vein clearing virus from wild Vitis rupestris. Phytopathology 2017, 107, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Maliogka, V.I.; Olmos, A.; Pappi, P.G.; Lotos, L.; Efthimiou, K.; Grammatikaki, G.; Candresse, T.; Katis, N.I.; Avgelis, A.D. A novel grapevine badnavirus is associated with the Roditis leaf discoloration disease. Virus Res. 2015, 203, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Sudarshana, M.R.; Perry, K.L.; Fuchs, M.F. Grapevine red blotch-associated virus, an emerging threat to the grapevine industry. Phytopathology 2015, 105, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Yepes, L.M.; Cieniewicz, E.; Krenz, B.; McLane, H.; Thompson, J.R.; Perry, K.L.; Fuchs, M. Causative role of grapevine red blotch virus in red blotch disease. Phytopathology 2018, 108, 902–909. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, J.; Vélez, D. A microsatellite-based system for the identification and legal protection of grapevine varieties Cost action FA1003: East-West Cillaboration for Grapevine Diversity Exploration and Mobilization of Adaptive Traits for Breeding View project. Bull. l’OIV 2007, 80, 177–185. [Google Scholar]
- Pertot, I.; Caffi, T.; Rossi, V.; Mugnai, L.; Hoffmann, C.; Grando, M.S.; Gary, C.; Lafond, D.; Duso, C.; Thiery, D.; et al. A critical review of plant protection tools for reducing pesticide use on grapevine and new perspectives for the implementation of IPM in viticulture. Crop Prot. 2017, 97, 70–84. [Google Scholar] [CrossRef]
- Alleweldt, G.; Possingham, J.V. Progress in grapevine breeding. Theor. Appl. Genet. 1988, 75, 669–673. [Google Scholar] [CrossRef]
- Akkurt, M.; Tahmaz, H.; Veziroğlu, S. Recent Developments in Seedless Grapevine Breeding. S. Afr. J. Enol. Vitic. 2019, 40, 1. [Google Scholar] [CrossRef]
- Eibach, R.; Töpfer, R. Traditional grapevine breeding techniques. In Grapevine Breeding Programs for the Wine Industry; Andrew Reynolds, Ed.; Woodhead Publishing: Sawston, UK, 2015; pp. 3–22. ISBN 9781782420804. [Google Scholar]
- Gardiman, M.; Bavaresco, L. The Vitis germplasm repository at the CRA-VIT, Conegliano (Italy): Conservation, characterization and valorisation of grapevine genetic resources. Acta Hortic. 2015, 1082, 239–244. [Google Scholar] [CrossRef]
- De Oliveira, G.L.; de Souza, A.P.; de Oliveira, F.A.; Zucchi, M.I.; de Souza, L.M.; Moura, M.F. Genetic structure and molecular diversity of Brazilian grapevine germplasm: Management and use in breeding programs. PLoS ONE 2020, 15, e0240665. [Google Scholar] [CrossRef] [PubMed]
- Sargolzaei, M.; Rustioni, L.; Cola, G.; Ricciardi, V.; Bianco, P.A.; Maghradze, D.; Failla, O.; Quaglino, F.; Toffolatti, S.L.; De Lorenzis, G. Georgian Grapevine Cultivars: Ancient Biodiversity for Future Viticulture. Front. Plant Sci. 2021, 12, 94. [Google Scholar] [CrossRef]
- El Oualkadi, A.; Ater, M.; Messaoudi, Z.; Laucou, V.; Boursiquot, J.M.; Lacombe, T.; This, P. Molecular characterization of Moroccan grapevine germplasm using SSR markers for the establishment of a reference collection. OENO One 2009, 43, 135–148. [Google Scholar] [CrossRef]
- Shvets, D.; Porotikova, E.; Sandomirsky, K.; Vinogradova, S. Virome of Grapevine Germplasm from the Anapa Ampelographic Collection (Russia). Viruses 2022, 14, 1314. [Google Scholar] [CrossRef] [PubMed]
- Portal of Bioresource Collections: Plant Collections. Available online: http://www.biores.cytogen.ru/portal_plants_collections (accessed on 15 September 2022).
- Ilnitskaya, E.T.; Suprun, I.I.; Naumova, L.G.; Tokmakov, S.V.; Ganich, V.A. Characterisation of native Dagestan grape cultivars using SSR-analysis and the main ampelographic features of the leaves. Vavilov J. Genet. Breed. 2017, 21, 617–622. [Google Scholar] [CrossRef][Green Version]
- Khalilova, E.A.; Kotenko, S.T.; Islammagomedova, E.A.; Abakarova, A.A.; Aliverdieva, D.A. Influence of Soil and Climate on the Biological Potential of “Kara-Koisu” Dry Red Wines from Cabernet-Sauvignon Grapes (Republic of Dagestan). Sci. Study Res. Chem. Chem. Eng. Biotechnol. Food Ind. 2022, 23, 141–154. [Google Scholar]
- Kislin, E.N.; Nosulchak, V.A.; Dzyubenko, N.I. Ampelographic collection of the Vavilov Institute: Past, present and future. Magarach. Vitic. Winemak. 2015, 3, 14–16. [Google Scholar]
- Agakhanov, M.M.; Volkov, V.A.; Ulianich, P.S.; Abdullaev, K.M.; Kislin, E.N. Polymorphism of microsattelite loci within the grape germplasm collection maintained at the Dagestan experiment station of VIR. Proc. Appl. Bot. Genet. Breed. 2018, 179, 224–234. [Google Scholar] [CrossRef]
- The Dagestan Experimental Selection Station of Viticulture and Olericulture. Available online: https://dagsosvio.ru/ (accessed on 15 September 2022).
- Feyzullaev, B.A.; Kazakhmedov, R.E. The main results of scientific research work of the Daghestan Breeding Experimental Stationof Viticulture and Horticulture in 2017. Sci. Work. North Cauc. Fed. Sci. Cent. Hortic. Vitic. Wine-Mak. 2018, 16, 34–38. [Google Scholar] [CrossRef]
- Kazakhmedov, R.E.; Agakhanov, A.K.; Abdullaeva, T.I. New promising hybrid forms of wine direction selected by Dagestan Breeding Experimental Station for Viticulture and Vegeculture. Magarach. Vitic. Winemak. 2020, 22, 100–104. [Google Scholar] [CrossRef]
- Morante-Carriel, J.; Sellés-Marchart, S.; Martínez-Márquez, A.; Martínez-Esteso, M.J.; Luque, I.; Bru-Martínez, R. RNA isolation from loquat and other recalcitrant woody plants with high quality and yield. Anal. Biochem. 2014, 452, 46–53. [Google Scholar] [CrossRef]
- FinchTV. Available online: https://digitalworldbiology.com/FinchTV (accessed on 15 September 2022).
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis Version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef] [PubMed]
- Shvets, D.; Vinogradova, S. Occurrence and Genetic Characterization of Grapevine Pinot Gris Virus in Russia. Plants 2022, 11, 1061. [Google Scholar] [CrossRef]
- Lefkowitz, E.J.; Dempsey, D.M.; Hendrickson, R.C.; Orton, R.J.; Siddell, S.G.; Smith, D.B. Virus taxonomy: The database of the International Committee on Taxonomy of Viruses (ICTV). Nucleic Acids Res. 2018, 46, D708–D717. [Google Scholar] [CrossRef] [PubMed]
- Schoch, C.L.; Ciufo, S.; Domrachev, M.; Hotton, C.L.; Kannan, S.; Khovanskaya, R.; Leipe, D.; McVeigh, R.; O’Neill, K.; Robbertse, B.; et al. NCBI Taxonomy: A comprehensive update on curation, resources and tools. Database 2020, 2020, baaa062. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef]
- Muhire, B.M.; Varsani, A.; Martin, D.P. SDT: A Virus Classification Tool Based on Pairwise Sequence Alignment and Identity Calculation. PLoS ONE 2014, 9, e108277. [Google Scholar] [CrossRef] [PubMed]
- Open Reading Frame Finder. Available online: https://www.ncbi.nlm.nih.gov/orffinder/ (accessed on 23 October 2022).
- InterPro—Classification of Protein Families. Available online: https://www.ebi.ac.uk/interpro/ (accessed on 23 October 2022).
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [CrossRef] [PubMed]
- Andret-Link, P.; Laporte, C.; Valat, L.; Ritzenthaler, C.; Demangeat, G.; Vigne, E.; Laval, V.; Pfeiffer, P.; Stussi-Garaud, C.; Fuchs, M. Grapevine fanleaf virus: Still a major threat to the grapevine industry. J. Plant Pathol. 2004, 86, 183–195. [Google Scholar]
- Gambino, G.; Gribaudo, I.; Leopold, S.; Schartl, A.; Laimer, M. Molecular characterization of grapevine plants transformed with GFLV resistance genes: I. Plant Cell Rep. 2005, 24, 655–662. [Google Scholar] [CrossRef]
- Volodin, V.; Gorislavets, S.; Risovannaya, V.; Stranishevskaya, E.; Shadura, N.; Volkov, Y.; Matveykina, E. Detection of a Viral Infection Complex (GLRAV-1, -3 and GFLV) in Vineyards of Crimea. Vitic. Winemak. 2020, 49, 124–126. [Google Scholar]
- Porotikova, E.; Terehova, U.; Volodin, V.; Yurchenko, E.; Vinogradova, S. Distribution and Genetic Diversity of Grapevine Viruses in Russia. Plants 2021, 10, 1080. [Google Scholar] [CrossRef]
- Navrotskaya, E.; Porotikova, E.; Yurchenko, E.; Galbacs, Z.N.; Varallyay, E.; Vinogradova, S. High-Throughput Sequencing of Small RNAs for Diagnostics of Grapevine Viruses and Viroids in Russia. Viruses 2021, 13, 2432. [Google Scholar] [CrossRef] [PubMed]
- Digiaro, M.; Elbeaino, T.; Martelli, G.P. Grapevine fanleaf virus and other old world nepoviruses. In Grapevine Viruses: Molecular Biology, Diagnostics and Management; Meng, B., Martelli, G., Golino, D., Fuchs, M., Eds.; Springer: Cham, Switzerland, 2017; pp. 47–82. ISBN 9783319577067. [Google Scholar]
- Wei, T.; Clover, G. Use of primers with 5′ non-complementary sequences in RT-PCR for the detection of nepovirus subgroups A and B. J. Virol. Methods 2008, 153, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Elbeaino, T.; Digiaro, M.; Ghebremeskel, S.; Martelli, G.P. Grapevine deformation virus: Completion of the sequence and evidence on its origin from recombination events between Grapevine fanleaf virus and Arabis mosaic virus. Virus Res. 2012, 166, 136–140. [Google Scholar] [CrossRef]
- Gottula, J.; Lapato, D.; Cantilina, K.; Saito, S.; Bartlett, B.; Fuchs, M. Genetic Variability, Evolution, and Biological Effects of Grapevine fanleaf virus Satellite RNAs. Phytopathology 2013, 103, 1180–1187. [Google Scholar] [CrossRef]
- Fuchs, M.; Schmitt-Keichinger, C.; Sanfaçon, H. A Renaissance in Nepovirus Research Provides New Insights into Their Molecular Interface with Hosts and Vectors. In Advances in Virus Research; Kielian, M., Mettenleiter, T., Roossinck, M., Eds.; Academic Press: London, UK, 2017; Volume 97, pp. 61–105. [Google Scholar]
- Dmitrenko, U.D.; Porotikova, E.V.; Gorislavets, S.M.; Risovannaya, V.I.; Volkov, Y.A.; Stranishevskaya, E.P.; Kamionskaya, A.M.; Vinogradova, S.V. First Report of Grapevine rupestris stem pitting-associated virus in Russia. Plant Dis. 2016, 100, 2542. [Google Scholar] [CrossRef]
- Hily, J.M.; Beuve, M.; Vigne, E.; Demangeat, G.; Candresse, T.; Lemaire, O. A genome-wide diversity study of grapevine rupestris stem pitting-associated virus. Arch. Virol. 2018, 163, 3105–3111. [Google Scholar] [CrossRef] [PubMed]
- Tarquini, G.; Ermacora, P.; Firrao, G. Polymorphisms at the 3′ end of the movement protein (MP) gene of grapevine Pinot gris virus (GPGV) affect virus titre and small interfering RNA accumulation in GLMD disease. Virus Res. 2021, 302, 198482. [Google Scholar] [CrossRef]
- Saldarelli, P.; Giampetruzzi, A.; Morelli, M.; Malossini, U.; Pirolo, C.; Bianchedi, P.; Gualandri, V. Genetic variability of Grapevine Pinot gris virus and its association with Grapevine leaf mottling and deformation. Phytopathology 2015, 105, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Morán, F.; Olmos, A.; Lotos, L.; Predajňa, L.; Katis, N.; Glasa, M.; Maliogka, V.; Ruiz-García, A.B. A novel specific duplex real-time RT-PCR method for absolute quantitation of Grapevine Pinot gris virus in plant material and single mites. PLoS ONE 2018, 13, e0197237. [Google Scholar] [CrossRef]
- Le Maguet, J.; Hommay, G.; Beuve, M.M.; Lemaire, O.O.; Herrbach, E.E.; Hommay, G.; Beuve, M.; Lemaire, O.; Herrbach, E. New Progress in the Study of Grapevine Leafroll Disease in France. In Proceedings of the 17th Congress of the International Council for the Study of Virus and Virus-like Diseases of the Grapevine (ICVG), Davis, CA, USA, 7–14 October 2012; pp. 46–47. [Google Scholar]
- Song, Y.; Hanner, R.H.; Meng, B. Probing into the Effects of Grapevine Leafroll-Associated Viruses on the Physiology, Fruit Quality and Gene Expression of Grapes. Viruses 2021, 13, 593. [Google Scholar] [CrossRef]
- Reynard, J.-S.; Schneeberger, P.H.H.; Frey, J.E.; Schaerer, S. Biological, Serological, and Molecular Characterization of a Highly Divergent Strain of Grapevine leafroll-associated virus 4 Causing Grapevine Leafroll Disease. Phytopathology 2015, 105, 1262–1269. [Google Scholar] [CrossRef]
- Porotikova, E.V.; Risovannaya, V.I.; Volkov, Y.A.; Dmitrenko, U.D.; Volodin, V.A.; Gorislavets, S.M.; Stranishevskaya, E.P.; Agranovsky, A.A.; Kamionskaya, A.M.; Vinogradova, S.V. Occurrence of grapevine leafroll-associated viruses-1 and -3 in Crimea. Moscow Univ. Biol. Sci. Bull. 2016, 71, 76–79. [Google Scholar] [CrossRef][Green Version]
- Porotikova, E.V.; Dmitrenko, U.D.; Yurchenko, E.G.; Vinogradova, S.V. First Report of Grapevine leafroll-associated virus 2 in Russian Grapevines (Vitis vinifera). Plant Dis. 2018, 103, 164. [Google Scholar] [CrossRef]
- Al Rwahnih, M.; Daubert, S.; Sudarshana, M.R.; Rowhani, A. Gene from a novel plant virus satellite from grapevine identifies a viral satellite lineage. Virus Genes 2013, 47, 114–118. [Google Scholar] [CrossRef]
- Czotter, N.; Molnar, J.; Szabó, E.; Demian, E.; Kontra, L.; Baksa, I.; Szittya, G.; Kocsis, L.; Deak, T.; Bisztray, G.; et al. NGS of virus-derived small RNAs as a diagnostic method used to determine viromes of Hungarian Vineyards. Front. Microbiol. 2018, 9, 122. [Google Scholar] [CrossRef]
- Miljanić, V.; Beber, A.; Rusjan, D.; Stajner, N. First report of grapevine satellite virus in Slovenia. J. Plant Pathol. 2021, 103, 1329–1330. [Google Scholar] [CrossRef]
- Flores, R.; Randles, J.; Bar-Joseph, M.; Diener, T.O. A proposed scheme for viroid classification and nomenclature. Arch. Virol. 1998, 143, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Fajardo, T.V.M.; Eiras, M.; Nickel, O. Detection and molecular characterization of Grapevine yellow speckle viroid 1 isolates infecting grapevines in Brazil. Trop. Plant Pathol. 2016, 41, 246–253. [Google Scholar] [CrossRef]
- Faggioli, F.; Duran-Vila, N.; Tsagris, M.; Pallás, V. Geographical Distribution of Viroids in Europe. In Viroids and Satellites; Academic Press: Cambridge, MA, USA, 2017; pp. 473–484. [Google Scholar] [CrossRef]
- Jiang, D.; Zhang, Z.; Wu, Z.; Guo, R.; Wang, H.; Fan, P.; Li, S. Molecular characterization of grapevine yellow speckle viroid-2 (GYSVd-2). Virus Genes 2009, 38, 515–520. [Google Scholar] [CrossRef][Green Version]
- Adkar-Purushothama, C.R.; Kanchepalli, P.R.; Yanjarappa, S.M.; Zhang, Z.; Sano, T. Detection, distribution, and genetic diversity of Australian grapevine viroid in grapevines in India. Virus Genes 2014, 49, 304–311. [Google Scholar] [CrossRef]
- Ryabov, E.V.; Taliansky, M.E.; Robinson, D.J.; Waterhouse, P.M.; Murant, A.F.; de Zoeten, G.A.; Falk, B.W.; Vetten, H.J.; Gibbs, M.J. Genus Umbravirus. In Virus Taxonomy Classification and Nomenclature of Viruses Ninth Report of the International Committee on Taxonomy of Viruses; King, A.M.Q., Adams, M.J., Carstens, E.B., Lefkowitz, E.J., Eds.; Academic Press: London, UK, 2011; Volume 9, pp. 1191–1195. ISBN 1865843830. [Google Scholar]
- Ryabov, E.V.; Taliansky, M.E. Umbraviruses (Calvusvirinae, Tombusviridae). In Encyclopedia of Virology; Academic Press: Cambridge, MA, USA, 2021; ISBN 9780128096338. [Google Scholar]
- Geering, A.D.W.; Hull, R. Genus Caulimovirus. In Virus Taxonomy Classification and Nomenclature of Viruses Ninth Report of the International Committee on Taxonomy of Viruses; King, A.M.Q., Adams, M.J., Carstens, E.B., Lefkowitz, E.J., Eds.; Academic Press: London, UK, 2011; Volume 9, pp. 432–434. ISBN 1865843830. [Google Scholar]
- Teycheney, P.Y.; Geering, A.D.W.; Dasgupta, I.; Hull, R.; Kreuze, J.F.; Lockhart, B.; Muller, E.; Olszewski, N.; Pappu, H.; Pooggin, M.M.; et al. ICTV Virus Taxonomy Profile: Caulimoviridae. J. Gen. Virol. 2020, 101, 1025. [Google Scholar] [CrossRef]
- Hu, R.; Dias, N.P.; Soltani, N.; Vargas-Asencio, J.; Hensley, D.D.; Perry, K.L.; Domier, L.L.; Hajimorad, M.R. Cultivated and wild grapevines in tennessee possess overlapping but distinct virus populations. Plant Dis. 2021, 105, 2785–2791. [Google Scholar] [CrossRef] [PubMed]
- Pankin, M.I.; Petrov, V.S.; Lukianova, A.A.; Ilnitskaya, E.T.; Nikulushkina, G.E.; Kovalenko, A.G.; Bolshakov, V.A. The Anapa ampelographic collection is the largest center of vine gene pool accumulation and research in Russia. Vavilovskii Zhurnal Genet. Selektsii 2018, 22, 54–59. [Google Scholar] [CrossRef]
- Nooh, S. An overview of oilseed rape (canola) virus diseases in Iran. Int. Res. J. Microbiol. 2012, 3, 24–28. [Google Scholar]
- Yadav, P.K.; Kumar, S.; Yadav, S.; Kumar, S. Role of aptamers in plant defense mechanism against viral diseases. In Aptamers; Springer: Singapore, 2019; pp. 169–174. [Google Scholar] [CrossRef]
- Şevik, M.A. Impact of Cauliflower mosaic virus Infection on Morphological Parameters of Radish Plants. Alinteri J. Agric. Sci. 2020, 35, 27–29. [Google Scholar] [CrossRef]
- Anitha, S.; Monyo, E.S.; Okori, P. Simultaneous detection of groundnut rosette assistor virus (GRAV), groundnut rosette virus (GRV) and satellite RNA (satRNA) in groundnuts using multiplex RT-PCR. Arch. Virol. 2014, 159, 3059–3062. [Google Scholar] [CrossRef]
- Vinogradova, S.V.; Porotikova, E.V.; Navrotskaya, E.V.; Massart, S.; Varallyay, E. The First Virome of Russian Vineyard. Plants 2022, in press. [Google Scholar]
- Reynard, J.S.; Brodard, J.; Dubuis, N.; Yobregat, O.; Kominek, P.; Schumpp, O.; Schaerer, S. First Report of Grapevine rupestris vein feathering virus in Swiss Grapevines. Plant Dis. 2017, 101, 1062. [Google Scholar] [CrossRef]
- Gambino, G.; Gribaudo, I. Simultaneous Detection of Nine Grapevine Viruses by Multiplex Reverse Transcription-Polymerase Chain Reaction with Coamplification of a Plant RNA as Internal Control. Phytopathology 2007, 96, 1223–1229. [Google Scholar] [CrossRef]
- Hajizadeh, M.; Navarro, B.; Bashir, N.S.; Torchetti, E.M.; Di Serio, F. Development and validation of a multiplex RT-PCR method for the simultaneous detection of five grapevine viroids. J. Virol. Methods 2012, 179, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Gambino, G.; Bondaz, J.; Gribaudo, I. Detection and Elimination of Viruses in Callus, Somatic Embryos and Regenerated Plantlets of Grapevine. Eur. J. Plant Pathol. 2006, 114, 397–404. [Google Scholar] [CrossRef]
- Daldoul, S.; Rheinpfalz, D.L.R.; Massart, S.; Ruiz-García, A.B.; Olmos, A.; Wetzel, T. First Report of Grapevine rupestris vein feathering virus in grapevine in Germany. Plant Dis. 2018, 102, 2029. [Google Scholar] [CrossRef]
- Digiaro, M.; Elbeaino, T.; Martelli, G.P. Development of degenerate and species-specific primers for the differential and simultaneous RT-PCR detection of grapevine-infecting nepoviruses of subgroups A, B and C. J. Virol. Methods 2007, 141, 34–40. [Google Scholar] [CrossRef]
- Osman, F.; Rowhani, A. Application of a spotting sample preparation technique for the detection of pathogens in woody plants by RT-PCR and real-time PCR (TaqMan). J. Virol. Methods 2006, 133, 130–136. [Google Scholar] [CrossRef]
- Čepin, U.; Gutiérrez-Aguirre, I.; Ravnikar, M.; Pompe-Novak, M. Frequency of occurrence and genetic variability of Grapevine fanleaf virus satellite RNA. Plant Pathol. 2016, 65, 510–520. [Google Scholar] [CrossRef]
- Osman, F.; Rowhani, A. Real-time RT-PCR (TaqMan®) assays for the detection of viruses associated with Rugose wood complex of grapevine. J. Virol. Methods 2008, 154, 69–75. [Google Scholar] [CrossRef]
- Glasa, M.; Predajňa, L.; Komínek, P.; Nagyová, A.; Candresse, T.; Olmos, A. Molecular characterization of divergent grapevine Pinot gris virus isolates and their detection in Slovak and Czech grapevines. Arch. Virol. 2014, 159, 2103–2107. [Google Scholar] [CrossRef] [PubMed]
- Goszczynski, D.E.; Jooste, A.E.C. Identification of divergent variants of Grapevine virus A. Eur. J. Plant Pathol. 2003, 109, 397–403. [Google Scholar] [CrossRef]
- Bianchi, G.L.; De Amicis, F.; De Sabbata, L.; Di Bernardo, N.; Governatori, G.; Nonino, F.; Prete, G.; Marrazzo, T.; Versolatto, S.; Frausin, C. Occurrence of grapevine pinot gris virus in friuli venezia giulia (Italy): Field monitoring and virus quantification by real-time RT-PCR. EPPO Bull. 2015, 45, 22–32. [Google Scholar] [CrossRef]
- Bruisson, S.; Lebel, S.; Walter, B.; Prevotat, L.; Seddas, S.; Schellenbaum, P. Comparative detection of a large population of grapevine viruses by TaqMan® RT-qPCR and ELISA. J. Virol. Methods 2017, 240, 73–77. [Google Scholar] [CrossRef]
- Sabella, E.; Pierro, R.; Luvisi, A.; Panattoni, A.; D’Onofrio, C.; Scalabrelli, G.; Nutricati, E.; Aprile, A.; De Bellis, L.; Materazzi, A. Phylogenetic analysis of viruses in Tuscan Vitis vinifera sylvestris (Gmeli) Hegi. PLoS One 2018, 13, e0200875. [Google Scholar] [CrossRef] [PubMed]
- Zarghani, S.N.; Hily, J.M.; Glasa, M.; Marais, A.; Wetzel, T.; Faure, C.; Vigne, E.; Velt, A.; Lemaire, O.; Boursiquot, J.M.; et al. Grapevine virus T diversity as revealed by full-length genome sequences assembled from high-throughput sequence data. PLoS One 2018, 13, e0206010. [Google Scholar] [CrossRef]
- Beuve, M.; Hily, J.M.; Alliaume, A.; Reinbold, C.; Le Maguet, J.; Candresse, T.; Herrbach, E.; Lemaire, O. A complex virome unveiled by deep sequencing analysis of RNAs from a French Pinot Noir grapevine exhibiting strong leafroll symptoms. Arch. Virol. 2018, 163, 2937–2946. [Google Scholar] [CrossRef]
- Maddahian, M.; Massumi, H.; Heydarnejad, J.; Hosseinipour, A.; Khezri, A.; Sano, T. Biological and molecular characterization of hop stunt viroid variants from pistachio trees in Iran. J. Phytopathol. 2019, 167, 163–173. [Google Scholar] [CrossRef]
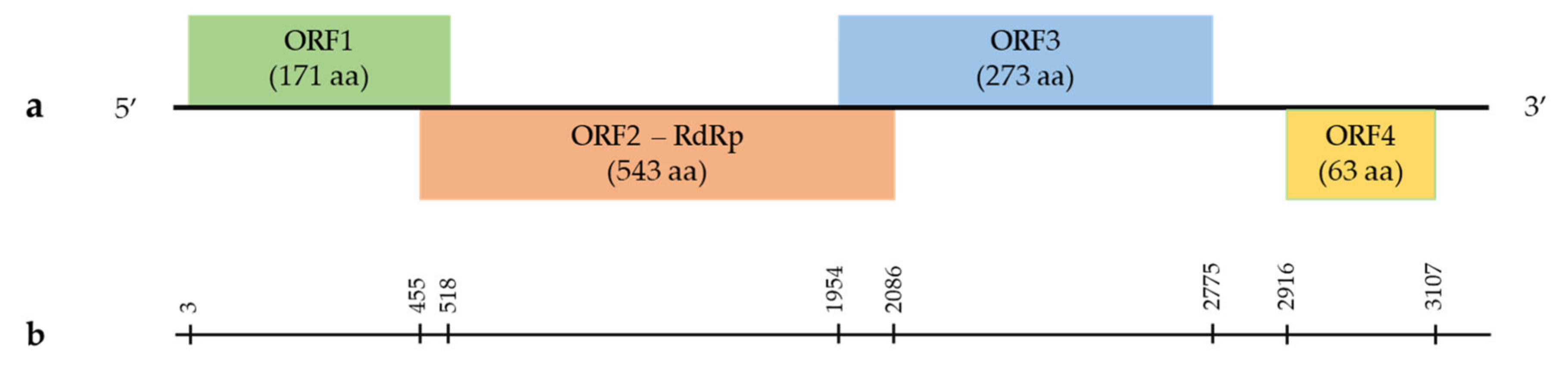
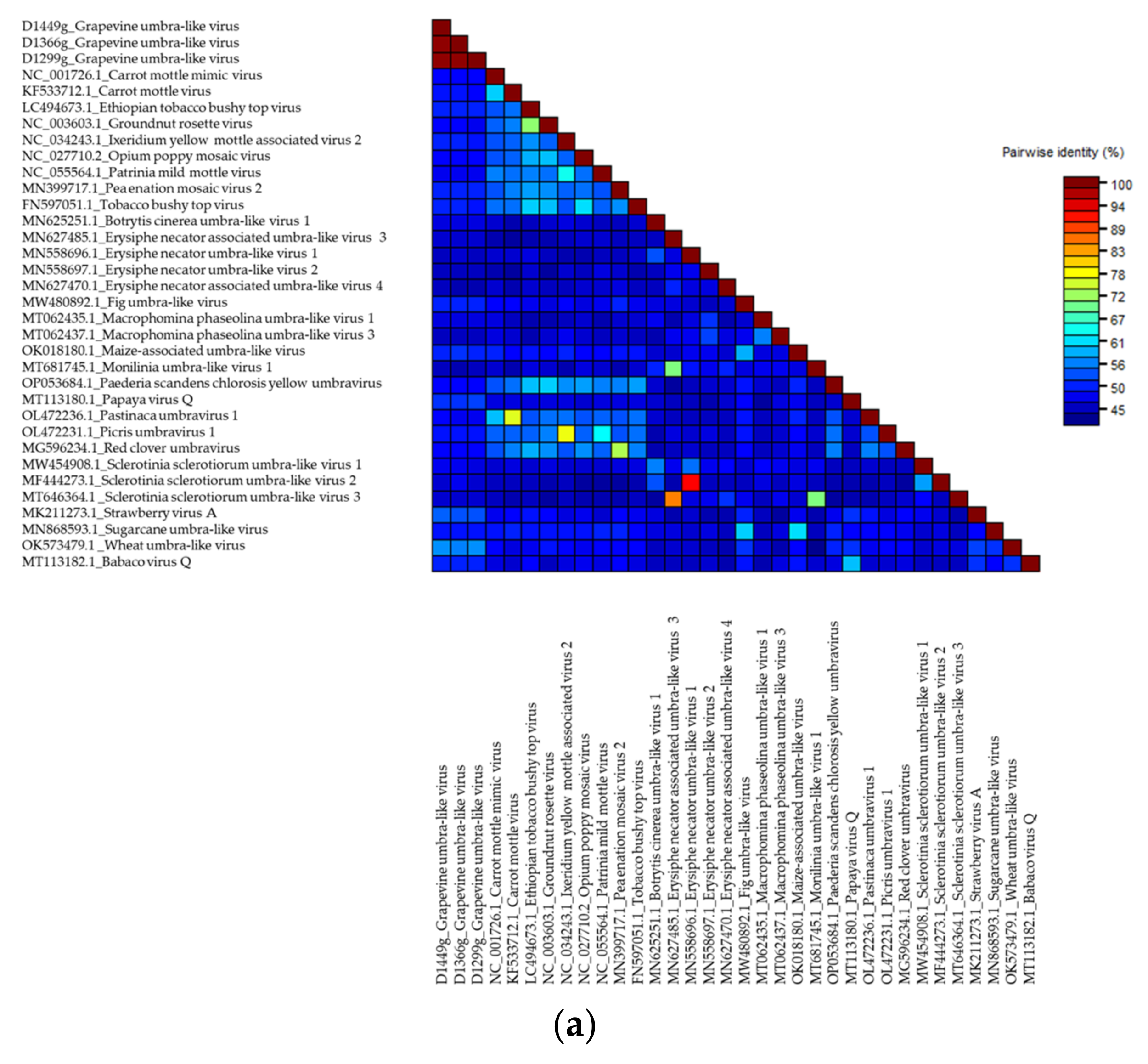
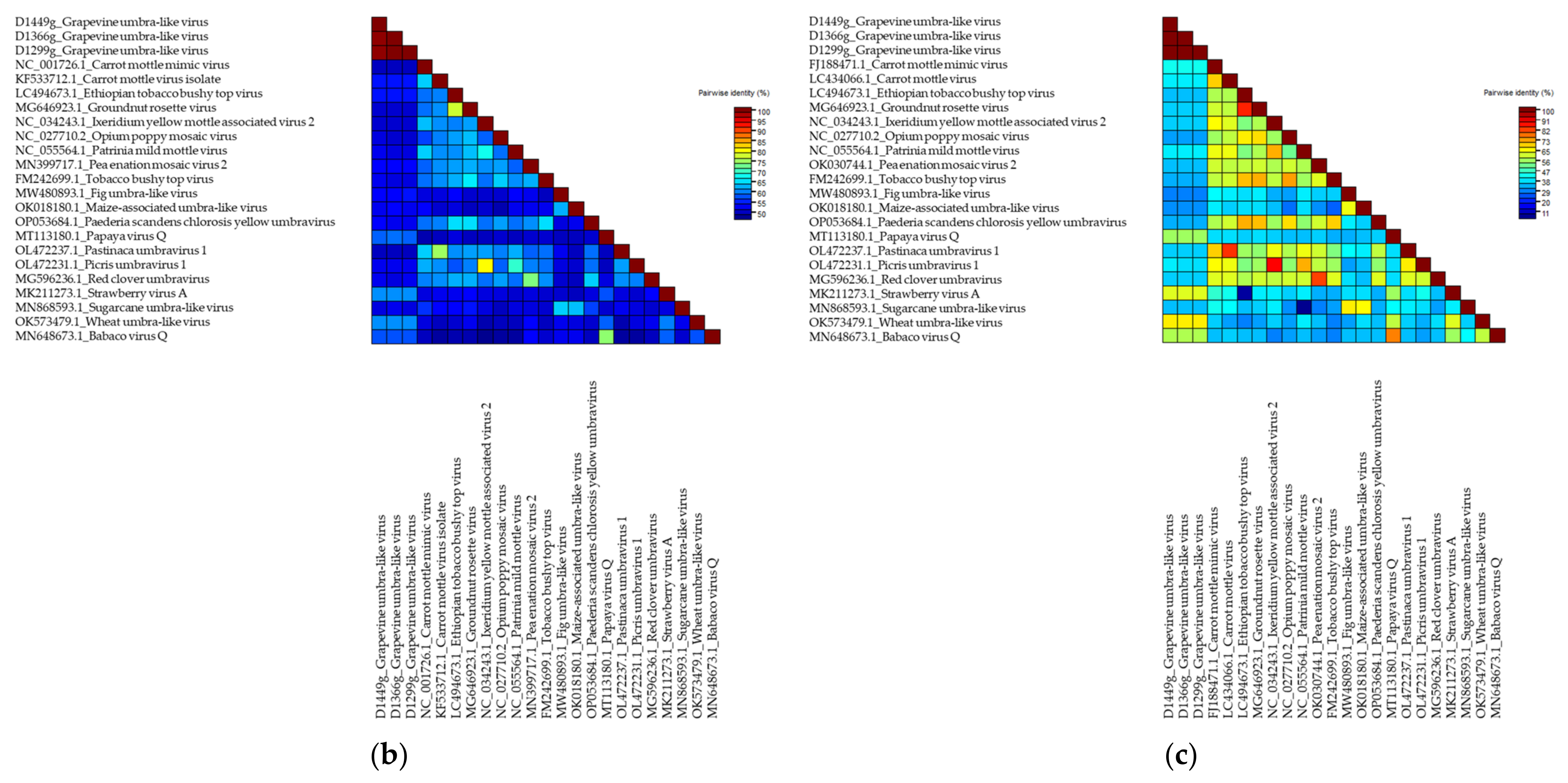

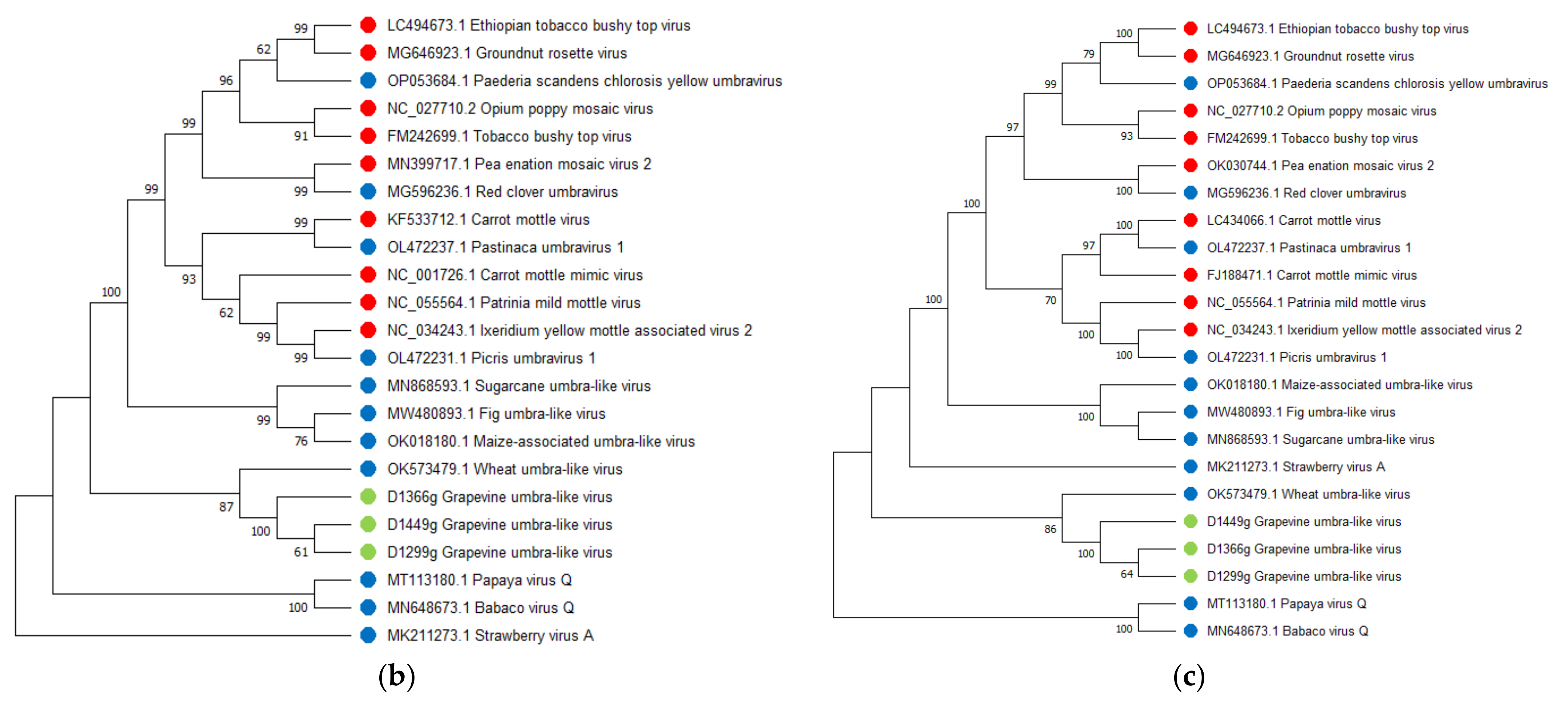
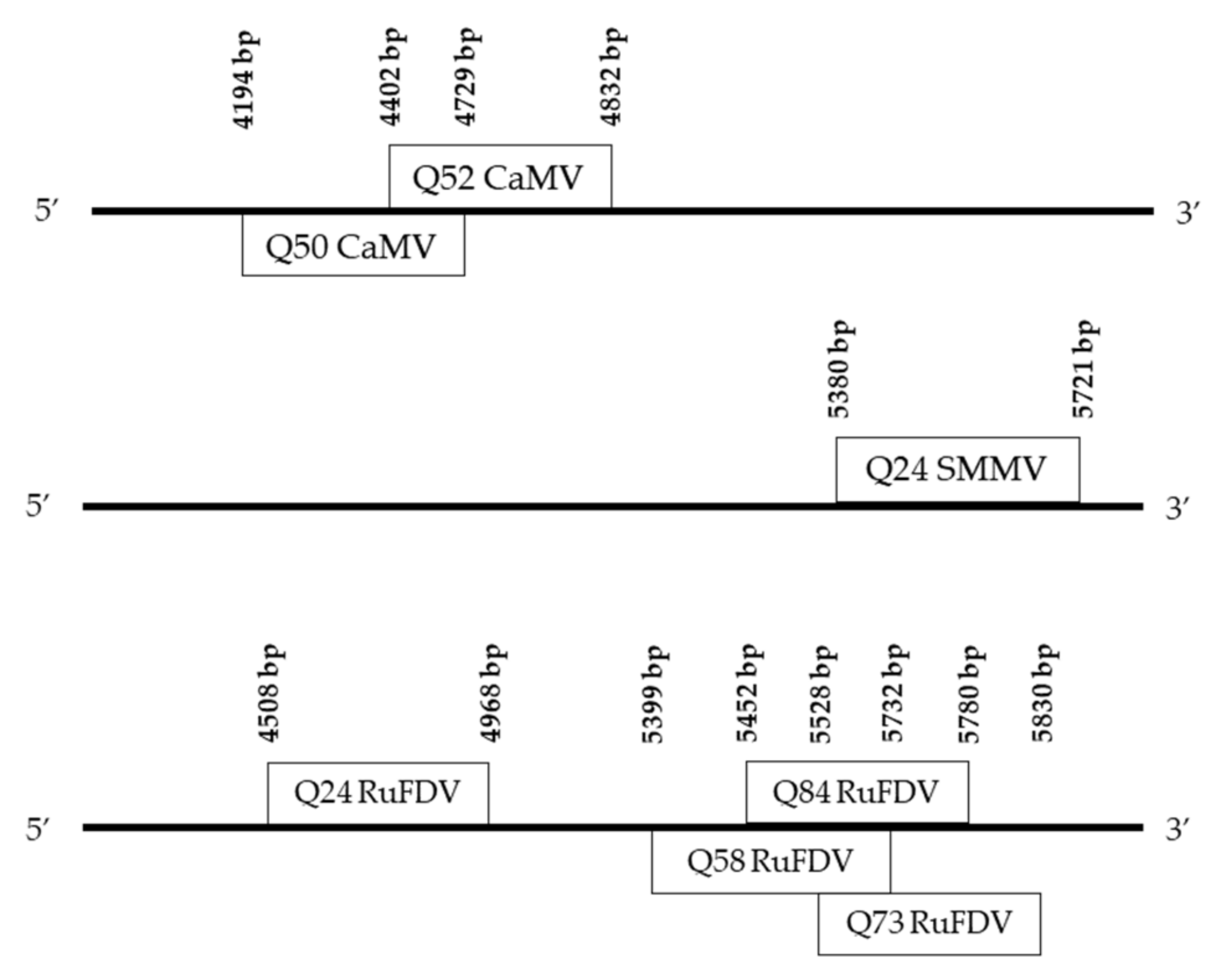
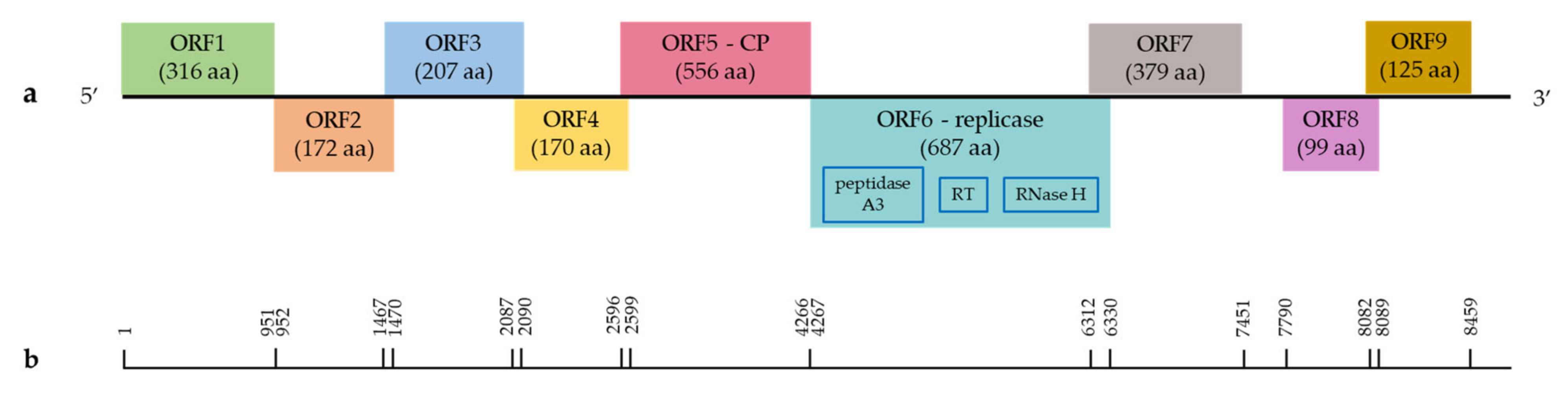
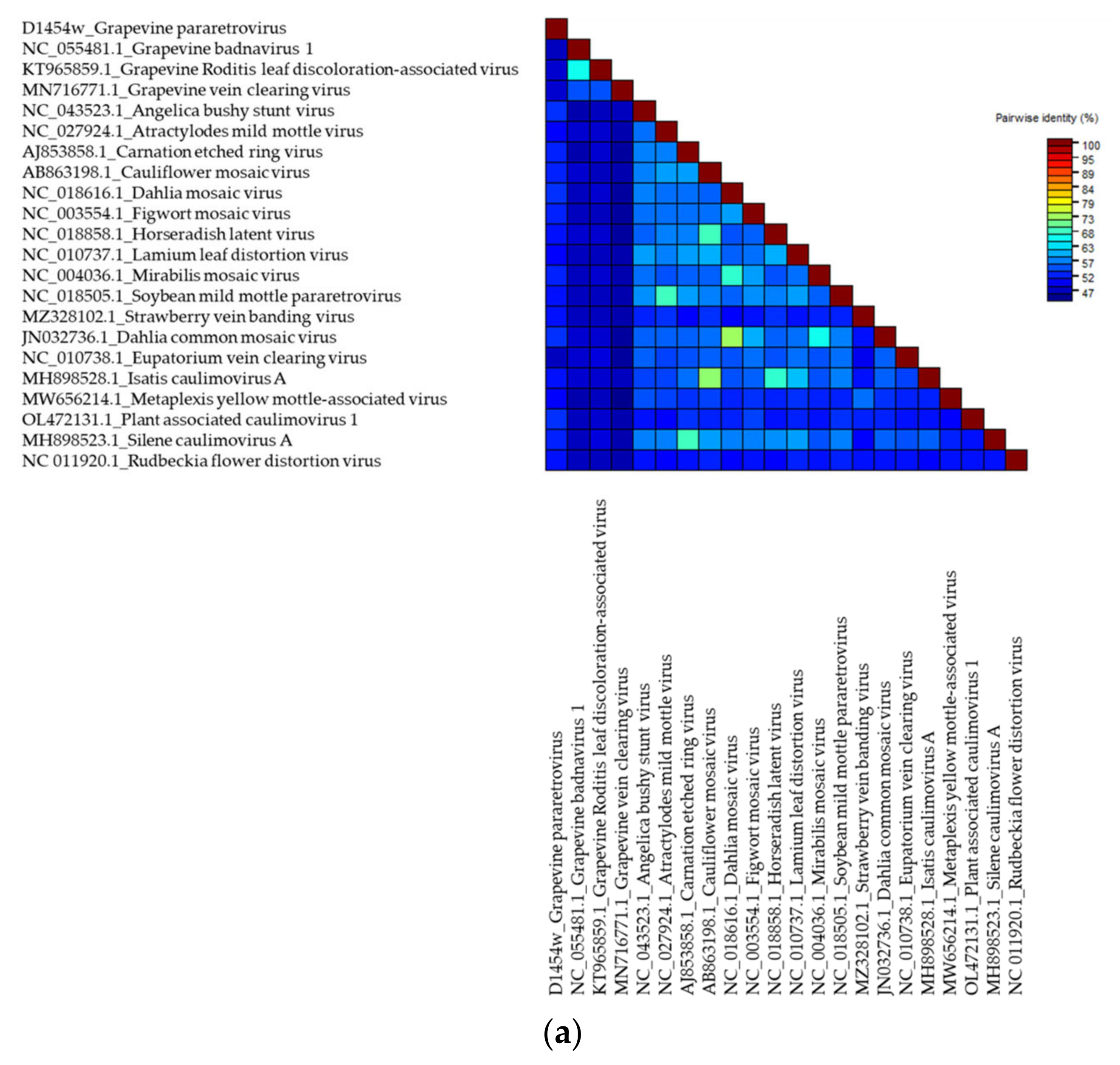
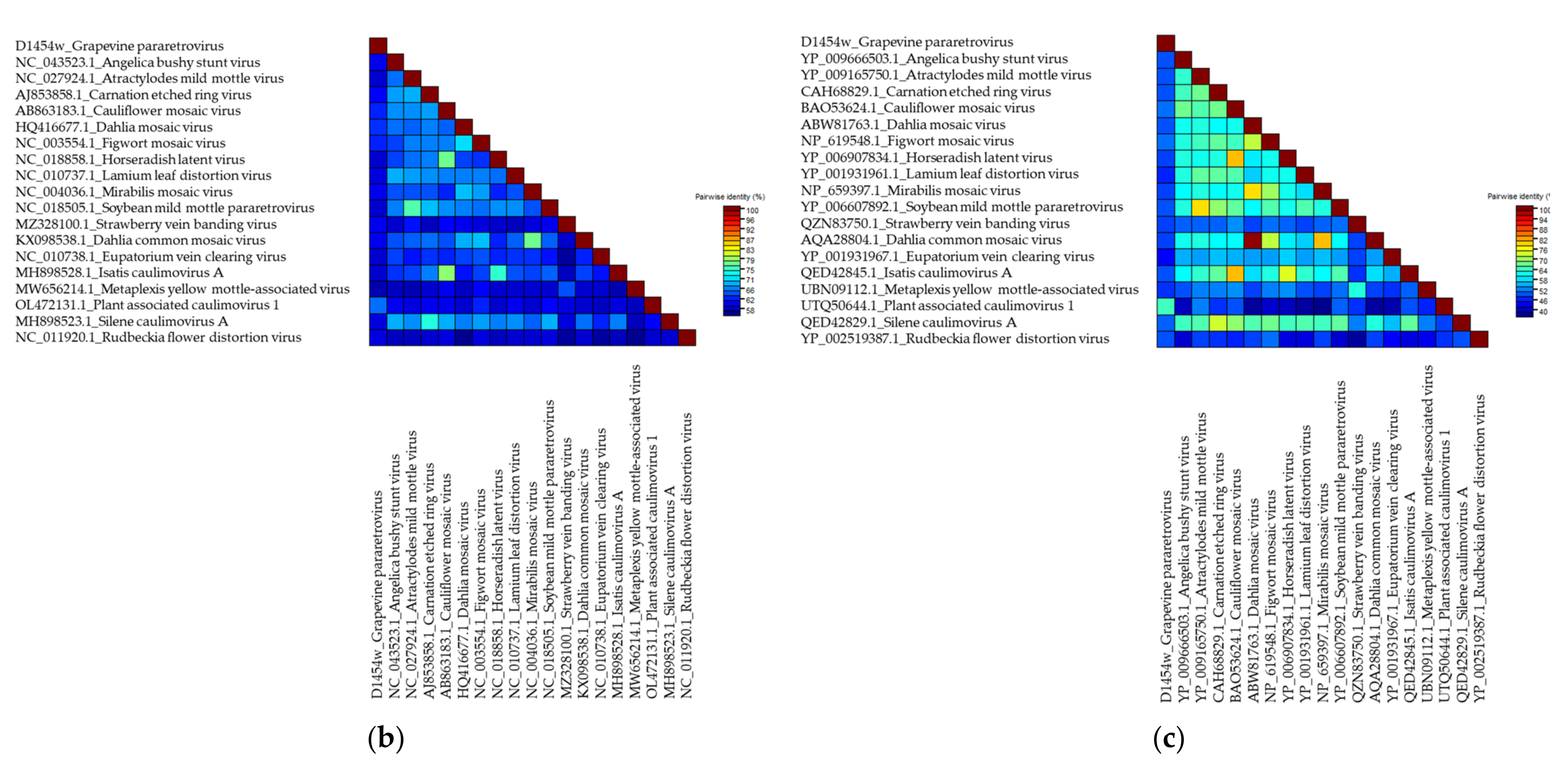

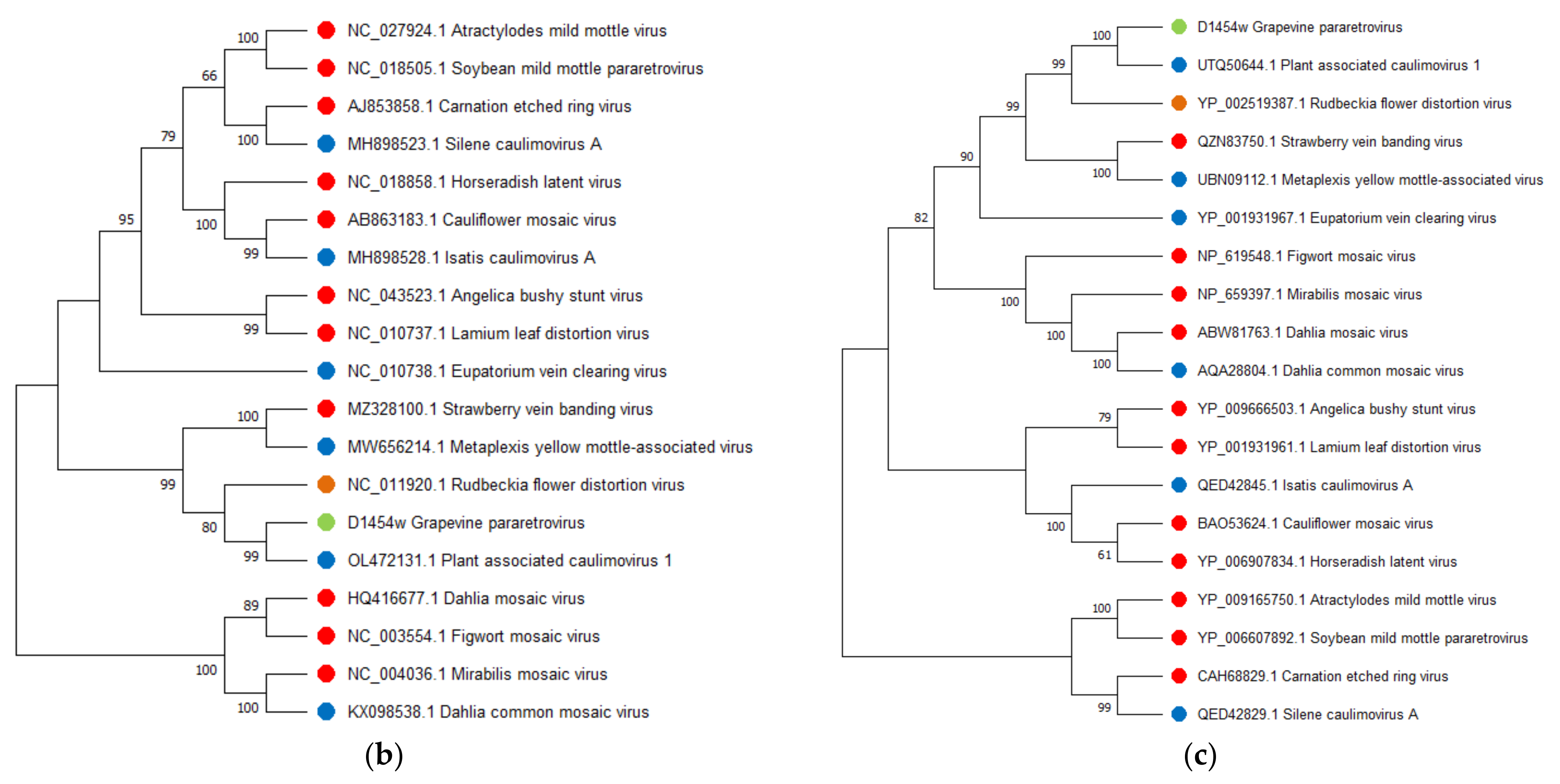
 .
.
 .
.
| Library | tblastx of Contigs | blastn (or blastx for Q73) of Contigs by NCBI Tool | |||||
|---|---|---|---|---|---|---|---|
| Virus Name | Contig E-Value | Contig Length, bp | Closest Isolate | Percent Identity, % | Query Cover, % | GenBank Identifier | |
| Q24 | RuFDV * | –51 | 461 | PaCV-1 | 70.26% | 91% | OL472131.1 |
| CaMV | 68.49% | 86% | AB863188.1 | ||||
| SMMP | –38 | 342 | DMV | 67.93% | 84% | HQ416677.1 | |
| AnBSV | 66.67% | 92% | NC_043523.1 | ||||
| Q50 | CaMV | –36 | 432 | DMV | 71.08% | 47% | EF203675.1 |
| CaMV | 70.26% | 61% | LC632935.1 | ||||
| HRLV | 68.34% | 46% | JX429923.1 | ||||
| MMV | 69.15% | 43% | AF454635.1 | ||||
| FMV | 67.65% | 47% | X06166.1 | ||||
| Q52 | CaMV | –58 | 431 | PaCV-1 | 74.84% | 72% | OL472131.1 |
| CaMV | 70.39% | 89% | KY810770.1 | ||||
| Q58 | RuFDV | –37 | 318 | PaCV-1 | 72.77% | 73% | OL472131.1 |
| AMMV | 68.42% | 82% | KR080327.1 | ||||
| FMV | 66.41% | 81% | X06166.1 | ||||
| AnBSV | 66.67% | 77% | NC_043523.1 | ||||
| Q73 | RuFDV | –16 | 265 | PaCV-1 | 68.09% | 53% | UTQ50644.1 |
| RuFDV | 64.81% | 61% | YP_002519387.1 | ||||
| CaMV | 64.81% | 61% | AB863188.1 | ||||
| DMV | 61.70% | 53% | AEB54984.1 | ||||
| LLDV | 63.64% | 49% | YP_001931961.1 | ||||
| Q84 | RuFDV | –30 | 299 | PaCV-1 | 69.85% | 66% | OL472131.1 |
| FMV | 70.76% | 57% | X06166.1 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shvets, D.; Sandomirsky, K.; Porotikova, E.; Vinogradova, S. Metagenomic Analysis of Ampelographic Collections of Dagestan Revealed the Presence of Two Novel Grapevine Viruses. Viruses 2022, 14, 2623. https://doi.org/10.3390/v14122623
Shvets D, Sandomirsky K, Porotikova E, Vinogradova S. Metagenomic Analysis of Ampelographic Collections of Dagestan Revealed the Presence of Two Novel Grapevine Viruses. Viruses. 2022; 14(12):2623. https://doi.org/10.3390/v14122623
Chicago/Turabian StyleShvets, Darya, Kirill Sandomirsky, Elena Porotikova, and Svetlana Vinogradova. 2022. "Metagenomic Analysis of Ampelographic Collections of Dagestan Revealed the Presence of Two Novel Grapevine Viruses" Viruses 14, no. 12: 2623. https://doi.org/10.3390/v14122623
APA StyleShvets, D., Sandomirsky, K., Porotikova, E., & Vinogradova, S. (2022). Metagenomic Analysis of Ampelographic Collections of Dagestan Revealed the Presence of Two Novel Grapevine Viruses. Viruses, 14(12), 2623. https://doi.org/10.3390/v14122623







