Antiviral Potential of Natural Resources against Influenza Virus Infections
Abstract
1. Introduction
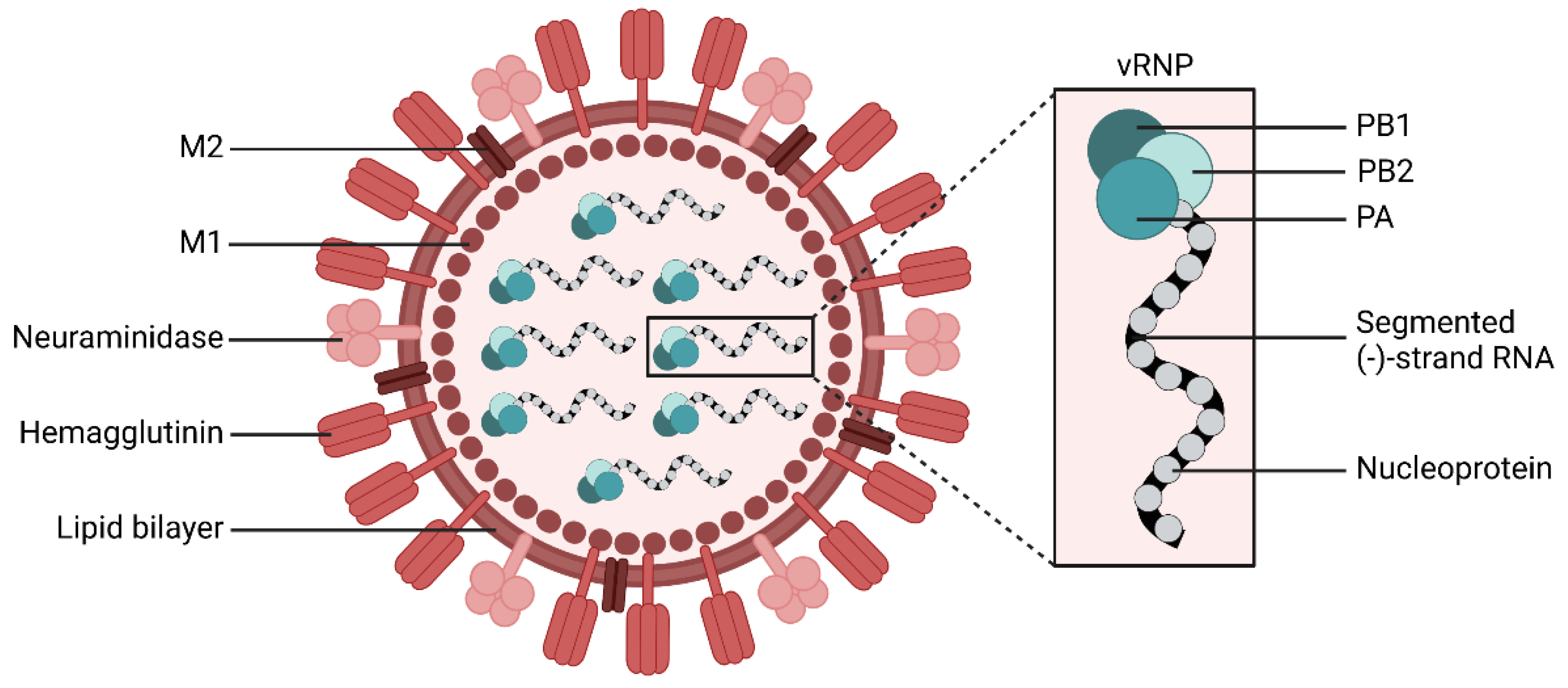
1.1. Biology of Influenza Viruses
1.2. Antiviral Therapy of Influenza Infections
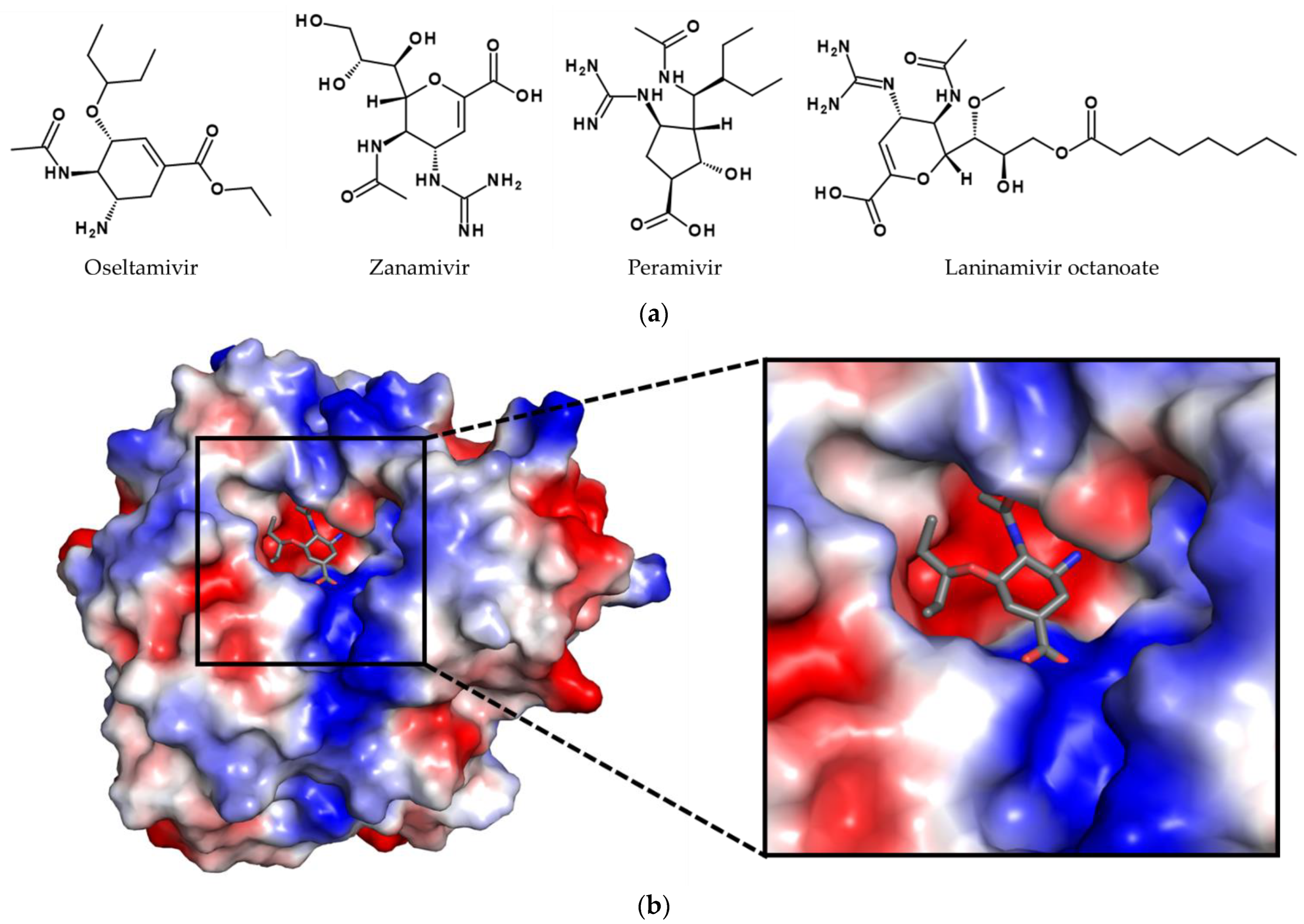
2. Natural Resources in Drug Development
3. Toxins and Defensive Peptides from Animals Tested for Antiviral Properties
4. Fungal Extracts and Compounds with Anti-Influenza Properties
4.1. Terrestrial Fungi

4.2. Marine and Limnic Fungi
5. Bacterial Extracts and Compounds with Anti-Influenza Properties
5.1. Bacterial Extract Screenings
5.2. Cyanobacteria
5.3. Pseudomonadota
5.4. Actinomycetota
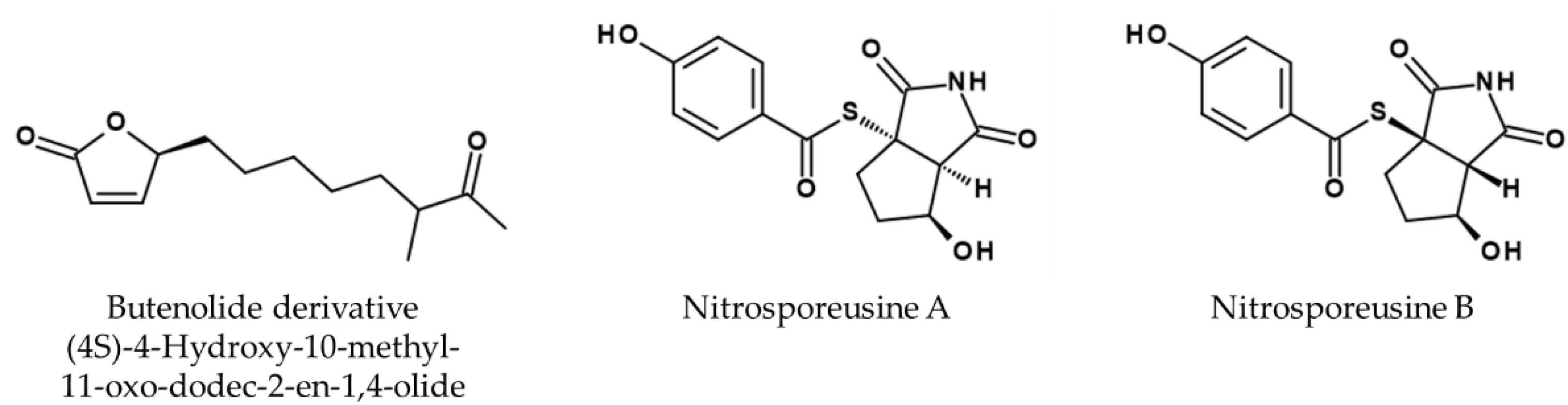
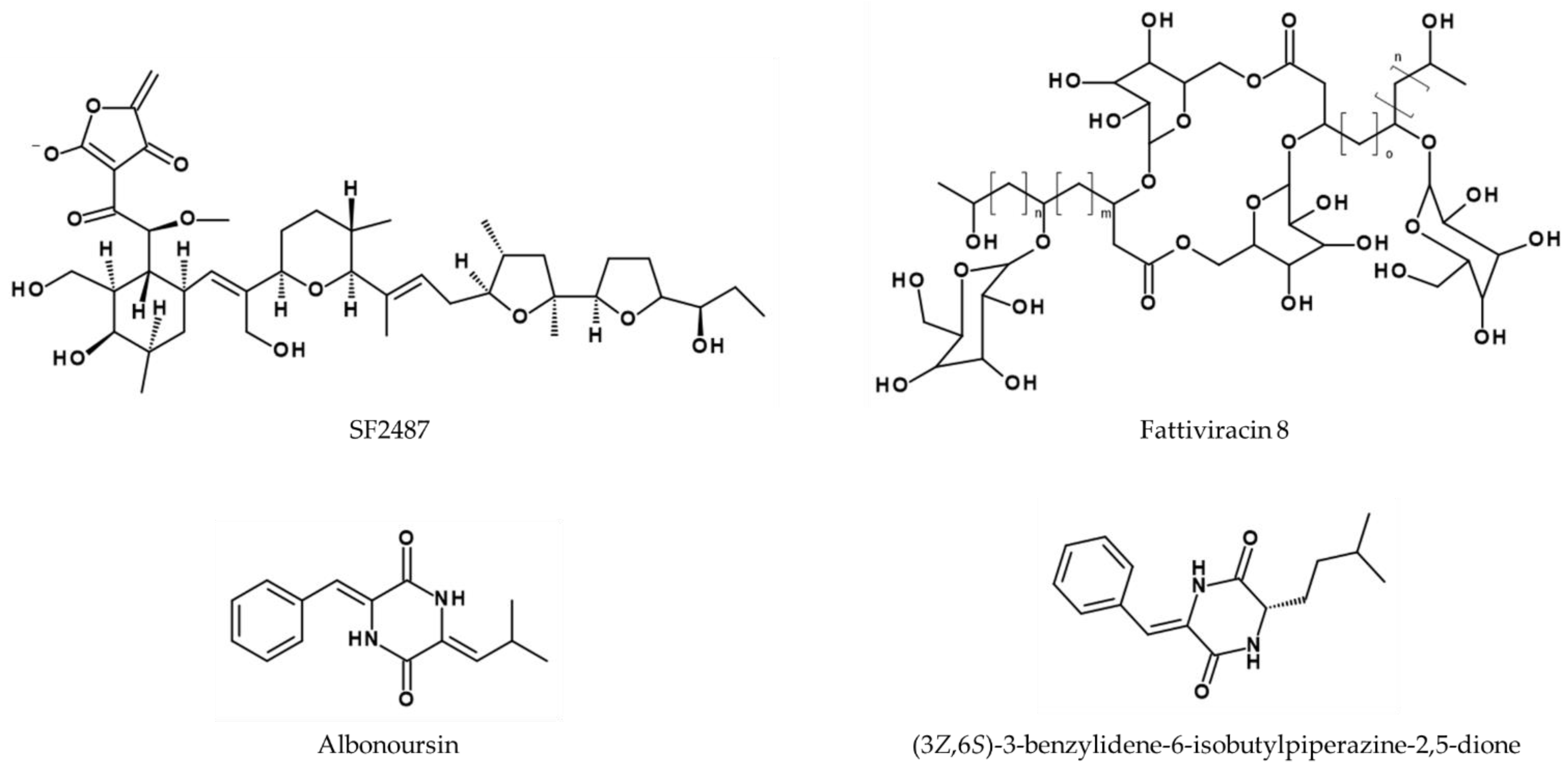
6. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Nomenclature
- EC50—Effective concentration of a compound where the infectivity is reduced by half
- IC50—Inhibitory concentration of an inhibitor where the response is reduced by half
- ID50—Virus induced cytopathic effect of an inhibitor is reduced by half
- MLD50—Murine lethal dose of a substance needed to kill half the members of the tested population
- TD50—Median toxic dose of a drug at which toxicity occurs in 50% of the cases
References
- Javanian, M.; Barary, M.; Ghebrehewet, S.; Koppolu, V.; Vasigala, V.; Ebrahimpour, S. A brief review of influenza virus infection. J. Med. Virol. 2021, 93, 4638–4646. [Google Scholar] [CrossRef]
- Krammer, F.; Smith, G.J.D.; Fouchier, R.A.M.; Peiris, M.; Kedzierska, K.; Doherty, P.C.; Palese, P.; Shaw, M.L.; Treanor, J.; Webster, R.G.; et al. Influenza. Nat. Rev. Dis. Primers 2018, 4, 3. [Google Scholar] [CrossRef]
- Szucs, T. The socio-economic burden of influenza. J. Antimicrob. Chemother. 1999, 44 (Suppl. B), 11–15. [Google Scholar] [CrossRef]
- Gasparini, R.; Amicizia, D.; Lai, P.L.; Panatto, D. Clinical and socioeconomic impact of seasonal and pandemic influenza in adults and the elderly. Hum. Vaccines Immunother. 2012, 8, 21–28. [Google Scholar] [CrossRef]
- Pfleiderer, M. Grippe-Impfstoffe. Pharm. Unserer Zeit 2011, 40, 126–134. [Google Scholar] [CrossRef]
- Girard, M.P.; Tam, J.S.; Assossou, O.M.; Kieny, M.P. The 2009 A (H1N1) influenza virus pandemic: A review. Vaccine 2010, 28, 4895–4902. [Google Scholar] [CrossRef]
- Webster, R.G.; Bean, W.J.; Gorman, O.T.; Chambers, T.M.; Kawaoka, Y. Evolution and ecology of influenza A viruses. Microbiol. Rev. 1992, 56, 152–179. [Google Scholar] [CrossRef]
- Olsen, B.; Munster, V.J.; Wallensten, A.; Waldenström, J.; Osterhaus, A.D.M.E.; Fouchier, R.A.M. Global patterns of influenza a virus in wild birds. Science 2006, 312, 384–388. [Google Scholar] [CrossRef]
- Medina, R.A.; García-Sastre, A. Influenza A viruses: New research developments. Nat. Rev. Microbiol. 2011, 9, 590–603. [Google Scholar] [CrossRef]
- Gamblin, S.J.; Skehel, J.J. Influenza hemagglutinin and neuraminidase membrane glycoproteins. J. Biol. Chem. 2010, 285, 28403–28409. [Google Scholar] [CrossRef]
- Matrosovich, M.; Stech, J.; Klenk, H.D. Influenza receptors, polymerase and host range. Rev. Sci. Tech. 2009, 28, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Marjuki, H.; Gornitzky, A.; Marathe, B.M.; Ilyushina, N.A.; Aldridge, J.R.; Desai, G.; Webby, R.J.; Webster, R.G. Influenza A virus-induced early activation of ERK and PI3K mediates V-ATPase-dependent intracellular pH change required for fusion. Cell. Microbiol. 2011, 13, 587–601. [Google Scholar] [CrossRef] [PubMed]
- Zambon, M.C. Epidemiology and pathogenesis of influenza. J. Antimicrob. Chemother. 1999, 44 (Suppl. B), 3–9. [Google Scholar] [CrossRef] [PubMed]
- Garten, W.; Klenk, H.D. Understanding influenza virus pathogenicity. Trends Microbiol. 1999, 7, 99–100. [Google Scholar] [CrossRef]
- Garten, W.; Klenk, H.-D. Cleavage Activation of the Influenza Virus Hemagglutinin and Its Role in Pathogenesis. In Avian Influenza; Klenk, H.-D., Matrosovich, M.N., Stech, J., Eds.; KARGER: Basel, Switzerland, 2008; pp. 156–167. ISBN 978-3-8055-8501-9. [Google Scholar]
- Gong, J.; Fang, H.; Li, M.; Liu, Y.; Yang, K.; Liu, Y.; Xu, W. Potential targets and their relevant inhibitors in anti-influenza fields. Curr. Med. Chem. 2009, 16, 3716–3739. [Google Scholar] [CrossRef]
- Gabriel, G.; Klingel, K.; Otte, A.; Thiele, S.; Hudjetz, B.; Arman-Kalcek, G.; Sauter, M.; Shmidt, T.; Rother, F.; Baumgarte, S.; et al. Differential use of importin-α isoforms governs cell tropism and host adaptation of influenza virus. Nat. Commun. 2011, 2, 156. [Google Scholar] [CrossRef]
- Hay, A.J.; Wolstenholme, A.J.; Skehel, J.J.; Smith, M.H. The molecular basis of the specific anti-influenza action of amantadine. EMBO J. 1985, 4, 3021–3024. [Google Scholar] [CrossRef]
- Duwe, S. Influenza viruses—Antiviral therapy and resistance. GMS Infect. Dis. 2017, 5, Doc04. [Google Scholar] [CrossRef]
- Bright, R.A.; Medina, M.; Xu, X.; Perez-Oronoz, G.; Wallis, T.R.; Davis, X.M.; Povinelli, L.; Cox, N.J.; Klimov, A.I. Incidence of adamantane resistance among influenza A (H3N2) viruses isolated worldwide from 1994 to 2005: A cause for concern. Lancet 2005, 366, 1175–1181. [Google Scholar] [CrossRef]
- Hayden, F. Developing new antiviral agents for influenza treatment: What does the future hold? Clin. Infect. Dis. 2009, 48 (Suppl. 1), S3–S13. [Google Scholar] [CrossRef]
- Dong, G.; Peng, C.; Luo, J.; Wang, C.; Han, L.; Wu, B.; Ji, G.; He, H. Adamantane-resistant influenza a viruses in the world (1902–2013): Frequency and distribution of M2 gene mutations. PLoS ONE 2015, 10, e0119115. [Google Scholar] [CrossRef]
- Stouffer, A.L.; Acharya, R.; Salom, D.; Levine, A.S.; Di Costanzo, L.; Soto, C.S.; Tereshko, V.; Nanda, V.; Stayrook, S.; DeGrado, W.F. Structural basis for the function and inhibition of an influenza virus proton channel. Nature 2008, 451, 596–599. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Chisti, Y.; Banerjee, U.C. Production of shikimic acid. Biotechnol. Adv. 2012, 30, 1425–1431. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed]
- McAuley, J.L.; Gilbertson, B.P.; Trifkovic, S.; Brown, L.E.; McKimm-Breschkin, J.L. Influenza Virus Neuraminidase Structure and Functions. Front. Microbiol. 2019, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Steinmetzer, T.; Hardes, K.; Böttcher-Friebertshäuser, E.; Garten, W. Strategies for the Development of Influenza Drugs: Basis for New Efficient Combination Therapies. In Therapy of Viral Infections; Diederich, W.E., Steuber, H., Eds.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 143–181. ISBN 978-3-662-46758-9. [Google Scholar]
- Edmond, J.D.; Johnston, R.G.; Kidd, D.; Rylance, H.J.; Sommerville, R.G. The inhibition of neuraminidase and antiviral action. Br. J. Pharmacol. Chemother. 1966, 27, 415–426. [Google Scholar] [CrossRef]
- Nath, N.K.; Yasuda, N.; Rabeh, W.M.; Sahoo, S.C.; Naumov, P. Structural Elucidation of the Neuraminidase Inhibitor Zanamivir (Relenza): Creeping and Diffusion for Polymorph Separation. Cryst. Growth Des. 2014, 14, 770–774. [Google Scholar] [CrossRef]
- Holmes, E.H.; Devalapally, H.; Li, L.; Perdue, M.L.; Ostrander, G.K. Permeability enhancers dramatically increase zanamivir absolute bioavailability in rats: Implications for an orally bioavailable influenza treatment. PLoS ONE 2013, 8, e61853. [Google Scholar] [CrossRef]
- Shie, J.-J.; Fang, J.-M. Development of effective anti-influenza drugs: Congeners and conjugates—A review. J. Biomed. Sci. 2019, 26, 84. [Google Scholar] [CrossRef]
- Dharan, N.J.; Gubareva, L.V.; Meyer, J.J.; Okomo-Adhiambo, M.; McClinton, R.C.; Marshall, S.A.; St George, K.; Epperson, S.; Brammer, L.; Klimov, A.I.; et al. Infections with oseltamivir-resistant influenza A(H1N1) virus in the United States. JAMA 2009, 301, 1034–1041. [Google Scholar] [CrossRef]
- Lampejo, T. Influenza and antiviral resistance: An overview. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 1201–1208. [Google Scholar] [CrossRef] [PubMed]
- Mishin, V.P.; Hayden, F.G.; Gubareva, L.V. Susceptibilities of antiviral-resistant influenza viruses to novel neuraminidase inhibitors. Antimicrob. Agents Chemother. 2005, 49, 4515–4520. [Google Scholar] [CrossRef] [PubMed]
- Moscona, A. Oseltamivir resistance--disabling our influenza defenses. N. Engl. J. Med. 2005, 353, 2633–2636. [Google Scholar] [CrossRef]
- Principi, N.; Camilloni, B.; Alunno, A.; Polinori, I.; Argentiero, A.; Esposito, S. Drugs for Influenza Treatment: Is There Significant News? Front. Med. 2019, 6, 109. [Google Scholar] [CrossRef] [PubMed]
- Treanor, J.J.; Hayden, F.G.; Vrooman, P.S.; Barbarash, R.; Bettis, R.; Riff, D.; Singh, S.; Kinnersley, N.; Ward, P.; Mills, R.G. Efficacy and safety of the oral neuraminidase inhibitor oseltamivir in treating acute influenza: A randomized controlled trial. US Oral Neuraminidase Study Group. JAMA 2000, 283, 1016–1024. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.J.; Haire, L.F.; Stevens, D.J.; Collins, P.J.; Lin, Y.P.; Blackburn, G.M.; Hay, A.J.; Gamblin, S.J.; Skehel, J.J. The structure of H5N1 avian influenza neuraminidase suggests new opportunities for drug design. Nature 2006, 443, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Todd, B.; Tchesnokov, E.P.; Götte, M. The active form of the influenza cap-snatching endonuclease inhibitor baloxavir marboxil is a tight binding inhibitor. J. Biol. Chem. 2021, 296, 100486. [Google Scholar] [CrossRef]
- Omoto, S.; Speranzini, V.; Hashimoto, T.; Noshi, T.; Yamaguchi, H.; Kawai, M.; Kawaguchi, K.; Uehara, T.; Shishido, T.; Naito, A.; et al. Characterization of influenza virus variants induced by treatment with the endonuclease inhibitor baloxavir marboxil. Sci. Rep. 2018, 8, 9633. [Google Scholar] [CrossRef]
- Uehara, T.; Hayden, F.G.; Kawaguchi, K.; Omoto, S.; Hurt, A.C.; de Jong, M.D.; Hirotsu, N.; Sugaya, N.; Lee, N.; Baba, K.; et al. Treatment-Emergent Influenza Variant Viruses With Reduced Baloxavir Susceptibility: Impact on Clinical and Virologic Outcomes in Uncomplicated Influenza. J. Infect. Dis. 2020, 221, 346–355. [Google Scholar] [CrossRef]
- Furuta, Y.; Gowen, B.B.; Takahashi, K.; Shiraki, K.; Smee, D.F.; Barnard, D.L. Favipiravir (T-705), a novel viral RNA polymerase inhibitor. Antivir. Res. 2013, 100, 446–454. [Google Scholar] [CrossRef]
- Hayden, F.G.; Lenk, R.P.; Stonis, L.; Oldham-Creamer, C.; Kang, L.L.; Epstein, C. Favipiravir Treatment of Uncomplicated Influenza in Adults: Results of Two Phase 3, Randomized, Double-Blind, Placebo-Controlled Trials. J. Infect. Dis. 2022. [Google Scholar] [CrossRef] [PubMed]
- Madelain, V.; Mentré, F.; Baize, S.; Anglaret, X.; Laouénan, C.; Oestereich, L.; Nguyen, T.H.T.; Malvy, D.; Piorkowski, G.; Graw, F.; et al. Modeling Favipiravir Antiviral Efficacy Against Emerging Viruses: From Animal Studies to Clinical Trials. CPT Pharmacomet. Syst. Pharmacol. 2020, 9, 258–271. [Google Scholar] [CrossRef] [PubMed]
- Tırmıkçıoğlu, Z. Favipiravir exposure and pregnancy outcome of COVID-19 patients. Eur. J. Obstet. Gynecol. Reprod. Biol. 2022, 268, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Ueda, M.; Tanimoto, T.; Murayama, A.; Ozaki, A.; Kami, M. Japan’s Drug Regulation During the COVID-19 Pandemic: Lessons From a Case Study of Favipiravir. Clin. Pharmacol. Ther. 2022, 111, 545–547. [Google Scholar] [CrossRef]
- Gatherer, D. The 2014 Ebola virus disease outbreak in West Africa. J. Gen. Virol. 2014, 95, 1619–1624. [Google Scholar] [CrossRef]
- Oestereich, L.; Lüdtke, A.; Wurr, S.; Rieger, T.; Muñoz-Fontela, C.; Günther, S. Successful treatment of advanced Ebola virus infection with T-705 (favipiravir) in a small animal model. Antivir. Res. 2014, 105, 17–21. [Google Scholar] [CrossRef]
- Delang, L.; Abdelnabi, R.; Neyts, J. Favipiravir as a potential countermeasure against neglected and emerging RNA viruses. Antivir. Res. 2018, 153, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Sissoko, D.; Laouenan, C.; Folkesson, E.; M’Lebing, A.-B.; Beavogui, A.-H.; Baize, S.; Camara, A.-M.; Maes, P.; Shepherd, S.; Danel, C.; et al. Experimental Treatment with Favipiravir for Ebola Virus Disease (the JIKI Trial): A Historically Controlled, Single-Arm Proof-of-Concept Trial in Guinea. PLoS Med. 2016, 13, e1001967. [Google Scholar] [CrossRef]
- Kerber, R.; Lorenz, E.; Duraffour, S.; Sissoko, D.; Rudolf, M.; Jaeger, A.; Cisse, S.D.; Camara, A.-M.; Miranda, O.; Castro, C.M.; et al. Laboratory Findings, Compassionate Use of Favipiravir, and Outcome in Patients With Ebola Virus Disease, Guinea, 2015-A Retrospective Observational Study. J. Infect. Dis. 2019, 220, 195–202. [Google Scholar] [CrossRef]
- Cai, Q.; Yang, M.; Liu, D.; Chen, J.; Shu, D.; Xia, J.; Liao, X.; Gu, Y.; Cai, Q.; Yang, Y.; et al. Experimental Treatment with Favipiravir for COVID-19: An Open-Label Control Study. Engineering 2020, 6, 1192–1198. [Google Scholar] [CrossRef]
- Udwadia, Z.F.; Singh, P.; Barkate, H.; Patil, S.; Rangwala, S.; Pendse, A.; Kadam, J.; Wu, W.; Caracta, C.F.; Tandon, M. Efficacy and safety of favipiravir, an oral RNA-dependent RNA polymerase inhibitor, in mild-to-moderate COVID-19: A randomized, comparative, open-label, multicenter, phase 3 clinical trial. Int. J. Infect. Dis. 2021, 103, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Doi, Y.; Hibino, M.; Hase, R.; Yamamoto, M.; Kasamatsu, Y.; Hirose, M.; Mutoh, Y.; Homma, Y.; Terada, M.; Ogawa, T.; et al. A Prospective, Randomized, Open-Label Trial of Early versus Late Favipiravir Therapy in Hospitalized Patients with COVID-19. Antimicrob. Agents Chemother. 2020, 64, e01897-20. [Google Scholar] [CrossRef]
- Leneva, I.A.; Russell, R.J.; Boriskin, Y.S.; Hay, A.J. Characteristics of arbidol-resistant mutants of influenza virus: Implications for the mechanism of anti-influenza action of arbidol. Antivir. Res. 2009, 81, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Haviernik, J.; Štefánik, M.; Fojtíková, M.; Kali, S.; Tordo, N.; Rudolf, I.; Hubálek, Z.; Eyer, L.; Ruzek, D. Arbidol (Umifenovir): A Broad-Spectrum Antiviral Drug That Inhibits Medically Important Arthropod-Borne Flaviviruses. Viruses 2018, 10, 184. [Google Scholar] [CrossRef] [PubMed]
- Dias, D.A.; Urban, S.; Roessner, U. A historical overview of natural products in drug discovery. Metabolites 2012, 2, 303–336. [Google Scholar] [CrossRef]
- Heinrich, M.; Gibbons, S. Ethnopharmacology in drug discovery: An analysis of its role and potential contribution. J. Pharm. Pharmacol. 2001, 53, 425–432. [Google Scholar] [CrossRef]
- Yi, M.; Lin, S.; Zhang, B.; Jin, H.; Ding, L. Antiviral potential of natural products from marine microbes. European J. Med. Chem. 2020, 207, 112790. [Google Scholar] [CrossRef]
- Langeder, J.; Grienke, U.; Chen, Y.; Kirchmair, J.; Schmidtke, M.; Rollinger, J.M. Natural products against acute respiratory infections: Strategies and lessons learned. J. Ethnopharmacol. 2020, 248, 112298. [Google Scholar] [CrossRef]
- Grienke, U.; Schmidtke, M.; von Grafenstein, S.; Kirchmair, J.; Liedl, K.R.; Rollinger, J.M. Influenza neuraminidase: A druggable target for natural products. Nat. Prod. Rep. 2012, 29, 11–36. [Google Scholar] [CrossRef]
- Zhang, Z.-J.; Morris-Natschke, S.L.; Cheng, Y.-Y.; Lee, K.-H.; Li, R.-T. Development of anti-influenza agents from natural products. Med. Res. Rev. 2020, 40, 2290–2338. [Google Scholar] [CrossRef]
- Wang, X.; Jia, W.; Zhao, A.; Wang, X. Anti-influenza agents from plants and traditional Chinese medicine. Phytother. Res. 2006, 20, 335–341. [Google Scholar] [CrossRef]
- Mousa, H.A.-L. Prevention and Treatment of Influenza, Influenza-Like Illness, and Common Cold by Herbal, Complementary, and Natural Therapies. J. Evid. Based Complement. Altern. Med. 2017, 22, 166–174. [Google Scholar] [CrossRef]
- Ge, H.; Wang, Y.-F.; Xu, J.; Gu, Q.; Liu, H.-B.; Xiao, P.-G.; Zhou, J.; Liu, Y.; Yang, Z.; Su, H. Anti-influenza agents from Traditional Chinese Medicine. Nat. Prod. Rep. 2010, 27, 1758–1780. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; Supuran, C.T. Natural products in drug discovery: Advances and opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Kitazato, K.; Wang, Y.; Kobayashi, N. Viral infectious disease and natural products with antiviral activity. Drug Discov. Ther. 2007, 1, 14–22. [Google Scholar]
- Sokmen, M.; Angelova, M.; Krumova, E.; Pashova, S.; Ivancheva, S.; Sokmen, A.; Serkedjieva, J. In vitro antioxidant activity of polyphenol extracts with antiviral properties from Geranium sanguineum L. Life Sci. 2005, 76, 2981–2993. [Google Scholar] [CrossRef]
- Miki, K.; Nagai, T.; Suzuki, K.; Tsujimura, R.; Koyama, K.; Kinoshita, K.; Furuhata, K.; Yamada, H.; Takahashi, K. Anti-influenza virus activity of biflavonoids. Bioorg. Med. Chem. Lett. 2007, 17, 772–775. [Google Scholar] [CrossRef]
- Kernan, M.R.; Sendl, A.; Chen, J.L.; Jolad, S.D.; Blanc, P.; Murphy, J.T.; Stoddart, C.A.; Nanakorn, W.; Balick, M.J.; Rozhon, E.J. Two new lignans with activity against influenza virus from the medicinal plant Rhinacanthus nasutus. J. Nat. Prod. 1997, 60, 635–637. [Google Scholar] [CrossRef]
- Pennington, M.W.; Czerwinski, A.; Norton, R.S. Peptide therapeutics from venom: Current status and potential. Bioorganic Med. Chem. 2018, 26, 2738–2758. [Google Scholar] [CrossRef]
- Plumlee, K.H. Clinical Veterinary Toxicology; Elsevier: Amsterdam, The Netherlands, 2004; ISBN 9780323011259. [Google Scholar]
- Zhang, Y. Why do we study animal toxins? Dongwuxue Yanjiu 2015, 36, 183–222. [Google Scholar] [CrossRef]
- Kachel, H.S.; Buckingham, S.D.; Sattelle, D.B. Insect toxins—Selective pharmacological tools and drug/chemical leads. Curr. Opin. Insect Sci. 2018, 30, 93–98. [Google Scholar] [CrossRef]
- da Mata, É.C.G.; Mourão, C.B.F.; Rangel, M.; Schwartz, E.F. Antiviral activity of animal venom peptides and related compounds. J. Venom. Anim. Toxins Incl. Trop. Dis. 2017, 23, 3. [Google Scholar] [CrossRef]
- Chernysh, S.; Kim, S.I.; Bekker, G.; Pleskach, V.A.; Filatova, N.A.; Anikin, V.B.; Platonov, V.G.; Bulet, P. Antiviral and antitumor peptides from insects. Proc. Natl. Acad. Sci. USA 2002, 99, 12628–12632. [Google Scholar] [CrossRef]
- Dai, C.; Ma, Y.; Zhao, Z.; Zhao, R.; Wang, Q.; Wu, Y.; Cao, Z.; Li, W. Mucroporin, the first cationic host defense peptide from the venom of Lychas mucronatus. Antimicrob. Agents Chemother. 2008, 52, 3967–3972. [Google Scholar] [CrossRef]
- Li, Q.; Zhao, Z.; Zhou, D.; Chen, Y.; Hong, W.; Cao, L.; Yang, J.; Zhang, Y.; Shi, W.; Cao, Z.; et al. Virucidal activity of a scorpion venom peptide variant mucroporin-M1 against measles, SARS-CoV and influenza H5N1 viruses. Peptides 2011, 32, 1518–1525. [Google Scholar] [CrossRef]
- Uddin, M.B.; Lee, B.-H.; Nikapitiya, C.; Kim, J.-H.; Kim, T.-H.; Lee, H.-C.; Kim, C.G.; Lee, J.-S.; Kim, C.-J. Inhibitory effects of bee venom and its components against viruses in vitro and in vivo. J. Microbiol. 2016, 54, 853–866. [Google Scholar] [CrossRef]
- Holthausen, D.J.; Lee, S.H.; Kumar, V.T.; Bouvier, N.M.; Krammer, F.; Ellebedy, A.H.; Wrammert, J.; Lowen, A.C.; George, S.; Pillai, M.R.; et al. An Amphibian Host Defense Peptide Is Virucidal for Human H1 Hemagglutinin-Bearing Influenza Viruses. Immunity 2017, 46, 587–595. [Google Scholar] [CrossRef]
- Xu, J.; Chen, S.; Jin, J.; Ma, L.; Guo, M.; Zhou, C.; Dou, J. Inhibition of peptide BF-30 on influenza A virus infection in vitro/vivo by causing virion membrane fusion. Peptides 2019, 112, 14–22. [Google Scholar] [CrossRef]
- Lüddecke, T.; Vilcinskas, A.; Lemke, S. Phylogeny-Guided Selection of Priority Groups for Venom Bioprospecting: Harvesting Toxin Sequences in Tarantulas as a Case Study. Toxins 2019, 11, 488. [Google Scholar] [CrossRef]
- von Reumont, B.M.; Campbell, L.I.; Jenner, R.A. Quo vadis venomics? A roadmap to neglected venomous invertebrates. Toxins 2014, 6, 3488–3551. [Google Scholar] [CrossRef] [PubMed]
- von Reumont, B.M.; Anderluh, G.; Antunes, A.; Ayvazyan, N.; Beis, D.; Caliskan, F.; Crnković, A.; Damm, M.; Dutertre, S.; Ellgaard, L.; et al. Modern venomics-Current insights, novel methods, and future perspectives in biological and applied animal venom research. Gigascience 2022, 11, giac048. [Google Scholar] [CrossRef] [PubMed]
- Watkinson, S.C.; Boddy, L.; Money, N. The Fungi, 3rd ed.; Academic Press: Amsterdam, The Netherlands, 2016; ISBN 9780123820358. [Google Scholar]
- Jakubczyk, D.; Dussart, F. Selected Fungal Natural Products with Antimicrobial Properties. Molecules 2020, 25, 911. [Google Scholar] [CrossRef] [PubMed]
- Krupodorova, T.; Rybalko, S.; Barshteyn, V. Antiviral activity of Basidiomycete mycelia against influenza type A (serotype H1N1) and herpes simplex virus type 2 in cell culture. Virol. Sin. 2014, 29, 284–290. [Google Scholar] [CrossRef]
- Linnakoski, R.; Reshamwala, D.; Veteli, P.; Cortina-Escribano, M.; Vanhanen, H.; Marjomäki, V. Antiviral Agents From Fungi: Diversity, Mechanisms and Potential Applications. Front. Microbiol. 2018, 9, 2325. [Google Scholar] [CrossRef]
- Liu, X.; Zhao, Z.; Li, Z.; Xu, C.; Sun, L.; Chen, J.; Liu, W. Cyclosporin A inhibits the influenza virus replication through cyclophilin A-dependent and -independent pathways. PLoS ONE 2012, 7, e37277. [Google Scholar] [CrossRef]
- Hamamoto, I.; Harazaki, K.; Inase, N.; Takaku, H.; Tashiro, M.; Yamamoto, N. Cyclosporin A inhibits the propagation of influenza virus by interfering with a late event in the virus life cycle. Jpn. J. Infect. Dis. 2013, 66, 276–283. [Google Scholar] [CrossRef][Green Version]
- Ma, C.; Li, F.; Musharrafieh, R.G.; Wang, J. Discovery of cyclosporine A and its analogs as broad-spectrum anti-influenza drugs with a high in vitro genetic barrier of drug resistance. Antivir. Res. 2016, 133, 62–72. [Google Scholar] [CrossRef]
- Minagawa, K.; Kouzuki, S.; Yoshimoto, J.; Kawamura, Y.; Tani, H.; Iwata, T.; Terui, Y.; Nakai, H.; Yagi, S.; Hattori, N.; et al. Stachyflin and acetylstachyflin, novel anti-influenza A virus substances, produced by Stachybotrys sp. RF-7260. I. Isolation, structure elucidation and biological activities. J. Antibiot. 2002, 55, 155–164. [Google Scholar] [CrossRef]
- Yoshimoto, J.; Kakui, M.; Iwasaki, H.; Fujiwara, T.; Sugimoto, H.; Hattori, N. Identification of a novel HA conformational change inhibitor of human influenza virus. Arch. Virol. 1999, 144, 865–878. [Google Scholar] [CrossRef]
- Sacramento, C.Q.; Marttorelli, A.; Fintelman-Rodrigues, N.; de Freitas, C.S.; de Melo, G.R.; Rocha, M.E.N.; Kaiser, C.R.; Rodrigues, K.F.; Da Costa, G.L.; Alves, C.M.; et al. Aureonitol, a Fungi-Derived Tetrahydrofuran, Inhibits Influenza Replication by Targeting Its Surface Glycoprotein Hemagglutinin. PLoS ONE 2015, 10, e0139236. [Google Scholar] [CrossRef]
- Awadh Ali, N.A.; Mothana, R.; Lesnau, A.; Pilgrim, H.; Lindequist, U. Antiviral activity of Inonotus hispidus. Fitoterapia 2003, 74, 483–485. [Google Scholar] [CrossRef]
- Piraino, F.; Brandt, C.R. Isolation and partial characterization of an antiviral, RC-183, from the edible mushroom Rozites caperata. Antivir. Res. 1999, 43, 67–78. [Google Scholar] [CrossRef]
- Stancu, C.; Sima, A. Statins: Mechanism of action and effects. J. Cell. Mol. Med. 2001, 5, 378–387. [Google Scholar] [CrossRef]
- Zeiser, R. Immune modulatory effects of statins. Immunology 2018, 154, 69–75. [Google Scholar] [CrossRef]
- Frost, F.J.; Petersen, H.; Tollestrup, K.; Skipper, B. Influenza and COPD mortality protection as pleiotropic, dose-dependent effects of statins. Chest 2007, 131, 1006–1012. [Google Scholar] [CrossRef]
- Vandermeer, M.L.; Thomas, A.R.; Kamimoto, L.; Reingold, A.; Gershman, K.; Meek, J.; Farley, M.M.; Ryan, P.; Lynfield, R.; Baumbach, J.; et al. Association between use of statins and mortality among patients hospitalized with laboratory-confirmed influenza virus infections: A multistate study. J. Infect. Dis. 2012, 205, 13–19. [Google Scholar] [CrossRef]
- Carrillo-Esper, R.; Sosa-García, J.O.; Arch-Tirado, E. Experience in the management of the severe form of human influenza A H1N1 pneumonia in an intensive care unit. Cir. Cir. 2011, 79, 409–416. [Google Scholar]
- Kwong, J.C.; Li, P.; Redelmeier, D.A. Influenza morbidity and mortality in elderly patients receiving statins: A cohort study. PLoS ONE 2009, 4, e8087. [Google Scholar] [CrossRef]
- Laidler, M.R.; Thomas, A.; Baumbach, J.; Kirley, P.D.; Meek, J.; Aragon, D.; Morin, C.; Ryan, P.A.; Schaffner, W.; Zansky, S.M.; et al. Statin treatment and mortality: Propensity score-matched analyses of 2007-2008 and 2009-2010 laboratory-confirmed influenza hospitalizations. Open Forum Infect. Dis. 2015, 2, ofv028. [Google Scholar] [CrossRef]
- Peng, J.; Zhang, D.; Ma, Y.; Wang, G.; Guo, Z.; Lu, J. Protective effect of fluvastatin on influenza virus infection. Mol. Med. Rep. 2014, 9, 2221–2226. [Google Scholar] [CrossRef]
- Belser, J.A.; Szretter, K.J.; Katz, J.M.; Tumpey, T.M. Simvastatin and oseltamivir combination therapy does not improve the effectiveness of oseltamivir alone following highly pathogenic avian H5N1 influenza virus infection in mice. Virology 2013, 439, 42–46. [Google Scholar] [CrossRef]
- Radigan, K.A.; Urich, D.; Misharin, A.V.; Chiarella, S.E.; Soberanes, S.; Gonzalez, A.; Perlman, H.; Wunderink, R.G.; Budinger, G.R.S.; Mutlu, G.M. The effect of rosuvastatin in a murine model of influenza A infection. PLoS ONE 2012, 7, e35788. [Google Scholar] [CrossRef]
- Wang, H.; Wang, Y.; Wang, W.; Fu, P.; Liu, P.; Zhu, W. Anti-influenza virus polyketides from the acid-tolerant fungus Penicillium purpurogenum JS03-21. J. Nat. Prod. 2011, 74, 2014–2018. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Zhang, X.; Du, L.; Wang, W.; Zhu, T.; Gu, Q.; Li, D. Sorbicatechols A and B, antiviral sorbicillinoids from the marine-derived fungus Penicillium chrysogenum PJX-17. J. Nat. Prod. 2014, 77, 424–428. [Google Scholar] [CrossRef]
- Yamamoto, T.; Izumi, N.; Ui, H.; Sueki, A.; Masuma, R.; Nonaka, K.; Hirose, T.; Sunazuka, T.; Nagai, T.; Yamada, H.; et al. Wickerols A and B: Novel anti-influenza virus diterpenes produced by Trichoderma atroviride FKI-3849. Tetrahedron 2012, 68, 9267–9271. [Google Scholar] [CrossRef]
- Chiba, T.; Asami, Y.; Suga, T.; Watanabe, Y.; Nagai, T.; Momose, F.; Nonaka, K.; Iwatsuki, M.; Yamada, H.; Ōmura, S.; et al. Herquline A, produced by Penicillium herquei FKI-7215, exhibits anti-influenza virus properties. Biosci. Biotechnol. Biochem. 2017, 81, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Liu, D.; Proksch, P.; Zhou, D.; Lin, W. Truncateols O-V, further isoprenylated cyclohexanols from the sponge-associated fungus Truncatella angustata with antiviral activities. Phytochemistry 2018, 155, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.-X.; Yang, J.-L.; Qi, Q.-Y.; Bao, L.; Yang, X.-L.; Liu, M.-M.; Huang, P.; Zhang, L.-X.; Chen, J.-L.; Cai, L.; et al. 3-Anhydro-6-hydroxy-ophiobolin A, a new sesterterpene inhibiting the growth of methicillin-resistant Staphylococcus aureus and inducing the cell death by apoptosis on K562, from the phytopathogenic fungus Bipolaris oryzae. Bioorg. Med. Chem. Lett. 2013, 23, 3547–3550. [Google Scholar] [CrossRef]
- Wang, S.; Luo, X.; Yan, R.; Wang, Q.; Qi, Q.; Chi, X.; Zhang, L.; Yu, Z.; Cai, B.; Chen, J.-L.; et al. 3-Anhydro-6-hydroxy-ophiobolin A displays high in vitro and in vivo efficacy against influenza A virus infection. Protein Cell 2016, 7, 839–843. [Google Scholar] [CrossRef]
- Bentley, R. Mycophenolic Acid: A one hundred year odyssey from antibiotic to immunosuppressant. Chem. Rev. 2000, 100, 3801–3826. [Google Scholar] [CrossRef] [PubMed]
- Regueira, T.B.; Kildegaard, K.R.; Hansen, B.G.; Mortensen, U.H.; Hertweck, C.; Nielsen, J. Molecular Basis for Mycophenolic Acid Biosynthesis in Penicillium brevicompactum. Appl. Environ. Microbiol. 2011, 77, 3035–3043. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.F.W.; Chan, K.-H.; Kao, R.Y.T.; To, K.K.W.; Zheng, B.-J.; Li, C.P.Y.; Li, P.T.W.; Dai, J.; Mok, F.K.Y.; Chen, H.; et al. Broad-spectrum antivirals for the emerging Middle East respiratory syndrome coronavirus. J. Infect. 2013, 67, 606–616. [Google Scholar] [CrossRef] [PubMed]
- To, K.K.W.; Mok, K.-Y.; Chan, A.S.F.; Cheung, N.N.; Wang, P.; Lui, Y.-M.; Chan, J.F.W.; Chen, H.; Chan, K.-H.; Kao, R.Y.T.; et al. Mycophenolic acid, an immunomodulator, has potent and broad-spectrum in vitro antiviral activity against pandemic, seasonal and avian influenza viruses affecting humans. J. Gen. Virol. 2016, 97, 1807–1817. [Google Scholar] [CrossRef]
- Miyake, Y.; Kozutsumi, Y.; Nakamura, S.; Fujita, T.; Kawasaki, T. Serine palmitoyltransferase is the primary target of a sphingosine-like immunosuppressant, ISP-1/myriocin. Biochem. Biophys. Res. Commun. 1995, 211, 396–403. [Google Scholar] [CrossRef]
- Tafesse, F.G.; Sanyal, S.; Ashour, J.; Guimaraes, C.P.; Hermansson, M.; Somerharju, P.; Ploegh, H.L. Intact sphingomyelin biosynthetic pathway is essential for intracellular transport of influenza virus glycoproteins. Proc. Natl. Acad. Sci. USA 2013, 110, 6406–6411. [Google Scholar] [CrossRef]
- Hidari, K.I.P.J.; Suzuki, Y.; Suzuki, T. Suppression of the biosynthesis of cellular sphingolipids results in the inhibition of the maturation of influenza virus particles in MDCK cells. Biol. Pharm. Bull. 2006, 29, 1575–1579. [Google Scholar] [CrossRef]
- Gladfelter, A.S.; James, T.Y.; Amend, A.S. Marine fungi. Curr. Biol. 2019, 29, R191–R195. [Google Scholar] [CrossRef]
- Gogineni, V.; Schinazi, R.F.; Hamann, M.T. Role of Marine Natural Products in the Genesis of Antiviral Agents. Chem. Rev. 2015, 115, 9655–9706. [Google Scholar] [CrossRef]
- Peng, J.; Lin, T.; Wang, W.; Xin, Z.; Zhu, T.; Gu, Q.; Li, D. Antiviral alkaloids produced by the mangrove-derived fungus Cladosporium sp. PJX-41. J. Nat. Prod. 2013, 76, 1133–1140. [Google Scholar] [CrossRef]
- Wu, G.; Sun, X.; Yu, G.; Wang, W.; Zhu, T.; Gu, Q.; Li, D. Cladosins A-E, hybrid polyketides from a deep-sea-derived fungus, Cladosporium sphaerospermum. J. Nat. Prod. 2014, 77, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Wang, Y.; Liu, P.; Fu, P.; Zhu, T.; Wang, W.; Zhu, W. Indole-diterpenoids with anti-H1N1 activity from the aciduric fungus Penicillium camemberti OUCMDZ-1492. J. Nat. Prod. 2013, 76, 1328–1336. [Google Scholar] [CrossRef]
- Wang, J.-F.; Lin, X.-P.; Qin, C.; Liao, S.-R.; Wan, J.-T.; Zhang, T.-Y.; Liu, J.; Fredimoses, M.; Chen, H.; Yang, B.; et al. Antimicrobial and antiviral sesquiterpenoids from sponge-associated fungus, Aspergillus sydowii ZSDS1-F6. J. Antibiot. 2014, 67, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.; Chen, Z.; Liu, P.; Wang, Y.; Xin, Z.; Zhu, W. New rubrolides from the marine-derived fungus Aspergillus terreus OUCMDZ-1925. J. Antibiot. 2014, 67, 315–318. [Google Scholar] [CrossRef] [PubMed]
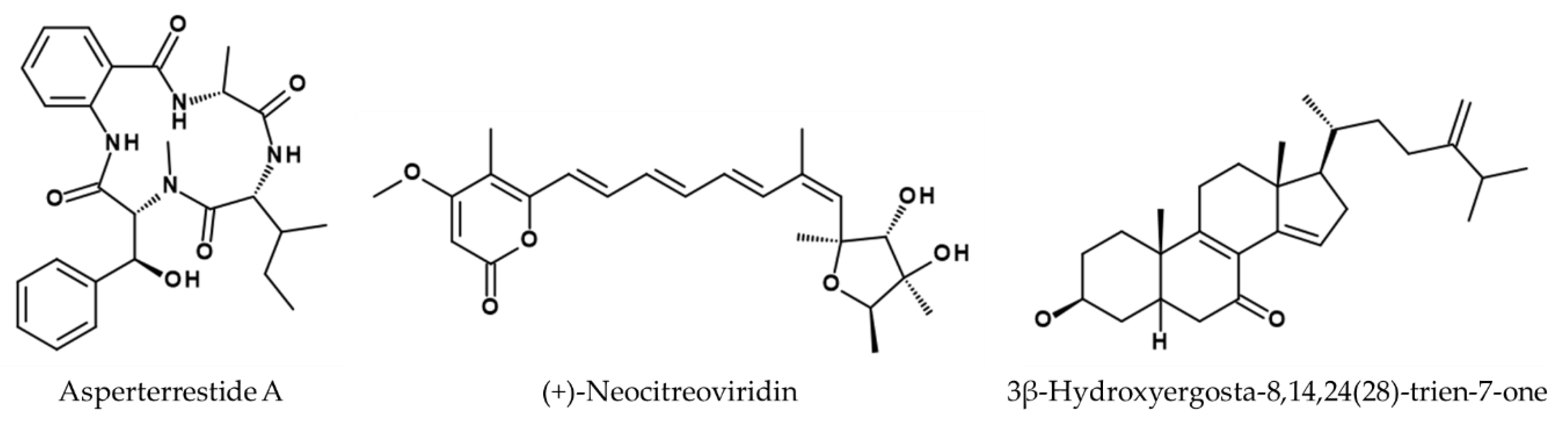
- He, F.; Bao, J.; Zhang, X.-Y.; Tu, Z.-C.; Shi, Y.-M.; Qi, S.-H. Asperterrestide A, a cytotoxic cyclic tetrapeptide from the marine-derived fungus Aspergillus terreus SCSGAF0162. J. Nat. Prod. 2013, 76, 1182–1186. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Guo, W.; Wang, Q.; Zhang, L.; Zhu, M.; Zhu, T.; Gu, Q.; Wang, W.; Li, D. Aspulvinones from a mangrove rhizosphere soil-derived fungus Aspergillus terreus Gwq-48 with anti-influenza A viral (H1N1) activity. Bioorg. Med. Chem. Lett. 2013, 23, 1776–1778. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, Y.; Hao, X.; Li, S.; Jia, J.; Guan, Y.; Peng, Z.; Bi, H.; Xiao, C.; Cen, S.; et al. Broad-Spectrum Antiviral Natural Products from the Marine-Derived Penicillium sp. IMB17-046. Molecules 2019, 24, 2821. [Google Scholar] [CrossRef]
- Zhang, G.; Sun, S.; Zhu, T.; Lin, Z.; Gu, J.; Li, D.; Gu, Q. Antiviral isoindolone derivatives from an endophytic fungus Emericella sp. associated with Aegiceras corniculatum. Phytochemistry 2011, 72, 1436–1442. [Google Scholar] [CrossRef]
- Wang, J.; Chen, F.; Liu, Y.; Liu, Y.; Li, K.; Yang, X.; Liu, S.; Zhou, X.; Yang, J. Spirostaphylotrichin X from a Marine-Derived Fungus as an Anti-influenza Agent Targeting RNA Polymerase PB2. J. Nat. Prod. 2018, 81, 2722–2730. [Google Scholar] [CrossRef]
- Luo, X.; Yang, J.; Chen, F.; Lin, X.; Chen, C.; Zhou, X.; Liu, S.; Liu, Y. Structurally Diverse Polyketides From the Mangrove-Derived Fungus Diaporthe sp. SCSIO 41011 With Their Anti-influenza A Virus Activities. Front. Chem. 2018, 6, 282. [Google Scholar] [CrossRef]
- Chen, X.; Si, L.; Liu, D.; Proksch, P.; Zhang, L.; Zhou, D.; Lin, W. Neoechinulin B and its analogues as potential entry inhibitors of influenza viruses, targeting viral hemagglutinin. Eur. J. Med. Chem. 2015, 93, 182–195. [Google Scholar] [CrossRef] [PubMed]
- Bovio, E.; Garzoli, L.; Poli, A.; Luganini, A.; Villa, P.; Musumeci, R.; McCormack, G.P.; Cocuzza, C.E.; Gribaudo, G.; Mehiri, M.; et al. Marine Fungi from the Sponge Grantia compressa: Biodiversity, Chemodiversity, and Biotechnological Potential. Mar. Drugs 2019, 17, 220. [Google Scholar] [CrossRef] [PubMed]
- Niu, S.; Si, L.; Liu, D.; Zhou, A.; Zhang, Z.; Shao, Z.; Wang, S.; Zhang, L.; Zhou, D.; Lin, W. Spiromastilactones: A new class of influenza virus inhibitors from deep-sea fungus. Eur. J. Med. Chem. 2016, 108, 229–244. [Google Scholar] [CrossRef] [PubMed]
- Bandoro, C.; Runstadler, J.A. Bacterial Lipopolysaccharide Destabilizes Influenza Viruses. mSphere 2017, 2, e00267-17. [Google Scholar] [CrossRef]
- Niedermeyer, T.H.J. Anti-infective Natural Products from Cyanobacteria. Planta Med. 2015, 81, 1309–1325. [Google Scholar] [CrossRef]
- Nowotny, A.; Mentel, R.; Wegner, U.; Mundt, S.; Lindequist, U. Antiviral Activity of an Aqueous Extract of the Cyanobacterium Microcystis aeruginosa. Phytother. Res. 1997, 11, 93–96. [Google Scholar] [CrossRef]
- Zainuddin, E.N.; Mentel, R.; Wray, V.; Jansen, R.; Nimtz, M.; Lalk, M.; Mundt, S. Cyclic depsipeptides, ichthyopeptins A and B, from Microcystis ichthyoblabe. J. Nat. Prod. 2007, 70, 1084–1088. [Google Scholar] [CrossRef]
- O’Keefe, B.R.; Smee, D.F.; Turpin, J.A.; Saucedo, C.J.; Gustafson, K.R.; Mori, T.; Blakeslee, D.; Buckheit, R.; Boyd, M.R. Potent anti-influenza activity of cyanovirin-N and interactions with viral hemagglutinin. Antimicrob. Agents Chemother. 2003, 47, 2518–2525. [Google Scholar] [CrossRef]
- Smee, D.F.; Bailey, K.W.; Wong, M.-H.; O’Keefe, B.R.; Gustafson, K.R.; Mishin, V.P.; Gubareva, L.V. Treatment of influenza A (H1N1) virus infections in mice and ferrets with cyanovirin-N. Antivir. Res. 2008, 80, 266–271. [Google Scholar] [CrossRef]
- Smee, D.F.; Wandersee, M.K.; Checketts, M.B.; O’Keefe, B.R.; Saucedo, C.; Boyd, M.R.; Mishin, V.P.; Gubareva, L.V. Influenza A (H1N1) virus resistance to cyanovirin-N arises naturally during adaptation to mice and by passage in cell culture in the presence of the inhibitor. Antivir. Chem. Chemother. 2007, 18, 317–327. [Google Scholar] [CrossRef]
- Kanekiyo, K.; Lee, J.-B.; Hayashi, K.; Takenaka, H.; Hayakawa, Y.; Endo, S.; Hayashi, T. Isolation of an antiviral polysaccharide, nostoflan, from a terrestrial cyanobacterium, Nostoc flagelliforme. J. Nat. Prod. 2005, 68, 1037–1041. [Google Scholar] [CrossRef]
- Morimoto, K.; Sato, Y. Anti-influenza virus activity of high-mannose binding lectins derived from genus Pseudomonas. Virus Res. 2016, 223, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Nagai, T.; Makino, S.; Ikegami, S.; Itoh, H.; Yamada, H. Effects of oral administration of yogurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1 and its exopolysaccharides against influenza virus infection in mice. Int. Immunopharmacol. 2011, 11, 2246–2250. [Google Scholar] [CrossRef] [PubMed]
- Al Kassaa, I.; Hober, D.; Hamze, M.; Chihib, N.E.; Drider, D. Antiviral potential of lactic acid bacteria and their bacteriocins. Probiotics Antimicrob. Proteins 2014, 6, 177–185. [Google Scholar] [CrossRef]
- Jung, Y.-J.; Lee, Y.-T.; Le Ngo, V.; Cho, Y.-H.; Ko, E.-J.; Hong, S.-M.; Kim, K.-H.; Jang, J.-H.; Oh, J.-S.; Park, M.-K.; et al. Heat-killed Lactobacillus casei confers broad protection against influenza A virus primary infection and develops heterosubtypic immunity against future secondary infection. Sci. Rep. 2017, 7, 17360. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Jiang, N.; Shi, W.; Chi, X.; Liu, S.; Chen, J.-L.; Wang, S. Development and Effects of Influenza Antiviral Drugs. Molecules 2021, 26, 810. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Chen, D.; Lu, S.; Yang, G.; Zhang, X.; Chen, Z.; Fan, S.; Wu, S.; He, J. Anti-Influenza A Viral Butenolide from Streptomyces sp. Smu03 Inhabiting the Intestine of Elephas maximus. Viruses 2018, 10, 356. [Google Scholar] [CrossRef]
- Berezin, V.; Abdukhakimova, D.; Trenozhnikova, L.; Bogoyavlenskiy, A.; Turmagambetova, A.; Issanov, A.; Azizan, A. Antiviral activities of extremophilic actinomycetes extracts from Kazakhstan’s unique ecosystems against influenza viruses and paramyxoviruses. Virol. J. 2019, 16, 150. [Google Scholar] [CrossRef]
- Yang, A.; Si, L.; Shi, Z.; Tian, L.; Liu, D.; Zhou, D.; Proksch, P.; Lin, W. Nitrosporeusines A and B, unprecedented thioester-bearing alkaloids from the Arctic Streptomyces nitrosporeus. Org. Lett. 2013, 15, 5366–5369. [Google Scholar] [CrossRef]
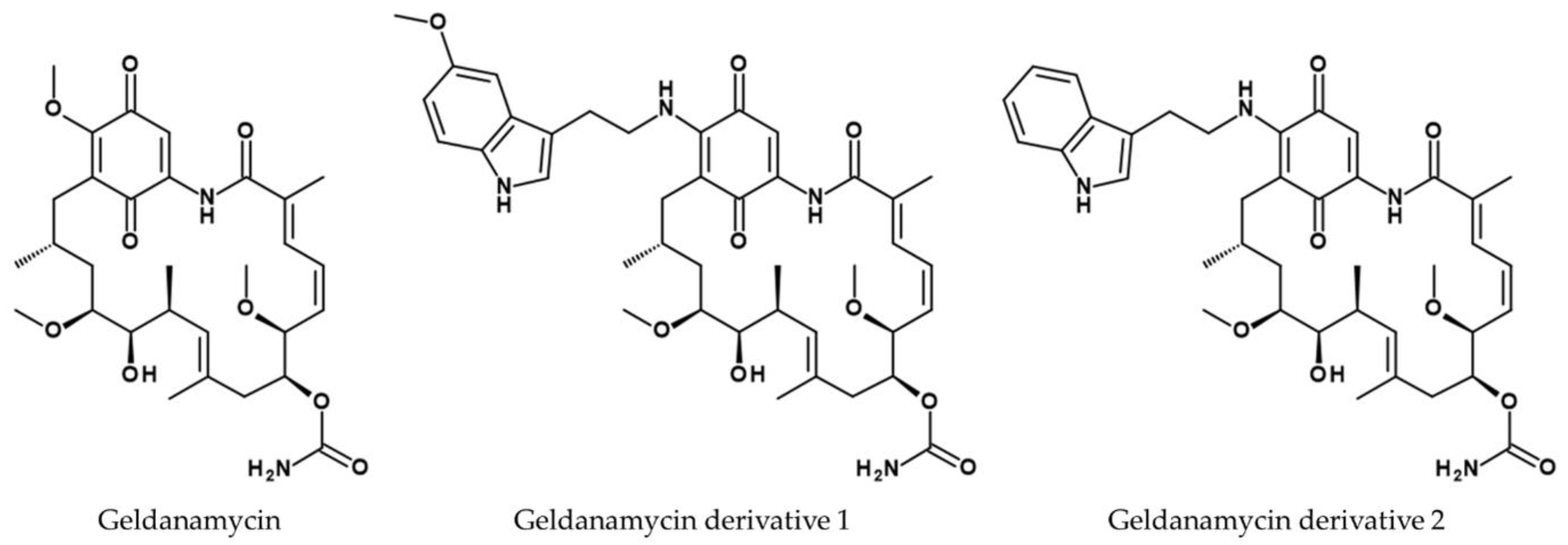
- Qin, H.-L.; Panek, J.S. Total synthesis of the Hsp90 inhibitor geldanamycin. Org. Lett. 2008, 10, 2477–2479. [Google Scholar] [CrossRef]
- DeBoer, C.; Meulman, P.A.; Wnuk, R.J.; Peterson, D.H. Geldanamycin, a new antibiotic. J. Antibiot. 1970, 23, 442–447. [Google Scholar] [CrossRef] [PubMed]
- Taechowisan, T.; Puckdee, W.; Phutdhawong, W.S. Streptomyces zerumbet, a Novel Species from Zingiber zerumbet (L.) Smith and Isolation of Its Bioactive Compounds. AiM 2019, 09, 194–219. [Google Scholar] [CrossRef]
- Taechowisan, T.; Samsawat, T.; Puckdee, W.; Phutdhawong, W.S. Antiviral activity of geldanamycin and its derivatives against influenza virus. J. App. Pharm. Sci. 2020, 10, 113–120. [Google Scholar] [CrossRef]
- Naruse, N.; Tenmyo, O.; Kawano, K.; Tomita, K.; Ohgusa, N.; Miyaki, T.; Konishi, M.; Oki, T. Fluvirucins A1, A2, B1, B2, B3, B4 and B5, new antibiotics active against influenza A virus. I. Production, isolation, chemical properties and biological activities. J. Antibiot. 1991, 44, 733–740. [Google Scholar] [CrossRef]
- Hatsu, M.; Sasaki, T.; Miyadoh, S.; Watabe, H.; Takeuchi, Y.; Kodama, Y.; Orikasa, Y.; Kajii, K.; Shomura, T.; Yamamoto, H. SF2487, a new polyether antibiotic produced by Actinomadura. J. Antibiot. 1990, 43, 259–266. [Google Scholar] [CrossRef]
- Uyeda, M. Fattiviracins, Antiviral Antibiotics Produced by an Actinomycete. Actinomycetologica 2004, 18, 62. [Google Scholar] [CrossRef]
- Habib, E.S.; Yokomizo, K.; Nagao, K.; Harada, S.; Uyeda, M. Antiviral activity of fattiviracin FV-8 against human immunodeficiency virus type 1 (HIV-1). Biosci. Biotechnol. Biochem. 2001, 65, 683–685. [Google Scholar] [CrossRef]
- Uyeda, M. Metabolites produced by actinomycetes--antiviral antibiotics and enzyme inhibitors. Yakugaku Zasshi 2004, 124, 469–479. [Google Scholar] [CrossRef]
- Wang, P.; Xi, L.; Liu, P.; Wang, Y.; Wang, W.; Huang, Y.; Zhu, W. Diketopiperazine derivatives from the marine-derived actinomycete Streptomyces sp. FXJ7.328. Mar. Drugs 2013, 11, 1035–1049. [Google Scholar] [CrossRef]
- Groupe, V.; Frankel, J.W.; Leche-Valier, M.P.; Waksman, S.A. Antiviral Properties of Ehrlichin, an Antibiotic Produced by Streptomyces Lavendulae. J. Immunol. 1951, 67, 471–482. [Google Scholar]
- Jordan, P.C.; Stevens, S.K.; Deval, J. Nucleosides for the treatment of respiratory RNA virus infections. Antivir. Chem. Chemother. 2018, 26, 2040206618764483. [Google Scholar] [CrossRef]
- Kataev, V.E.; Garifullin, B.F. Antiviral nucleoside analogs. Chem. Heterocycl. Comp. 2021, 57, 326–341. [Google Scholar] [CrossRef] [PubMed]
- Takizawa, N.; Yamasaki, M. Current landscape and future prospects of antiviral drugs derived from microbial products. J. Antibiot. 2017, 71, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Buchanan, J.G.; Hamblin, M.R.; Sood, G.R.; Wightman, R.H. The biosynthesis of pyrazofurin and formycin. J. Chem. Soc. Chem. Commun. 1980, 19, 917. [Google Scholar] [CrossRef]
- Kunimoto, T.; Wakashiro, T.; Okamura, I.; Asajima, T.; Hori, M. Structural requirements for formycin activity. J. Antibiot. 1968, 21, 468–470. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Takizawa, N.; Takada, H.; Umekita, M.; Igarashi, M.; Takahashi, Y. Anti-influenza Virus Activity of Methylthio-Formycin Distinct From That of T-705. Front. Microbiol. 2022, 13, 802671. [Google Scholar] [CrossRef]
- Malakhov, M.P.; Aschenbrenner, L.M.; Smee, D.F.; Wandersee, M.K.; Sidwell, R.W.; Gubareva, L.V.; Mishin, V.P.; Hayden, F.G.; Kim, D.H.; Ing, A.; et al. Sialidase fusion protein as a novel broad-spectrum inhibitor of influenza virus infection. Antimicrob. Agents Chemother. 2006, 50, 1470–1479. [Google Scholar] [CrossRef]
- Belser, J.A.; Lu, X.; Szretter, K.J.; Jin, X.; Aschenbrenner, L.M.; Lee, A.; Hawley, S.; Kim, D.H.; Malakhov, M.P.; Yu, M.; et al. DAS181, a novel sialidase fusion protein, protects mice from lethal avian influenza H5N1 virus infection. J. Infect. Dis. 2007, 196, 1493–1499. [Google Scholar] [CrossRef]
- Moss, R.B.; Hansen, C.; Sanders, R.L.; Hawley, S.; Li, T.; Steigbigel, R.T. A phase II study of DAS181, a novel host directed antiviral for the treatment of influenza infection. J. Infect. Dis. 2012, 206, 1844–1851. [Google Scholar] [CrossRef]
- Marjuki, H.; Mishin, V.P.; Chesnokov, A.P.; de La Cruz, J.A.; Fry, A.M.; Villanueva, J.; Gubareva, L.V. An investigational antiviral drug, DAS181, effectively inhibits replication of zoonotic influenza A virus subtype H7N9 and protects mice from lethality. J. Infect. Dis. 2014, 210, 435–440. [Google Scholar] [CrossRef]
- Salvatore, M.; Satlin, M.J.; Jacobs, S.E.; Jenkins, S.G.; Schuetz, A.N.; Moss, R.B.; van Besien, K.; Shore, T.; Soave, R. DAS181 for Treatment of Parainfluenza Virus Infections in Hematopoietic Stem Cell Transplant Recipients at a Single Center. Biol. Blood Marrow Transplant. 2016, 22, 965–970. [Google Scholar] [CrossRef]
- Nguyen, J.T.; Hoopes, J.D.; Le, M.H.; Smee, D.F.; Patick, A.K.; Faix, D.J.; Blair, P.J.; de Jong, M.D.; Prichard, M.N.; Went, G.T. Triple combination of amantadine, ribavirin, and oseltamivir is highly active and synergistic against drug resistant influenza virus strains in vitro. PLoS ONE 2010, 5, e9332. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, J.T.; Hoopes, J.D.; Smee, D.F.; Prichard, M.N.; Driebe, E.M.; Engelthaler, D.M.; Le, M.H.; Keim, P.S.; Spence, R.P.; Went, G.T. Triple combination of oseltamivir, amantadine, and ribavirin displays synergistic activity against multiple influenza virus strains in vitro. Antimicrob. Agents Chemother. 2009, 53, 4115–4126. [Google Scholar] [CrossRef] [PubMed]
- Hoopes, J.D.; Driebe, E.M.; Kelley, E.; Engelthaler, D.M.; Keim, P.S.; Perelson, A.S.; Rong, L.; Went, G.T.; Nguyen, J.T. Triple combination antiviral drug (TCAD) composed of amantadine, oseltamivir, and ribavirin impedes the selection of drug-resistant influenza A virus. PLoS ONE 2011, 6, e29778. [Google Scholar] [CrossRef] [PubMed]
- Govorkova, E.A.; Webster, R.G. Combination chemotherapy for influenza. Viruses 2010, 2, 1510–1529. [Google Scholar] [CrossRef]
- Park, S.; Kim, J.I.; Lee, I.; Lee, S.; Hwang, M.-W.; Bae, J.-Y.; Heo, J.; Kim, D.; Jang, S.-I.; Kim, H.; et al. Combination effects of peramivir and favipiravir against oseltamivir-resistant 2009 pandemic influenza A(H1N1) infection in mice. PLoS ONE 2014, 9, e101325. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eichberg, J.; Maiworm, E.; Oberpaul, M.; Czudai-Matwich, V.; Lüddecke, T.; Vilcinskas, A.; Hardes, K. Antiviral Potential of Natural Resources against Influenza Virus Infections. Viruses 2022, 14, 2452. https://doi.org/10.3390/v14112452
Eichberg J, Maiworm E, Oberpaul M, Czudai-Matwich V, Lüddecke T, Vilcinskas A, Hardes K. Antiviral Potential of Natural Resources against Influenza Virus Infections. Viruses. 2022; 14(11):2452. https://doi.org/10.3390/v14112452
Chicago/Turabian StyleEichberg, Johanna, Elena Maiworm, Markus Oberpaul, Volker Czudai-Matwich, Tim Lüddecke, Andreas Vilcinskas, and Kornelia Hardes. 2022. "Antiviral Potential of Natural Resources against Influenza Virus Infections" Viruses 14, no. 11: 2452. https://doi.org/10.3390/v14112452
APA StyleEichberg, J., Maiworm, E., Oberpaul, M., Czudai-Matwich, V., Lüddecke, T., Vilcinskas, A., & Hardes, K. (2022). Antiviral Potential of Natural Resources against Influenza Virus Infections. Viruses, 14(11), 2452. https://doi.org/10.3390/v14112452





