Evaluation of the Efficacy and Safety of Silver Nanoparticles in the Treatment of Non-Neurological and Neurological Distemper in Dogs: A Randomized Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Good Animal Handling Practices
2.2. Objective
2.3. Hypothesis
2.4. Study Design
2.5. Sample Size
2.6. Enrollment Criteria
2.7. Exclusion Criteria
2.8. Randomization
2.9. Blinding
2.10. Trial Medication
2.11. Population Analyzed
2.12. Experimental Animals
2.13. Experimental Procedures
2.14. Concomitant Treatments
2.15. Schedule of Events
2.16. Rescue Therapy
2.17. Premature Completion and Follow-Up
2.18. Monitoring of Adverse Effects
2.19. Efficacy Assessments
2.20. Safety Assessments
2.21. Indirect Immunofluorescence
2.22. Hemi-Nested RT-PCR Assay for CDV Detection
2.23. Statistical Analysis
3. Results
3.1. Study Population
3.2. Monitoring of Adverse Effects
3.3. Adverse Reactions after Administration of AgNPs
3.4. Rescue Therapy and Concomitant Treatments
3.5. Immune Response
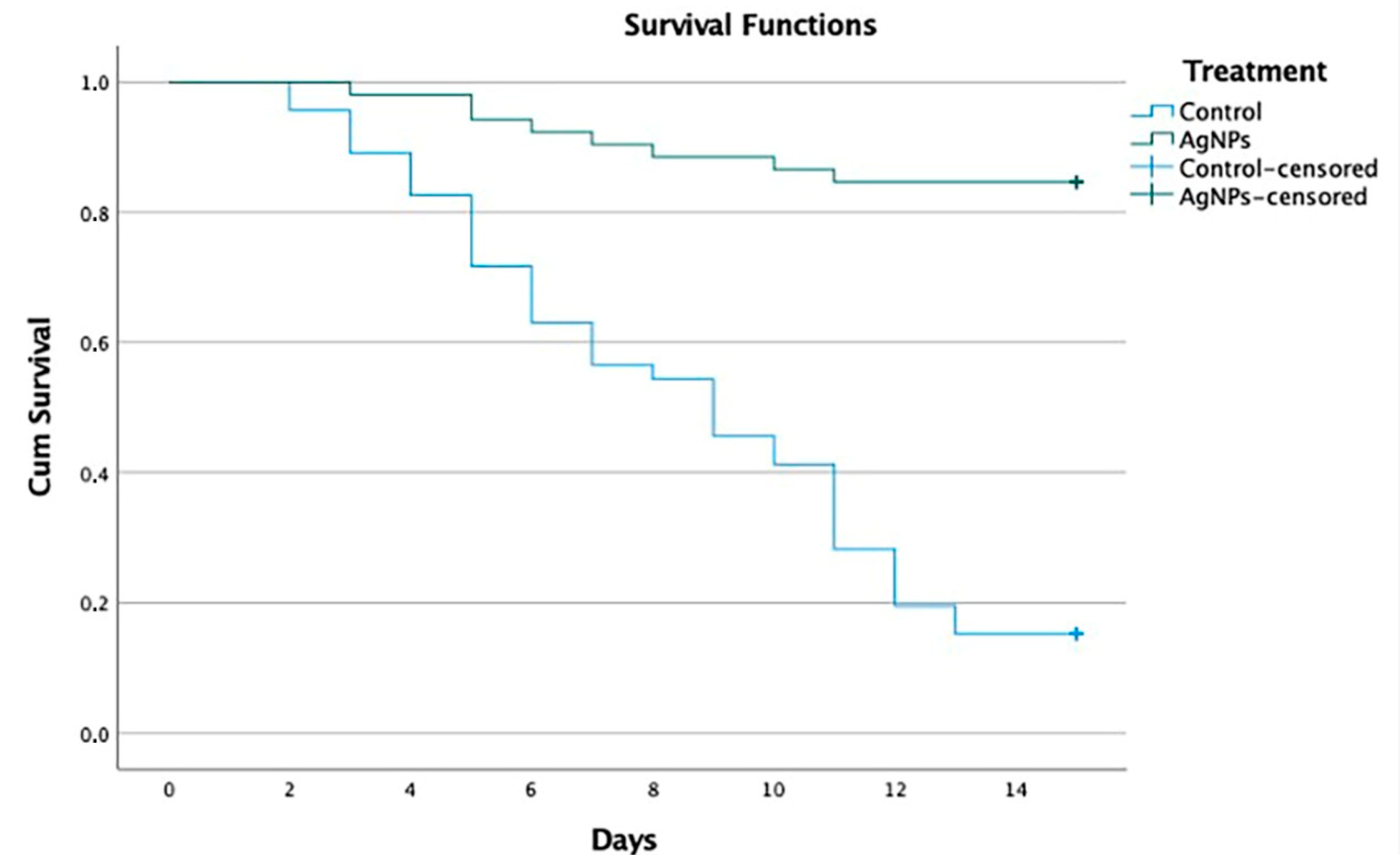
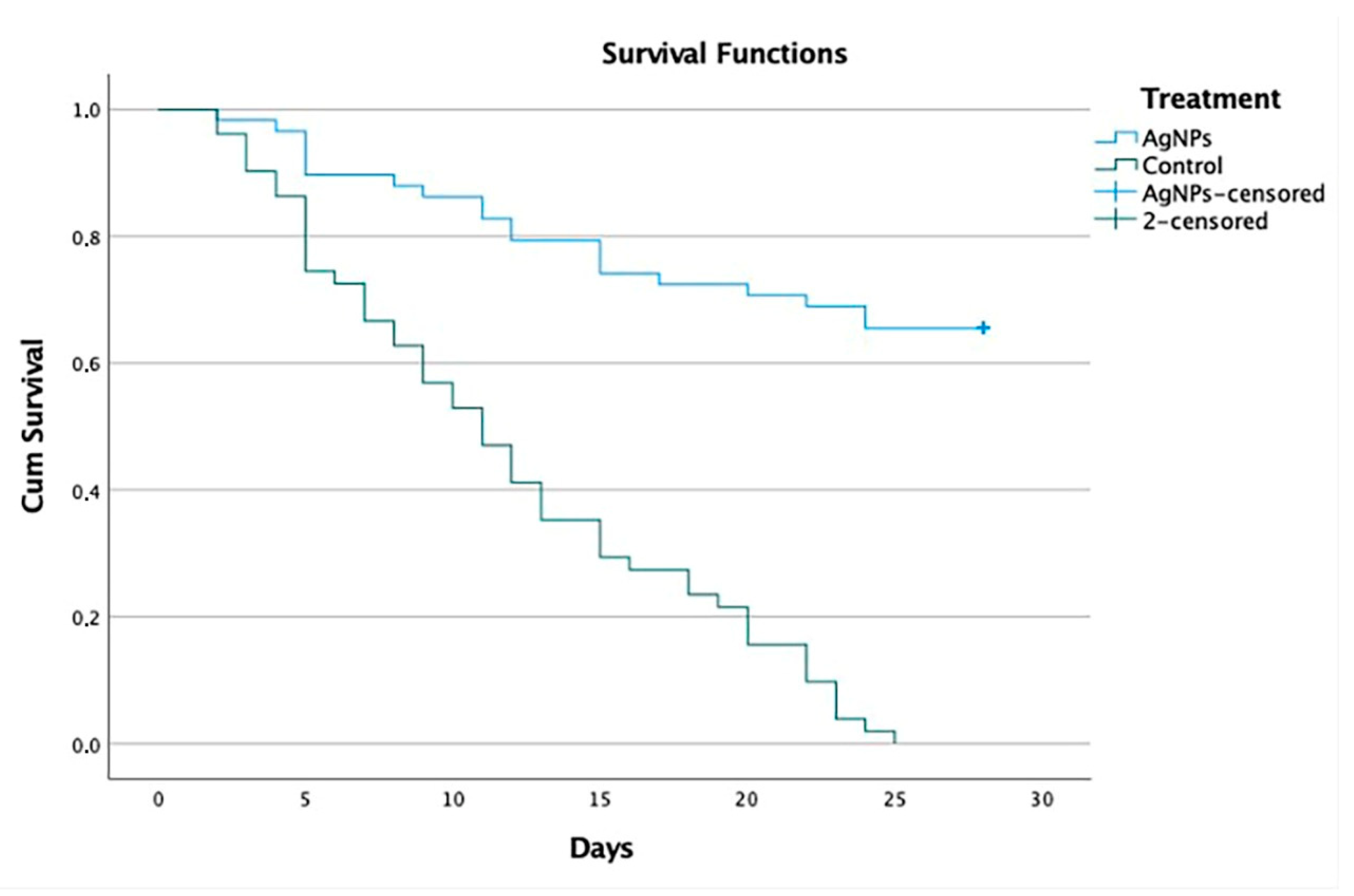
3.6. Survival
4. Discussion
4.1. Immunological Biomarkers
4.2. Treatment
- The previous work included three groups with a small number of 40 dogs [24], while the present work was designed as one of the largest prospective, randomized, double-blind, multicenter, four-arm clinical studies in veterinary medicine involving 207 naturally infected dogs.
- 2.
- The need for rescue therapy: the veterinarians were authorized to provide rescue therapy at any moment after the application of AgNPs, but none of the dogs within the protocol received rescue therapy (Table 1). Most of them only received supportive therapies, including the anesthetic drugs allowed in the protocol, mainly fluids and intravenous or oral antibiotics, and anticonvulsants such as phenobarbital, pregabalin, levetiracetam and diazepam, with no differences between groups (Table S1).
- 3.
- Premature withdrawal due to adverse effects: there were no significant differences in the frequency of reported adverse events in the two groups in which AgNPs were applied; this treatment was well-tolerated in both groups in this study. Although adverse effects were reported, most of them were mild or benign and classified as unrelated to the treatment. Only two dogs diagnosed with neurological distemper (Group 2a) were withdrawn from the study after two days of treatment with oral and nasal AgNPs, since they presented liquid diarrhea, vomiting and a temperature that reached 40.0 °C in both cases. Treatment was stopped and the dogs recovered on supportive therapy for 3 days and in 24 h they were again included in the AgNP treatment protocol. Apart from these two dogs, no other clinically significant adverse effects were observed following the administration of AgNPs, except for a slight increase in liver parameters over time in some dogs that never exceeded normal values. These slight changes were attributed to the combination of AgNPs and supportive therapy during treatment. The serum biochemical evaluation profile provides supporting evidence for the toxicity and safety of treatment with AgNPs. These findings indicate that the chronic administration of AgNPs in this population is safe.
- 4.
- Evaluate the disease progression after the assessment of clinical signs and sequelae presented by the dogs: the results referring to the clinical signs show that all animals presented with one or more systemic signs of disease caused by CDV. Most of these occurred before they presented neurological signs, after recovery from respiratory and gastrointestinal clinical signs. The neurological manifestations of CDV and its clinical expressions can vary due to the CNS being involved with the white matter as well as the gray matter [46]. A wide variety of neurological clinical signs (abnormal behavior, ataxia, paralysis, spasms, tics, prostration, paresis, cachexia, coma, myoclonus and convulsions) can be observed in the animal and a high percentage of patients are killed or euthanized due to poor disease prognosis [55]. In Group 1a, 44 of 52 (84.6%) dogs survived, 39 (75.0%) without sequelae and 5 (9.6%) with sequelae, whereas 7 of 46 (15.2%) dogs survived in Group 1b, 3 (6.5%) without sequelae and 4 (8.7%) with sequelae (Table S5, Figure 1). In Group 2a, 38 of 58 (65.5%) dogs survived, 28 (48.3%) without sequelae and 10 (17.2%) with sequelae, whereas 0 of 51 (0%) dogs survived in Group 2b (Table S5, Figure 2). In the experimental group 1a, five dogs were reported to survive with mild neurological sequelae, such as slight myoclonus or jaw tics, in addition to digestive, respiratory, and ophthalmological signs. In control group 1b, four dogs recovered with neurological sequelae, being mainly parietal, and jaw tics, myoclonus, prostration, and ataxia. In group 2a, 10 dogs presented signs of neurological lesions, such as myoclonus, ataxia and jaw and parietal tics, seizures, and claudication. In group 2b, where all dogs died, the neurological signs were parietal and jaw tics, myoclonus, seizures, paralysis of the swallowing reflex, spasm, prostration, coma, and ataxia. Regarding the onset of neurological signs, our results correspond to those reported by Koutinas et al. (2002); in most of our cases, these were gradual onset with a progressive chronic course, and in those animals that died were progressive and multifocal, the predominant neurological sign was the presence of myoclonus, both localized and generalized, in 13 of the 19 dogs [56]. Regarding the percentage of clinical signs and sequelae presented by the dogs with respect to age, our results coincided with a study carried out in Brazil, which included 70 dogs aged between 2 months and 13 years; in almost half (33; 47.1%), the dogs were up to one year of age, and in about a third (24; 34.3%) of the cases, the dogs were up to six months. Thirty cases (42.8%) occurred in dogs that were between one and six years of age; only seven (10.0%) cases occurred in dogs that were seven years of age or older [57]. These results are similar to those of a North American study that collected data on more than 100 cases of distemper with neurological manifestations, noting that just over half of the cases occurred in dogs up to one year of age, approximately one third of these dogs were less than four months old and most cases occurred in adult dogs [12].
- 5.
- Percentage of dogs for which euthanasia was indicated: in Group 1a, 8 of 52 dogs (15.38%) died, 7 (13.46%) due to the disease and 1 (1.92%) due to euthanasia; in Group 1b, 39 of 46 dogs died (84.78%), 31 (67.39%) died due to the disease and 8 (17.39%) due to euthanasia. In Group 2a, 20 of 58 dogs (34.48%) died, 12 (20.69%) due to the disease and 8 (13.79%) due to euthanasia; in group 2b, 51 of 51 dogs (100.0%) died, 36 (70.59%) due to the disease and 15 (29.41%) due to euthanasia. Regarding the percentage of dogs that were euthanized, our results correspond with those reported by Ranjithkumar et al. (2021), who recorded that the neurological forms of CDV were found in the group of 1–3 years of age. This coincides with the two proposed forms of disease progression, where the primary form states that the progress of cases of distemper encephalomyelitis was initially catarrhal signs, then the appearance of epilepsy, followed by paraplegia, and finally death/euthanasia. The second form presents epilepsy as an initial sign, which can progress to status epilepticus, and then to coma, and finally death/euthanasia [37].
- 6.
- Percentage of dogs that presented sudden death: regarding the percentage of dogs that presented spontaneous deaths, according to the diagnosis of non-neurological distemper and [38,41] neurological distemper, our results presented with mild manifestations of the disease, which varied from practically no clinical signs to severe effects, with a mortality rate between 50 and 100%, as reported by different studies [37,38,58,59,60,61]. The study shows, for the first time, convincing evidence of the benefit of treatment based on AgNPs in dogs naturally infected with CDV that showed clinical signs in non-neurological and neurological stages of the disease. Dogs that were administered with oral and nasal AgNPs showed a higher survival rate, reaching 84.6% in dogs with non-neurological distemper (Group 1a) and 65.5% in dogs with neurological distemper (Group 2a), delaying the appearance of clinical signs and spontaneous deaths related to distemper or euthanasia. Comparing these with the survival rates of control group 1b, at 15.2% with non-neurological distemper, and control group 2b, at 0% with neurological distemper, statistically demonstrates the difference between the survival rates. In addition, it was shown that, with a 15-day oral and nasal treatment of AgNPs in dogs that survived with a diagnosis of non-neurological distemper, 75% did not present neurological sequelae and only 9.6% survived with neurological sequelae. Likewise, it was shown that, with a 30-day oral treatment of AgNPs in dogs that survived with a diagnosis of neurological distemper, 48.3% did not present neurological sequelae and only 17.2% survived with neurological sequelae. The statistical analysis shows a significant difference at 99% reliability in the survival results obtained for both dogs with non-neurological distemper and, most importantly, dogs with neurological distemper. The survival rate decreases from 85 to 65% when compared to the control group, to 15 and 0%, respectively. These differences are relevant, and we can conclude that both neurological and non-neurological distemper treatment with AgNPs are effective.
4.3. General Strengths and Limitations
4.3.1. Strengths
4.3.2. Limitations
- Clinical and epidemiological information for each dog was available to the reviewers.
- Clinical signs of naturally infected dogs with non-neurological and neurological distemper, with a positive serological test by Indirect Immunofluorescent Assay, were registered.
- To confirm an acute CDV infection, the virus-specific IgM titer was determined.
- Diagnosis was confirmed with Reverse Transcription Polymerase Chain Reaction (RT-PCR).
- Subsequently, after clinical treatment, an RT-PCR test was performed, resulting in a negative for all cases.
- Dogs were assessed before and during treatment to evaluate clinical manifestations before inclusion in the group of dogs diagnosed with non-neurological or neurological distemper.
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Willi, B.; Spiri, A.M.; Meli, M.L.; Grimm, F.; Beatrice, L.; Riond, B.; Bley, T.; Jordi, R.; Dennler, M.; Hofmann-Lehmann, R. Clinical and Molecular Investigation of a Canine Distemper Outbreak and Vector-Borne Infections in a Group of Rescue Dogs Imported from Hungary to Switzerland. BMC Vet. Res. 2015, 11, 154. [Google Scholar] [CrossRef] [PubMed]
- Lednicky, J.A.; Dubach, J.; Kinsel, M.J.; Meehan, T.P.; Bocchetta, M.; Hungerford, L.L.; Sarich, N.A.; Witecki, K.E.; Braid, M.D.; Pedrak, C.; et al. Genetically Distant American Canine Distemper Virus Lineages Have Recently Caused Epizootics with Somewhat Different Characteristics in Raccoons Living around a Large Suburban Zoo in the USA. Virol. J. 2004, 1, 2. [Google Scholar] [CrossRef] [PubMed]
- Beineke, A.; Puff, C.; Seehusen, F.; Baumgärtner, W. Pathogenesis and Immunopathology of Systemic and Nervous Canine Distemper. Vet. Immunol. Immunopathol. 2009, 127, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Rendon-Marin, S.; Da Fontoura Budaszewski, R.; Canal, C.W.; Ruiz-Saenz, J. Tropism and Molecular Pathogenesis of Canine Distemper Virus. Virol. J. 2019, 16, 30. [Google Scholar] [CrossRef] [PubMed]
- Deem, S.L.; Spelman, L.H.; Yates, R.A.; Montali, R.J. Canine Distemper in Terrestrial Carnivores: A Review. J. Zoo Wildl. Med. 2000, 31, 441–451. [Google Scholar] [CrossRef] [PubMed]
- Koutinas, A.F.; Baumgärtner, W.; Tontis, D.; Polizopoulou, Z.; Saridomichelakis, M.N.; Lekkas, S. Histopathology and Immunohistochemistry of Canine Distemper Virus-Induced Footpad Hyperkeratosis (Hard Pad Disease) in Dogs with Natural Canine Distemper. Vet. Pathol. 2004, 41, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Beineke, A.; Baumgärtner, W.; Wohlsein, P. Cross-Species Transmission of Canine Distemper Virus-an Update. One Health 2015, 1, 49–59. [Google Scholar] [CrossRef]
- Liu, P.C.; Chen, C.A.; Chen, C.M.; Yen, C.H.; Lee, M.H.; Chuang, C.K.; Tu, C.F.; Su, B.L. Application of Xenogeneic Anti-Canine Distemper Virus Antibodies in Treatment of Canine Distemper Puppies. J. Small Anim. Pract. 2016, 57, 626–630. [Google Scholar] [CrossRef]
- Martella, V.; Elia, G.; Buonavoglia, C. Canine Distemper Virus. Vet. Clin. N. Am. Small Anim. Pract. 2008, 38, 787–797. [Google Scholar] [CrossRef]
- Gonçalves, D.S.V.; Gomes, M.V.S.; Guterra, V.L.P.; Lucchi-Rodrigues, A.F.; Mathias, C.H.T.; Maestri, L.F.P.; Argôlo-Neto, N.M.; Monteiro, B.S. Mesenchymal Stem Cell Infusion for the Treatment of Neurological Sequelae of Canine Distemper Virus: A Clinical Study. Genet. Mol. Res. 2018, 17, GMR18088. [Google Scholar] [CrossRef]
- Loots, A.K.; Mitchell, E.; Dalton, D.L.; Kotzé, A.; Venter, E.H. Advances in Canine Distemper Virus Pathogenesis Research: A Wildlife Perspective. J. Gen. Virol. 2017, 98, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Tipold, A.; Vandevelde, M.; Jaggy, A. Neurological Manifestations of Canine Distemper Virus Infection. J. Small Anim. Pract. 1992, 33, 466–470. [Google Scholar] [CrossRef]
- Rubin, S.; Carr, A. Canine Internal Medicine Secrets E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Cunha, R.D.S.; da Silva Junior, C.L.; Costa, C.A.; de Aguiar, H.M.; Junqueira Júnior, D.G. Comparison of Immunity against Canine Distemper, Adenovirus and Parvovirus after Vaccination with Two Multivalent Canine Vaccines. Vet. Med. Sci. 2020, 6, 330–334. [Google Scholar] [CrossRef] [PubMed]
- Zacarias, J.; Dimande, A.; Achá, S.; Dias, P.T.; Leonel, E.M.; Messa, A.; Macucule, B.; Júnior, J.L.; Bila, C.G. Severe Canine Distemper Outbreak in Unvaccinated Dogs in Mozambique. J. S. Afr. Vet. Assoc. 2016, 87, 2–3. [Google Scholar] [CrossRef] [PubMed]
- Garde, E.; Pérez, G.; Acosta-Jamett, G.; Bronsvoort, B.M. Characteristics of a Canine Distemper Virus Outbreak in Dichato, Chile Following the February 2010 Earthquake. Animals 2013, 3, 843–854. [Google Scholar] [CrossRef] [PubMed]
- Elia, G.; Belloli, C.; Cirone, F.; Lucente, M.S.; Caruso, M.; Martella, V.; Decaro, N.; Buonavoglia, C.; Ormas, P. In Vitro Efficacy of Ribavirin against Canine Distemper Virus. Antivir. Res. 2008, 77, 108–113. [Google Scholar] [CrossRef]
- Lanave, G.; Cavalli, A.; Martella, V.; Fontana, T.; Losappio, R.; Tempesta, M.; Decaro, N.; Buonavoglia, D.; Camero, M. Ribavirin and Boceprevir Are Able to Reduce Canine Distemper Virus Growth in Vitro. J. Virol. Methods 2017, 248, 207–211. [Google Scholar] [CrossRef]
- Carvalho, O.V.; Saraiva, G.L.; Ferreira, C.G.T.; Felix, D.M.; Fietto, J.L.R.; Bressan, G.C.; Almeida, M.R.; Silva Júnior, A. In-Vitro Antiviral Efficacy of Ribavirin and Interferon-Alpha against Canine Distemper Virus. Can. J. Vet. Res. 2014, 78, 283–289. [Google Scholar]
- Wang, H.; Jia, X.; Yang, L.; Sun, L.; Wang, H.; Liu, W. [Comparison of antiviral activity between FeIFN-omega and FeIFN-alpha]. Sheng Wu Gong Cheng Xue Bao 2008, 24, 1556–1560. [Google Scholar]
- Xue, X.; Zhu, Y.; Yan, L.; Wong, G.; Sun, P.; Zheng, X.; Xia, X. Antiviral Efficacy of Favipiravir against Canine Distemper Virus Infection in Vitro. BMC Vet. Res. 2019, 15, 316. [Google Scholar] [CrossRef]
- Carvalho, O.V.; Botelho, C.V.; Ferreira, C.G.T.; Ferreira, H.C.C.; Santos, M.R.; Diaz, M.A.N.; Oliveira, T.T.; Soares-Martins, J.A.P.; Almeida, M.R.; Silva Júnior, A. In Vitro Inhibition of Canine Distemper Virus by Flavonoids and Phenolic Acids: Implications of Structural Differences for Antiviral Design. Res. Vet. Sci. 2013, 95, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Cui, D.; Zuo, Y.; Zheng, Z.; Wu, F.; Li, W.; Zhang, Y.; Huo, S.; Li, N.; Li, L.; et al. Donkey-Derived Anti-CDV IgG, as a Passive Immunotherapy Agent, Can Effectively Increase Survival Rates of the Experimental CDV-Infected Dogs. BMC Vet. Res. 2021, 17, 266. [Google Scholar] [CrossRef] [PubMed]
- Bogdanchikova, N.; Vázquez-Muñoz, R.; Huerta-saquero, A.; Pena-Jasso, A.; Aguilar-Uzcanga, G.; Picos-díaz, P.L.; Pestryakov, A.; Burmistrov, V.; Martynyuk, O.; Luna-Vázquez-Gómez, R.; et al. Silver Nanoparticles Composition for Treatment of Distemper in Dogs. Int. J. Nanotechnol. 2016, 13, 227–237. [Google Scholar] [CrossRef]
- Youssef, F.S.; El-Banna, H.A.; Elzorba, H.Y.; Galal, A.M. Application of Some Nanoparticles in the Field of Veterinary Medicine. Int. J. Vet. Sci. Med. 2019, 7, 78–93. [Google Scholar] [CrossRef]
- Percie du Sert, N.; Hurst, V.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; et al. The ARRIVE Guidelines 2.0: Updated Guidelines for Reporting Animal Research*. J. Cereb. Blood Flow Metab. 2020, 40, 1769–1777. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, A.O.; Cardoso, M.T.; Vidane, A.S.; Casals, J.B.; Passarelli, D.; Alencar, A.L.F.; Sousa, R.L.M.; Fantinato-Neto, P.; Oliveira, V.C.; Lara, V.M.; et al. Controversial Results of Therapy with Mesenchymal Stem Cells in the Acute Phase of Canine Distemper Disease. Genet. Mol. Res. 2016, 15, 2. [Google Scholar] [CrossRef] [PubMed]
- Sedgwick, P. Why Randomise in Clinical Trials? BMJ 2012, 345, 22–23. [Google Scholar] [CrossRef][Green Version]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated Guidelines for Reporting Parallel Group Randomised Trials. J. Pharmacol. Pharmacother. 2010, 1, 100–107. [Google Scholar] [CrossRef]
- Kendall, M.J. Designing a Research Project: Randomised Controlled Trials and Their Principles. Emerg. Med. J. 2003, 20, 164–168. [Google Scholar] [CrossRef]
- Ranjan, R.; Kumar Jha, A.; Kumar, S. Canine Distemper: A Fatal Disease Seeking Special Intervention. J. Entomol. Zool. Stud. 2021, 9, 1411–1418. [Google Scholar]
- Sykes, J. Greene’s Infectious Diseases of the Dog and Cat; Elsevier: Amsterdam, The Netherlands, 2022; ISBN 9780323509343. [Google Scholar]
- Nelson, R.; Couto, C. Small Animal Internal Medicine-E-Book; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Galán, A.; Gamito, A.; Carletti, B.E.; Guisado, A.; De Las Mulas, J.M.; Pérez, J.; Martín, E.M. Case Report Rapport de Cas Uncommon Acute Neurologic Presentation of Canine Distemper in 4 Adult Dogs. Can. Vet. J. 2014, 55, 373–378. [Google Scholar] [PubMed]
- Couto, R.M.; França, S.A.; Rios, M.A.; Rosado, I.R.; Costa, P.M.; Ecco, R. Clinical and Pathological Findings of Necrotizing Meningoencephalitis in a Maltese Dog. Braz. J. Vet. Pathol. 2013, 6, 31–36. [Google Scholar]
- Candini, D.; Biasato, I.; Dell’Armelina Rocha, P.R.; Grego, E.; Capucchio, M.T.; Vercelli, C. How Behavioral Changes Can Indicate Serious Cerebral Pathology: A Case Report of Concomitant Olfactory Neuroblastoma and Distemper Virus Encephalitis in a Swiss Shepherd Dog. Vet. Sci. 2017, 4, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Ranjithkumar, M. Clinical Progression of Naturally Occurring Canine Distemper Encephalomyelitis Cases. Indian J. Vet. Med. 2021, 41, 22–26. [Google Scholar]
- Richards, T.R.; Whelan, N.C.; Pinard, C.L.; Alcala, F.C.; Wolfe, K.C. Optic Neuritis Caused by Canine Distemper Virus in a Jack Russell Terrier. Can. Vet. J. 2011, 52, 398–402. [Google Scholar]
- di Francesco, C.E.; di Francesco, D.; di Martino, B.; Speranza, R.; Santori, D.; Boari, A.; Marsilio, F. Detection by Hemi-Nested Reverse Transcription Polymerase Chain Reaction and Genetic Characterization of Wild Type Strains of Canine Distemper Virus in Suspected Infected Dogs. J. Vet. Diagn. Investig. 2012, 24, 107–115. [Google Scholar] [CrossRef][Green Version]
- Zhang, H.; Shan, F.; Zhou, X.; Li, B.; Zhai, J.Q.; Zou, S.Z.; Wu, M.F.; Chen, W.; Zhai, S.L.; Luo, M.L. Outbreak and Genotyping of Canine Distemper Virus in Captive Siberian Tigers and Red Pandas. Sci. Rep. 2017, 7, 8132. [Google Scholar] [CrossRef]
- Sun, L.; Singh, A.; Vig, K.; Pillai, S.R.; Singh, S.R. Silver Nanoparticles Inhibit Replication of Respiratory Syncytial Virus. J. Biomed. Nanotechnol. 2008, 4, 149–158. [Google Scholar] [CrossRef]
- Morris, D.; Ansar, M.; Speshock, J.; Ivanciuc, T.; Qu, Y.; Casola, A.; Garofalo, R. Antiviral and Immunomodulatory Activity of Silver Nanoparticles in Experimental Rsv Infection. Viruses 2019, 11, 732. [Google Scholar] [CrossRef]
- Gaikwad, S.; Ingle, A.; Gade, A.; Rai, M.; Falanga, A.; Incoronato, N.; Russo, L.; Galdiero, S.; Galdiero, M. Antiviral Activity of Mycosynthesized Silver Nanoparticles against Herpes Simplex Virus and Human Parainfluenza Virus Type 3. Int. J. Nanomed. 2013, 8, 4303–4314. [Google Scholar] [CrossRef]
- Yin, J.J.; Li, X.J.; Zheng, C.L. Potential Mechanism and Inhibitory Effects of Silver Nanoparticles on Parainfluenza Virus Type 3. J. Jiangsu Univ. 2013, 3, 191. [Google Scholar]
- Khandelwal, N.; Kaur, G.; Chaubey, K.K.; Singh, P.; Sharma, S.; Tiwari, A.; Singh, S.V.; Kumar, N. Silver Nanoparticles Impair Peste Des Petits Ruminants Virus Replication. Virus Res. 2014, 190, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Summers, B.A.; Greisen, H.A.; Appel, M.J. Canine Distemper Encephalomyelitis: Variation with Virus Strain. J. Comp. Pathol. 1984, 94, 65–75. [Google Scholar] [CrossRef]
- Krakowka, S.; Olsen, R.; Confer, A.; Koestner, A.; Mccullough, B. Serologic Response to Canine Distemper Viral Antigens in Gnotobiotic Dogs Infected with Canine Distemper Virus. J. Infect. Dis. 1975, 132, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Vandevelde, M.; Zurbriggen, A. The Neurobiology of Canine Distemper Virus Infection. Vet. Microbiol. 1995, 44, 271–280. [Google Scholar] [CrossRef]
- Blixenkrone-Moller, M.; Pedersen, I.R.; Appel, M.J.; Griot, C. Detection of IgM Antibodies against Canine Distemper Virus in Dog and Mink Sera Employing Enzyme-Linked Immunosorbent Assay (ELISA). J. Vet. Diagn. Investig. 1991, 3, 3–9. [Google Scholar] [CrossRef]
- von Messling, V.; Harder, T.C.; Moennig, V.; Rautenberg, P.; Nolte, I.; Haas, L. Rapid and Sensitive Detection of Immunoglobulin M (IgM) and IgG Antibodies against Canine Distemper Virus by a New Recombinant Nucleocapsid Protein-Based Enzyme-Linked Immunosorbent Assay. J. Clin. Microbiol. 1999, 37, 1049–1056. [Google Scholar] [CrossRef]
- Jóźwik, A.; Frymus, T.; Mizak, B.; Rzezutka, A. Antibody Titres against Canine Distemper Virus in Vaccinated and Unvaccinated Dogs. J. Vet. Med. Ser. B Infect. Dis. Vet. Public Health 2004, 51, 99–103. [Google Scholar] [CrossRef]
- Borrego, B.; Lorenzo, G.; Mota-Morales, J.D.; Almanza-Reyes, H.; Mateos, F.; López-Gil, E.; de la Losa, N.; Burmistrov, V.A.; Pestryakov, A.N.; Brun, A.; et al. Potential Application of Silver Nanoparticles to Control the Infectivity of Rift Valley Fever Virus in Vitro and in Vivo. Nanomedicine 2016, 12, 1185–1192. [Google Scholar] [CrossRef]
- Nefedova, E.; Koptev, V.; Bobikova, A.S.; Cherepushkina, V.; Mironova, T.; Afonyushkin, V.; Shkil, N.; Donchenko, N.; Kozlova, Y.; Sigareva, N.; et al. The Infectious Bronchitis Coronavirus Pneumonia Model Presenting a Novel Insight for the Sars-Cov-2 Dissemination Route. Vet. Sci. 2021, 8, 239. [Google Scholar] [CrossRef]
- Almanza-Reyes, H.; Moreno, S.; Plascencia-López, I.; Alvarado-Vera, M.; Patrón-Romero, L.; Borrego, B.; Reyes-Escamilla, A.; Valencia-Manzo, D.; Brun, A.; Pestryakov, A.; et al. Evaluation of Silver Nanoparticles for the Prevention of SARS-CoV-2 Infection in Health Workers: In Vitro and in Vivo. PLoS ONE 2021, 16, e0256401. [Google Scholar] [CrossRef] [PubMed]
- Fighera, R.A.; Souza, T.M.; Silva, M.C.; Brum, J.S.; Graça, D.L.; Kommers, G.D.; Irigoyen, L.F.; Barros, C.S.L. Causas de Morte e Razões Para Eutanásia de Cães Da Mesorregião Do Centro Ocidental Rio-Grandense. Pesqui. Veterinária Bras. 2008, 28, 223–230. [Google Scholar] [CrossRef]
- Koutinas, A.F.; Polizopoulou, Z.S.; Baumgaertner, W.; Lekkas, S.; Kontos, V. Relation of Clinical Signs to Pathological Changes in 19 Cases of Canine Distemper Encephalomyelitis. J. Comp. Pathol. 2002, 126, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.C.; Fighera, R.A.; Mazzanti, A.; Brum, J.S.; Pierezan, F.; Barros, C.S.L. Neuropatologia Da Cinomose Canina: 70 Casos (2005–2008)1. Pesq. Vet. Bras. 2009, 29, 643–652. [Google Scholar] [CrossRef]
- Krakowka, S.; Koestner, A. Age-Related Susceptibility to Infection with Canine Distemper Virus in Gnotobiotic Dogs. J. Infect. Dis. 1976, 134, 629–632. [Google Scholar] [CrossRef]
- Study, A.I.; Vandevelde, M.; Fankhauser, R.; Kristensen, F.; Kristensen, B. Acta Neuropathologica Immunoglobulins in Demyelinating Lesions in Canine Distemper Encephalitis. Acta Neuropathol. 1981, 54, 31–41. [Google Scholar]
- Giulia, D.; Francesca, V.; Sandra, S.; Alessandra, S.; Gualtiero, G.; Luciana, G.; Laura, C. A Molecular Study of Hippocampus in Dogs with Convulsion during Canine Distemper Virus Encephalitis. Brain Res. 2006, 1098, 186–195. [Google Scholar] [CrossRef]
- Vandevelde, M.; Zurbriggen, A.; Higgins, R.J.; Palmer, D. Spread and Distribution of Viral Antigen in Nervous Canine Distemper*; Springer: Berlin/Heidelberg, Germany, 1985; Volume 67. [Google Scholar]
- Clausen, M.B.; Bandholm, T.; Rathleff, M.S.; Christensen, K.B.; Zebis, M.K.; Graven-Nielsen, T.; Hölmich, P.; Thorborg, K. The Strengthening Exercises in Shoulder Impingement Trial (The SExSI-Trial) Investigating the Effectiveness of a Simple Add-on Shoulder Strengthening Exercise Programme in Patients with Long-Lasting Subacromial Impingement Syndrome: Study Protocol for a Pragmatic, Assessor Blinded, Parallel-Group, Randomised, Controlled Trial. Trials 2018, 19. [Google Scholar] [CrossRef]
- Andersen, A.L.; Houlind, M.B.; Nielsen, R.L.; Jørgensen, L.M.; Treldal, C.; Damgaard, M.; Bengaard, A.K.; Juul-Larsen, H.G.; Laursen, L.B.; Iversen, E.; et al. Optimization of Nutrition And Medication (OptiNAM) for Acutely Admitted Older Patients: Protocol for a Randomized Single-Blinded Controlled Trial. Trials 2021, 22, 616. [Google Scholar] [CrossRef]
- Bhandari, M.; Busse, J.W.; Jackowski, D.; Montori, V.M.; Schünemann, H.; Sprague, S.; Mears, D.; Schemitsch, E.H.; Heels-Ansdell, D.; Devereaux, P.J. Association between Industry Funding and Statistically Significant Pro-Industry Findings in Medical and Surgical Randomized Trials. Can. Med. Assoc. J. 2004, 17, 477–480. [Google Scholar]
- Anderson, M.L.; Chiswell, K.; Peterson, E.D.; Tasneem, A.; Topping, J.; Califf, R.M. Compliance with Results Reporting at ClinicalTrials.Gov. N. Engl. J. Med. 2015, 372, 1031–1039. [Google Scholar] [CrossRef]
- Lexchin, J.; Bero, L.A.; Djulbegovic, B.; Clark, O. Pharmaceutical Industry Sponsorship and Research Outcome and Quality: Systematic Review. BMJ 2003, 326, 1167–1170. [Google Scholar] [CrossRef] [PubMed]
- Lundh, A.; Lexchin, J.; Mintzes, B.; Schroll, J.B.; Bero, L. Industry Sponsorship and Research Outcome. Cochrane Database Syst. Rev. 2017, 2, MR000033. [Google Scholar] [CrossRef] [PubMed]
- Okike, K.; Kocher, M.S.; Wei, E.X.; Mehlman, C.T.; Bhandari, M. Accuracy of Conflict-of-Interest Disclosures Reported by Physicians. N. Engl. J. Med. 2009, 361, 1466–1474. [Google Scholar] [CrossRef] [PubMed]
- Dickersin, K.; Chalmers, I. Recognizing, Investigating and Dealingwith Incomplete and Biased Reporting of Clinical Research: From Francis Bacon to the WHO. J. R. Soc. Med. 2011, 104, 532–538. [Google Scholar] [CrossRef] [PubMed]
- Schott, G.; Pachl, H.; Limbach, U.; Gundert-Remy, U.; Ludwig, W.D.; Lieb, K. Finanzierung von Arzneimittelstudien Durch Pharmazeutische Unternehmen Und Die Folgen. Teil 2010, 107, 279–285. [Google Scholar] [CrossRef]
- Harmon, C.P.; Chalmers, P.N.; Carpiniello, M.F.; Cvetanovich, G.L.; Cole, B.J.; Bach, B.R. Inconsistencies between Physician-Reported Disclosures at the AAOS Annual Meeting and Industry-Reported Financial Disclosures in the Open Payments Database. J. Bone Jt. Surg. Am. 2016, 98, e90. [Google Scholar]
- Lane, P. Handling Drop-out in Longitudinal Clinical Trials: A Comparison of the LOCF and MMRM Approaches. Pharm. Stat. J. Appl. Stat. 2008, 7, 93–106. [Google Scholar] [CrossRef]
| Medications * | Description |
|---|---|
| Interferons | Feline interferon alpha (FeIFN-α) and recombinant feline interferon omega (rFeIFN-Ω). |
| Immunotherapy | Autologous hyperimmune sera and heterologous hyperimmune sera. |
| Antivirals | Ribavirin, Boceprevir, Favipiravir and Azatioprin. |
| Investigational therapies | Sulfated polysaccharide, flavonoids (quercetin, morin, rutin and hesperidin), phenolic acids (cinnamic, trans-cinnamic and ferulic), mesenchymal stem cells and dialyzed leukocyte extract (DLE). |
| Other | Immunostimulants based on the lyophilized culture of inactivated Parapoxvirus ovis. |
| Study | Group 1a | Group 1b | Group 2a | Group 2b | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Week | Week | Week | Week | |||||||
| 0 | 2 | 0 | 2 | 0 | 2 | 4 | 0 | 2 | 4 | |
| Questionnaire | X | X | X | X | X | X | X | X | X | a |
| Clinical evaluation | X | X | X | X | X | X | X | X | X | a |
| Clinical chemistry | X | X | X | X | X | X | X | X | X | a |
| Study | Reference | Group 1a | Group 1b | Group 2a | Group 2b | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Week 0 | Week 2 | Week 0 | Week 2 | Week 0 | Week 2 | Week 4 | Week 0 | Week 2 | Week 4 | ||
| Glucose | 90–150 mg/dL | 107 ± 10 | 103 ± 10 | 102 ± 10 | 105 ± 10 | 101 ± 10 | 99 ± 10 | 102 ± 10 | 105 ± 10 | 101 ± 10 | ----- |
| BUN | 10–58 mg/dL | 23 ± 8 | 22 ± 8 | 25 ± 8 | 27 ± 8 | 24 ± 8 | 26 ± 8 | 29 ± 8 | 20 ± 8 | 25 ± 8 | ----- |
| Creatinine | 0.3–1.4 mg/dL | 0.9 ± 0.3 | 1.0 ± 0.3 | 1.1 ± 0.3 | 0.9 ± 0.3 | 0.8 ± 0.3 | 1.0 ± 0.3 | 1.2 ± 0.3 | 1.0 ± 0.3 | 1.2 ± 0.3 | ----- |
| Cholesterol | 125–310 mg/dL | 299 ± 60 | 290 ± 60 | 291 ± 60 | 295 ± 60 | 289 ± 60 | 292 ± 60 | 298 ± 60 | 291 ± 60 | 295 ± 60 | ----- |
| Triglycerides | 40–150 mg/dL | 65 ± 15 | 62 ± 15 | 61 ± 15 | 64 ± 15 | 60 ± 15 | 62 ± 15 | 65 ± 15 | 62 ± 15 | 64 ± 15 | ----- |
| Total protein | 4.0–5.8 g/dL | 4.5 ± 0.3 | 4.7 ± 0.3 | 5.1 ± 0.3 | 5.0 ± 0.3 | 4.9 ± 0.3 | 4.6 ± 0.3 | 4.7 ± 0.3 | 4.8 ± 0.3 | 5.0 ± 0.3 | ----- |
| Albumin | 2.2–2.9 g/dL | 2.4 ± 0.3 | 2.7 ± 0.3 | 2.5 ± 0.3 | 2.6 ± 0.3 | 2.7 ± 0.3 | 2.8 ± 0.3 | 2.6 ± 0.3 | 2.6 ± 0.3 | 2.8 ± 0.3 | ----- |
| Globulin | 2.1–4.4 g/dL | 2.5 ± 0.3 | 2.7 ± 0.3 | 2.4 ± 0.3 | 2.6 ± 0.3 | 2.6 ± 0.3 | 2.5 ± 0.3 | 2.7 ± 0.3 | 2.7 ± 0.3 | 2.9 ± 0.3 | ----- |
| ALP | 75–450 U/L | 136 ± 90 | 155 ± 95 | 140 ± 90 | 163 ± 100 | 146 ± 90 | 165 ± 95 | 189 ± 105 | 145 ± 90 | 149 ± 90 | ----- |
| ALT | 10–60 U/L | 30 ± 25 | 35 ± 28 | 40 ± 25 | 48 ± 25 | 39 ± 25 | 47 ± 30 | 56 ± 30 | 39 ± 25 | 43 ± 25 | ----- |
| AST | 10–65 U/L | 33 ± 20 | 43 ± 30 | 36 ± 20 | 39 ± 20 | 29 ± 20 | 38 ± 25 | 45 ± 30 | 39 ± 20 | 46 ± 20 | ----- |
| GGT | 3–9 U/L | 5 ± 2.5 | 7 ± 2.5 | 4.5 ± 2.5 | 6.5 ± 2.5 | 4.9 ± 2.5 | 6.1 ± 2.5 | 7.5 ± 3 | 4.8 ± 2.5 | 5.8 ± 2.5 | ----- |
| Total Bilirubin | 0.1–1.0 mg/dL | 0.5 ± 0.2 | 0.6 ± 0.2 | 0.4 ± 0.2 | 0.5 ± 0.2 | 0.5 ± 0.2 | 0.4 ± 0.2 | 0.5 ± 0.2 | 0.7 ± 0.2 | 0.6 ± 0.2 | ----- |
| Uric acid | 0.2–1.6 mg/dL | 0.9 ± 0.2 | 1.1 ± 0.2 | 0.6 ± 0.2 | 0.8 ± 0.2 | 0.7 ± 0.2 | 0.9 ± 0.2 | 0.8 ± 0.2 | 0.5 ± 0.2 | 0.7 ± 0.2 | ----- |
| Phosphorus | 4.8–10.7 mg/dL | 6.8 ± 0.6 | 7.1 ± 0.6 | 5.8 ± 0.6 | 6.1 ± 0.6 | 5.9 ± 0.6 | 6.1 ± 0.6 | 6.9 ± 0.6 | 7.8 ± 0.6 | 8.1 ± 0.6 | ----- |
| Amylase | 230–500 U/L | 290 ± 60 | 300 ± 60 | 310 ± 60 | 300 ± 60 | 315 ± 60 | 340 ± 60 | 320 ± 60 | 290 ± 60 | 310 ± 60 | ----- |
| Lipase | <550 U/L | 380 ± 80 | 397 ± 80 | 401 ± 80 | 390 ± 80 | 389 ± 80 | 397 ± 80 | 409 ± 80 | 310 ± 80 | 370 ± 80 | ----- |
| Group 1a | Group 1b | Group 2a | Group 2b | |
|---|---|---|---|---|
| Total survival rate | 44/52 (84.6%) | 7/46 (15.2%) | 38/58(65.5%) | 0/51(0%) |
| Survival rate of dogs without sequels | 39/52 (75.0%) | 3/46 (6.5%) | 28/58(48.3%) | 0/51(0%) |
| Survival rate of dogs with sequels | 5/52 (9.6%) | 4/46 (8.7%) | 10/58(17.2%) | 0/51(0%) |
| Percentage of dogs died | 8/52 (15.38%) | 39/46 (84.78%) | 20/58 (34.48%) | 51/51 (100%) |
| Percentage of dogs died of the disease | 7/52 (13.46%) | 31/46 (67.39%) | 12/58 (20.69%) | 36/51 (70.59%) |
| Percentage of dogs died by euthanasia * | 1/52 (1.92%) | 8/46 (17.93%) | 8/58 (13.79%) | 15/51 (29.41%) |
| Overall Comparisons | |||
|---|---|---|---|
| Chi-Square | df | Sig. | |
| Log Rank (Mantel–Cox) | 48.187 | 1 | <0.001 |
| Breslow (Generalized Wilcoxon) | 41.249 | 1 | <0.001 |
| Tarone–Ware | 44.785 | 1 | <0.001 |
| Overall Comparisons | |||
|---|---|---|---|
| Chi-Square | df | Sig. | |
| Log Rank (Mantel–Cox) | 57.062 | 1 | <0.001 |
| Case Processing Summary | ||||
|---|---|---|---|---|
| Censored | ||||
| Treatment | Total N | N of Events | N | Percent |
| Control | 46 | 39 | 7 | 15.2% |
| AgNPs | 52 | 8 | 44 | 84.6% |
| Overall | 98 | 47 | 51 | 52.0% |
| Case Processing Summary | ||||
|---|---|---|---|---|
| Censored | ||||
| Treatment | Total N | N of Events | N | Percent |
| Control | 58 | 20 | 38 | 65.5% |
| AgNPs | 51 | 51 | 0 | 0.0% |
| Overall | 109 | 71 | 38 | 34.9% |
| Mean a | ||||
|---|---|---|---|---|
| 95% Confidence Interval | ||||
| Estimate | Std. Error | Lower Bound | Upper Bound | |
| Control | 8.783 | 0.597 | 7.613 | 9.952 |
| AgNPs | 13.750 | 0.429 | 12.909 | 14.591 |
| Overall | 11.418 | 0.439 | 10.557 | 12.279 |
| Mean a | ||||
|---|---|---|---|---|
| 95% Confidence Interval | ||||
| Estimate | Std. Error | Lower Bound | Upper Bound | |
| Control | 22.500 | 1.121 | 2.304 | 24.696 |
| AgNPs | 11.941 | 0.966 | 10.047 | 13.835 |
| Overall | 17.560 | 0.900 | 15.795 | 19.324 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gastelum-Leyva, F.; Pena-Jasso, A.; Alvarado-Vera, M.; Plascencia-López, I.; Patrón-Romero, L.; Loera-Castañeda, V.; Gándara-Mireles, J.A.; Lares-Asseff, I.; Leal-Ávila, M.Á.; Alvelais-Palacios, J.A.; et al. Evaluation of the Efficacy and Safety of Silver Nanoparticles in the Treatment of Non-Neurological and Neurological Distemper in Dogs: A Randomized Clinical Trial. Viruses 2022, 14, 2329. https://doi.org/10.3390/v14112329
Gastelum-Leyva F, Pena-Jasso A, Alvarado-Vera M, Plascencia-López I, Patrón-Romero L, Loera-Castañeda V, Gándara-Mireles JA, Lares-Asseff I, Leal-Ávila MÁ, Alvelais-Palacios JA, et al. Evaluation of the Efficacy and Safety of Silver Nanoparticles in the Treatment of Non-Neurological and Neurological Distemper in Dogs: A Randomized Clinical Trial. Viruses. 2022; 14(11):2329. https://doi.org/10.3390/v14112329
Chicago/Turabian StyleGastelum-Leyva, Fabian, Antonio Pena-Jasso, Martha Alvarado-Vera, Ismael Plascencia-López, Leslie Patrón-Romero, Verónica Loera-Castañeda, Jesús Alonso Gándara-Mireles, Ismael Lares-Asseff, María Ángeles Leal-Ávila, J. A. Alvelais-Palacios, and et al. 2022. "Evaluation of the Efficacy and Safety of Silver Nanoparticles in the Treatment of Non-Neurological and Neurological Distemper in Dogs: A Randomized Clinical Trial" Viruses 14, no. 11: 2329. https://doi.org/10.3390/v14112329
APA StyleGastelum-Leyva, F., Pena-Jasso, A., Alvarado-Vera, M., Plascencia-López, I., Patrón-Romero, L., Loera-Castañeda, V., Gándara-Mireles, J. A., Lares-Asseff, I., Leal-Ávila, M. Á., Alvelais-Palacios, J. A., Almeida-Pérez, J., Bogdanchikova, N., Pestryakov, A., & Almanza-Reyes, H. (2022). Evaluation of the Efficacy and Safety of Silver Nanoparticles in the Treatment of Non-Neurological and Neurological Distemper in Dogs: A Randomized Clinical Trial. Viruses, 14(11), 2329. https://doi.org/10.3390/v14112329






