Breaking Entry-and Species Barriers: LentiBOOST® Plus Polybrene Enhances Transduction Efficacy of Dendritic Cells and Monocytes by Adenovirus 5
Abstract
1. Introduction
2. Materials and Methods
2.1. Mice
2.2. Generation of Murine Bone Marrow-Derived DCs
2.3. Generation of Human Monocytes, Monocyte-Derived DCs and T Cells
2.4. Recombinant Adenoviruses
2.5. Adenoviral Transduction
2.6. Flow Cytometry
2.7. Cell Imaging
2.8. Dimethylthiazol–Diphenyltetrazolium (MTT) Assay
2.9. Mixed Lymphocyte Reaction (MLR)
2.10. Cytometric Bead Array
2.11. In-Vitro RNA Transcription and Electroporation of Human DCs
2.12. Priming of CD8+ T Cells
2.13. Statistical Analysis
3. Results
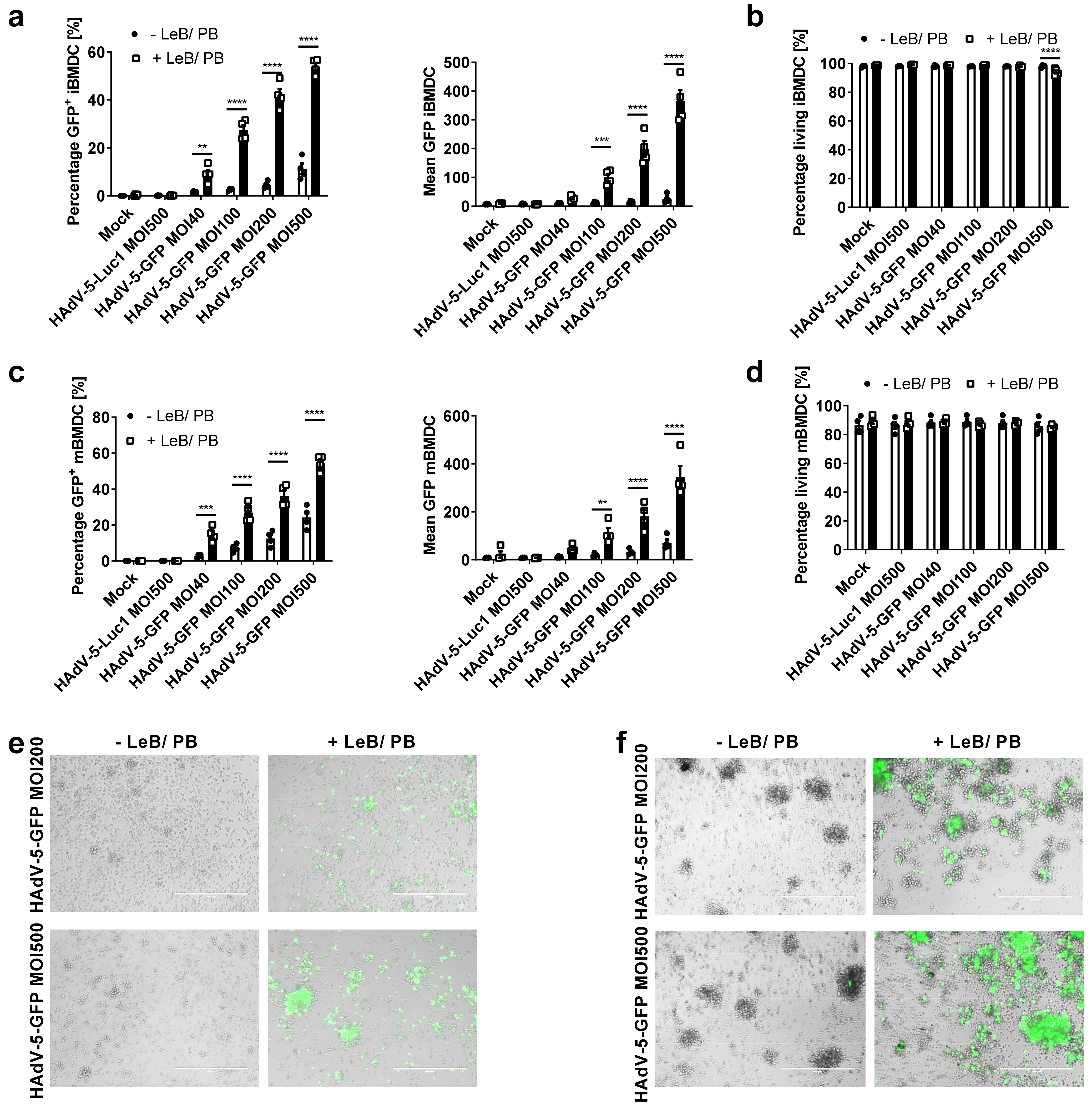
3.1. LentiBOOST® in Combination with Polybrene Enables Transduction of Murine BMDCs with HAdV-5
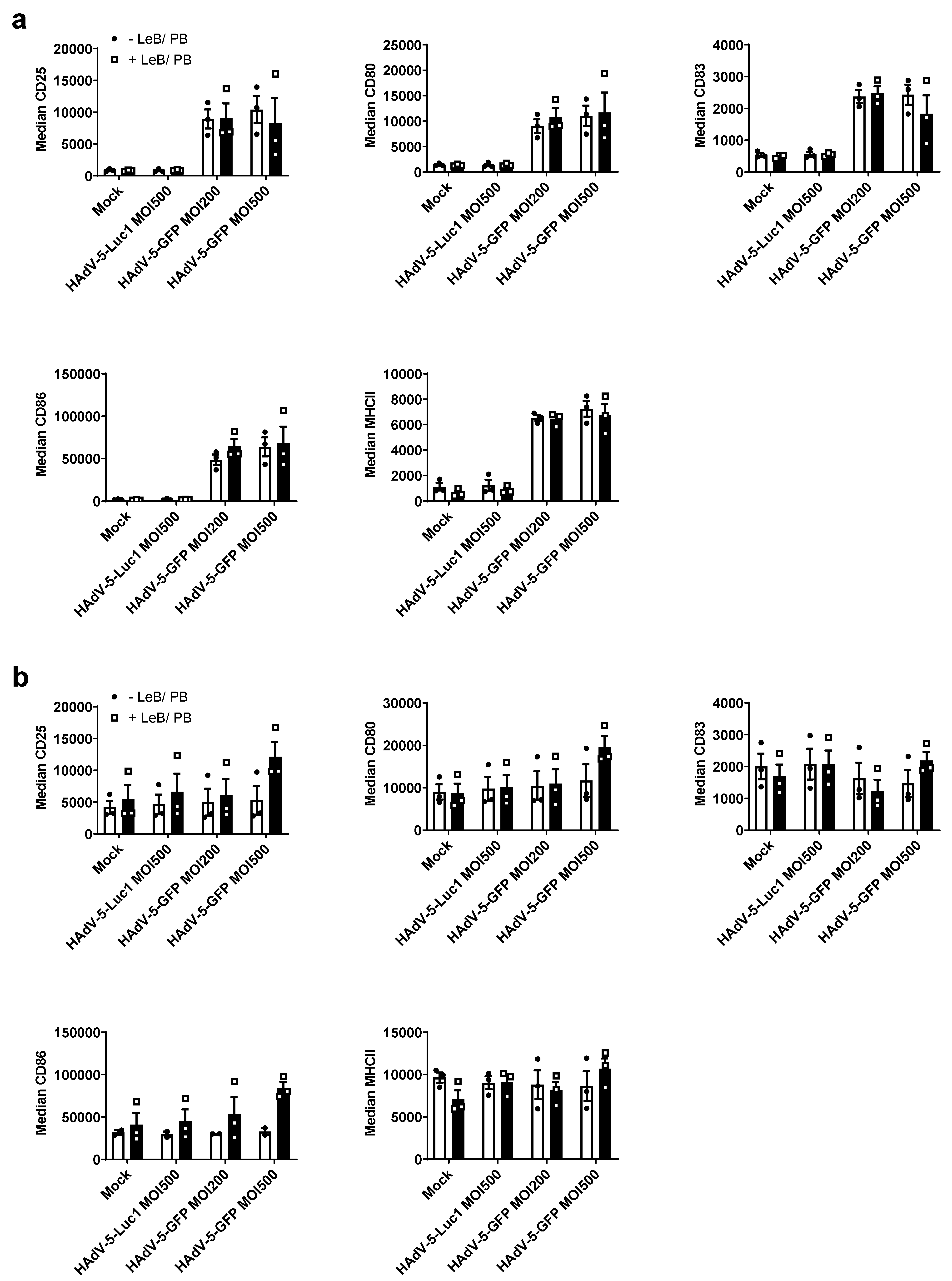
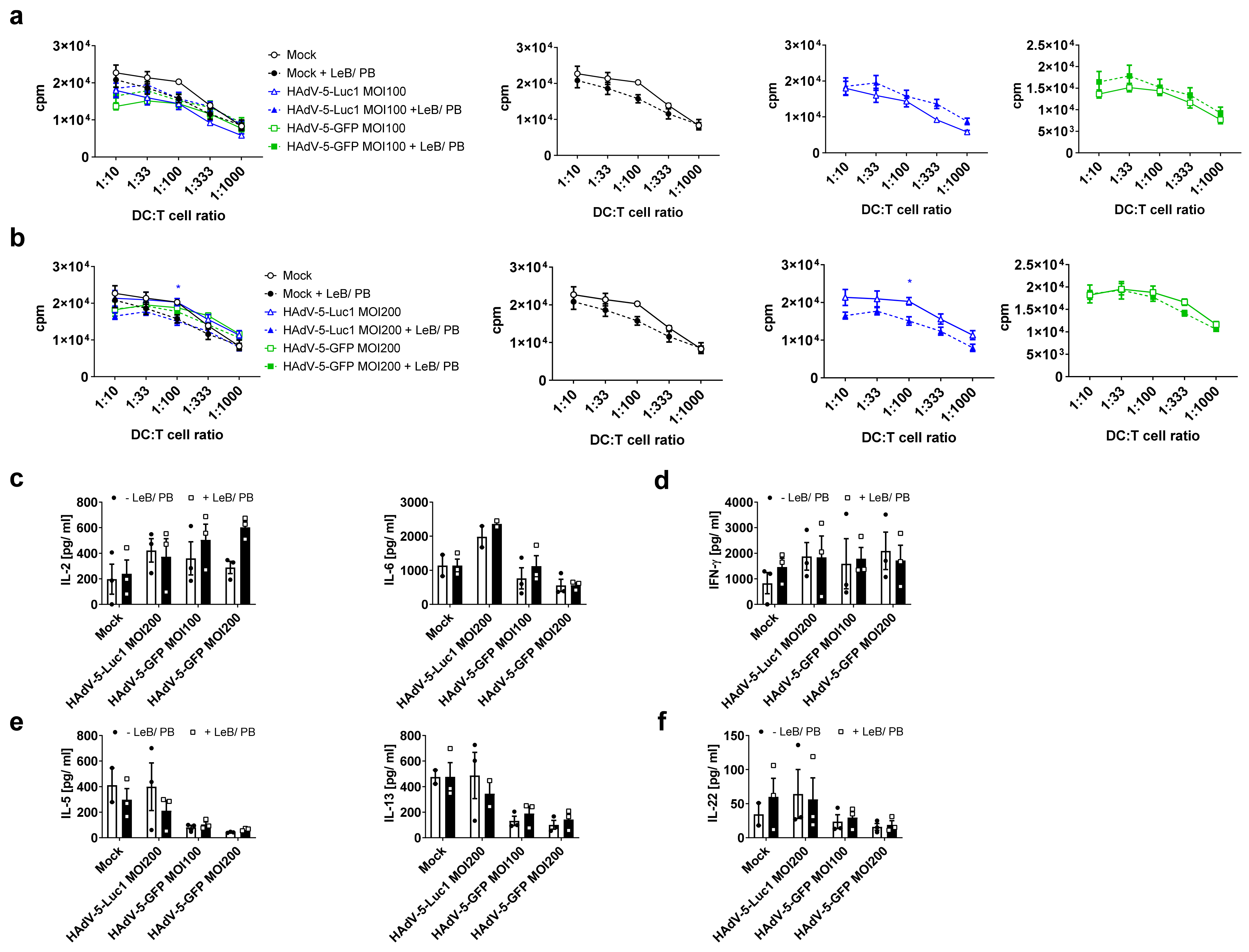
3.2. LentiBOOST®/Polybrene Does Not Alter the Phenotype and Function of Murine BMDCs
3.3. LentiBOOST®/Polybrene Increases Transduction Efficiency of Human Monocyte-Derived DCs at Low HAdV-5 Virus Titers
3.4. LentiBOOST®/Polybrene Induces Maturation of Immature Human DCs but Does Not Alter DC Function
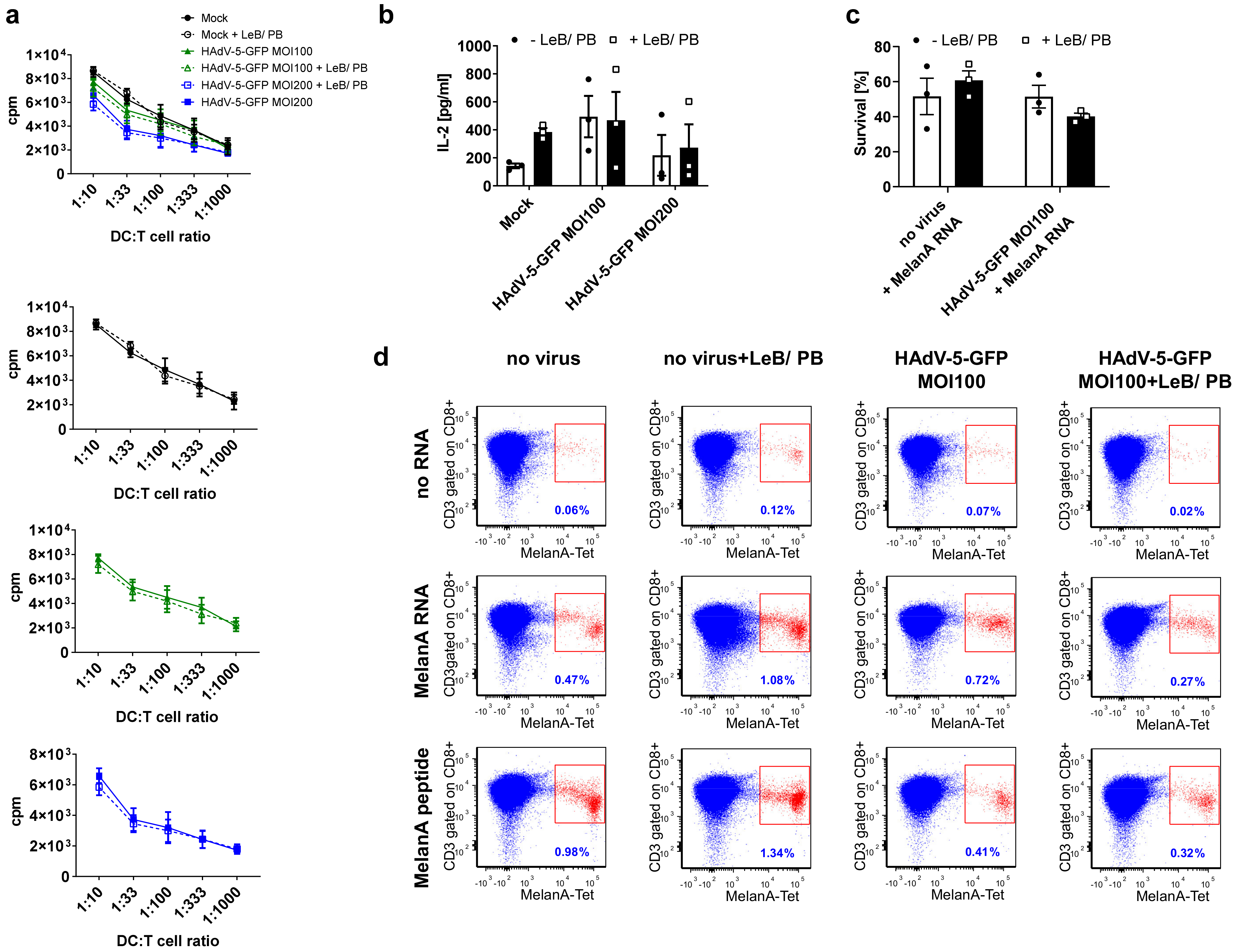
3.5. Efficient HAdV-5-Mediated Gene Transfer into Monocytes by LentiBOOST®/Polybrene Allows for Proper Subsequent iDCs and mDCs Differentiation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lee, C.S.; Bishop, E.S.; Zhang, R.; Yu, X.; Farina, E.M.; Yan, S.; Zhao, C.; Zheng, Z.; Shu, Y.; Wu, X.; et al. Adenovirus-Mediated Gene Delivery: Potential Applications for Gene and Cell-Based Therapies in the New Era of Personalized Medicine. Genes Dis. 2017, 4, 43–63. [Google Scholar] [CrossRef] [PubMed]
- Crenshaw, B.J.; Jones, L.B.; Bell, C.R.; Kumar, S.; Matthews, Q.L. Perspective on Adenoviruses: Epidemiology, Pathogenicity, and Gene Therapy. Biomedicines 2019, 7, 61. [Google Scholar] [CrossRef]
- King, R.G.; Silva-Sanchez, A.; Peel, J.N.; Botta, D.; Meza-Perez, S.; Allie, R.; Schultz, M.D.; Liu, M.; Bradley, J.E.; Qiu, S.; et al. Single-dose intranasal administration of AdCOVID elicits systemic and mucosal immunity against SARS-CoV-2 in mice. bioRxiv 2020. Preprint. [Google Scholar] [CrossRef]
- Gao, J.; Zhang, W.; Ehrhardt, A. Expanding the Spectrum of Adenoviral Vectors for Cancer Therapy. Cancers 2020, 12, 1139. [Google Scholar] [CrossRef] [PubMed]
- Wei, C.J.; Boyington, J.C.; McTamney, P.M.; Kong, W.P.; Pearce, M.B.; Xu, L.; Andersen, H.; Rao, S.; Tumpey, T.M.; Yang, Z.Y.; et al. Induction of broadly neutralizing H1N1 influenza antibodies by vaccination. Science 2010, 329, 1060–1064. [Google Scholar] [CrossRef]
- Alexander, J.; Ward, S.; Mendy, J.; Manayani, D.J.; Farness, P.; Avanzini, J.B.; Guenther, B.; Garduno, F.; Jow, L.; Snarsky, V.; et al. Pre-clinical evaluation of a replication-competent recombinant adenovirus serotype 4 vaccine expressing influenza H5 hemagglutinin. PLoS ONE 2012, 7, e31177. [Google Scholar] [CrossRef]
- Vannucci, L.; Lai, M.; Chiuppesi, F.; Ceccherini-Nelli, L.; Pistello, M. Viral vectors: A look back and ahead on gene transfer technology. New Microbiol. 2013, 36, 1–22. [Google Scholar]
- Alonso-Padilla, J.; Papp, T.; Kajan, G.L.; Benko, M.; Havenga, M.; Lemckert, A.; Harrach, B.; Baker, A.H. Development of Novel Adenoviral Vectors to Overcome Challenges Observed With HAdV-5-based Constructs. Mol. Ther. 2016, 24, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.S.; Natchiar, S.K.; Stewart, P.L.; Nemerow, G.R. Crystal structure of human adenovirus at 3.5 A resolution. Science 2010, 329, 1071–1075. [Google Scholar] [CrossRef]
- Liu, H.; Jin, L.; Koh, S.B.; Atanasov, I.; Schein, S.; Wu, L.; Zhou, Z.H. Atomic structure of human adenovirus by cryo-EM reveals interactions among protein networks. Science 2010, 329, 1038–1043. [Google Scholar] [CrossRef]
- Reddy, V.S.; Nemerow, G.R. Structures and organization of adenovirus cement proteins provide insights into the role of capsid maturation in virus entry and infection. Proc. Natl. Acad. Sci. USA 2014, 111, 11715–11720. [Google Scholar] [CrossRef]
- Benevento, M.; Di Palma, S.; Snijder, J.; Moyer, C.L.; Reddy, V.S.; Nemerow, G.R.; Heck, A.J.R. Adenovirus composition, proteolysis, and disassembly studied by in-depth qualitative and quantitative proteomics. J. Biol. Chem. 2014, 289, 11421–11430. [Google Scholar] [CrossRef]
- Arnberg, N. Adenovirus receptors: Implications for targeting of viral vectors. Trends Pharmacol. Sci. 2012, 33, 442–448. [Google Scholar] [CrossRef]
- Boucher, P.; Cui, X.; Curiel, D.T. Adenoviral vectors for In Vivo delivery of CRISPR-Cas gene editors. J. Control. Release 2020, 327, 788–800. [Google Scholar] [CrossRef]
- Stein, M.F.; Lang, S.; Winkler, T.H.; Deinzer, A.; Erber, S.; Nettelbeck, D.M.; Naschberger, E.; Jochmann, R.; Sturzl, M.; Slany, R.K.; et al. Multiple interferon regulatory factor and NF-kappaB sites cooperate in mediating cell-type- and maturation-specific activation of the human CD83 promoter in dendritic cells. Mol. Cell. Biol. 2013, 33, 1331–1344. [Google Scholar] [CrossRef]
- Diao, J.; Smythe, J.A.; Smyth, C.; Rowe, P.B.; Alexander, I.E. Human PBMC-derived dendritic cells transduced with an adenovirus vector induce cytotoxic T-lymphocyte responses against a vector-encoded antigen In Vitro. Gene Ther. 1999, 6, 845–853. [Google Scholar] [CrossRef]
- Dietz, A.B.; Vuk-Pavlovic, S. High Efficiency Adenovirus-Mediated Gene Transfer to Human Dendritic Cells. Blood 1998, 91, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Tan, P.H.; Beutelspacher, S.C.; Xue, S.-A.; Wang, Y.-H.; Mitchell, P.; McAlister, J.C.; Larkin, D.F.P.; McClure, M.O.; Stauss, H.J.; Ritter, M.A.; et al. Modulation of human dendritic-cell function following transduction with viral vectors: Implications for gene therapy. Blood 2005, 105, 3824–3832. [Google Scholar] [CrossRef]
- Tatsis, N.; Ertl, H.C. Adenoviruses as vaccine vectors. Mol. Ther. 2004, 10, 616–629. [Google Scholar] [CrossRef] [PubMed]
- Gregory, L.G.; Harbottle, R.P.; Lawrence, L.; Knapton, H.J.; Themis, M.; Coutelle, C. Enhancement of adenovirus-mediated gene transfer to the airways by DEAE dextran and sodium caprate In Vivo. Mol. Ther. 2003, 7, 19–26. [Google Scholar] [CrossRef]
- McKay, T.R.; MacVinish, L.J.; Carpenter, B.; Themis, M.; Jezzard, S.; Goldin, R.; Pavirani, A.; Hickman, M.E.; Cuthbert, A.W.; Coutelle, C. Selective In Vivo transfection of murine biliary epithelia using polycation-enhanced adenovirus. Gene Ther. 2000, 7, 644–652. [Google Scholar] [CrossRef]
- Anastasov, N.; Hofig, I.; Mall, S.; Krackhardt, A.M.; Thirion, C. Optimized Lentiviral Transduction Protocols by Use of a Poloxamer Enhancer, Spinoculation, and scFv-Antibody Fusions to VSV-G. Methods Mol. Biol. 2016, 1448, 49–61. [Google Scholar] [CrossRef]
- Delville, M.; Soheili, T.; Bellier, F.; Durand, A.; Denis, A.; Lagresle-Peyrou, C.; Cavazzana, M.; Andre-Schmutz, I.; Six, E. A Nontoxic Transduction Enhancer Enables Highly Efficient Lentiviral Transduction of Primary Murine T Cells and Hematopoietic Stem Cells. Mol. Ther. Methods Clin. Dev. 2018, 10, 341–347. [Google Scholar] [CrossRef]
- Schott, J.W.; Leon-Rico, D.; Ferreira, C.B.; Buckland, K.F.; Santilli, G.; Armant, M.A.; Schambach, A.; Cavazza, A.; Thrasher, A.J. Enhancing Lentiviral and Alpharetroviral Transduction of Human Hematopoietic Stem Cells for Clinical Application. Mol. Ther. Methods Clin. Dev. 2019, 14, 134–147. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.; Kim, Y.-S.; Wielgosz, M.M.; Ferrara, F.; Ma, Z.; Condori, J.; Palmer, L.E.; Zhao, X.; Kang, G.; Rawlings, D.J.; et al. Optimizing lentiviral vector transduction of hematopoietic stem cells for gene therapy. Gene Ther. 2020, 27, 545–556. [Google Scholar] [CrossRef]
- Davis, H.E.; Rosinski, M.; Morgan, J.R.; Yarmush, M.L. Charged Polymers Modulate Retrovirus Transduction via Membrane Charge Neutralization and Virus Aggregation. Biophys. J. 2004, 86, 1234–1242. [Google Scholar] [CrossRef]
- Wang, J.; Segatori, L.; Biswal, S.L. Probing the association of triblock copolymers with supported lipid membranes using microcantilevers. Soft Matter 2014, 10, 6417–6424. [Google Scholar] [CrossRef] [PubMed]
- Lutz, M.B.; Kukutsch, N.; Ogilvie, A.L.; Rossner, S.; Koch, F.; Romani, N.; Schuler, G. An advanced culture method for generating large quantities of highly pure dendritic cells from mouse bone marrow. J. Immunol. Methods 1999, 223, 77–92. [Google Scholar] [CrossRef]
- Zal, T.; Volkmann, A.; Stockinger, B. Mechanisms of tolerance induction in major histocompatibility complex class II-restricted T cells specific for a blood-borne self-antigen. J. Exp. Med. 1994, 180, 2089–2099. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, I.A.; Zinser, E.; Strasser, E.; Stein, M.F.; Dorrie, J.; Schaft, N.; Steinkasserer, A.; Knippertz, I. Leukoreduction system chambers are an efficient, valid, and economic source of functional monocyte-derived dendritic cells and lymphocytes. Immunobiology 2013, 218, 1392–1401. [Google Scholar] [CrossRef] [PubMed]
- He, T.C. Adenoviral vectors. Curr. Protoc. Hum. Genet. 2004, 40. [Google Scholar] [CrossRef] [PubMed]
- Rodenko, B.; Toebes, M.; Hadrup, S.R.; van Esch, W.J.; Molenaar, A.M.; Schumacher, T.N.; Ovaa, H. Generation of peptide-MHC class I complexes through UV-mediated ligand exchange. Nat. Protoc. 2006, 1, 1120–1132. [Google Scholar] [CrossRef] [PubMed]
- Gerer, K.F.; Hoyer, S.; Dorrie, J.; Schaft, N. Electroporation of mRNA as Universal Technology Platform to Transfect a Variety of Primary Cells with Antigens and Functional Proteins. Methods Mol. Biol. 2017, 1499, 165–178. [Google Scholar] [CrossRef] [PubMed]
- Bosch, N.C.; Voll, R.E.; Voskens, C.J.; Gross, S.; Seliger, B.; Schuler, G.; Schaft, N.; Dorrie, J. NF-kappaB activation triggers NK-cell stimulation by monocyte-derived dendritic cells. Ther. Adv. Med. Oncol. 2019, 11, 1758835919891622. [Google Scholar] [CrossRef]
- Zhao, J.; Zhao, S.; Ou, J.; Zhang, J.; Lan, W.; Guan, W.; Wu, X.; Yan, Y.; Zhao, W.; Wu, J.; et al. COVID-19: Coronavirus Vaccine Development Updates. Front. Immunol. 2020, 11, 602256. [Google Scholar] [CrossRef] [PubMed]
- Mercier, S.; Gahery-Segard, H.; Monteil, M.; Lengagne, R.; Guillet, J.G.; Eloit, M.; Denesvre, C. Distinct roles of adenovirus vector-transduced dendritic cells, myoblasts, and endothelial cells in mediating an immune response against a transgene product. J. Virol. 2002, 76, 2899–2911. [Google Scholar] [CrossRef]
- Kaygisiz, K.; Synatschke, C.V. Materials promoting viral gene delivery. Biomater. Sci. 2020, 8, 6113–6156. [Google Scholar] [CrossRef]
- Arcasoy, S.M.; Latoche, J.D.; Gondor, M.; Pitt, B.R.; Pilewski, J.M. Polycations increase the efficiency of adenovirus-mediated gene transfer to epithelial and endothelial cells In Vitro. Gene Ther. 1997, 4, 32–38. [Google Scholar] [CrossRef]
- Zhao, C.; Wu, N.; Deng, F.; Zhang, H.; Wang, N.; Zhang, W.; Chen, X.; Wen, S.; Zhang, J.; Yin, L.; et al. Adenovirus-mediated gene transfer in mesenchymal stem cells can be significantly enhanced by the cationic polymer polybrene. PLoS ONE 2014, 9, e92908. [Google Scholar] [CrossRef]
- Davis, H.E.; Morgan, J.R.; Yarmush, M.L. Polybrene increases retrovirus gene transfer efficiency by enhancing receptor-independent virus adsorption on target cell membranes. Biophys. Chem. 2002, 97, 159–172. [Google Scholar] [CrossRef]
- Hofig, I.; Atkinson, M.J.; Mall, S.; Krackhardt, A.M.; Thirion, C.; Anastasov, N. Poloxamer synperonic F108 improves cellular transduction with lentiviral vectors. J. Gene Med. 2012, 14, 549–560. [Google Scholar] [CrossRef]
- Simon, B.; Harrer, D.C.; Thirion, C.; Schuler-Thurner, B.; Schuler, G.; Uslu, U. Enhancing lentiviral transduction to generate melanoma-specific human T cells for cancer immunotherapy. J. Immunol. Methods 2019, 472, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Becker, M.; Cotena, A.; Gordon, S.; Platt, N. Expression of the class A macrophage scavenger receptor on specific subpopulations of murine dendritic cells limits their endotoxin response. Eur. J. Immunol. 2006, 36, 950–960. [Google Scholar] [CrossRef]
- Ulyanova, T.; Scott, L.M.; Priestley, G.V.; Jiang, Y.; Nakamoto, B.; Koni, P.A.; Papayannopoulou, T. VCAM-1 expression in adult hematopoietic and nonhematopoietic cells is controlled by tissue-inductive signals and reflects their developmental origin. Blood 2005, 106, 86–94. [Google Scholar] [CrossRef]
- Ansari, A.M.; Ahmed, A.K.; Matsangos, A.E.; Lay, F.; Born, L.J.; Marti, G.; Harmon, J.W.; Sun, Z. Cellular GFP Toxicity and Immunogenicity: Potential Confounders in In Vivo Cell Tracking Experiments. Stem. Cell Rev. Rep. 2016, 12, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Skelton, D.; Satake, N.; Kohn, D.B. The enhanced green fluorescent protein (eGFP) is minimally immunogenic in C57BL/6 mice. Gene Ther. 2001, 8, 1813–1814. [Google Scholar] [CrossRef] [PubMed]
- Gambotto, A.; Dworacki, G.; Cicinnati, V.; Kenniston, T.; Steitz, J.; Tuting, T.; Robbins, P.D.; DeLeo, A.B. Immunogenicity of enhanced green fluorescent protein (EGFP) in BALB/c mice: Identification of an H2-Kd-restricted CTL epitope. Gene Ther. 2000, 7, 2036–2040. [Google Scholar] [CrossRef]
- Stripecke, R.; Carmen Villacres, M.; Skelton, D.; Satake, N.; Halene, S.; Kohn, D. Immune response to green fluorescent protein: Implications for gene therapy. Gene Ther. 1999, 6, 1305–1312. [Google Scholar] [CrossRef] [PubMed]
- Re, F.; Srinivasan, R.; Igarashi, T.; Marincola, F.; Childs, R. Green fluorescent protein expression in dendritic cells enhances their immunogenicity and elicits specific cytotoxic T-cell responses in humans. Exp. Hematol. 2004, 32, 210–217. [Google Scholar] [CrossRef]
- Limberis, M.P.; Bell, C.L.; Wilson, J.M. Identification of the murine firefly luciferase-specific CD8 T-cell epitopes. Gene Ther. 2009, 16, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Mangeot, P.E.; Duperrier, K.; Negre, D.; Boson, B.; Rigal, D.; Cosset, F.L.; Darlix, J.L. High levels of transduction of human dendritic cells with optimized SIV vectors. Mol. Ther. 2002, 5, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Rouas, R.; Uch, R.; Cleuter, Y.; Jordier, F.; Bagnis, C.; Mannoni, P.; Lewalle, P.; Martiat, P.; Van den Broeke, A. Lentiviral-mediated gene delivery in human monocyte-derived dendritic cells: Optimized design and procedures for highly efficient transduction compatible with clinical constraints. Cancer Gene Ther. 2002, 9, 715–724. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Huang, S.; Endo, R.I.; Nemerow, G.R. Upregulation of integrins alpha v beta 3 and alpha v beta 5 on human monocytes and T lymphocytes facilitates adenovirus-mediated gene delivery. J. Virol. 1995, 69, 2257–2263. [Google Scholar] [CrossRef] [PubMed]
- Schneider, S.D.; Rusconi, S.; Seger, R.A.; Hossle, J.P. Adenovirus-mediated gene transfer into monocyte-derived macrophages of patients with X-linked chronic granulomatous disease: Ex vivo correction of deficient respiratory burst. Gene Ther. 1997, 4, 524–532. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Burke, B.; Sumner, S.; Maitland, N.; Lewis, C.E. Macrophages in gene therapy: Cellular delivery vehicles and In Vivo targets. J. Leukoc. Biol. 2002, 72, 417–428. [Google Scholar] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Strack, A.; Deinzer, A.; Thirion, C.; Schrödel, S.; Dörrie, J.; Sauerer, T.; Steinkasserer, A.; Knippertz, I. Breaking Entry-and Species Barriers: LentiBOOST® Plus Polybrene Enhances Transduction Efficacy of Dendritic Cells and Monocytes by Adenovirus 5. Viruses 2022, 14, 92. https://doi.org/10.3390/v14010092
Strack A, Deinzer A, Thirion C, Schrödel S, Dörrie J, Sauerer T, Steinkasserer A, Knippertz I. Breaking Entry-and Species Barriers: LentiBOOST® Plus Polybrene Enhances Transduction Efficacy of Dendritic Cells and Monocytes by Adenovirus 5. Viruses. 2022; 14(1):92. https://doi.org/10.3390/v14010092
Chicago/Turabian StyleStrack, Astrid, Andrea Deinzer, Christian Thirion, Silke Schrödel, Jan Dörrie, Tatjana Sauerer, Alexander Steinkasserer, and Ilka Knippertz. 2022. "Breaking Entry-and Species Barriers: LentiBOOST® Plus Polybrene Enhances Transduction Efficacy of Dendritic Cells and Monocytes by Adenovirus 5" Viruses 14, no. 1: 92. https://doi.org/10.3390/v14010092
APA StyleStrack, A., Deinzer, A., Thirion, C., Schrödel, S., Dörrie, J., Sauerer, T., Steinkasserer, A., & Knippertz, I. (2022). Breaking Entry-and Species Barriers: LentiBOOST® Plus Polybrene Enhances Transduction Efficacy of Dendritic Cells and Monocytes by Adenovirus 5. Viruses, 14(1), 92. https://doi.org/10.3390/v14010092





