Host Range of Carp Edema Virus (CEV) during a Natural Mortality Event in a Minnesota Lake and Update of CEV Associated Mortality Events in the USA
Abstract
1. Introduction
2. Materials and Methods
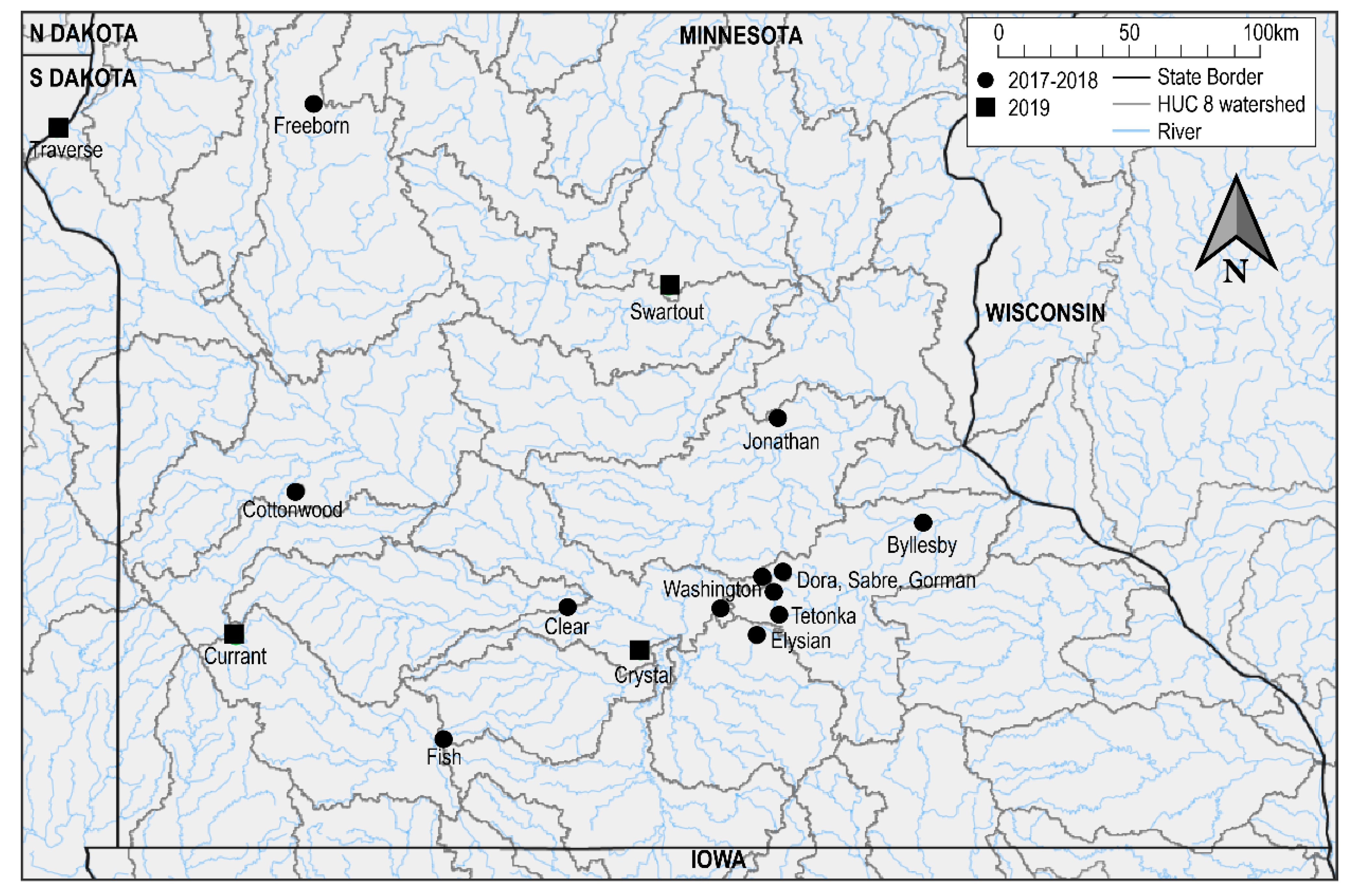
2.1. Mortality Event Investigation at Lake Swartout
2.2. Additional Mortality Event Investigations
2.3. Diagnostic Methods for CEV and KHV Detection
3. Results
3.1. Mortality Event Investigation on Lake Swartout
3.2. Investigation of CEV Species Specificity
3.3. Additional Mortality Events Investigations
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gozlan, R.E.; Peeler, E.J.; Longshaw, M.; St-Hilaire, S.; Feist, S.W. Effect of Microbial Pathogens on the Diversity of Aquatic Populations, Notably in Europe. Microbes Infect. 2006, 8, 1358–1364. [Google Scholar] [CrossRef]
- Peeler, E.J.; Oidtmann, B.C.; Midtlyng, P.J.; Miossec, L.; Gozlan, R.E. Non-Native Aquatic Animals Introductions Have Driven Disease Emergence in Europe. Biol. Invasions 2011, 13, 1291–1303. [Google Scholar] [CrossRef]
- Spikmans, F.; Lemmers, P.; op den Camp, H.J.M.; van Haren, E.; Kappen, F.; Blaakmeer, A.; van der Velde, G.; van Langevelde, F.; Leuven, R.S.E.W.; van Alen, T.A. Impact of the Invasive Alien Topmouth Gudgeon (Pseudorasbora Parva) and Its Associated Parasite Sphaerothecum Destruens on Native Fish Species. Biol. Invasions 2020, 22, 587–601. [Google Scholar] [CrossRef]
- Rodgers, C.J.; Mohan, C.V.; Peeler, E.J. The Spread of Pathogens through Trade in Aquatic Animals and Their Products. Rev. Sci. Tech. De L’oie 2011, 30, 241–256. [Google Scholar] [CrossRef]
- Whittington, R.J.; Chong, R. Global Trade in Ornamental Fish from an Australian Perspective: The Case for Revised Import Risk Analysis and Management Strategies. Prev. Vet. Med. 2007, 81, 92–116. [Google Scholar] [CrossRef] [PubMed]
- Girisha, S.K.; Kushala, K.B.; Nithin, M.S.; Puneeth, T.G.; Naveen Kumar, B.T.; Vinay, T.N.; Suresh, T.; Ajay, S.K.; Venugopal, M.N.; Ramesh, K.S. First Report of the Infectious Spleen and Kidney Necrosis Virus (ISKNV) Infection in Ornamental Fishes in India. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef]
- Phelps, N.B.D.; Armién, A.G.; Mor, S.K.; Goyal, S.M.; Warg, J.V.; Bhagyam, R.; Monahan, T. Spring Viremia of Carp Virus in Minnehaha Creek, Minnesota. J. Aquat. Anim. Health 2012, 24, 232–237. [Google Scholar] [CrossRef]
- Lovy, J.; Friend, S.; Al-Hussinee, L.; Waltzek, T. First Report of Carp Edema Virus in the Mortality of Wild Common Carp Cyprinus Carpio in North America. Dis. Aquat. Org. 2018, 131, 177–186. [Google Scholar] [CrossRef]
- Padhi, S.K.; Tolo, I.; McEachran, M.; Primus, A.; Mor, S.K.; Phelps, N.B.D. Koi Herpesvirus and Carp Oedema Virus: Infections and Coinfections during Mortality Events of Wild Common Carp in the United States. J. Fish Dis. 2019, 42, 1609–1621. [Google Scholar] [CrossRef]
- Thresher, R.E.; Allman, J.; Stremick-Thompson, L. Impacts of an Invasive Virus (CyHV-3) on Established Invasive Populations of Common Carp (Cyprinus Carpio) in North America. Biol. Invasions 2018, 20, 1703–1718. [Google Scholar] [CrossRef]
- Klein, Z.B.; Quist, M.C.; Miranda, L.E.; Marron, M.M.; Steuck, M.J.; Hansen, K.A. Commercial Fisheries of the Upper Mississippi River: A Century of Sustained Harvest. Fisheries 2018, 43, 563–574. [Google Scholar] [CrossRef]
- Rehman, T.; Yin, L.; Latif, M.B.; Zhou, Y.; Wang, K.; Geng, Y.; Huang, X.; Chen, D.; Fang, J.; Chen, Z.; et al. Current Findings on Carp Edema Virus, Control Challenges, and Future Outlook. Aquac. Int. 2020, 28, 2015–2026. [Google Scholar] [CrossRef]
- Matras, M.; Stachnik, M.; Borzym, E.; Maj-Paluch, J.; Reichert, M. Potential Vector Species of Carp Edema Virus (CEV). J. Fish Dis. 2019. [Google Scholar] [CrossRef] [PubMed]
- Adamek, M.; Oschilewski, A.; Wohlsein, P.; Jung-Schroers, V.; Teitge, F.; Dawson, A.; Gela, D.; Piackova, V.; Kocour, M.; Adamek, J.; et al. Experimental Infections of Different Carp Strains with the Carp Edema Virus (CEV) Give Insights into the Infection Biology of the Virus and Indicate Possible Solutions to Problems Caused by Koi Sleepy Disease (KSD) in Carp Aquaculture. Vet. Res. 2017, 48. [Google Scholar] [CrossRef]
- Adamek, M.; Teitge, F.; Steinhagen, D. Quantitative Diagnostics of Gill Diseases in Common Carp: Not as Simple as It Seems. Dis. Aquat. Org. 2019, 134, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, P.; Yang, R.; Chen, J.; Wang, K.; Geng, Y.; Lai, W.; Huang, X.; Chen, D.; Fang, J.; Chen, Z.; et al. First Detection of Carp Edema Virus in Association with Cyprinid Herpesvirus 3 in Cultured Ornamental Koi, Cyprinus Carpio L., in China. Aquaculture 2018, 490, 162–168. [Google Scholar] [CrossRef]
- Toffan, A.; Marsella, A.; Abbadi, M.; Abass, S.; Al-Adhadh, B.; Wood, G.; Stone, D.M. First Detection of Koi Herpesvirus and Carp Oedema Virus in Iraq Associated with a Mass Mortality in Common Carp (Cyprinus Carpio). Transbound. Emerg. Dis. 2020, 67, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.W.; Giri, S.S.; Kim, S.G.; Kwon, J.; Oh, W.T.; Park, S.C. Carp Edema Virus and Cyprinid Herpesvirus-3 Coinfection Is Associated with Mass Mortality of Koi (Cyprinus Carpio Haematopterus) in the Republic of Korea. Pathogens 2020, 9, 222. [Google Scholar] [CrossRef] [PubMed]
- Epitools—Epidemiological Calculators. Available online: https://epitools.ausvet.com.au/ (accessed on 17 February 2021).
- Zida, S.; Kolia-Diafouka, P.; Kania, D.; Sotto, A.; Foulongne, V.; Bolloré, K.; Ouangraoua, S.; Méda, N.; Carrère-Kremer, S.; Van de Perre, P.; et al. Combined Testing for Herpes Simplex Virus and Mycobacterium Tuberculosis DNA in Cerebrospinal Fluid of Patients with Aseptic Meningitis in Burkina Faso, West Africa. J. Clin. Lab. Anal. 2019, 33, e22719. [Google Scholar] [CrossRef]
- Gilad, O.; Yun, S.; Zagmutt-Vergara, F.; Leutenegger, C.; Bercovier, H.; Hedrick, R. Concentrations of a Koi Herpesvirus (KHV) in Tissues of Experimentally-Infected Cyprinus Carpio Koi as Assessed by Real-Time TaqMan PCR. Dis. Aquat. Org. 2004, 60, 179–187. [Google Scholar] [CrossRef]
- Matras, M.; Borzym, E.; Stone, D.; Way, K.; Stachnik, M.; Maj-Paluch, J.; Palusińska, M.; Reichert, M. Carp Edema Virus in Polish Aquaculture—Evidence of Significant Sequence Divergence and a New Lineage in Common Carp Cyprinus Carpio (L.). J. Fish Dis. 2017, 40, 319–325. [Google Scholar] [CrossRef]
- Tan, M.; Armbruster, J.W. Phylogenetic Classification of Extant Genera of Fishes of the Order Cypriniformes (Teleostei: Ostariophysi). Zootaxa 2018, 4476, 6. [Google Scholar] [CrossRef]
- Hirt, M.V.; Arratia, G.; Chen, W.-J.; Mayden, R.L.; Tang, K.L.; Wood, R.M.; Simons, A.M. Effects of Gene Choice, Base Composition and Rate Heterogeneity on Inference and Estimates of Divergence Times in Cypriniform Fishes. Biol. J. Linn. Soc. 2017, 121, 319–339. [Google Scholar] [CrossRef]
- McAllister, P.E.; Owens, W.J. Recovery of Infectious Pancreatic Necrosis Virus from the Faeces of Wild Piscivorous Birds. Aquaculture 1992, 106, 227–232. [Google Scholar] [CrossRef]
- Willumsen, B. Birds and Wild Fish as Potential Vectors of Yersinia ruckeri. J. Fish Dis. 1989, 12, 275–277. [Google Scholar] [CrossRef]
- Taylor, P.W. Fish-Eating Birds as Potential Vectors of Edwardsiella ictaluri. J. Aquat. Anim. Health 1992, 4, 240–243. [Google Scholar] [CrossRef]
- Torres-Meza, O.A.; Loza-Rubio, E.; Martínez-Maya, J.J.; García-Espinosa, G. The First Detection of Koi Herpesvirus (Cy HV 3) in Migratory Wild Ducks in North America. J. Aquat. Anim. Health 2020, 32, 28–31. [Google Scholar] [CrossRef]
- McColl, K.A.; Sunarto, A.; Neave, M.J. Biocontrol of Carp: More than Just a Herpesvirus. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]

| Date | Water Temp (°C) | Species | No. Individuals (No.Pools) | Disposition, Pooling | Clinical Signs | Tissue Type | CEV Detection | CEV Avg Log Copy No. (SD) |
|---|---|---|---|---|---|---|---|---|
| 24-Jun | 24 | C. carpio (adult) | 5 | live, no pooling | gill lesions | brain | 5/5 | 4.84 (0.62) |
| gill | 5/5 | 7.24 (0.22) | ||||||
| kidney | 5/5 | 6.12 (0.04) | ||||||
| 28-Jun | 26 | C. carpio (adult) | 2 | live, no pooling | gill lesions | brain | 2/2 | 3.89 (0.09) |
| gill | 2/2 | 4.86 (0.05) | ||||||
| kidney | 1/2 | 3.99 | ||||||
| C. carpio (juvenile) | 45 (9) | dead, pools of 5 individuals | skin lesions | whole fish | 9/9 | 7.22 (0.16) | ||
| P. promelas | 3 (1) | dead, pool of 3 individuals | none | whole fish | 0/1 | negative | ||
| L. pipiens | 3 (1) | dead, pool of 3 individuals | none | whole tadpole | 0/1 | negative | ||
| P. auritus | 5 (1) | feces, pool of 5 | na | feces | 1/1 | 4.39 | ||
| regurgitant, pool of 5 | na | regurgitant | 1/1 | 4.67 | ||||
| 2-Jul | 28 | C. carpio (juvenile) | 33 (11) | live, pool of 3 individuals | skin lesions | whole fish | 11/11 | 5.77 (0.74) |
| C. carpio (juvenile) | 21 (7) | live, pool of 3 individuals | none | brain | 6/7 | 4.43 (0.47) | ||
| gill | 7/7 | 4.61 (0.90) | ||||||
| kidney | 6/7 | 3.91 (0.25) | ||||||
| P. promelas | 36 (12) | live, pool of 3 individuals | none | whole fish | 0/12 | negative | ||
| P. promelas | 33 (11) | live, pool of 3 individuals | none | brain | 0/11 | negative | ||
| gill | 0/11 | negative | ||||||
| kidney | 0/11 | negative | ||||||
| L. pipiens | 21 (7) | live, pool of 3 individuals | none | brain | 0/7 | negative | ||
| gill | 0/7 | negative | ||||||
| kidney | 0/7 | negative | ||||||
| C. inconstans | 4 (1) | live, pool of 4 individuals | none | whole fish | 0/1 | negative | ||
| L. cyanellus | 2 (1) | live, pool of 2 individuals | none | whole fish | 0/1 | negative | ||
| L. gibbosus | 54 (18) | live, pool of 3 individuals | none | brain | 0/18 | negative | ||
| gill | 0/18 | negative | ||||||
| kidney | 0/18 | negative | ||||||
| 5-Jul | 27 | P. auritus | 36 (12) | feces, pool of 3 | na | feces | 0/12 | negative |
| regurgitant, pool of 3 | na | regurgitant | 3/12 | 5.77 (0.64) | ||||
| 10-Jul | 27 | P. auritus | 36 (12) | feces, pool of 3 | na | feces | 0/12 | negative |
| regurgitant, pool of 3 | na | regurgitant | 1/12 | 3.53 |
| Location | Carp Sampling | Virus Load | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lake Name (County) | Lake Size (ha) | Mortality Event History | Sampling Date | Water Temp (°C) | Estimated Mortality | Disposition (Condition) | Size (cm) | No. Carp | CEV Avg Log Copy No. (SD) | KHV Avg Log Copy No. (SD) | ||||
| Brain | Gill | Kidney | Brain | Gill | Kidney | |||||||||
| Pymatuning (Crawford) | 6915 | KHV, 2017 | 30-April | 11 | few | live (good) | nr | 5 | not tested | 5.53 (0.20) | negative | not tested | negative | negative |
| Currant (Murray) | 164 | KHV + CEV, 2018 | 4-June | 20 | hundreds | live (fair) | 45–48 | 2 | 5.03 (0.42) | 5.64 (0.40) | 5.00 (0.07) | 3.65 (0.31) | negative | negative |
| Crystal (Blue Earth) | 13 | none | 7-June | 25 | hundreds | dead (poor) | nr | 2 | 3.46 | 4.40 | 3.37 (0.20) | negative | negative | negative |
| Traverse (Traverse) | 4390 | none | 12-June | 18 | hundreds | dead (poor) | 50–70 | 2 | negative | 4.01 (0.09) | negative | negative | negative | negative |
| Swartout (Wright) | 12 | none | 24-June | 24 | 405 a | live (excellent) | 63–70 | 5 | 4.84 (0.62) | 7.24 (0.22) | 6.12 (0.04) | negative | negative | negative |
| Primer Name | Target | Primer Sequence (5′-3′) | References | Target Gene (bp-Length) |
|---|---|---|---|---|
| KHV-86f | KHV | GAC-GCC-GGA-GAC-CTT-GTG | [21] | ORF 89 (78) |
| KHV-163r | CGG-GTT-GTT-ATT-TTT-GTC-CTT-GTT | |||
| KHV-109p | [TAMRA] CTT-CCT-CTG-CTC-GGC-GAG-CAC-G-[IBRQ] | |||
| CEFAS_qF | CEV | AGT-TTT-GTA-KAT-TGT-AGC-ATT-TCC | [22] | p4a (76) |
| CEFAS_qR | GAT-TCC-TCA-AGG-AGT-TDC-AGT-AAA | |||
| CEV qProbe1 | [FAM]-AGA-GTT-TGT-TTC-TTG-CCA-TAC-AAA-CT-[BHQ1] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tolo, I.E.; K. Padhi, S.; Hundt, P.J.; Bajer, P.G.; K. Mor, S.; Phelps, N.B.D. Host Range of Carp Edema Virus (CEV) during a Natural Mortality Event in a Minnesota Lake and Update of CEV Associated Mortality Events in the USA. Viruses 2021, 13, 400. https://doi.org/10.3390/v13030400
Tolo IE, K. Padhi S, Hundt PJ, Bajer PG, K. Mor S, Phelps NBD. Host Range of Carp Edema Virus (CEV) during a Natural Mortality Event in a Minnesota Lake and Update of CEV Associated Mortality Events in the USA. Viruses. 2021; 13(3):400. https://doi.org/10.3390/v13030400
Chicago/Turabian StyleTolo, Isaiah E., Soumesh K. Padhi, Peter J. Hundt, Przemyslaw G. Bajer, Sunil K. Mor, and Nicholas B. D. Phelps. 2021. "Host Range of Carp Edema Virus (CEV) during a Natural Mortality Event in a Minnesota Lake and Update of CEV Associated Mortality Events in the USA" Viruses 13, no. 3: 400. https://doi.org/10.3390/v13030400
APA StyleTolo, I. E., K. Padhi, S., Hundt, P. J., Bajer, P. G., K. Mor, S., & Phelps, N. B. D. (2021). Host Range of Carp Edema Virus (CEV) during a Natural Mortality Event in a Minnesota Lake and Update of CEV Associated Mortality Events in the USA. Viruses, 13(3), 400. https://doi.org/10.3390/v13030400







