Mental Health and Substance Use Associated with Hospitalization among People with COVID-19: A Population-Based Cohort Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Comorbidities
2.3. Statistical Analysis
3. Results
3.1. Demographic and Clinical Characteristics
3.2. Chronic Comorbidities as Risk Factors
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Disclaimer
References
- Phua, J.; Weng, L.; Ling, L.; Egi, M.; Lim, C.M.; Divatia, J.V.; Shrestha, B.R.; Arabi, Y.M.; Ng, J.; Gomersall, C.D.; et al. Intensive care management of coronavirus disease 2019 (COVID-19): Challenges and recommendations. Lancet Respir. Med. 2020, 8, 506–517. [Google Scholar] [CrossRef]
- Holman, N.; Knighton, P.; Kar, P.; O’Keefe, J.; Curley, M.; Weaver, A.; Barron, M.; Bakhai, C.; Khunti, K.; Wareham, N.J.; et al. Risk factors for COVID-19-related mortality in people with type 1 and type 2 diabetes in England: A population-based cohort study. Lancet Diabetes Endocrinol. 2020, 8, 823–833. [Google Scholar] [CrossRef]
- Carenzo, L.; Costantini, E.; Greco, M.; Barra, F.L.; Rendiniello, V.; Mainetti, M.; Bui, R.; Zanella, A.; Grasselli, G.; Lagioia, M.; et al. Hospital surge capacity in a tertiary emergency referral centre during the COVID-19 outbreak in Italy. Anaesthesia 2020, 75, 928–934. [Google Scholar] [CrossRef] [PubMed]
- Himmels, J.P.W.; Borge, T.C.; Brurberg, K.G.; Gravningen, K.M.; Feruglio, S.L.; Berild, J.D. COVID-19 and Risk Factors for Hospital Admission, Severe Disease and Death 3rd Update. Norwegian Institute of Public Health. 15 November 2020. Available online: https://www.fhi.no/en/publ/2020/COVID19-and-risk-factors-for-hospital-admission-severe-disease-and-death-3rd_update/ (accessed on 18 May 2021).
- Wang, Q.Q.; Kaelber, D.C.; Xu, R.; Volkow, N.D. COVID-19 risk and outcomes in patients with substance use disorders: Analyses from electronic health records in the United States. Mol. Psychiatry 2021, 26, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Baillargeon, J.; Polychronopoulou, E.; Kuo, Y.F.; Raji, M.A. The impact of substance use disorder on COVID-19 outcomes. Psychiatr. Serv. 2021, 72, 578–581. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.Y.; Danielson, M.L.; Town, M.; Derado, G.; Greenlund, K.J.; Kirley, P.D.; Alden, N.B.; Yousey-Hindes, K.; Anderson, E.J.; Ryan, P.A.; et al. Risk Factors for Coronavirus Disease 2019 (COVID-19)–Associated Hospitalization: COVID-19–Associated Hospitalization Surveillance Network and Behavioral Risk Factor Surveillance System. Clin. Infect. Dis. 2020, 72, e695–e703. [Google Scholar] [CrossRef]
- Owusu, D.; Kim, L.; O’Halloran, A.; Whitaker, M.; Piasecki, A.M.; Reingold, A.; Alden, N.B.; Maslar, A.; Anderson, E.J.; Ryan, P.A.; et al. Characteristics of Adults aged 18–49 Years without Underlying Conditions Hospitalized with Laboratory-Confirmed COVID-19 in the United States, COVID-NET—March–August 2020. Clin. Infect. Dis. 2020, 72, e162–e166. [Google Scholar] [CrossRef]
- Sheikh, A.; McMenamin, J.; Taylor, B.; Robertson, C. SARS-CoV-2 Delta VOC in Scotland: Demographics, risk of hospital admission, and vaccine effectiveness. Lancet 2021, 397, 2461–2462. [Google Scholar] [CrossRef]
- Zou, G. A Modified Poisson Regression Approach to Prospective Studies with Binary Data. Am. J. Epidemiol. 2004, 159, 702–706. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. 2020. Available online: https://www.R-project.org/ (accessed on 18 May 2021).
- Sundaram, M.E.; Calzavara, A.; Mishra, S.; Kustra, R.; Chan, A.K.; Hamilton, M.A.; Djebli, M.; Rosella, L.C.; Watson, T.; Chen, H.; et al. Individual and social determinants of SARS-CoV-2 testing and positivity in Ontario, Canada: A population-wide study. CMAJ 2021, 193, E723–E734. [Google Scholar] [CrossRef]
- Manitoba Centre for Health Policy. Concept: Intellectual Disability (ID) (Mental Retardation)/Developmental Disability (DD)/Developmental Disorders. University of Manitoba, 9 July 2020. Available online: http://mchp-appserv.cpe.umanitoba.ca/viewConcept.php?conceptID=1365 (accessed on 8 February 2021).
- Janjua, N.Z.; Islam, N.; Kuo, M.; Yu, A.; Wong, S.; Butt, Z.A.; Gilbert, M.; Buxton, J.; Chapinal, N.; Samji, H.; et al. Identifying injection drug use and estimating population size of people who inject drugs using healthcare administrative datasets. Int. J. Drug Policy 2018, 55, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Janjua, N.Z.; Kuo, M.; Yu, A.; Alvarez, M.; Wong, S.; Cook, D.; Wong, J.; Grebely, J.; Butt, Z.A.; Samji, H.; et al. The Population Level Cascade of Care for Hepatitis C in British Columbia, Canada: The BC Hepatitis Testers Cohort (BC-HTC). EBioMedicine 2016, 12, 189–195. [Google Scholar] [CrossRef] [Green Version]
- Vai, B.; Mazza, M.G.; Delli Colli, C.; Foiselle, M.; Allen, B.; Benedetti, F.; Borsini, A.; Dias, M.C.; Tamouza, R.; Leboyer, M.; et al. Mental disorders and risk of COVID-19-related mortality, hospitalisation, and intensive care unit admission: A systematic review and meta-analysis. Lancet Psychiatry 2021, 8, 797–812. [Google Scholar] [CrossRef]
- Government of British Columbia. COVID-19 Immunization Plan. Available online: https://www2.gov.bc.ca/gov/content/covid-19/vaccine/plan (accessed on 27 June 2021).
- Atkins, J.L.; Masoli, J.A.H.; Delgado, J.; Pilling, L.C.; Kuo, C.L.; Kuchel, G.A.; Melzer, D. Preexisting Comorbidities Predicting COVID-19 and Mortality in the UK Biobank Community Cohort. J. Gerontol.—Ser. A Biol. Sci. Med. Sci. 2020, 75, 2224–2230. [Google Scholar] [CrossRef] [PubMed]
- Galbadage, T.; Peterson, B.M.; Awada, J.; Buck, A.S.; Ramirez, D.A.; Wilson, J.; Gunasekera, R.S. Systematic Review and Meta-Analysis of Sex-Specific COVID-19 Clinical Outcomes. Front. Med. 2020, 7, 348. [Google Scholar] [CrossRef]
- O’Brien, J.; Du, K.Y.; Peng, C. Incidence, clinical features, and outcomes of COVID-19 in Canada: Impact of sex and age. J. Ovarian Res. 2020, 13, 137. [Google Scholar] [CrossRef] [PubMed]
- Petrilli, C.M.; Jones, S.A.; Yang, J.; Rajagopalan, H.; O’Donnell, L.; Chernyak, Y.; Tobin, K.A.; Cerfolio, R.J.; Francois, F.; Horwitz, L.I. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: Prospective cohort study. BMJ 2020, 369, m1966. [Google Scholar] [CrossRef]
- Van Gerwen, M.; Alsen, M.; Little, C.; Barlow, J.; Genden, E.; Naymagon, L.; Tremblay, D. Risk factors and outcomes of COVID-19 in New York City; A retrospective cohort study. J. Med. Virol. 2021, 93, 907–915. [Google Scholar] [CrossRef]
- Booth, A.; Reed, A.B.; Ponzo, S.; Yassaee, A.; Aral, M.; Plans, D.; Labrique, A.; Mohan, D. Population risk factors for severe disease and mortality in COVID-19: A global systematic review and meta-analysis. PLoS ONE 2021, 16, e0247461. [Google Scholar] [CrossRef]
- Haitao, T.; Vermunt, J.V.; Abeykoon, J.; Ghamrawi, R.; Gunaratne, M.; Jayachandran, M.; Narang, K.; Parashuram, S.; Suvakov, S.; Garovic, V.D. COVID-19 and Sex Differences: Mechanisms and Biomarkers. Mayo Clin. Proc. 2020, 95, 2189–2203. [Google Scholar] [CrossRef]
- Takahashi, T.; Ellingson, M.K.; Wong, P.; Israelow, B.; Lucas, C.; Klein, J.; Silva, J.; Mao, T.; Oh, J.E.; Tokuyama, M.; et al. Sex differences in immune responses that underlie COVID-19 disease outcomes. Nature 2020, 588, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.L.; Pekosz, A.; Park, H.S.; Ursin, R.L.; Shapiro, J.R.; Benner, S.E.; Littlefield, K.; Kumar, S.; Naik, H.M.; Betenbaugh, M.J.; et al. Sex, age, and hospitalization drive antibody responses in a COVID-19 convalescent plasma donor population. J. Clin. Investig. 2020, 130, 6141–6150. [Google Scholar] [CrossRef]
- Zambrano, L.D.; Ellington, S.; Strid, P.; Galang, R.R.; Oduyebo, T.; Tong, V.T.; Woodworth, K.R.; Nahabedian, J.F., III; Azziz-Baumgartner, E.; Gilboa, M.; et al. Update: Characteristics of Symptomatic Women of Reproductive Age with Laboratory-Confirmed SARS-CoV-2 Infection by Pregnancy Status—United States, January 22–October 3, 2020. Morb. Mortal. Wkly. Rep. 2020, 69, 1641–1647. [Google Scholar] [CrossRef] [PubMed]
- Badr, D.A.; Mattern, J.; Carlin, A.; Cordier, A.G.; Maillart, E.; El Hachem, L.; El Kenz, H.; Andronikof, M.; De Bels, D.; Damoisel, C.; et al. Are clinical outcomes worse for pregnant women at ≥20 weeks’ gestation infected with coronavirus disease 2019? A multicenter case-control study with propensity score matching. Am. J. Obstet. Gynecol. 2020, 223, 764–768. [Google Scholar] [CrossRef] [PubMed]
- Oakes, M.C.; Kernberg, A.S.; Carter, E.B.; Foeller, M.E.; Palanisamy, A.; Raghuraman, N.; Kelly, J.C. Pregnancy as a risk factor for severe coronavirus 2019 (COVID-19) disease using standardized clinical criteria. Am. J. Obstet. Gynecol. MFM 2021, 3, 100319. [Google Scholar] [CrossRef] [PubMed]
- Allotey, J.; Stallings, E.; Bonet, M.; Yap, M.; Chatterjee, S.; Kew, T.; Debenham, L.; Llavall, A.C.; Dixit, A.; Zhou, D.; et al. Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: Living systematic review and meta-analysis. BMJ 2020, 370, 3320. [Google Scholar] [CrossRef]
- BC Centre for Disease Control. Overdose Response Indicators. Available online: http://www.bccdc.ca/health-professionals/data-reports/overdose-response-indicators (accessed on 6 July 2021).
- British Columbia Coroner’s Service. Llicit Drug Toxicity Deaths in BC: January 1, 2011–March 31, 2021. Minister of Public Safety and Solicitor General, Burnaby, BC. 29 April 2021. Available online: https://www2.gov.bc.ca/assets/gov/birth-adoption-death-marriage-and-divorce/deaths/coroners-service/statistical/illicit-drug.pdf (accessed on 6 July 2021).
- BC Centre for Disease Control. Phases of COVID-19 Testing in BC. 2021. Available online: http://www.bccdc.ca/health-info/diseases-conditions/covid-19/testing/phases-of-covid-19-testing-in-bc (accessed on 6 July 2021).
- Yanes-Lane, M.; Winters, N.; Fregonese, F.; Bastos, M.; Perlman-Arrow, S.; Campbell, J.R.; Menzies, D. Proportion of asymptomatic infection among COVID-19 positive persons and their transmission potential: A systematic review and meta-analysis. PLoS ONE 2020, 15, e0241536. [Google Scholar] [CrossRef]
| Variable | Category | Total * n = 56,874 | Non-Hospitalized ⁰ n = 54,576 | Hospitalized ⁰ n = 2298 | Hospitalized Row % | Crude Incidence Rate Ratio (95% Conf. Interv.) | p† |
|---|---|---|---|---|---|---|---|
| Sex | Female | 27,769 (48.8%) | 26,816 (49.1%) | 953 (41.5%) | 3.4% | Reference | <0.0001 |
| Male | 29,105 (51.2%) | 27,760 (50.9%) | 1345 (58.5%) | 4.6% | 1.35 (1.24–1.46) | ||
| Age (years) | N/A | 36 (28) ⁱ | 35 (26) ⁱ | 66 (25) ⁱ | - | 1.07 (1.06–1.07) | <0.0001 |
| Age group ‡ | <20 years | 7683 (13.5%) | 7665 (14.0%) | 18 (0.8%) | 0.2% | 0.45 (0.27–0.77) | 0.0031 |
| 20–29 years | 13,490 (23.7%) | 13,421 (24.6%) | 69 (3.0%) | 0.5% | Reference | - | |
| 30–39 years | 10,681 (18.8%) | 10,507 (19.3%) | 174 (7.6%) | 1.6% | 3.18 (2.41–4.20) | <0.0001 | |
| 40–49 years | 8818 (15.8%) | 8604 (15.8%) | 214 (9.3%) | 2.4% | 4.74 (3.62–6.22) | <0.0001 | |
| 50–59 years | 7565 (13.3%) | 7201 (13.2%) | 364 (15.8%) | 4.8% | 9.41 (7.28–12.15) | <0.0001 | |
| 60–69 years | 4793 (8.4%) | 4330 (7.9%) | 463 (20.1%) | 9.7% | 18.89 (14.70–24.27) | <0.0001 | |
| 70–79 years | 2425 (4.3%) | 1915 (3.5%) | 510 (22.2%) | 21.0% | 41.12 (32.10–52.67) | <0.0001 | |
| 80+ years | 1419 (2.5%) | 933 (1.7%) | 486 (21.1%) | 34.2% | 66.96 (52.35–85.65) | <0.0001 | |
| Pregnant (female population, ages 15 to 49; n = 17,693) | No | 16,638 (94.0%) | 16,457 (94.1%) | 181 (86.6%) | 1.1% | Reference | - |
| Yes | 398 (2.2%) | 388 (2.2%) | 10 (4.8%) | 2.5% | 2.31 (1.23–4.33) | 0.0091 | |
| Unknown | 657 (3.7%) | 639 (3.7%) | 18 (8.6%) | 2.7% | 2.52 (1.56–4.06) | 0.0002 | |
| Angina | No | 55,915 (98.3%) | 53,850 (98.7%) | 2065 (89.9%) | 3.7% | Reference | <0.0001 |
| Yes | 959 (1.7%) | 726 (1.3%) | 233 (10.1%) | 24.3% | 6.58 (5.84–7.41) | ||
| Chronic heart disease 1 | No | 53,840 (94.7%) | 52,161 (95.6%) | 1679 (73.1%) | 3.1% | Reference | <0.0001 |
| Yes | 3034 (5.3%) | 2415 (4.4%) | 619 (26.9%) | 20.4% | 6.54 (6.01–7.12) | ||
| Heart failure | No | 56,003 (98.5%) | 53,975 (98.9%) | 2028 (88.3%) | 3.6% | Reference | <0.0001 |
| Yes | 871 (1.5%) | 601 (1.1%) | 270 (11.7%) | 31.0% | 8.56 (7.68–8.54) | ||
| Hypertension | No | 48,009 (84.4%) | 46,955 (86.0%) | 1054 (45.9%) | 2.2% | Reference | <0.0001 |
| Yes | 8865 (15.6%) | 7621 (14.0%) | 1244 (54.1%) | 14.0% | 6.39 (5.91–6.92) | ||
| Ischemic heart disease | No | 54,139 (95.2%) | 52,378 (96.0%) | 1761 (76.6%) | 3.3% | Reference | <0.0001 |
| Yes | 2735 (4.8%) | 2198 (4.0%) | 537 (23.4%) | 19.6% | 6.04 (5.52–6.59) | ||
| Myocardial infarct | No | 56,273 (98.9%) | 54,109 (99.1%) | 2164 (94.2%) | 3.8% | Reference | <0.0001 |
| Yes | 601 (1.1%) | 467 (0.9%) | 134 (5.8%) | 22.3% | 5.80 (4.97–6.77) | ||
| Immunosuppression 2 | No | 55,541 (97.7%) | 53,380 (97.8%) | 2161 (94.0%) | 3.9% | Reference | <0.0001 |
| Yes | 1333 (2.3%) | 1196 (2.2%) | 137 (6.0%) | 10.3% | 2.64 (2.24–3.11) | ||
| Rheumatoid arthritis | No | 56,198 (98.8%) | 53,998 (98.9%) | 2200 (95.7%) | 3.9% | Reference | <0.0001 |
| Yes | 676 (1.2%) | 578 (1.1%) | 98 (4.3%) | 14.5% | 3.70 (3.07–4.47) | ||
| Depression | No | 44,541 (78.3%) | 43,124 (79.0%) | 1417 (61.7%) | 3.2% | Reference | <0.0001 |
| Yes | 12,333 (21.7%) | 11,452 (21.0%) | 881 (38.3%) | 7.1% | 2.25 (2.07–2.44) | ||
| Intellectual and developmental disability 3 | No | 56,444 (99.2%) | 54,177 (99.3%) | 2267 (98.7%) | 4.0% | Reference | 0.0008 |
| Yes | 430 (0.8%) | 399 (0.7%) | 31 (1.3%) | 7.2% | 1.79 (1.28–2.53) | ||
| Mood and anxiety disorders | No | 41,590 (73.1%) | 40,322 (73.9%) | 1268 (55.2%) | 3.0% | Reference | <0.0001 |
| Yes | 15,284 (26.9%) | 14,254 (26.1%) | 1030 (44.8%) | 6.7% | 2.21 (2.04–2.39) | ||
| Schizophrenia and psychotic disorders | No | 56,112 (98.7%) | 53,927 (98.8%) | 2185 (95.1%) | 3.9% | Reference | <0.0001 |
| Yes | 762 (1.3%) | 649 (1.2%) | 113 (4.9%) | 14.8% | 3.81 (3.20–4.53) | ||
| Diabetes | No | 51,487 (90.5%) | 50,011 (91.6%) | 1476 (64.2%) | 2.9% | Reference | - |
| Non-insulin | 5141 (9.1%) | 4541 (8.3%) | 600 (26.1%) | 11.7% | 4.07 (3.72–4.46) | <0.0001 | |
| Insulin 5 | 246 (0.4%) | 24 (0.0%) | 222 (9.7) | 90.2% | 31.48 (29.50–33.59) | <0.0001 | |
| Gout | No | 55,707 (97.9%) | 53,622 (98.3%) | 2085 (90.7%) | 3.7% | Reference | <0.0001 |
| Yes | 1167 (2.1%) | 964 (1.7%) | 213 (9.3%) | 18.3% | 4.88 (4.29–5.55) | ||
| Chronic kidney disease 4 | No | 54,686 (96.2%) | 52,928 (97.0%) | 1758 (76.5%) | 3.2% | Reference | <0.0001 |
| Yes | 2188 (3.8%) | 1648 (3.0%) | 540 (23.5%) | 24.7% | 7.68 (7.04–8.37) | ||
| Osteoarthritis | No | 52,645 (92.6%) | 50,975 (93.4%) | 1670 (72.7%) | 3.2% | Reference | <0.0001 |
| Yes | 4229 (7.4%) | 3601 (6.6%) | 628 (27.3%) | 14.8% | 4.68 (4.29–5.10) | ||
| Osteoporosis | No | 55,831 (98.2%) | 53,771 (98.5%) | 2060 (89.6%) | 3.7% | Reference | <0.0001 |
| Yes | 1043 (1.8%) | 805 (1.5%) | 238 (10.4%) | 22.8% | 6.18 (5.49–6.97) | ||
| Alzheimer/dementia | No | 56,624 (99.6%) | 54,411 (99.7%) | 2213 (93.3%) | 3.9% | Reference | <0.0001 |
| Yes | 250 (0.4%) | 165 (0.3%) | 85 (3.7%) | 34.0% | 8.70 (7.28–10.39) | ||
| Epilepsy | No | 56,465 (99.3%) | 54,216 (99.3%) | 2249 (97.9%) | 4.0% | Reference | <0.0001 |
| Yes | 409 (0.7%) | 360 (0.7%) | 49 (2.1%) | 12.0% | 3.01 (2.31–3.92) | ||
| Parkinsonism | No | 56,806 (99.9%) | 54,534 (99.9%) | 2272 (98.9%) | 4.0% | Reference | <0.0001 |
| Yes | 68 (0.1%) | 42 (0.1%) | 26 (1.1%) | 38.2% | 9.56 (7.05–12.97) | ||
| Stroke (combined) 5 | No | 56,571 (99.5%) | 54,351 (99.6%) | 2220 (96.6%) | 3.9% | Reference | <0.0001 |
| Yes | 303 (0.5%) | 225 (0.4%) | 78 (3.4%) | 25.7% | 6.56 (5.39–7.98) | ||
| Stroke (hemorrhagic) | No | 56,817 (99.9%) | 54,528 (99.9%) | 2289 (99.6%) | 4.0% | Reference | <0.0001 |
| Yes | 57 (0.1%) | 48 (0.1%) | 9 (0.4%) | 15.8% | 3.92 (2.15–7.15) | ||
| Stroke (ischemic) | No | 56,705 (99.7%) | 54,453(99.8%) | 2252 (98.0%) | 4.0% | Reference | <0.0001 |
| Yes | 169 (0.3%) | 123 (0.2%) | 46 (2.0%) | 27.2% | 6.85 (5.34–8.80) | ||
| Transitory ischemic attack | No | 56,773 (99.8%) | 54,504 (99.9%) | 2269 (98.7%) | 4.0% | Reference | <0.0001 |
| Yes | 101 (0.2%) | 72 (0.1%) | 29 (1.3%) | 28.7% | 7.18 (5.27–9.79) | ||
| Asthma | No | 49,270 (86.6%) | 47,435 (86.9%) | 1835 (79.9%) | 3.7% | Reference | <0.0001 |
| Yes | 7604 (13.4%) | 7141 (13.1%) | 463 (20.1%) | 6.1% | 1.63 (1.48–1.81) | ||
| Chronic obstructive pulmonary disease | No | 55,800 (98.1%) | 53,767 (98.5%) | 2033 (88.5%) | 3.6% | Reference | <0.0001 |
| Yes | 1074 (1.9%) | 809 (1.5%) | 265 (11.5%) | 24.7% | 6.77 (6.05–7.58) | ||
| Injection drug use 6 | No | 54,574 (96.0%) | 52,506 (96.2%) | 2068 (90.0%) | 3.8% | Reference | <0.0001 |
| Yes | 2300 (4.0%) | 2070 (3.8%) | 230 (10.0%) | 10.0% | 2.64 (2.32–3.00) | ||
| Problematic alcohol use 7 | No | 54,322 (95.5%) | 52,336 (95.9%) | 1986 (86.4%) | 3.7% | Reference | <0.0001 |
| Yes | 2552 (4.5%) | 2240 (4.1%) | 312 (13.6%) | 12.2% | 3.34 (2.99–3.74) | ||
| Cancer 8 | No | 50,865 (89.4%) | 49,182 (90.1%) | 1683 (73.2%) | 3.3% | Reference | <0.0001 |
| Yes | 6009 (10.6%) | 5394 (9.9%) | 615 (26.8%) | 10.2% | 3.09 (2.83–3.38) | ||
| Cirrhosis 7 | No | 56,635 (99.6%) | 54,402 (99.7%) | 2233 (97.2%) | 3.9% | Reference | <0.0001 |
| Yes | 239 (0.4%) | 174 (0.3%) | 65 (2.8%) | 27.2% | 6.90 (5.58–8.52) |
| Variable | Category | 26 January 2020–15 January 2021 (n = 56,874; Hospitalized = 2298) | |
|---|---|---|---|
| aRR (95% CI) * | p† | ||
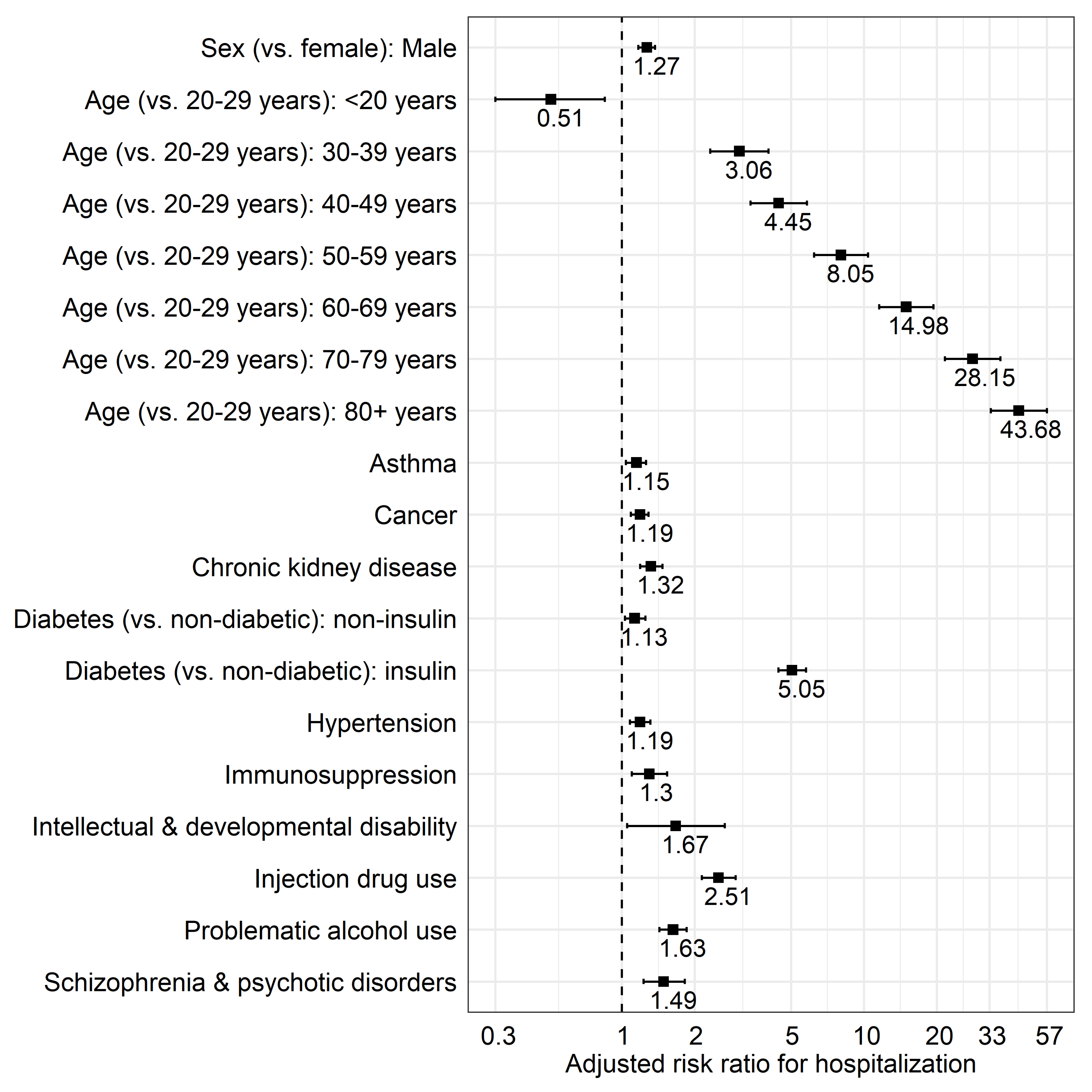
| Sex (vs. female) | Male | 1.27 (1.17–1.37) | <0.0001 |
| Age ‡ (Reference group: 20–29 years) | <20 years | 0.51 (0.30–0.85) | 0.0103 |
| 30–39 years | 3.06 (2.32–4.03) | <0.0001 | |
| 40–49 years | 4.45 (3.40–5.82) | <0.0001 | |
| 50–59 years | 8.05 (6.22–10.41) | <0.0001 | |
| 60–69 years | 14.98 (11.58–19.37) | <0.0001 | |
| 70–79 years | 28.15 (21.64–36.61) | <0.0001 | |
| 80+ years | 43.68 (33.41–57.10) | <0.0001 | |
| Asthma | 1.15 (1.04–1.26) | 0.0049 | |
| Cancer 1 | 1.19 (1.09–1.29) | 0.0001 | |
| Chronic kidney disease 2 | 1.32 (1.19–1.47) | <0.0001 | |
| Diabetes (vs. non-diabetic) | Non-insulin | 1.13 (1.03–1.25) | 0.0112 |
| Insulin 3 | 5.05 (4.43–5.76) | <0.0001 | |
| Hypertension | 1.19 (1.08–1.31) | 0.0007 | |
| Immunosuppression 4 | 1.30 (1.10–1.54) | 0.0019 | |
| Injection drug use 5 | 2.51 (2.14–2.95) | <0.0001 | |
| Intellectual and developmental disability 6 | 1.67 (1.05–2.66) | 0.0307 | |
| Problematic alcohol use 7 | 1.63 (1.43–1.85) | <0.0001 | |
| Schizophrenia and psychotic disorders | 1.49 (1.23–1.82) | <0.0001 | |
| Variable | Category | 26 January 2020–15 January 2021 (n = 17,036; Hospitalized = 191) | |
|---|---|---|---|
| aRR (95% CI) * | p† | ||
| Age ** (Reference group: 20–29 years) | <20 years | 0.24 (0.06–0.95) | 0.0424 |
| 30–39 years | 1.99 (1.35–2.94) | 0.0005 | |
| 40–49 years | 2.32 (1.54–3.48) | <0.0001 | |
| Asthma | 1.80 (1.29–2.52) | 0.0005 | |
| Diabetes (vs. non-diabetic) | Non-insulin | 2.39 (1.46–3.89) | 0.0005 |
| Insulin 1 | 31.89 (16.78–60.60) | <0.0001 | |
| Hypertension | 2.02 (1.29–3.16) | 0.0020 | |
| Injection drug use 2 | 3.97 (2.44–6.43) | <0.0001 | |
| Pregnancy | 2.69 (1.42–5.07) | 0.0023 | |
| Problematic alcohol use 3 | 3.05 (1.86–5.02) | <0.0001 | |
| Variable | Age Group | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| All Age Groups ⁰ (n = 56,874; Hospitalized = 2298) | <40 Years (n = 31,854; Hospitalized = 261) | 40–59 Years (n = 16,383; Hospitalized = 578) | 60+ Years (n = 8637; Hospitalized = 1459) | ||||||
| aRR (95% CI) * | p † | aRR (95% CI) * | p† | aRR (95% CI) * | p† | aRR (95% CI) * | p† | ||
| Sex (male vs. female) | 1.27 (1.17–1.37) | <0.0001 | 0.99 (0.77–1.27) | 0.93 | 1.46 (1.24–1.72) | <0.0001 | 1.26 (1.15–1.38) | <0.0001 | |
| Asthma | 1.15 (1.04–1.26) | 0.0049 | 1.10 (0.78–1.54) | 0.59 | 1.41 (1.16–1.71) | 0.0005 | 1.10 (0.98–1.23) | 0.09 | |
| Cancer 1 | 1.19 (1.09–1.29) | 0.0001 | 1.47 (0.99–2.17) | 0.054 | 1.24 (1.00–1.54) | 0.0459 | 1.31 (1.19–1.44) | <0.0001 | |
| Chronic kidney disease 2 | 1.32 (1.19–1.47) | <0.0001 | 2.68 (1.22–5.90) | 0.0141 | 1.32 (0.97–1.80) | 0.08 | 1.70 (1.53–1.88) | <0.0001 | |
| Diabetes (vs. non-diabetic) | Non-insulin | 1.13 (1.03–1.25) | 0.0112 | 2.03 (0.98–4.17) | 0.055 | 1.62 (1.31–1.99) | <0.0001 | 1.01 (0.91–1.12) | 0.87 |
| Insulin 3 | 5.05 (4.43–5.76) | <0.0001 | 20.29 (4.69–87.90) | <0.0001 | 19.30 (14.66–25.40) | <0.0001 | 3.94 (3.52–4.41) | <0.0001 | |
| Hypertension | 1.19 (1.08–1.31) | 0.0007 | 1.87 (0.98–3.57) | 0.057 | 1.46 (1.21–1.75) | <0.0001 | 1.31 (1.18–1.46) | <0.0001 | |
| Immunosuppression 4 | 1.30 (1.10–1.54) | 0.0019 | 2.56 (1.67–3.94) | <0.0001 | 1.30 (0.95–1.76) | 0.10 | 1.14 (0.93–1.38) | 0. 21 | |
| Injection drug use 5 | 2.51 (2.14–2.95) | <0.0001 | 2.95 (1.78–4.90) | <0.0001 | 2.55 (1.97–3.30) | <0.0001 | 1.01 (0.76–1.32) | 0.97 | |
| Intellectual and developmental disability 6 | 1.67 (1.05–2.66) | 0.0307 | 1.17 (0.56–2.42) | 0.68 | 4.28 (2.29–8.01) | <0.0001 | 1.21 (0.71–2.04) | 0.49 | |
| Problematic alcohol use 7 | 1.63 (1.43–1.85) | <0.0001 | 2.84 (1.72–4.72) | <0.0001 | 1.62 (1.26–2.10) | 0.0002 | 1.53 (1.30–1.79) | <0.0001 | |
| Schizophrenia and psychotic disorders | 1.49 (1.23–1.82) | <0.0001 | 2.33 (1.40–3.87) | 0.0012 | 1.19 (0.80–1.76) | 0.38 | 1.56 (1.21–2.00) | 0.0005 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Velásquez García, H.A.; Wilton, J.; Smolina, K.; Chong, M.; Rasali, D.; Otterstatter, M.; Rose, C.; Prystajecky, N.; David, S.; Galanis, E.; et al. Mental Health and Substance Use Associated with Hospitalization among People with COVID-19: A Population-Based Cohort Study. Viruses 2021, 13, 2196. https://doi.org/10.3390/v13112196
Velásquez García HA, Wilton J, Smolina K, Chong M, Rasali D, Otterstatter M, Rose C, Prystajecky N, David S, Galanis E, et al. Mental Health and Substance Use Associated with Hospitalization among People with COVID-19: A Population-Based Cohort Study. Viruses. 2021; 13(11):2196. https://doi.org/10.3390/v13112196
Chicago/Turabian StyleVelásquez García, Héctor Alexander, James Wilton, Kate Smolina, Mei Chong, Drona Rasali, Michael Otterstatter, Caren Rose, Natalie Prystajecky, Samara David, Eleni Galanis, and et al. 2021. "Mental Health and Substance Use Associated with Hospitalization among People with COVID-19: A Population-Based Cohort Study" Viruses 13, no. 11: 2196. https://doi.org/10.3390/v13112196
APA StyleVelásquez García, H. A., Wilton, J., Smolina, K., Chong, M., Rasali, D., Otterstatter, M., Rose, C., Prystajecky, N., David, S., Galanis, E., McKee, G., Krajden, M., & Janjua, N. Z. (2021). Mental Health and Substance Use Associated with Hospitalization among People with COVID-19: A Population-Based Cohort Study. Viruses, 13(11), 2196. https://doi.org/10.3390/v13112196







