Transcutaneous Administration of Dengue Vaccines
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Mice
2.3. Cell Lines and Virus
2.4. Purification of the Heat Labile Toxin (LT1)
2.5. DENV2 Harvesting and Concentration
2.6. DENV2 Purification
2.7. Transcutaneous (TC) and Intradermal (ID) Immunization Procedures
2.8. Analysis of Specific Antibody Responses
2.9. Analysis of Cytokine Production
2.10. Lethal Challenges with the DENV2 NGC Strain
2.11. Statistical Analyses
3. Results
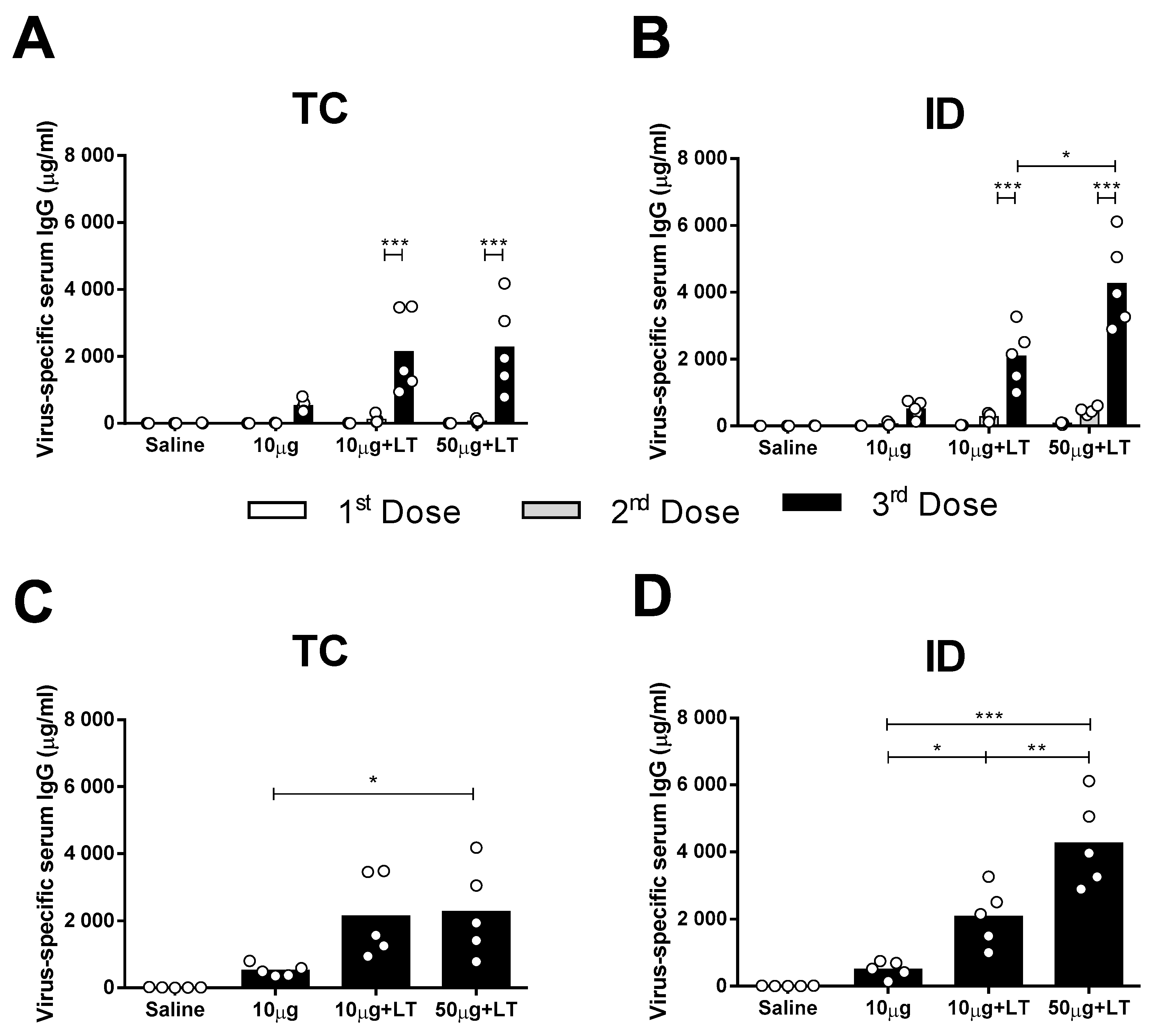
3.1. Induction of DENV2-Specific Serum Antibody Responses in Mice Immunized via the TC or ID Route
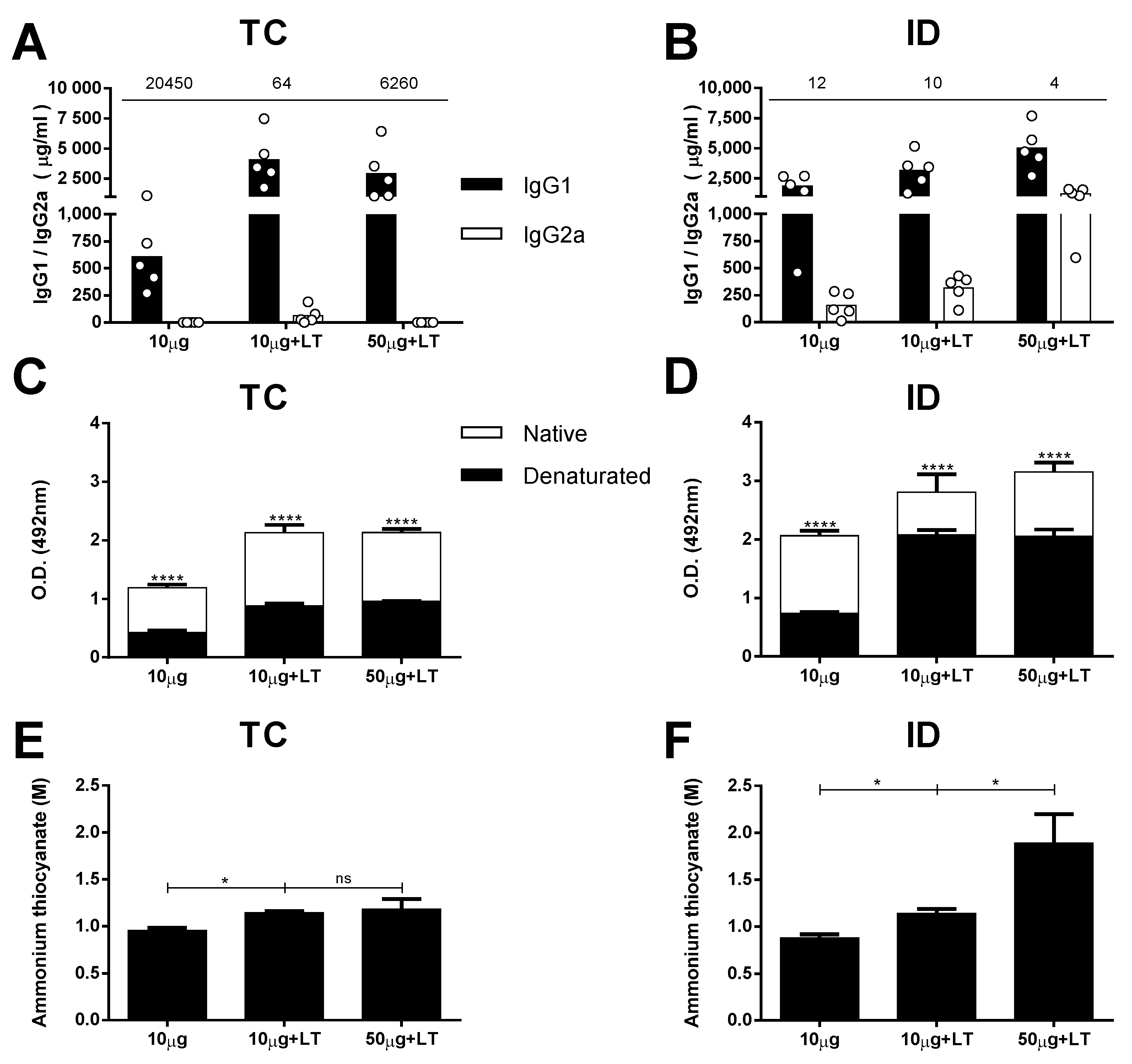
3.2. Characterization of the Antibody Responses in the Mice Immunized via the TC or ID Route
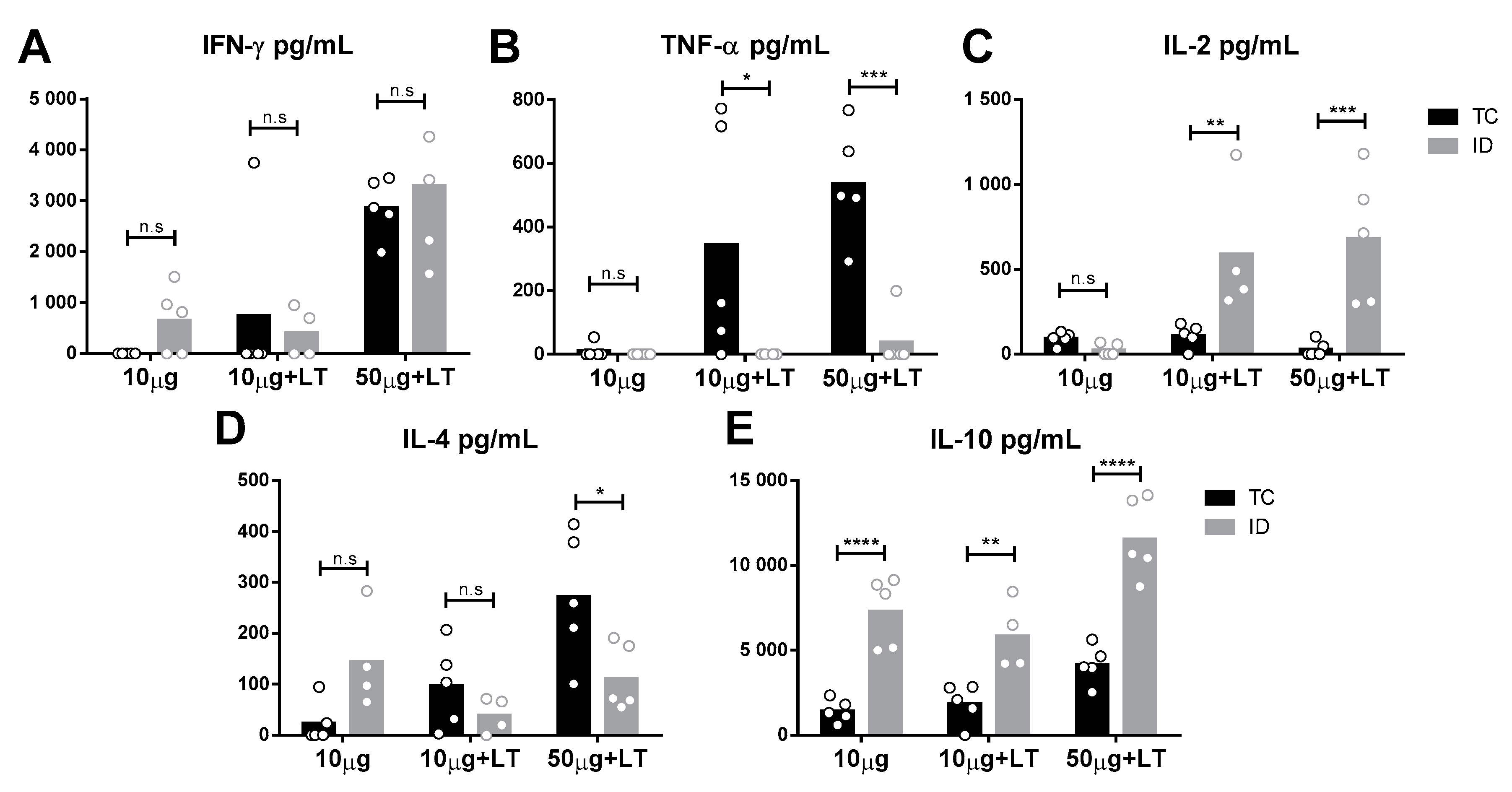
3.3. Detection of Cytokine Secretion Profiles in the Mice Immunized with the Different Vaccines Tested
3.4. Protective Immunity Conferred by the Anti-DENV2 Vaccines Inoculated via the TC or ID Route
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stahl, H.-C.; Butenschoen, V.M.; Tran, H.T.; Gozzer, E.; Skewes, R.; Mahendradhata, Y.; Runge-Ranzinger, S.; Kroeger, A.; Farlow, A. Cost of dengue outbreaks: Literature review and country case studies. BMC Public Health 2013, 13, 1048. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Constenla, D.; Garcia, C.; Lefcourt, N. Assessing the Economics of Dengue: Results from a Systematic Review of the Literature and Expert Survey. Pharmacoeconomics 2015, 33, 1107–1135. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO) Dengue and severe dengue. Available online: https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue (accessed on 8 May 2019).
- Glenn, G.M.; Scharton-Kersten, T.; Vassell, R.; Mallett, C.P.; Hale, T.L.; Alving, C.R. Transcutaneous immunization with cholera toxin protects mice against lethal mucosal toxin challenge. J. Immunol. 1998, 161, 3211–3214. [Google Scholar] [PubMed]
- Glenn, G.M.; Taylor, D.N.; Li, X.; Frankel, S.; Montemarano, A.; Alving, C.R. Transcutaneous immunization: A human vaccine delivery strategy using a patch. Nat. Med. 2000, 6, 1403–1406. [Google Scholar] [CrossRef]
- Matsuo, K.; Yokota, Y.; Zhai, Y.; Quan, Y.S.; Kamiyama, F.; Mukai, Y.; Okada, N.; Nakagawa, S. A low-invasive and effective transcutaneous immunization system using a novel dissolving microneedle array for soluble and particulate antigens. J. Control. Release 2012, 161, 10–17. [Google Scholar] [CrossRef]
- Zhang, J.; Shi, Z.; Kong, F.K.; Jex, E.; Huang, Z.; Watt, J.M.; Van Kampen, K.R.; Tang, D.C.C. Topical application of Escherichia coli-vectored vaccine as a simple method for eliciting protective immunity. Infect. Immun. 2006, 74, 3607–3617. [Google Scholar] [CrossRef]
- Clark, R. a Skin-resident T cells: The ups and downs of on site immunity. J. Invest. Dermatol. 2010, 130, 362–370. [Google Scholar] [CrossRef]
- Combadiere, B.; Liard, C. Transcutaneous and intradermal vaccination. Hum. Vaccin. 2011, 7, 811–827. [Google Scholar] [CrossRef]
- Lee, M.Y.; Shin, M.C.; Yang, V.C. Transcutaneous antigen delivery system. BMB Rep. 2013, 46, 17–24. [Google Scholar] [CrossRef]
- Giudice, E.L.; Campbell, J.D. Needle-free vaccine delivery. Adv. Drug Deliv. Rev. 2006, 58, 68–89. [Google Scholar] [CrossRef] [PubMed]
- Bal, S.M.; Ding, Z.; Van Riet, E.; Jiskoot, W.; Bouwstra, J.A. Advances in transcutaneous vaccine delivery: Do all ways lead to Rome? J. Control. Release 2010, 148, 266–282. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Qiu, Y.; Chen, J.; Zhang, S.; Xu, B.; Gao, Y. Effective transcutaneous immunization against hepatitis B virus by a combined approach of hydrogel patch formulation and microneedle arrays. Biomed. Microdevices 2013, 15, 1077–1085. [Google Scholar] [CrossRef] [PubMed]
- Belyakov, I.M.; Ahlers, J.D. Simultaneous Approach Using Systemic, Mucosal and Transcutaneous Routes of Immunization For Development of Protective HIV-1 Vaccines. Curr. Med. Chem. 2011, 18, 3953–3962. [Google Scholar] [CrossRef]
- Rancan, F.; Amselgruber, S.; Hadam, S.; Munier, S.; Pavot, V.; Verrier, B.; Hackbarth, S.; Combadiere, B.; Blume-Peytavi, U.; Vogt, A. Particle-based transcutaneous administration of HIV-1 p24 protein to human skin explants and targeting of epidermal antigen presenting cells. J. Control. Release 2014, 176, 115–122. [Google Scholar] [CrossRef]
- Cheng, J.Y.; Huang, H.N.; Tseng, W.C.; Li, T.L.; Chan, Y.L.; Cheng, K.C.; Wu, C.J. Transcutaneous immunization by lipoplex-patch based DNA vaccines is effective vaccination against Japanese encephalitis virus infection. J. Control. Release 2009, 135, 242–249. [Google Scholar] [CrossRef]
- El-Ghorr, A.A.; Williams, R.M.; Heap, C.; Norval, M. Transcutaneous immunisation with herpes simplex virus stimulates immunity in mice. FEMS Immunol. Med. Microbiol. 2000, 29, 255–261. [Google Scholar] [CrossRef]
- Hirschberg, H.; Van Kuijk, S.; Loch, J.; Jiskoot, W.; Bouwstra, J.; Kersten, G.; Amorij, J.P. A combined approach of vesicle formulations and microneedle arrays for transcutaneous immunization against hepatitis B virus. Eur. J. Pharm. Sci. 2012, 46, 1–7. [Google Scholar] [CrossRef]
- Johnson, A.J.; Roehrig, J.T. New mouse model for dengue virus vaccine testing. J. Virol. 1999, 73, 783–786. [Google Scholar] [CrossRef]
- Rodrigues, J.F.; Mathias-Santos, C.; Sbrogio-Almeida, M.E.; Amorim, J.H.; Cabrera-Crespo, J.; Balan, A.; Ferreira, L.C.S. Functional diversity of heat-labile toxins (LT) produced by enterotoxigenic Escherichia coli: Differential enzymatic and immunological activities of LT1 (hLT) and LT4 (pLT). J. Biol. Chem. 2011, 286, 5222–5233. [Google Scholar] [CrossRef]
- Lasaro, M.A.S.; Rodrigues, J.F.; Mathias-Santos, C.; Guth, B.E.C.; Régua-Mangia, A.; Ferreira, A.J.P.; Takagi, M.; Cabrera-Crespo, J.; Sbrogio-Almeida, M.E.; Ferreira, L.C.D.S. Production and release of heat-labile toxin by wild-type human-derived enterotoxigenic Escherichia coli. FEMS Immunol. Med. Microbiol. 2006, 48, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Lasaro, M.A.; Mathias-Santos, C.; Rodrigues, J.F.; Ferreira, L.C.S. Functional and immunological characterization of a natural polymorphic variant of a heat-labile toxin (LT-I) produced by enterotoxigenic Escherichia coli (ETEC): RESEARCH ARTICLE. FEMS Immunol. Med. Microbiol. 2009, 55, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Amorim, J.H.; Pereira Bizerra, R.S.; dos Santos Alves, R.P.; Sbrogio-Almeida, M.E.; Levi, J.E.; Capurro, M.L.; de Souza Ferreira, L.C. A Genetic and Pathologic Study of a DENV2 Clinical Isolate Capable of Inducing Encephalitis and Hematological Disturbances in Immunocompetent Mice. PLoS ONE 2012, 7, e044984. [Google Scholar] [CrossRef] [PubMed]
- Henriques, H.R.; Rampazo, E.V.; Gonçalves, A.J.S.; Vicentin, E.C.M.; Amorim, J.H.; Panatieri, R.H.; Amorim, K.N.S.; Yamamoto, M.M.; Ferreira, L.C.S.; Alves, A.M.B.; et al. Targeting the non-structural protein 1 from dengue virus to a dendritic cell population confers protective immunity to lethal virus challenge. PLoS Negl. Trop. Dis. 2013, 7, e2330. [Google Scholar] [CrossRef]
- Andreata-Santos, R. No Other Adverse Reactions, Such as Weight Loss or Altered Animal Behavior, Were Observed in the Mice in Which the Vaccine Regimens Were Tested; University of São Paulo: São Paulo, Brazil, 2020. [Google Scholar]
- Combadière, B.; Vogt, A.; Mahé, B.; Costagliola, D.; Hadam, S.; Bonduelle, O.; Sterry, W.; Staszewski, S.; Schaefer, H.; van der Werf, S.; et al. Preferential amplification of CD8 effector-T cells after transcutaneous application of an inactivated influenza vaccine: A randomized phase I trial. PLoS ONE 2010, 5, e10818. [Google Scholar] [CrossRef]
- Slifka, M.K.; Leung, D.Y.M.; Hammarlund, E.; Raué, H.P.; Simpson, E.L.; Tofte, S.; Baig-Lewis, S.; David, G.; Lynn, H.; Woolson, R.; et al. Transcutaneous yellow fever vaccination of subjects with or without atopic dermatitis. J. Allergy Clin. Immunol. 2014, 133, 439–447. [Google Scholar] [CrossRef]
- Vrdoljak, A.; McGrath, M.G.; Carey, J.B.; Draper, S.J.; Hill, A.V.S.; O’Mahony, C.; Crean, A.M.; Moore, A.C. Coated microneedle arrays for transcutaneous delivery of live virus vaccines. J. Control. Release 2012, 159, 34–42. [Google Scholar] [CrossRef]
- Vogt, A.; Mahé, B.; Costagliola, D.; Bonduelle, O.; Hadam, S.; Schaefer, G.; Schaefer, H.; Katlama, C.; Sterry, W.; Autran, B.; et al. Transcutaneous anti-influenza vaccination promotes both CD4 and CD8 T cell immune responses in humans. J. Immunol. 2008, 180, 1482–1489. [Google Scholar] [CrossRef]
- Muller, D.A.; Depelsenaire, A.C.I.; Shannon, A.E.; Watterson, D.; Corrie, S.R.; Owens, N.S.; Agyei-Yeboah, C.; Cheung, S.T.M.; Zhang, J.; Fernando, G.J.P.; et al. Efficient Delivery of Dengue Virus Subunit Vaccines to the Skin by Microprojection Arrays. Vaccines 2019, 7, 189. [Google Scholar] [CrossRef]
- Turvey, M.E.; Uppu, D.S.S.M.; Mohamed Sharif, A.R.; Bidet, K.; Alonso, S.; Ooi, E.E.; Hammond, P.T. Microneedle-based intradermal delivery of stabilized dengue virus. Bioeng. Transl. Med. 2019, 4. [Google Scholar] [CrossRef]
- Scharton-Kersten, T.; Yu, J.M.; Vassell, R.; O’Hagan, D.; Alving, C.R.; Glenn, G.M. Transcutaneous immunization with bacterial ADP-ribosylating exotoxins, subunits, and unrelated adjuvants. Infect. Immun. 2000, 68, 5306–5313. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Frolov, V.G.; Seid, R.C.; Odutayo, O.; Al-Khalili, M.; Yu, J.; Frolova, O.Y.; Vu, H.; Butler, B.A.; Look, J.L.; Ellingsworth, L.R.; et al. Transcutaneous delivery and thermostability of a dry trivalent inactivated influenza vaccine patch. Influenza Other Respi. Viruses 2008, 2, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Mawas, F.; Peyre, M.; Beignon, A.; Frost, L.; Del Giudice, G.; Rappuoli, R.; Muller, S.; Sesardic, D.; Partidos, C.D. Successful Induction of Protective Antibody Responses against Haemophilus influenzae Type b and Diphtheria after Transcutaneous Immunization with the Glycoconjugate Polyribosyl Ribitol Phosphate–Cross-Reacting Material 197 Vaccine. J. Infect. Dis. 2004, 190, 1177–1182. [Google Scholar] [CrossRef] [PubMed]
- Lasaro, M.A.; Rodrigues, J.F.; Mathias-Santos, C.; Guth, B.E.C.; Balan, A.; Sbrogio-Almeida, M.E.; Ferreira, L.C.S. Genetic Diversity of Heat-Labile Toxin Expressed by Enterotoxigenic Escherichia coli Strains Isolated from Humans. J. Bacteriol. 2008, 190, 2400–2410. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.; Verbaan, F.J.; Bivas-Benita, M.; Bungener, L.; Huckriede, A.; van den Berg, D.J.; Kersten, G.; Bouwstra, J.A. Microneedle arrays for the transcutaneous immunization of diphtheria and influenza in BALB/c mice. J. Control. Release 2009, 136, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Costin, J.M.; Zaitseva, E.; Kahle, K.M.; Nicholson, C.O.; Rowe, D.K.; Graham, A.S.; Bazzone, L.E.; Hogancamp, G.; Figueroa Sierra, M.; Fong, R.H.; et al. Mechanistic Study of Broadly Neutralizing Human Monoclonal Antibodies against Dengue Virus That Target the Fusion Loop. J. Virol. 2013, 87, 52–66. [Google Scholar] [CrossRef]
- Tsai, W.-Y.; Lai, C.-Y.; Wu, Y.-C.; Lin, H.-E.; Edwards, C.; Jumnainsong, A.; Kliks, S.; Halstead, S.; Mongkolsapaya, J.; Screaton, G.R.; et al. High-Avidity and Potently Neutralizing Cross-Reactive Human Monoclonal Antibodies Derived from Secondary Dengue Virus Infection. J. Virol. 2013, 87, 12562–12575. [Google Scholar] [CrossRef]
- Elong Ngono, A.; Chen, H.-W.; Tang, W.W.; Joo, Y.; King, K.; Weiskopf, D.; Sidney, J.; Sette, A.; Shresta, S. Protective Role of Cross-Reactive CD8 T Cells Against Dengue Virus Infection. EBioMedicine 2016, 13, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Zellweger, R.M.; Tang, W.W.; Eddy, W.E.; King, K.; Sanchez, M.C.; Shresta, S. CD8 + T Cells Can Mediate Short-Term Protection against Heterotypic Dengue Virus Reinfection in Mice. J. Virol. 2015, 89, 6494–6505. [Google Scholar] [CrossRef]
- Liu, T.; Khanna, K.M.; Carriere, B.N.; Hendricks, R.L. Gamma Interferon Can Prevent Herpes Simplex Virus Type 1 Reactivation from Latency in Sensory Neurons. J. Virol. 2001, 75, 11178–11184. [Google Scholar] [CrossRef]
- Amorim, J.H.; dos Santos Alves, R.P.; Bizerra, R.; Araújo Pereira, S.; Ramos Pereira, L.; Nascimento Fabris, D.L.; Santos, R.A.; Romano, C.M.; de Souza Ferreira, L.C. Antibodies are not required to a protective immune response against dengue virus elicited in a mouse encephalitis model. Virology 2016, 487, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Yauch, L.E.; Zellweger, R.M.; Kotturi, M.F.; Qutubuddin, A.; Sidney, J.; Peters, B.; Prestwood, T.R.; Sette, A.; Shresta, S. A Protective Role for Dengue Virus-Specific CD8 + T Cells. J. Immunol. 2009, 182, 4865–4873. [Google Scholar] [CrossRef] [PubMed]
- Yauch, L.E.; Prestwood, T.R.; May, M.M.; Morar, M.M.; Zellweger, R.M.; Peters, B.; Sette, A.; Shresta, S. CD4 + T Cells Are Not Required for the Induction of Dengue Virus-Specific CD8 + T Cell or Antibody Responses but Contribute to Protection after Vaccination. J. Immunol. 2010, 185, 5405–5416. [Google Scholar] [CrossRef] [PubMed]
- Hatch, S.; Endy, T.P.; Thomas, S.; Mathew, A.; Potts, J.; Pazoles, P.; Libraty, D.H.; Gibbons, R.; Rothman, A.L. Intracellular Cytokine Production by Dengue Virus–specific T cells Correlates with Subclinical Secondary Infection. J. Infect. Dis. 2011, 203, 1282–1291. [Google Scholar] [CrossRef]
- Lindow, J.C.; Borochoff-Porte, N.; Durbin, A.P.; Whitehead, S.S.; Fimlaid, K.A.; Bunn, J.Y.; Kirkpatrick, B.D. Primary Vaccination with Low Dose Live Dengue 1 Virus Generates a Proinflammatory, Multifunctional T Cell Response in Humans. PLoS Negl. Trop. Dis. 2012, 6, e1742. [Google Scholar] [CrossRef]
- Boaz, M.J.; Waters, A.; Murad, S.; Easterbrook, P.J.; Vyakarnam, A. Presence of HIV-1 Gag-Specific IFN-γ + IL-2 + and CD28 + IL-2 + CD4 T Cell Responses Is Associated with Nonprogression in HIV-1 Infection. J. Immunol. 2002, 169, 6376–6385. [Google Scholar] [CrossRef]
- Basu, A.; Chaturvedi, U.C. Vascular endothelium: the battlefield of dengue viruses. FEMS Immunol. Med. Microbiol. 2008, 53, 287–299. [Google Scholar] [CrossRef]
- Libraty, D.H.; Endy, T.P.; Houng, H.H.; Green, S.; Kalayanarooj, S.; Suntayakorn, S.; Chansiriwongs, W.; Vaughn, D.W.; Nisalak, A.; Ennis, F.A.; et al. Differing Influences of Virus Burden and Immune Activation on Disease Severity in Secondary Dengue-3 Virus Infections. J. Infect. Dis. 2002, 185, 1213–1221. [Google Scholar] [CrossRef]
- Schexneider, K.I.; Reedy, E.A. Thrombocytopenia in dengue fever. Curr. Hematol. Rep. 2005, 4, 145–148. [Google Scholar]
- Schaeffer, E.; Flacher, V.; Papageorgiou, V.; Decossas, M.; Fauny, J.-D.; Krämer, M.; Mueller, C.G. Dermal CD14 + Dendritic Cell and Macrophage Infection by Dengue Virus Is Stimulated by Interleukin-4. J. Invest. Dermatol. 2015, 135, 1743–1751. [Google Scholar] [CrossRef]
- Pérez, A.B.; García, G.; Sierra, B.; Alvarez, M.; Vázquez, S.; Cabrera, M.V.; Rodríguez, R.; Rosario, D.; Martínez, E.; Denny, T.; et al. IL-10 levels in Dengue patients: Some findings from the exceptional epidemiological conditions in Cuba. J. Med. Virol. 2004, 73, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Shen, B.-Q.; Lee, D.Y.; Cortopassi, K.M.; Damico, L.A.; Zioncheck, T.F. Vascular Endothelial Growth Factor KDR Receptor Signaling Potentiates Tumor Necrosis Factor-induced Tissue Factor Expression in Endothelial Cells. J. Biol. Chem. 2001, 276, 5281–5286. [Google Scholar] [CrossRef] [PubMed]
- Martins, M.Â.; Silva, M.L.; Elói-Santos, S.M.; Ribeiro, J.G.L.; Peruhype-Magalhães, V.; Marciano, A.P.V.; Homma, A.; Kroon, E.G.; Teixeira-Carvalho, A.; Martins-Filho, O.A. Innate immunity phenotypic features point toward simultaneous raise of activation and modulation events following 17DD live attenuated yellow fever first-time vaccination. Vaccine 2008, 26, 1173–1184. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.L.; Martins, M.A.; Espírito-Santo, L.R.; Campi-Azevedo, A.C.; Silveira-Lemos, D.; Ribeiro, J.G.L.; Homma, A.; Kroon, E.G.; Teixeira-Carvalho, A.; Elói-Santos, S.M.; et al. Characterization of main cytokine sources from the innate and adaptive immune responses following primary 17DD yellow fever vaccination in adults. Vaccine 2011, 29, 583–592. [Google Scholar] [CrossRef] [PubMed]
- Trandem, K.; Zhao, J.; Fleming, E.; Perlman, S. Highly Activated Cytotoxic CD8 T Cells Express Protective IL-10 at the Peak of Coronavirus-Induced Encephalitis. J. Immunol. 2011, 186, 3642–3652. [Google Scholar] [CrossRef]
- Costa, S.M.; Paes, M.V.; Barreto, D.F.; Pinhão, A.T.; Barth, O.M.; Queiroz, J.L.S.; Armôa, G.R.G.; Freire, M.S.; Alves, A.M.B. Protection against dengue type 2 virus induced in mice immunized with a DNA plasmid encoding the non-structural 1 (NS1) gene fused to the tissue plasminogen activator signal sequence. Vaccine 2006, 24, 195–205. [Google Scholar] [CrossRef]
- dos Santos Alves, R.P.; Pereira, L.R.; Fabris, D.L.N.; Salvador, F.S.; Santos, R.A.; de Andrade Zanotto, P.M.; Romano, C.M.; Amorim, J.H.; de Souza Ferreira, L.C. Production of a Recombinant Dengue Virus 2 NS5 Protein and Potential Use as a Vaccine Antigen. Clin. Vaccine Immunol. 2016, 23, 460–469. [Google Scholar] [CrossRef]
- Putnak, R.; Cassidy, K.; Conforti, N.; Lee, R.; Sollazzo, D.; Truong, T.; Ing, E.; Dubois, D.; Sparkuhl, J.; Gastle, W.; et al. Immunogenic and protective response in mice immunized with a purified, inactivated, dengue-2 virus vaccine prototype made in fetal rhesus lung cells. Am. J. Trop. Med. Hyg. 1996, 55, 504–510. [Google Scholar] [CrossRef]
- Andreata-Santos, R. Flow-Cytometry Assays Were also Performed with Spleen Cells after the Immunization Protocols Prior to Challenge, But No Significant Difference Among Immunized Versus Nonimmunized Mice Was Observed; University of São Paulo: São Paulo, Brazil, 2020. [Google Scholar]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andreata-Santos, R.; Alves, R.P.d.S.; Pereira, S.A.; Pereira, L.R.; Freitas, C.L.d.; Pereira, S.S.; Venceslau-Carvalho, A.A.; Castro-Amarante, M.F.; Favaro, M.T.P.; Mathias-Santos, C.; et al. Transcutaneous Administration of Dengue Vaccines. Viruses 2020, 12, 514. https://doi.org/10.3390/v12050514
Andreata-Santos R, Alves RPdS, Pereira SA, Pereira LR, Freitas CLd, Pereira SS, Venceslau-Carvalho AA, Castro-Amarante MF, Favaro MTP, Mathias-Santos C, et al. Transcutaneous Administration of Dengue Vaccines. Viruses. 2020; 12(5):514. https://doi.org/10.3390/v12050514
Chicago/Turabian StyleAndreata-Santos, Robert, Rúbens Prince dos Santos Alves, Sara Araujo Pereira, Lennon Ramos Pereira, Carla Longo de Freitas, Samuel Santos Pereira, Alexia Adrianne Venceslau-Carvalho, Maria Fernanda Castro-Amarante, Marianna Teixeira Pinho Favaro, Camila Mathias-Santos, and et al. 2020. "Transcutaneous Administration of Dengue Vaccines" Viruses 12, no. 5: 514. https://doi.org/10.3390/v12050514
APA StyleAndreata-Santos, R., Alves, R. P. d. S., Pereira, S. A., Pereira, L. R., Freitas, C. L. d., Pereira, S. S., Venceslau-Carvalho, A. A., Castro-Amarante, M. F., Favaro, M. T. P., Mathias-Santos, C., Amorim, J. H., & Ferreira, L. C. d. S. (2020). Transcutaneous Administration of Dengue Vaccines. Viruses, 12(5), 514. https://doi.org/10.3390/v12050514





