Multisubunit RNA Polymerases of Jumbo Bacteriophages
Abstract
1. Multisubunit RNA Polymerases of Cellular Organisms
2. A Historical Expose: The First Multisubunit Phage RNA Polymerase
3. Jumbo Phages Encoding RNA Polymerases and Their Features
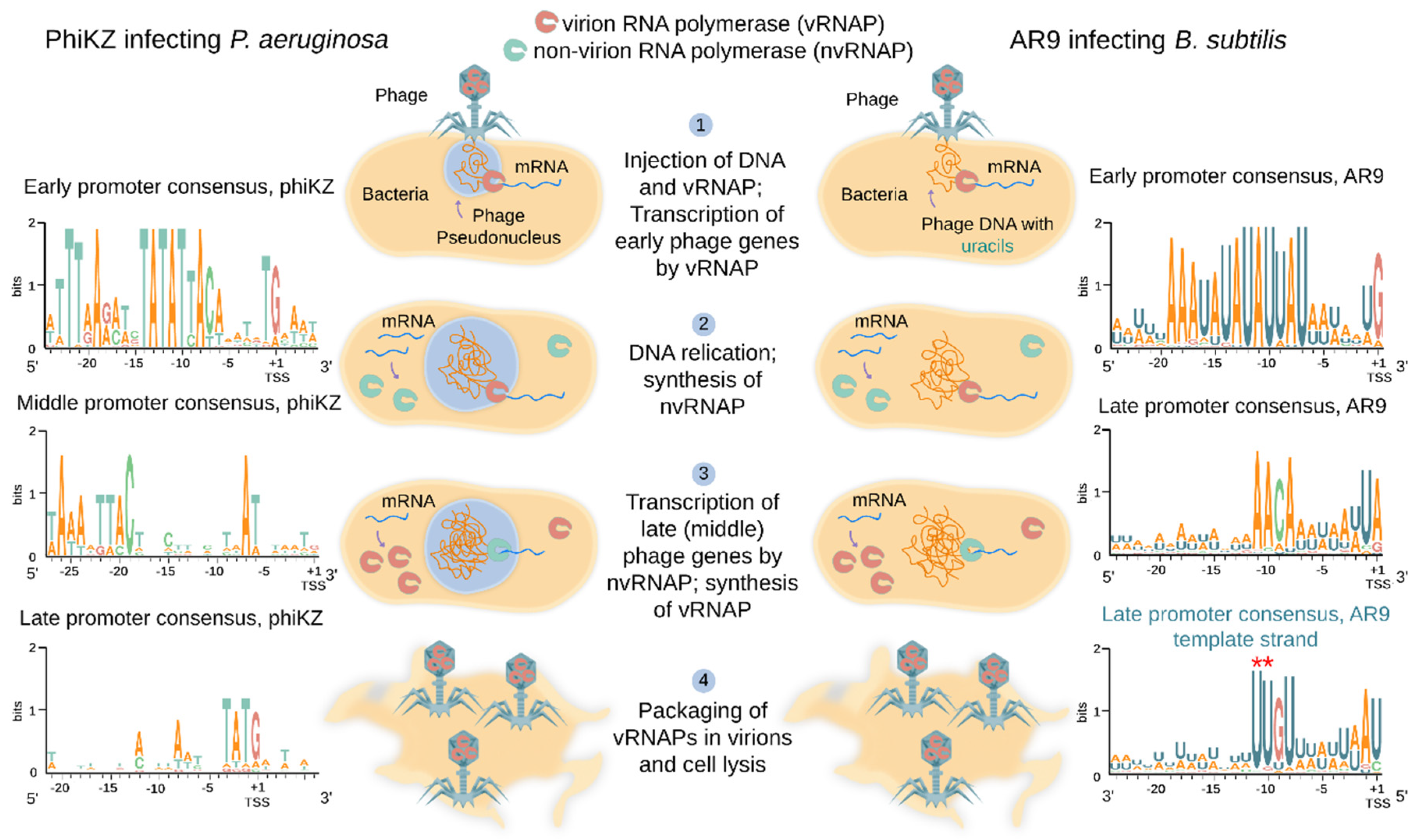
4. Transcriptional Strategy of Jumbo Phages Encoding RNA Polymerases
5. Analysis of RNA Polymerases Genes of Jumbo Phages and Their Origins
6. In Vitro Properties of Non-Virion RNA Polymerases of Jumbo Phages
7. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Werner, F.; Grohmann, D. Evolution of multisubunit RNA polymerases in the three domains of life. Nat. Rev. Microbiol. 2011, 9, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Law, J.A. RNA Pol IV and V in gene silencing: Rebel polymerases evolving away from Pol II’s rules. Curr. Opin. Plant Biol. 2015, 27, 154–164. [Google Scholar] [CrossRef] [PubMed]
- Hirata, A.; Klein, B.J.; Murakami, K.S. The X-ray crystal structure of RNA polymerase from Archaea. Nature 2008, 451, 851–854. [Google Scholar] [CrossRef] [PubMed]
- Werner, F. Structure and function of archaeal RNA polymerases. Mol. Microbiol. 2007, 65, 1395–1404. [Google Scholar] [CrossRef] [PubMed]
- Werner, F. Structural evolution of multisubunit RNA polymerases. Trends Microbiol. 2008, 16, 247–250. [Google Scholar] [CrossRef]
- Ishihama, A. Subunit of assembly of Escherichia coli RNA polymerase. Adv. Biophys. 1981, 14, 1–35. [Google Scholar]
- Lane, W.J.; Darst, S.A. Molecular evolution of multisubunit RNA polymerases: Structural analysis. J. Mol. Biol. 2010, 395, 686–704. [Google Scholar] [CrossRef]
- Minakhin, L.; Bhagat, S.; Brunning, A.; Campbell, E.A.; Darst, S.A.; Ebright, R.H.; Severinov, K. Bacterial RNA polymerase subunit omega and eukaryotic RNA polymerase subunit RPB6 are sequence, structural, and functional homologs and promote RNA polymerase assembly. Proc. Natl. Acad. Sci. USA 2001, 98, 892–897. [Google Scholar] [CrossRef]
- Vassylyev, D.G.; Sekine, S.; Laptenko, O.; Lee, J.; Vassylyeva, M.N.; Borukhov, S.; Yokoyama, S. Crystal structure of a bacterial RNA polymerase holoenzyme at 2.6 A resolution. Nature 2002, 417, 712–719. [Google Scholar] [CrossRef]
- Zhang, G.; Campbell, E.A.; Minakhin, L.; Richter, C.; Severinov, K.; Darst, S.A. Crystal structure of Thermus aquaticus core RNA polymerase at 3.3 A resolution. Cell 1999, 98, 811–824. [Google Scholar] [CrossRef]
- Vassylyev, D.G.; Vassylyeva, M.N.; Zhang, J.; Palangat, M.; Artsimovitch, I.; Landick, R. Structural basis for substrate loading in bacterial RNA polymerase. Nature 2007, 448, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Kettenberger, H.; Armache, K.J.; Cramer, P. Complete RNA polymerase II elongation complex structure and its interactions with NTP and TFIIS. Mol. Cell. 2004, 16, 955–965. [Google Scholar] [CrossRef] [PubMed]
- Paget, M.S. Bacterial Sigma Factors and Anti-Sigma Factors: Structure, Function and Distribution. Biomolecules 2015, 5, 1245–1265. [Google Scholar] [CrossRef]
- Blombach, F.; Smollett, K.L.; Grohmann, D.; Werner, F. Molecular Mechanisms of Transcription Initiation-Structure, Function, and Evolution of TFE/TFIIE-Like Factors and Open Complex Formation. J. Mol. Biol. 2016, 428, 2592–2606. [Google Scholar] [CrossRef] [PubMed]
- Hantsche, M.; Cramer, P. Conserved RNA polymerase II initiation complex structure. Curr. Opin. Struct. Biol. 2017, 47, 17–22. [Google Scholar] [CrossRef]
- Berdygulova, Z.; Westblade, L.F.; Florens, L.; Koonin, E.V.; Chait, B.T.; Ramanculov, E.; Washburn, M.P.; Darst, S.A.; Severinov, K.; Minakhin, L. Temporal regulation of gene expression of the Thermus thermophilus bacteriophage P23-45. J. Mol. Biol. 2011, 405, 125–142. [Google Scholar] [CrossRef]
- Iyer, L.M.; Aravind, L. Insights from the architecture of the bacterial transcription apparatus. J. Struct. Biol. 2012, 179, 299–319. [Google Scholar] [CrossRef]
- Lavysh, D.; Sokolova, M.; Minakhin, L.; Yakunina, M.; Artamonova, T.; Kozyavkin, S.; Makarova, K.S.; Koonin, E.V.; Severinov, K. The genome of AR9, a giant transducing Bacillus phage encoding two multisubunit RNA polymerases. Virology 2016, 495, 185–196. [Google Scholar] [CrossRef]
- Mirzakhanyan, Y.; Gershon, P.D. Multisubunit DNA-Dependent RNA Polymerases from Vaccinia Virus and Other Nucleocytoplasmic Large-DNA Viruses: Impressions from the Age of Structure. Microbiol. Mol. Biol. Rev. 2017, 81, e00010–e00017. [Google Scholar] [CrossRef]
- Ruprich-Robert, G.; Thuriaux, P. Non-canonical DNA transcription enzymes and the conservation of two-barrel RNA polymerases. Nucleic Acids Res. 2010, 38, 4559–4569. [Google Scholar] [CrossRef]
- Sykora, M.; Pospisek, M.; Novak, J.; Mrvova, S.; Krasny, L.; Vopalensky, V. Transcription apparatus of the yeast virus-like elements: Architecture, function, and evolutionary origin. PLoS Pathog. 2018, 14, e1007377. [Google Scholar] [CrossRef]
- Yutin, N.; Makarova, K.S.; Gussow, A.B.; Krupovic, M.; Segall, A.; Edwards, R.A.; Koonin, E.V. Discovery of an expansive bacteriophage family that includes the most abundant viruses from the human gut. Nat. Microbiol. 2018, 3, 38–46. [Google Scholar] [CrossRef]
- Ahn, B.Y.; Moss, B. RNA polymerase-associated transcription specificity factor encoded by vaccinia virus. Proc. Natl. Acad. Sci. USA 1992, 89, 3536–3540. [Google Scholar] [CrossRef]
- Forrest, D.; James, K.; Yuzenkova, Y.; Zenkin, N. Single-peptide DNA-dependent RNA polymerase homologous to multi-subunit RNA polymerase. Nat. Commun. 2017, 8, 15774. [Google Scholar] [CrossRef]
- Guarino, L.A.; Dong, W.; Jin, J. In vitro activity of the baculovirus late expression factor LEF-5. J. Virol. 2002, 76, 12663–12675. [Google Scholar] [CrossRef]
- Sokolova, M.; Borukhov, S.; Lavysh, D.; Artamonova, T.; Khodorkovskii, M.; Severinov, K. A non-canonical multisubunit RNA polymerase encoded by the AR9 phage recognizes the template strand of its uracil-containing promoters. Nucleic Acids Res. 2017, 45, 5958–5967. [Google Scholar] [CrossRef]
- Spencer, E.; Shuman, S.; Hurwitz, J. Purification and properties of vaccinia virus DNA-dependent RNA polymerase. J. Biol. Chem. 1980, 255, 5388–5395. [Google Scholar]
- Yakunina, M.; Artamonova, T.; Borukhov, S.; Makarova, K.S.; Severinov, K.; Minakhin, L. A non-canonical multisubunit RNA polymerase encoded by a giant bacteriophage. Nucleic Acids Res. 2015, 43, 10411–10420. [Google Scholar] [CrossRef]
- Rima, B.K.; Takahashi, I. The synthesis of nucleic acids in Bacillus subtilis infected with phage PBS 1. Can. J. Biochem. 1973, 51, 1219–1224. [Google Scholar] [CrossRef]
- Price, A.R.; Frabotta, M. Resistance of bacteriophage PBS2 infection to rifampicin, an inhibitor of Bacillus subtilis RNA synthesis. Biochem. Biophys. Res. Commun. 1972, 48, 1578–1585. [Google Scholar] [CrossRef]
- Geiduschek, E.P.; Sklar, J. Continual requirement for a host RNA polymerase component in a bacteriophage development. Nature 1969, 221, 833–836. [Google Scholar] [CrossRef] [PubMed]
- Haselkorn, R.; Vogel, M.; Brown, R.D. Conservation of the rifamycin sensitivity of transcription during T4 development. Nature 1969, 221, 836–838. [Google Scholar] [CrossRef] [PubMed]
- Summers, W.C.; Siegel, R.B. Control of template specificity of E. coli RNA polymerase by a phage-coded protein. Nature 1969, 223, 1111–1113. [Google Scholar] [CrossRef]
- Chamberlin, M.; McGrath, J.; Waskell, L. New RNA polymerase from Escherichia coli infected with bacteriophage T7. Nature 1970, 228, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Clark, S.; Losick, R.; Pero, J. New RNA polymerase from Bacillus subtilis infected with phage PBS2. Nature 1974, 252, 21–24. [Google Scholar] [CrossRef]
- Clark, S. Transcriptional specificity of a multisubunit RNA polymerase induced by Bacillus subtilis bacteriophage PBS2. J. Virol. 1978, 25, 224–237. [Google Scholar] [CrossRef] [PubMed]
- Mesyanzhinov, V.V.; Robben, J.; Grymonprez, B.; Kostyuchenko, V.A.; Bourkaltseva, M.V.; Sykilinda, N.N.; Krylov, V.N.; Volckaert, G. The genome of bacteriophage phiKZ of Pseudomonas aeruginosa. J. Mol. Biol. 2002, 317, 1–19. [Google Scholar] [CrossRef]
- Hertveldt, K.; Lavigne, R.; Pleteneva, E.; Sernova, N.; Kurochkina, L.; Korchevskii, R.; Robben, J.; Mesyanzhinov, V.; Krylov, V.N.; Volckaert, G. Genome comparison of Pseudomonas aeruginosa large phages. J. Mol. Biol. 2005, 354, 536–545. [Google Scholar] [CrossRef]
- Thomas, J.A.; Rolando, M.R.; Carroll, C.A.; Shen, P.S.; Belnap, D.M.; Weintraub, S.T.; Serwer, P.; Hardies, S.C. Characterization of Pseudomonas chlororaphis myovirus 201varphi2-1 via genomic sequencing, mass spectrometry, and electron microscopy. Virology 2008, 376, 330–338. [Google Scholar] [CrossRef]
- Krylov, V.N.; Dela Cruz, D.M.; Hertveldt, K.; Ackermann, H.W. “phiKZ-like viruses”, a proposed new genus of myovirus bacteriophages. Arch. Virol. 2007, 152, 1955–1959. [Google Scholar] [CrossRef]
- Bhunchoth, A.; Blanc-Mathieu, R.; Mihara, T.; Nishimura, Y.; Askora, A.; Phironrit, N.; Leksomboon, C.; Chatchawankanphanich, O.; Kawasaki, T.; Nakano, M.; et al. Two asian jumbo phages, phiRSL2 and phiRSF1, infect Ralstonia solanacearum and show common features of phiKZ-related phages. Virology 2016, 494, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Monson, R.; Foulds, I.; Foweraker, J.; Welch, M.; Salmond, G.P.C. The Pseudomonas aeruginosa generalized transducing phage phiPA3 is a new member of the phiKZ-like group of ‘jumbo’ phages, and infects model laboratory strains and clinical isolates from cystic fibrosis patients. Microbiology 2011, 157 Pt 3, 859–867. [Google Scholar] [CrossRef]
- Lee, J.H.; Shin, H.; Kim, H.; Ryu, S. Complete genome sequence of Salmonella bacteriophage SPN3US. J. Virol. 2011, 85, 13470–13471. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, A.; Hardies, S.C.; Shaburova, O.V.; Krylov, V.N.; Mattheus, W.; Kropinski, A.M.; Lavigne, R. Complete genome sequence of the giant virus OBP and comparative genome analysis of the diverse PhiKZ-related phages. J. Virol. 2012, 86, 1844–1852. [Google Scholar] [CrossRef] [PubMed]
- Skurnik, M.; Hyytiainen, H.J.; Happonen, L.J.; Kiljunen, S.; Datta, N.; Mattinen, L.; Williamson, K.; Kristo, P.; Szeliga, M.; Kalin-Manttari, L.; et al. Characterization of the genome, proteome, and structure of yersiniophage varphiR1-37. J. Virol. 2012, 86, 12625–12642. [Google Scholar] [CrossRef]
- Domotor, D.; Becsagh, P.; Rakhely, G.; Schneider, G.; Kovacs, T. Complete genomic sequence of Erwinia amylovora phage PhiEaH2. J. Virol. 2012, 86, 10899. [Google Scholar] [CrossRef]
- Meczker, K.; Domotor, D.; Vass, J.; Rakhely, G.; Schneider, G.; Kovacs, T. The genome of the Erwinia amylovora phage PhiEaH1 reveals greater diversity and broadens the applicability of phages for the treatment of fire blight. FEMS Microbiol. Lett. 2014, 350, 25–27. [Google Scholar] [CrossRef]
- Yagubi, A.I.; Castle, A.J.; Kropinski, A.M.; Banks, T.W.; Svircev, A.M. Complete Genome Sequence of Erwinia amylovora Bacteriophage vB_EamM_Ea35-70. Genome Announc. 2014, 2, e00413–e00414. [Google Scholar] [CrossRef]
- Yuan, Y.; Gao, M. Proteomic Analysis of a Novel Bacillus Jumbo Phage Revealing Glycoside Hydrolase As Structural Component. Front. Microbiol. 2016, 7, 745. [Google Scholar] [CrossRef]
- Danis-Wlodarczyk, K.; Vandenheuvel, D.; Jang, H.B.; Briers, Y.; Olszak, T.; Arabski, M.; Wasik, S.; Drabik, M.; Higgins, G.; Tyrrell, J.; et al. A proposed integrated approach for the preclinical evaluation of phage therapy in Pseudomonas infections. Sci. Rep. 2016, 6, 28115. [Google Scholar] [CrossRef]
- Lee, J.H.; Bai, J.; Shin, H.; Kim, Y.; Park, B.; Heu, S.; Ryu, S. A Novel Bacteriophage Targeting Cronobacter sakazakii Is a Potential Biocontrol Agent in Foods. Appl. Environ. Microbiol. 2016, 82, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Esplin, I.N.D.; Berg, J.A.; Sharma, R.; Allen, R.C.; Arens, D.K.; Ashcroft, C.R.; Bairett, S.R.; Beatty, N.J.; Bickmore, M.; Bloomfield, T.J.; et al. Genome Sequences of 19 Novel Erwinia amylovora Bacteriophages. Genome Announc. 2017, 5. [Google Scholar] [CrossRef]
- Matsui, T.; Yoshikawa, G.; Mihara, T.; Chatchawankanphanich, O.; Kawasaki, T.; Nakano, M.; Fujie, M.; Ogata, H.; Yamada, T. Replications of Two Closely Related Groups of Jumbo Phages Show Different Level of Dependence on Host-encoded RNA Polymerase. Front. Microbiol. 2017, 8, 1010. [Google Scholar] [CrossRef] [PubMed]
- Wojtus, J.K.; Fitch, J.L.; Christian, E.; Dalefield, T.; Lawes, J.K.; Kumar, K.; Peebles, C.L.; Altermann, E.; Hendrickson, H.L. Complete Genome Sequences of Three Novel Pseudomonas fluorescens SBW25 Bacteriophages, Noxifer, Phabio, and Skulduggery. Genome Announc. 2017, 5. [Google Scholar] [CrossRef] [PubMed]
- Latz, S.; Kruttgen, A.; Hafner, H.; Buhl, E.M.; Ritter, K.; Horz, H.P. Differential Effect of Newly Isolated Phages Belonging to PB1-Like, phiKZ-Like and LUZ24-Like Viruses against Multi-Drug Resistant Pseudomonas aeruginosa under Varying Growth Conditions. Viruses 2017, 9, 315. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.G.; Jun, J.W.; Giri, S.S.; Yun, S.; Kim, H.J.; Kim, S.W.; Kang, J.W.; Han, S.J.; Jeong, D.; Park, S.C. Isolation and characterisation of pVa-21, a giant bacteriophage with anti-biofilm potential against Vibrio alginolyticus. Sci. Rep. 2019, 9, 6284. [Google Scholar] [CrossRef]
- Wojtus, J.K.; Frampton, R.A.; Warring, S.; Hendrickson, H.; Fineran, P.C. Genome Sequence of a Jumbo Bacteriophage That Infects the Kiwifruit Phytopathogen Pseudomonas syringae pv. actinidiae. Microbiol. Resour. Announc. 2019, 8. [Google Scholar] [CrossRef]
- Korf, I.H.E.; Meier-Kolthoff, J.P.; Adriaenssens, E.M.; Kropinski, A.M.; Nimtz, M.; Rohde, M.; van Raaij, M.J.; Wittmann, J. Still Something to Discover: Novel Insights intoEscherichia coli Phage Diversity and Taxonomy. Viruses 2019, 11, 454. [Google Scholar] [CrossRef]
- Price, L.; Rohren, M.; Newkirk, H.; Liu, M.; Ramsey, J. Complete Genome Sequence of Serratia marcescens Myophage Moabite. Microbiol. Resour. Announc. 2019, 8. [Google Scholar] [CrossRef]
- Lewis, R.; Clooney, A.G.; Stockdale, S.R.; Buttimer, C.; Draper, L.A.; Ross, R.P.; Hill, C. Isolation of a Novel Jumbo Bacteriophage Effective Against Klebsiella aerogenes. Front. Med. 2020, 7, 67. [Google Scholar] [CrossRef]
- Ong, S.P.; Azam, A.H.; Sasahara, T.; Miyanaga, K.; Tanji, Y. Characterization of Pseudomonas lytic phages and their application as a cocktail with antibiotics in controlling Pseudomonas aeruginosa. J. Biosci. Bioeng. 2020, 129, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Malone, L.M.; Warring, S.L.; Jackson, S.A.; Warnecke, C.; Gardner, P.P.; Gumy, L.F.; Fineran, P.C. A jumbo phage that forms a nucleus-like structure evades CRISPR-Cas DNA targeting but is vulnerable to type III RNA-based immunity. Nat. Microbiol. 2020, 5, 48–55. [Google Scholar] [CrossRef] [PubMed]
- Simoliunas, E.; Kaliniene, L.; Truncaite, L.; Zajanckauskaite, A.; Staniulis, J.; Kaupinis, A.; Ger, M.; Valius, M.; Meskys, R. Klebsiella phage vB_KleM-RaK2-a giant singleton virus of the family Myoviridae. PLoS ONE 2013, 8, e60717. [Google Scholar] [CrossRef]
- Abbasifar, R.; Griffiths, M.W.; Sabour, P.M.; Ackermann, H.W.; Vandersteegen, K.; Lavigne, R.; Noben, J.P.; Alanis Villa, A.; Abbasifar, A.; Nash, J.H.; et al. Supersize me: Cronobacter sakazakii phage GAP32. Virology 2014, 460–461, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.J.; Lin, T.L.; Chen, C.C.; Tsai, Y.T.; Cheng, Y.H.; Chen, Y.Y.; Hsieh, P.F.; Lin, Y.T.; Wang, J.T. Klebsiella Phage PhiK64-1 Encodes Multiple Depolymerases for Multiple Host Capsular Types. J. Virol. 2017, 91. [Google Scholar] [CrossRef]
- Buttimer, C.; Hendrix, H.; Oliveira, H.; Casey, A.; Neve, H.; McAuliffe, O.; Ross, R.P.; Hill, C.; Noben, J.P.; O’Mahony, J.; et al. Things Are Getting Hairy: Enterobacteria Bacteriophage vB_PcaM_CBB. Front. Microbiol. 2017, 8, 44. [Google Scholar] [CrossRef]
- Evseev, P.; Sykilinda, N.; Gorshkova, A.; Kurochkina, L.; Ziganshin, R.; Drucker, V.; Miroshnikov, K. Pseudomonas Phage PaBG-A Jumbo Member of an Old Parasite Family. Viruses 2020, 12, 721. [Google Scholar] [CrossRef]
- Takahashi, I.; Marmur, J. Replacement of thymidylic acid by deoxyuridylic acid in the deoxyribonucleic acid of a transducing phage for Bacillus subtilis. Nature 1963, 197, 794–795. [Google Scholar] [CrossRef]
- Uchiyama, J.; Takemura-Uchiyama, I.; Sakaguchi, Y.; Gamoh, K.; Kato, S.; Daibata, M.; Ujihara, T.; Misawa, N.; Matsuzaki, S. Intragenus generalized transduction in Staphylococcus spp. by a novel giant phage. ISME J. 2014, 8, 1949–1952. [Google Scholar] [CrossRef]
- Chaikeeratisak, V.; Nguyen, K.; Khanna, K.; Brilot, A.F.; Erb, M.L.; Coker, J.K.; Vavilina, A.; Newton, G.L.; Buschauer, R.; Pogliano, K.; et al. Assembly of a nucleus-like structure during viral replication in bacteria. Science 2017, 355, 194–197. [Google Scholar] [CrossRef]
- Chaikeeratisak, V.; Nguyen, K.; Egan, M.E.; Erb, M.L.; Vavilina, A.; Pogliano, J. The Phage Nucleus and Tubulin Spindle Are Conserved among Large Pseudomonas Phages. Cell Rep. 2017, 20, 1563–1571. [Google Scholar]
- Mendoza, S.D.; Nieweglowska, E.S.; Govindarajan, S.; Leon, L.M.; Berry, J.D.; Tiwari, A.; Chaikeeratisak, V.; Pogliano, J.; Agard, D.A.; Bondy-Denomy, J. A bacteriophage nucleus-like compartment shields DNA from CRISPR nucleases. Nature 2020, 577, 244–248. [Google Scholar]
- Ceyssens, P.J.; Minakhin, L.; Van den Bossche, A.; Yakunina, M.; Klimuk, E.; Blasdel, B.; De Smet, J.; Noben, J.P.; Blasi, U.; Severinov, K.; et al. Development of giant bacteriophage varphiKZ is independent of the host transcription apparatus. J. Virol. 2014, 88, 10501–10510. [Google Scholar]
- Lavysh, D.; Sokolova, M.; Slashcheva, M.; Forstner, K.U.; Severinov, K. Transcription Profiling of Bacillus subtilis Cells Infected with AR9, a Giant Phage Encoding Two Multisubunit RNA Polymerases. MBio 2017, 8. [Google Scholar] [CrossRef]
- Coote, J.G.; Wood, D.A.; Mandelstam, J. Lethal effect of rifampicin in Bacillus subtilis as a complicating factor in the assessment of the lifetime of messenger ribonucleic acid. Biochem. J. 1973, 134, 263–270. [Google Scholar]
- Leskinen, K.; Blasdel, B.G.; Lavigne, R.; Skurnik, M. RNA-Sequencing Reveals the Progression of Phage-Host Interactions between phiR1-37 and Yersinia enterocolitica. Viruses 2016, 8, 111. [Google Scholar]
- Lecoutere, E.; Ceyssens, P.J.; Miroshnikov, K.A.; Mesyanzhinov, V.V.; Krylov, V.N.; Noben, J.P.; Robben, J.; Hertveldt, K.; Volckaert, G.; Lavigne, R. Identification and comparative analysis of the structural proteomes of phiKZ and EL, two giant Pseudomonas aeruginosa bacteriophages. Proteomics 2009, 9, 3215–3219. [Google Scholar]
- Thomas, J.A.; Weintraub, S.T.; Hakala, K.; Serwer, P.; Hardies, S.C. Proteome of the large Pseudomonas myovirus 201 phi 2-1: Delineation of proteolytically processed virion proteins. Mol. Cell. Proteom. 2010, 9, 940–951. [Google Scholar]
- Lane, W.J.; Darst, S.A. Molecular evolution of multisubunit RNA polymerases: Sequence analysis. J. Mol. Biol. 2010, 395, 671–685. [Google Scholar]
- Thomas, J.A.; Benitez Quintana, A.D.; Bosch, M.A.; Coll De Pena, A.; Aguilera, E.; Coulibaly, A.; Wu, W.; Osier, M.V.; Hudson, A.O.; Weintraub, S.T.; et al. Identification of Essential Genes in the Salmonella Phage SPN3US Reveals Novel Insights into Giant Phage Head Structure and Assembly. J. Virol. 2016, 90, 10284–10298. [Google Scholar]
- Aravind, L.; Koonin, E.V. The HD domain defines a new superfamily of metal-dependent phosphohydrolases. Trends Biochem. Sci. 1998, 23, 469–472. [Google Scholar] [CrossRef]
- Xie, W.Q.; Jager, K.; Potts, M. Cyanobacterial RNA polymerase genes rpoC1 and rpoC2 correspond to rpoC of Escherichia coli. J. Bacteriol. 1989, 171, 1967–1973. [Google Scholar] [CrossRef]
- Sokolova, M. Functional and Structural Analysis of a Non-Canonical Multisubunit RNA Polymerase Encoded by Giant Bacteriophage AR9. Ph.D. Thesis, Skolkovo Institute of Science and Technology, Moscow, Russia, 2018. [Google Scholar]
- Feklistov, A. RNA polymerase: In search of promoters. Ann. N. Y. Acad. Sci. 2013, 1293, 25–32. [Google Scholar] [CrossRef]
- Feklistov, A.; Darst, S.A. Structural basis for promoter-10 element recognition by the bacterial RNA polymerase sigma subunit. Cell 2011, 147, 1257–1269. [Google Scholar] [CrossRef]
- Bae, B.; Feklistov, A.; Lass-Napiorkowska, A.; Landick, R.; Darst, S.A. Structure of a bacterial RNA polymerase holoenzyme open promoter complex. Elife 2015, 4, e08504. [Google Scholar]
- Campagne, S.; Marsh, M.E.; Capitani, G.; Vorholt, J.A.; Allain, F.H. Structural basis for -10 promoter element melting by environmentally induced sigma factors. Nat. Struct. Mol. Biol. 2014, 21, 269–276. [Google Scholar]
- Zhilina, E.; Esyunina, D.; Brodolin, K.; Kulbachinskiy, A. Structural transitions in the transcription elongation complexes of bacterial RNA polymerase during sigma-dependent pausing. Nucleic Acids Res. 2012, 40, 3078–3091. [Google Scholar]
- Toulokhonov, I.; Landick, R. The role of the lid element in transcription by E. coli RNA polymerase. J. Mol. Biol. 2006, 361, 644–658. [Google Scholar]
- Naryshkina, T.; Kuznedelov, K.; Severinov, K. The role of the largest RNA polymerase subunit lid element in preventing the formation of extended RNA-DNA hybrid. J. Mol. Biol. 2006, 361, 634–643. [Google Scholar]
- Weinheimer, A.R.; Aylward, F.O. A distinct lineage of Caudovirales that encodes a deeply branching multi-subunit RNA polymerase. Nat. Commun. 2020, 11, 4506. [Google Scholar]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sokolova, M.L.; Misovetc, I.; V. Severinov, K. Multisubunit RNA Polymerases of Jumbo Bacteriophages. Viruses 2020, 12, 1064. https://doi.org/10.3390/v12101064
Sokolova ML, Misovetc I, V. Severinov K. Multisubunit RNA Polymerases of Jumbo Bacteriophages. Viruses. 2020; 12(10):1064. https://doi.org/10.3390/v12101064
Chicago/Turabian StyleSokolova, Maria L., Inna Misovetc, and Konstantin V. Severinov. 2020. "Multisubunit RNA Polymerases of Jumbo Bacteriophages" Viruses 12, no. 10: 1064. https://doi.org/10.3390/v12101064
APA StyleSokolova, M. L., Misovetc, I., & V. Severinov, K. (2020). Multisubunit RNA Polymerases of Jumbo Bacteriophages. Viruses, 12(10), 1064. https://doi.org/10.3390/v12101064




