Insights into Thymus Development and Viral Thymic Infections
Abstract
1. Introduction
2. Thymocyte Development and Related Transcription Factors
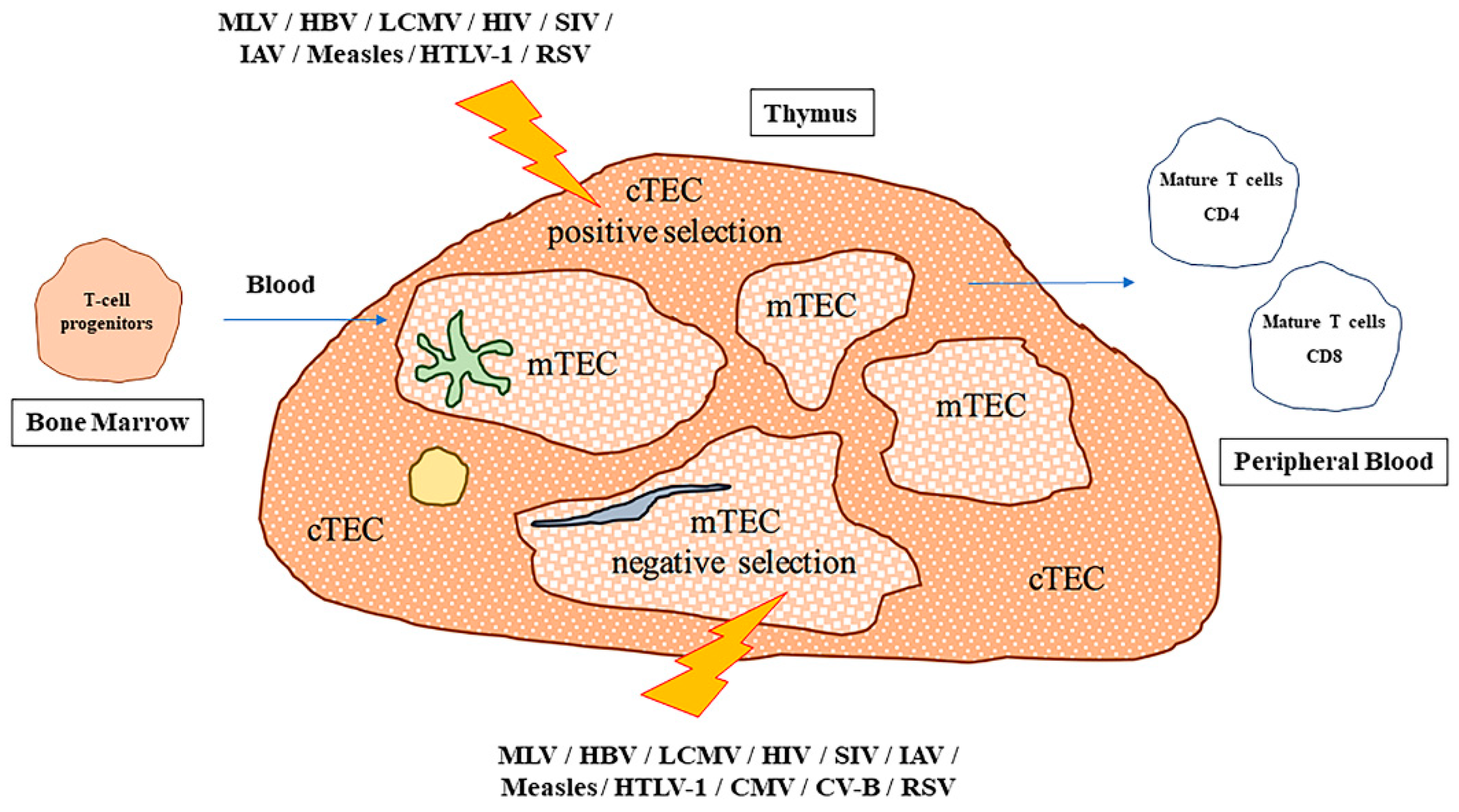
3. Immunological Consequences of Viral Infections of the Thymus
4. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Zdrojewicz, Z.; Pachura, E.; Pachura, P. The thymus: A forgotten, but very important organ. Adv. Clin. Exp. Med. 2016, 25, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Flores, K.G.; Li, J.; Sempowski, G.D.; Haynes, B.F.; Hale, L.P. Analysis of the human thymic perivascular space during aging. J. Clin. Investig. 1999, 104, 1031–1039. [Google Scholar] [CrossRef] [PubMed]
- Vosshenrich, C.A.; García-Ojeda, M.E.; Samson-Villéger, S.I.; Pasqualetto, V.; Enault, L.; Richard-Le Goff, O.; Corcuff, E.; Guy-Grand, D.; Rocha, B.; Cumano, A.; et al. A thymic pathway of mouse natural killer cell development characterized by expression of gata-3 and cd127. Nat. Immunol. 2006, 7, 1217–1224. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, V.S.; Hasan, M.; Wilson, A.; Boucontet, L.; Pereira, P.; Lesjean-Pottier, S.; Satoh-Takayama, N.; Di Santo, J.P.; Vosshenrich, C.A. Cutting edge: Thymic nk cells develop independently from t cell precursors. J. Immunol. 2010, 185, 4993–4997. [Google Scholar] [CrossRef] [PubMed]
- Vargas, C.L.; Poursine-Laurent, J.; Yang, L.; Yokoyama, W.M. Development of thymic nk cells from double negative 1 thymocyte precursors. Blood 2011, 118, 3570–3578. [Google Scholar] [CrossRef] [PubMed]
- White, A.J.; Baik, S.; Parnell, S.M.; Holland, A.M.; Brombacher, F.; Jenkinson, W.E.; Anderson, G. A type 2 cytokine axis for thymus emigration. J. Exp. Med. 2017, 214, 2205–2216. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Wang, H.; Starrett, G.J.; Phuong, V.; Jameson, S.C.; Hogquist, K.A. Tissue-specific distribution of inkt cells impacts their cytokine response. Immunity 2015, 43, 566–578. [Google Scholar] [CrossRef]
- Ferlazzo, G.; Morandi, B. Cross-talks between natural killer cells and distinct subsets of dendritic cells. Front. Immunol. 2014, 5, 159. [Google Scholar] [CrossRef]
- Liu, L.T.; Lang, Z.F.; Li, Y.; Zhu, Y.J.; Zhang, J.T.; Guo, S.F.; Wang, J.X.; Wang, H.W.; Xu, Y.D. Composition and characteristics of distinct macrophage subpopulations in the mouse thymus. Mol. Med. Rep. 2013, 7, 1850–1854. [Google Scholar] [CrossRef][Green Version]
- Soga, H.; Nakamura, M.; Yagi, H.; Kayaba, S.; Ishii, T.; Gotoh, T.; Itoh, T. Heterogeneity of mouse thymic macrophages: I. Immunohistochemical analysis. Arch. Histol. Cytol. 1997, 60, 53–63. [Google Scholar] [CrossRef]
- Kroger, C.J.; Spidale, N.A.; Wang, B.; Tisch, R. Thymic dendritic cell subsets display distinct efficiencies and mechanisms of intercellular mhc transfer. J. Immunol. 2017, 198, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Shortman, K. Heterogeneity of thymic dendritic cells. Semin. Immunol. 2005, 17, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Rodewald, H.R.; Paul, S.; Haller, C.; Bluethmann, H.; Blum, C. Thymus medulla consisting of epithelial islets each derived from a single progenitor. Nature 2001, 414, 763–768. [Google Scholar] [CrossRef] [PubMed]
- Hamazaki, Y.; Fujita, H.; Kobayashi, T.; Choi, Y.; Scott, H.S.; Matsumoto, M.; Minato, N. Medullary thymic epithelial cells expressing aire represent a unique lineage derived from cells expressing claudin. Nat. Immunol. 2007, 8, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.W.; Jenkinson, W.E.; Anderson, G.; Jenkinson, E.J. Clonal analysis reveals a common progenitor for thymic cortical and medullary epithelium. Nature 2006, 441, 988–991. [Google Scholar] [CrossRef] [PubMed]
- Shakib, S.; Desanti, G.E.; Jenkinson, W.E.; Parnell, S.M.; Jenkinson, E.J.; Anderson, G. Checkpoints in the development of thymic cortical epithelial cells. J. Immunol. 2009, 182, 130–137. [Google Scholar] [CrossRef]
- Bleul, C.C.; Corbeaux, T.; Reuter, A.; Fisch, P.; Monting, J.S.; Boehm, T. Formation of a functional thymus initiated by a postnatal epithelial progenitor cell. Nature 2006, 441, 992–996. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, N.; Wang, Y.H.; Lee, H.K.; Ito, T.; Wang, Y.H.; Cao, W.; Liu, Y.J. Hassall’s corpuscles instruct dendritic cells to induce cd4+cd25+ regulatory t cells in human thymus. Nature 2005, 436, 1181–1185. [Google Scholar] [CrossRef]
- Ucar, A.; Ucar, O.; Klug, P.; Matt, S.; Brunk, F.; Hofmann, T.G.; Kyewski, B. Adult thymus contains foxn1(-) epithelial stem cells that are bipotent for medullary and cortical thymic epithelial lineages. Immunity 2014, 41, 257–269. [Google Scholar] [CrossRef]
- Guyden, J.C.; Martinez, M.; Chilukuri, R.V.; Reid, V.; Kelly, F.; Samms, M.O. Thymic nurse cells participate in heterotypic internalization and repertoire selection of immature thymocytes; their removal from the thymus of autoimmune animals may be important to disease etiology. Curr. Mol. Med. 2015, 15, 828–835. [Google Scholar] [CrossRef]
- Wekerle, H.; Ketelsen, U.P. Thymic nurse cells--ia-bearing epithelium involved in t-lymphocyte differentiation? Nature 1980, 283, 402–404. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, Y.; Ohigashi, I.; Nitta, T.; Sakata, M.; Tanaka, K.; Murata, S.; Kanagawa, O.; Takahama, Y. Thymic nurse cells provide microenvironment for secondary t cell receptor alpha rearrangement in cortical thymocytes. Proc. Natl. Acad. Sci. USA 2012, 109, 20572–20577. [Google Scholar] [CrossRef] [PubMed]
- Akashi, K.; Richie, L.I.; Miyamoto, T.; Carr, W.H.; Weissman, I.L. B lymphopoiesis in the thymus. J. Immunol. 2000, 164, 5221–5226. [Google Scholar] [CrossRef] [PubMed]
- Radtke, F.; Wilson, A.; Stark, G.; Bauer, M.; van Meerwijk, J.; MacDonald, H.R.; Aguet, M. Deficient t cell fate specification in mice with an induced inactivation of notch1. Immunity 1999, 10, 547–558. [Google Scholar] [CrossRef]
- Wilson, A.; MacDonald, H.R.; Radtke, F. Notch 1-deficient common lymphoid precursors adopt a b cell fate in the thymus. J. Exp. Med. 2001, 194, 1003–1012. [Google Scholar] [CrossRef] [PubMed]
- Feyerabend, T.B.; Terszowski, G.; Tietz, A.; Blum, C.; Luche, H.; Gossler, A.; Gale, N.W.; Radtke, F.; Fehling, H.J.; Rodewald, H.R. Deletion of notch1 converts pro-t cells to dendritic cells and promotes thymic b cells by cell-extrinsic and cell-intrinsic mechanisms. Immunity 2009, 30, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Perera, J.; Huang, H. The development and function of thymic b cells. CMLS 2015, 72, 2657–2663. [Google Scholar] [CrossRef] [PubMed]
- Yamano, T.; Nedjic, J.; Hinterberger, M.; Steinert, M.; Koser, S.; Pinto, S.; Gerdes, N.; Lutgens, E.; Ishimaru, N.; Busslinger, M.; et al. Thymic b cells are licensed to present self antigens for central t cell tolerance induction. Immunity 2015, 42, 1048–1061. [Google Scholar] [CrossRef] [PubMed]
- Haddad, R.; Guimiot, F.; Six, E.; Jourquin, F.; Setterblad, N.; Kahn, E.; Yagello, M.; Schiffer, C.; Andre-Schmutz, I.; Cavazzana-Calvo, M.; et al. Dynamics of thymus-colonizing cells during human development. Immunity 2006, 24, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Six, E.M.; Bonhomme, D.; Monteiro, M.; Beldjord, K.; Jurkowska, M.; Cordier-Garcia, C.; Garrigue, A.; Dal Cortivo, L.; Rocha, B.; Fischer, A.; et al. A human postnatal lymphoid progenitor capable of circulating and seeding the thymus. J. Exp. Med. 2007, 204, 3085–3093. [Google Scholar] [CrossRef] [PubMed]
- Famili, F.; Wiekmeijer, A.S.; Staal, F.J. The development of t cells from stem cells in mice and humans. Future Sci. OA 2017, 3, FSO186. [Google Scholar] [CrossRef] [PubMed]
- Takada, K.; Takahama, Y. Positive-selection-inducing self-peptides displayed by cortical thymic epithelial cells. Adv. Immunol. 2015, 125, 87–110. [Google Scholar] [PubMed]
- Anderson, G.; Takahama, Y. Thymic epithelial cells: Working class heroes for t cell development and repertoire selection. Trends Immunol. 2012, 33, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Takahama, Y.; Takada, K.; Murata, S.; Tanaka, K. Beta5t-containing thymoproteasome: Specific expression in thymic cortical epithelial cells and role in positive selection of cd8+ t cells. Curr. Opin. Immunol. 2012, 24, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Singer, A.; Adoro, S.; Park, J.H. Lineage fate and intense debate: Myths, models and mechanisms of cd4- versus cd8-lineage choice. Nat. Rev. Immunol. 2008, 8, 788–801. [Google Scholar] [CrossRef] [PubMed]
- Blom, B.; Verschuren, M.C.; Heemskerk, M.H.; Bakker, A.Q.; van Gastel-Mol, E.J.; Wolvers-Tettero, I.L.; van Dongen, J.J.; Spits, H. Tcr gene rearrangements and expression of the pre-t cell receptor complex during human t-cell differentiation. Blood 1999, 93, 3033–3043. [Google Scholar] [PubMed]
- Carrasco, Y.R.; Navarro, M.N.; de Yebenes, V.G.; Ramiro, A.R.; Toribio, M.L. Regulation of surface expression of the human pre-t cell receptor complex. Semin. Immunol. 2002, 14, 325–334. [Google Scholar] [CrossRef]
- Taghon, T.; Yui, M.A.; Pant, R.; Diamond, R.A.; Rothenberg, E.V. Developmental and molecular characterization of emerging beta- and gammadelta-selected pre-t cells in the adult mouse thymus. Immunity 2006, 24, 53–64. [Google Scholar] [CrossRef]
- Csaba, G. The immunoendocrine thymus as a pacemaker of lifespan. Acta Microbiol. Immunol. Hung. 2016, 63, 139–158. [Google Scholar] [CrossRef]
- Miller, J.F. The golden anniversary of the thymus. Nat. Rev. Immunol. 2011, 11, 489–495. [Google Scholar] [CrossRef]
- Davis, M.M.; Boniface, J.J.; Reich, Z.; Lyons, D.; Hampl, J.; Arden, B.; Chien, Y. Ligand recognition by alpha beta t cell receptors. Annu. Rev. Immunol. 1998, 16, 523–544. [Google Scholar] [CrossRef] [PubMed]
- Takaba, H.; Takayanagi, H. The mechanisms of t cell selection in the thymus. Trends Immunol. 2017, 38, 805–816. [Google Scholar] [CrossRef] [PubMed]
- Murata, S.; Sasaki, K.; Kishimoto, T.; Niwa, S.; Hayashi, H.; Takahama, Y.; Tanaka, K. Regulation of cd8+ t cell development by thymus-specific proteasomes. Science 2007, 316, 1349–1353. [Google Scholar] [CrossRef] [PubMed]
- Forster, R.; Davalos-Misslitz, A.C.; Rot, A. Ccr7 and its ligands: Balancing immunity and tolerance. Nat. Rev. Immunol. 2008, 8, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Klein, L.; Kyewski, B.; Allen, P.M.; Hogquist, K.A. Positive and negative selection of the t cell repertoire: What thymocytes see (and don’t see). Nat. Rev. Immunol. 2014, 14, 377–391. [Google Scholar] [CrossRef] [PubMed]
- Stritesky, G.L.; Jameson, S.C.; Hogquist, K.A. Selection of self-reactive t cells in the thymus. Annu. Rev. Immunol. 2012, 30, 95–114. [Google Scholar] [CrossRef]
- McCaughtry, T.M.; Baldwin, T.A.; Wilken, M.S.; Hogquist, K.A. Clonal deletion of thymocytes can occur in the cortex with no involvement of the medulla. J. Exp. Med. 2008, 205, 2575–2584. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Rudra, D. Emerging functions of regulatory t cells in tissue homeostasis. Front. Immunol. 2018, 9, 883. [Google Scholar] [CrossRef]
- Moran, A.E.; Hogquist, K.A. T-cell receptor affinity in thymic development. Immunology 2012, 135, 261–267. [Google Scholar] [CrossRef]
- Nemazee, D. Receptor editing in lymphocyte development and central tolerance. Nat. Rev. Immunol. 2006, 6, 728–740. [Google Scholar] [CrossRef]
- Kreslavsky, T.; Kim, H.J.; Koralov, S.B.; Ghitza, D.; Buch, T.; Cantor, H.; Rajewsky, K.; von Boehmer, H. Negative selection, not receptor editing, is a physiological response of autoreactive thymocytes. J. Exp. Med. 2013, 210, 1911–1918. [Google Scholar] [CrossRef] [PubMed]
- Surh, C.D.; Sprent, J. T-cell apoptosis detected in situ during positive and negative selection in the thymus. Nature 1994, 372, 100–103. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P.L.; Eisenberg, R.A. Lpr and gld: Single gene models of systemic autoimmunity and lymphoproliferative disease. Annu. Rev. Immunol. 1991, 9, 243–269. [Google Scholar] [CrossRef] [PubMed]
- Miller, C.N.; Proekt, I.; von Moltke, J.; Wells, K.L.; Rajpurkar, A.R.; Wang, H.; Rattay, K.; Khan, I.S.; Metzger, T.C.; Pollack, J.L.; et al. Thymic tuft cells promote an il-4-enriched medulla and shape thymocyte development. Nature 2018, 559, 627–631. [Google Scholar] [CrossRef] [PubMed]
- Bornstein, C.; Nevo, S.; Giladi, A.; Kadouri, N.; Pouzolles, M.; Gerbe, F.; David, E.; Machado, A.; Chuprin, A.; Toth, B.; et al. Single-cell mapping of the thymic stroma identifies il-25-producing tuft epithelial cells. Nature 2018, 559, 622–626. [Google Scholar] [CrossRef] [PubMed]
- Weerkamp, F.; Baert, M.R.; Brugman, M.H.; Dik, W.A.; de Haas, E.F.; Visser, T.P.; de Groot, C.J.; Wagemaker, G.; van Dongen, J.J.; Staal, F.J. Human thymus contains multipotent progenitors with t/b lymphoid, myeloid, and erythroid lineage potential. Blood 2006, 107, 3131–3137. [Google Scholar] [CrossRef] [PubMed]
- Kelly, A.P.; Finlay, D.K.; Hinton, H.J.; Clarke, R.G.; Fiorini, E.; Radtke, F.; Cantrell, D.A. Notch-induced t cell development requires phosphoinositide-dependent kinase 1. EMBO J. 2007, 26, 3441–3450. [Google Scholar] [CrossRef] [PubMed]
- Love, P.E.; Bhandoola, A. Signal integration and crosstalk during thymocyte migration and emigration. Nat. Rev. Immunol. 2011, 11, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Radtke, F.; Fasnacht, N.; Macdonald, H.R. Notch signaling in the immune system. Immunity 2010, 32, 14–27. [Google Scholar] [CrossRef]
- Weber, B.N.; Chi, A.W.; Chavez, A.; Yashiro-Ohtani, Y.; Yang, Q.; Shestova, O.; Bhandoola, A. A critical role for tcf-1 in t-lineage specification and differentiation. Nature 2011, 476, 63–68. [Google Scholar] [CrossRef]
- Germar, K.; Dose, M.; Konstantinou, T.; Zhang, J.; Wang, H.; Lobry, C.; Arnett, K.L.; Blacklow, S.C.; Aifantis, I.; Aster, J.C.; et al. T-cell factor 1 is a gatekeeper for t-cell specification in response to notch signaling. Proc. Natl. Acad. Sci. USA 2011, 108, 20060–20065. [Google Scholar] [CrossRef] [PubMed]
- Rothenberg, E.V. Transcriptional control of early t and b cell developmental choices. Annu. Rev. Immunol. 2014, 32, 283–321. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.K.; Hernandez-Hoyos, G.; Dionne, C.J.; Arias, A.M.; Chen, D.; Rothenberg, E.V. Definition of regulatory network elements for t cell development by perturbation analysis with pu.1 and gata-3. Dev. Biol. 2002, 246, 103–121. [Google Scholar] [CrossRef] [PubMed]
- Scripture-Adams, D.D.; Damle, S.S.; Li, L.; Elihu, K.J.; Qin, S.; Arias, A.M.; Butler, R.R., 3rd; Champhekar, A.; Zhang, J.A.; Rothenberg, E.V. Gata-3 dose-dependent checkpoints in early t cell commitment. J. Immunol. 2014, 193, 3470–3491. [Google Scholar] [CrossRef] [PubMed]
- Taghon, T.; Yui, M.A.; Rothenberg, E.V. Mast cell lineage diversion of t lineage precursors by the essential t cell transcription factor gata–3. Nat. Immunol. 2007, 8, 845–855. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Leid, M.; Rothenberg, E.V. An early t cell lineage commitment checkpoint dependent on the transcription factor bcl11b. Science 2010, 329, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhang, J.A.; Dose, M.; Kueh, H.Y.; Mosadeghi, R.; Gounari, F.; Rothenberg, E.V. A far downstream enhancer for murine bcl11b controls its t-cell specific expression. Blood 2013, 122, 902–911. [Google Scholar] [CrossRef]
- Ikawa, T.; Hirose, S.; Masuda, K.; Kakugawa, K.; Satoh, R.; Shibano-Satoh, A.; Kominami, R.; Katsura, Y.; Kawamoto, H. An essential developmental checkpoint for production of the t cell lineage. Science 2010, 329, 93–96. [Google Scholar] [CrossRef]
- Rothenberg, E.V.; Champhekar, A.; Damle, S.; Del Real, M.M.; Kueh, H.Y.; Li, L.; Yui, M.A. Transcriptional establishment of cell-type identity: Dynamics and causal mechanisms of t-cell lineage commitment. Cold Spring Harb. Symp. Quant. Biol. 2013, 78, 31–41. [Google Scholar] [CrossRef]
- Hosokawa, H.; Rothenberg, E.V. Cytokines, transcription factors, and the initiation of t-cell development. Cold Spring Harb. Perspec. Biol. 2018, 10, a028621. [Google Scholar] [CrossRef]
- Steinke, F.C.; Yu, S.; Zhou, X.; He, B.; Yang, W.; Zhou, B.; Kawamoto, H.; Zhu, J.; Tan, K.; Xue, H.H. Tcf-1 and lef-1 act upstream of th-pok to promote the cd4 (+) t cell fate and interact with runx3 to silence cd4 in cd8 (+) t cells. Nat. Immunol. 2014, 15, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Stros, M.; Launholt, D.; Grasser, K.D. The hmg-box: A versatile protein domain occurring in a wide variety of DNA-binding proteins. Cell Mol. Life Sci. 2007, 64, 2590–2606. [Google Scholar] [CrossRef] [PubMed]
- Donato, G.; Martinez Hoyos, J.; Amorosi, A.; Maltese, L.; Lavano, A.; Volpentesta, G.; Signorelli, F.; Pentimalli, F.; Pallante, P.; Ferraro, G.; et al. High mobility group a1 expression correlates with the histological grade of human glial tumors. Oncol. Rep. 2004, 11, 1209–1213. [Google Scholar] [CrossRef] [PubMed]
- Mucida, D.; Husain, M.M.; Muroi, S.; van Wijk, F.; Shinnakasu, R.; Naoe, Y.; Reis, B.S.; Huang, Y.; Lambolez, F.; Docherty, M.; et al. Transcriptional reprogramming of mature cd4 (+) helper t cells generates distinct mhc class ii-restricted cytotoxic t lymphocytes. Nat. Immunol. 2013, 14, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Setoguchi, R.; Tachibana, M.; Naoe, Y.; Muroi, S.; Akiyama, K.; Tezuka, C.; Okuda, T.; Taniuchi, I. Repression of the transcription factor th-pok by runx complexes in cytotoxic t cell development. Science 2008, 319, 822–825. [Google Scholar] [CrossRef] [PubMed]
- Muroi, S.; Naoe, Y.; Miyamoto, C.; Akiyama, K.; Ikawa, T.; Masuda, K.; Kawamoto, H.; Taniuchi, I. Cascading suppression of transcriptional silencers by thpok seals helper t cell fate. Nat. Immunol. 2008, 9, 1113–1121. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, S.; Hainberger, D.; Tizian, C.; Tanaka, H.; Okuda, T.; Taniuchi, I.; Ellmeier, W. Mazr and runx factors synergistically repress thpok during cd8+ t cell lineage development. J. Immunol. 2015, 195, 2879–2887. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, S.; Hombauer, M.; Bilic, I.; Naoe, Y.; Schebesta, A.; Taniuchi, I.; Ellmeier, W. The zinc-finger protein mazr is part of the transcription factor network that controls the cd4 versus cd8 lineage fate of double-positive thymocytes. Nat. Immunol. 2010, 11, 442–448. [Google Scholar] [CrossRef] [PubMed]
- Xing, Y.; Wang, X.; Jameson, S.C.; Hogquist, K.A. Late stages of t cell maturation in the thymus involve nf-kappab and tonic type i interferon signaling. Nat. Immunol. 2016, 17, 565–573. [Google Scholar] [CrossRef]
- Fiume, G.; Rossi, A.; de Laurentiis, A.; Falcone, C.; Pisano, A.; Vecchio, E.; Pontoriero, M.; Scala, I.; Scialdone, A.; Masci, F.F.; et al. Eukaryotic initiation factor 4 h is under transcriptional control of p65/nf-kappab. PLoS ONE 2013, 8, e66087. [Google Scholar] [CrossRef]
- Pontoriero, M.; Fiume, G.; Vecchio, E.; de Laurentiis, A.; Albano, F.; Iaccino, E.; Mimmi, S.; Pisano, A.; Agosti, V.; Giovannone, E.; et al. Activation of nf-kappab in b cell receptor signaling through bruton’s tyrosine kinase-dependent phosphorylation of ikappab-alpha. J. Mol. Med. 2019, 97, 675–690. [Google Scholar] [CrossRef] [PubMed]
- Tai, T.S.; Pai, S.Y.; Ho, I.C. Gata-3 regulates the homeostasis and activation of cd8+ t cells. J. Immunol. 2013, 190, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Castro, E.; Yagi, R.; Zhu, J.; Lesourne, R.; Love, P.E.; Feigenbaum, L.; Bosselut, R. Thpok-independent repression of runx3 by gata3 during cd4+ t-cell differentiation in the thymus. Eur. J. Immunol. 2013, 43, 918–928. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Adoro, S.; Guinter, T.; Erman, B.; Alag, A.S.; Catalfamo, M.; Kimura, M.Y.; Cui, Y.; Lucas, P.J.; Gress, R.E.; et al. Signaling by intrathymic cytokines, not t cell antigen receptors, specifies cd8 lineage choice and promotes the differentiation of cytotoxic-lineage t cells. Nat. Immunol. 2010, 11, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Zamisch, M.; Tian, L.; Grenningloh, R.; Xiong, Y.; Wildt, K.F.; Ehlers, M.; Ho, I.C.; Bosselut, R. The transcription factor ets1 is important for cd4 repression and runx3 up-regulation during cd8 t cell differentiation in the thymus. J. Exp. Med. 2009, 206, 2685–2699. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, H.J.; Briones Leon, A.; Blackburn, C.C. Foxn1 in thymus organogenesis and development. Eur. J. Immunol. 2016, 46, 1826–1837. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.; Littman, D.R.; Taniuchi, I. Runx proteins in transcription factor networks that regulate t-cell lineage choice. Nat. Rev. Immunol. 2009, 9, 106–115. [Google Scholar] [CrossRef]
- Taniuchi, I.; Osato, M.; Egawa, T.; Sunshine, M.J.; Bae, S.C.; Komori, T.; Ito, Y.; Littman, D.R. Differential requirements for runx proteins in cd4 repression and epigenetic silencing during t lymphocyte development. Cell 2002, 111, 621–633. [Google Scholar] [CrossRef]
- McDonald, B.D.; Bunker, J.J.; Erickson, S.A.; Oh-Hora, M.; Bendelac, A. Crossreactive alphabeta t cell receptors are the predominant targets of thymocyte negative selection. Immunity 2015, 43, 859–869. [Google Scholar] [CrossRef]
- Woolf, E.; Xiao, C.; Fainaru, O.; Lotem, J.; Rosen, D.; Negreanu, V.; Bernstein, Y.; Goldenberg, D.; Brenner, O.; Berke, G.; et al. Runx3 and runx1 are required for cd8 t cell development during thymopoiesis. Proc. Natl. Acad. Sci. USA 2003, 100, 7731–7736. [Google Scholar] [CrossRef]
- Pobezinsky, L.A.; Angelov, G.S.; Tai, X.; Jeurling, S.; Van Laethem, F.; Feigenbaum, L.; Park, J.H.; Singer, A. Clonal deletion and the fate of autoreactive thymocytes that survive negative selection. Nat. Immunol. 2012, 13, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Egawa, T.; Tillman, R.E.; Naoe, Y.; Taniuchi, I.; Littman, D.R. The role of the runx transcription factors in thymocyte differentiation and in homeostasis of naive t cells. J. Exp. Med. 2007, 204, 1945–1957. [Google Scholar] [CrossRef] [PubMed]
- Bansal, K.; Yoshida, H.; Benoist, C.; Mathis, D. The transcriptional regulator aire binds to and activates super-enhancers. Nat. Immunol. 2017, 18, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Passos, G.A.; Speck-Hernandez, C.A.; Assis, A.F.; Mendes-da-Cruz, D.A. Update on aire and thymic negative selection. Immunology 2018, 153, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Takaba, H.; Morishita, Y.; Tomofuji, Y.; Danks, L.; Nitta, T.; Komatsu, N.; Kodama, T.; Takayanagi, H. Fezf2 orchestrates a thymic program of self-antigen expression for immune tolerance. Cell 2015, 163, 975–987. [Google Scholar] [CrossRef] [PubMed]
- Eckler, M.J.; Chen, B. Fez family transcription factors: Controlling neurogenesis and cell fate in the developing mammalian nervous system. BioEssays News Rev. Zmol. Cell. Dev. Biol. 2014, 36, 788–797. [Google Scholar] [CrossRef] [PubMed]
- Savino, W. The thymus is a common target organ in infectious diseases. PLoS Pathog. 2006, 2, e62. [Google Scholar] [CrossRef] [PubMed]
- Nunez, S.; Moore, C.; Gao, B.; Rogers, K.; Hidalgo, Y.; Del Nido, P.J.; Restaino, S.; Naka, Y.; Bhagat, G.; Madsen, J.C.; et al. The human thymus perivascular space is a functional niche for viral-specific plasma cells. Sci. Immunol. 2016, 1, e4447. [Google Scholar] [CrossRef]
- Korostoff, J.M.; Nakada, M.T.; Faas, S.J.; Blank, K.J.; Gaulton, G.N. Neonatal exposure to thymotropic gross murine leukemia virus induces virus-specific immunologic nonresponsiveness. J. Exp. Med. 1990, 172, 1765–1775. [Google Scholar] [CrossRef]
- Nunes-Alves, C.; Nobrega, C.; Behar, S.M.; Correia-Neves, M. Tolerance has its limits: How the thymus copes with infection. Trends Immunol. 2013, 34, 502–510. [Google Scholar] [CrossRef]
- Ahmed, R.; King, C.C.; Oldstone, M.B. Virus-lymphocyte interaction: T cells of the helper subset are infected with lymphocytic choriomeningitis virus during persistent infection in vivo. J. Virol. 1987, 61, 1571–1576. [Google Scholar] [PubMed]
- Gossmann, J.; Lohler, J.; Lehmanngrube, F. Entry of antivirally active lymphocytes-t into the thymus of virus-infected mice. J. Immunol. 1991, 146, 293–297. [Google Scholar] [PubMed]
- Jamieson, B.D.; Ahmed, R. T-cell tolerance: Exposure to virus in utero does not cause a permanent deletion of specific t cells. Proc. Natl. Acad. Sci. USA 1988, 85, 2265–2268. [Google Scholar] [CrossRef] [PubMed]
- McCune, J.M. Thymic function in hiv-1 disease. Semin. Immunol. 1997, 9, 397–404. [Google Scholar] [CrossRef] [PubMed]
- Joshi, V.V.; Oleske, J.M.; Minnefor, A.B.; Singh, R.; Bokhari, T.; Rapkin, R.H. Pathology of suspected acquired immune deficiency syndrome in children: A study of eight cases. Pediatr. Pathol. 1984, 2, 71–87. [Google Scholar] [CrossRef] [PubMed]
- Rosenzweig, M.; Clark, D.P.; Gaulton, G.N. Selective thymocyte depletion in neonatal hiv-1 thymic infection. AIDS 1993, 7, 1601–1605. [Google Scholar] [CrossRef]
- Reimann, K.A.; Li, J.T.; Veazey, R.; Halloran, M.; Park, I.W.; Karlsson, G.B.; Sodroski, J.; Letvin, N.L. A chimeric simian/human immunodeficiency virus expressing a primary patient human immunodeficiency virus type 1 isolate env causes an aids-like disease after in vivo passage in rhesus monkeys. J. Virol. 1996, 70, 6922–6928. [Google Scholar]
- Joshi, V.V.; Oleske, J.M. Pathologic appraisal of the thymus gland in acquired immunodeficiency syndrome in children. A study of four cases and a review of the literature. Arch. Pathol. Lab. Med. 1985, 109, 142–146. [Google Scholar]
- Kourtis, A.P.; Ibegbu, C.; Nahmias, A.J.; Lee, F.K.; Clark, W.S.; Sawyer, M.K.; Nesheim, S. Early progression of disease in hiv-infected infants with thymus dysfunction. N. Engl. J. Med. 1996, 335, 1431–1436. [Google Scholar] [CrossRef]
- Muller, J.G.; Krenn, V.; Schindler, C.; Czub, S.; Stahl-Hennig, C.; Coulibaly, C.; Hunsmann, G.; Kneitz, C.; Kerkau, T.; Rethwilm, A.; et al. Alterations of thymus cortical epithelium and interdigitating dendritic cells but no increase of thymocyte cell death in the early course of simian immunodeficiency virus infection. Am. J. Pathol. 1993, 143, 699–713. [Google Scholar]
- Wykrzykowska, J.J.; Rosenzweig, M.; Veazey, R.S.; Simon, M.A.; Halvorsen, K.; Desrosiers, R.C.; Johnson, R.P.; Lackner, A.A. Early regeneration of thymic progenitors in rhesus macaques infected with simian immunodeficiency virus. J. Exp. Med. 1998, 187, 1767–1778. [Google Scholar] [CrossRef] [PubMed]
- Fiume, G.; Scialdone, A.; Albano, F.; Rossi, A.; Tuccillo, F.M.; Rea, D.; Palmieri, C.; Caiazzo, E.; Cicala, C.; Bellevicine, C.; et al. Impairment of t cell development and acute inflammatory response in hiv-1 tat transgenic mice. Sci. Rep. 2015, 5, 13864. [Google Scholar] [CrossRef] [PubMed]
- Vogel, A.B.; Haasbach, E.; Reiling, S.J.; Droebner, K.; Klingel, K.; Planz, O. Highly pathogenic influenza virus infection of the thymus interferes with t lymphocyte development. J. Immunol. 2010, 185, 4824–4834. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zhang, X.; Deng, W.; Liu, J.; Li, H.; Wen, M.; Bao, L.; Qu, J.; Liu, Y.; Li, F.; et al. Severe influenza a (h1n1) pdm09 infection induces thymic atrophy through activating innate cd8 (+) cd44 (hi) t cells by upregulating ifn-gamma. Cell Death Dis. 2014, 5, e1440. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.; Lu, J.; Zhou, K.; Wang, J.; Wu, J.; Gao, G.F.; Fang, M. Nk-cells are involved in thymic atrophy induced by influenza a virus infection. J. Gen. Virol. 2015, 96, 3223–3235. [Google Scholar] [CrossRef] [PubMed]
- Auwaerter, P.G.; Kaneshima, H.; McCune, J.M.; Wiegand, G.; Griffin, D.E. Measles virus infection of thymic epithelium in the scid-hu mouse leads to thymocyte apoptosis. J. Virol. 1996, 70, 3734–3740. [Google Scholar] [PubMed]
- Valentin, H.; Azocar, O.; Horvat, B.; Williems, R.; Garrone, R.; Evlashev, A.; Toribio, M.L.; Rabourdin-Combe, C. Measles virus infection induces terminal differentiation of human thymic epithelial cells. J. Virol. 1999, 73, 2212–2221. [Google Scholar] [PubMed]
- Vidalain, P.O.; Laine, D.; Zaffran, Y.; Azocar, O.; Servet-Delprat, C.; Wild, T.F.; Rabourdin-Combe, C.; Valentin, H. Interferons mediate terminal differentiation of human cortical thymic epithelial cells. J. Virol. 2002, 76, 6415–6424. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Vricella, L.A.; Moss, W.J.; Griffin, D.E. Immature cd4+cd8+ thymocytes are preferentially infected by measles virus in human thymic organ cultures. PLoS ONE 2012, 7, e45999. [Google Scholar] [CrossRef]
- Carvalho Barros, L.R.; Linhares-Lacerda, L.; Moreira-Ramos, K.; Ribeiro-Alves, M.; Machado Motta, M.C.; Bou-Habib, D.C.; Savino, W. Htlv-1-infected thymic epithelial cells convey the virus to cd4 (+) t lymphocytes. Immunobiology 2017, 222, 1053–1063. [Google Scholar] [CrossRef]
- Mocarski, E.S.; Bonyhadi, M.; Salimi, S.; McCune, J.M.; Kaneshima, H. Human cytomegalovirus in a scid-hu mouse: Thymic epithelial cells are prominent targets of viral replication. Proc. Natl. Acad. Sci. USA 1993, 90, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Brilot, F.; Chehadeh, W.; Charlet-Renard, C.; Martens, H.; Geenen, V.; Hober, D. Persistent infection of human thymic epithelial cells by coxsackievirus b4. J. Virol. 2002, 76, 5260–5265. [Google Scholar] [CrossRef] [PubMed]
- Jaidane, H.; Caloone, D.; Lobert, P.E.; Sane, F.; Dardenne, O.; Naquet, P.; Gharbi, J.; Aouni, M.; Geenen, V.; Hober, D. Persistent infection of thymic epithelial cells with coxsackievirus b4 results in decreased expression of type 2 insulin-like growth factor. J. Virol. 2012, 86, 11151–11162. [Google Scholar] [CrossRef] [PubMed]
- Michaux, H.; Martens, H.; Jaidane, H.; Halouani, A.; Hober, D.; Geenen, V. How does thymus infection by coxsackievirus contribute to the pathogenesis of type 1 diabetes? Front. Immunol. 2015, 6, 338. [Google Scholar] [CrossRef] [PubMed]
- Gul, K.A.; Sonerud, T.; Fjaerli, H.O.; Nakstad, B.; Abrahamsen, T.G.; Inchley, C.S. Thymus activity measured by t-cell receptor excision circles in patients with different severities of respiratory syncytial virus infection. BMC Infect. Dis. 2017, 17, 18. [Google Scholar] [CrossRef] [PubMed]
- Milich, D.R.; Jones, J.E.; Hughes, J.L.; Price, J.; Raney, A.K.; McLachlan, A. Is a function of the secreted hepatitis b e antigen to induce immunologic tolerance in utero? Proc. Natl. Acad. Sci. USA 1990, 87, 6599–6603. [Google Scholar] [CrossRef] [PubMed]
- Stevens, C.E.; Beasley, R.P.; Tsui, J.; Lee, W.C. Vertical transmission of hepatitis b antigen in taiwan. N. Engl. J. Med. 1975, 292, 771–774. [Google Scholar] [CrossRef]
- Lamontagne, L.; Jolicoeur, P. Low-virulent mouse hepatitis viruses exhibiting various tropisms in macrophages, t and b cell subpopulations, and thymic stromal cells. Lab. Anim. Sci. 1994, 44, 17–24. [Google Scholar] [PubMed]
- Takamura, S.; Kajiwara, E.; Tsuji-Kawahara, S.; Masumoto, T.; Fujisawa, M.; Kato, M.; Chikaishi, T.; Kawasaki, Y.; Kinoshita, S.; Itoi, M.; et al. Infection of adult thymus with murine retrovirus induces virus-specific central tolerance that prevents functional memory cd8+ t cell differentiation. PLoS Pathog. 2014, 10, e1003937. [Google Scholar] [CrossRef]
- Fiume, G.; Vecchio, E.; De Laurentiis, A.; Trimboli, F.; Palmieri, C.; Pisano, A.; Falcone, C.; Pontoriero, M.; Rossi, A.; Scialdone, A.; et al. Human immunodeficiency virus-1 tat activates nf-kappab via physical interaction with ikappab-alpha and p65. Nucleic Acids Res. 2012, 40, 3548–3562. [Google Scholar] [CrossRef]
- Puca, A.; Fiume, G.; Palmieri, C.; Trimboli, F.; Olimpico, F.; Scala, G.; Quinto, I. Ikappab-alpha represses the transcriptional activity of the hiv-1 tat transactivator by promoting its nuclear export. J. Biol. Chem. 2007, 282, 37146–37157. [Google Scholar] [CrossRef] [PubMed]
- Vitagliano, L.; Fiume, G.; Scognamiglio, P.L.; Doti, N.; Cannavo, R.; Puca, A.; Pedone, C.; Scala, G.; Quinto, I.; Marasco, D. Structural and functional insights into ikappab-alpha/hiv-1 tat interaction. Biochimie 2011, 93, 1592–1600. [Google Scholar] [CrossRef] [PubMed]
- Schiavone, M.; Fiume, G.; Caivano, A.; de Laurentiis, A.; Falcone, C.; Masci, F.F.; Iaccino, E.; Mimmi, S.; Palmieri, C.; Pisano, A.; et al. Design and characterization of a peptide mimotope of the hiv-1 gp120 bridging sheet. Int. J. Mol. Sci. 2012, 13, 5674–5699. [Google Scholar] [CrossRef] [PubMed]
- Gilhus, N.E.; Romi, F.; Hong, Y.; Skeie, G.O. Myasthenia gravis and infectious disease. J. Neurol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Cron, M.A.; Maillard, S.; Villegas, J.; Truffault, F.; Sudres, M.; Dragin, N.; Berrih-Aknin, S.; Le Panse, R. Thymus involvement in early-onset myasthenia gravis. Ann. N. Y. Acad. Sci. 2018, 1412, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Cavalcante, P.; Maggi, L.; Colleoni, L.; Caldara, R.; Motta, T.; Giardina, C.; Antozzi, C.; Berrih-Aknin, S.; Bernasconi, P.; Mantegazza, R. Inflammation and epstein-barr virus infection are common features of myasthenia gravis thymus: Possible roles in pathogenesis. Autoimmune Dis. 2011, 2011, 213092. [Google Scholar] [CrossRef] [PubMed]
- Cavalcante, P.; Serafini, B.; Rosicarelli, B.; Maggi, L.; Barberis, M.; Antozzi, C.; Berrih-Aknin, S.; Bernasconi, P.; Aloisi, F.; Mantegazza, R. Epstein-barr virus persistence and reactivation in myasthenia gravis thymus. Ann. Neurol. 2010, 67, 726–738. [Google Scholar] [CrossRef] [PubMed]
- Cavalcante, P.; Barzago, C.; Baggi, F.; Antozzi, C.; Maggi, L.; Mantegazza, R.; Bernasconi, P. Toll-like receptors 7 and 9 in myasthenia gravis thymus: Amplifiers of autoimmunity? Ann. N. Y. Acad. Sci. 2018, 1413, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Cavalcante, P.; Galbardi, B.; Franzi, S.; Marcuzzo, S.; Barzago, C.; Bonanno, S.; Camera, G.; Maggi, L.; Kapetis, D.; Andreetta, F.; et al. Increased expression of toll-like receptors 7 and 9 in myasthenia gravis thymus characterized by active epstein-barr virus infection. Immunobiology 2016, 221, 516–527. [Google Scholar] [CrossRef]
- Gong, L.; Li, Y.; Li, X.; Tu, Q.; Mou, X.; Wang, S.; Wan, Y.; Lu, Q.; Wang, J.; Zhang, W.; et al. Detection of human parvovirus b19 infection in the thymus of patients with thymic hyperplasia-associated myasthenia gravis. Clin. Microbiol. Infect. 2018, 25, 109.e7–109.e12. [Google Scholar] [CrossRef]
| Cell Type | Maturation State/Phenotype | References |
|---|---|---|
| Dendritic macrophages | CD45+, F4/80+, Mac-2+ | [9,10] |
| Flat-shaped macrophages | F4/80+, CD32/16+, Mac-2− | [9,10] |
| Small oval macrophages | Mac-2+ | [9,10] |
| Type 1 Conventional dendritic cells | CD11chigh, MHC II+, CD45RA−, CD8α+ | [11,12] |
| Type 2 Conventional dendritic cells | CD11chigh, MHC II+, CD45RA−, SIRPα+ | [11,12] |
| Plasmacytoid dendritic cells | CD11cmedium MHC IIlow, CD45RAhigh, CD45Rhigh | [11,12] |
| cTECs | CD45–, EpCAM+, Ly51+, K8+, K5–, K14– | [13,14,15,16,17,18] |
| mTECs | CD45–, EpCAM+, Ly51–/low, CD80high, K8–, K5+, K14+ | [13,14,15,16,17,18] |
| Sphere-forming TEC | FoxN1- | [19] |
| Thymic nurse cells | K5+, K8+, pH91+ | [20,21,22] |
| Cell Type | Phenotype | References |
|---|---|---|
| Thymic NK cells | CD127 (IL-7 receptor α) | [3,4,5,6,7,8] |
| iNKT cells | CD1d-restricted Thymic NK cells | [6,7] |
| TSP type 1—Fetal BM-derived hematopoietic progenitors as thymus colonizing cells | CD34high, CD45RAhigh, CD7+ | [29] |
| TSP type 2—Have the capacity to develop into B cells, NK cells and T cells using in vitro co-culture systems | Lin−, CD34+, CD10+, CD24− | [30] |
| TSP type 3—Possess full lymphoid and monocytic potential, but lack erythroid potential | Lin−, CD34+, CD10−, CD45RA+, CD62Lhigh | [31] |
| Double Negative 1 | CD34−, CD38−, CD1a+ | [31] |
| Double Negative 2 | CD34−, CD38+, CD1a+ | [31] |
| Double Negative 3 | CD34+, CD38+, CD1a+ | [31] |
| Double Negative 4—Immature single positive | CD3−, CD4+ | [31] |
| Double Positive | CD3+, CD4+, CD8+ | [16,17,19,32,33,34,35] |
| Single Positive | CD3+, CD4+, CD8−/CD3+, CD4−, CD8+ | [31,32,33,34,35,36,37,38] |
| B-cells | CD19+ | [23,24,25,26,27,28] |
| Progenitors | DN1 | DN2 | DN3 | DN4 | DP | SP | |
|---|---|---|---|---|---|---|---|
| Human | CD34high, CD45RAhigh, CD7+ | CD34−, CD38−, CD1a+ | CD34−, CD38+, CD1a+ | CD34+, CD38+, CD1a+ | CD3−, CD4+ | CD4+, CD8+ | CD4+, CD8− or CD4−, CD8+ |
| Mouse | Lin−, IL-7R+, Thy-1−, Sca-1low c-Kitlow | CD117/c-KIT+, CD44+, CD25− | CD117/c-KIT+, CD44+, CD25+ | CD117/c-KIT−, CD44−, CD25+ | CD117/c-Kit−, CD44−, CD25− | CD4+, CD8+ | CD4+, CD8− or CD4−, CD8+ |
| Transcriptional Factor | Cell Type | Function | References |
|---|---|---|---|
| GATA-3 | Thymic NK cells | Drive NK development from early thymocyte precursors | [3,4,5] |
| Notch | DN3 cells | Triggers the β-selection process | [57,58,59] |
| GATA-3, Tcf7, Bcl11b | T-cells | Drives the early stage of T-cells development | [60,61,62,63,64,65,81,82] |
| Bcl11b | DN2 cells | Completes the exclusion process, limits non-T-cell fates, inhibits NK lineages, decrease Kit expression | [66,67,68,69,70] |
| PU.1, Pax5 | Immature lymphoid cells | If not inhibited by Gata-3, drive B-cell and myeloid differentiation | [63,64] |
| Myb, Tcf1 | Immature lymphoid cells | Gata-3 positive regulators | [65] |
| E2A, HEB, Gata-3, Tox, Tcf1, Lef | CD4 cells | Drive CD4 lineage | [71] |
| ThPOK | CD4+/CD8+ | Drives CD4+ Th fate and prevents the differentiation of thymocytes in CD8+ CTLs | [71] |
| Mature T-cells | Blocking the cytotoxic fate of MHC class II-restricted CD4+ T cells | [74] | |
| Runx3 | CD4+/CD8+ | Abolishes CD4 expression inducing CTL-lineage differentiation | [71] |
| Runx1, Runx3, Mazr | CD8 cells | Inhibit the expression of ThPOK | [75,76,77,78] |
| NF-κB | Positive selection survived T-cells | Required for late maturation | [79,80] |
| Transcriptional Factor | Cell Type | Function | References |
|---|---|---|---|
| FoxN1 | TEPC | Development of TEPC in mTECs and cTECs; maintains postnatal TEC homeostasis; its lack arrests TEPC differentiation | [82,83,84,85,86] |
| p63 | TEPC | Required in epithelial development of thymus and epidermis; required for proliferation and differentiation | [87,88] |
| AIRE | CD80hi MHC-IIhi mTECs | Critical for thymus development, required for clonal deletion of self-reactive T cells; involved in expression of TRA | [93,94] |
| Fezf2 | mTECs | Regulates TRAs expression different from those of AIRE | [95,96] |
| Pou2f3 | Thymic tuft cells | Essential for the maturation of thymic tuft cells | [54] |
| Virus | Target | Effect | Reference |
|---|---|---|---|
| MLV/HBV/LCMV | TEC/TEC stem cells | Impaired tolerance | [99,100,101,102,103] |
| HIV/SIV | DP cells, thymocytes | Thymic atrophy; DN to DP block | [104,105,106,107,108,109,110,111,112] |
| H7N7/HSN1/H1N1 | Respiratory DCs in lung before thymic migration | Thymic atrophy | [113,114] |
| IAV | Thymocytes | Increased INFγ production by NKs; increased T-cells apoptosis | [115] |
| Measles | TEC/Myelomonocytic cells | Reduced DP cells | [116,117,118,119] |
| HTLV-1 | TEC | Virus transmission to CD4; impaired gene expression | [120] |
| CMV | mTEC | Infects epithelial cells rather than hematopoietic cells | [121] |
| CV-B | TEC/mTEC | Increased IL-6/GM-CSF/LIF production; increased IgF2 production and autoimmune diabetes | [122,123,124] |
| RSV | Thymocytes | Reduced TREC | [125] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Albano, F.; Vecchio, E.; Renna, M.; Iaccino, E.; Mimmi, S.; Caiazza, C.; Arcucci, A.; Avagliano, A.; Pagliara, V.; Donato, G.; et al. Insights into Thymus Development and Viral Thymic Infections. Viruses 2019, 11, 836. https://doi.org/10.3390/v11090836
Albano F, Vecchio E, Renna M, Iaccino E, Mimmi S, Caiazza C, Arcucci A, Avagliano A, Pagliara V, Donato G, et al. Insights into Thymus Development and Viral Thymic Infections. Viruses. 2019; 11(9):836. https://doi.org/10.3390/v11090836
Chicago/Turabian StyleAlbano, Francesco, Eleonora Vecchio, Maurizio Renna, Enrico Iaccino, Selena Mimmi, Carmen Caiazza, Alessandro Arcucci, Angelica Avagliano, Valentina Pagliara, Giuseppe Donato, and et al. 2019. "Insights into Thymus Development and Viral Thymic Infections" Viruses 11, no. 9: 836. https://doi.org/10.3390/v11090836
APA StyleAlbano, F., Vecchio, E., Renna, M., Iaccino, E., Mimmi, S., Caiazza, C., Arcucci, A., Avagliano, A., Pagliara, V., Donato, G., Palmieri, C., Mallardo, M., Quinto, I., & Fiume, G. (2019). Insights into Thymus Development and Viral Thymic Infections. Viruses, 11(9), 836. https://doi.org/10.3390/v11090836







