Long-Term Immunogenicity and Efficacy of the Oral Rabies Virus Vaccine Strain SPBN GASGAS in Foxes
Abstract
1. Introduction
2. Material & Methods
2.1. Study Animals and Housing
2.2. Ethics Statement
2.3. Vaccination and Challenge
2.4. Diagnostic Assays and Statistical Analysis
3. Results
3.1. Bait Acceptance, Vaccination, and General Health
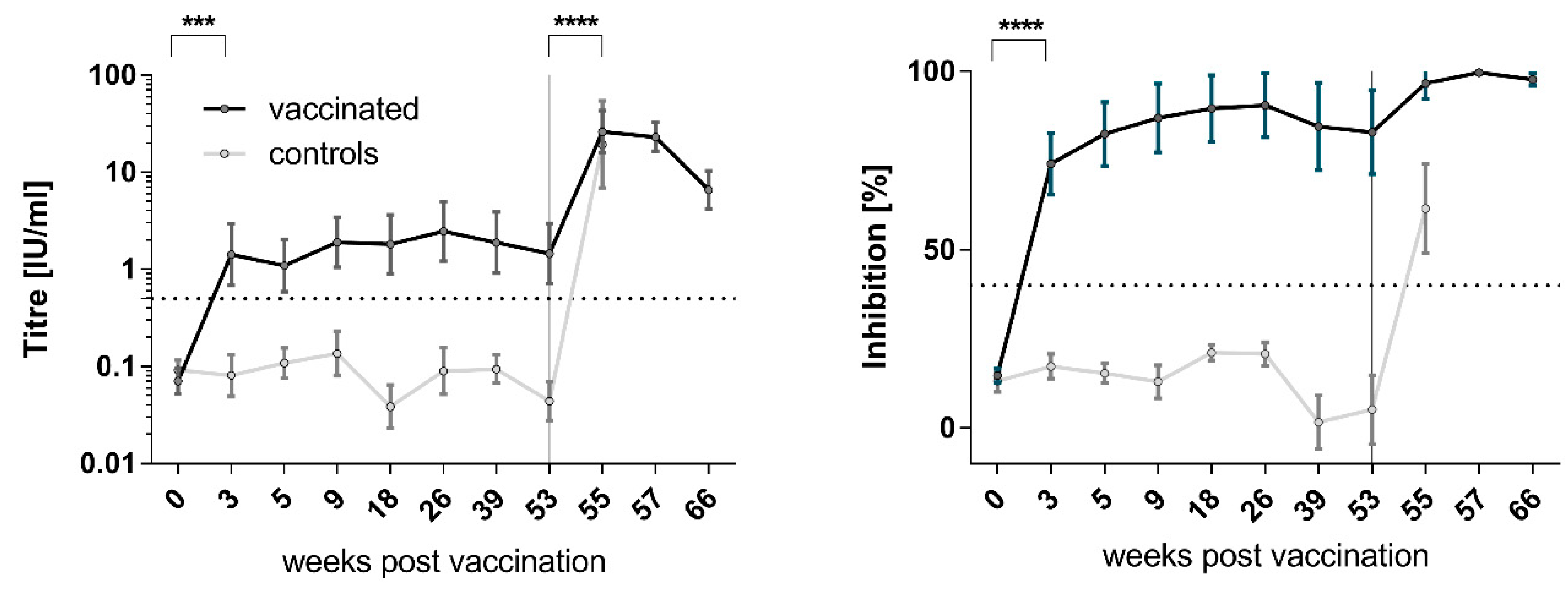
3.2. Immunogenicity
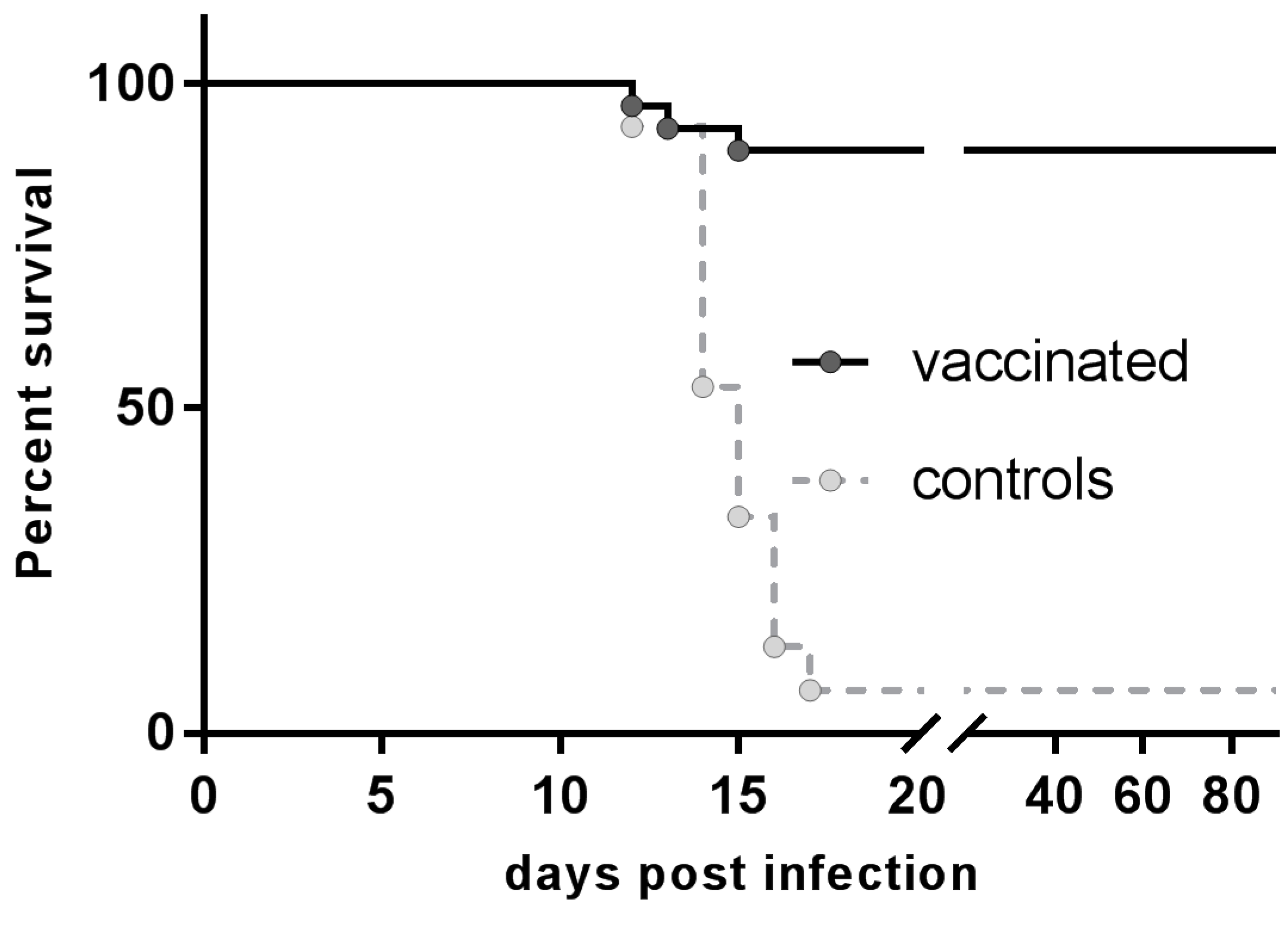
3.3. Resistance to Rabies Challenge
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Slate, D.; Algeo, T.P.; Nelson, K.M.; Chipman, R.B.; Donovan, D.; Blanton, J.D.; Niezgoda, M.; Rupprecht, C.E. Oral Rabies Vaccination in North America: Opportunities, Complexities, and Challenges. PLoS Negl. Trop. Dis. 2009, 3, e549. [Google Scholar] [CrossRef] [PubMed]
- Rosatte, R.C.; Donovan, D.; Davies, J.C.; Brown, L.; Allan, M.; von Zuben, V.; Bachmann, P.; Sobey, K.; Silver, A.; Bennett, K.; et al. High-density baiting with ONRAB(R) rabies vaccine baits to control Arctic-variant rabies in striped skunks in Ontario, Canada. J. Wildl. Dis. 2011, 47, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Freuling, C.M.; Hampson, K.; Selhorst, T.; Schröder, R.; Meslin, F.X.; Mettenleiter, T.C.; Müller, T. The elimination of fox rabies from Europe: Determinants of success and lessons for the future. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20120142. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.J.; Rosatte, R.C.; Fehlner-Gardiner, C.; Bachmann, P.; Ellison, J.A.; Jackson, F.R.; Taylor, J.S.; Davies, C.; Donovan, D. Oral vaccination and protection of red foxes (Vulpes vulpes) against rabies using ONRAB(R), an adenovirus-rabies recombinant vaccine. Vaccine 2014, 32, 984–989. [Google Scholar] [CrossRef] [PubMed]
- Müller, T.F.; Schröder, R.; Wysocki, P.; Mettenleiter, T.C.; Freuling, C.M. Spatio-temporal Use of Oral Rabies Vaccines in Fox Rabies Elimination Programmes in Europe. PLoS Negl. Trop. Dis. 2015, 9, e0003953. [Google Scholar] [CrossRef] [PubMed]
- Fehlner-Gardiner, C.; Nadin-Davis, S.A.; Armstrong, J.; Muldoon, F.; Bachmann, P.; Wandeler, A.I. ERA Vaccine-derivied cases of rabies in wildlife and domestic animals in Ontario, Canada, 1989–2004. J. Wildl. Dis. 2008, 44, 71–85. [Google Scholar] [CrossRef]
- Mahl, P.; Cliquet, F.; Guiot, A.L.; Niin, E.; Fournials, E.; Saint-Jean, N.; Aubert, M.; Rupprecht, E.C.; Gueguen, S. Twenty year experience of the oral rabies vaccine SAG2 in wildlife: A global review. Veter. Res. 2014, 45, 77. [Google Scholar] [CrossRef]
- World Health Organization. Expert Consultation on Rabies, Second report. World Health Organ Tech. Rep. Ser. 2013, 982, 1–150. [Google Scholar]
- Schneider, L.G.; Cox, J.H. Ein Feldversuch zur oralen Immunisierung von Füchsen gegen die Tollwut in der Bundesrepublik Deutschland: Unschädlichkeit, Wirksamkeit und Stabilität der Vakzine SAD B 19. Tierarztl Umsch. 1983, 38, 315–324. [Google Scholar]
- Artois, M.; Guittré, C.; Thomas, I.; Leblois, H.; Brochier, B.; Barrat, J. Potential pathogenicity for rodents of vaccines intended for oral vaccination against rabies: A comparison. Vaccine 1992, 10, 524–528. [Google Scholar] [CrossRef]
- Güzel, T.; Aylan, O.; Vos, A. Innocuity tests with SAD B19 in Turkish non-target species. J. Etlik Vet. Microbiol. 1998, 9, 103–112. [Google Scholar]
- Vos, A.; Neubert, A.; Aylan, O.; Schuster, P.; Pommerening, E.; Muller, T.; Chivatsi, D.C. An update on safety studies of SAD B19 rabies virus vaccine in target and non-target species. Epidemiol. Infect. 1999, 123, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Lawson, K.F.; Hertler, R.; Charlton, K.M.; Campbell, J.B.; Rhodes, A.J. Safety and immunogenicity of ERA strain of rabies virus propagated in a BHK-21 cell line. Can. J. Veter. Res. Rev. Can. Rech. Veter. 1989, 53, 438–444. [Google Scholar]
- Müller, T.; Bätza, H.J.; Beckert, A.; Bunzenthal, C.; Cox, J.H.; Freuling, C.M.; Fooks, A.R.; Frost, J.; Geue, L.; Hoeflechner, A.; et al. Analysis of vaccine-virus-associated rabies cases in red foxes (Vulpes vulpes) after oral rabies vaccination campaigns in Germany and Austria. Arch. Virol. 2009, 154, 1081–1091. [Google Scholar] [CrossRef] [PubMed]
- Hostnik, P.; Picard-Meyer, E.; Rihtaric, D.; Toplak, I.; Cliquet, F. Vaccine-induced Rabies in a Red Fox (Vulpes vulpes): Isolation of Vaccine Virus in Brain Tissue and Salivary Glands. J. Wildl. Dis. 2014, 50, 397–401. [Google Scholar] [CrossRef]
- Vuta, V.; Picard-Meyer, E.; Robardet, E.; Barboi, G.; Motiu, R.; Barbuceanu, F.; Vlagioiu, C.; Cliquet, F. Vaccine-induced rabies case in a cow (Bos taurus): Molecular characterisation of vaccine strain in brain tissue. Vaccine 2016, 34, 5021–5025. [Google Scholar] [CrossRef]
- Pfaff, F.; Muller, T.; Freuling, C.M.; Fehlner-Gardiner, C.; Nadin-Davis, S.; Robardet, E.; Cliquet, F.; Vuta, V.; Hostnik, P.; Mettenleiter, T.C.; et al. In-depth genome analyses of viruses from vaccine-derived rabies cases and corresponding live-attenuated oral rabies vaccines. Vaccine 2018, 37, 4758–4765. [Google Scholar] [CrossRef]
- Cliquet, F.; Robardet, E.; Meyer, E.P. Genetic strain modification of a live rabies virus vaccine widely used in Europe for wildlife oral vaccination. Antivir. Res. 2013, 100, 84–89. [Google Scholar] [CrossRef]
- Cliquet, F.; Picard-Meyer, E.; Mojzis, M.; Dirbáková, Z.; Muižniece, Z.; Jaceviciene, I.; Mutinelli, F.; Matulova, M.; Frolichova, J.; Rychlík, I.; et al. In-Depth Characterization of Live Vaccines Used in Europe for Oral Rabies Vaccination of Wildlife. PLoS ONE 2015, 10, e0141537. [Google Scholar] [CrossRef]
- Höper, D.; Freuling, C.M.; Müller, T.; Hanke, D.; Von Messling, V.; Duchow, K.; Beer, M.; Mettenleiter, T.C. High definition viral vaccine strain identity and stability testing using full-genome population data – The next generation of vaccine quality control. Vaccine 2015, 33, 5829–5837. [Google Scholar] [CrossRef]
- Rupprecht, C.E.; Charlton, K.M.; Artois, M.; Casey, G.A.; Webster, W.A.; Campbell, J.B.; Lawson, K.F.; Schneider, L.G. Ineffectiveness and Comparative Pathogenicity of Attenuated Rabies Virus Vaccines for the Striped Skunk (Mephitis mephitis). J. Wildl. Dis. 1990, 26, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Tolson, N.D.; Charlton, K.M.; Lawson, K.F.; Campbell, J.B.; Stewart, R.B. Studies of ERA/BHK-21 rabies vaccine in skunks and mice. Can. J. Veter. Res. Rev. Can. Rech. Veter. 1988, 52, 58–62. [Google Scholar]
- Rupprecht, C.E.; Dietzschold, B.; Cox, J.H.; Schneider, L.Q. Oral vaccination of raccoons (Procyon lotor) with an attenuated (sad-b 19) rabies virus vaccine. J. Wildl. Dis. 1989, 25, 548–554. [Google Scholar] [CrossRef] [PubMed]
- Kieny, M.P.; Lathe, R.; Drillien, R.; Spehner, D.; Skory, S.; Schmitt, D.; Wiktor, T.; Koprowski, H.; Lecocq, J.P. Expression of rabies virus glycoprotein from a recombinant vaccinia virus. Nature 1984, 312, 163–166. [Google Scholar] [CrossRef] [PubMed]
- Blancou, J.; Kieny, M.P.; Lathe, R.; Lecocq, J.P.; Pastore, P.P.; Soulebot, J.P.; Desmettre, P. Oral vaccination of the fox against rabies using a live recombinant vaccinia virus. Nature 1986, 322, 373–375. [Google Scholar] [CrossRef]
- Prevec, L.; Campbell, J.B.; Christie, B.S.; Belbeck, L.; Graham, F.L. A Recombinant Human Adenovirus Vaccine against Rabies. J. Infect. Dis. 1990, 161, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Z.Q.; Yang, Y.; Wilson, J.M.; Ertl, H.C. A Replication-Defective Human Adenovirus Recombinant Serves as a Highly Efficacious Vaccine Carrier. Virology 1996, 219, 220–227. [Google Scholar] [CrossRef]
- Yarosh, O.K.; Wandeler, A.I.; Graham, F.L.; Campbell, J.B.; Prevec, L. Human adenovirus type 5 vectors expressing rabies glycoprotein. Vaccine 1996, 14, 1257–1264. [Google Scholar] [CrossRef]
- Knowles, M.K.; Nadin-Davis, S.A.; Sheen, M.; Rosatte, R.; Mueller, R.; Beresford, A. Safety studies on an adenovirus recombinant vaccine for rabies (AdRG1.3-ONRAB®) in target and non-target species. Vaccine 2009, 27, 6619–6626. [Google Scholar] [CrossRef]
- Slate, D.; Chipman, R.B.; Algeo, T.P.; Mills, S.A.; Nelson, K.M.; Croson, C.K.; Dubovi, E.J.; Vercauteren, K.; Renshaw, R.W.; Atwood, T.; et al. Safety and immunogenicity of ontario rabies vaccine bait (Onrab) in the first us field trial in raccoons (Procyon lotor). J. Wildl. Dis. 2014, 50, 582–595. [Google Scholar] [CrossRef]
- Brochier, B.; Dechamps, P.; Costy, F.; Hallet, L.; Villers, M. Élimination de la rage en Belgique par la vaccination du renard roux (Vulpes vulpes). Ann. Med. Vet. 2001, 145, 293–305. [Google Scholar]
- Cliquet, F.; Combes, B.; Barrat, J. Means used for terrestrial rabies elimination in France and policy for rabies surveillance in case of re-emergence. Dev. Biol. 2006, 125, 119–126. [Google Scholar]
- Fehlner-Gardiner, C.; Rudd, R.; Donovan, D.; Slate, D.; Kempf, L.; Badcock, J. Comparing ONRAB(R) AND RABORAL V-RG(R) oral rabies vaccine field performance in raccoons and striped skunks, New Brunswick, Canada, and Maine, USA. J. Wildl. Dis. 2012, 48, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Mainguy, J.; Fehlner-Gardiner, C.; Slate, D.; Rudd, R.J. Oral rabies vaccination in raccoons: Comparison of ONRAB(R) and RABORAL V-RG(R) vaccine-bait field performance in Quebec, Canada and Vermont, USA. J. Wildl. Dis. 2013, 49, 190–193. [Google Scholar] [CrossRef] [PubMed]
- Rosatte, R.C.; Donovan, D.; Davies, J.C.; Allan, M.; Bachmann, P.; Stevenson, B.; Sobey, K.; Brown, L.; Silver, A.; Bennett, K.; et al. Aerial distribution of onrab® baits as a tactic to control rabies in raccoons and striped skunks in ontario, canada. J. Wildl. Dis. 2009, 45, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Schnell, M.; Mebatsion, T.; Conzelmann, K. Infectious rabies viruses from cloned cDNA. EMBO J. 1994, 13, 4195–4203. [Google Scholar] [CrossRef] [PubMed]
- Faber, M.; Dietzschold, B.; Li, J. Immunogenicity and Safety of Recombinant Rabies Viruses Used for Oral Vaccination of Stray Dogs and Wildlife. Zoonoses Public Health 2009, 56, 262–269. [Google Scholar] [CrossRef]
- Bankovskiy, D.; Safonov, G.; Kurilchuk, Y. Immunogenicity of the ERA G 333 rabies virus strain in foxes and raccoon dogs. Dev. Biol. 2008, 131, 461–466. [Google Scholar]
- Faber, M.; Faber, M.-L.; Papaneri, A.; Bette, M.; Weihe, E.; Dietzschold, B.; Schnell, M.J. A Single Amino Acid Change in Rabies Virus Glycoprotein Increases Virus Spread and Enhances Virus Pathogenicity. J. Virol. 2005, 79, 14141–14148. [Google Scholar] [CrossRef]
- Freuling, C.M.; Eggerbauer, E.; Finke, S.; Kaiser, C.; Kaiser, C.; Kretzschmar, A.; Nolden, T.; Ortmann, S.; Schröder, C.; Teifke, J.P.; et al. Efficacy of the oral rabies virus vaccine strain SPBN GASGAS in foxes and raccoon dogs. Vaccine 2017, 37, 4750–4757. [Google Scholar] [CrossRef]
- Rieder, M.; Brzozka, K.; Pfaller, C.K.; Cox, J.H.; Stitz, L.; Conzelmann, K.K. Genetic dissection of interferon-antagonistic functions of rabies virus phosphoprotein: Inhibition of interferon regulatory factor 3 activation is important for pathogenicity. J. Virol. 2011, 85, 842–852. [Google Scholar] [CrossRef] [PubMed]
- Mebatsion, T. Extensive Attenuation of Rabies Virus by Simultaneously Modifying the Dynein Light Chain Binding Site in the P Protein and Raplacing Arg333 in the G Protein. J. Virol. 2001, 75, 11496–11502. [Google Scholar] [CrossRef] [PubMed]
- Mebatsion, T.; Kilari, S.; Visser, N. Development of an oral rabies vaccine with improved safety and efficacy to control canine rabies in ownerless and stray dogs. Virol. Res. Dev. 2001, 1, 1–8. [Google Scholar]
- Zhang, Y.; Zhou, M.; Li, Y.; Luo, Z.; Chen, H.; Cui, M.; Fu, Z.F.; Zhao, L. Recombinant rabies virus with the glycoprotein fused with a DC-binding peptide is an efficacious rabies vaccine. Oncotarget 2018, 9, 831–841. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Li, M.; Zhou, M.; Zhang, Y.; Yang, J.; Cao, Y.; Wang, K.; Cui, M.; Chen, H.; Fu, Z.F.; et al. A Novel Rabies Vaccine Expressing CXCL13 Enhances Humoral Immunity by Recruiting both T Follicular Helper and Germinal Center B Cells. J. Virol. 2017, 91, e01956-16. [Google Scholar] [CrossRef] [PubMed]
- Gaskell, R.M.; Dawson, S.; Radford, A.D. Duration of immunity (DOI)—The regulatory issues. Veter. Microbiol. 2006, 117, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Lawson, K.F.; Crawley, J.F. Era Strain of Rabies Vaccine. Can. J. Comp. Med. 1972, 36, 339–344. [Google Scholar] [PubMed]
- Larghi, O.P.; Savy, V.L.; E Nebel, A.; Rodríguez, A. Rabies vaccine inactivated with ethylenimine. Duration of the immunity in dogs. Rev. Argent. Microbiol. 1979, 11, 102–107. [Google Scholar] [PubMed]
- Jas, D.; Coupier, C.; Toulemonde, C.E.; Guigal, P.M.; Poulet, H. Three-year duration of immunity in cats vaccinated with a canarypox-vectored recombinant rabies virus vaccine. Vaccine 2012, 30, 6991–6996. [Google Scholar] [CrossRef] [PubMed]
- Lakshmanan, N.; Gore, T.C.; Duncan, K.L.; Coyne, M.J.; Lum, M.A.; Sterner, F.J. Three-year rabies duration of immunity in dogs following vaccination with a core combination vaccine against canine distemper virus, canine adenovirus type-1, canine parvovirus, and rabies virus. Vet. Ther. Res. Appl. Vet. Med. 2006, 7, 223–231. [Google Scholar]
- Lawson, K.F.; Chiu, H.; Crosgrey, S.J.; Matson, M.; A Casey, G.; Campbell, J.B. Duration of immunity in foxes vaccinated orally with ERA vaccine in a bait. Can. J. Veter. Res. Rev. Can. Rech. Veter. 1997, 61, 39–42. [Google Scholar]
- Artois, M.; Cliquet, F.; Barrat, J.; Schumacher, C.L. Effectiveness of SAG1 oral vaccine for the long-term protection of red foxes (Vulpes vulpes) against rabies. Veter. Rec. 1997, 140, 57–59. [Google Scholar] [CrossRef] [PubMed]
- Black, J.G.; Lawson, K.F. The safety and efficacy of immunizing foxes (Vulpes vulpes) using bait containing attenuated rabies virus vaccine. Can. J. Comp. Med. Rev. Can. Med. Comp. 1980, 44, 169–176. [Google Scholar]
- European Food Safety Authority. Scientific opinion-Update on oral vaccination of foxes and raccoon dogs against rabies. EFSA J. 2015, 13, 4164. [Google Scholar]
- Un, H.; Eskiizmirliler, S.; Unal, N.; Freuling, C.M.; Johnson, N.; Fooks, A.R.; Müller, T.; Vos, A.; Aylan, O. Oral vaccination of foxes against rabies in Turkey between 2008 and 2010. Berl. Munch. Tierarztl. Wochenschr. 2012, 125, 203–208. [Google Scholar] [PubMed]
- Yakobson, B.; Goga, I.; Freuling, C.M.; Fooks, A.R.; Gjinovci, V.; Hulaj, B.; Horton, D.; Johnson, N.; Muhaxhiri, J.; Recica, I.; et al. Implementation and monitoring of oral rabies vaccination of foxes in Kosovo between 2010 and 2013—An international and intersectorial effort. Int. J. Med Microbiol. 2014, 304, 902–910. [Google Scholar] [CrossRef] [PubMed]
- European Union. Rabies Eradication in the EU; European Union: Brussels, Belgium, 2017.
- Anonymous. Guidelines for the veterinary care of laboratory animals: Report of the FELASA/ECLAM/ESLAV Joint Working Group on Veterinary Care. Lab. Anim. 2008, 42, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hartinger, J.; Foltz, T.; Cussler, K. Clinical endpoints during rabies vaccine control tests. Altex 2000, 18, 37–40. [Google Scholar]
- European Medicines Agency. ICH Guideline for good clinical practice E6(R2) Committee for Human Medicinal Products; EMA/CHMP/ICH: Amsterdam, The Netherlands; London, UK; Geneva, Switzerland, 2016.
- Dean, D.J.; Abelseth, M.K.; Athanasiu, P. The Fluorescence Antibody Test. In Laboratory Techniques in Rabies, 4th ed.; Meslin, F.X., Kaplan, M.M., Koprowski, H., Eds.; World Health Organization: Geneva, Switzerland, 1996; pp. 88–93. [Google Scholar]
- Hoffmann, B.; Freuling, C.M.; Wakeley, P.R.; Rasmussen, T.B.; Leech, S.; Fooks, A.R.; Beer, M.; Müller, T. Improved Safety for Molecular Diagnosis of Classical Rabies Viruses by Use of a TaqMan Real-Time Reverse Transcription-PCR “Double Check” Strategy. J. Clin. Microbiol. 2010, 48, 3970–3978. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.M.; Gilbert, A.; Vos, A.; Freuling, C.M.; Ellis, C.; Kliemt, J.; Müller, T. Rabies Virus Antibodies from Oral Vaccination as a Correlate of Protection against Lethal Infection in Wildlife. Trop. Med. Infect. Dis. 2017, 2, 31. [Google Scholar] [CrossRef]
- Wasniewski, M.; Guiot, A.; Schereffer, J.; Tribout, L.; Mahar, K.; Cliquet, F. Evaluation of an ELISA to detect rabies antibodies in orally vaccinated foxes and raccoon dogs sampled in the field. J. Virol. Methods 2013, 187, 264–270. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. Rabies Vaccine (Live, Oral) for Foxes and Raccoon Dogs; European Directorate for the Quality of Medicines/European Council: Strasbourg, France, 2014.
- Anonymous. Electronic Code of Federal Regulations (e-CFR) Title 9. Animals and Animal Products Chapter I. Animal and Plant Health Inspection Service, US Department of Agriculture Subchapter e. Viruses, Serums, Toxins, and Analogous Products; Live Virus Vaccines: 2007.
- Cliquet, F.; Guiot, A.; Munier, M.; Bailly, J.; Rupprecht, C.; Barrat, J. Safety and efficacy of the oral rabies vaccine SAG2 in raccoon dogs. Vaccine 2006, 24, 4386–4392. [Google Scholar] [CrossRef] [PubMed]
- Pommerening, E.; Neubert, A.; Schuster, P.; Muller, T.; Vos, A. Immunogenicity and Efficacy of the Oral Rabies Vaccine SAD B19 in Foxes. J. Veter. Med. Ser. B 2001, 48, 179–183. [Google Scholar]
- Schuster, P.; Muller, T.; Vos, A.; Selhorst, T.; Neubert, L.; Pommerening, E.; Information, R. Comparative immunogenicity and efficacy studies with oral rabies virus vaccine SAD P5/88 in raccoon dogs and red foxes. Acta Veter. Hung. 2001, 49, 285–290. [Google Scholar]
- Blancou, J.; Artois, M.; Brochier, B.; Thomas, I.; Pastoret, P.P.; Desmettre, P.; Languet, B.; Kieny, M.P. Innocuite et efficacite d’un vaccin antirabique recombinant des virus de la vaccine et de la rage administre par voie orale au renard, au chiery et au chat. Ann. Rech. Vet. 1989, 20, 195–204. [Google Scholar] [PubMed]
- Brochier, B.M.; Languet, B.; Artois, M.; Zanker, S.; Guittre, C.; Blancou, J.; Chappuis, G.; Desmettre, P.; Pastoret, P.P. Efficacy of a baiting system for vaccinating foxes against rabies with vaccinia-rabies recombinant virus. Veter. Rec. 1990, 127, 165–167. [Google Scholar]
- Cliquet, F.; Guiot, A.L.; Schumacher, C.; Maki, J.; Cael, N.; Barrat, J. Efficacy of a square presentation of V-RG vaccine baits in red fox, domestic dog and raccoon dog. Dev. Biol. 2008, 131, 257–264. [Google Scholar]
- Brown, L.J.; Rosatte, R.C.; Fehlner-Gardiner, C.; Ellison, J.A.; Jackson, F.R.; Bachmann, P.; Taylor, J.S.; Franka, R.; Donovan, D. Oral vaccination and protection of striped skunks (Mephitis mephitis) against rabies using ONRAB(R). Vaccine 2014, 32, 3675–3679. [Google Scholar] [CrossRef]
- Gilbert, A.; Johnson, S.; Walker, N.; Wickham, C.; Beath, A.; Vercauteren, K. Efficacy of Ontario Rabies Vaccine Baits (ONRAB) against rabies infection in raccoons. Vaccine 2018, 36, 4919–4926. [Google Scholar] [CrossRef]
- World Health Organization. Oral Vaccination of Dogs against Rabies-Guidance for Research on Oral Rabies Vaccines and Field Application of Oral Vaccination of Dogs against Rabies; WHO: Geneva, Switzerland, 2007.
- Fekadu, M.; Nesby, S.; Shaddock, J.; Schumacher, C.; Linhart, S.; Sanderlin, D. Immunogenicity, efficacy and safety of an oral rabies vaccine (SAG-2) in dogs. Vaccine 1996, 14, 465–468. [Google Scholar] [CrossRef]
- Zhugunissov, K.; Bulatov, Y.; Taranov, D.; Yershebulov, Z.; Koshemetov, Z.; Abduraimov, Y.; Kondibayeva, Z.; Samoltyrova, A.; Amanova, Z.; Khairullin, B.; et al. Protective immune response of oral rabies vaccine in stray dogs, corsacs and steppe wolves after a single immunization. Arch. Virol. 2017, 162, 3363–3370. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, Y.; Fooks, A.R.; Zhang, F.; Hu, R. Oral vaccination of dogs (Canis familiaris) with baits containing the recombinant rabies-canine adenovirus type-2 vaccine confers long-lasting immunity against rabies. Vaccine 2008, 26, 345–350. [Google Scholar] [CrossRef]
- Hanlon, C.A.; Niezgoda, M.; Morrill, P.; Rupprecht, C.E. Oral Efficacy of an Attenuated Rabies Virus Vaccine in Skunks and Raccoons. J. Wildl. Dis. 2002, 38, 420–427. [Google Scholar] [CrossRef]
- Hsu, A.P.; Tseng, C.H.; Barrat, J.; Lee, S.H.; Shih, Y.H.; Wasniewski, M.; Mähl, P.; Chang, C.C.; Lin, C.T.; Chen, R.S.; et al. Safety, efficacy and immunogenicity evaluation of the SAG2 oral rabies vaccine in Formosan ferret badgers. PLoS ONE 2017, 12, e0184831. [Google Scholar] [CrossRef]
- European Commission. The oral vaccination of foxes against rabies. Report of the Scientific Committee on Animal Health and Animal Welfare. 2002; 1–55. [Google Scholar]
- Thomas, I.; Brochier, B.; Levaux, T.; Bauduin, B.; Costy, F.; Peharpre, D. Results of the 1988 Campaign of Fox Vaccination Against Rabies Using Sad B19 Vaccine in Belgium. Ann. Med. Vet. 1989, 133, 403–412. [Google Scholar]
- Brochier, B.; Iokem, A.; Ginter, A.; Lejeune, E.; Costy, F.; Marchal, A. Premiere campagne de vaccination antirabique du renard par voie orale menee en Belgique. Controles d’efficacite et d’innocuite chez le renard roux (Vulpes vulpes L.). Ann. Med. Vet. 1987, 131, 463–472. [Google Scholar]
- Brown, L.J.; Rosatte, R.C.; Fehlner-Gardiner, C.; Taylor, J.S.; Davies, J.C.; Donovan, D. Immune response and protection in raccoons (Procyon lotor) following consumption of baits containing ONRAB(R), a human adenovirus rabies glycoprotein recombinant vaccine. J. Wildl. Dis. 2012, 48, 1010–1020. [Google Scholar] [CrossRef]
- Weinberg, G.A.; Szilagyi, P.G. Vaccine Epidemiology: Efficacy, Effectiveness, and the Translational Research Roadmap. J. Infect. Dis. 2010, 201, 1607–1610. [Google Scholar] [CrossRef]
- Shim, E.; Galvani, A.P. Distinguishing vaccine efficacy and effectiveness. Vaccine 2012, 30, 6700–6705. [Google Scholar] [CrossRef]
- Knobel, D.L.; Lembo, T.; Morters, M.; Townsend, S.E.; Cleaveland, S.; Hampson, K. Chapter 17-Dog Rabies and Its Control; Jackson, A.C., Ed.; Academic Press: Boston, MA, USA, 2013; pp. 591–615. [Google Scholar]
- Thulke, H.H.; Eisinger, D. The strength of 70%: Revision of a Standard Threshold of Rabies Control. Dev. Biol. 2008, 131, 291–298. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freuling, C.M.; te Kamp, V.; Klein, A.; Günther, M.; Zaeck, L.; Potratz, M.; Eggerbauer, E.; Bobe, K.; Kaiser, C.; Kretzschmar, A.; et al. Long-Term Immunogenicity and Efficacy of the Oral Rabies Virus Vaccine Strain SPBN GASGAS in Foxes. Viruses 2019, 11, 790. https://doi.org/10.3390/v11090790
Freuling CM, te Kamp V, Klein A, Günther M, Zaeck L, Potratz M, Eggerbauer E, Bobe K, Kaiser C, Kretzschmar A, et al. Long-Term Immunogenicity and Efficacy of the Oral Rabies Virus Vaccine Strain SPBN GASGAS in Foxes. Viruses. 2019; 11(9):790. https://doi.org/10.3390/v11090790
Chicago/Turabian StyleFreuling, Conrad M., Verena te Kamp, Antonia Klein, Maria Günther, Luca Zaeck, Madlin Potratz, Elisa Eggerbauer, Katharina Bobe, Christian Kaiser, Antje Kretzschmar, and et al. 2019. "Long-Term Immunogenicity and Efficacy of the Oral Rabies Virus Vaccine Strain SPBN GASGAS in Foxes" Viruses 11, no. 9: 790. https://doi.org/10.3390/v11090790
APA StyleFreuling, C. M., te Kamp, V., Klein, A., Günther, M., Zaeck, L., Potratz, M., Eggerbauer, E., Bobe, K., Kaiser, C., Kretzschmar, A., Ortmann, S., Schuster, P., Vos, A., Finke, S., & Müller, T. (2019). Long-Term Immunogenicity and Efficacy of the Oral Rabies Virus Vaccine Strain SPBN GASGAS in Foxes. Viruses, 11(9), 790. https://doi.org/10.3390/v11090790




