A Novel RNA Virus Related to Sobemoviruses Confers Hypovirulence on the Phytopathogenic Fungus Sclerotinia sclerotiorum
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains and Culture Conditions
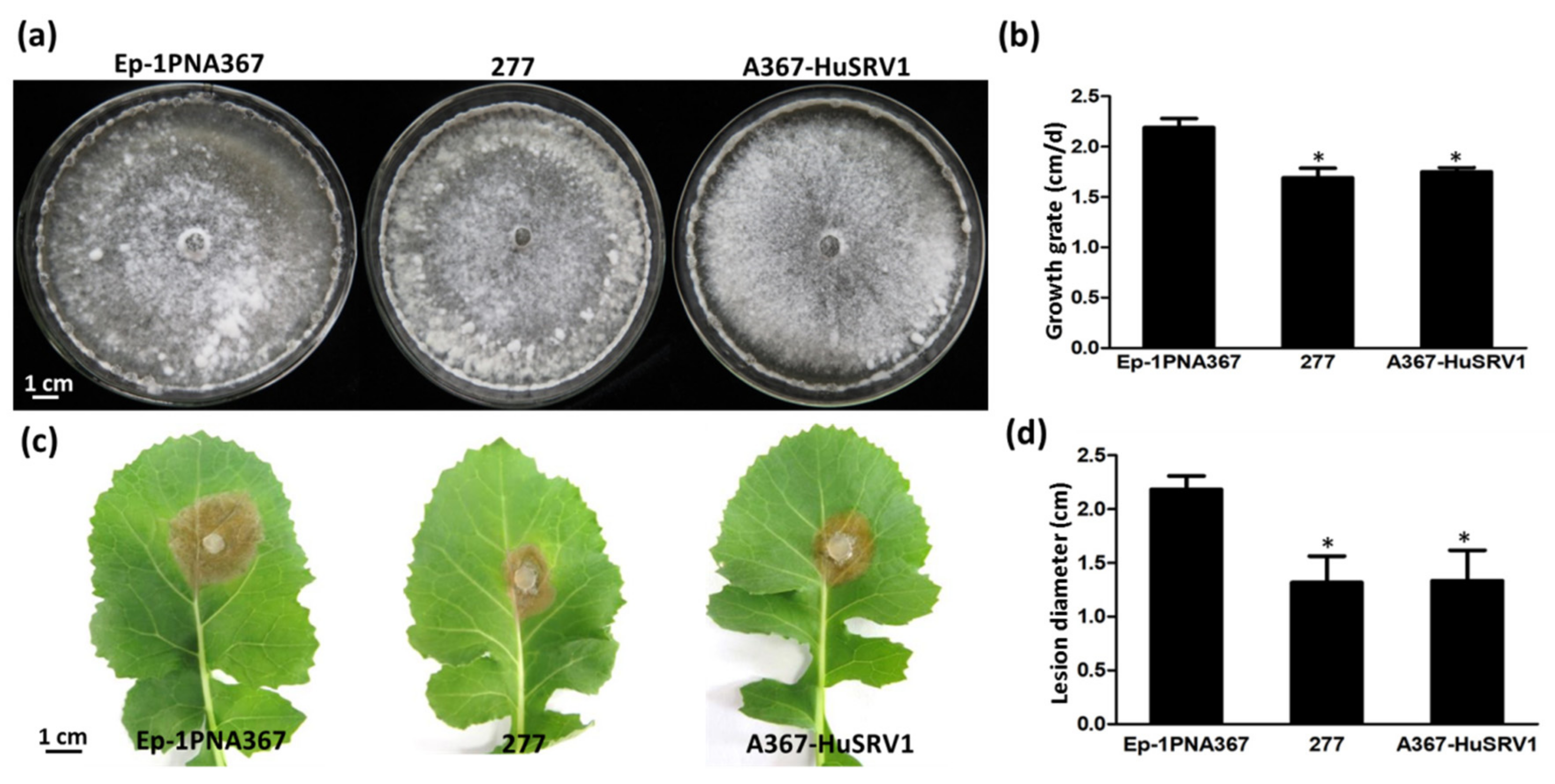
2.2. Comparison of Biological Characteristics
2.3. Purification and Observation of Virions
2.4. Nucleic Acid Extraction, Metatranscriptomic Sequencing, and Full-Length cDNA Cloning
2.5. Sequence and Phylogenetic Analyses
2.6. Protoplast Preparation and Transfection
2.7. Horizontal Transmission of Hypovirulence Traits
3. Results
3.1. Biological Characteristics of Strain 277
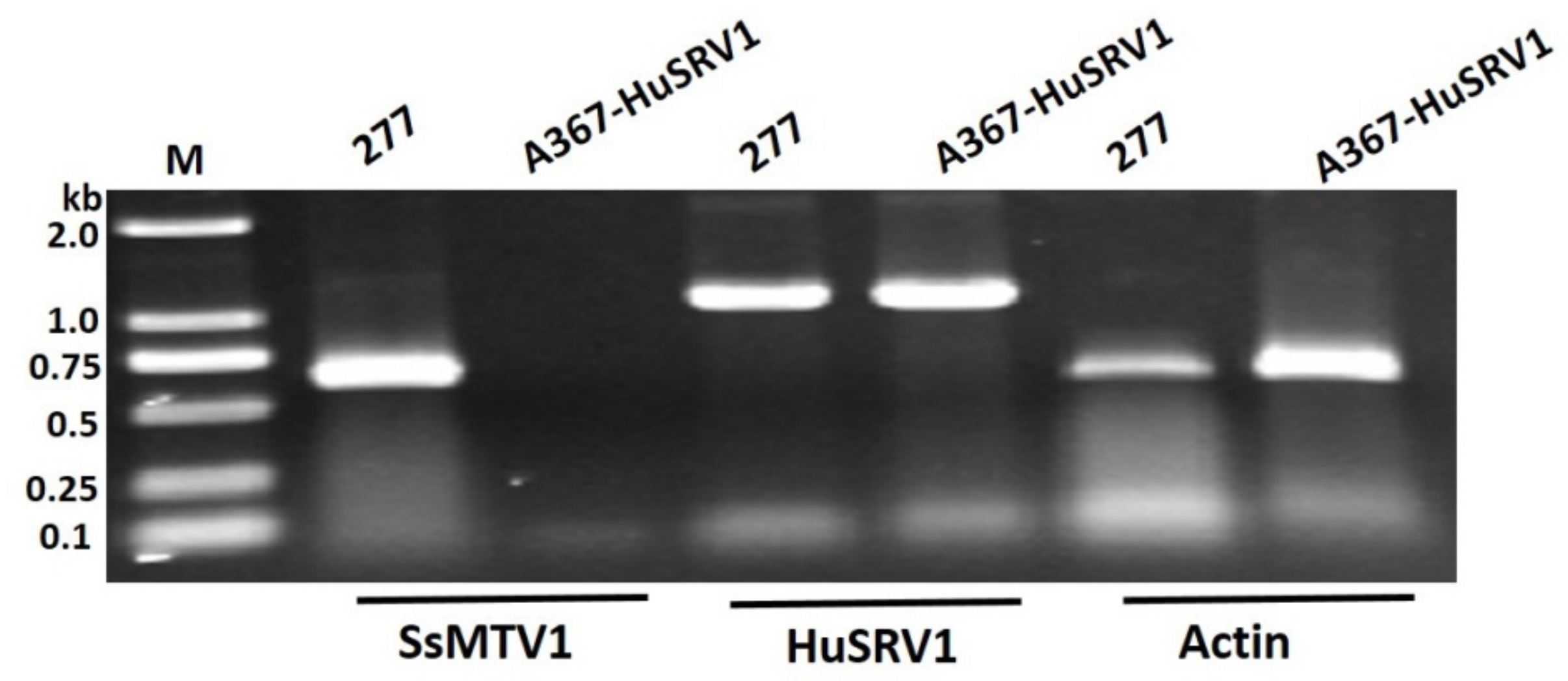
3.2. Complete Sequence and Organization of the HuSRV1 Genome
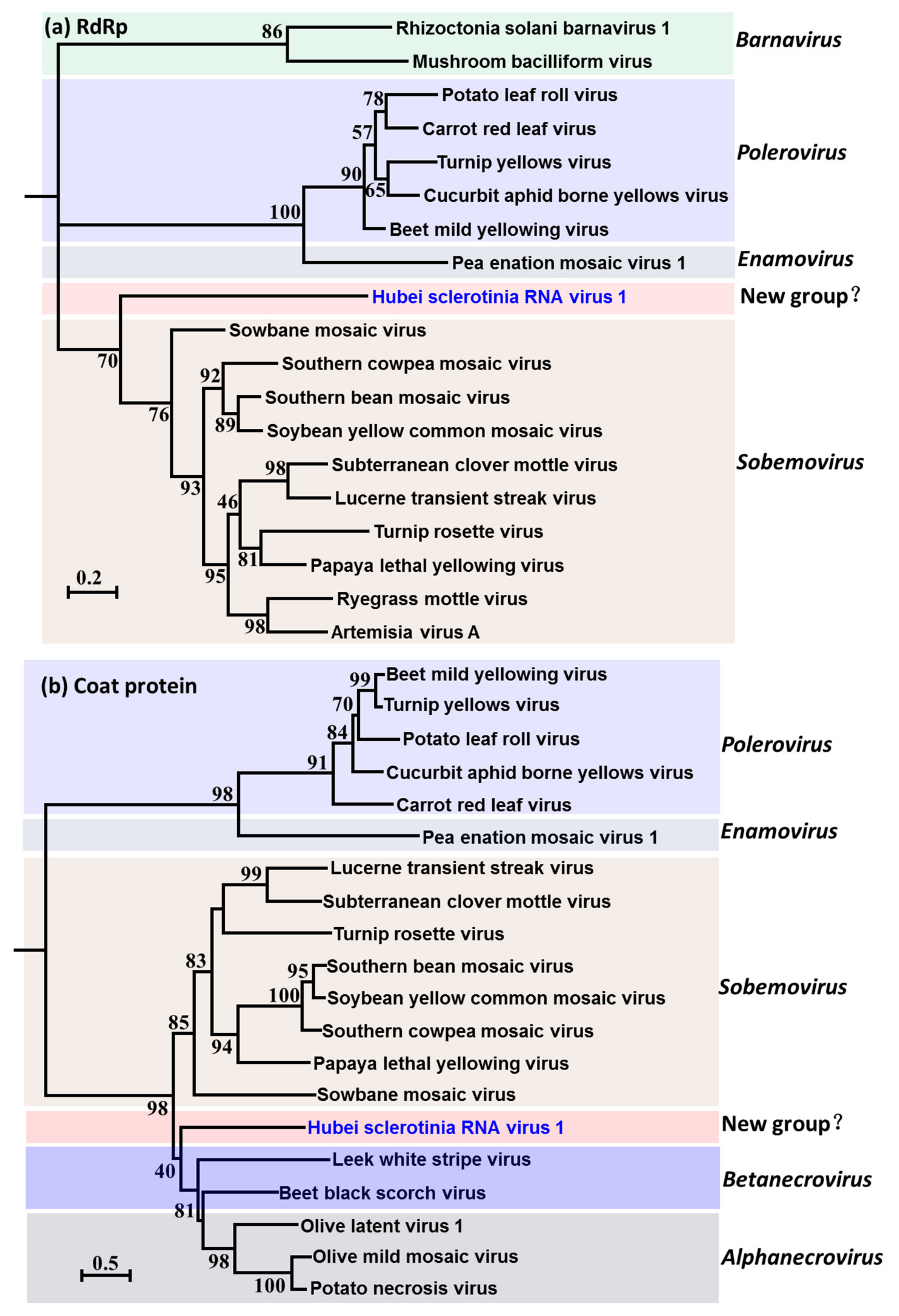
3.3. Evolutionary Relationship of HuSRV1
3.4. Transfection Assay and Biological Traits of S. sclerotiorum Infected by HuSRV1
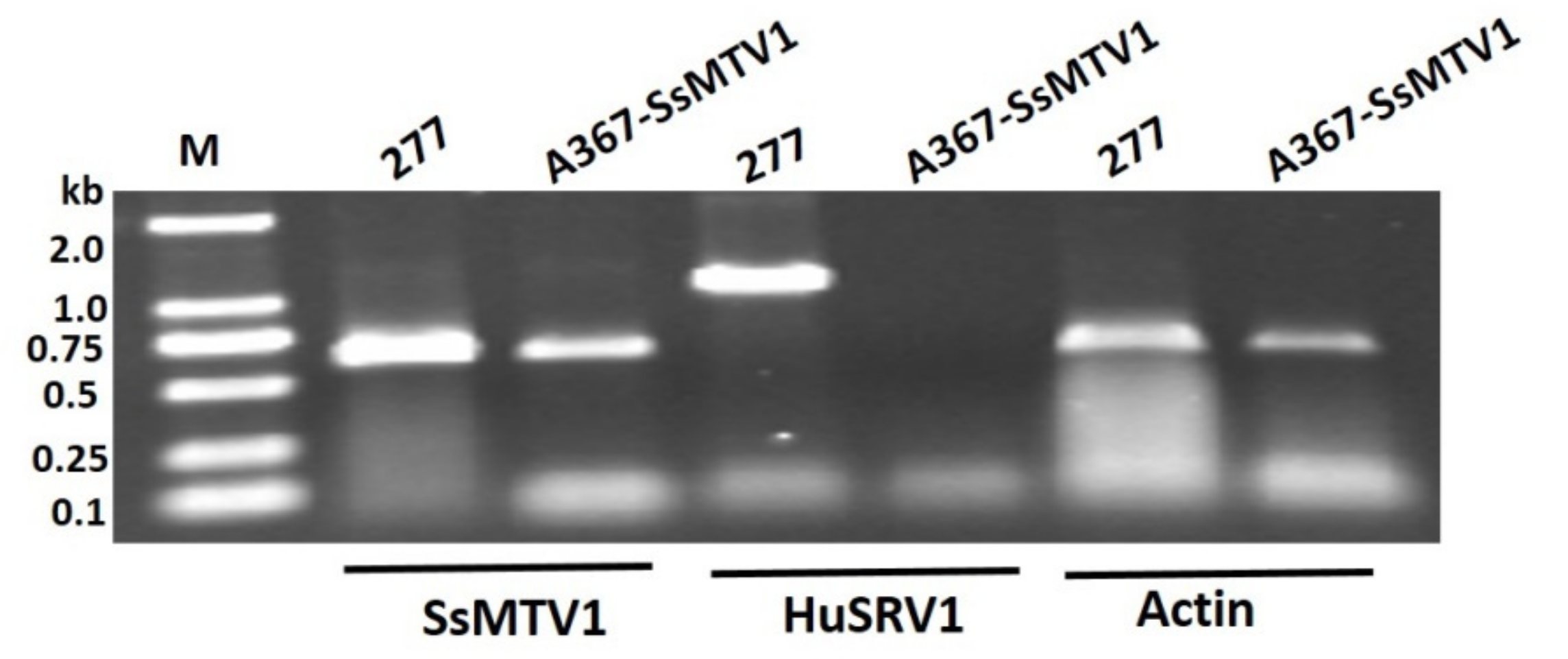
3.5. Transmission Assay of the Dual Culture of Strain 277 and Ep-1PNA367R
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Strange, R.N.; Scott, P.R. Plant disease: A threat to global food security. Annu. Rev. Phytopathol. 2005, 43, 83–116. [Google Scholar] [CrossRef] [PubMed]
- Anderson, P.K.; Cunningham, A.A.; Patel, N.G.; Morales, F.J.; Epstein, P.R.; Daszak, P. Emerging infectious diseases of plants: Pathogen pollution, climate change and agrotechnology drivers. Trends Ecol. Evol. 2004, 19, 53–544. [Google Scholar] [CrossRef] [PubMed]
- Ghabrial, S.A.; Suzuki, N. Viruses of plant pathogenic fungi. Annu. Rev. Phytopathol. 2009, 47, 353–384. [Google Scholar] [CrossRef] [PubMed]
- Pearson, M.N.; Beever, R.E.; Boine, B.; Arthur, K. Mycoviruses of filamentous fungi and their relevance to plant pathology. Mol. Plant Pathol. 2009, 10, 115–128. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Jiang, D. New insights into mycoviruses and exploration for the biological control of crop fungal diseases. Annu. Rev. Phytopathol. 2014, 52, 45–68. [Google Scholar] [CrossRef]
- Kotta-Loizou, I.; Coutts, R.H. Studies on the virome of the entomopathogenic fungus Beauveria bassiana reveal novel dsRNA elements and mild hypervirulence. PLoS Pathog. 2017, 13, 1006183. [Google Scholar] [CrossRef] [PubMed]
- Nuss, D. Hypovirulence: Mycoviruses at the fungal-plant interface. Nat. Rev. Microbiol. 2005, 3, 632–642. [Google Scholar] [CrossRef]
- Liu, S.; Xie, J.; Cheng, J.; Li, B.; Chen, T.; Fu, Y.; Li, G.; Wang, M.; Jin, H.; Wan, H.; et al. Fungal DNA virus infects a mycophagous insect and utilizes it as a transmission vector. Proc. Natl. Acad. Sci. USA 2016, 113, 12803–12808. [Google Scholar] [CrossRef]
- Boland, G.J.; Hall, R. Index of plant hosts of Sclerotinia sclerotiorum. Can. J. Plant Pathol. 1994, 16, 93–108. [Google Scholar] [CrossRef]
- Bolton, M.D.; Thomma, B.P.H.J.; Nelson, B.D. Sclerotinia sclerotiorum (Lib.) de Bary: Biology and molecular traits of a cosmopolitan pathogen. Mol. Plant Pathol. 2006, 7, 1–16. [Google Scholar] [CrossRef]
- Ma, H.; Feng, X.; Chen, Y.; Chen, C.; Zhou, M. Occurrence and characterization of dimethachlon insensitivity in Sclerotinia sclerotiorum in Jiangsu Province of China. Plant Dis. 2008, 93, 36–42. [Google Scholar] [CrossRef]
- Gossen, B.D.; Rimmer, S.R.; Holley, J.D. First report of resistance to benomyl fungicide in Sclerotinia sclerotiorum. Plant Dis. 2001, 85, 1206. [Google Scholar] [CrossRef]
- Kuang, J.; Wang, J.; Zhou, M. Monitoring on carbendazim and dimethachlon-resistance of Sclerotinia sclerotiorum obtained from the blight stems of rape in Jiangsu Province. Chin. Agric. Sci. Bull. 2011, 27, 28–291. [Google Scholar]
- Li, H.; Fu, Y.; Jiang, D.; Li, G.; Ghabrial, S.A.; Yi, X. Down-regulation of Sclerotinia sclerotiorum gene expression in response to infection with Sclerotinia sclerotiorum debilitation-associated RNA virus. Virus Res. 2008, 135, 95–106. [Google Scholar] [CrossRef]
- Jiang, D.; Fu, Y.; Li, G.; Ghabrial, S.A. Viruses of the plant pathogenic fungus Sclerotinia sclerotiorum. Adv. Virus Res. 2013, 86, 21–48. [Google Scholar]
- Boland, G.J. Hypovirulence and double-stranded RNA in Sclerotinia sclerotiorum. Can. J. Plant Pathol. 1992, 14, 10–17. [Google Scholar] [CrossRef]
- Yu, X.; Li, B.; Fu, Y.; Jiang, D.; Ghabrial, S.A.; Li, G.; Peng, Y.; Xie, J.; Cheng, J.; Huang, J.; et al. A geminivirus-related DNA mycovirus that confers hypovirulence to a plant pathogenic fungus. Proc. Natl. Acad. Sci. USA 2010, 107, 8387–8392. [Google Scholar] [CrossRef]
- Khalifa, M.E.; Pearson, M.N. Molecular characterization of three mitoviruses co-infecting a hypovirulent isolate of Sclerotinia sclerotiorum fungus. Virology 2013, 441, 22–30. [Google Scholar] [CrossRef]
- Xiao, X.; Cheng, J.; Tang, J.; Fu, Y.; Jiang, D.; Baker, T.S.; Ghabrial, S.A.; Xie, J. A novel partitivirus that confers hypovirulence on plant pathogenic fungi. J. Virol. 2014, 88, 10120–10133. [Google Scholar] [CrossRef]
- Yu, X.; Li, B.; Fu, Y.; Xie, J.; Cheng, J.; Ghabrial, S.A.; Li, G.; Yi, X.; Jiang, D. Extracellular transmission of a DNA mycovirus and its use as a natural fungicide. Proc. Natl. Acad. Sci. USA 2013, 110, 1452–1457. [Google Scholar] [CrossRef]
- Liu, L.; Xie, J.; Cheng, J.; Fu, Y.; Li, G.; Yi, X.; Jiang, D. Fungal negative-stranded RNA virus that is related to bornaviruses and nyaviruses. Proc. Natl. Acad. Sci. USA 2014, 111, 12205–12210. [Google Scholar] [CrossRef]
- Li, P.; Lin, Y.; Zhang, H.; Wang, S.; Qiu, D.; Guo, L. Molecular characterization of a novel mycovirus of the family Tymoviridae isolated from the plant pathogenic fungus Fusarium graminearum. Virology 2016, 489, 86–94. [Google Scholar] [CrossRef]
- Xie, J.; Wei, D.; Jiang, D.; Fu, Y.; Li, G.; Ghabrial, S.; Peng, Y. Characterization of debilitation-associated mycovirus infecting the plant-pathogenic fungus Sclerotinia sclerotiorum. J. Gen. Virol. 2006, 87, 241–249. [Google Scholar] [CrossRef]
- Potgieter, A.C.; Page, N.A.; Liebenberg, J.; Wright, I.M.; Landt, O.; van Dijk, A.A. Improved strategies for sequence-independent amplification and sequencing of viral double-stranded RNA genomes. J. Gen. Virol. 2009, 90, 1423–1432. [Google Scholar] [CrossRef]
- Sperschneider, J.; Datta, A. DotKnot: Pseudoknot prediction using the probability dot plot under a refined energy model. Nucleic Acids Res. 2010, 38, e103. [Google Scholar] [CrossRef]
- Darty, K.; Denise, A.; Ponty, Y. VARNA: Interactive drawing and editing of the RNA secondary structure. Bioinformatics. 2009, 25, 1974–1975. [Google Scholar] [CrossRef]
- Kelley, L.A.; Sternberg, M.J.E. Protein structure prediction on the web: A case study using the Phyre server. Nat. Protoc. 2009, 4, 363–371. [Google Scholar] [CrossRef]
- Zhang, L.; Fu, Y.; Xie, J.; Jiang, D.; Li, G.; Yi, X. A novel virus that infecting hypovirulent strain XG36-1 of plant fungal pathogen Sclerotinia sclerotiorum. Virol. J. 2009, 6, 96. [Google Scholar] [CrossRef]
- Wang, Q.; Cheng, S.; Xiao, X.; Cheng, J.; Fu, Y.; Chen, T.; Jiang, D.; Xie, J. Discovery of two mycoviruses by high-throughput sequencing and assembly of mycovirus-derived small silencing RNAs from a hypovirulent strain of Sclerotinia sclerotiorum. Front. Microbiol. 2019, 10, 1415. [Google Scholar] [CrossRef]
- Sõmera, M.; Sarmiento, C.; Truve, E. Overview on sobemoviruses and a proposal for the creation of the family Sobemoviridae. Viruses 2015, 7, 3076–3115. [Google Scholar] [CrossRef]
- Truve, E.; Fargette, D. Genus Sobemovirus. In In Virus Taxonomy: Classification and Nomenclature of Viruses; Elsevier Academic Press: Waltham, MA, USA, 2012; pp. 1185–1190. [Google Scholar]
- Ling, R.; Pate, A.E.; Carr, J.P.; Firth, A.E. An essential fifth coding ORF in the sobemoviruses. Virology 2013, 446, 397–408. [Google Scholar] [CrossRef]
- Sérémé, D.; Lacombe, S.; Konaté, M.; Pinel-Galzi, A.; Traoré, V.S.E.; Hébrard, E.; Traoré, O.; Brugidou, C.; Fargette, D.; Konaté, G. Biological and molecular characterization of a putative new sobemovirus infecting Imperata cylindrica and maize in Africa. Arch. Virol. 2008, 153, 1813–1820. [Google Scholar] [CrossRef]
- Mäkinen, K.; Tamm, T.; Naess, V.; Truve, E.; Puurand, U.; Munthe, T.; Saarma, M. Characterization of cocksfoot mottle sobemovirus genomic RNA and sequence comparison with related viruses. J. Gen. Virol. 1995, 76, 2817–2825. [Google Scholar] [CrossRef]
- McGavin, W.J.; Macfarlane, S.A. Rubus chlorotic mottle virus, a new sobemovirus infecting raspberry and bramble. Virus Res. 2009, 139, 10–13. [Google Scholar] [CrossRef]
- Dwyer, G.I.; Njeru, R.; Williamson, S.; Fosu-Nyarko, J.; Hopkins, R.; Jones, R.A.C.; Waterhouse, P.M.; Jones, M.G.K. The complete nucleotide sequence of Subterranean clover mottle virus. Arch. Virol. 2003, 148, 2237–2247. [Google Scholar] [CrossRef]
- Tamm, T.; Suurväli, J.; Lucchesi, J.; Olspert, A.; Truve, E. Stem-loop structure of Cocksfoot mottle virus RNA is indispensable for programmed -1 ribosomal frameshifting. Virus Res. 2009, 146, 73–80. [Google Scholar] [CrossRef]
- Zirkel, F.; Kurth, A.; Quan, P.L.; Briese, T.; Ellerbrok, H.; Pauli, G.; Leendertz, F.H.; Lipkin, W.I.; Ziebuhr, J.; Drosten, C.; et al. An insect nidovirus emerging from a primary tropical rainforest. mBio 2011, 2, e00077-11. [Google Scholar] [CrossRef]
- Plevka, P.; Tars, K.; Zeltins, A.; Balke, I.; Truve, E.; Liljas, L. The three-dimensional structure of ryegrass mottle virus at 2.9 A resolution. Virology 2007, 369, 364–374. [Google Scholar] [CrossRef]
- Hong, Y.; Cole, T.E.; Brasier, C.M.; Buck, K.W. Evolutionary Relationships among putative RNA-dependent RNA polymerases encoded by a mitochondrial virus-like RNA in the Dutch elm disease fungus, Ophiostoma novo-ulmi, by other viruses and virus-like RNAs and by the Arabidopsis mitochondrial genome. Virology 1998, 246, 158–169. [Google Scholar] [CrossRef]
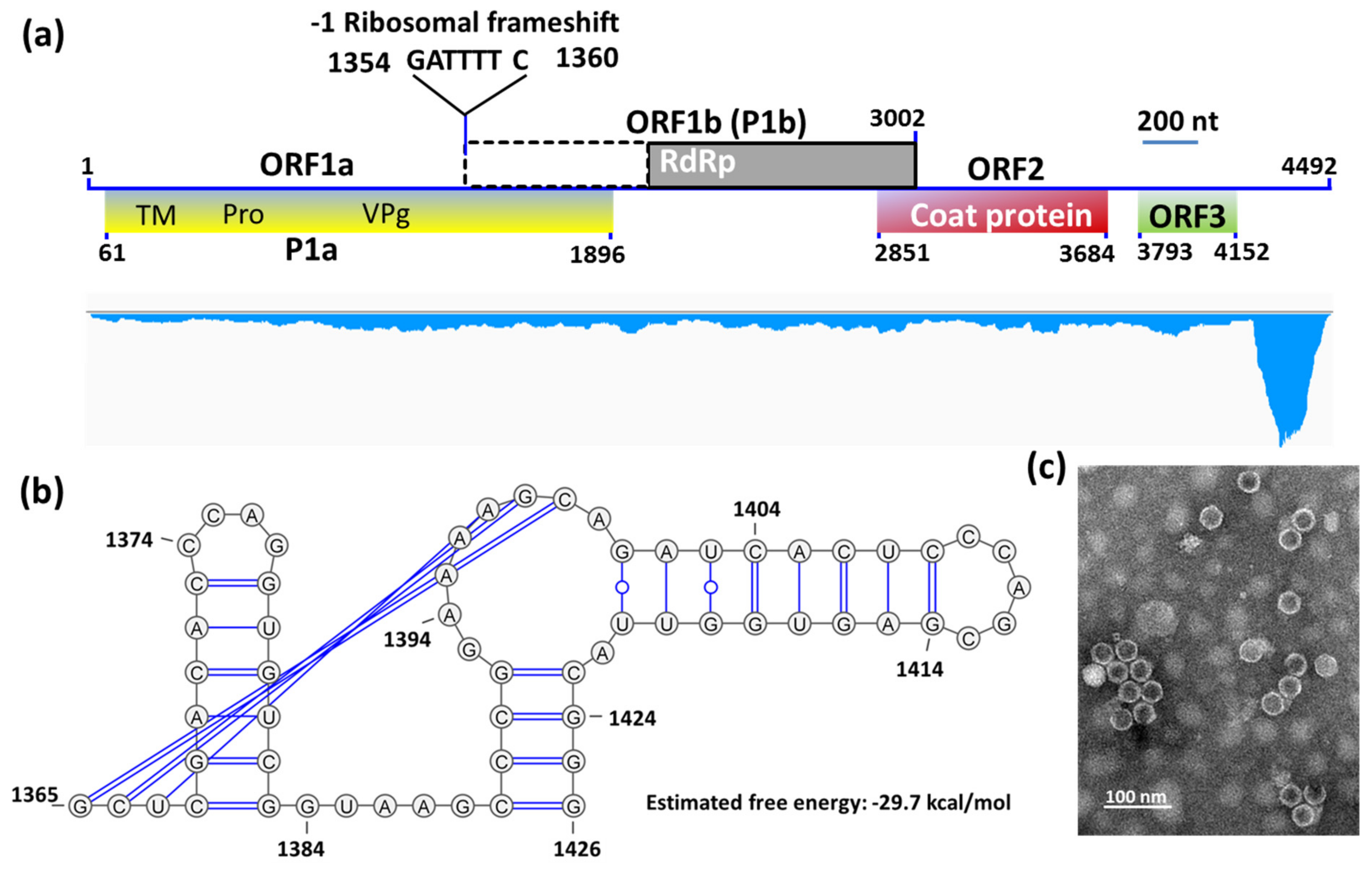

| Nucleotide Sequence motifs | ||
| Motif | Nucleotide positions | Description |
| ACAAAA | 1–6 | Precedes the P1a start codon |
| GATTTTC | 1354–1360 | −1 ribosomal frameshifting signal |
| Amino acid (AA) sequence motifs | ||
| Motif | AA positions | Description |
| Transmembrane helix | 21–43 (P1a) | Predicted transmembrane domain |
| HX36DX62TXXGXSG | 186–292 (P1a) | Pro: Serine protease |
| WADLDDEDEDX3DD | 393–407 (P1a) | VPg: ED-rich region |
| RdRp motifs I-VII | I: 241–252 II: 265–290 III: 301–309 IV: 330–342 V: 387–411 VI: 417–428 VII: 455–464 | Motifs that classify RdRp genes into superfamilies |
| GX3TX3NX19GDD | 395–425 (RdRp) | Conserved region |
| S (shell) domain | 43–272 (CP) | Shell domain involved in subunit interactions |
| P144, G152, D161, D164, P239, G257, N276 | 144–276 (CP) | Conserved invariant amino acid residues |
| D161, D164, T220, N276 | 161–276 (CP) | Ca2+ binding sites |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azhar, A.; Mu, F.; Huang, H.; Cheng, J.; Fu, Y.; Hamid, M.R.; Jiang, D.; Xie, J. A Novel RNA Virus Related to Sobemoviruses Confers Hypovirulence on the Phytopathogenic Fungus Sclerotinia sclerotiorum. Viruses 2019, 11, 759. https://doi.org/10.3390/v11080759
Azhar A, Mu F, Huang H, Cheng J, Fu Y, Hamid MR, Jiang D, Xie J. A Novel RNA Virus Related to Sobemoviruses Confers Hypovirulence on the Phytopathogenic Fungus Sclerotinia sclerotiorum. Viruses. 2019; 11(8):759. https://doi.org/10.3390/v11080759
Chicago/Turabian StyleAzhar, Ayesha, Fan Mu, Huang Huang, Jiasen Cheng, Yanping Fu, Muhammad Rizwan Hamid, Daohong Jiang, and Jiatao Xie. 2019. "A Novel RNA Virus Related to Sobemoviruses Confers Hypovirulence on the Phytopathogenic Fungus Sclerotinia sclerotiorum" Viruses 11, no. 8: 759. https://doi.org/10.3390/v11080759
APA StyleAzhar, A., Mu, F., Huang, H., Cheng, J., Fu, Y., Hamid, M. R., Jiang, D., & Xie, J. (2019). A Novel RNA Virus Related to Sobemoviruses Confers Hypovirulence on the Phytopathogenic Fungus Sclerotinia sclerotiorum. Viruses, 11(8), 759. https://doi.org/10.3390/v11080759






