Identification of a Novel Equine Papillomavirus in Semen from a Thoroughbred Stallion with a Penile Lesion
Abstract
1. Introduction
2. Materials and Methods
Sample Collection, RNA Sequencing and Virus Discovery
3. Results and Discussion
3.1. Identification of a Novel Equine Papillomavirus
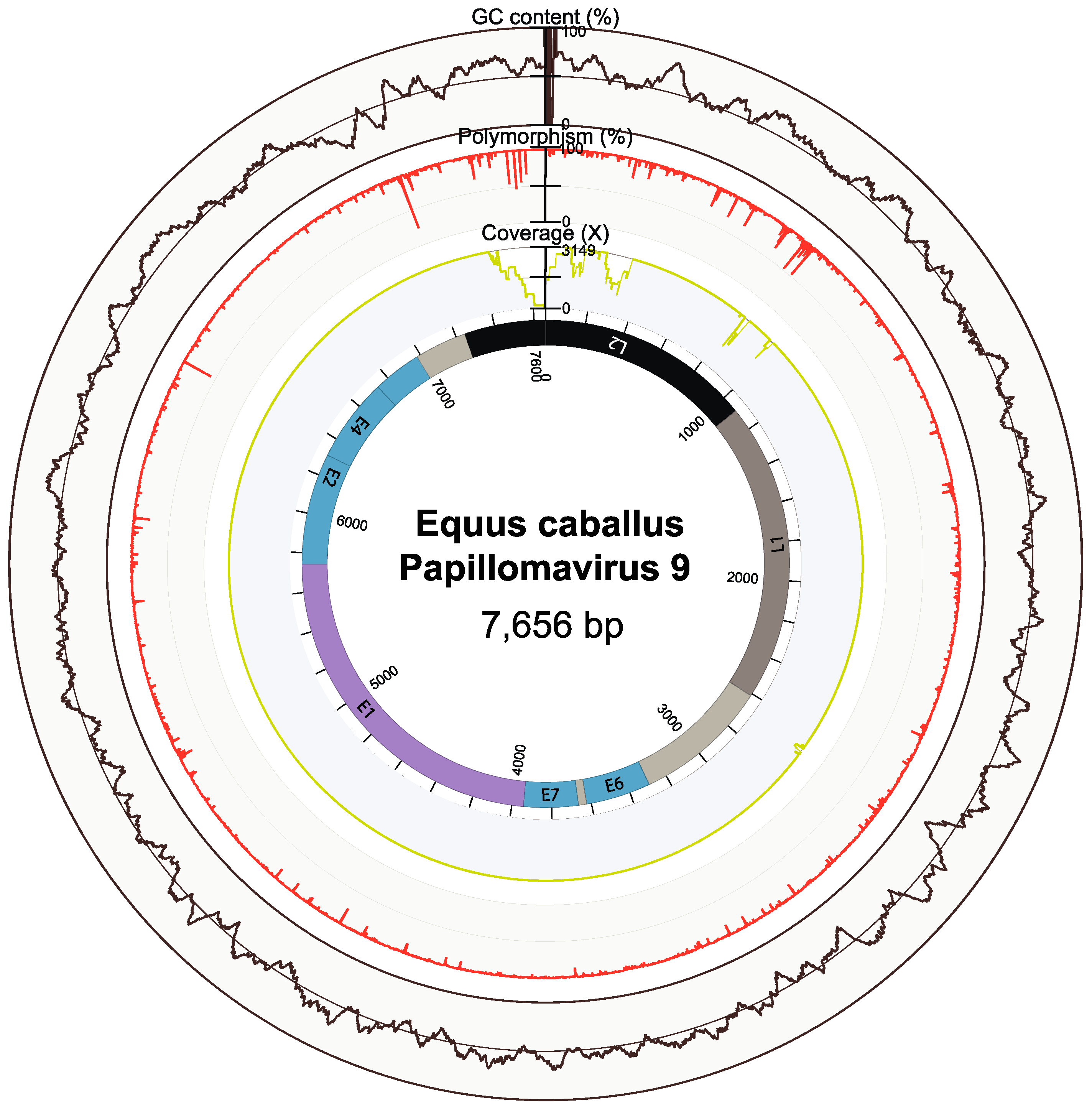
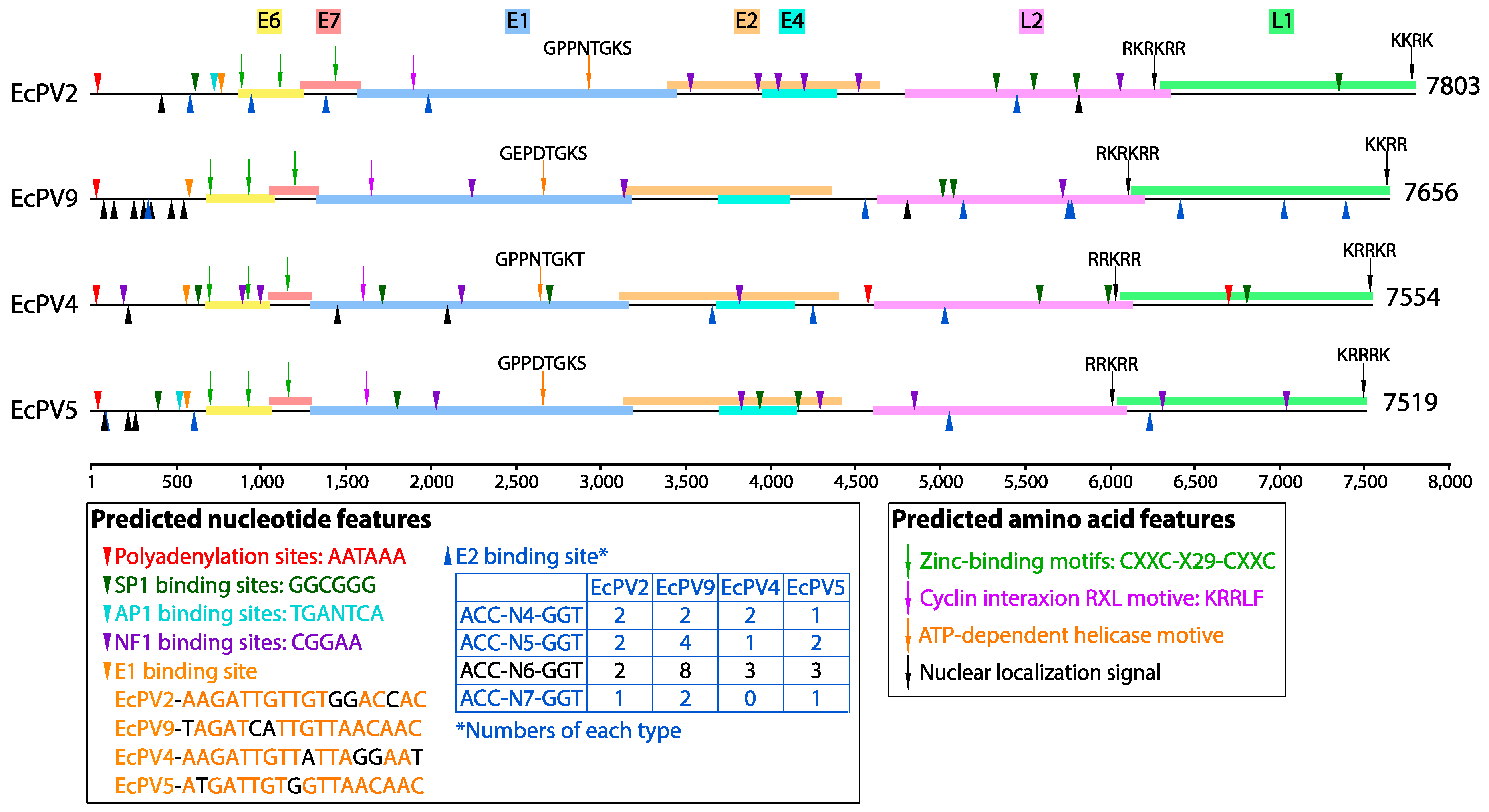
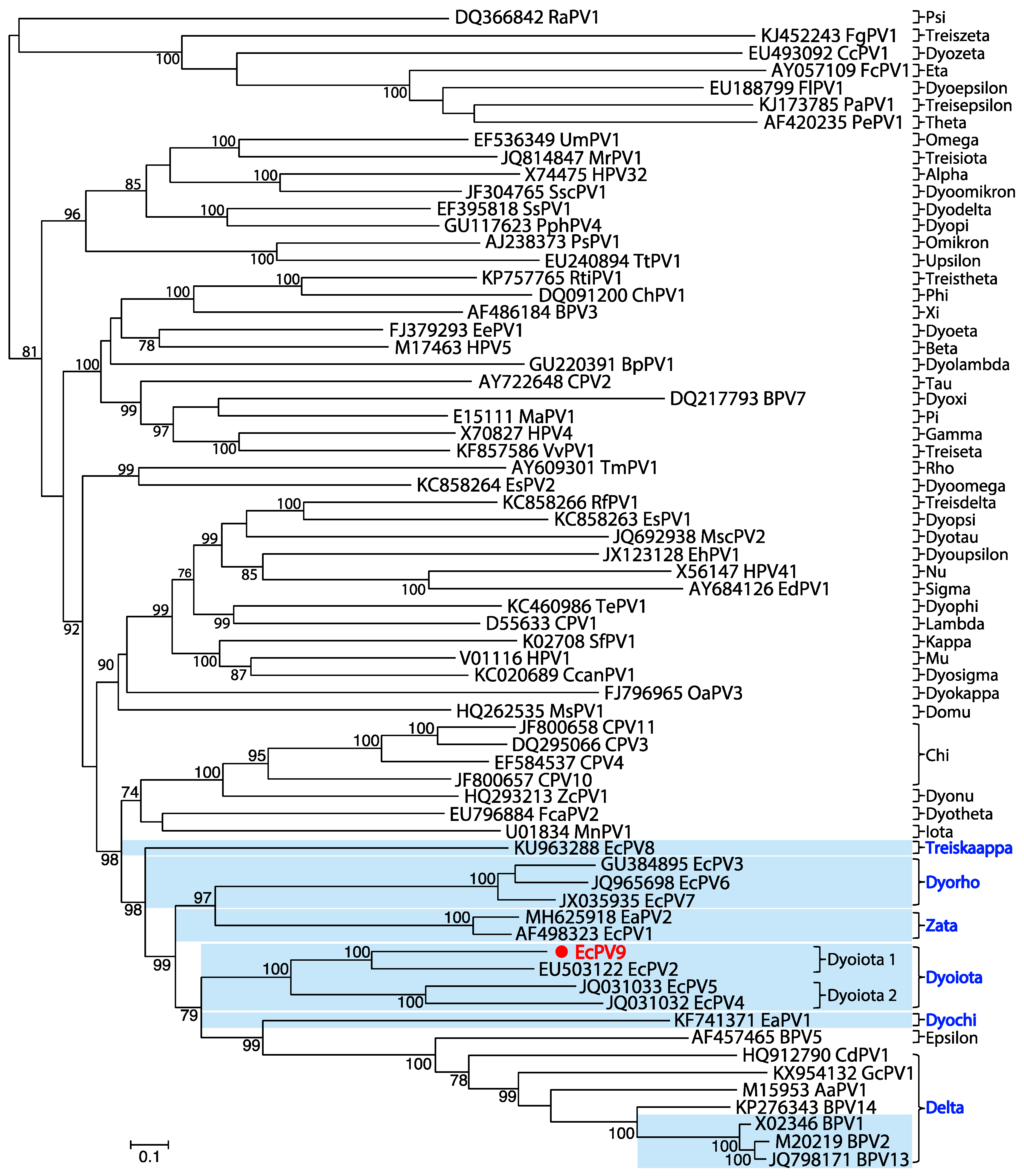
3.2. Genomic Properties and Evolutionary Relationships of EcPV9
3.3. Disease Association
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Nasir, L.; Brandt, S. Papillomavirus associated diseases of the horse. Vet. Microbiol. 2013, 167, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Lunardi, M.; de Alcântara, B.K.; Otonel, R.A.; Rodrigues, W.B.; Alfieri, A.F.; Alfieri, A.A. Bovine papillomavirus type 13 DNA in equine sarcoids. J. Clin. Microbiol. 2013, 51, 2167–2171. [Google Scholar] [CrossRef] [PubMed]
- Torres, S.M.; Koch, S.N. Papillomavirus-associated diseases. Vet. Clin. N. Am. Equine. Pract. 2013, 29, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Sykora, S.; Brandt, S. Papillomavirus infection and squamous cell carcinoma in horses. Vet. J. 2017, 223, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Lange, C.E.; Tobler, K.; Ackermann, M.; Favrot, C. Identification of two novel equine papillomavirus sequences suggests three genera in one cluster. Vet. Microbiol. 2011, 149, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Lange, C.E.; Vetsch, E.; Ackermann, M.; Favrot, C.; Tobler, K. Four novel papillomavirus sequences support a broad diversity among equine papillomaviruses. J. Gen. Virol. 2013, 94, 1365–1372. [Google Scholar] [CrossRef] [PubMed]
- Linder, K.E.; Bizikova, P.; Luff, J.; Zhou, D.; Yuan, H.; Breuhaus, B.; Nelson, E.; Mackay, R. Generalized papillomatosis in three horses associated with a novel equine papillomavirus (EcPV8). Vet. Dermatol. 2018, 29, 72-e30. [Google Scholar] [CrossRef] [PubMed]
- Scase, T.; Brandt, S.; Kainzbauer, C.; Sykora, S.; Bijmholt, S.; Hughes, K.; Sharpe, S.; Foote, A. Equus caballus papillomavirus-2 (EcPV-2): An infectious cause for equine genital cancer? Equine. Vet. J. 2010, 42, 738–745. [Google Scholar] [CrossRef] [PubMed]
- Lecis, R.; Tore, G.; Scagliarini, A.; Antuofermo, E.; Dedola, C.; Cacciotto, C.; Dore, G.M.; Coradduzza, E.; Gallina, L.; Battilani, M.; et al. Equus asinus papillomavirus (EaPV1) provides new insights into equine papillomavirus diversity. Vet. Microbiol. 2014, 170, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Buchfink, B.; Xie, C.; Huson, D.H. Fast and sensitive protein alignment using diamond. Nat. Meth. 2015, 12, 59–60. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Luo, R.; Liu, C.M.; Leung, C.M.; Ting, H.F.; Sadakane, K.; Yamashita, H.; Lam, T.W. MEGAHIT v1.0: A fast and scalable metagenome assembler driven by advanced methodologies and community practices. Methods 2016, 102, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Liu, C.M.; Luo, R.; Sadakane, K.; Lam, T.W. MEGAHIT: An ultra-fast single-node solution for large and complex metagenomics assembly via succinct de Bruijn graph. Bioinformatics 2015, 31, 1674–1676. [Google Scholar] [CrossRef] [PubMed]
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; Buxton, S.; Cooper, A.; Markowitz, S.; Duran, C.; et al. Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef] [PubMed]
- Peters-Kennedy, J.; Lange, C.E.; Rine, S.L.; Hacket, R.P. Equus caballus papillomavirus 8 (EcPV8) associated with multiple viral plaques, viral papillomas, and squamous cell carcinoma in a horse. Equine Vet. J. 2019, 51, 470–474. [Google Scholar] [CrossRef] [PubMed]
- Hans-Ulrich, B.; Burk, R.D.; Chen, Z.; Van Doorslaer, K.; zur Hausen, H.; De Villiers, E.-M. Classification of papillomaviruses (PVs) based on 189 PV types and proposal and taxonomic amendments. Virology 2010, 401, 70–79. [Google Scholar]
- Gardiol, D.; Kühne, C.; Glaunsinger, B.; Lee, S.S.; Javier, R.; Banks, L. Oncogenic human papillomavirus E6 proteins target the discs large tumour suppressor for proteasome-mediated degradation. Oncogene 1999, 18, 5487–5496. [Google Scholar] [CrossRef] [PubMed]
- Wise-Draper, T.M.; Wells, S.I. Papillomavirus E6 and E7 proteins and their cellular targets. Front. Biosci. 2008, 13, 1003–1017. [Google Scholar] [CrossRef] [PubMed]
- Narechania, A.; Terai, M.; Chen, C.Z.; DeSalle, R.; Burk, R.D. Lack of the canonical pRB-binding domain in the E7 ORF of artiodactyl papillomaviruses is associated with the development of fibropapillomas. J. Gen. Virol. 2004, 85, 1243–1250. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Standley, D.M. Mafft multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Gascuel, O. A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst. Biol. 2003, 52, 696–704. [Google Scholar] [CrossRef] [PubMed]
| EcPV9 | EcPV2 (%) | EcPV4 (%) | EcPV5 (%) | |
|---|---|---|---|---|
| E6 | nt | 52.85 | 42.93 | 44.17 |
| aa | 41.86 | 35.16 | 34.88 | |
| E7 | nt | 40.23 | 43.02 | 50.74 |
| aa | 33.04 | 27.18 | 33.98 | |
| E1 | nt | 62.20 | 51.12 | 53.81 |
| aa | 58.63 | 44.97 | 46.56 | |
| E2 | nt | 51.05 | 49.54 | 48.77 |
| aa | 42.79 | 35.31 | 36.38 | |
| E4 | nt | 49.32 | 40.83 | 42.19 |
| aa | 40.14 | 33.33 | 32.24 | |
| L2 | nt | 59.49 | 46.60 | 49.63 |
| aa | 59.17 | 42.55 | 45.07 | |
| L1 | nt | 69.35 | 59.25 | 58.00 |
| aa | 70.44 | 57.74 | 59.88 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, C.-X.; Chang, W.-S.; Mitsakos, K.; Rodger, J.; Holmes, E.C.; Hudson, B.J. Identification of a Novel Equine Papillomavirus in Semen from a Thoroughbred Stallion with a Penile Lesion. Viruses 2019, 11, 713. https://doi.org/10.3390/v11080713
Li C-X, Chang W-S, Mitsakos K, Rodger J, Holmes EC, Hudson BJ. Identification of a Novel Equine Papillomavirus in Semen from a Thoroughbred Stallion with a Penile Lesion. Viruses. 2019; 11(8):713. https://doi.org/10.3390/v11080713
Chicago/Turabian StyleLi, Ci-Xiu, Wei-Shan Chang, Katerina Mitsakos, James Rodger, Edward C. Holmes, and Bernard J. Hudson. 2019. "Identification of a Novel Equine Papillomavirus in Semen from a Thoroughbred Stallion with a Penile Lesion" Viruses 11, no. 8: 713. https://doi.org/10.3390/v11080713
APA StyleLi, C.-X., Chang, W.-S., Mitsakos, K., Rodger, J., Holmes, E. C., & Hudson, B. J. (2019). Identification of a Novel Equine Papillomavirus in Semen from a Thoroughbred Stallion with a Penile Lesion. Viruses, 11(8), 713. https://doi.org/10.3390/v11080713





