Animal Models for Hepatitis E Virus
Abstract
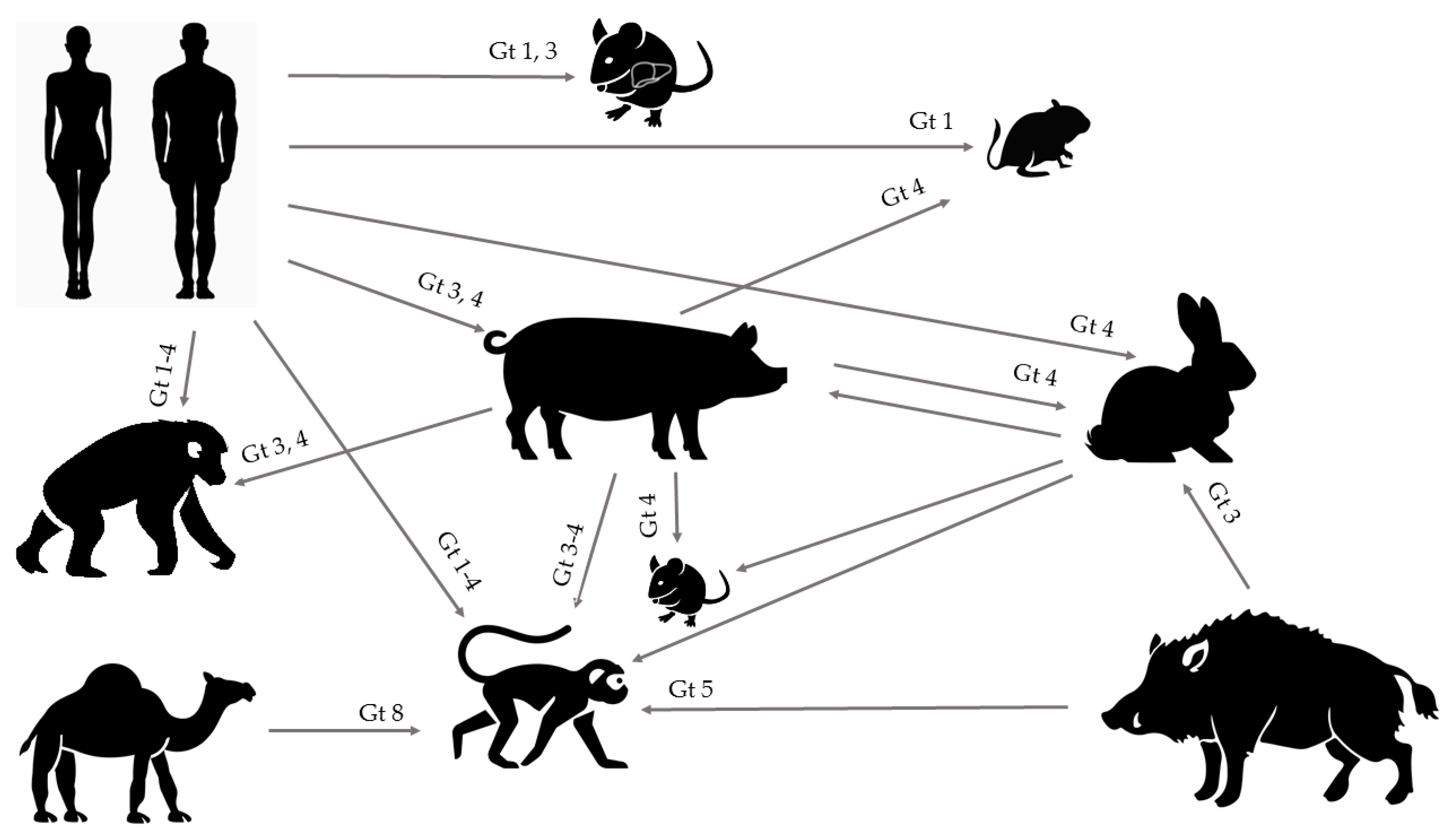
1. Introduction
2. Non-Human Primate Model
2.1. Experimental Infection of Non-Human Primates
2.2. NHP as Animal Model for HEV
2.3. Limitations of NHP Models
3. Pig Model
3.1. Swine HEV and Course of Infection in Swine
3.2. Pigs as an Animal Model for HEV
3.3. Limitations of the Pig Model
4. Rabbit Model
4.1. Rabbit HEV and Course of Infection in Rabbits
4.2. Rabbit as Animal Model for HEV
4.3. Limitations of the Rabbit Model
5. Rat Model
5.1. Rat HEV and Course of Infection in Rats
5.2. Rat as Animal Model for HEV and the Associated Limitations
6. Mouse Model
6.1. Mouse as Animal Model for HEV Infection
6.2. The Limitations of Mice as HEV Animal Model
7. Chicken Model
7.1. Avian HEV and Course of Infection in Chickens
7.2. Chicken as an Animal Model and Its Limitations
8. Other Small Animal Models
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Debing, Y.; Moradpour, D.; Neyts, J.; Gouttenoire, J. Update on hepatitis E virology: Implications for clinical practice. J. Hepatol. 2016, 65, 200–212. [Google Scholar] [CrossRef] [PubMed]
- Wong, D.C.; Purcell, R.H.; Sreenivasan, M.A.; Prasad, S.R.; Pavri, K.M. Epidemic and endemic hepatitis in India: Evidence for a non-A, non-B hepatitis virus aetiology. Lancet 1980, 2, 876–879. [Google Scholar] [CrossRef]
- Koonin, E.V.; Gorbalenya, A.E.; Purdy, M.A.; Rozanov, M.N.; Reyes, G.R.; Bradley, D.W. Computer-assisted assignment of functional domains in the nonstructural polyprotein of hepatitis E virus: Delineation of an additional group of positive-strand RNA plant and animal viruses. Proc. Natl. Acad. Sci. USA 1992, 89, 8259–8263. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, S.; Teng, J.L.L.; Chiu, T.H.; Lau, S.K.P.; Woo, P.C.Y. Hepatitis E Virus Genotypes and Evolution: Emergence of Camel Hepatitis E Variants. Int. J. Mol. Sci. 2017, 18, 869. [Google Scholar] [CrossRef] [PubMed]
- Li, T.C.; Wakita, T. Small Animal Models of Hepatitis E Virus Infection. Cold Spring Harb. Perspect. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Kenney, S.P.; Meng, X.J. Hepatitis E Virus: Animal Models and Zoonosis. Annu. Rev. Anim. Biosci. 2019, 7, 427–448. [Google Scholar] [CrossRef]
- Xing, L.; Wang, J.C.; Li, T.C.; Yasutomi, Y.; Lara, J.; Khudyakov, Y.; Schofield, D.; Emerson, S.U.; Purcell, R.H.; Takeda, N.; et al. Spatial configuration of hepatitis E virus antigenic domain. J. Virol. 2011, 85, 1117–1124. [Google Scholar] [CrossRef]
- Montpellier, C.; Wychowski, C.; Sayed, I.M.; Meunier, J.C.; Saliou, J.M.; Ankavay, M.; Bull, A.; Pillez, A.; Abravanel, F.; Helle, F.; et al. Hepatitis E Virus Lifecycle and Identification of 3 Forms of the ORF2 Capsid Protein. Gastroenterology 2018, 154, 211–223. [Google Scholar] [CrossRef]
- Yin, X.; Ying, D.; Lhomme, S.; Tang, Z.; Walker, C.M.; Xia, N.; Zheng, Z.; Feng, Z. Origin, antigenicity, and function of a secreted form of ORF2 in hepatitis E virus infection. Proc. Natl. Acad. Sci. USA 2018, 115, 4773–4778. [Google Scholar] [CrossRef]
- Ding, Q.; Heller, B.; Capuccino, J.M.; Song, B.; Nimgaonkar, I.; Hrebikova, G.; Contreras, J.E.; Ploss, A. Hepatitis E virus ORF3 is a functional ion channel required for release of infectious particles. Proc. Natl. Acad. Sci. USA 2017, 114, 1147–1152. [Google Scholar] [CrossRef]
- Smith, D.B.; Simmonds, P.; International Committee on Taxonomy of Viruses Hepeviridae Study Group; Jameel, S.; Emerson, S.U.; Harrison, T.J.; Meng, X.J.; Okamoto, H.; Van der Poel, W.H.; Purdy, M.A. Consensus proposals for classification of the family Hepeviridae. J. Gen. Virol. 2014, 95 Pt 10, 2223–2232. [Google Scholar]
- von Nordheim, M.; Boinay, M.; Leisi, R.; Kempf, C.; Ros, C. Cutthroat Trout Virus-Towards a Virus Model to Support Hepatitis E Research. Viruses 2016, 8, 289. [Google Scholar] [CrossRef] [PubMed]
- Kamar, N.; Dalton, H.R.; Abravanel, F.; Izopet, J. Hepatitis E virus infection. Clin. Microbiol. Rev. 2014, 27, 116–138. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, R. Hepatitis E: Clinical presentation in disease-endemic areas and diagnosis. Semin. Liver Dis. 2013, 33, 30–40. [Google Scholar] [PubMed]
- Sayed, I.M.; Vercouter, A.S.; Abdelwahab, S.F.; Vercauteren, K.; Meuleman, P. Is hepatitis E virus an emerging problem in industrialized countries? Hepatology 2015, 62, 1883–1892. [Google Scholar] [CrossRef] [PubMed]
- Lack, J.B.; Volk, K.; Van Den Bussche, R.A. Hepatitis E virus genotype 3 in wild rats, United States. Emerg. Infect. Dis. 2012, 18, 1268–1273. [Google Scholar] [CrossRef] [PubMed]
- Simanavicius, M.; Juskaite, K.; Verbickaite, A.; Jasiulionis, M.; Tamosiunas, P.L.; Petraityte-Burneikiene, R.; Zvirbliene, A.; Ulrich, R.G.; Kucinskaite-Kodze, I. Detection of rat hepatitis E virus, but not human pathogenic hepatitis E virus genotype 1-4 infections in wild rats from Lithuania. Vet. Microbiol. 2018, 221, 129–133. [Google Scholar] [CrossRef]
- Brassard, J.; Gagne, M.J.; Genereux, M.; Cote, C. Detection of human food-borne and zoonotic viruses on irrigated, field-grown strawberries. Appl. Environ. Microbiol. 2012, 78, 3763–3766. [Google Scholar] [CrossRef]
- Kokkinos, P.; Kozyra, I.; Lazic, S.; Bouwknegt, M.; Rutjes, S.; Willems, K.; Moloney, R.; de Roda Husman, A.M.; Kaupke, A.; Legaki, E.; et al. Harmonised investigation of the occurrence of human enteric viruses in the leafy green vegetable supply chain in three European countries. Food Environ. Virol. 2012, 4, 179–191. [Google Scholar] [CrossRef]
- Yugo, D.M.; Meng, X.J. Hepatitis E virus: Foodborne, waterborne and zoonotic transmission. Int. J. Environ. Res. Public Health 2013, 10, 4507–4533. [Google Scholar] [CrossRef]
- Cacopardo, B.; Russo, R.; Preiser, W.; Benanti, F.; Brancati, G.; Nunnari, A. Acute hepatitis E in Catania (eastern Sicily) 1980–1994. The role of hepatitis E virus. Infection 1997, 25, 313–316. [Google Scholar] [CrossRef] [PubMed]
- Ijaz, S.; Arnold, E.; Banks, M.; Bendall, R.P.; Cramp, M.E.; Cunningham, R.; Dalton, H.R.; Harrison, T.J.; Hill, S.F.; Macfarlane, L.; et al. Non-travel-associated hepatitis E in England and Wales: Demographic, clinical, and molecular epidemiological characteristics. J. Infect. Dis. 2005, 192, 1166–1172. [Google Scholar] [CrossRef] [PubMed]
- Renou, C.; Moreau, X.; Pariente, A.; Cadranel, J.F.; Maringe, E.; Morin, T.; Causse, X.; Payen, J.L.; Izopet, J.; Nicand, E.; et al. A national survey of acute hepatitis E in France. Aliment. Pharmacol. Ther. 2008, 27, 1086–1093. [Google Scholar] [CrossRef] [PubMed]
- Said, B.; Ijaz, S.; Kafatos, G.; Booth, L.; Thomas, H.L.; Walsh, A.; Ramsay, M.; Morgan, D.; Hepatitis, E.I.I.T. Hepatitis E outbreak on cruise ship. Emerg. Infect. Dis. 2009, 15, 1738–1744. [Google Scholar] [CrossRef] [PubMed]
- Hunter, J.G.; Madden, R.G.; Stone, A.M.; Osborne, N.; Wheeler, B.; Vine, L.; Dickson, A.; Barlow, M.; Lewis, J.; Bendall, R.P.; et al. Coastal clustering of HEV; Cornwall, UK. Eur. J. Gastroenterol. Hepatol. 2016, 28, 323–327. [Google Scholar] [CrossRef]
- Doceul, V.; Bagdassarian, E.; Demange, A.; Pavio, N. Zoonotic Hepatitis E Virus: Classi fi cation, Animal Reservoirs and Transmission Routes. Viruses 2016, 8, 270. [Google Scholar] [CrossRef]
- Kamar, N.; Bendall, R.; Legrand-Abravanel, F.; Xia, N.S.; Ijaz, S.; Izopet, J.; Dalton, H.R. Hepatitis E. Lancet 2012, 379, 2477–2488. [Google Scholar] [CrossRef]
- Pischke, S.; Peron, J.M.; von Wulffen, M.; von Felden, J.; Honer Zu Siederdissen, C.; Fournier, S.; Lutgehetmann, M.; Iking-Konert, C.; Bettinger, D.; Par, G.; et al. Chronic Hepatitis E in Rheumatology and Internal Medicine Patients: A Retrospective Multicenter European Cohort Study. Viruses 2019, 11, 186. [Google Scholar] [CrossRef]
- Pavio, N.; Meng, X.J.; Renou, C. Zoonotic hepatitis E: Animal reservoirs and emerging risks. Vet. Res. 2010, 41, 46. [Google Scholar] [CrossRef]
- Garcia-Bocanegra, I.; Rivero, A.; Caballero-Gomez, J.; Lopez-Lopez, P.; Cano-Terriza, D.; Frias, M.; Jimenez-Ruiz, S.; Risalde, M.A.; Gomez-Villamandos, J.C.; Rivero-Juarez, A. Hepatitis E virus infection in equines in Spain. Transbound. Emerg. Dis. 2019, 66, 66–71. [Google Scholar] [CrossRef]
- Saad, M.D.; Hussein, H.A.; Bashandy, M.M.; Kamel, H.H.; Earhart, K.C.; Fryauff, D.J.; Younan, M.; Mohamed, A.H. Hepatitis E virus infection in work horses in Egypt. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2007, 7, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Shen, Q.; Mou, J.; Gong, G.; Yang, Z.; Cui, L.; Zhu, J.; Ju, G.; Hua, X. Hepatitis E virus infection among domestic animals in eastern China. Zoonoses Public Health 2008, 55, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.B.; Simmonds, P. Classification and Genomic Diversity of Enterically Transmitted Hepatitis Viruses. Cold Spring Harb. Perspect. Med. 2018, 8, a031880. [Google Scholar] [CrossRef] [PubMed]
- Sridhar, S.; Yip, C.C.Y.; Wu, S.; Cai, J.; Zhang, A.J.; Leung, K.H.; Chung, T.W.H.; Chan, J.F.W.; Chan, W.M.; Teng, J.L.L.; et al. Rat Hepatitis E Virus as Cause of Persistent Hepatitis after Liver Transplant. Emerg. Infect. Dis. 2018, 24, 2241–2250. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Wen, Y.; Xiong, Y.; Zhang, M.; Cheng, M.; Chen, Q. The prevalence and genomic characteristics of hepatitis E virus in murine rodents and house shrews from several regions in China. BMC Vet. Res. 2018, 14, 414. [Google Scholar] [CrossRef]
- Dao Thi, V.L.; Wu, X.; Rice, C.M. Stem Cell-Derived Culture Models of Hepatitis E Virus Infection. Cold Spring Harb. Perspect. Med. 2019, 9, a031799. [Google Scholar] [CrossRef]
- Tanaka, T.; Takahashi, M.; Kusano, E.; Okamoto, H. Development and evaluation of an efficient cell-culture system for Hepatitis E virus. J. Gen. Virol. 2007, 88 Pt 3, 903–911. [Google Scholar] [CrossRef]
- Tanaka, T.; Takahashi, M.; Takahashi, H.; Ichiyama, K.; Hoshino, Y.; Nagashima, S.; Mizuo, H.; Okamoto, H. Development and characterization of a genotype 4 hepatitis E virus cell culture system using a HE-JF5/15F strain recovered from a fulminant hepatitis patient. J. Clin. Microbiol. 2009, 47, 1906–1910. [Google Scholar] [CrossRef]
- Okamoto, H. Hepatitis E virus cell culture models. Virus Res. 2011, 161, 65–77. [Google Scholar] [CrossRef]
- Shukla, P.; Nguyen, H.T.; Faulk, K.; Mather, K.; Torian, U.; Engle, R.E.; Emerson, S.U. Adaptation of a genotype 3 hepatitis E virus to efficient growth in cell culture depends on an inserted human gene segment acquired by recombination. J. Virol. 2012, 86, 5697–5707. [Google Scholar] [CrossRef]
- Shukla, P.; Nguyen, H.T.; Torian, U.; Engle, R.E.; Faulk, K.; Dalton, H.R.; Bendall, R.P.; Keane, F.E.; Purcell, R.H.; Emerson, S.U. Cross-species infections of cultured cells by hepatitis E virus and discovery of an infectious virus-host recombinant. Proc. Natl. Acad. Sci. USA 2011, 108, 2438–2443. [Google Scholar] [CrossRef] [PubMed]
- Adachi, A.; Miura, T. Animal model studies on viral infections. Front. Microbiol. 2014, 5, 672. [Google Scholar] [CrossRef] [PubMed]
- Krawczynski, K.; Meng, X.J.; Rybczynska, J. Pathogenetic elements of hepatitis E and animal models of HEV infection. Virus Res. 2011, 161, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Purcell, R.H.; Engle, R.E.; Govindarajan, S.; Herbert, R.; St Claire, M.; Elkins, W.R.; Cook, A.; Shaver, C.; Beauregard, M.; Swerczek, J.; et al. Pathobiology of hepatitis E: Lessons learned from primate models. Emerg. Microbes Infect. 2013, 2, e9. [Google Scholar] [CrossRef] [PubMed]
- Tsarev, S.A.; Tsareva, T.S.; Emerson, S.U.; Govindarajan, S.; Shapiro, M.; Gerin, J.L.; Purcell, R.H. Successful passive and active immunization of cynomolgus monkeys against hepatitis E. Proc. Natl. Acad. Sci. USA 1994, 91, 10198–10202. [Google Scholar] [CrossRef] [PubMed]
- Tsarev, S.A.; Tsareva, T.S.; Emerson, S.U.; Yarbough, P.O.; Legters, L.J.; Moskal, T.; Purcell, R.H. Infectivity titration of a prototype strain of hepatitis E virus in cynomolgus monkeys. J. Med Virol. 1994, 43, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Balayan, M.S.; Andjaparidze, A.G.; Savinskaya, S.S.; Ketiladze, E.S.; Braginsky, D.M.; Savinov, A.P.; Poleschuk, V.F. Evidence for a virus in non-A, non-B hepatitis transmitted via the fecal-oral route. Intervirology 1983, 20, 23–31. [Google Scholar] [PubMed]
- Ticehurst, J.; Rhodes, L.L., Jr.; Krawczynski, K.; Asher, L.V.; Engler, W.F.; Mensing, T.L.; Caudill, J.D.; Sjogren, M.H.; Hoke, C.H., Jr.; LeDuc, J.W.; et al. Infection of owl monkeys (Aotus trivirgatus) and cynomolgus monkeys (Macaca fascicularis) with hepatitis E virus from Mexico. J. Infect. Dis. 1992, 165, 835–845. [Google Scholar] [CrossRef]
- Tsarev, S.A.; Tsareva, T.S.; Emerson, S.U.; Kapikian, A.Z.; Ticehurst, J.; London, W.; Purcell, R.H. ELISA for antibody to hepatitis E virus (HEV) based on complete open-reading frame-2 protein expressed in insect cells: Identification of HEV infection in primates. J. Infect. Dis. 1993, 168, 369–378. [Google Scholar] [CrossRef]
- Bradley, D.W.; Krawczynski, K.; Cook, E.H., Jr.; McCaustland, K.A.; Humphrey, C.D.; Spelbring, J.E.; Myint, H.; Maynard, J.E. Enterically transmitted non-A, non-B hepatitis: Serial passage of disease in cynomolgus macaques and tamarins and recovery of disease-associated 27- to 34-nm viruslike particles. Proc. Natl. Acad. Sci. USA 1987, 84, 6277–6281. [Google Scholar] [CrossRef]
- Wang, L.; Wang, L. Animal Models for Hepatitis E Virus. Adv. Exp. Med. Boil. 2016, 948, 161–173. [Google Scholar]
- Yugo, D.M.; Cossaboom, C.M.; Meng, X.J. Naturally occurring animal models of human hepatitis E virus infection. ILAR J. 2014, 55, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Bu, Q.N.; Wang, L.; Han, J.; Du, R.J.; Lei, Y.X.; Ouyang, Y.Q.; Li, J.; Zhu, Y.H.; Lu, F.M.; et al. Transmission of hepatitis E virus from rabbits to cynomolgus macaques. Emerg. Infect. Dis. 2013, 19, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Li, T.C.; Bai, H.; Yoshizaki, S.; Ami, Y.; Suzaki, Y.; Doan, Y.H.; Takahashi, K.; Mishiro, S.; Takeda, N.; Wakita, T. Genotype 5 Hepatitis E Virus Produced by a Reverse Genetics System Has the Potential for Zoonotic Infection. Hepatol. Commun. 2019, 3, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Teng, J.L.L.; Lau, S.K.P.; Sridhar, S.; Fu, H.; Gong, W.; Li, M.; Xu, Q.; He, Y.; Zhuang, H.; et al. Transmission of a Novel Genotype of Hepatitis E Virus from Bactrian Camels to Cynomolgus Macaques. J. Virol. 2019, 93, e02014-18. [Google Scholar] [CrossRef]
- Li, T.C.; Yoshizaki, S.; Ami, Y.; Suzaki, Y.; Yang, T.; Takeda, N.; Takaji, W. Monkeys and Rats Are Not Susceptible to Ferret Hepatitis E Virus Infection. Intervirology 2015, 58, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Purcell, R.H.; Engle, R.E.; Rood, M.P.; Kabrane-Lazizi, Y.; Nguyen, H.T.; Govindarajan, S.; St Claire, M.; Emerson, S.U. Hepatitis E virus in rats, Los Angeles, California, USA. Emerg. Infect. Dis. 2011, 17, 2216–2222. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.F.; Sun, Z.F.; Emerson, S.U.; Purcell, R.H.; Shivaprasad, H.L.; Pierson, F.W.; Toth, T.E.; Meng, X.J. Determination and analysis of the complete genomic sequence of avian hepatitis E virus (avian HEV) and attempts to infect rhesus monkeys with avian HEV. J. Gen. Virol. 2004, 85 Pt 6, 1609–1618. [Google Scholar] [CrossRef]
- Emerson, S.U.; Zhang, M.; Meng, X.J.; Nguyen, H.; St Claire, M.; Govindarajan, S.; Huang, Y.K.; Purcell, R.H. Recombinant hepatitis E virus genomes infectious for primates: Importance of capping and discovery of a cis-reactive element. Proc. Natl. Acad. Sci. USA 2001, 98, 15270–15275. [Google Scholar] [CrossRef]
- Tsarev, S.A.; Tsareva, T.S.; Emerson, S.U.; Rippy, M.K.; Zack, P.; Shapiro, M.; Purcell, R.H. Experimental hepatitis E in pregnant rhesus monkeys: Failure to transmit hepatitis E virus (HEV) to offspring and evidence of naturally acquired antibodies to HEV. J. Infect. Dis. 1995, 172, 31–37. [Google Scholar] [CrossRef]
- Gardinali, N.R.; Guimaraes, J.R.; Melgaco, J.G.; Kevorkian, Y.B.; Bottino, F.O.; Vieira, Y.R.; da Silva, A.C.; Pinto, D.P.; da Fonseca, L.B.; Vilhena, L.S.; et al. Cynomolgus monkeys are successfully and persistently infected with hepatitis E virus genotype 3 (HEV-3) after long-term immunosuppressive therapy. PLoS ONE 2017, 12, e0174070. [Google Scholar] [CrossRef] [PubMed]
- Kamili, S. Toward the development of a hepatitis E vaccine. Virus Res. 2011, 161, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Im, S.W.; Zhang, J.Z.; Zhuang, H.; Che, X.Y.; Zhu, W.F.; Xu, G.M.; Li, K.; Xia, N.S.; Ng, M.H. A bacterially expressed peptide prevents experimental infection of primates by the hepatitis E virus. Vaccine 2001, 19, 3726–3732. [Google Scholar] [CrossRef]
- Li, S.W.; Zhang, J.; Li, Y.M.; Ou, S.H.; Huang, G.Y.; He, Z.Q.; Ge, S.X.; Xian, Y.L.; Pang, S.Q.; Ng, M.H.; et al. A bacterially expressed particulate hepatitis E vaccine: Antigenicity, immunogenicity and protectivity on primates. Vaccine 2005, 23, 2893–2901. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, X.F.; Huang, S.J.; Wu, T.; Hu, Y.M.; Wang, Z.Z.; Wang, H.; Jiang, H.M.; Wang, Y.J.; Yan, Q.; et al. Long-term efficacy of a hepatitis E vaccine. New Engl. J. Med. 2015, 372, 914–922. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.C.; Zhang, J.; Zhang, X.F.; Zhou, C.; Wang, Z.Z.; Huang, S.J.; Wang, H.; Yang, C.L.; Jiang, H.M.; Cai, J.P.; et al. Efficacy and safety of a recombinant hepatitis E vaccine in healthy adults: A large-scale, randomised, double-blind placebo-controlled, phase 3 trial. Lancet 2010, 376, 895–902. [Google Scholar] [CrossRef]
- Purcell, R.H.; Nguyen, H.; Shapiro, M.; Engle, R.E.; Govindarajan, S.; Blackwelder, W.C.; Wong, D.C.; Prieels, J.P.; Emerson, S.U. Pre-clinical immunogenicity and efficacy trial of a recombinant hepatitis E vaccine. Vaccine 2003, 21, 2607–2615. [Google Scholar] [CrossRef]
- Zhang, M.; Emerson, S.U.; Nguyen, H.; Engle, R.; Govindarajan, S.; Blackwelder, W.C.; Gerin, J.; Purcell, R.H. Recombinant vaccine against hepatitis E: Duration of protective immunity in rhesus macaques. Vaccine 2002, 20, 3285–3291. [Google Scholar] [CrossRef]
- Shrestha, M.P.; Scott, R.M.; Joshi, D.M.; Mammen, M.P., Jr.; Thapa, G.B.; Thapa, N.; Myint, K.S.; Fourneau, M.; Kuschner, R.A.; Shrestha, S.K.; et al. Safety and efficacy of a recombinant hepatitis E vaccine. New Engl. J. Med. 2007, 356, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.J.; Purcell, R.H.; Halbur, P.G.; Lehman, J.R.; Webb, D.M.; Tsareva, T.S.; Haynes, J.S.; Thacker, B.J.; Emerson, S.U. A novel virus in swine is closely related to the human hepatitis E virus. Proc. Natl. Acad. Sci. USA 1997, 94, 9860–9865. [Google Scholar] [CrossRef]
- Meng, X.J. From barnyard to food table: The omnipresence of hepatitis E virus and risk for zoonotic infection and food safety. Virus Res. 2011, 161, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Kasorndorkbua, C.; Guenette, D.K.; Huang, F.F.; Thomas, P.J.; Meng, X.J.; Halbur, P.G. Routes of transmission of swine hepatitis E virus in pigs. J. Clin. Microbiol. 2004, 42, 5047–5052. [Google Scholar] [CrossRef] [PubMed]
- Bouwknegt, M.; Rutjes, S.A.; Reusken, C.B.; Stockhofe-Zurwieden, N.; Frankena, K.; de Jong, M.C.; de Roda Husman, A.M.; Poel, W.H. The course of hepatitis E virus infection in pigs after contact-infection and intravenous inoculation. BMC Vet. Res. 2009, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Feagins, A.R.; Cordoba, L.; Sanford, B.J.; Dryman, B.A.; Huang, Y.W.; LeRoith, T.; Emerson, S.U.; Meng, X.J. Intergenotypic chimeric hepatitis E viruses (HEVs) with the genotype 4 human HEV capsid gene in the backbone of genotype 3 swine HEV are infectious in pigs. Virus Res. 2011, 156, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Feagins, A.R.; Opriessnig, T.; Huang, Y.W.; Halbur, P.G.; Meng, X.J. Cross-species infection of specific-pathogen-free pigs by a genotype 4 strain of human hepatitis E virus. J. Med. Virol. 2008, 80, 1379–1386. [Google Scholar] [CrossRef] [PubMed]
- Halbur, P.G.; Kasorndorkbua, C.; Gilbert, C.; Guenette, D.; Potters, M.B.; Purcell, R.H.; Emerson, S.U.; Toth, T.E.; Meng, X.J. Comparative pathogenesis of infection of pigs with hepatitis E viruses recovered from a pig and a human. J. Clin. Microbiol. 2001, 39, 918–923. [Google Scholar] [CrossRef] [PubMed]
- Cordoba, L.; Feagins, A.R.; Opriessnig, T.; Cossaboom, C.M.; Dryman, B.A.; Huang, Y.W.; Meng, X.J. Rescue of a genotype 4 human hepatitis E virus from cloned cDNA and characterization of intergenotypic chimeric viruses in cultured human liver cells and in pigs. J. Gen. Virol. 2012, 93 Pt 10, 2183–2194. [Google Scholar] [CrossRef]
- Cossaboom, C.M.; Cordoba, L.; Sanford, B.J.; Pineyro, P.; Kenney, S.P.; Dryman, B.A.; Wang, Y.; Meng, X.J. Cross-species infection of pigs with a novel rabbit, but not rat, strain of hepatitis E virus isolated in the United States. J. Gen. Virol. 2012, 93 Pt 8, 1687–1695. [Google Scholar] [CrossRef]
- Williams, T.P.; Kasorndorkbua, C.; Halbur, P.G.; Haqshenas, G.; Guenette, D.K.; Toth, T.E.; Meng, X.J. Evidence of extrahepatic sites of replication of the hepatitis E virus in a swine model. J. Clin. Microbiol. 2001, 39, 3040–3046. [Google Scholar] [CrossRef]
- Huang, Y.W.; Haqshenas, G.; Kasorndorkbua, C.; Halbur, P.G.; Emerson, S.U.; Meng, X.J. Capped RNA transcripts of full-length cDNA clones of swine hepatitis E virus are replication competent when transfected into Huh7 cells and infectious when intrahepatically inoculated into pigs. J. Virol. 2005, 79, 1552–1558. [Google Scholar] [CrossRef]
- Huang, Y.W.; Opriessnig, T.; Halbur, P.G.; Meng, X.J. Initiation at the third in-frame AUG codon of open reading frame 3 of the hepatitis E virus is essential for viral infectivity in vivo. J. Virol. 2007, 81, 3018–3026. [Google Scholar] [CrossRef] [PubMed]
- Pudupakam, R.S.; Huang, Y.W.; Opriessnig, T.; Halbur, P.G.; Pierson, F.W.; Meng, X.J. Deletions of the hypervariable region (HVR) in open reading frame 1 of hepatitis E virus do not abolish virus infectivity: Evidence for attenuation of HVR deletion mutants in vivo. J. Virol. 2009, 83, 384–395. [Google Scholar] [CrossRef] [PubMed]
- Pudupakam, R.S.; Kenney, S.P.; Cordoba, L.; Huang, Y.W.; Dryman, B.A.; Leroith, T.; Pierson, F.W.; Meng, X.J. Mutational analysis of the hypervariable region of hepatitis e virus reveals its involvement in the efficiency of viral RNA replication. J. Virol. 2011, 85, 10031–10040. [Google Scholar] [CrossRef] [PubMed]
- Sanford, B.J.; Dryman, B.A.; Huang, Y.W.; Feagins, A.R.; Leroith, T.; Meng, X.J. Prior infection of pigs with a genotype 3 swine hepatitis E virus (HEV) protects against subsequent challenges with homologous and heterologous genotypes 3 and 4 human HEV. Virus Res. 2011, 159, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Sanford, B.J.; Opriessnig, T.; Kenney, S.P.; Dryman, B.A.; Cordoba, L.; Meng, X.J. Assessment of the cross-protective capability of recombinant capsid proteins derived from pig, rat, and avian hepatitis E viruses (HEV) against challenge with a genotype 3 HEV in pigs. Vaccine 2012, 30, 6249–6255. [Google Scholar] [CrossRef] [PubMed]
- Cao, D.J.; Cao, Q.M.; Subramaniam, S.; Yugo, D.M.; Heffron, C.L.; Rogers, A.J.; Kenney, S.P.; Tian, D.B.; Matzinger, S.R.; Overend, C.; et al. Pig model mimicking chronic hepatitis E virus infection in immunocompromised patients to assess immune correlates during chronicity. Proc. Natl. Acad. Sci. USA 2017, 114, 6914–6923. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Ma, Z.; Harrison, T.J.; Feng, R.; Zhang, C.; Qiao, Z.; Fan, J.; Ma, H.; Li, M.; Song, A.; et al. novel genotype of hepatitis E virus prevalent among farmed rabbits in China. J. Med. Virol. 2009, 81, 1371–1379. [Google Scholar] [CrossRef] [PubMed]
- Ahn, H.S.; Park, B.J.; Han, S.H.; Kim, Y.H.; Kim, D.H.; Kim, B.S.; Lee, J.B.; Park, S.Y.; Song, C.S.; Lee, S.W.; et al. Prevalence and genetic features of rabbit hepatitis E virus in Korea. J. Med. Virol. 2017, 89, 1995–2002. [Google Scholar] [CrossRef]
- Cossaboom, C.M.; Cordoba, L.; Dryman, B.A.; Meng, X.J. Hepatitis E virus in rabbits, Virginia, USA. Emerg. Infect. Dis. 2011, 17, 2047–2049. [Google Scholar] [CrossRef]
- Hammerschmidt, F.; Schwaiger, K.; Dahnert, L.; Vina-Rodriguez, A.; Hoper, D.; Gareis, M.; Groschup, M.H.; Eiden, M. Hepatitis E virus in wild rabbits and European brown hares in Germany. Zoonoses Public Health 2017, 64, 612–622. [Google Scholar] [CrossRef]
- Izopet, J.; Dubois, M.; Bertagnoli, S.; Lhomme, S.; Marchandeau, S.; Boucher, S.; Kamar, N.; Abravanel, F.; Guerin, J.L. Hepatitis E virus strains in rabbits and evidence of a closely related strain in humans, France. Emerg. Infect. Dis. 2012, 18, 1274–1281. [Google Scholar] [CrossRef] [PubMed]
- Di Bartolo, I.; De Sabato, L.; Marata, A.; Martinelli, N.; Magistrali, C.F.; Monini, M.; Ponterio, E.; Ostanello, F.; Ruggeri, F.M. Serological survey of hepatitis E virus infection in farmed and pet rabbits in Italy. Arch. Virol. 2016, 161, 1343–1346. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Zheng, L.; Liu, Y.; Zhao, C.; Harrison, T.J.; Ma, Y.; Sun, S.; Zhang, J.; Wang, Y. Experimental infection of rabbits with rabbit and genotypes 1 and 4 hepatitis E viruses. PLoS ONE 2010, 5, e9160. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Lei, Y.; Liu, L.; Liu, P.; Xia, J.; Zhang, Y.; Zeng, H.; Wang, L.; Wang, L.; Zhuang, H. SPF rabbits infected with rabbit hepatitis E virus isolate experimentally showing the chronicity of hepatitis. PLoS ONE 2014, 9, e99861. [Google Scholar] [CrossRef] [PubMed]
- Kaba, M.; Richet, H.; Ravaux, I.; Moreau, J.; Poizot-Martin, I.; Motte, A.; Nicolino-Brunet, C.; Dignat-George, F.; Menard, A.; Dhiver, C.; et al. Hepatitis E virus infection in patients infected with the human immunodeficiency virus. J. Med. Virol. 2011, 83, 1704–1716. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xia, J.; Wang, L.; Wang, Y. Experimental infection of rabbits with genotype 3 hepatitis E virus produced both chronicity and kidney injury. Gut 2017, 66, 561–562. [Google Scholar] [CrossRef]
- Xia, J.; Liu, L.; Wang, L.; Zhang, Y.; Zeng, H.; Liu, P.; Zou, Q.; Wang, L.; Zhuang, H. Experimental infection of pregnant rabbits with hepatitis E virus demonstrating high mortality and vertical transmission. J. Viral Hepat. 2015, 22, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Abravanel, F.; Lhomme, S.; El Costa, H.; Schvartz, B.; Peron, J.M.; Kamar, N.; Izopet, J. Rabbit Hepatitis E Virus Infections in Humans, France. Emerg. Infect. Dis. 2017, 23, 1191–1193. [Google Scholar] [CrossRef]
- Cheng, X.F.; Wang, S.; Dai, X.; Shi, C.B.; Wen, Y.F.; Zhu, M.; Zhan, S.W.; Meng, J.H. Rabbit as a Novel Animal Model for Hepatitis E Virus Infection and Vaccine Evaluation. PLoS ONE 2012, 7, e51616. [Google Scholar] [CrossRef]
- Wu, Q.; An, J.; She, R.; Shi, R.; Hao, W.; Soomro, M.; Yuan, X.; Yang, J.; Wang, J. Detection of Genotype 4 Swine Hepatitis E Virus in Systemic Tissues in Cross-Species Infected Rabbits. PLoS ONE 2017, 12, e0171277. [Google Scholar] [CrossRef]
- Schlosser, J.; Dahnert, L.; Dremsek, P.; Tauscher, K.; Fast, C.; Ziegler, U.; Groner, A.; Ulrich, R.G.; Groschup, M.H.; Eiden, M. Different Outcomes of Experimental Hepatitis E Virus Infection in Diverse Mouse Strains, Wistar Rats, and Rabbits. Viruses 2018, 11, 1. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Das, V.; Agarwal, A.; Pandey, A.; Agrawal, S. Fetomaternal outcomes in pregnant women with hepatitis E infection; still an important fetomaternal killer with an unresolved mystery of increased virulence in pregnancy. Turk. J. Obstet. Gynecol. 2017, 14, 106–113. [Google Scholar] [CrossRef] [PubMed]
- Gautam, N.; Ganju, S.; Ganju, S.A.; Walia, S.; Kumar, A.K. Foetomaternal outcomes of hepatitis E infection outbreak in North India. Indian J. Med. Microbiol. 2018, 36, 121–123. [Google Scholar] [PubMed]
- Ahn, H.S.; Han, S.H.; Kim, Y.H.; Park, B.J.; Kim, D.H.; Lee, J.B.; Park, S.Y.; Song, C.S.; Lee, S.W.; Choi, C.; et al. Adverse fetal outcomes in pregnant rabbits experimentally infected with rabbit hepatitis E virus. Virology 2017, 512, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Knegendorf, L.; Drave, S.A.; Dao Thi, V.L.; Debing, Y.; Brown, R.J.P.; Vondran, F.W.R.; Resner, K.; Friesland, M.; Khera, T.; Engelmann, M.; et al. Hepatitis E virus replication and interferon responses in human placental cells. Hepatol. Commun. 2018, 2, 173–187. [Google Scholar] [CrossRef] [PubMed]
- Tian, J.; Shi, R.; Liu, T.; She, R.; Wu, Q.; An, J.; Hao, W.; Soomro, M.H. Brain Infection by Hepatitis E Virus Probably via Damage of the Blood-Brain Barrier Due to Alterations of Tight Junction Proteins. Front. Cell. Infect. Microbiol. 2019, 9, 52. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zeng, H.; Liu, P.; Liu, L.; Xia, J.; Wang, L.; Zou, Q.; Wang, L.; Zhuang, H. Hepatitis E vaccine immunization for rabbits to prevent animal HEV infection and zoonotic transmission. Vaccine 2015, 33, 4922–4928. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Gong, W.; Song, W.T.; Fu, H.; Wang, L.; Li, M.; Wang, L.; Zhuang, H. Different susceptibility and pathogenesis of rabbit genotype 3 hepatitis E virus (HEV-3) and human HEV-3 (JRC-HE3) in SPF rabbits. Vet. Microbiol. 2017, 207, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Johne, R.; Plenge-Bonig, A.; Hess, M.; Ulrich, R.G.; Reetz, J.; Schielke, A. Detection of a novel hepatitis E-like virus in faeces of wild rats using a nested broad-spectrum RT-PCR. J. Gen. Virol. 2010, 91 Pt 3, 750–758. [Google Scholar] [CrossRef]
- Mulyanto; Suparyatmo, J.B.; Andayani, I.G.; Khalid; Takahashi, M.; Ohnishi, H.; Jirintai, S.; Nagashima, S.; Nishizawa, T.; Okamoto, H. Marked genomic heterogeneity of rat hepatitis E virus strains in Indonesia demonstrated on a full-length genome analysis. Virus Res. 2014, 179, 102–112. [Google Scholar]
- Raj, V.S.; Smits, S.L.; Pas, S.D.; Provacia, L.B.; Moorman-Roest, H.; Osterhaus, A.D.; Haagmans, B.L. Novel hepatitis E virus in ferrets, the Netherlands. Emerg. Infect. Dis. 2012, 18, 1369–1370. [Google Scholar] [CrossRef] [PubMed]
- Debing, Y.; Mishra, N.; Verbeken, E.; Ramaekers, K.; Dallmeier, K.; Neyts, J. A rat model for hepatitis E virus. Dis. Model. Mech. 2016, 9, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Maneerat, Y.; Clayson, E.T.; Myint, K.S.; Young, G.D.; Innis, B.L. Experimental infection of the laboratory rat with the hepatitis E virus. J. Med. Virol. 1996, 48, 121–128. [Google Scholar] [CrossRef]
- Rolstad, B. The athymic nude rat: An animal experimental model to reveal novel aspects of innate immune responses? Immunol. Rev. 2001, 184, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Lai, F.; Chen, Q. Humanized Mouse Models for the Study of Infection and Pathogenesis of Human Viruses. Viruses 2018, 10, 643. [Google Scholar] [CrossRef] [PubMed]
- Takaki, H.; Oshiumi, H.; Shingai, M.; Matsumoto, M.; Seya, T. Development of mouse models for analysis of human virus infections. Microbiol. Immunol. 2017, 61, 107–113. [Google Scholar] [CrossRef]
- Sayed, I.M.; Verhoye, L.; Cocquerel, L.; Abravanel, F.; Foquet, L.; Montpellier, C.; Debing, Y.; Farhoudi, A.; Wychowski, C.; Dubuisson, J.; et al. Study of hepatitis E virus infection of genotype 1 and 3 in mice with humanised liver. Gut 2017, 66, 920–929. [Google Scholar] [CrossRef]
- van de Garde, M.D.B.; Pas, S.D.; van der Net, G.; de Man, R.A.; Osterhaus, A.D.M.E.; Haagmans, B.L.; Boonstra, A.; Vanwolleghem, T. Hepatitis E Virus (HEV) Genotype 3 Infection of Human Liver Chimeric Mice as a Model for Chronic HEV Infection. J. Virol. 2016, 90, 4394. [Google Scholar] [CrossRef]
- Allweiss, L.; Gass, S.; Giersch, K.; Groth, A.; Kah, J.; Volz, T.; Rapp, G.; Schobel, A.; Lohse, A.W.; Polywka, S.; et al. Human liver chimeric mice as a new model of chronic hepatitis E virus infection and preclinical drug evaluation. J. Hepatol. 2016, 64, 1033–1040. [Google Scholar] [CrossRef]
- Foquet, L.; Wilson, E.M.; Verhoye, L.; Grompe, M.; Leroux-Roels, G.; Bial, J.; Meuleman, P. Successful Engraftment of Human Hepatocytes in uPA-SCID and FRG((R)) KO Mice. Methods Mol. Boil. 2017, 1506, 117–130. [Google Scholar]
- Rhim, J.A.; Sandgren, E.P.; Degen, J.L.; Palmiter, R.D.; Brinster, R.L. Replacement of diseased mouse liver by hepatic cell transplantation. Science 1994, 263, 1149–1152. [Google Scholar] [CrossRef] [PubMed]
- Sandgren, E.P.; Palmiter, R.D.; Heckel, J.L.; Daugherty, C.C.; Brinster, R.L.; Degen, J.L. Complete hepatic regeneration after somatic deletion of an albumin-plasminogen activator transgene. Cell 1991, 66, 245–256. [Google Scholar] [CrossRef]
- Meuleman, P.; Vanlandschoot, P.; Leroux-Roels, G. A simple and rapid method to determine the zygosity of uPA-transgenic SCID mice. Biochem. Biophys. Res. Commun. 2003, 308, 375–378. [Google Scholar] [CrossRef]
- Azuma, H.; Paulk, N.; Ranade, A.; Dorrell, C.; Al-Dhalimy, M.; Ellis, E.; Strom, S.; Kay, M.A.; Finegold, M.; Grompe, M. Robust expansion of human hepatocytes in Fah−/−/Rag2−/−/Il2rg−/− mice. Nat. Biotechnol. 2007, 25, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Giersch, K.; Allweiss, L.; Volz, T.; Helbig, M.; Bierwolf, J.; Lohse, A.W.; Pollok, J.M.; Petersen, J.; Dandri, M.; Lutgehetmann, M. Hepatitis Delta co-infection in humanized mice leads to pronounced induction of innate immune responses in comparison to HBV mono-infection. J. Hepatol. 2015, 63, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Mikolajczak, S.A.; Vaughan, A.M.; Kangwanrangsan, N.; Roobsoong, W.; Fishbaugher, M.; Yimamnuaychok, N.; Rezakhani, N.; Lakshmanan, V.; Singh, N.; Kaushansky, A.; et al. Plasmodium vivax liver stage development and hypnozoite persistence in human liver-chimeric mice. Cell Host Microbe 2015, 17, 526–535. [Google Scholar] [CrossRef]
- Siu, E.; Ploss, A. Modeling malaria in humanized mice: Opportunities and challenges. Ann. N. Y. Acad. Sci. 2015, 1342, 29–36. [Google Scholar] [CrossRef]
- Vercauteren, K.; de Jong, Y.P.; Meuleman, P. HCV animal models and liver disease. J. Hepatol. 2014, 61 (Suppl. 1), S26–S33. [Google Scholar] [CrossRef]
- Sayed, I.M.; Foquet, L.; Verhoye, L.; Abravanel, F.; Farhoudi, A.; Leroux-Roels, G.; Izopet, J.; Meuleman, P. Transmission of hepatitis E virus infection to human-liver chimeric FRG mice using patient plasma. Antivir. Res. 2017, 141, 150–154. [Google Scholar] [CrossRef]
- Huang, F.; Zhang, W.; Gong, G.; Yuan, C.; Yan, Y.; Yang, S.; Cui, L.; Zhu, J.; Yang, Z.; Hua, X. Experimental infection of Balb/c nude mice with Hepatitis E virus. BMC Infect. Dis. 2009, 9, 93. [Google Scholar] [CrossRef]
- Yu, W.; Yang, C.; Hao, X.; Ma, T.; Huang, F. Successful infection of BALB/c mice by a swine hepatitis E virus clone constructed with reverse genetics. BMC Infect. Dis. 2018, 18, 687. [Google Scholar] [CrossRef]
- Sun, Y.; Lu, Q.; Liu, B.; Sheng, Y.; Du, T.; Hiscox, J.A.; Zhou, E.M.; Zhao, Q. Cross-species infection of mice by rabbit hepatitis E virus. Vet. Microbiol. 2018, 225, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Li, T.C.; Suzaki, Y.; Ami, Y.; Tsunemitsu, H.; Miyamura, T.; Takeda, N. Mice are not susceptible to hepatitis E virus infection. J. Vet. Med. Sci. 2008, 70, 1359–1362. [Google Scholar] [CrossRef] [PubMed]
- Dusseaux, M.; Masse-Ranson, G.; Darche, S.; Ahodantin, J.; Li, Y.; Fiquet, O.; Beaumont, E.; Moreau, P.; Riviere, L.; Neuveut, C.; et al. Viral Load Affects the Immune Response to HBV in Mice With Humanized Immune System and Liver. Gastroenterology 2017, 153, 1647–1661. [Google Scholar] [CrossRef] [PubMed]
- Strick-Marchand, H.; Dusseaux, M.; Darche, S.; Huntington, N.D.; Legrand, N.; Masse-Ranson, G.; Corcuff, E.; Ahodantin, J.; Weijer, K.; Spits, H.; et al. A novel mouse model for stable engraftment of a human immune system and human hepatocytes. PLoS ONE 2015, 10, e0119820. [Google Scholar] [CrossRef]
- Todt, D.; Moeller, N.; Praditya, D.; Kinast, V.; Friesland, M.; Engelmann, M.; Verhoye, L.; Sayed, I.M.; Behrendt, P.; Dao Thi, V.L.; et al. The natural compound silvestrol inhibits hepatitis E virus (HEV) replication in vitro and in vivo. Antivir. Res. 2018, 157, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Haqshenas, G.; Shivaprasad, H.L.; Woolcock, P.R.; Read, D.H.; Meng, X.J. Genetic identification and characterization of a novel virus related to human hepatitis E virus from chickens with hepatitis-splenomegaly syndrome in the United States. J. Gen. Virol. 2001, 82 Pt 10, 2449–2462. [Google Scholar] [CrossRef]
- Marek, A.; Bilic, I.; Prokofieva, I.; Hess, M. Phylogenetic analysis of Avian hepatitis E virus samples from European and Australian chicken flocks supports the existence of a different genus within the Hepeviridae comprising at least three different genotypes. Vet. Microbiol. 2010, 145, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Payne, C.J.; Ellis, T.M.; Plant, S.L.; Gregory, A.R.; Wilcox, G.E. Sequence data suggests big liver and spleen disease virus (BLSV) is genetically related to hepatitis E virus. Vet. Microbiol. 1999, 68, 119–125. [Google Scholar] [CrossRef]
- Su, Q.; Li, Y.; Zhang, Y.; Zhang, Z.; Meng, F.; Cui, Z.; Chang, S.; Zhao, P. Characterization of the novel genotype avian hepatitis E viruses from outbreaks of hepatic rupture haemorrhage syndrome in different geographical regions of China. Transbound. Emerg. Dis. 2018, 65, 2017–2026. [Google Scholar] [CrossRef]
- Peralta, B.; Biarnes, M.; Ordonez, G.; Porta, R.; Martin, M.; Mateu, E.; Pina, S.; Meng, X.J. Evidence of widespread infection of avian hepatitis E virus (avian HEV) in chickens from Spain. Vet. Microbiol. 2009, 137, 31–36. [Google Scholar] [CrossRef]
- Younus, M.; Nisa, Q.; Munir, M.T.; Jamil, T.; Kaboudi, K.; Rehman, Z.U.; Umar, S.; Shah, M.A. Viral hepatitis in chicken and turkeys. World Poult. Sci. J. 2017, 73, 379–394. [Google Scholar] [CrossRef]
- Billam, P.; Huang, F.F.; Sun, Z.F.; Pierson, F.W.; Duncan, R.B.; Elvinger, F.; Guenette, D.K.; Toth, T.E.; Meng, X.J. Systematic pathogenesis and replication of avian hepatitis E virus in specific-pathogen-free adult chickens. J. Virol. 2005, 79, 3429–3437. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Zhou, E.M.; Sun, Z.F.; Meng, X.J. Egg whites from eggs of chickens infected experimentally with avian hepatitis E virus contain infectious virus, but evidence of complete vertical transmission is lacking. J. Gen. Virol. 2007, 88 Pt 5, 1532–1537. [Google Scholar] [CrossRef]
- Guo, H.; Zhou, E.M.; Sun, Z.F.; Meng, X.J. Protection of chickens against avian hepatitis E virus (avian HEV) infection by immunization with recombinant avian HEV capsid protein. Vaccine 2007, 25, 2892–2899. [Google Scholar] [CrossRef]
- Li, T.C.; Yoshizaki, S.; Kataoka, M.; Ami, Y.; Suzaki, Y.; Doan, Y.H.; Haga, K.; Ishii, K.; Takeda, N.; Wakita, T. Genetic and physicochemical analyses of a novel ferret hepatitis E virus, and clinical signs of infection after birth. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2017, 51, 153–159. [Google Scholar] [CrossRef]
- Li, T.C.; Yonemitsu, K.; Terada, Y.; Takeda, N.; Takaji, W.; Maeda, K. Ferret hepatitis E virus infection in Japan. Jpn. J. Infect. Dis. 2015, 68, 60–62. [Google Scholar] [CrossRef] [PubMed]
- Li, T.C.; Yang, T.; Yoshizaki, S.; Ami, Y.; Suzaki, Y.; Ishii, K.; Kishida, N.; Shirakura, M.; Asanuma, H.; Takeda, N.; et al. Ferret hepatitis E virus infection induces acute hepatitis and persistent infection in ferrets. Vet. Microbiol. 2016, 183, 30–36. [Google Scholar] [CrossRef]
- Li, W.; Sun, Q.; She, R.; Wang, D.; Duan, X.; Yin, J.; Ding, Y. Experimental infection of Mongolian gerbils by a genotype 4 strain of swine hepatitis E virus. J. Med Virol. 2009, 81, 1591–1596. [Google Scholar] [CrossRef]
- Yang, Y.; Shi, R.; She, R.; Soomro, M.H.; Mao, J.; Du, F.; Zhao, Y.; Liu, C. Effect of swine hepatitis E virus on the livers of experimentally infected Mongolian gerbils by swine hepatitis E virus. Virus Res. 2015, 208, 171–179. [Google Scholar] [CrossRef]
- Hong, Y.; He, Z.J.; Tao, W.; Fu, T.; Wang, Y.K.; Chen, Y. Experimental infection of Z:ZCLA Mongolian gerbils with human hepatitis E virus. World J. Gastroenterol. 2015, 21, 862–867. [Google Scholar] [CrossRef] [PubMed]
- Shi, R.; Soomro, M.H.; She, R.; Yang, Y.; Wang, T.; Wu, Q.; Li, H.; Hao, W. Evidence of Hepatitis E virus breaking through the blood-brain barrier and replicating in the central nervous system. J. Viral Hepat. 2016, 23, 930–939. [Google Scholar] [CrossRef] [PubMed]
- Soomro, M.H.; Shi, R.; She, R.; Yang, Y.; Wang, T.; Wu, Q.; Li, H.; Hao, W. Molecular and structural changes related to hepatitis E virus antigen and its expression in testis inducing apoptosis in Mongolian gerbil model. J. Viral Hepat. 2017, 24, 696–707. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Xiao, P.; Li, R.; She, R.; Tian, J.; Wang, J.; Mao, J.; Yin, J.; Shi, R. Increased Mast Cell Activation in Mongolian Gerbils Infected by Hepatitis E Virus. Front. Microbiol. 2018, 9, 2226. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corneillie, L.; Banda, D.H.; Meuleman, P. Animal Models for Hepatitis E Virus. Viruses 2019, 11, 564. https://doi.org/10.3390/v11060564
Corneillie L, Banda DH, Meuleman P. Animal Models for Hepatitis E Virus. Viruses. 2019; 11(6):564. https://doi.org/10.3390/v11060564
Chicago/Turabian StyleCorneillie, Laura, Dominic H. Banda, and Philip Meuleman. 2019. "Animal Models for Hepatitis E Virus" Viruses 11, no. 6: 564. https://doi.org/10.3390/v11060564
APA StyleCorneillie, L., Banda, D. H., & Meuleman, P. (2019). Animal Models for Hepatitis E Virus. Viruses, 11(6), 564. https://doi.org/10.3390/v11060564



