The Virioneuston: A Review on Viral–Bacterial Associations at Air–Water Interfaces
Abstract
1. Introduction
2. Viruses as Overlooked Key Players in the SML Realm
3. Viral Abundance and Morphotypes in the SML
| Viral Abundance (×106 mL−1) | EF | n | Bacterial Abundance (×106 cells mL−1) | EF | n | Virus to Bacteria Ratio | Sampling Site | SML Sampling Device | ULW Depth (m) | Reference | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SML | ULW | SML/ULW | SML | ULW | SML/ULW | SML | ULW | ||||||
| 13.2 ± 1.9 | 16.8 ± 3.2 | 0.8 | 3 | 8.6 ± 0.018 | 7.3 ± 0.10 | 1.2 | 3 | 1.5 | 2.3 | Mesocosm experiment, day 7 (Pacific seawater) | Glass plate | 0.9 | [61] |
| 12.1 ± 2.2 | 6.0 ± 1.0 | 2.0 | 3 | 9.6 ± 0.015 | 9.9 ± 0.06 | 1.0 | 3 | 1.3 | 0.6 | Mesocosm experiment, day 8 (Pacific seawater) | Glass plate | 0.9 | [61] |
| 12.1 ± 2.2 | 19.3 ± 2.1 | 0.6 | 3 | 9.8 ± 0.081 | 8.8 ± 0.12 | 1.1 | 3 | 1.2 | 2.2 | Mesocosm experiment, day 9 (Pacific seawater) | Glass plate | 0.9 | [61] |
| 14.3 ± 2.8 | 11.0 ± 3.8 | 1.3 | 3 | 11.7 ± 0.11 | 10.0 ± 0.051 | 1.2 | 3 | 1.2 | 1.1 | Mesocosm experiment, day 10 (Pacific seawater) | Glass plate | 0.9 | [61] |
| 20.8 ± 2.7 | 29.9 ± 3.6 | 0.7 | 3 | 7.2 ± 0.015 | 10.0 ± 0.015 | 0.7 | 3 | 2.1 | 3.0 | Mesocosm experiment, day 16 (Pacific seawater) | Glass plate | 0.9 | [61] |
| 35.4 ± 4.2 | 23.6 ± 2.6 | 1.5 | 3 | 10.9 ± 0.17 | 9.8 ± 0.31 | 1.1 | 3 | 3.2 | 2.4 | Mesocosm experiment, day 17 (Pacific seawater) | Glass plate | 0.9 | [61] |
| 38.5 ± 5.1 | 29.1 ± 4.2 | 1.3 | 3 | 12.0 ± 0.35 | 11.0 ± 0.24 | 1.1 | 3 | 3.2 | 2.6 | Mesocosm experiment, day 18 (Pacific seawater) | Glass plate | 0.9 | [61] |
| 39.0 ± 4.2 | 43.6 ± 3.6 | 0.9 | 3 | 8.6 ± 0.071 | 8.4 ± 0.12 | 1.0 | 3 | 4.5 | 5.2 | Mesocosm experiment, day 21 (Pacific seawater) | Glass plate | 0.9 | [61] |
| 30.4 ± 4.3 | 79.7 ± 7.9 | 0.4 | 3 | 14.7 ± 0.091 | 12.4 ± 0.44 | 1.2 | 3 | 2.1 | 6.4 | Mesocosm experiment, day 22 (Pacific seawater) | Glass plate | 0.9 | [61] |
| 36.6 ± 3.5 | 47.6 ± 6.5 | 0.8 | 3 | 33.5 ± 0.26 | 18.6 ± 0.75 | 1.8 | 3 | 1.1 | 2.6 | Mesocosm experiment, day 23 (Pacific seawater) | Glass plate | 0.9 | [61] |
| 39.6 ± 5.3 | 36.3 ± 5.2 | 1.1 | 3 | 33.3 ± 0.039 | 29.8 ± 1.27 | 1.1 | 3 | 1.2 | 1.2 | Mesocosm experiment, day 24 (Pacific seawater) | Glass plate | 0.9 | [61] |
| 7.3 (4.1–18.4) | 3.1 (2.0–4.9) | 2.4 | 16 | 1.3 (0.5–2.9) | 0.6 (0.5–1.0) | 2.2 | 16 | 6.4 | 5.0 | Halong Bay, Vietnam, Oct. 2012 | Glass plate | 1.5 | [81] |
| 125 (46–204) | 25 (24–26) | 5.1 | 2 | 0.9 (0.9–0.9) | 0.9 (1.0–0.9) | 1.0 | 2 | 138.9 | 27.8 | Mediterranean Sea, Barcelona Site, Sept. 2001 | Metal screen | 0.5 | [74] |
| 1.8 (1.0–2.5) | 1.6 (1.1–2.1) | 1.1 | 2 | 2.6 (1.7–4.0) | 2.4 (1.3–3.9) | 1.2 | 2 | 0.7 | 0.7 | Mediterranean Sea, Barcelona Site, Mar. 2002 | Metal screen | 0.5 | [74] |
| 24.5 | 25.8 | 0.9 | 1 | 1.0 (0.9–1.1) | 0.9 (0.9–1.0) | 1.1 | 2 | 24.5 | 28.7 | Mediterranean Sea, Banyuls Site, Sept. 2001 | Metal screen | 0.5 | [74] |
| 0.4 (0.3–0.4) | 0.3 (0.3–0.4) | 1.1 | 3 | 0.9 (0.8–1.2) | 1.0 (0.8–1.1) | 0.9 | 3 | 0.4 | 0.3 | Mediterranean Sea, Banyuls Site, Mar. 2002 | Metal screen | 0.5 | [74] |
| 10.9 | 1.4 | 7.8 | 1 | 12.4 | 6.2 | 2.0 | 1 | 0.9 | 0.2 | Stony Brook Harbor, NY, June 2003 | Polyester screen | 0.15 | [83] |
| 2.4 | 3.1 | 0.8 | 1 | 2.0 | 1.7 | 1.2 | 1 | 1.2 | 1.8 | Stony Brook Harbor, NY, July 2003 | Polyester screen | 0.15 | [83] |
| 2.4 | 0.9 | 2.7 | 1 | 2.7 | 1.6 | 1.7 | 1 | 0.9 | 0.6 | North Atlantic Ocean, Sample No. 1, June 2001 | Polyester screen | 0.15 | [76] |
| 1.9 | 1.7 | 1.1 | 1 | 1.9 | 1.5 | 1.2 | 1 | 1.3 | 1.1 | North Atlantic Ocean, Sample No. 2, June 2001 | Polyester screen | 0.15 | [76] |
| 2.8 | 3.9 | 0.7 | 1 | 1.6 | 1.5 | 1.1 | 1 | 1.5 | 2.7 | North Atlantic Ocean, Sample No. 3, June 2001 | Polyester screen | 0.15 | [76] |
| 5.5 | 2.9 | 1.9 | 1 | 2.0 | 1.8 | 1.1 | 1 | 1.2 | 1.6 | North Atlantic Ocean, Sample No. 4, June 2001 | Rotating drum | 0.15 | [76] |
| 1.6 | 1.0 | 1.6 | 1 | 1.0 | 1.1 | 0.9 | 1 | 2.4 | 0.9 | North Atlantic Ocean, Sample No. 5, June 2001 | Polyester screen | 0.15 | [76] |
| 4.6 | 2.2 | 2.0 | 1 | 1.3 | 0.7 | 1.7 | 1 | 1.9 | 3.1 | North Atlantic Ocean, Sample No. 6, June 2001 | Polyester screen | 0.15 | [76] |
| 3.0 | 1.2 | 2.4 | 1 | 0.7 | 0.6 | 1.1 | 1 | 3.6 | 2.0 | North Atlantic Ocean, Sample No. 7, June 2001 | Polyester screen | 0.15 | [76] |
| 2.4 | 2.0 | 1.2 | 1 | 0.7 | 0.6 | 1.2 | 1 | 3.5 | 3.4 | North Atlantic Ocean, Sample No. 8, June 2001 | Polyester screen | 0.15 | [76] |
| 2.5 | 2.4 | 1.0 | 1 | 1.2 | 0.8 | 1.5 | 1 | 2.1 | 3.1 | North Atlantic Ocean, Sample No. 9, June 2001 | Polyester screen | 0.15 | [76] |
| 3.2 | 2.8 | 1.2 | 1 | 0.9 | 1.0 | 0.9 | 1 | 2.7 | 2.7 | North Atlantic Ocean, Sample No. 10, June 2001 | Rotating drum | 0.15 | [76] |
| 3.1 | 4.1 | 0.8 | 1 | 1.3 | 0.9 | 1.4 | 1 | 1.9 | 4.5 | North Atlantic Ocean, Sample No. 11, June 2001 | Polyester screen | 0.15 | [76] |
| 3.8 | 3.9 | 1.0 | 1 | 1.0 | 0.8 | 1.2 | 1 | 2.5 | 4.9 | North Atlantic Ocean, Sample No. 12, June 2001 | Polyester screen | 0.15 | [76] |
| 2.3 | 3.5 | 0.7 | 1 | 0.7 | 0.8 | 0.9 | 1 | 3.4 | 4.5 | North Atlantic Ocean, Sample No. 14, June 2001 | Rotating drum | 0.15 | [76] |
| 6.9 | 3.8 | 1.8 | 1 | 1.3 | 0.9 | 1.5 | 1 | 1.9 | 4.5 | North Atlantic Ocean, Sample No. 15, June 2001 | Polyester screen | 0.15 | [76] |
| 3.8 | 3.4 | 1.1 | 1 | 2.2 | 0.9 | 2.3 | 1 | 1.1 | 3.7 | North Atlantic Ocean, Sample No. 16, June 2001 | Polyester screen | 0.15 | [76] |
| 1.9 | 1.7 | 1.1 | 1 | 1.3 | 1.2 | 1.1 | 1 | 1.9 | 1.4 | North Atlantic Ocean, Sample No. 17, June 2001 | Rotating drum | 0.15 | [76] |
| 2.8 | 1.7 | 1.7 | 1 | 1.9 | 1.3 | 1.4 | 1 | 1.3 | 1.3 | North Atlantic Ocean, Sample No. 18, June 2001 | Polyester screen | 0.15 | [76] |
| 3.5 | 3.2 | 1.1 | 1 | 1.2 | 1.0 | 1.2 | 1 | 2.0 | 3.3 | North Atlantic Ocean, Sample No. 19, June 2001 | Polyester screen | 0.15 | [76] |
| 2.7 | 2.0 | 1.4 | 1 | 1.5 | 1.4 | 1.1 | 1 | 1.6 | 1.5 | North Atlantic Ocean, Sample No. 20, June 2001 | Rotating drum | 0.15 | [76] |
| 10.9 | 5.1 | 2.1 | 1 | 2.4 | 1.7 | 1.4 | 1 | 1.0 | 3.1 | North Atlantic Ocean, Sample No. 21, June 2001 | Polyester screen | 0.15 | [76] |
| 1.7 | 1.0 | 1.7 | 1 | 2.6 | 1.9 | 1.4 | 1 | 0.9 | 0.5 | North Atlantic Ocean, Sample No. 22, June 2001 | Rotating drum | 0.15 | [76] |
| 3.1 | 2.0 | 1.5 | 1 | 2.4 | 1.7 | 1.4 | 1 | 1.0 | 1.2 | North Atlantic Ocean, Sample No. 23, June 2001 | Polyester screen | 0.15 | [76] |
| 2.8 | 0.2 | 15.4 | 1 | 5.2 | 2.2 | 2.3 | 1 | 0.5 | 0.1 | Lake Superior, June 1993 | Teflon sheet | 20 | [73] |
| 9.2 | 0.9 | 10.7 | 1 | 18.3 | 1.2 | 15.4 | 1 | 0.1 | 0.7 | Lake Superior, July 1993 | Teflon sheet | 20 | [73] |
| 0.7 | 0.3 | 2.2 | 1 | 1.7 | 1.7 | 0.9 | 1 | 1.5 | 0.2 | Lake Superior, Aug. 1993 | Teflon sheet | 20 | [73] |
| 1.7 | 0.2 | 11.0 | 1 | 9.2 | 4.6 | 2.0 | 1 | 0.3 | 0.03 | Lake Superior, Oct. 1993 | Teflon sheet | 20 | [73] |
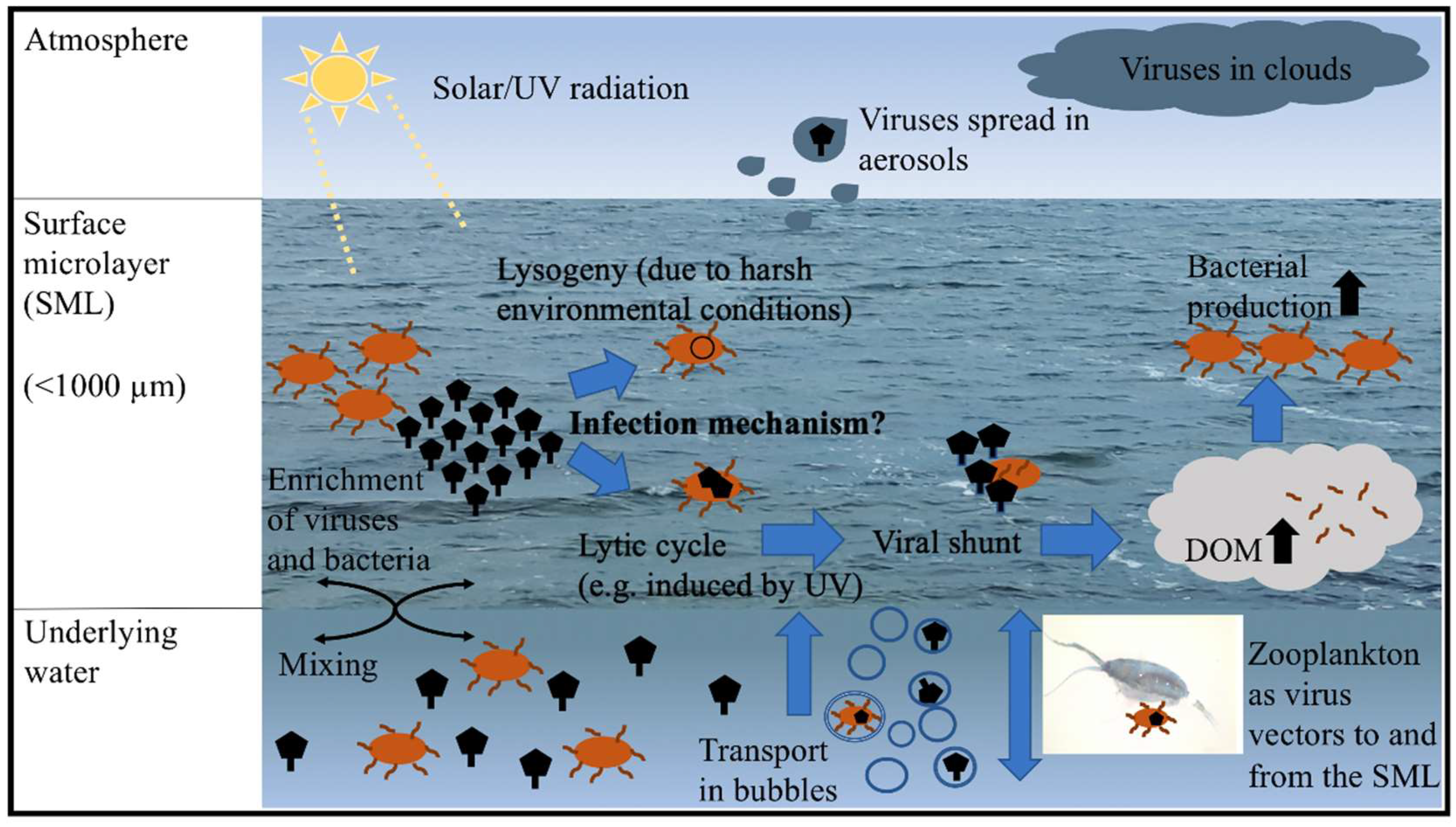
4. Viral Dispersal in and out of the SML
5. Infection Mechanisms of the Virioneuston
6. Future Perspectives and Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Cunliffe, M.; Engel, A.; Frka, S.; Gašparović, B.; Guitart, C.; Murrell, J.C.; Salter, M.; Stolle, C.; Upstill-Goddard, R.; Wurl, O. Sea surface microlayers: A unified physicochemical and biological perspective of the air–ocean interface. Progr. Oceanogr. 2013, 109, 104–116. [Google Scholar] [CrossRef]
- Liss, P.S.; Duce, R.A. The Sea Surface and Global Change; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Hardy, J.T. The sea surface microlayer: Biology, chemistry and anthropogenic enrichment. Progr. Oceanogr. 1982, 11, 307–328. [Google Scholar] [CrossRef]
- Sieburth, J.M.; Willis, P.J.; Johnson, K.M.; Burney, C.M.; Lavoie, D.M.; Hinga, K.R.; Caron, D.A.; French, F.W., 3rd; Johnson, P.W.; Davis, P.G. Dissolved organic matter and heterotrophic microneuston in the surface microlayers of the North Atlantic. Science 1976, 194, 1415–1418. [Google Scholar] [CrossRef] [PubMed]
- Wurl, O.; Miller, L.; Ruttgers, R.; Vagle, S. The distribution and fate of surface-active substances in the sea-surface microlayer and water column. Mar. Chem. 2009, 115, 1–9. [Google Scholar] [CrossRef]
- Wurl, O.; Holmes, M. The gelatinous nature of the sea-surface microlayer. Mar. Chem. 2008, 110, 89–97. [Google Scholar] [CrossRef]
- Cunliffe, M.; Murrell, J.C. The sea-surface microlayer is a gelatinous biofilm. ISME J. 2009, 3, 1001–1003. [Google Scholar] [CrossRef] [PubMed]
- Franklin, M.P.; McDonald, I.R.; Bourne, D.G.; Owens, N.J.; Upstill-Goddard, R.C.; Murrell, J.C. Bacterial diversity in the bacterioneuston (sea surface microlayer): The bacterioneuston through the looking glass. Environ. Microbiol. 2005, 7, 723–736. [Google Scholar] [CrossRef] [PubMed]
- Naumann, E. Beiträge zur Kenntnis des Teichnannoplanktons, II. Über das Neuston des Süsswassers. Biol. Centralblatt 1917, 37, 98–106. [Google Scholar]
- Flemming, H.C.; Wuertz, S. Bacteria and archaea on Earth and their abundance in biofilms. Nat. Rev. Microbiol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Maki, J. The air-water interface as an extreme environment. In Aquatic Microbiology: An Ecological Approach; Ford, T., Ed.; Blackwell Scientific Publication: Boston, MA, USA, 1993; pp. 409–439. [Google Scholar]
- Tsyban, A. Marine bacterioneuston. J. Oceanogr. Soc. Jpn. 1971, 27, 56–66. [Google Scholar] [CrossRef]
- Wurl, O.; Stolle, C.; Van Thuoc, C.; Thu, P.T.; Mari, X. Biofilm-like properties of the sea surface and predicted effects on air-sea CO2 exchange. Progr. Oceanogr. 2016, 144, 15–24. [Google Scholar] [CrossRef]
- Obernosterer, I.; Catala, P.; Reinthaler, T.; Herndl, G.J.; Lebaron, P. Enhanced heterotrophic activity in the surface microlayer of the Mediterranean Sea. Aquat. Microb. Ecol. 2005, 39, 293–302. [Google Scholar] [CrossRef]
- Reinthaler, T.; Sintes, E.; Herndl, G.J. Dissolved organic matter and bacterial production and respiration in the sea-surface microlayer of the open Atlantic and the western Mediterranean Sea. Limnol. Oceanogr. 2008, 53, 122–136. [Google Scholar] [CrossRef]
- Rahlff, J.; Stolle, C.; Wurl, O. SISI: A new device for in situ incubations at the ocean surface. J. Mar. Sci. Eng. 2017, 5, 46. [Google Scholar] [CrossRef]
- Williams, P.M.; Carlucci, A.F.; Henrichs, S.M.; Van Vleet, E.S.; Horrigan, S.G.; Reid, F.M.H.; Robertson, K.J. Chemical and microbiological studies of sea-surface films in the Southern Gulf of California and off the west coast of Baja California. Mar. Chem. 1986, 19, 17–98. [Google Scholar] [CrossRef]
- Albright, L.J. Photosynthetic activities of phytoneuston and phytoplankton. Can. J. Microbiol. 1980, 26, 389–392. [Google Scholar] [CrossRef]
- Yue, W.Z.; Sun, C.C.; Shi, P.; Engel, A.; Wang, Y.S.; He, W.H. Effect of temperature on the accumulation of marine biogenic gels in the surface microlayer near the outlet of nuclear power plants and adjacent areas in the Daya Bay, China. PLoS ONE 2018, 13, e0198735. [Google Scholar] [CrossRef]
- Hardy, J.T.; Coley, J.A.; Antrim, L.D.; Kiesser, S.L. A hydrophobic large-volume sampler for collecting aquatic surface microlayers: Characterization and comparison with the glass plate method. Can. J. Fish. Aquat. Sci. 1988, 45, 822–826. [Google Scholar] [CrossRef]
- Stolle, C.; Labrenz, M.; Meeske, C.; Jürgens, K. Bacterioneuston community structure in the southern Baltic Sea and its dependence on meteorological conditions. Appl. Environ. Microbiol. 2011, 77, 3726–3733. [Google Scholar] [CrossRef]
- Rahlff, J.; Stolle, C.; Giebel, H.A.; Brinkhoff, T.; Ribas-Ribas, M.; Hodapp, D.; Wurl, O. High wind speeds prevent formation of a distinct bacterioneuston community in the sea-surface microlayer. FEMS Microbiol. Ecol. 2017, 93. [Google Scholar] [CrossRef]
- Santos, A.L.; Lopes, S.; Baptista, I.; Henriques, I.; Gomes, N.C.; Almeida, A.; Correia, A.; Cunha, A. Diversity in UV sensitivity and recovery potential among bacterioneuston and bacterioplankton isolates. Lett. Appl. Microbiol. 2011, 52, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Agogué, H.; Joux, F.; Obernosterer, I.; Lebaron, P. Resistance of marine bacterioneuston to solar radiation. Appl. Environ. Microbiol. 2005, 71, 5282–5289. [Google Scholar] [CrossRef] [PubMed]
- Santos, A.L.; Oliveira, V.; Baptista, I.; Henriques, I.; Gomes, N.C.; Almeida, A.; Correia, A.; Cunha, A. Effects of UV-B radiation on the structural and physiological diversity of bacterioneuston and bacterioplankton. Appl. Environ. Microbiol. 2012, 78, 2066–2069. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.T. Phytoneuston ecology of a temperate marine lagoon. Limnol. Oceanogr. 1973, 18, 525–533. [Google Scholar] [CrossRef]
- Norkrans, B. Surface microlayers in aquatic environments. In Advances in Microbial Ecology; Alexander, M., Ed.; Springer: Boston, MA, USA, 1980; Volume 4, pp. 51–85. [Google Scholar]
- Rahlff, J.; Ribas-Ribas, M.; Brown, S.M.; Mustaffa, N.I.H.; Renz, J.; Peck, M.A.; Bird, K.; Cunliffe, M.; Melkonian, K.; Zappa, C.J. Blue pigmentation of neustonic copepods benefits exploitation of a prey-rich niche at the air-sea boundary. Sci. Rep. 2018, 8, 11510. [Google Scholar] [CrossRef]
- Blanchard, D.C. The ejection of drops from the sea and their enrichment with bacteria and other materials: A review. Estuaries Coasts 1989, 12, 127–137. [Google Scholar] [CrossRef]
- Cunliffe, M.; Whiteley, A.S.; Newbold, L.; Oliver, A.; Schäfer, H.; Murrell, J.C. Comparison of bacterioneuston and bacterioplankton dynamics during a phytoplankton bloom in a fjord mesocosm. Appl. Environ. Microbiol. 2009, 75, 7173–7181. [Google Scholar] [CrossRef]
- Cunliffe, M.; Murrell, J.C. Eukarya 18S rRNA gene diversity in the sea surface microlayer: Implications for the structure of the neustonic microbial loop. ISME J. 2010, 4, 455–458. [Google Scholar] [CrossRef]
- Hugoni, M.; Vellet, A.; Debroas, D. Unique and highly variable bacterial communities inhabiting the surface microlayer of an oligotrophic lake. Aquat. Microb. Ecol. 2017, 79, 115–125. [Google Scholar] [CrossRef]
- Hörtnagl, P.; Perez, M.T.; Zeder, M.; Sommaruga, R. The bacterial community composition of the surface microlayer in a high mountain lake. FEMS Microbiol. Ecol. 2010, 73, 458–467. [Google Scholar] [CrossRef]
- Stolle, C.; Nagel, K.; Labrenz, M.; Jürgens, K. Bacterial activity in the sea-surface microlayer: In situ investigations in the Baltic sea and the influence of sampling devices. Aquat. Microb. Ecol. 2009, 58, 67–78. [Google Scholar] [CrossRef]
- Agogué, H.; Casamayor, E.O.; Joux, F.; Obernosterer, I.; Dupuy, C.; Lantoine, F.; Catala, P.; Weinbauer, M.G.; Reinthaler, T.; Herndl, G.J.; et al. Comparison of samplers for the biological characterization of the sea surface microlayer. Limnol. Oceanogr.-Meth. 2004, 2, 213–225. [Google Scholar] [CrossRef]
- Cunliffe, M.; Harrison, E.; Salter, M.; Schäfer, H.; Upstill-Goddard, R.C.; Murrell, J.C. Comparison and validation of sampling strategies for the molecular microbial analysis of surface microlayers. Aquat. Microb. Ecol. 2009, 57, 69–77. [Google Scholar] [CrossRef]
- Brussaard, C.P. Viral control of phytoplankton populations—A review. J. Eukaryot. Microbiol. 2004, 51, 125–138. [Google Scholar] [CrossRef]
- Rohwer, F.; Thurber, R.V. Viruses manipulate the marine environment. Nature 2009, 459, 207–212. [Google Scholar] [CrossRef]
- Engel, A.; Bange, H.W.; Cunliffe, M.; Burrows, S.M.; Friedrichs, G.; Galgani, L.; Herrmann, H.; Hertkorn, N.; Johnson, M.; Liss, P.S.; et al. The ocean’s vital skin: Toward an integrated understanding of the sea surface microlayer. Front. Mar. Sci. 2017, 4. [Google Scholar] [CrossRef]
- Suttle, C.A. Viruses in the sea. Nature 2005, 437, 356–361. [Google Scholar] [CrossRef]
- Fuhrman, J.A. Marine viruses and their biogeochemical and ecological effects. Nature 1999, 399, 541. [Google Scholar] [CrossRef]
- Weinbauer, M.G.; Rassoulzadegan, F. Are viruses driving microbial diversification and diversity? Environ. Microbiol. 2004, 6, 1–11. [Google Scholar] [CrossRef]
- Thingstad, T.F.; Heldal, M.; Bratbak, G.; Dundas, I. Are viruses important partners in pelagic food webs? Trends Ecol. Evol. 1993, 8, 209–213. [Google Scholar] [CrossRef]
- Baylor, E.; Baylor, M.; Blanchard, D.C.; Syzdek, L.D.; Appel, C. Virus transfer from surf to wind. Science 1977, 198, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Mari, X.; Kerros, M.E.; Weinbauer, M.G. Virus attachment to transparent exopolymeric particles along trophic gradients in the southwestern lagoon of New Caledonia. Appl. Environ. Microbiol. 2007, 73, 5245–5252. [Google Scholar] [CrossRef] [PubMed]
- Wilson, T.W.; Ladino, L.A.; Alpert, P.A.; Breckels, M.N.; Brooks, I.M.; Browse, J.; Burrows, S.M.; Carslaw, K.S.; Huffman, J.A.; Judd, C.; et al. A marine biogenic source of atmospheric ice-nucleating particles. Nature 2015, 525, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.W.; Suttle, C.A. Viruses and nutrient cycles in the sea: Viruses play critical roles in the structure and function of aquatic food webs. Bioscience 1999, 49, 781–788. [Google Scholar] [CrossRef]
- Wells, M.L.; Goldberg, E.D. Marine submicron particles. Mar. Chem. 1992, 40, 5–18. [Google Scholar] [CrossRef]
- Alldredge, A.L.; Passow, U.; Logan, B.E. The abundance and significance of a class of large, transparent organic particles in the ocean. Deep Sea Res. Part I Oceanogr. Res. Pap. 1993, 40, 1131–1140. [Google Scholar] [CrossRef]
- Bigg, E.K.; Leck, C.; Tranvik, L. Particulates of the surface microlayer of open water in the central Arctic Ocean in summer. Mar. Chem. 2004, 91, 131–141. [Google Scholar] [CrossRef]
- Chattopadhyay, S.; Puls, R.W. Forces dictating colloidal interactions between viruses and soil. Chemosphere 2000, 41, 1279–1286. [Google Scholar] [CrossRef]
- Weinbauer, M.G.; Bettarel, Y.; Cattaneo, R.; Luef, B.; Maier, C.; Motegi, C.; Peduzzi, P.; Mari, X. Viral ecology of organic and inorganic particles in aquatic systems: Avenues for further research. Aquat. Microb. Ecol. 2009, 57, 321–341. [Google Scholar] [CrossRef]
- Peduzzi, P.; Luef, B. Viruses, bacteria and suspended particles in a backwater and main channel site of the Danube (Austria). Aquat. Sci. 2008, 70, 186–194. [Google Scholar] [CrossRef]
- Wurl, O.; Miller, L.; Vagle, S. Production and fate of transparent exopolymer particles in the ocean. J. Geophys. Res.-Oceans 2011, 116. [Google Scholar] [CrossRef]
- Mari, X.; Passow, U.; Migon, C.; Burd, A.B.; Legendre, L. Transparent exopolymer particles: Effects on carbon cycling in the ocean. Progr. Oceanogr. 2017, 151, 13–37. [Google Scholar] [CrossRef]
- Sabbaghzadeh, B.; Upstill-Goddard, R.C.; Beale, R.; Pereira, R.; Nightingale, P.D. The Atlantic Ocean surface microlayer from 50°N to 50°S is ubiquitously enriched in surfactants at wind speeds up to 13 m s−1. Geophys. Res. Lett. 2017, 44, 2852–2858. [Google Scholar] [CrossRef]
- Satpute, S.K.; Banat, I.M.; Dhakephalkar, P.K.; Banpurkar, A.G.; Chopade, B.A. Biosurfactants, bioemulsifiers and exopolysaccharides from marine microorganisms. Biotechnol. Adv. 2010, 28, 436–450. [Google Scholar] [CrossRef] [PubMed]
- Kurata, N.; Vella, K.; Hamilton, B.; Shivji, M.; Soloviev, A.; Matt, S.; Tartar, A.; Perrie, W. Surfactant-associated bacteria in the near-surface layer of the ocean. Sci. Rep. 2016, 6, 19123. [Google Scholar] [CrossRef]
- Naruse, N.; Tenmyo, O.; Kobaru, S.; Kamei, H.; Miyaki, T.; Konishi, M.; Oki, T. Pumilacidin, a complex of new antiviral antibiotics. Production, isolation, chemical properties, structure and biological activity. J. Antibiot. 1990, 43, 267–280. [Google Scholar] [CrossRef] [PubMed]
- Vollenbroich, D.; Ozel, M.; Vater, J.; Kamp, R.M.; Pauli, G. Mechanism of inactivation of enveloped viruses by the biosurfactant surfactin from Bacillus subtilis. Biologicals 1997, 25, 289–297. [Google Scholar] [CrossRef]
- Michaud, J.M.; Thompson, L.R.; Kaul, D.; Espinoza, J.L.; Richter, R.A.; Xu, Z.Z.; Lee, C.; Pham, K.M.; Beall, C.M.; Malfatti, F.; et al. Taxon-specific aerosolization of bacteria and viruses in an experimental ocean-atmosphere mesocosm. Nat. Commun. 2018, 9, 2017. [Google Scholar] [CrossRef] [PubMed]
- Frew, N.M.; Goldman, J.C.; Dennett, M.R.; Johnson, A.S. Impact of phytoplankton-generated surfactants on air-sea gas exchange. J. Geophys. Res.-Oceans 1990, 95, 3337–3352. [Google Scholar] [CrossRef]
- Goldman, J.C.; Dennett, M.R.; Frew, N.M. Surfactant effects on air-sea gas exchange under turbulent conditions. Deep Sea Res. Part I Oceanogr. Res. Pap. 1988, 35, 1953–1970. [Google Scholar] [CrossRef]
- Bratbak, G.; Egge, J.K.; Heldal, M. Viral mortality of the marine alga Emiliania huxleyi (Haptophyceae) and termination of algal blooms. Mar. Ecol. Prog. Ser. 1993, 93, 39–48. [Google Scholar] [CrossRef]
- Pereira, R.; Ashton, I.; Sabbaghzadeh, B.; Shutler, J.D.; Upstill-Goddard, R.C. Reduced air–sea CO2 exchange in the atlantic ocean due to biological surfactants. Nat. Geosci. 2018, 11, 492–496. [Google Scholar] [CrossRef]
- Romano, J.C. Sea-surface slick occurrence in the open sea (Mediterranean, Red Sea, Indian Ocean) in relation to wind speed. Deep Sea Res. Part I Oceanogr. Res. Pap. 1996, 43, 411–423. [Google Scholar] [CrossRef]
- Sieburth, J.M.; Conover, J.T. Slicks associated with Trichodesmium blooms in the Sargasso Sea. Nature 1965, 205, 830. [Google Scholar] [CrossRef]
- Wurl, O.; Bird, K.; Cunliffe, M.; Landing, W.M.; Miller, U.; Mustaffa, N.I.H.; Ribas-Ribas, M.; Witte, C.; Zappa, C.J. Warming and inhibition of salinization at the ocean’s surface by cyanobacteria. Geophys. Res. Lett. 2018, 45, 4230–4237. [Google Scholar] [CrossRef] [PubMed]
- Maynard, N.G. Aquatic foams as an ecological habitat. Zeitschrift für allgemeine Mikrobiologie 1968, 8, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Eisenreich, S.J.; Elzerman, A.W.; Armstrong, D.E. Enrichment of micronutrients, heavy metals, and chlorinated hydrocarbons in wind-generated lake foam. Environ. Sci. Technol. 1978, 12, 413–417. [Google Scholar] [CrossRef]
- Napolitano, G.E.; Cicerone, D.S. Lipids in water-surface microlayers and foams. In Lipids in Freshwater Ecosystems; Springer: New York, NY, USA, 1999; pp. 235–262. [Google Scholar]
- Thingstad, T.F. Elements of a theory for the mechanisms controlling abundance, diversity, and biogeochemical role of lytic bacterial viruses in aquatic systems. Limnol. Oceanogr. 2000, 45, 1320–1328. [Google Scholar] [CrossRef]
- Tapper, M.A.; Hicks, R.E. Temperate viruses and lysogeny in lake superior bacterioplankton. Limnol. Oceanogr. 1998, 43, 95–103. [Google Scholar] [CrossRef]
- Joux, F.; Agogué, H.; Obernosterer, I.; Dupuy, C.; Reinthaler, T.; Herndl, G.J.; Lebaron, P. Microbial community structure in the sea surface microlayer at two contrasting coastal sites in the northwestern Mediterranean Sea. Aquat. Microb. Ecol. 2006, 42, 91–104. [Google Scholar] [CrossRef]
- GESAMP (IMO/FAO/UNESCO-IOC/WMO/WHO/IAEA/UN/UNEP Joint Group of Experts on the Scientific Aspects of Marine Environemental Protection). Biological effects of chemical and radiative change in the sea surface. In The Sea-Surface Microlayer and Its Role in Global Change; WMO: Geneva, Switzerland, 1995; Volume 59, pp. 27–51. [Google Scholar]
- Kuznetsova, M.; Lee, C.; Aller, J.; Frew, N. Enrichment of amino acids in the sea surface microlayer at coastal and open ocean sites in the North Atlantic Ocean. Limnol. Oceanogr. 2004, 49, 1605–1619. [Google Scholar] [CrossRef]
- Culley, A.I.; Welschmeyer, N.A. The abundance, distribution, and correlation of viruses, phytoplankton, and prokaryotes along a Pacific Ocean transect. Limnol. Oceanogr. 2002, 47, 1508–1513. [Google Scholar] [CrossRef]
- Maranger, R.; Bird, D.F. Viral abundance in aquatic systems:A comparison between marine and fresh waters. Mar. Ecol. Progr. Ser. 1995, 121, 217–226. [Google Scholar] [CrossRef]
- Rahlff, J.; Stolle, C.; Giebel, H.A.; Ribas-Ribas, M.; Damgaard, L.R.; Wurl, O. Oxygen profiles across the sea-surface microlayer-effects of diffusion and biological activity. Front. Mar. Sci. 2019, 6. [Google Scholar] [CrossRef]
- Knowles, B.; Silveira, C.B.; Bailey, B.A.; Barott, K.; Cantu, V.A.; Cobian-Guemes, A.G.; Coutinho, F.H.; Dinsdale, E.A.; Felts, B.; Furby, K.A.; et al. Lytic to temperate switching of viral communities. Nature 2016, 531, 466–470. [Google Scholar] [CrossRef] [PubMed]
- Ram, A.S.P.; Mari, X.; Brune, J.; Torreton, J.P.; Chu, V.T.; Raimbault, P.; Niggemann, J.; Sime-Ngando, T. Bacterial-viral interactions in the sea surface microlayer of a black carbon-dominated tropical coastal ecosystem (Halong Bay, Vietnam). Elementa-Sci. Anthrop. 2018, 6, 13. [Google Scholar]
- Sullivan, M.B.; Waterbury, J.B.; Chisholm, S.W. Cyanophages infecting the oceanic cyanobacterium Prochlorococcus. Nature 2003, 424, 1047–1051. [Google Scholar] [CrossRef]
- Kuznetsova, M.; Lee, C.; Aller, J. Characterization of the proteinaceous matter in marine aerosols. Mar. Chem. 2005, 96, 359–377. [Google Scholar] [CrossRef]
- Aller, J.Y.; Kuznetsova, M.R.; Jahns, C.J.; Kemp, P.F. The sea surface microlayer as a source of viral and bacterial enrichment in marine aerosols. J. Aerosol Sci. 2005, 36, 801–812. [Google Scholar] [CrossRef]
- Woodcock, A.H. Subsurface pelagic Sargassum. J. Mar. Res. 1950, 9, 77–92. [Google Scholar]
- Castello, J.D. Detection of infectious tomato mosaic tobamovirus in fog and clouds. Phytopathology 1995, 85, 1409–1412. [Google Scholar] [CrossRef]
- Rastelli, E.; Corinaldesi, C.; Dell’Anno, A.; Lo Martire, M.; Greco, S.; Cristina Facchini, M.; Rinaldi, M.; O’Dowd, C.; Ceburnis, D.; Danovaro, R. Transfer of labile organic matter and microbes from the ocean surface to the marine aerosol: An experimental approach. Sci. Rep. 2017, 7, 11475. [Google Scholar] [CrossRef] [PubMed]
- Sharoni, S.; Trainic, M.; Schatz, D.; Lehahn, Y.; Flores, M.J.; Bidle, K.D.; Ben-Dor, S.; Rudich, Y.; Koren, I.; Vardi, A. Infection of phytoplankton by aerosolized marine viruses. Proc. Natl. Acad. Sci. USA 2015, 112, 6643–6647. [Google Scholar] [CrossRef] [PubMed]
- Reche, I.; D’Orta, G.; Mladenov, N.; Winget, D.M.; Suttle, C.A. Deposition rates of viruses and bacteria above the atmospheric boundary layer. ISME J. 2018, 12, 1154–1162. [Google Scholar] [CrossRef] [PubMed]
- Short, C.M.; Suttle, C.A. Nearly identical bacteriophage structural gene sequences are widely distributed in both marine and freshwater environments. Appl. Environ. Microbiol. 2005, 71, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Breitbart, M.; Rohwer, F. Here a virus, there a virus, everywhere the same virus? Trends Microbiol. 2005, 13, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Mojib, N.; Thimma, M.; Kumaran, M.; Sougrat, R.; Irigoien, X. Comparative metatranscriptomics reveals decline of a neustonic planktonic population. Limnol. Oceanogr. 2017, 62, 299–310. [Google Scholar] [CrossRef]
- Zaitsev, Y.P. Marine Neustonology; National Technical Information Service: Springfield, VA, USA, 1971. [Google Scholar]
- Frada, M.J.; Schatz, D.; Farstey, V.; Ossolinski, J.E.; Sabanay, H.; Ben-Dor, S.; Koren, I.; Vardi, A. Zooplankton may serve as transmission vectors for viruses infecting algal blooms in the ocean. Curr. Biol. 2014, 24, 2592–2597. [Google Scholar] [CrossRef] [PubMed]
- Wilhelm, S.W.; Weinbauer, M.G.; Suttle, C.A.; Pledger, R.J.; Mitchell, D.L. Measurements of DNA damage and photoreactivation imply that most viruses in marine surface waters are infective. Aquat. Microb. Ecol. 1998, 14, 215–222. [Google Scholar] [CrossRef]
- Jiang, S.C.; Paul, J.H. Significance of lysogeny in the marine environment: Studies with isolates and a model of lysogenic phage production. Microb. Ecol. 1998, 35, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Wilcox, R.M.; Fuhrman, J.A. Bacterial-viruses in coastal seawater—Lytic rather than lysogenic production. Mar. Ecol. Prog. Ser. 1994, 114, 35–45. [Google Scholar] [CrossRef]
- Jiang, S.C.; Paul, J.H. Occurrence of lysogenic bacteria in marine microbial communities as determined by prophage induction. Mar. Ecol. Progr. Ser. 1996, 142, 27–38. [Google Scholar] [CrossRef]
- Guitart, C.; Garcia-Flor, N.; Bayona, J.M.; Albaiges, J. Occurrence and fate of polycyclic aromatic hydrocarbons in the coastal surface microlayer. Mar. Pollut. Bull. 2007, 54, 186–194. [Google Scholar] [CrossRef] [PubMed]
- Payet, J.P.; Suttle, C.A. To kill or not to kill: The balance between lytic and lysogenic viral infection is driven by trophic status. Limnol. Oceanogr. 2013, 58, 465–474. [Google Scholar] [CrossRef]
- Roux, S.; Enault, F.; Hurwitz, B.L.; Sullivan, M.B. VirSorter: Mining viral signal from microbial genomic data. PeerJ 2015, 3, e985. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Ahlgren, N.A.; Lu, Y.Y.; Fuhrman, J.A.; Sun, F. VirFinder: A novel k-mer based tool for identifying viral sequences from assembled metagenomic data. Microbiome 2017, 5, 69. [Google Scholar] [CrossRef] [PubMed]
- Ahlgren, N.A.; Ren, J.; Lu, Y.Y.; Fuhrman, J.A.; Sun, F. Alignment-free oligonucleotide frequency dissimilarity measure improves prediction of hosts from metagenomically-derived viral sequences. Nucleic Acids Res. 2017, 45, 39–53. [Google Scholar] [CrossRef]
- Hurwitz, B.L.; Ponsero, A.; Thornton, J.; U’Ren, J.M. Phage hunters: Computational strategies for finding phages in large-scale ’omics datasets. Virus Res. 2018, 244, 110–115. [Google Scholar] [CrossRef]
- Andersson, A.F.; Banfield, J.F. Virus population dynamics and acquired virus resistance in natural microbial communities. Science 2008, 320, 1047–1050. [Google Scholar] [CrossRef]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rahlff, J. The Virioneuston: A Review on Viral–Bacterial Associations at Air–Water Interfaces. Viruses 2019, 11, 191. https://doi.org/10.3390/v11020191
Rahlff J. The Virioneuston: A Review on Viral–Bacterial Associations at Air–Water Interfaces. Viruses. 2019; 11(2):191. https://doi.org/10.3390/v11020191
Chicago/Turabian StyleRahlff, Janina. 2019. "The Virioneuston: A Review on Viral–Bacterial Associations at Air–Water Interfaces" Viruses 11, no. 2: 191. https://doi.org/10.3390/v11020191
APA StyleRahlff, J. (2019). The Virioneuston: A Review on Viral–Bacterial Associations at Air–Water Interfaces. Viruses, 11(2), 191. https://doi.org/10.3390/v11020191





