Suggestive Serological Evidence of Infection with Shrew-Borne Imjin Virus (Hantaviridae) in Humans
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Expression of Recombinant MJNV NP
2.3. Immunization of Mice
2.4. ELISA
2.5. RT-PCR
2.6. Dot Blot and Western blot
3. Results
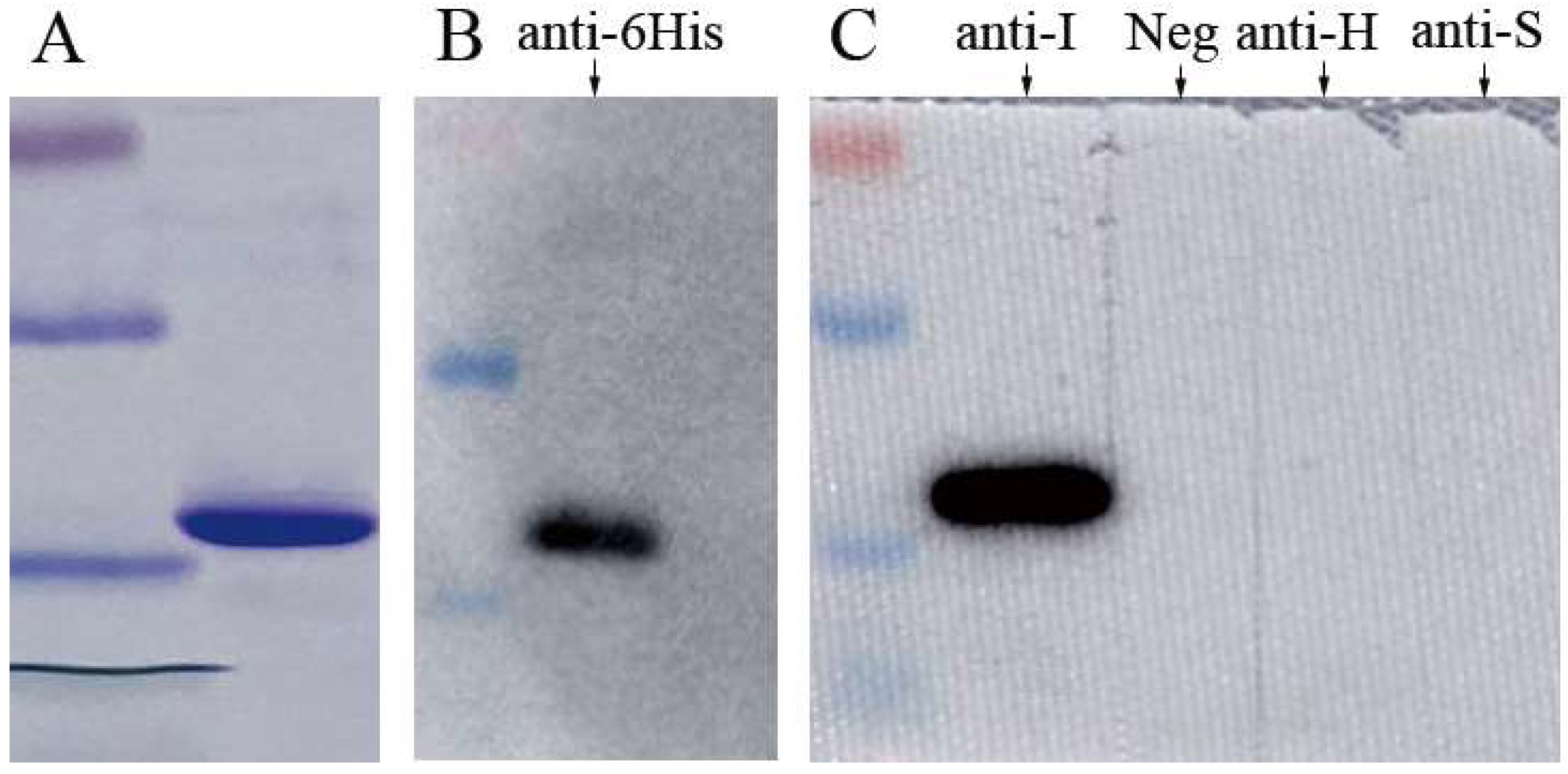
3.1. Establishing ELISA Diagnostic Assay with MJNV NP
3.2. Specificity of MJNV NP
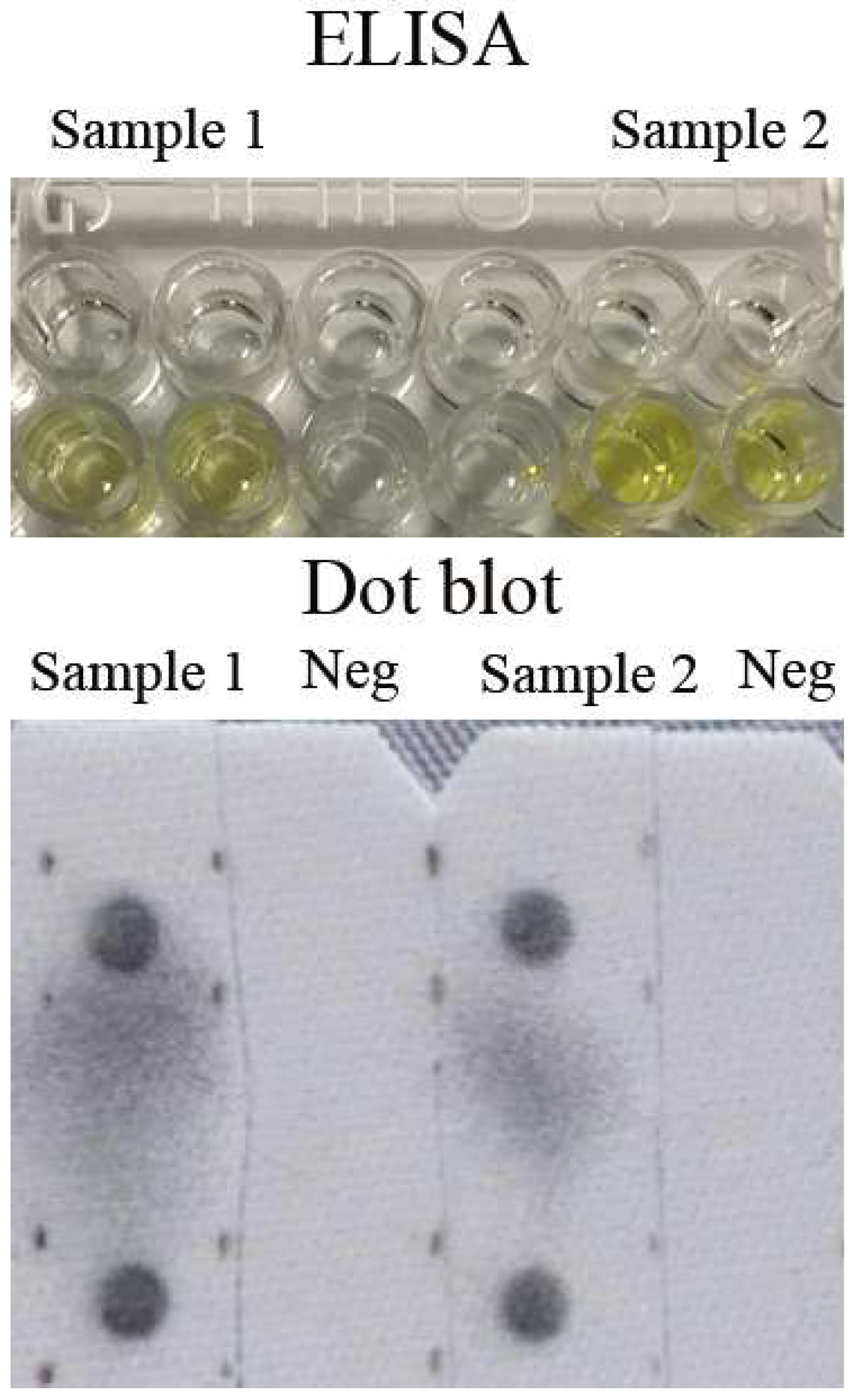
3.3. Prevalence of MJNV in Endemic Area
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jonsson, C.B.; Figueiredo, L.T.; Vapalahti, O. A global perspective on hantavirus ecology, epidemiology, and disease. Clin. Microbiol. Rev. 2010, 23, 412–441. [Google Scholar] [CrossRef] [PubMed]
- Krautkramer, E.; Zeier, M.; Plyusnin, A. Hantavirus infection: An emerging infectious disease causing acute renal failure. Kidney Int. 2013, 83, 23–27. [Google Scholar] [CrossRef] [PubMed]
- King, A.M.Q.; Lefkowitz, E.J.; Mushegian, A.R.; Adams, M.J.; Dutilh, B.E.; Gorbalenya, A.E.; Harrach, B.; Harrison, R.L.; Junglen, S.; Knowles, N.J.; et al. Changes to taxonomy and the International Code of Virus Classification and Nomenclature ratified by the International Committee on Taxonomy of Viruses (2018). Arch. Virol. 2018, 163, 2601–2631. [Google Scholar] [CrossRef] [PubMed]
- Forbes, K.M.; Sironen, T.; Plyusnin, A. Hantavirus maintenance and transmission in reservoir host populations. Curr. Opin. Virol. 2018, 28, 1–6. [Google Scholar] [CrossRef]
- Arai, S.; Ohdachi, S.D.; Asakawa, M.; Kang, H.J.; Mocz, G.; Arikawa, J.; Okabe, N.; Yanagihara, R. Molecular phylogeny of a newfound hantavirus in the Japanese shrew mole (Urotrichus talpoides). Proc. Natl. Acad. Sci. USA 2008, 105, 16296–16301. [Google Scholar] [CrossRef]
- Clement, J.; Maes, P.; Van Ranst, M. Hemorrhagic Fever with Renal Syndrome in the New, and Hantavirus Pulmonary Syndrome in the Old World: Paradi(se)gm lost or regained? Virus Res. 2014, 187, 55–58. [Google Scholar] [CrossRef]
- Chinese Center for Disease Control and Prevention. HFRS surveillance programs (trial). 10 August 2005. Available online: http://www.chinacdc.cn/jkzt/crb/lxxcxr/cxrjc/200508/t20050810_24189.htm (accessed on 26 February 2019).
- Clement, J.; LeDuc, J.W.; Lloyd, G.; Reynes, J.M.; McElhinney, L.; Van Ranst, M.; Lee, H.W. Wild Rats, Laboratory Rats, Pet Rats: Global Seoul Hantavirus Disease Revisited. Viruses 2019, 11, 652. [Google Scholar] [CrossRef]
- Qi, R.; Qin, X.; Wang, L.; Han, H.; Cui, F.; Yu, H.; Yu, X.J. Severe fever with thrombocytopenia syndrome can masquerade as hemorrhagic fever with renal syndrome. PLoS Negl. Trop. Dis. 2013, 3, e0007308. [Google Scholar] [CrossRef]
- Wang, L.; Wang, T.; Cui, F.; Zhai, S.Y.; Zhang, L.; Yang, S.X.; Wang, Z.Q.; Yu, X.J. Hemorrhagic Fever with Renal Syndrome, Zibo City, China, 2006-2014. Emerg. Infect. Dis. 2016, 22, 274–276. [Google Scholar] [CrossRef]
- Jiang, F.; Zhang, Z.; Dong, L.; Hao, B.; Xue, Z.; Ma, D.; Su, H.; Wen, H.L.; Yu, H.; Yu, X.J. Prevalence of hemorrhagic fever with renal syndrome in Qingdao City, China, 2010-2014. Sci. Rep. 2016, 6, 36081. [Google Scholar] [CrossRef]
- Fang, L.Z.; Zhao, L.; Wen, H.L.; Zhang, Z.T.; Liu, J.W.; He, S.T.; Xue, Z.F.; Ma, D.Q.; Zhang, X.S.; Zhang, Y.; et al. Reservoir host expansion of hantavirus, China. Emerg. Infect. Dis. 2015, 21, 170–171. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.F.; Zhao, L.; Zhang, Z.T.; Liu, M.M.; Xue, Z.F.; Wen, H.L.; Ma, D.Q.; Huang, Y.T.; Sun, Y.; Zhou, C.M.; et al. Detection of Imjin Virus and Seoul Virus in Crocidurine Shrews in Shandong Province, China. Vector Borne. Zoonotic Dis. 2017, 17, 425–431. [Google Scholar] [CrossRef] [PubMed]
- Song, J.W.; Kang, H.J.; Gu, S.H.; Moon, S.S.; Bennett, S.N.; Song, K.J.; Baek, L.J.; Kim, H.C.; O’Guinn, M.L.; Chong, S.T.; et al. Characterization of Imjin virus, a newly isolated hantavirus from the Ussuri white-toothed shrew (Crocidura lasiura). J. Virol. 2009, 83, 6184–6191. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Wang, L.; Wang, S.; Zhu, L.; Dong, L.; Zhang, Z.; Hao, B.; Yang, F.; Liu, W.; Deng, Y.; et al. Meteorological factors affect the epidemiology of hemorrhagic fever with renal syndrome via altering the breeding and hantavirus-carrying states of rodents and mites: A 9 years’ longitudinal study. Emerg. Microbes. Infect. 2017, 6, e104. [Google Scholar] [CrossRef] [PubMed]
- Schlegel, M.; Tegshduuren, E.; Yoshimatsu, K.; Petraityte, R.; Sasnauskas, K.; Hammerschmidt, B.; Friedrich, R.; Mertens, M.; Groschup, M.H.; Arai, S.; et al. Novel serological tools for detection of Thottapalayam virus, a Soricomorpha-borne hantavirus. Arch. Virol. 2012, 157, 2179–2187. [Google Scholar] [CrossRef] [PubMed]
- Saasa, N.; Yoshida, H.; Shimizu, K.; Sanchez-Hernandez, C.; Romero-Almaraz Mde, L.; Koma, T.; Sanada, T.; Seto, T.; Yoshii, K.; Ramos, C.; et al. The N-terminus of the Montano virus nucleocapsid protein possesses broadly cross-reactive conformation-dependent epitopes conserved in rodent-borne hantaviruses. Virology 2012, 428, 48–57. [Google Scholar] [CrossRef]
- Tischler, N.D.; Rosemblatt, M.; Valenzuela, P.D. Characterization of cross-reactive and serotype-specific epitopes on the nucleocapsid proteins of hantaviruses. Virus. Res. 2008, 135, 1–9. [Google Scholar] [CrossRef]
- Elgh, F.; Lundkvist, A.; Alexeyev, O.A.; Wadell, G.; Juto, P. A major antigenic domain for the human humoral response to Puumala virus nucleocapsid protein is located at the amino-terminus. J. Virol. Methods 1996, 59, 161–172. [Google Scholar] [CrossRef]
- Yoshimatsu, K.; Arikawa, J.; Tamura, M.; Yoshida, R.; Lundkvist, A.; Niklasson, B.; Kariwa, H.; Azuma, I. Characterization of the nucleocapsid protein of Hantaan virus strain 76-118 using monoclonal antibodies. J. Gen. Virol. 1996, 77, 695–704. [Google Scholar] [CrossRef]
- Yoshimatsu, K.; Arikawa, J. Antigenic properties of N protein of hantavirus. Viruses 2014, 6, 3097–3109. [Google Scholar] [CrossRef]
- Elgh, F.; Lundkvist, A.; Alexeyev, O.A.; Stenlund, H.; Avsic-Zupanc, T.; Hjelle, B.; Lee, H.W.; Smith, K.J.; Vainionpaa, R.; Wiger, D.; et al. Serological diagnosis of hantavirus infections by an enzyme-linked immunosorbent assay based on detection of immunoglobulin G and M responses to recombinant nucleocapsid proteins of five viral serotypes. J. Clin. Microbiol. 1997, 35, 1122–1130. [Google Scholar] [PubMed]
- Lundkvist, A.; Meisel, H.; Koletzki, D.; Lankinen, H.; Cifire, F.; Geldmacher, A.; Sibold, C.; Gott, P.; Vaheri, A.; Kruger, D.H.; et al. Mapping of B-cell epitopes in the nucleocapsid protein of Puumala hantavirus. Viral. Immunol. 2002, 15, 177–192. [Google Scholar] [CrossRef] [PubMed]
- Gott, P.; Zoller, L.; Darai, G.; Bautz, E.K. A major antigenic domain of hantaviruses is located on the aminoproximal site of the viral nucleocapsid protein. Virus Genes 1997, 14, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Hjelle, B.; Lanzi, R.; Morris, C.; Anderson, B.; Jenison, S. Antibody responses to Four Corners hantavirus infections in the deer mouse (Peromyscus maniculatus): Identification of an immunodominant region of the viral nucleocapsid protein. J. Virol. 1995, 69, 1939–1943. [Google Scholar] [PubMed]
- Yanagihara, R.; Gu, S.H.; Song, J.W. Expanded host diversity and global distribution of hantaviruses: Implications for identifying and investigating previously unrecognized hantaviral diseases. In Global Virology I-identifying and Investigating Viral Diseases; Shapshak, P., Sinnott, J.T., Eds.; Springer: New York, USA, 2015; pp. 185–186. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qi, R.; Sun, X.-F.; Qin, X.-R.; Wang, L.-J.; Zhao, M.; Jiang, F.; Wang, L.; Lei, X.-Y.; Liu, J.-W.; Yu, X.-J. Suggestive Serological Evidence of Infection with Shrew-Borne Imjin Virus (Hantaviridae) in Humans. Viruses 2019, 11, 1128. https://doi.org/10.3390/v11121128
Qi R, Sun X-F, Qin X-R, Wang L-J, Zhao M, Jiang F, Wang L, Lei X-Y, Liu J-W, Yu X-J. Suggestive Serological Evidence of Infection with Shrew-Borne Imjin Virus (Hantaviridae) in Humans. Viruses. 2019; 11(12):1128. https://doi.org/10.3390/v11121128
Chicago/Turabian StyleQi, Rui, Xi-Feng Sun, Xiang-Rong Qin, Li-Jun Wang, Min Zhao, Fachun Jiang, Ling Wang, Xiao-Ying Lei, Jian-Wei Liu, and Xue-Jie Yu. 2019. "Suggestive Serological Evidence of Infection with Shrew-Borne Imjin Virus (Hantaviridae) in Humans" Viruses 11, no. 12: 1128. https://doi.org/10.3390/v11121128
APA StyleQi, R., Sun, X.-F., Qin, X.-R., Wang, L.-J., Zhao, M., Jiang, F., Wang, L., Lei, X.-Y., Liu, J.-W., & Yu, X.-J. (2019). Suggestive Serological Evidence of Infection with Shrew-Borne Imjin Virus (Hantaviridae) in Humans. Viruses, 11(12), 1128. https://doi.org/10.3390/v11121128




