Natural Infection of Aedes aegypti by Chikungunya and Dengue type 2 Virus in a Transition Area of North-Northeast Brazil
Abstract
1. Introduction
2. Materials and Methods
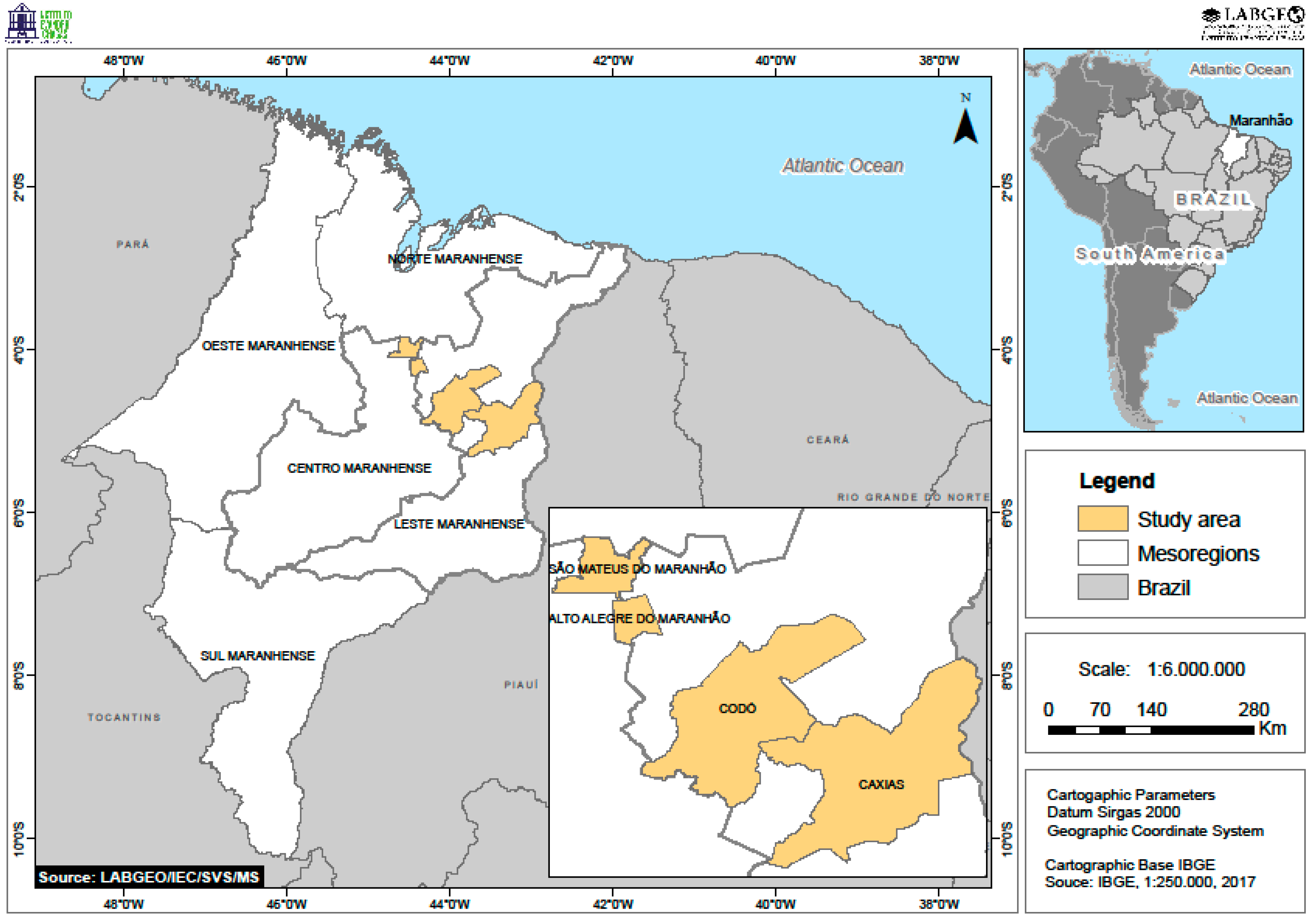
2.1. Study Area
2.2. Insect Collection
2.3. Identification
2.4. Sample Preparation for Analysis
2.5. Viral RNA Extraction
2.6. Real Time Reverse Transcription Polymerase Chain Reaction (RT-qPCR) Assay
2.7. Viral Isolation
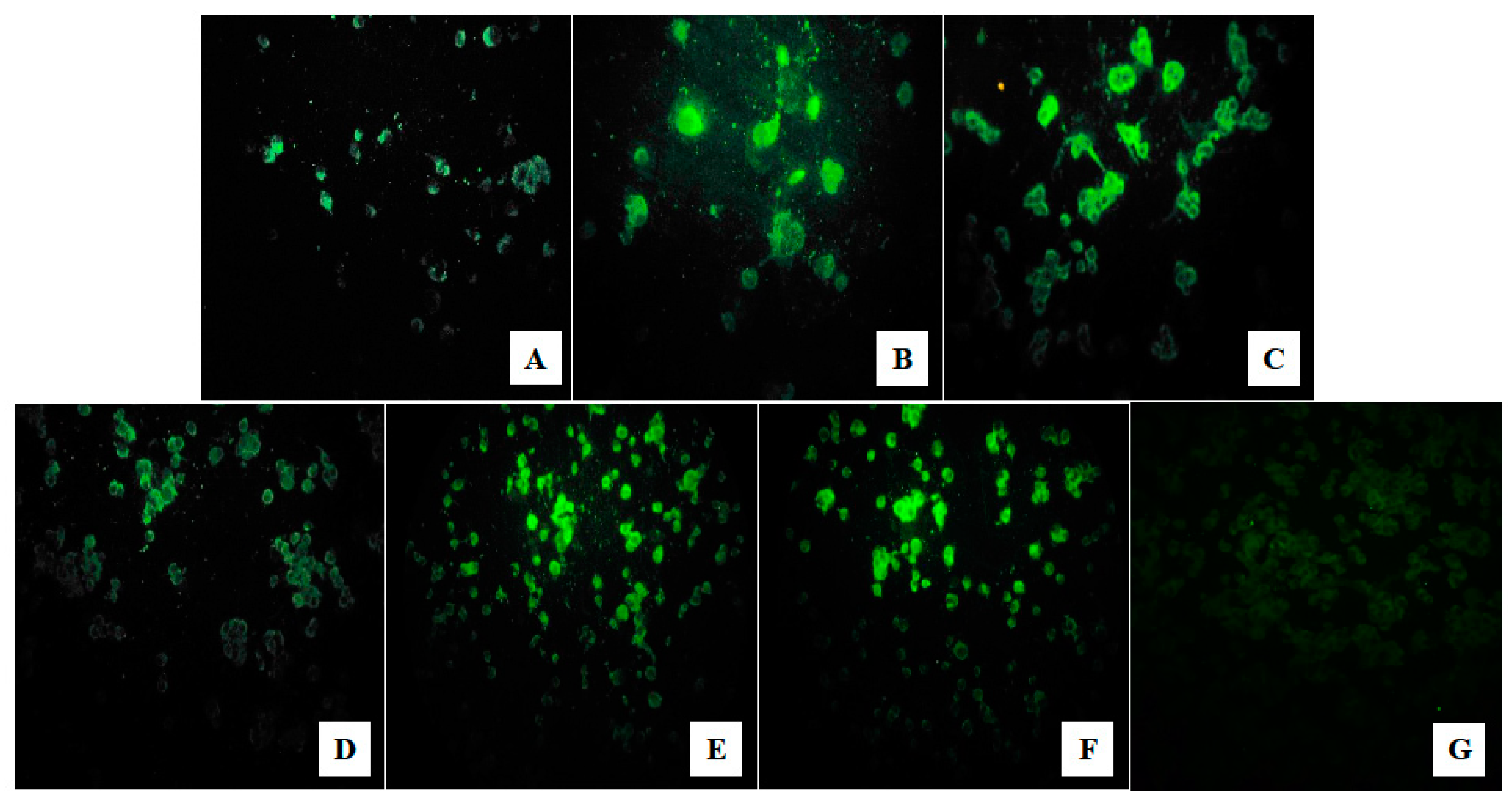
2.8. Indirect Immunofluorescence (IIF) Test
2.9. Nucleotide Sequencing
2.9.1. RNA Extraction
2.9.2. Genomic Sequencing
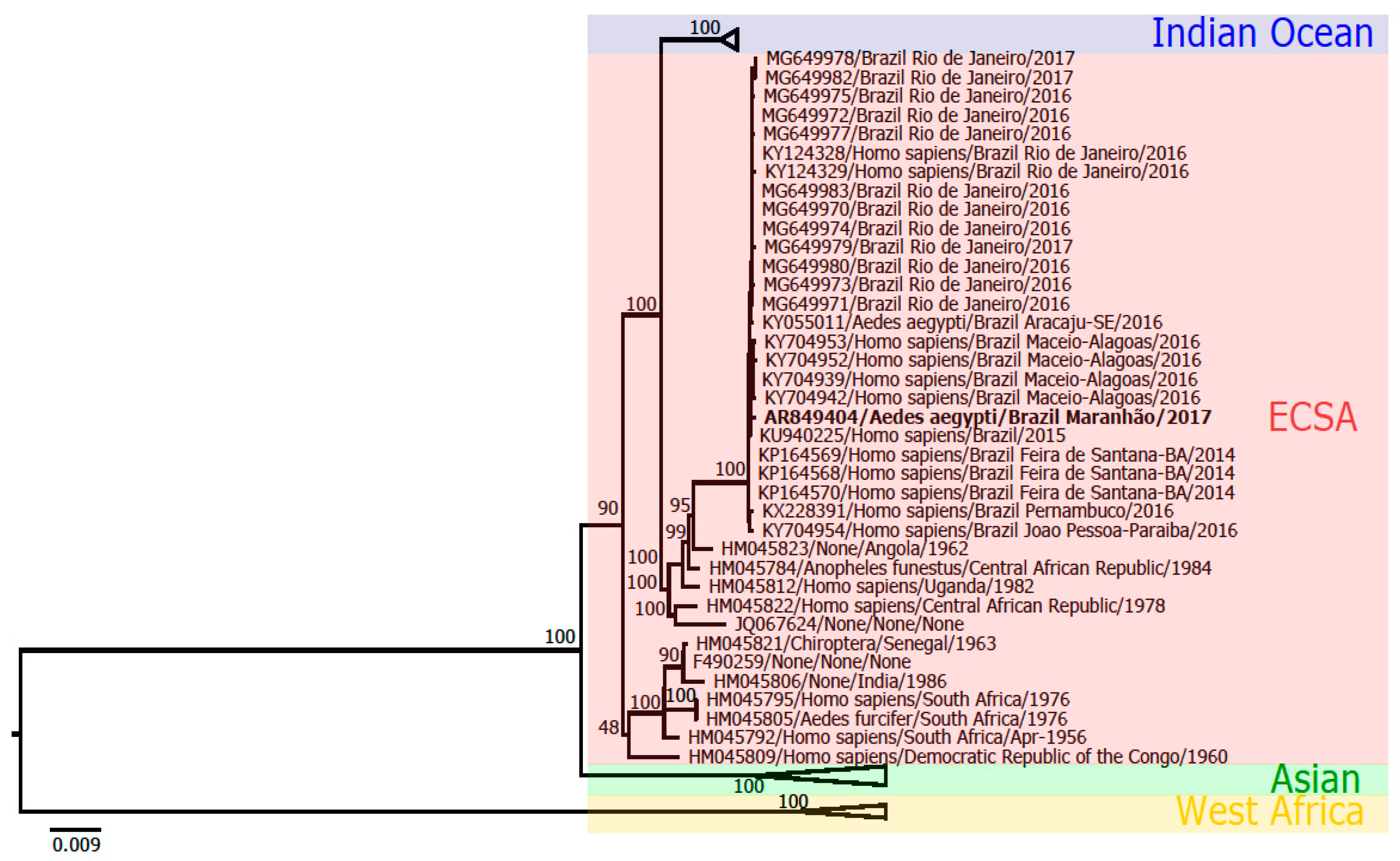
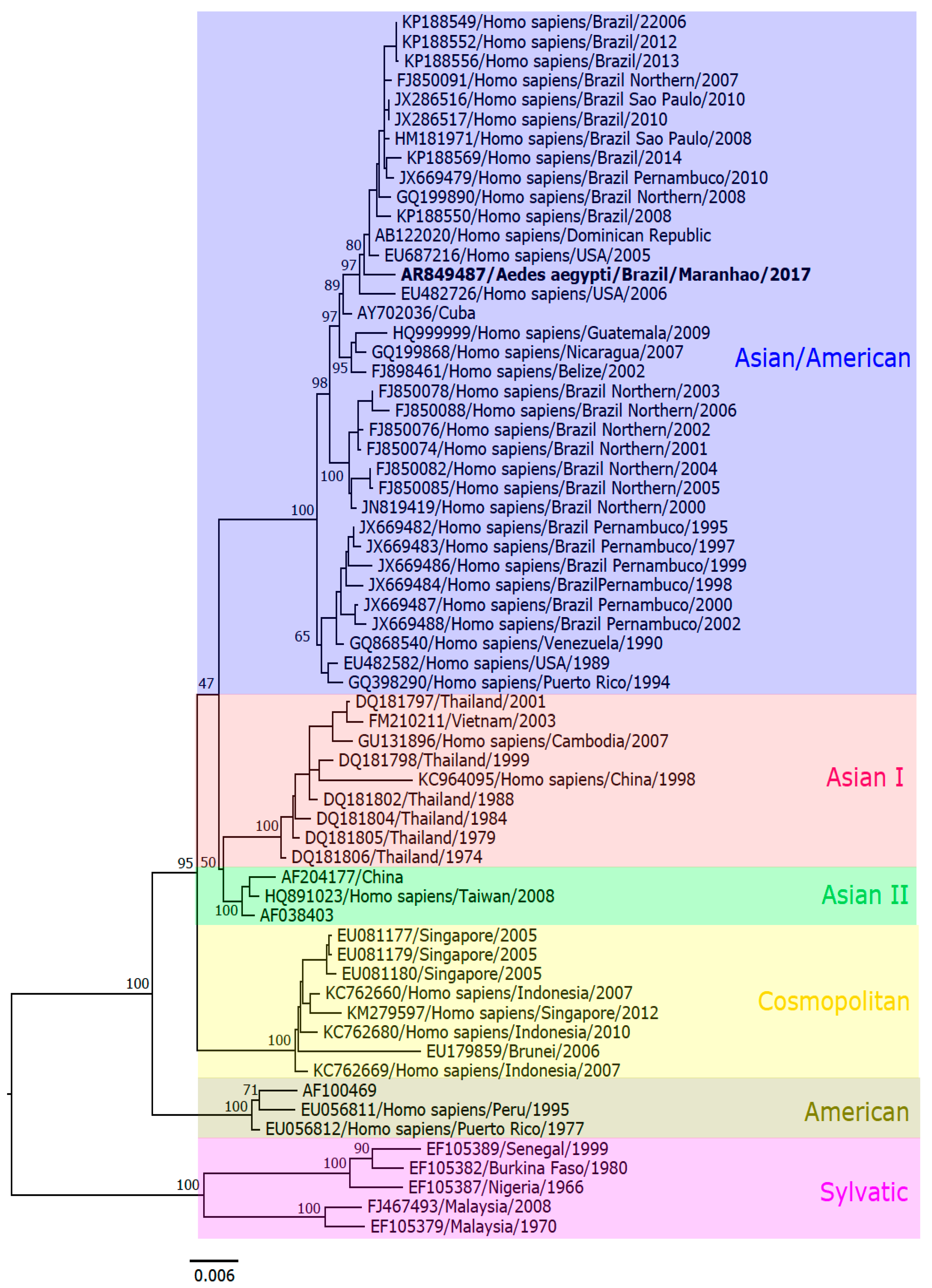
2.9.3. Genome Assembling and Phylogenetic Analysis
3. Results
3.1. Collection and Identification
3.2. RT-qPCR Assay
3.3. Viral Isolation
3.4. Nucleotide Sequencing
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Azevedo, R.; Rodrigues, S.G.; Martins, L.C.; Chiang, J.O.; Rosa, A.T.; Vasconcelos, P.F.d.C. Arboviroses. In Medicina Tropical e Infectologia na Amazônia; Leão, R.N.Q., Bichara, C.N.C., Fraiha Neto, H., Vasconcelos, P.F.d.C., Eds.; Samauma Editorial: Belém, PA, Brasil, 2013; pp. 481–503. [Google Scholar]
- Weaver, S.C. Urbanization and geographic expansion of zoonotic arboviral diseases: mechanisms and potential strategies for prevention. Trends Microbiol. 2013, 21, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, L.T.M. Large outbreaks of Chikungunya virus in Brazil reveal uncommon clinical features and fatalities. Rev. Soc. Bras. Med. Trop. 2017, 50, 583–584. [Google Scholar] [CrossRef] [PubMed]
- Casseb, A.d.R.; Casseb, L.M.N.; Silva, S.P.; Vasconcelos, P.F.d.C. Arbovírus: importante zoonose na Amazônia brasileira. Veterinária e Zootec. 2013, 20, 9–21. [Google Scholar]
- Lopes, N.; Nozawa, C.; Linhares, R.E.C. Características gerais e epidemiologia dos arbovírus emergentes no Brasil. Rev. Pan-Amazônica Saúde 2014, 5, 55–64. [Google Scholar] [CrossRef]
- Donalisio, M.R.; Freitas, A.R.R.; Zuben, A.P.B. Von Arboviroses emergentes no Brasil: desafios para a clínica e implicações para a saúde pública. Rev. Saude Publica 2017, 51, 1–7. [Google Scholar]
- Brasil. Ministério da Saúde. Secretaria de Vigilância e Saúde Monitoramento dos casos de arboviroses urbanas transmitidas pelo Aedes (dengue, chikungunya e Zika): Semanas Epidemiológicas 1 a 36. Available online: https://portalarquivos2.saude.gov.br/images/pdf/2019/outubro/04/BE-multitematico-n28.pdf (accessed on 20 September 2019).
- Villabona-Arenas, C.J.; Zanotto, P.M.A. Worldwide Spread of Dengue Virus Type 1. PLoS ONE 2013, 8, e62649. [Google Scholar] [CrossRef]
- Pyke, A.T.; Moore, P.R.; Taylor, C.T.; Hall-Mendelin, S.; Cameron, J.N.; Hewitson, G.R.; Pukallus, D.S.; Huang, B.; Warrilow, D.; Van Den Hurk, A.F. Highly divergent dengue virus type 1 genotype sets a new distance record. Sci. Rep. 2016, 6, 22356. [Google Scholar] [CrossRef]
- Wei, K.; Li, Y. Global evolutionary history and spatio-temporal dynamics of dengue virus type 2. Sci. Rep. 2017, 7, 45505. [Google Scholar] [CrossRef]
- Araújo, J.M.G.; Bello, G.; Schatzmayr, H.G.; Santos, F.B.; Nogueira, R.M.R. Dengue virus type 3 in Brazil: A phylogenetic perspective. Mem. Inst. Oswaldo Cruz 2009, 104, 526–529. [Google Scholar] [CrossRef]
- Gallichotte, E.N.; Baric, T.J.; Nivarthi, U.; Delacruz, M.J.; Graham, R.; Widman, D.G.; Yount, B.L.; Durbin, A.P.; Whitehead, S.S.; Silva, A.M.; et al. Genetic Variation between Dengue Virus Type 4 Strains Impacts Human Antibody Binding and Neutralization. Cell Rep. 2018, 25, 1214–1224. [Google Scholar] [CrossRef]
- Brasil. Ministério da Saúde. Secretaria de Vigilância e Saúde Monitoramento dos casos de dengue até a semana epidemiológica (SE) 38 e febre de chikungunya até a SE 39 de 2014. Available online: http://portalarquivos2.saude.gov.br/images/pdf/2014/outubro/03/BE-2014-45--23----Dengue--SE38--e-CHIKV--SE39-.pdf (accessed on 13 December 2017).
- Zanluca, C.; De Melo, V.C.A.; Mosimann, A.L.P.; Dos Santos, G.I.V.; dos Santos, C.N.D.; Luz, K. First report of autochthonous transmission of Zika virus in Brazil. Mem. Inst. Oswaldo Cruz 2015, 110, 569–572. [Google Scholar] [CrossRef] [PubMed]
- Nunes, M.R.T.; Faria, N.R.; Vasconcelos, J.M.; Golding, N.; Kraemer, M.U.; Oliveira, L.F.; Azevedo, R.d.S.d.S.; Silva, D.E.A.; Silva, E.V.P.; Silva, S.P.; et al. Emergence and potential for spread of Chikungunya virus in Brazil. BMC Med. 2015, 13, 102. [Google Scholar] [CrossRef] [PubMed]
- Bandeira, A.C.; Campos, G.S.; Sardi, S.I.; Rocha, V.F.D.; Rocha, G.C.M. Neonatal encephalitis due to Chikungunya vertical transmission: First report in Brazil. IDCases 2016, 5, 57–59. [Google Scholar] [CrossRef] [PubMed]
- Rosário, M.S.; Jesus, P.A.P.; Vasilakis, N.; Farias, D.S.; Novaes, M.A.C.; Rodrigues, S.G.; Martins, L.C.; Vasconcelos, P.F.d.C.; Ko, A.I.; Alcântara, L.C.J.; et al. Case report: Guillain-barré syndrome after zika virus infection in Brazil. Am. J. Trop. Med. Hyg. 2016, 95, 1157–1160. [Google Scholar]
- Silva, I.R.F.; Frontera, J.A.; Filippis, A.M.B.; Nascimento, O.J.M. Neurologic complications associated with the Zika virus in Brazilian adults. JAMA Neurol. 2017, 74, 1190–1198. [Google Scholar] [CrossRef]
- Styczynski, A.R.; Malta, J.M.A.S.; Krow-Lucal, E.R.; Percio, J.; Nóbrega, M.E.; Vargas, A.; Lanzieri, T.M.; Leite, P.L.; Staples, J.E.; Fischer, M.X.; et al. Increased rates of Guillain-Barré syndrome associated with Zika virus outbreak in the Salvador metropolitan area, Brazil. PLoS Negl. Trop. Dis. 2017, 11, e0005869. [Google Scholar] [CrossRef]
- Araújo, T.V.B.; Ximenes, R.A.d.A.; Miranda-Filho, D.d.B.; Souza, W.V.; Montarroyos, U.R.; Melo, A.P.L.; Valongueiro, S.; Albuquerque, M.d.F.P.M.; Braga, C.; Filho, S.P.B.; et al. Association between microcephaly, Zika virus infection, and other risk factors in Brazil: final report of a case-control study. Lancet. Infect. Dis. 2018, 18, 328–336. [Google Scholar]
- Mehta, R.; Soares, C.N.; Medialdea-Carrera, R.; Ellul, M.; Silva, M.T.T.; Rosala-Hallas, A.; Jardim, M.R.; Burnside, G.; Pamplona, L.; Bhojak, M.; et al. The spectrum of neurological disease associated with Zika and chikungunya viruses in adults in Rio de Janeiro, Brazil: A case series. PLoS Negl. Trop. Dis. 2018, 12, e0006212. [Google Scholar] [CrossRef]
- Vissoci, J.R.N.; Rocha, T.A.H.; Silva, N.C.d.; Queiroz, R.C.d.S.; Thomaz, E.B.A.F.; Amaral, P.V.M.; Lein, A.; Branco, M.d.R.F.C.; Aquino, J.; Rodrigues, Z.M.R.; et al. Zika virus infection and microcephaly: Evidence regarding geospatial associations. PLoS Negl. Trop. Dis. 2018, 12, e0006392. [Google Scholar] [CrossRef]
- Simonin, Y.; van Riel, D.; Van de Perre, P.; Rockx, B.; Salinas, S. Differential virulence between Asian and African lineages of Zika virus. PLoS Negl. Trop. Dis. 2017, 11, e0005821. [Google Scholar] [CrossRef]
- Pettersson, J.H.-O.; Eldholm, V.; Seligman, S.J.; Lundkvist, Å.; Falconar, A.K.; Gaunt, M.W.; Musso, D.; Nougairède, A.; Charrel, R.; Gould, E.A.; et al. How Did Zika Virus Emerge in the Pacific Islands and Latin America? MBio 2016, 7, e01239-16. [Google Scholar] [CrossRef]
- Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Monitoramento dos casos de dengue, febre de chikungunya e doença aguda pelo vírus Zika até a Semana Epidemiológica 52 de 2018. Available online: http://portalarquivos2.saude.gov.br/images/pdf/2019/janeiro/28/2019-002.pdf (accessed on 2 February 2019).
- Wahid, B.; Ali, A.; Rafique, S.; Idrees, M. Global expansion of chikungunya virus: mapping the 64-year history. Int. J. Infect. Dis. 2017, 58, 69–76. [Google Scholar] [CrossRef]
- Carvalho, F.D.; Moreira, L.A. Why is Aedes aegypti Linnaeus so Successful as a Species? Neotrop. Entomol. 2017, 46, 243–255. [Google Scholar] [CrossRef]
- Kotsakiozi, P.; Gloria-Soria, A.; Caccone, A.; Evans, B.; Schama, R.; Martins, A.J.; Powell, J.R. Tracking the return of Aedes aegypti to Brazil, the major vector of the dengue, chikungunya and Zika viruses. PLoS Negl. Trop. Dis. 2017, 11, e0005653. [Google Scholar] [CrossRef] [PubMed]
- Instituto Brasileiro de Geografia e Estatística. Síntese de Indicadores Sociais: Pobreza aumenta e atinge 54,8 milhões de pessoas em 2017. Available online: https://agenciadenoticias.ibge.gov.br/agencia-noticias/2012-agencia-de-noticias/noticias/23299-pobreza-aumenta-e-atinge-54-8-milhoes-de-pessoas-em-2017 (accessed on 2 February 2019).
- LIRAa aponta 357 municípios em situação de Risco para dengue, zika e chikungunya. Available online: http://www.planejamento.gov.br/aedes/liraa-aponta-357-municipios-em-situacao-de-risco-para-dengue-zika-e-chikungunya (accessed on 23 June 2019).
- Soares-da-Silva, J.; Ibiapina, S.S.; Bezerra, J.M.T.; Tadei, W.P.; Pinheiro, V.C.S. Variation in Aedes aegypti (Linnaeus) (Diptera, Culicidae) infestation in artificial containers in Caxias, state of Maranhão, Brazil. Rev. Soc. Bras. Med. Trop. 2012, 45, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Bezerra, J.M.T.; Miranda, J.P.; Nunes-Neto, J.P.; Cruz, A.C.R.; Tadei, W.P.; Pinheiro, V.C.S. Occurrence of Aedes aegypti (Diptera, Culicidae) in a Dengue Transmission Area at Coastal Maranhão State, Brazil. Open Trop. Med. J. 2013, 6, 5–10. [Google Scholar] [CrossRef]
- Bezerra, J.M.T.; Santana, I.N.S.; Miranda, J.P.; Tadei, W.P.; Pinheiro, V.C.S. Breeding Sites of Aedes aegypti (Linnaeus) (Diptera, Culicidae): study about the containers in dry and rainy seasons in dengue-endemic city. Rev. Pesqui. em Saúde 2017, 8, 102–107. [Google Scholar]
- Sousa, A.A.d.; Fraga, E.; Sampaio, I.; Schneider, H.; Barros, M.C. Genetic differentiation in populations of Aedes aegypti (Diptera, Culicidae) dengue vector from the Brazilian state of Maranhão. Rev. Bras. Entomol. 2017, 61, 51–59. [Google Scholar] [CrossRef][Green Version]
- Andrade, A.T.d.S.; Lobo, K.d.S.; Soares-da-Silva, J.; Pinheiro, V.C.S. Densidade populacional de Aedes aegypti (Linnaeus, 1762) e Aedes albopictus (Skuse, 1894) (Diptera: Culicidae) nos criadouros artificiais do município de Caxias, Maranhão. In Avanços das Ciências Biológicas no Centro de Estudos Superiores de Caxias (CESC); Fonseca, R.S., Barros, M.C., Eds.; UEMA: São Luís, MA, Brasil, 2018; pp. 135–152. ISBN 9788582271339. [Google Scholar]
- Aragão, C.F.; Cruz, A.C.R.; Nunes-Neto, J.P.; Monteiro, H.A.d.O.; Silva, E.V.P.; Silva, S.P.; Andrade, A.T.d.S.; Tadei, W.P.; Pinheiro, V.C.S. Circulation of Chikungunya virus in Aedes aegypti in Maranhão, Northeast Brazil. Acta Trop. 2018, 186, 1–4. [Google Scholar]
- Instituto Brasileiro de Geografia e Estatística. Maranhão. Available online: https://cidades.ibge.gov.br/brasil/ma/panorama (accessed on 2 February 2019).
- Nasci, R.S. A light weight battery powered aspirator for collecting resting mosquitoes in the field. Mosq. News 1981, 41, 808–811. [Google Scholar]
- Lane, J. Neotropical Culicidae; Edusp: São Paulo, SP, Brasil, 1953; Volume 1. [Google Scholar]
- Lane, J. Neotropical Culicidae; Edusp: São Paulo, SP, Brasil, 1953; Volume 2. [Google Scholar]
- Forattini, O.P. Entomologia Médica. Parte geral, Diptera, Anophelini; Faculdade de Higiene e Saúde Pública: São Paulo, SP, Brasil, 1962; Volume 1. [Google Scholar]
- Forattini, O.P. Entomologia médica. Culicini: Culex, Aedes e Psorophora; Editora da Universidade de São Paulo: São Paulo, SP, Brasil, 1965; Volume 2. [Google Scholar]
- Forattini, O.P. Entomologia médica. Culicini: Haemagogus, Mansonia, Culiseta, Sabethini, Toxorhynchitini, Arboviroses, Filariose bancroftiana, Genética; Editora da Universidade de São Paulo: São Paulo, SP, Brasil, 1965; Volume 3. [Google Scholar]
- Forattini, O.P. Culicidologia Médica; Universidade de São Paulo: São Paulo, Brazil, 2002; Volume 2, ISBN 85-314-0699-4. [Google Scholar]
- Consoli, R.A.G.B.; Lourenço-de-Oliveira, R. Principais mosquitos de importância sanitária no Brasil; Editora Fiocruz: Rio de Janeiro, RJ, Brazil, 1994; ISBN 85-85676-03-5. [Google Scholar]
- Wirth, W.W.; Blanton, F.S. A review of the maruins or biting midges of the genus Culicoides (Diptera: Ceratopogonidae) in the Amazon Basin. Amazoniana 1973, 4, 405–470. [Google Scholar]
- Tesh, R.B. A method for the isolation and identification of dengue viruses, using mosquito cell cultures. Am. J. Trop. Med. Hyg. 1979, 28, 1053–1059. [Google Scholar] [CrossRef] [PubMed]
- Menting, S.; Thai, K.T.D.; Nga, T.T.T.; Phuong, H.L.; Klatser, P.; Wolthers, K.C.; Binh, T.Q.; de Vries, P.J.; Beld, M. Internally Controlled, Generic Real-Time PCR for Quantification and Multiplex Real-Time PCR with Serotype-Specific Probes for Serotyping of Dengue Virus Infections. Adv. Virol. 2011, 2011, 1–9. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Santiago, G.A.; Vergne, E.; Quiles, Y.; Cosme, J.; Vazquez, J.; Medina, J.F.; Medina, F.; Colón, C.; Margolis, H.; Muñoz-Jordán, J.L. Analytical and Clinical Performance of the CDC Real Time RT-PCR Assay for Detection and Typing of Dengue Virus. PLoS Negl. Trop. Dis. 2013, 7, e2311. [Google Scholar] [CrossRef]
- Faye, O.; Faye, O.; Diallo, D.; Diallo, M.; Weidmann, M.; Sall, A.A. Quantitative real-time PCR detection of Zika virus and evaluation with field-caught Mosquitoes. Virol. J. 2013, 10, 1. [Google Scholar] [CrossRef]
- Lanciotti, R.S.; Kosoy, O.L.; Laven, J.J.; Panella, A.J.; Velez, J.O.; Lambert, A.J.; Campbell, G.L. Chikungunya virus in US travelers returning from India, 2006. Emerg. Infect. Dis. 2007, 13, 764–767. [Google Scholar] [CrossRef]
- Beaty, B.J.; Calisher, C.H.; Shope, R.E. Arboviruses. In Diagnostic Procedures for Viral Rickettsial and Chlamydial Infections; Lennette, E.H., Lunette, D.A., Lenette, E.T., Eds.; American Public Health Association: Washington, DC, USA, 1995; pp. 189–212. [Google Scholar]
- Gubler, D.J.; Kuno, G.; Sather, G.E.; Velez, M.; Oliver, A. Mosquito Cell Cultures and Specific Monoclonal Antibodies in Surveillance for Dengue Viruses. Am. J. Trop. Med. Hyg. 1984, 33, 158–165. [Google Scholar] [CrossRef]
- Peng, Y.; Leung, H.C.M.; Yiu, S.M.; Chin, F.Y.L. IDBA-UD: a de novo assembler for single-cell and metagenomic sequencing data with highly uneven depth. Bioinformatics 2012, 28, 1420–1428. [Google Scholar] [CrossRef]
- Buchfink, B.; Xie, C.; Huson, D.H. Fast and sensitive protein alignment using DIAMOND. Nat. Methods 2015, 12, 59–60. [Google Scholar] [CrossRef]
- Huson, D.H.; Auch, A.F.; Qi, J.; Schuster, S.C. MEGAN analysis of metagenomic data. Genome Res. 2007, 17, 377–386. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT Multiple Sequence Alignment Software Version 7: Improvements in Performance and Usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [PubMed]
- Abascal, F.; Zardoya, R.; Posada, D. ProtTest: selection of best-fit models of protein evolution. Bioinformatics 2005, 21, 2104–2105. [Google Scholar] [CrossRef] [PubMed]
- Myung, I.J. Tutorial on maximum likelihood estimation. J. Math. Psychol. 2003, 47, 90–100. [Google Scholar] [CrossRef]
- Stamatakis, A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef]
- Felsenstein, J. Confidence limits on phylogenies: an approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef]
- Souza, S.S.; Silva, I.G.; Silva, H.H.G. Associação entre incidência de dengue, pluviosidade e densidade larvária de Aedes aegypti, no Estado de Goiás. Rev. Soc. Bras. Med. Trop. 2010, 43, 152–155. [Google Scholar] [CrossRef][Green Version]
- Ferreira, A.C.; Chiaravalloti-Neto, F.; Mondini, A. Dengue in Araraquara, state of São Paulo: epidemiology, climate and Aedes aegypti infestation. Rev. Saude Publica 2018, 52, 18. [Google Scholar] [CrossRef]
- Soares, V.A.R.C.; Rodrigues, W.C.; Cabral, M.M.d.O. Estudo de áreas e depósitos preferenciais de Aedes albopictus (Skuse, 1894) e Aedes aegypti (Linnaeus, 1762) no Município de Paracambi – Rio de Janeiro, Brasil. EntomoBrasilis 2008, 1, 63–68. [Google Scholar] [CrossRef][Green Version]
- Ayllón, T.; Câmara, D.C.P.; Morone, F.C.; Silva Gonçalves, L.; Barros, F.S.M.; Brasil, P.; Carvalho, M.S.; Honório, N.A. Dispersion and oviposition of Aedes albopictus in a Brazilian slum: Initial evidence of Asian tiger mosquito domiciliation in urban environments. PLoS ONE 2018, 13, e0195014. [Google Scholar] [CrossRef]
- Figueiredo, M.L.G.; Gomes, A.d.C.; Amarilla, A.A.; Leandro, A.d.S.; Orrico, A.d.S.; Araujo, R.F.d.; Castro, J.d.S.M.; Durigon, E.L.; Aquino, V.H.; Figueiredo, L.T.M. Mosquitoes infected with dengue viruses in Brazil. Virol. J. 2010, 7, 152. [Google Scholar] [CrossRef]
- Martins, V.E.P.; Alencar, C.H.; Kamimura, M.T.; Araújo, F.M.d.C.; Simone, S.G.; Dutra, R.F.; Guedes, M.I.F. Occurrence of natural vertical transmission of dengue-2 and dengue-3 viruses in Aedes aegypti and Aedes albopictus in Fortaleza, Ceará, Brazil. PLoS ONE 2012, 7, 1–9. [Google Scholar] [CrossRef]
- Smartt, C.T.; Stenn, T.M.S.; Chen, T.; Teixeira, M.G.; Queiroz, E.P.; Santos, L.S.; Queiroz, G.A.N.; Souza, K.R.; Silva, L.K.; Shin, D.; et al. Evidence of Zika Virus RNA Fragments in Aedes albopictus (Diptera: Culicidae) Field-Collected Eggs From Camaçari, Bahia, Brazil. J. Med. Entomol. 2017, 54, 1085–1087. [Google Scholar] [CrossRef] [PubMed]
- Castro, M.G.; Nogueira, R.M.R.; Schatzmayr, H.G.; Miagostovich, M.P.; Lourenço-de-Oliveira, R. Dengue Virus Detection by Using Reverse Transcription- Polymerase Chain Reaction in Saliva and Progeny of Experimentally Infected Aedes albopictus from Brazil. Mem. Inst. Oswaldo Cruz 2004, 99, 809–814. [Google Scholar] [CrossRef] [PubMed]
- Vega-Rua, A.; Zouache, K.; Girod, R.; Failloux, A.-B.; Lourenco-de-Oliveira, R. High Level of Vector Competence of Aedes aegypti and Aedes albopictus from Ten American Countries as a Crucial Factor in the Spread of Chikungunya Virus. J. Virol. 2014, 88, 6294–6306. [Google Scholar] [CrossRef] [PubMed]
- Honório, N.A.; Wiggins, K.; Câmara, D.C.P.; Eastmond, B.; Alto, B.W. Chikungunya virus vector competency of Brazilian and Florida mosquito vectors. PLoS Negl. Trop. Dis. 2018, 12, e0006521. [Google Scholar] [CrossRef]
- Effler, P.V.; Pang, L.; Kitsutani, P.; Vorndam, V.; Nakata, M.; Ayers, T.; Elm, J.; Tom, T.; Reiter, P.; Ringau-Perez, J.G.; et al. Dengue Fever, Hawaii, 2001-2002. Emerg. Infect. Dis. 2005, 11, 742–749. [Google Scholar] [CrossRef]
- Almeida, A.P.G.; Baptista, S.S.S.G.; Sousa, C.A.G.C.C.; Novo, M.T.L.M.; Ramos, H.C.; Panella, N.A.; Godsey, M.; Simões, M.J.; Anselmo, M.L.; Komar, N.; et al. Bioecology and Vectorial Capacity of Aedes albopictus (Diptera: Culicidae) in Macao, China, in Relation to Dengue Virus Transmission. J. Med. Entomol. 2005, 42, 419–428. [Google Scholar] [CrossRef]
- Xu, G.; Dong, H.; Shi, N.; Liu, S.; Zhou, A.; Cheng, Z.; Chen, G.; Liu, J.; Fang, T.; Zhang, H.; et al. An outbreak os Dengue virus serotype 1 infection in Cixi, Ningbo, people’s republic of China, 2004, associated with atraveler from Thailand and high density of Aedes albopictus. Am. J. Trop. Med. Hyg. 2007, 76, 1182–1188. [Google Scholar] [CrossRef]
- Delatte, H.; Paupy, C.; Dehecq, J.S.; Thiria, J.; Failloux, A.B.; Fontenille, D. Aedes albopictus, vecteur des virus du chikungunya et de la dengue à la Réunion: biologie et contrôle. Parasite 2008, 15, 3–13. [Google Scholar] [CrossRef]
- Leroy, E.M.; Nkoghe, D.; Ollomo, B.; Nze-nkogue, C.; Becquart, P.; Grard, G.; Pourrut, X.; Charrel, R.; Moureau, G.; Ndjoyi-Mbguino, A.; et al. Concurrent chikungunya and dengue virus infections during simultaneous outbreaks, Gabon, 2007. Emerg. Infect. Dis. 2009, 15, 591–593. [Google Scholar] [CrossRef]
- Issack, M.I.; Pursem, V.N.; Barkham, T.M.S.; Ng, L.; Inoue, M.; Manraj, S.S. Reemergence of Dengue in Mauritius. Emerg. Infect. Dis. 2010, 16, 716–718. [Google Scholar] [CrossRef] [PubMed]
- Grard, G.; Caron, M.; Mombo, I.M.; Nkoghe, D.; Ondo, S.M.; Jiolle, D.; Fontenille, D.; Paupy, C.; Leroy, E.M. Zika Virus in Gabon (Central Africa) - 2007: A New Threat from Aedes albopictus? PLoS Negl. Trop. Dis. 2014, 8, e2681. [Google Scholar] [CrossRef] [PubMed]
- Reiter, P.; Fontenille, D.; Paupy, C. Aedes albopictus as an epidemic vector of chikungunya virus: another emerging problem? Lancet Infect. Dis. 2006, 6, 463–464. [Google Scholar] [CrossRef]
- Ratsitorahina, M.; Harisoa, J.; Ratovonjato, J.; Biacabe, S.; Reynes, J.-M.; Zeller, H.; Raoelina, Y.; Talarmin, A.; Richard, V.; Soares, J.L. Outbreak of Dengue and Chikungunya Fevers, Toamasina, Madagascar, 2006. Emerg. Infect. Dis. 2008, 14, 1135–1137. [Google Scholar] [CrossRef] [PubMed]
- Pagès, F.; Peyrefitte, C.N.; Mve, M.T.; Jarjaval, F.; Brisse, S.; Iteman, I.; Gravier, P.; Nkoghe, D.; Grandadam, M. Aedes albopictus Mosquito: The Main Vector of the 2007 Chikungunya Outbreak in Gabon. PLoS Negl. Trop. Dis. 2009, 4, e4691. [Google Scholar]
- Li, Y.; Kamara, F.; Zhou, G.; Puthiyakunnon, S.; Li, C.; Liu, Y.; Zhou, Y.; Yao, L.; Yan, G.; Chen, X. Urbanization Increases Aedes albopictus Larval Habitats and Accelerates Mosquito Development and Survivorship. PLoS Negl. Trop. Dis. 2014, 8, e3301. [Google Scholar] [CrossRef] [PubMed]
- Ramasamy, R.; Surendran, S.N. Mosquito vectors developing in atypical anthropogenic habitats: Global overview of recent observations, mechanisms and impact on disease transmission. J. Vector Borne Dis. 2016, 53, 91–98. [Google Scholar]
- SINAN: Sistema de Informação de Agravos de Notificação. Available online: http://sinan.saude.gov.br/sinan/login/login.jsf (accessed on 8 May 2019).
- Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Monitoramento dos casos de dengue, febre de chikungunya e febre pelo vírus Zika até a Semana Epidemiológica 7. 2016. Available online: http://portalarquivos2.saude.gov.br/images/pdf/2016/marco/23/2016-008---DengueSE8-publica----o.pdf (accessed on 13 December 2017).
- Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Monitoramento dos casos de dengue, febre de chikungunya e febre pelo vírus Zika até a Semana Epidemiológica 52. 2017. Available online: http://portalarquivos2.saude.gov.br/images/pdf/2018/janeiro/23/Boletim-2018-001-Dengue.pdf (accessed on 15 February 2019).
- Costa-da-Silva, A.L.; Ioshino, R.S.; Petersen, V.; Lima, A.F.; Cunha, M.d.P.; Wiley, M.R.; Ladner, J.T.; Prieto, K.; Palacios, G.; Costa, D.D.; et al. First report of naturally infected Aedes aegypti with chikungunya virus genotype ECSA in the Americas. PLoS Negl. Trop. Dis. 2017, 11, e0005630. [Google Scholar] [CrossRef]
- Costa, A.C.d.; Thézé, J.; Komninakis, S.C.V.; Sanz-Duro, R.L.; Felinto, M.R.L.; Moura, L.C.C.; Barroso, I.M.d.O.; Santos, L.E.C.; Nunes, M.A.d.L.; Moura, A.A.; et al. Spread of Chikungunya Virus East/Central/South African Genotype in Northeast Brazil. Emerg. Infect. Dis. 2017, 23, 1742–1744. [Google Scholar] [CrossRef]
- Tanabe, E.L.d.L.; Tanabe, I.S.B.; Santos, E.C.d.; Marques, J.P.d.S.; Borges, A.A.; Lima, M.C.d.; Anderson, L.; Bassi, Ê.J. Report of East-Central South African Chikungunya virus genotype during the 2016 outbreak in the Alagoas State, Brazil. Rev. Inst. Med. Trop. Sao Paulo 2018, 60, e19. [Google Scholar] [CrossRef]
- Tauro, L.B.; Cardoso, C.W.; Souza, R.L.; Nascimento, L.C.J.; Santos, D.R.; Campos, G.S.; Sardi, S.; Reis, O.B.; Reis, M.G.; Kitron, U.; et al. A localized outbreak of Chikungunya virus in Salvador, Bahia, Brazil. Mem. Inst. Oswaldo Cruz 2019, 114, e180597. [Google Scholar] [CrossRef] [PubMed]
- Conteville, L.C.; Zanella, L.; Marín, M.A.; de Filippis, A.M.B.; Nogueira, R.M.R.; Vicente, A.C.P.; de Mendonça, M.C.L. Phylogenetic analyses of chikungunya virus among travelers in Rio De Janeiro, Brazil, 2014-2015. Mem. Inst. Oswaldo Cruz 2016, 111, 347–348. [Google Scholar] [CrossRef] [PubMed]
- Cunha, M.S.; Cruz, N.V.G.; Schnellrath, L.C.; Medaglia, M.L.G.; Casotto, M.E.; Albano, R.M.; Costa, L.J.; Damaso, C.R. Autochthonous Transmission of East/Central/South African Genotype Chikungunya Virus, Brazil. Emerg. Infect. Dis. 2017, 23, 1737–1739. [Google Scholar] [CrossRef] [PubMed]
- Naveca, F.G.; Claro, I.; Giovanetti, M.; Jesus, J.G.; Xavier, J.; Iani, F.C.d.M.; Nascimento, V.A.; Souza, V.C.; Silveira, P.P.; Lourenço, J.; et al. Genomic, epidemiological and digital surveillance of Chikungunya virus in the Brazilian Amazon. PLoS Negl. Trop. Dis. 2019, 13, e0007065. [Google Scholar] [CrossRef] [PubMed]
- Souza, T.M.A.; Azeredo, E.L.; Badolato-Corrêa, J.; Damasco, P.V.; Santos, C.; Petitinga-Paiva, F.; Nunes, P.C.G.; Barbosa, L.S.; Cipitelli, M.C.; Chouin-Carneiro, T.; et al. First Report of the East-Central South African Genotype of Chikungunya Virus in Rio de Janeiro, Brazil. PLOS Curr. 2017, 9. [Google Scholar] [CrossRef]
- Lessa-Aquino, C.; Trinta, K.S.; Pestana, C.P.; Ribeiro, M.O.; Sucupira, M.V.F.; Boia, M.N.; Baptista, P.A.; Cunha, R.V.; Medeiros, M.A. Detection of East/Central/South African genotype Chikungunya virus during an outbreak in a southeastern state of Brazil. Epidemiol. Infect. 2018, 146, 2056–2058. [Google Scholar] [CrossRef]
- Machado, L.C.; de Morais-Sobral, M.C.; Campos, T.d.L.; Pereira, M.R.; de Albuquerque, M.d.F.P.M.; Gilbert, C.; Franca, R.F.O.; Wallau, G.L. Genome sequencing reveals coinfection by multiple chikungunya virus genotypes in a recent outbreak in Brazil. PLoS Negl. Trop. Dis. 2019, 13, e0007332. [Google Scholar] [CrossRef]
- Brasil. Ministério da Saúde. Secretaria de Vigilância em Saúde. Monitoramento dos casos de dengue, febre de chikungunya e febre pelo vírus Zika até a Semana Epidemiológica 5 de 2018. Available online: http://portalarquivos2.saude.gov.br/images/pdf/2018/fevereiro/20/2018-007.pdf (accessed on 15 February 2019).
- Gonçalves-Neto, V.S.; Rebêlo, J.M.M. Aspectos epidemiológicos do dengue no Município de São Luís, Maranhão, Brasil, 1997-2002. Caderno 2004, 20, 1424–1431. [Google Scholar] [CrossRef]
- Faria, N.R.d.C.; Nogueira, R.M.R.; Filippis, A.M.B.d.; Simões, J.B.S.; Nogueira, F.d.B.; Lima, M.d.R.Q.; Santos, F.B.d. Twenty Years of DENV-2 Activity in Brazil: Molecular Characterization and Phylogeny of Strains Isolated from 1990 to 2010. PLoS Negl. Trop. Dis. 2013, 7, e2095. [Google Scholar] [CrossRef]
- Dias-Júnior, J.d.J.; Branco, M.d.R.F.C.; Queiroz, R.C.d.S.; Santos, A.M.; Moreira, E.P.B.; Silva, M.d.S. Analysis of dengue cases according to clinical severity, São Luís, Maranhão, Brazil. Rev. Inst. Med. Trop. Sao Paulo 2017, 59, e71. [Google Scholar]
- Cruz, A.C.R.; Galler, R.; Silva, E.V.P.; Silva, M.d.O.; Carneiro, A.R.; Rosa, E.S.T.d.; Vasconcelos, H.B.; Sá, E.L.R.d.; Vasconcelos, P.F.d.C. Molecular epidemiology of dengue virus serotypes 2 and 3 isolated in Brazil from 1991 to 2008. Rev. Pan-Amazônica Saúde 2010, 1, 25–34. [Google Scholar] [CrossRef]
- Barcelos-Figueiredo, L.; Sakamoto, T.; Coelho, L.F.L.; Rocha, E.S.d.O.; Cota, M.M.G.; Ferreira, G.P.; Oliveira, J.G.; Kroon, E.G. Dengue Virus 2 American-Asian Genotype Identified during the 2006/2007 Outbreak in Piauí Caribbean Route of Introduction and Dissemination of Dengue Virus in Brazil. PLoS ONE 2014, 9, e104516. [Google Scholar] [CrossRef]
- Oliveira, M.F.; Araújo, J.M.G.; Júnior, O.C.F.; Ferreira, D.F.; Lima, D.B.m.; Santos, F.B.; Gonçalves, H.; Schatzmayr, A.T.; Nogueira, R.M.R. Two Lineages of Dengue Virus Type 2, Brazil. Emerg. Infect. Dis. 2010, 16, 576–578. [Google Scholar] [CrossRef] [PubMed]
- Romano, C.M.; Matos, A.M.; Araújo, E.S.A.; Villas-Boas, L.S.; Silva, W.C.; Oliveira, O.M.N.P.F.; Carvalho, K.I.; Souza, A.C.M.; Rodrigues, C.L.; Levi, J.E.; et al. Characterization of Dengue Virus Type 2: New Insights on the 2010 Brazilian Epidemic. PLoS ONE 2010, 5, e11811. [Google Scholar] [CrossRef] [PubMed]
- Drumond, B.P.; Mondini, A.; Schmidt, D.J.; Bronzoni, R.V.d.M.; Bosch, I.; Nogueira, M.L. Circulation of Different Lineages of Dengue Virus 2, Genotype American/Asian in Brazil: Dynamics and Molecular and Phylogenetic Characterization. PLoS ONE 2013, 8, e59422. [Google Scholar] [CrossRef] [PubMed]
| Specification | Municipalities | Total | ||||
|---|---|---|---|---|---|---|
| Alto Alegre do Maranhão | Caxias | Codó | São Mateus do Maranhão | N | % | |
| Aedes spp. | 6 | 0 | 1 | 0 | 7 | 0.22 |
| Aedes aegypti | 49 | 55 | 213 | 31 | 348 | 10.99 |
| Aedes albopictus | 0 | 1 | 11 | 0 | 12 | 0.38 |
| Aedes scapularis | 3 | 2 | 1 | 0 | 6 | 0.19 |
| Aedes taeniorhynchus | 0 | 0 | 2 | 0 | 2 | 0.06 |
| Anopheles (Nyssorhynchus) sp. | 0 | 0 | 1 | 0 | 1 | 0.03 |
| Ceratopogonidae | 1 | 0 | 0 | 0 | 1 | 0.03 |
| Culex (Culex) spp. | 3 | 0 | 0 | 0 | 3 | 0.09 |
| Culex spp. | 93 | 0 | 0 | 0 | 93 | 2.94 |
| Culex (Melanoconion) spp. | 2 | 0 | 2 | 0 | 4 | 0.13 |
| Culex quinquefasciatus | 713 | 238 | 1063 | 630 | 2644 | 83.51 |
| Culex spissipes | 0 | 13 | 0 | 0 | 13 | 0.41 |
| Culicoides paraensis group | 1 | 0 | 0 | 0 | 1 | 0.03 |
| Mansonia (Mansonia) spp. | 9 | 0 | 1 | 1 | 11 | 0.35 |
| Mansonia titillans | 2 | 0 | 0 | 0 | 2 | 0.06 |
| Phlebotominae | 11 | 2 | 2 | 1 | 16 | 0.51 |
| Uranotaenia hystera | 1 | 0 | 0 | 0 | 1 | 0.03 |
| Uranotaenia lowii | 0 | 0 | 1 | 0 | 1 | 0.03 |
| Total | 894 | 311 | 1298 | 663 | 3166 | 100.00 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aragão, C.F.; Pinheiro, V.C.S.; Nunes Neto, J.P.; Silva, E.V.P.d.; Pereira, G.J.G.; Nascimento, B.L.S.d.; Castro, K.d.S.; Maia, A.M.; Catete, C.P.; Martins, L.C.; et al. Natural Infection of Aedes aegypti by Chikungunya and Dengue type 2 Virus in a Transition Area of North-Northeast Brazil. Viruses 2019, 11, 1126. https://doi.org/10.3390/v11121126
Aragão CF, Pinheiro VCS, Nunes Neto JP, Silva EVPd, Pereira GJG, Nascimento BLSd, Castro KdS, Maia AM, Catete CP, Martins LC, et al. Natural Infection of Aedes aegypti by Chikungunya and Dengue type 2 Virus in a Transition Area of North-Northeast Brazil. Viruses. 2019; 11(12):1126. https://doi.org/10.3390/v11121126
Chicago/Turabian StyleAragão, Carine Fortes, Valéria Cristina Soares Pinheiro, Joaquim Pinto Nunes Neto, Eliana Vieira Pinto da Silva, Glennda Juscely Galvão Pereira, Bruna Laís Sena do Nascimento, Karoline da Silva Castro, Ariadne Mendonça Maia, Clistenes Pamplona Catete, Lívia Carício Martins, and et al. 2019. "Natural Infection of Aedes aegypti by Chikungunya and Dengue type 2 Virus in a Transition Area of North-Northeast Brazil" Viruses 11, no. 12: 1126. https://doi.org/10.3390/v11121126
APA StyleAragão, C. F., Pinheiro, V. C. S., Nunes Neto, J. P., Silva, E. V. P. d., Pereira, G. J. G., Nascimento, B. L. S. d., Castro, K. d. S., Maia, A. M., Catete, C. P., Martins, L. C., Tadei, W. P., Silva, S. P. d., & Cruz, A. C. R. (2019). Natural Infection of Aedes aegypti by Chikungunya and Dengue type 2 Virus in a Transition Area of North-Northeast Brazil. Viruses, 11(12), 1126. https://doi.org/10.3390/v11121126








