Interferon γ and α Have Differential Effects on SAMHD1, a Potent Antiviral Protein, in Feline Lymphocytes
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. RNA Extraction and Real-Time Quantitative PCR
2.3. Flow Cytometry
2.4. Western Blotting
2.5. Statistical Analysis
3. Results
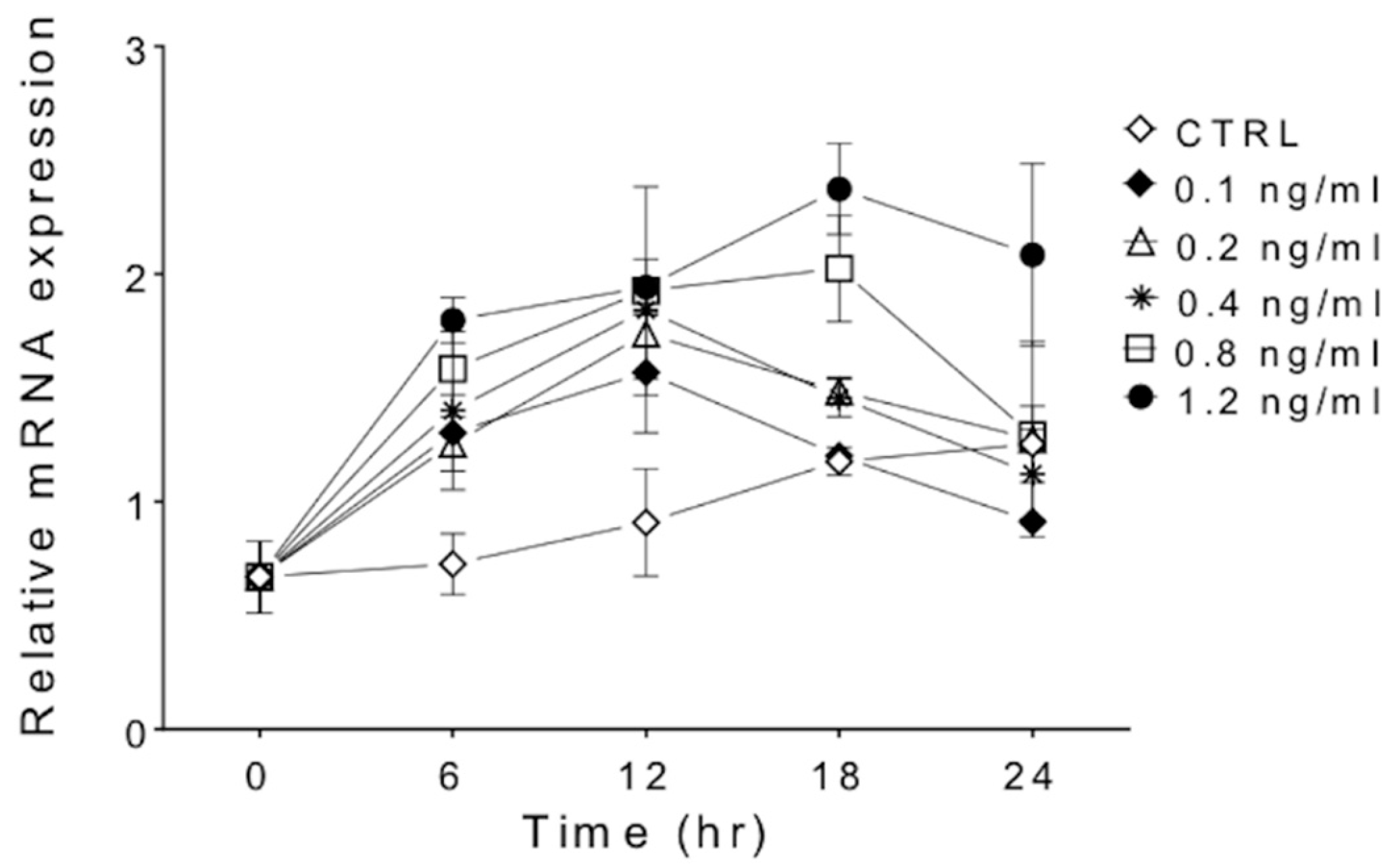
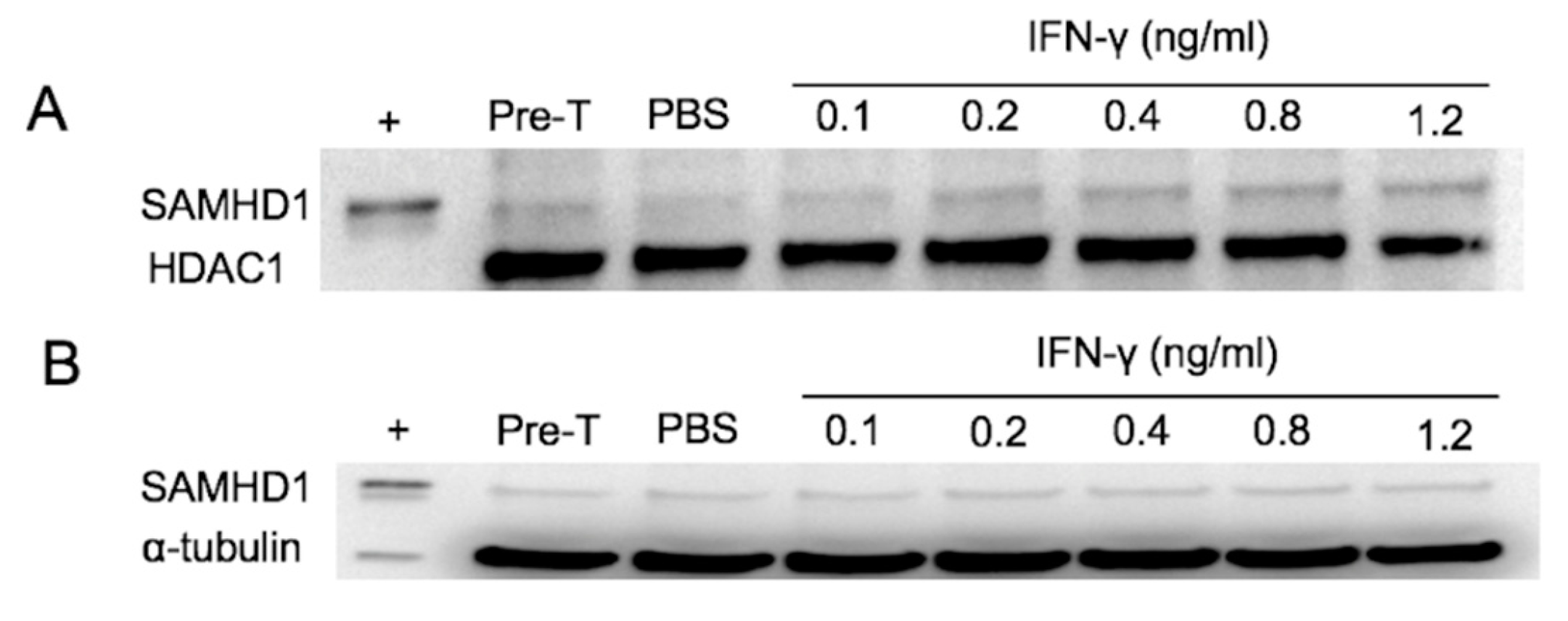
3.1. IFNγ Treatment Increases SAMHD1 mRNA
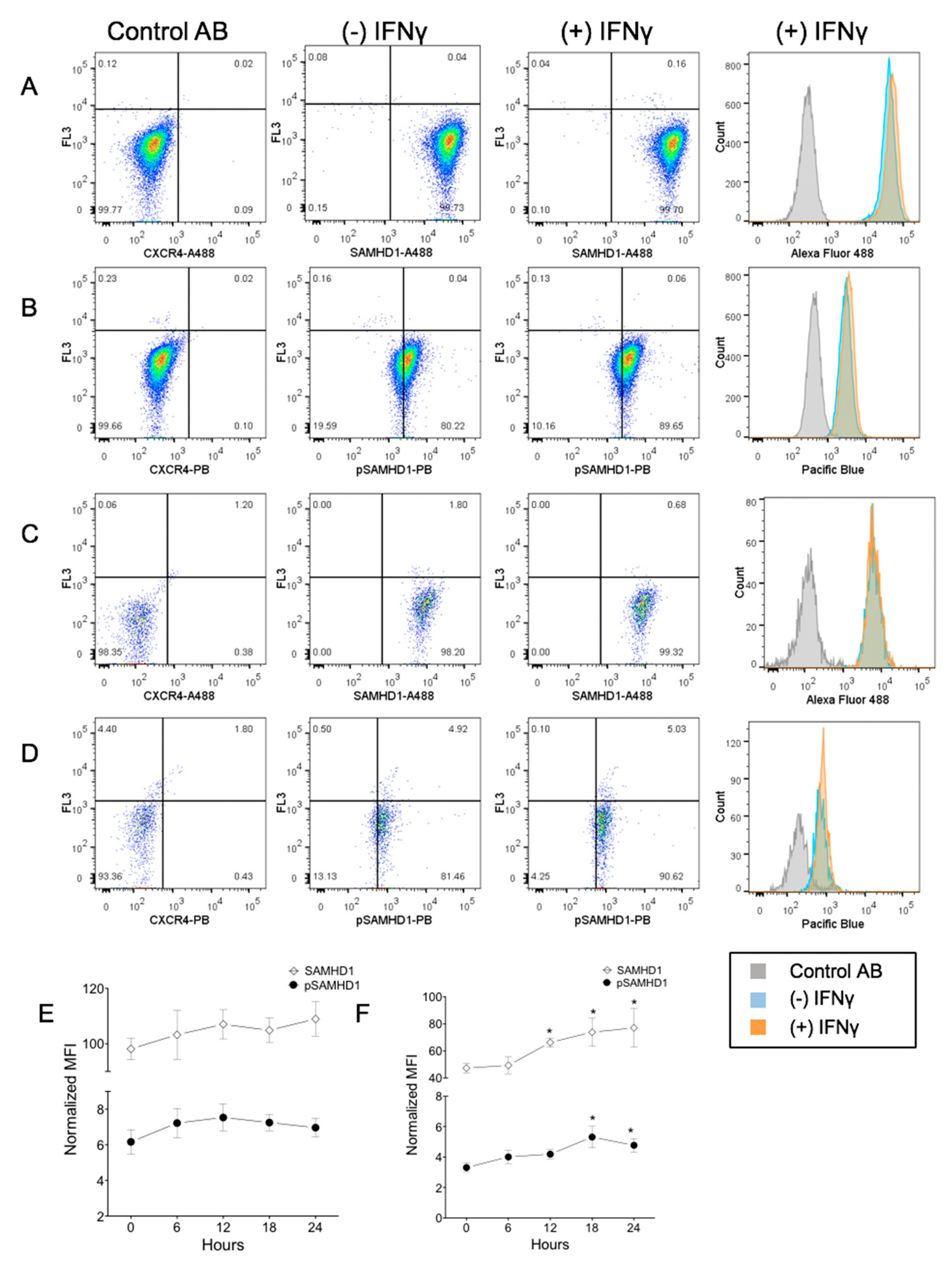
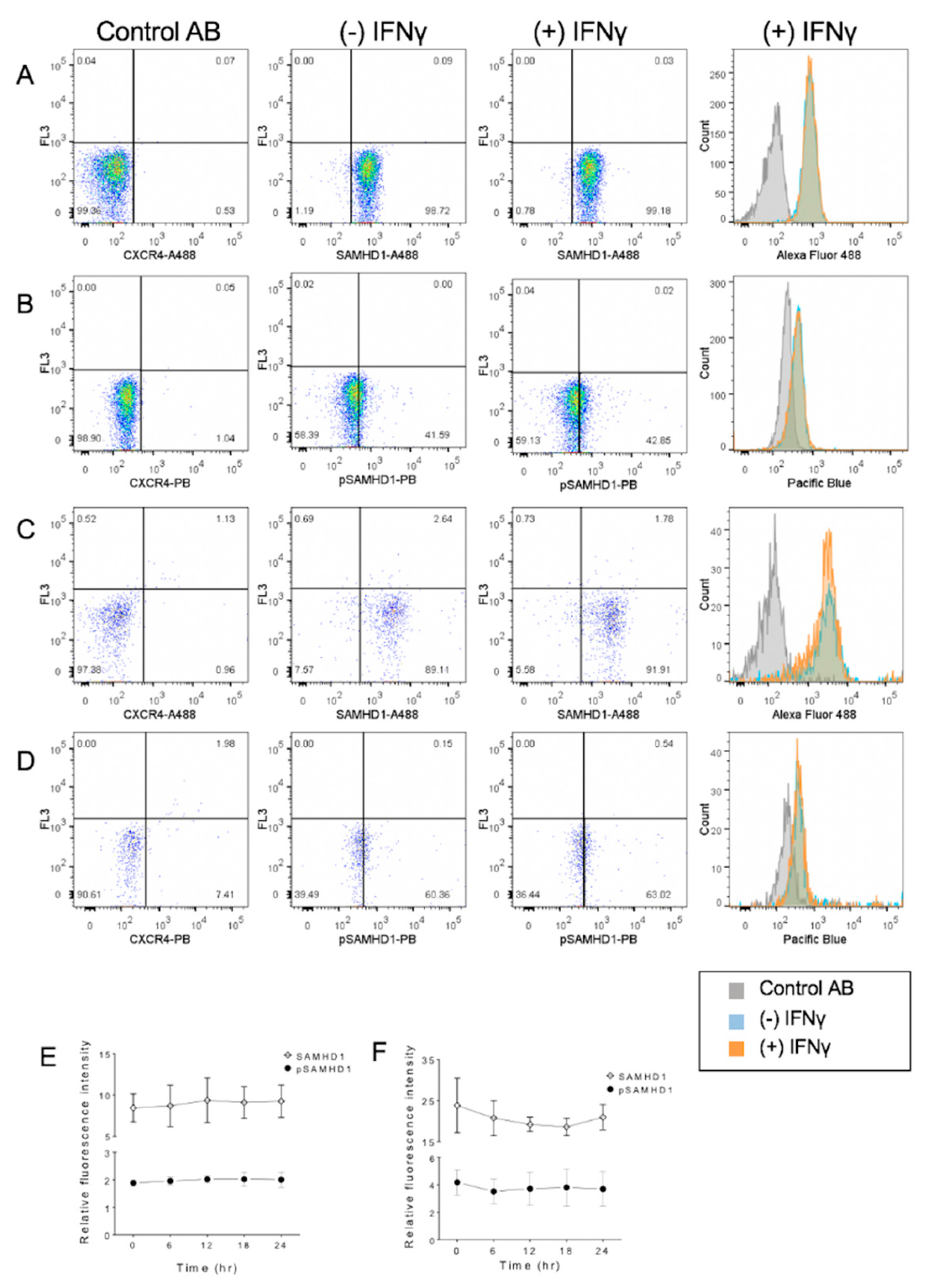
3.2. IFNγ Increases SAMHD1 Nuclear Translocation in FeTJ Cells
3.3. IFNγ Treatment Increases Nuclear pSAMHD1 but Not Nuclear or Whole Cell SAMHD1
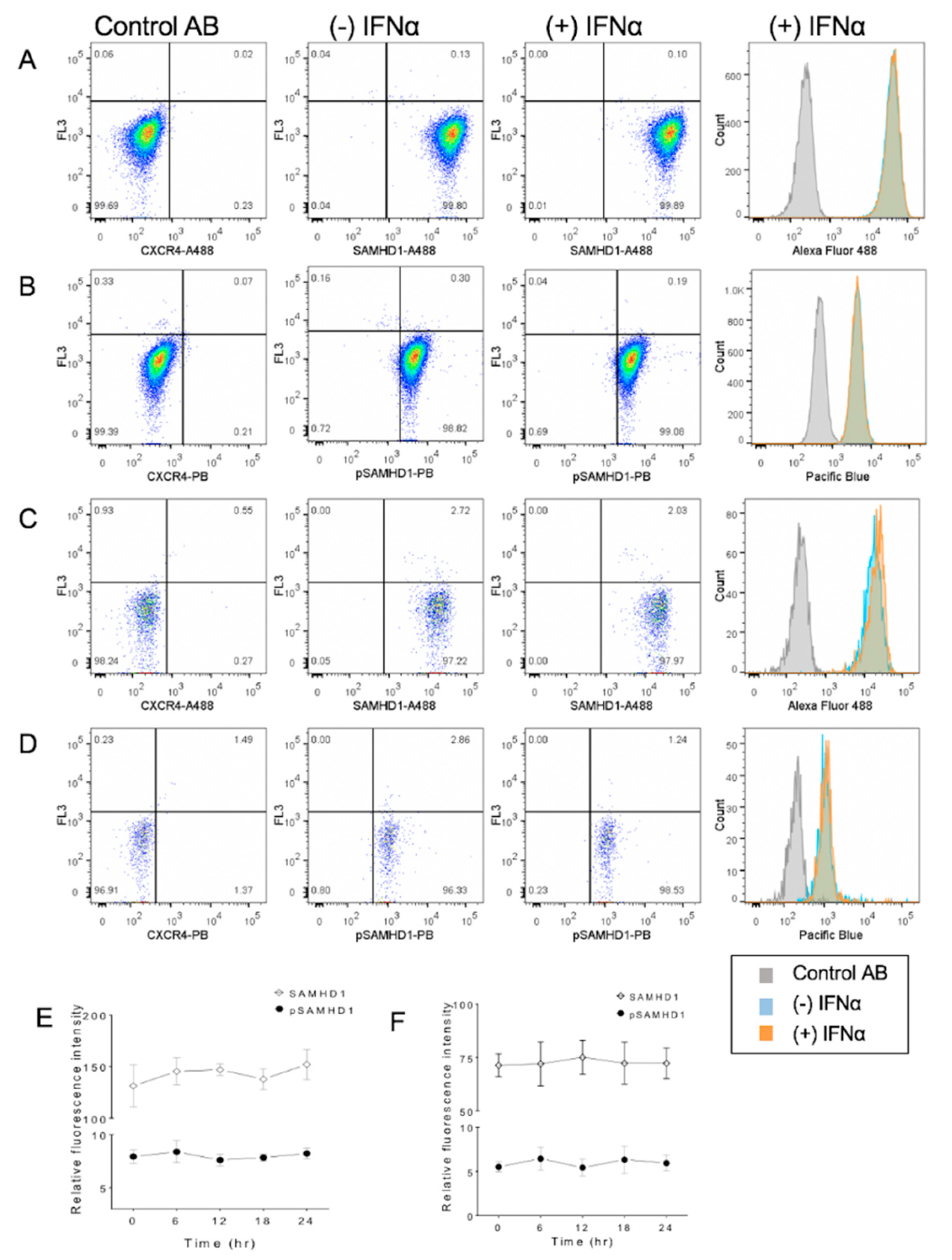
3.4. IFNα Treatment Does Not Significantly Change SAMHD1 Phosphorylation or Localization
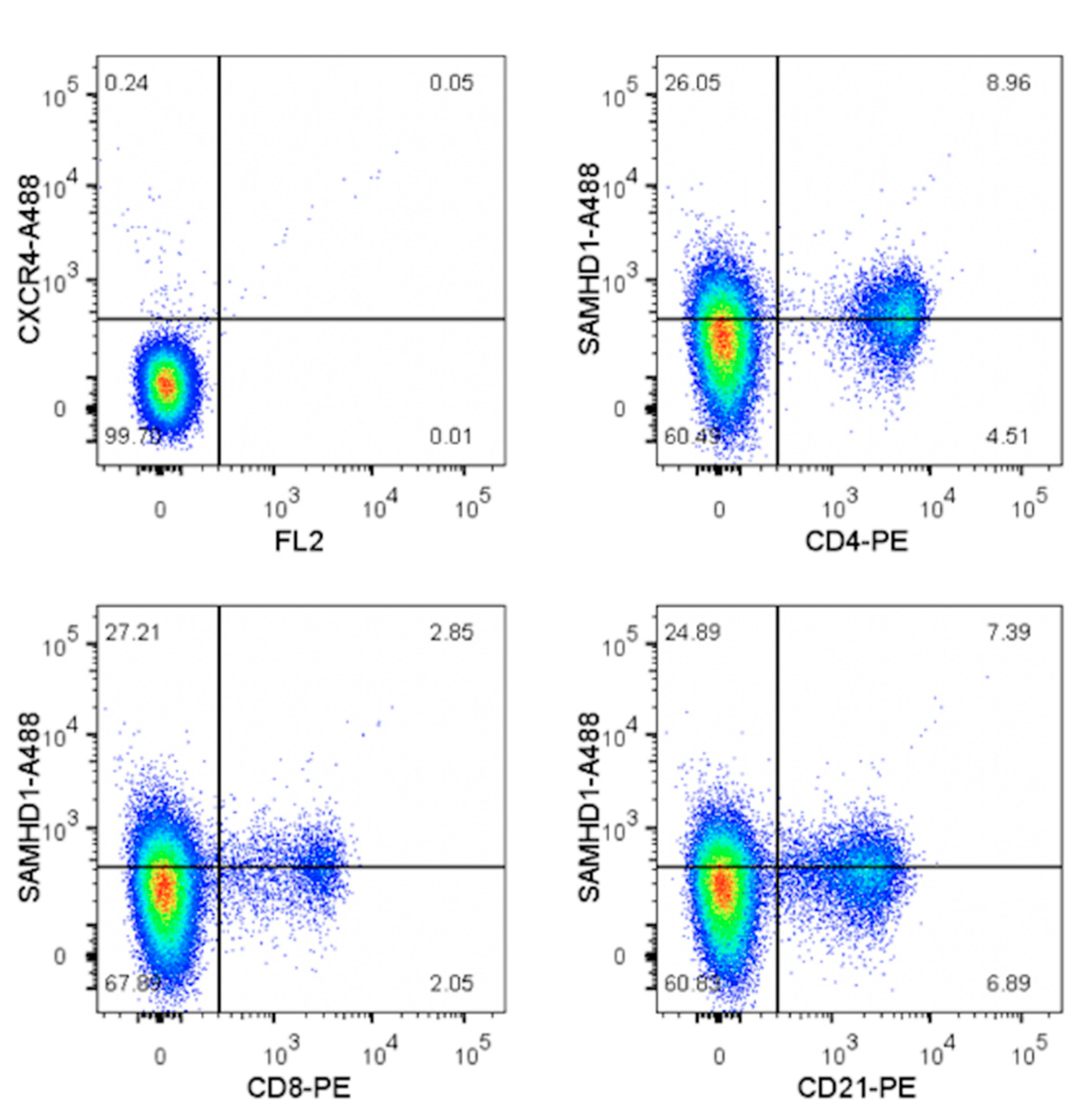
3.5. Primary Lymphocytes Contain Abundant SAMHD1 Protein That Is Not Further Increased by IFNγ Treatment
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mauney, C.H.; Hollis, T. SAMHD1: Recurring roles in cell cycle, viral restriction, cancer, and innate immunity. Autoimmunity 2018, 51, 96–110. [Google Scholar] [CrossRef]
- Pai, C.C.; Kearsey, S.E. A Critical Balance: dNTPs and the Maintenance of Genome Stability. Genes (Basel) 2017, 8, 57. [Google Scholar] [CrossRef] [PubMed]
- Pajalunga, D.; Franzolin, E.; Stevanoni, M.; Zribi, S.; Passaro, N.; Gurtner, A.; Donsante, S.; Loffredo, D.; Losanno, L.; Bianchi, V.; et al. A defective dNTP pool hinders DNA replication in cell cycle-reactivated terminally differentiated muscle cells. Cell Death Differ. 2017, 24, 774–784. [Google Scholar] [CrossRef] [PubMed]
- Goldstone, D.C.; Ennis-Adeniran, V.; Hedden, J.J.; Groom, H.C.; Rice, G.I.; Christodoulou, E.; Walker, P.A.; Kelly, G.; Haire, L.F.; Yap, M.W.; et al. HIV-1 restriction factor SAMHD1 is a deoxynucleoside triphosphate triphosphohydrolase. Nature 2011, 480, 379–382. [Google Scholar] [CrossRef]
- Tramentozzi, E.; Ferraro, P.; Hossain, M.; Stillman, B.; Bianchi, V.; Pontarin, G. The dNTP triphosphohydrolase activity of SAMHD1 persists during S-phase when the enzyme is phosphorylated at T592. Cell Cycle 2018, 17, 1102–1114. [Google Scholar] [CrossRef] [PubMed]
- Sze, A.; Olagnier, D.; Lin, R.; van Grevenynghe, J.; Hiscott, J. SAMHD1 host restriction factor: a link with innate immune sensing of retrovirus infection. J. Mol. Biol. 2013, 425, 4981–4994. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Gao, W.; Zhao, K.; Qin, X.; Zhang, Y.; Peng, X.; Zhang, L.; Dong, Y.; Zhang, W.; Li, P.; et al. Structural insight into dGTP-dependent activation of tetrameric SAMHD1 deoxynucleoside triphosphate triphosphohydrolase. Nat. Commun. 2013, 4, 2722. [Google Scholar] [CrossRef]
- Baldauf, H.-M.; Stegmann, L.; Schwarz, S.-M.; Ambiel, I.; Trotard, M.; Martin, M.; Burggraf, M.; Lenzi, G.M.; Lejk, H.; Pan, X. Vpx overcomes a SAMHD1-independent block to HIV reverse transcription that is specific to resting CD4 T cells. Proc. Natl. Acad. Sci. USA 2017, 114, 2729–2734. [Google Scholar] [CrossRef]
- Hofmann, H.; Logue, E.C.; Bloch, N.; Daddacha, W.; Polsky, S.B.; Schultz, M.L.; Kim, B.; Landau, N.R. The Vpx lentiviral accessory protein targets SAMHD1 for degradation in the nucleus. J. Virol. 2012, 86, 12552–12560. [Google Scholar] [CrossRef]
- Schmidt, S.; Schenkova, K.; Adam, T.; Erikson, E.; Lehmann-Koch, J.; Sertel, S.; Verhasselt, B.; Fackler, O.T.; Lasitschka, F.; Keppler, O.T. SAMHD1’s protein expression profile in humans. J. Leukoc. Biol. 2015, 98, 5–14. [Google Scholar] [CrossRef]
- Baldauf, H.M.; Pan, X.; Erikson, E.; Schmidt, S.; Daddacha, W.; Burggraf, M.; Schenkova, K.; Ambiel, I.; Wabnitz, G.; Gramberg, T.; et al. SAMHD1 restricts HIV-1 infection in resting CD4(+) T cells. Nat. Med. 2012, 18, 1682–1687. [Google Scholar] [CrossRef] [PubMed]
- Hollenbaugh, J.A.; Gee, P.; Baker, J.; Daly, M.B.; Amie, S.M.; Tate, J.; Kasai, N.; Kanemura, Y.; Kim, D.-H.; Ward, B.M. Host factor SAMHD1 restricts DNA viruses in non-dividing myeloid cells. PLoS Pathog. 2013, 9, e1003481. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.T.; Roche, K.L.; Kulej, K.; Spruce, L.A.; Seeholzer, S.H.; Coen, D.M.; Diaz-Griffero, F.; Murphy, E.A.; Weitzman, M.D. SAMHD1 Modulates Early Steps during Human Cytomegalovirus Infection by Limiting NF-κB Activation. Cell Rep. 2019, 28, 434–448. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.T.; White, T.E.; Brandariz-Núñez, A.; Diaz-Griffero, F.; Weitzman, M.D. SAMHD1 restricts herpes simplex virus 1 in macrophages by limiting DNA replication. J. Virol. 2013, 87, 12949–12956. [Google Scholar] [CrossRef] [PubMed]
- White, T.E.; Brandariz-Nunez, A.; Valle-Casuso, J.C.; Amie, S.; Nguyen, L.; Kim, B.; Brojatsch, J.; Diaz-Griffero, F. Contribution of SAM and HD domains to retroviral restriction mediated by human SAMHD1. Virology 2013, 436, 81–90. [Google Scholar] [CrossRef]
- Sommer, A.F.; Rivière, L.; Qu, B.; Schott, K.; Riess, M.; Ni, Y.; Shepard, C.; Schnellbächer, E.; Finkernagel, M.; Himmelsbach, K. Restrictive influence of SAMHD1 on Hepatitis B Virus life cycle. Sci. Rep. 2016, 6, 26616. [Google Scholar] [CrossRef] [PubMed]
- Daep, C.A.; Muñoz-Jordán, J.L.; Eugenin, E.A. Flaviviruses, an expanding threat in public health: focus on dengue, West Nile, and Japanese encephalitis virus. J. Neurovirol. 2014, 20, 539–560. [Google Scholar] [CrossRef] [PubMed]
- Wichit, S.; Hamel, R.; Zanzoni, A.; Diop, F.; Cribier, A.; Talignani, L.; Diack, A.; Ferraris, P.; Liegeois, F.; Urbach, S. SAMHD1 Enhances Chikungunya and Zika Virus Replication in Human Skin Fibroblasts. Int. J. Mol. Sci. 2019, 20, 1695. [Google Scholar] [CrossRef]
- Gelais, C.S.; Kim, S.H.; Maksimova, V.V.; Buzovetsky, O.; Knecht, K.M.; Shepard, C.; Kim, B.; Xiong, Y.; Wu, L. A cyclin-binding motif in human SAMHD1 is required for its HIV-1 restriction, dNTPase activity, tetramer formation, and efficient phosphorylation. J. Virol. 2018, 92, e01787-01717. [Google Scholar] [CrossRef]
- St Gelais, C.; Kim, S.H.; Ding, L.; Yount, J.S.; Ivanov, D.; Spearman, P.; Wu, L. A Putative Cyclin-binding Motif in Human SAMHD1 Contributes to Protein Phosphorylation, Localization, and Stability. J. Biol. Chem. 2016, 291, 26332–26342. [Google Scholar] [CrossRef]
- Cribier, A.; Descours, B.; Valadão, A.L.C.; Laguette, N.; Benkirane, M. Phosphorylation of SAMHD1 by cyclin A2/CDK1 regulates its restriction activity toward HIV-1. Cell Rep. 2013, 3, 1036–1043. [Google Scholar] [CrossRef] [PubMed]
- White, T.E.; Brandariz-Nuñez, A.; Martinez-Lopez, A.; Knowlton, C.; Lenzi, G.; Kim, B.; Ivanov, D.; Diaz-Griffero, F. A SAMHD1 mutation associated with Aicardi-Goutières Syndrome uncouples the ability of SAMHD1 to restrict HIV-1 from its ability to downmodulate type I interferon in humans. Hum. Mutat. 2017, 38, 658–668. [Google Scholar] [CrossRef] [PubMed]
- White, T.E.; Brandariz-Nunez, A.; Valle-Casuso, J.C.; Amie, S.; Nguyen, L.A.; Kim, B.; Tuzova, M.; Diaz-Griffero, F. The retroviral restriction ability of SAMHD1, but not its deoxynucleotide triphosphohydrolase activity, is regulated by phosphorylation. Cell Host Microbe 2013, 13, 441–451. [Google Scholar] [CrossRef] [PubMed]
- Beloglazova, N.; Flick, R.; Tchigvintsev, A.; Brown, G.; Popovic, A.; Nocek, B.; Yakunin, A.F. Nuclease activity of the human SAMHD1 protein implicated in the Aicardi-Goutieres syndrome and HIV-1 restriction. J. Biol. Chem. 2013, 288, 8101–8110. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, A.; Karayel, E.; Rice, G.I.; Bennett, K.L.; Crow, Y.J.; Superti-Furga, G.; Burckstummer, T. SAMHD1 is a nucleic-acid binding protein that is mislocalized due to aicardi-goutieres syndrome-associated mutations. Hum. Mutat. 2012, 33, 1116–1122. [Google Scholar] [CrossRef] [PubMed]
- Ryoo, J.; Hwang, S.-Y.; Choi, J.; Oh, C.; Ahn, K. SAMHD1, the Aicardi-Goutieres syndrome gene and retroviral restriction factor, is a phosphorolytic ribonuclease rather than a hydrolytic ribonuclease. Biochem. Biophys. Res. Commun. 2016, 477, 977–981. [Google Scholar] [CrossRef]
- Li, T.; Chen, Z.J. The cGAS–cGAMP–STING pathway connects DNA damage to inflammation, senescence, and cancer. J. Exp. Med. 2018, 215, 1287–1299. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Damania, B. The cGAS-STING Defense Pathway and Its Counteraction by Viruses. Cell Host Microbe 2016, 19, 150–158. [Google Scholar] [CrossRef]
- Coquel, F.; Silva, M.-J.; Técher, H.; Zadorozhny, K.; Sharma, S.; Nieminuszczy, J.; Mettling, C.; Dardillac, E.; Barthe, A.; Schmitz, A.-L. SAMHD1 acts at stalled replication forks to prevent interferon induction. Nature 2018, 557, 57. [Google Scholar] [CrossRef]
- Ishii, K.J.; Coban, C.; Kato, H.; Takahashi, K.; Torii, Y.; Takeshita, F.; Ludwig, H.; Sutter, G.; Suzuki, K.; Hemmi, H. Erratum: A Toll-like receptor–independent antiviral response induced by double-stranded B-form DNA. Nat. Immunol. 2006, 7, 427. [Google Scholar] [CrossRef]
- Herzner, A.-M.; Hagmann, C.A.; Goldeck, M.; Wolter, S.; Kübler, K.; Wittmann, S.; Gramberg, T.; Andreeva, L.; Hopfner, K.-P.; Mertens, C. Sequence-specific activation of the DNA sensor cGAS by Y-form DNA structures as found in primary HIV-1 cDNA. Nat. Immunol. 2015, 16, 1025. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Bonifati, S.; Qin, Z.; Gelais, C.S.; Wu, L. SAMHD1 suppression of antiviral immune responses. Trends Microbiol. 2019, 27, 254–267. [Google Scholar] [CrossRef] [PubMed]
- De Silva, S.; Hoy, H.; Hake, T.S.; Wong, H.K.; Porcu, P.; Wu, L. Promoter methylation regulates SAMHD1 gene expression in human CD4+ T cells. J. Biol. Chem. 2013, 288, 9284–9292. [Google Scholar] [CrossRef] [PubMed]
- Goujon, C.; Schaller, T.; Galao, R.P.; Amie, S.M.; Kim, B.; Olivieri, K.; Neil, S.J.; Malim, M.H. Evidence for IFNalpha-induced, SAMHD1-independent inhibitors of early HIV-1 infection. Retrovirology 2013, 10, 23. [Google Scholar] [CrossRef] [PubMed]
- St Gelais, C.; de Silva, S.; Amie, S.M.; Coleman, C.M.; Hoy, H.; Hollenbaugh, J.A.; Kim, B.; Wu, L. SAMHD1 restricts HIV-1 infection in dendritic cells (DCs) by dNTP depletion, but its expression in DCs and primary CD4+ T-lymphocytes cannot be upregulated by interferons. Retrovirology 2012, 9, 105. [Google Scholar] [CrossRef] [PubMed]
- Asadian, P.; Finnie, G.; Bienzle, D. The expression profile of sterile alpha motif and histidine-aspartate domain-containing protein 1 (SAMHD1) in feline tissues. Vet. Immunol. Immunopathol. 2018, 195, 7–18. [Google Scholar] [CrossRef]
- Golding, A.; Rosen, A.; Petri, M.; Akhter, E.; Andrade, F. Interferon-alpha regulates the dynamic balance between human activated regulatory and effector T cells: implications for antiviral and autoimmune responses. Immunology 2010, 131, 107–117. [Google Scholar] [CrossRef]
- Achleitner, A.; Clark, M.E.; Bienzle, D. T-regulatory cells infected with feline immunodeficiency virus up-regulate programmed death-1 (PD-1). Vet. Immunol. Immunopathol. 2011, 143, 307–313. [Google Scholar] [CrossRef]
- Penning, L.C.; Vrieling, H.E.; Brinkhof, B.; Riemers, F.M.; Rothuizen, J.; Rutteman, G.R.; Hazewinkel, H.A. A validation of 10 feline reference genes for gene expression measurements in snap-frozen tissues. Vet. Immunol. Immunopathol. 2007, 120, 212–222. [Google Scholar] [CrossRef]
- Schaller, T.; Pollpeter, D.; Apolonia, L.; Goujon, C.; Malim, M.H. Nuclear import of SAMHD1 is mediated by a classical karyopherin α/β1 dependent pathway and confers sensitivity to Vpx MAC induced ubiquitination and proteasomal degradation. Retrovirology 2014, 11, 29. [Google Scholar] [CrossRef]
- Lafuse, W.P.; Brown, D.; Castle, L.; Zwilling, B.S. Cloning and characterization of a novel cDNA that is IFN-γ-induced in mouse peritoneal macrophages and encodes a putative GTP-binding protein. J. Leukoc. Biol. 1995, 57, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Franzolin, E.; Salata, C.; Bianchi, V.; Rampazzo, C. The Deoxynucleoside Triphosphate Triphosphohydrolase Activity of SAMHD1 Protein Contributes to the Mitochondrial DNA Depletion Associated with Genetic Deficiency of Deoxyguanosine Kinase. J. Biol. Chem. 2015, 290, 25986–25996. [Google Scholar] [CrossRef] [PubMed]
- Lahouassa, H.; Daddacha, W.; Hofmann, H.; Ayinde, D.; Logue, E.C.; Dragin, L.; Bloch, N.; Maudet, C.; Bertrand, M.; Gramberg, T. SAMHD1 restricts the replication of human immunodeficiency virus type 1 by depleting the intracellular pool of deoxynucleoside triphosphates. Nat. Immunol. 2012, 13, 223. [Google Scholar] [CrossRef] [PubMed]
- Hansen, E.C.; Seamon, K.J.; Cravens, S.L.; Stivers, J.T. GTP activator and dNTP substrates of HIV-1 restriction factor SAMHD1 generate a long-lived activated state. Proc. Natl. Acad. Sci. USA 2014, 111, 1843–1851. [Google Scholar] [CrossRef] [PubMed]
- Crow, Y.J.; Manel, N. Aicardi–Goutières syndrome and the type I interferonopathies. Nat. Rev. Immunol. 2015, 15, 429. [Google Scholar] [CrossRef] [PubMed]
- The Catalogue of Somatic Mutations in Cancer (COSMIC). Available online: https://cancer.sanger.ac.uk/cosmic/gene/analysis?ln=SAMHD1 (accessed on 28 July 2019).
- Buzovetsky, O.; Tang, C.; Knecht, K.M.; Antonucci, J.M.; Wu, L.; Ji, X.; Xiong, Y. The SAM domain of mouse SAMHD1 is critical for its activation and regulation. Nat. Commun. 2018, 9, 411. [Google Scholar] [CrossRef] [PubMed]
- Schoggins, J.W. Recent advances in antiviral interferon-stimulated gene biology. F1000Research 2018, 7, 309. [Google Scholar] [CrossRef] [PubMed]
- Doyle, T.; Goujon, C.; Malim, M.H. HIV-1 and interferons: who’s interfering with whom? Nat. Rev. Microbiol. 2015, 13, 403. [Google Scholar] [CrossRef]
- Zheng, Y.H.; Jeang, K.T.; Tokunaga, K. Host restriction factors in retroviral infection: promises in virus-host interaction. Retrovirology 2012, 9, 112. [Google Scholar] [CrossRef]
- Peng, G.; Lei, K.J.; Jin, W.; Greenwell-Wild, T.; Wahl, S.M. Induction of APOBEC3 family proteins, a defensive maneuver underlying interferon-induced anti–HIV-1 activity. J. Exp. Med. 2006, 203, 41–46. [Google Scholar] [CrossRef]
- Carthagena, L.; Parise, M.C.; Ringeard, M.; Chelbi-Alix, M.K.; Hazan, U.; Nisole, S. Implication of TRIMalpha and TRIMCyp in interferon-induced anti-retroviral restriction activities. Retrovirology 2008, 5, 59. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, V.L.; Borba, H.H.L.; Bonetti, A.D.F.; Leonart, L.; Pontarolo, R. Cytokines and Interferons: Types and Functions. Available online: https://www.intechopen.com/books/autoantibodies-and-cytokines/cytokines-and-interferons-types-and-functions (accessed on 28 July 2019).
- Lee, A.J.; Ashkar, A.A. The dual nature of type I and type II interferons. Front. Immunol 2018, 9, 2061. [Google Scholar] [CrossRef] [PubMed]
- Welbourn, S.; Dutta, S.M.; Semmes, O.J.; Strebel, K. Restriction of virus infection but not catalytic dNTPase activity are regulated by phosphorylation of SAMHD1. J. Virol. 2013, 8, 11516–11524. [Google Scholar] [CrossRef] [PubMed]
- Tüngler, V.; Staroske, W.; Kind, B.; Dobrick, M.; Kretschmer, S.; Schmidt, F.; Krug, C.; Lorenz, M.; Chara, O.; Schwille, P. Single-stranded nucleic acids promote SAMHD1 complex formation. J. Mol. Med. 2013, 91, 759–770. [Google Scholar] [CrossRef] [PubMed]
- Seamon, K.J.; Sun, Z.; Shlyakhtenko, L.S.; Lyubchenko, Y.L.; Stivers, J.T. SAMHD1 is a single-stranded nucleic acid binding protein with no active site-associated nuclease activity. Nucleic Acids Res. 2015, 43, 6486–6499. [Google Scholar] [CrossRef] [PubMed]
- Ryoo, J.; Choi, J.; Oh, C.; Kim, S.; Seo, M.; Kim, S.Y.; Seo, D.; Kim, J.; White, T.E.; Brandariz-Nunez, A.; et al. The ribonuclease activity of SAMHD1 is required for HIV-1 restriction. Nat. Med. 2014, 20, 936–941. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Lv, D.-W.; Li, R. Conserved Herpesvirus Protein Kinases Target SAMHD1 to Facilitate Virus Replication. Cell Rep. 2019, 28, 449–459. [Google Scholar] [CrossRef]
- Jin, C.; Peng, X.; Liu, F.; Cheng, L.; Xie, T.; Lu, X.; Wu, H.; Wu, N. Interferon-induced SAMHD1 expression in astrocytes and microglia is mediated by miR-181a. AIDS 2016, 30, 2053–2064. [Google Scholar] [CrossRef]
- Jarry, A.; Malard, F.; Bou-Hanna, C.; Meurette, G.; Mohty, M.; Mosnier, J.-F.; Laboisse, C.L.; Bossard, C. Interferon-alpha promotes Th1 response and epithelial apoptosis via inflammasome activation in human intestinal mucosa. Cellular Mol. Gastroenterol. Hepatol. 2017, 3, 72–81. [Google Scholar] [CrossRef]
- Morrison, J.H.; Guevara, R.B.; Marcano, A.C.; Saenz, D.T.; Fadel, H.J.; Rogstad, D.K.; Poeschla, E.M. Feline immunodeficiency virus envelope glycoproteins antagonize tetherin through a distinctive mechanism that requires virion incorporation. J. Virol. 2014, 88, 3255–3272. [Google Scholar] [CrossRef]
- Jin, C.; Peng, X.; Liu, F.; Cheng, L.; Lu, X.; Yao, H.; Wu, H.; Wu, N. MicroRNA-181 expression regulates specific post-transcriptional level of SAMHD1 expression in vitro. Biochem. Biophys. Res. Commun. 2014, 452, 760–767. [Google Scholar] [CrossRef] [PubMed]
- Riess, M.; Fuchs, N.V.; Idica, A.; Hamdorf, M.; Flory, E.; Pedersen, I.M.; König, R. Interferons induce expression of SAMHD1 in monocytes through down-regulation of miR-181a and miR-30a. J. Biol. Chem. 2017, 292, 264–277. [Google Scholar] [CrossRef] [PubMed]
- Hoss, F.; Rolfes, V.; Davanso, M.R.; Braga, T.T.; Franklin, B.S. Detection of ASC speck formation by flow cytometry and chemical cross-linking. In Innate Immune Activation. Methods in Molecular Biology; De Nardo, D., De Nardo, C., Eds.; Springer: New York, NY, USA, 2018; pp. 149–165. [Google Scholar]
- Descours, B.; Cribier, A.; Chable-Bessia, C.; Ayinde, D.; Rice, G.; Crow, Y.; Yatim, A.; Schwartz, O.; Laguette, N.; Benkirane, M. SAMHD1 restricts HIV-1 reverse transcription in quiescent CD4(+) T-cells. Retrovirology 2012, 9, 87. [Google Scholar] [CrossRef] [PubMed]
- Overwijk, W.W.; Schluns, K.S. Functions of γC cytokines in immune homeostasis: Current and potential clinical applications. Clin. Immunol. 2009, 132, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Read, K.A.; Powell, M.D.; McDonald, P.W.; Oestreich, K.J. IL-2, IL-7, and IL-15: multistage regulators of CD4+ T helper cell differentiation. Exp. Hematol. 2016, 44, 799–808. [Google Scholar] [CrossRef]
- Coiras, M.; Bermejo, M.; Descours, B.; Mateos, E.; García-Pérez, J.; López-Huertas, M.-R.; Lederman, M.M.; Benkirane, M.; Alcamí, J. IL-7 Induces SAMHD1 Phosphorylation in CD4+ T Lymphocytes, Improving Early Steps of HIV-1 Life Cycle. Cell Rep. 2016, 14, 2100–2107. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asadian, P.; Bienzle, D. Interferon γ and α Have Differential Effects on SAMHD1, a Potent Antiviral Protein, in Feline Lymphocytes. Viruses 2019, 11, 921. https://doi.org/10.3390/v11100921
Asadian P, Bienzle D. Interferon γ and α Have Differential Effects on SAMHD1, a Potent Antiviral Protein, in Feline Lymphocytes. Viruses. 2019; 11(10):921. https://doi.org/10.3390/v11100921
Chicago/Turabian StyleAsadian, Peyman, and Dorothee Bienzle. 2019. "Interferon γ and α Have Differential Effects on SAMHD1, a Potent Antiviral Protein, in Feline Lymphocytes" Viruses 11, no. 10: 921. https://doi.org/10.3390/v11100921
APA StyleAsadian, P., & Bienzle, D. (2019). Interferon γ and α Have Differential Effects on SAMHD1, a Potent Antiviral Protein, in Feline Lymphocytes. Viruses, 11(10), 921. https://doi.org/10.3390/v11100921




