Feline Virome—A Review of Novel Enteric Viruses Detected in Cats
Abstract
1. Introduction
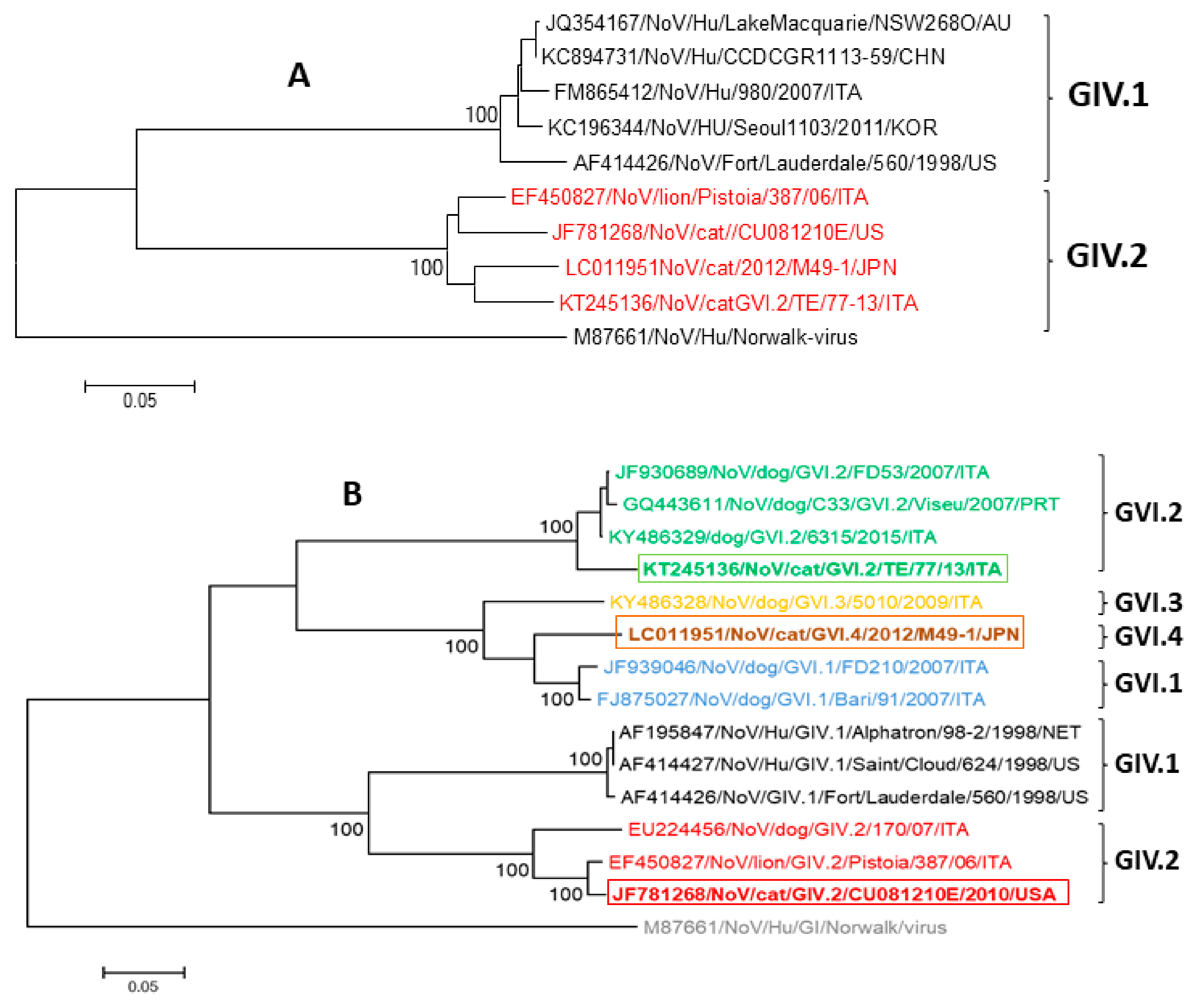
2. Feline Noroviruses
2.1. Etiology
2.2. Epidemiology and Pathogenesis
2.3. Diagnosis
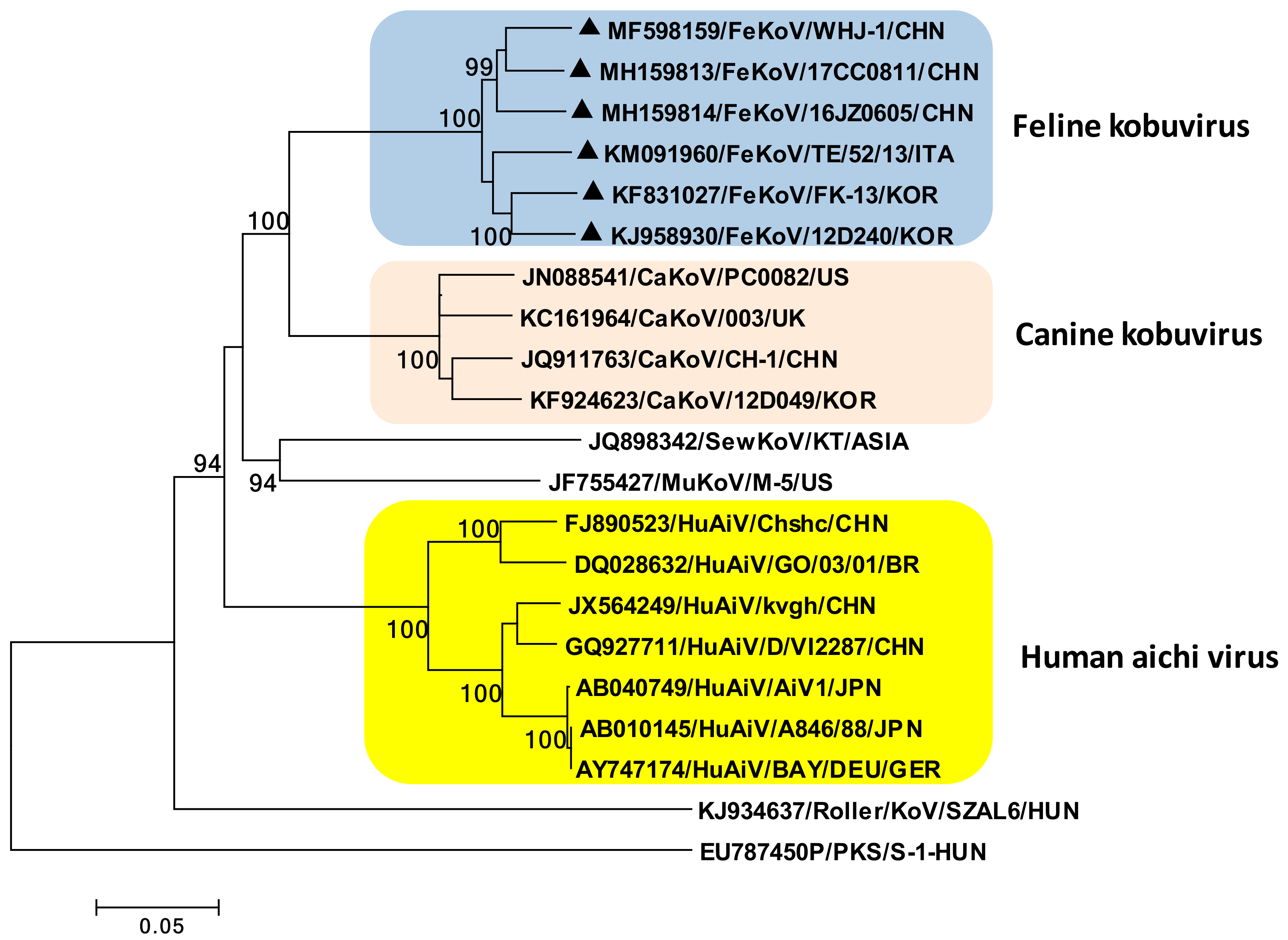
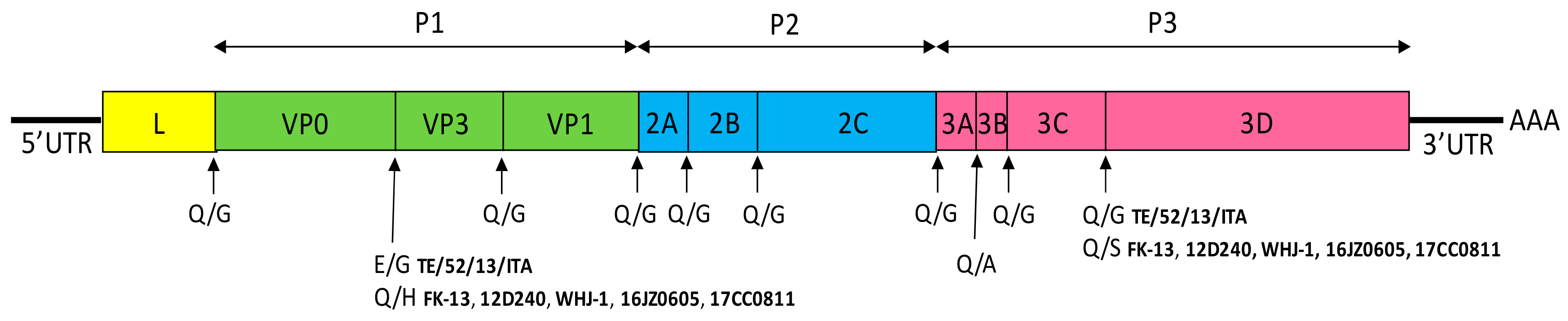
3. Feline Kobuvirus
3.1. Etiology
3.2. Epidemiology and Pathogenesis
3.3. Diagnosis
4. Feline Parvoviruses
4.1. Feline Bocaparvoviruses
4.2. Feline Protoparvoviruses
5. Other Viruses
6. Discussion and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Verge, J.; Cristoforoni, N. La gastroenterite infectieuse des chats estelle due a un virus filtrable? Comptes Rendus Seances Soc. Biol. (Paris) 1928, 99, 312–314. [Google Scholar]
- Parrish, C.R. Pathogenesis of feline panleukopenia virus and canine parvovirus. Baillieres Clin. Haematol. 1995, 8, 57–71. [Google Scholar] [CrossRef]
- Stuetzer, B.; Hartmann, K. Feline parvovirus infection and associated disease. Vet. J. 2014, 201, 150–155. [Google Scholar] [CrossRef] [PubMed]
- Addie, D.D.; Jarrett, O. A study of naturally occurring feline coronavirus infections in kittens. Vet. Rec. 1992, 130, 133–137. [Google Scholar] [CrossRef] [PubMed]
- Drechsler, Y.; Alcaraz, A.; Bossong, F.J.; Collisson, E.W.; Diniz, P.P. Feline coronavirus in multicat environments. Vet. Clin. N. Am. Small Anim. Pract. 2011, 41, 1133–1169. [Google Scholar] [CrossRef]
- Kipar, A.; Kremendahl, J.; Jackson, M.L.; Reinacher, M. Comparative examination of cats with feline leukemia virus-associated enteritis and other relevant forms of feline enteritis. Vet. Pathol. 2001, 38, 359–371. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, Y.; Zimmer, J.F.; Moise, N.S.; Scott, F.W. Detection of astroviruses in feces of a cat with diarrhea. Arch. Virol. 1981, 70, 373–376. [Google Scholar] [CrossRef]
- Rice, M.; Wilks, C.R.; Jones, B.R.; Beck, K.E.; Jones, J.M. Detection of astrovirus in the faeces of cats with diarrhoea. N. Z. Vet. J. 1993, 41, 96–97. [Google Scholar] [CrossRef] [PubMed]
- Marshall, J.A.; Kennett, M.L.; Rodger, S.M.; Studdert, M.J.; Thompson, W.L.; Gust, I.D. Virus and virus-like particles in the faeces of cats with and without diarrhoea. Aust. Vet. J. 1987, 64, 100–105. [Google Scholar] [CrossRef]
- Kennedy, F.A.; Mullaney, T.P. Disseminated adenovirus infection in a cat. J. Vet. Diagn. Investig. 1993, 5, 273–276. [Google Scholar] [CrossRef]
- Pinto, P.; Wang, Q.; Chen, N.; Dubovi, E.J.; Daniels, J.B.; Millward, L.M.; Buonavoglia, C.; Martella, V.; Saif, L.J. Discovery and genomic characterization of noroviruses from a gastroenteritis outbreak in domestic cats in the US. PLoS ONE 2012, 7, e32739. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.K.P.; Woo, P.C.Y.; Yeung, H.C.; Teng, J.L.L.; Wu, Y.; Bai, R.; Fan, R.Y.Y.; Chan, K.H.; Yuen, K.Y. Identification and characterization of bocaviruses in cats and dogs reveals a novel feline bocavirus and a novel genetic group of canine bocavirus. J. Gen. Virol. 2012, 93, 1573–1582. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.Y.; Kim, S.H.; Kim, Y.H.; Lee, M.H.; Lee, K.K.; Oem, J.K. Detection and genetic characterization of feline kobuviruses. Virus Genes 2013, 47, 559–562. [Google Scholar] [CrossRef] [PubMed]
- Diakoudi, G.; Lanave, G.; Capozza, P.; Di Profio, F.; Melegari, I.; Di Martino, B.; Pennisi, M.G.; Elia, G.; Cavalli, A.; Tempesta, M.; et al. Identification of a novel parvovirus in domestic cats. Vet. Microbiol. 2019, 228, 246–251. [Google Scholar] [CrossRef]
- Takano, T.; Yanai, Y.; Hiramatsu, K.; Doki, T.; Hohdatsu, T. Novel single-stranded, circular DNA virus identified in cats in Japan. Arch. Virol. 2018, 163, 3389–3393. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.F.F.; Mesquita, J.R.; Nascimento, M.S.J.; Kondov, N.O.; Wong, W.; Reuter, G.; Knowles, N.J.; Vega, E.; Esona, M.D.; Deng, X.; et al. Feline fecal virome reveals novel and prevalent enteric viruses. Vet. Microbiol. 2014, 17, 102–111. [Google Scholar] [CrossRef]
- Zhang, W.; Li, L.; Deng, X.; Kapusinszky, B.; Pesavento, P.A.; Delwart, E. Faecal virome of cats in an animal shelter. J. Gen. Virol. 2014, 95, 2553–2564. [Google Scholar] [CrossRef] [PubMed]
- Phan, T.G.; Leutenegger, C.M.; Chan, R.; Delwart, E. Rotavirus I in feces of a cat with diarrhea. Virus Genes 2017, 53, 487–490. [Google Scholar] [CrossRef]
- Fahsbender, E.; Altan, E.; Estrada, M.; Seguin, M.A.; Young, P.; Leutenegger, C.M.; Delwart, E. Lyon-IARC polyomavirus DNA in feces of diarrheic cats. Microbiol. Resour. Announc. 2019, 8, e00550-19. [Google Scholar] [CrossRef]
- Patel, M.M.; Widdowson, M.A.; Glass, R.I.; Akazawa, K.; Vinjé, J.; Parashar, U.D. Systematic literature review of role of noroviruses in sporadic gastroenteritis. Emerg. Infect. Dis. 2008, 14, 1224–1231. [Google Scholar] [CrossRef]
- Woode, G.N.; Bridger, J.C. Isolation of small viruses resembling astroviruses and caliciviruses from acute enteritis of calves. J. Med. Microbiol. 1978, 11, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Günther, H.; Otto, P. Diarrhea in young calves. ‘‘Zackenvirus” (Jena agent 117/80) a new diarrhea pathogen in calves. Arch. Exp. Vet. 1987, 41, 934–938. [Google Scholar]
- Sugieda, M.; Nagaoka, H.; Kakishima, Y.; Ohshita, T.; Nakamura, S.; Nakajima, S. Detection of Norwalk-like virus genes in the caecum contents of pigs. Arch. Virol. 1998, 143, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Van Der Poel, W.H.; Vinje, J.; van Der Heide, R.; Herrera, M.I.; Vivo, A.; Koopmans, M.P. Norwalk-like calicivirus genes in farm animals. Emerg. Infect. Dis. 2000, 6, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Martella, V.; Campolo, M.; Lorusso, E.; Cavicchio, P.; Camero, M.; Bellacicco, A.L.; Elia, G.; Greco, G.; Corrente, M.; Desario, C.; et al. Norovirus in captive lion cub (Panthera leo). Emerg. Infect. Dis. 2007, 13, 1071–1073. [Google Scholar] [CrossRef] [PubMed]
- Martella, V.; Lorusso, E.; Decaro, N.; Elia, G.; Radogna, A.; D’Abramo, M.; Desario, C.; Cavalli, A.; Corrente, M.; Germinaro, C.A.; et al. Detection and molecular characterization of a canine norovirus. Emerg. Infect. Dis. 2008, 14, 1306–1308. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Shan, T.; Wang, C.; Côté, C.; Kolman, J.; Onions, D.; Gulland, F.M.; Delwart, E. The fecal viral flora of California sea lions. J. Virol. 2011, 85, 9909–9917. [Google Scholar] [CrossRef]
- Wu, Z.; Yang, L.; Ren, X.; He, G.; Zhang, J.; Yang, J.; Qian, Z.; Dong, J.; Sun, L.; Zhu, Y.; et al. Deciphering the bat virome catalog to better understand the ecological diversity of bat viruses and the bat origin of emerging infectious diseases. ISME J. 2016, 10, 609–620. [Google Scholar] [CrossRef] [PubMed]
- De Graaf, M.; Bodewes, R.; van Elk, C.E.; van de Bildt, M.; Getu, S.; Aron, G.I.; Verjans, G.M.; Osterhaus, A.D.; van den Brand, J.M.; Kuiken, T.; et al. Norovirus Infection in Harbor Porpoises. Emerg. Infect. Dis. 2017, 23, 87–91. [Google Scholar] [CrossRef]
- Carstens, E.B. Ratification vote on taxonomic proposals to the International Commitee on Taxonomy of viruses (2009). Arch. Virol. 2010, 155, 133–146. [Google Scholar] [CrossRef]
- Green, K.Y. Caliciviridae: The Noroviruses, in Fields Virology. In Fields Virology, 5th ed.; Knipe, D.M., Howley, P.M., Griffin, D.E., Lamb, R.A., Martin, M.A., Roizman, B., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007; pp. 949–979. [Google Scholar]
- Thorne, L.G.; Goodfellow, I.G. Norovirus gene expression and replication. J. Gen. Virol. 2014, 95, 278–291. [Google Scholar] [CrossRef] [PubMed]
- Prasad, B.V.V. X-ray Crystallographic Structure of the Norwalk Virus Capsid. Science 1999, 286, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, E.F.; Lindesmith, L.C.; Lobue, A.D.; Baric, R.S. Viral shape-shifting: Norovirus evasion of the human immune system. Nat. Rev. Microbiol. 2010, 8, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Vongpunsawad, S.; Venkataram Prasad, B.V.; Estes, M.K. Norwalk virus minor capsid protein VP2 associates within the VP1 shell domain. J. Virol. 2013, 87, 4818–4825. [Google Scholar] [CrossRef] [PubMed]
- De Graaf, M.; van Beek, J.; Koopmans, M.P. Human norovirus transmission and evolution in a changing world. Nat. Rev. Microbiol. 2016, 14, 421–433. [Google Scholar] [CrossRef]
- Zheng, D.P.; Ando, T.; Frankhouser, R.L.; Beard, R.S.; Glass, R.I.; Monroe, S.S. Norovirus classification and proposed strain nomenclature. Virology 2006, 346, 312–323. [Google Scholar] [CrossRef]
- Wang, Q.H.; Han, M.G.; Cheetham, S.; Souza, M.; Funk, J.A.; Saif, L.J. Porcine noroviruses related to human noroviruses. Emerg. Infect. Dis. 2005, 11, 1874–1881. [Google Scholar] [CrossRef]
- Oliver, S.L.; Asobayire, E.; Dastjerdi, A.M.; Bridger, J.C. Genomic characterization of the unclassified bovine enteric virus Newbury agent-1 (Newbury1) endorses a new genus in the family Caliciviridae. Virology 2006, 350, 240–250. [Google Scholar] [CrossRef]
- Wolf, S.; Williamson, W.; Hewitt, J.; Lin, S.; Rivera-Aban, M.; Ball, A.; Scholes, P.; Savill, M.; Greening, G.E. Molecular detection of norovirus in sheep and pigs in New Zealand farms. Vet. Microbiol. 2009, 133, 184–189. [Google Scholar] [CrossRef]
- Karst, S.M.; Wobus, C.E.; Lay, M.; Davidson, J.; Virgin, H.W.T. STAT1-dependent innate immunity to a Norwalk-like virus. Science 2003, 299, 1575–1578. [Google Scholar] [CrossRef]
- Mesquita, J.R.; Barclay, L.; Nascimento, M.S.; Vinjé, J. Novel norovirus in dogs with diarrhea. Emerg. Infect. Dis. 2010, 16, 980–982. [Google Scholar] [CrossRef] [PubMed]
- Takano, T.; Kusuhara, H.; Kuroishi, A.; Takashina, M.; Doki, T.; Nishinaka, T.; Hohdatsu, T. Molecular characterization and pathogenicity of a genogroup GVI feline norovirus. Vet. Microbiol. 2015, 178, 201–217. [Google Scholar] [CrossRef] [PubMed]
- Tse, H.; Lau, S.K.; Chan, W.M.; Choi, G.K.; Woo, P.C.; Yuen, K.Y. Complete genome sequences of novel canine noroviruses in Hong Kong. J. Virol. 2012, 86, 9531–9532. [Google Scholar] [CrossRef]
- Martella, V.; Decaro, N.; Lorusso, E.; Radogna, A.; Moschidou, P.; Amorisco, F.; Lucente, M.S.; Desario, C.; Elia, G.; Banyai, K.; et al. Genetic heterogeneity and recombination in canine noroviruses. J. Virol. 2009, 83, 11391–11396. [Google Scholar] [CrossRef] [PubMed]
- Ntafis, V.; Xylouri, E.; Radogna, A.; Buonavoglia, C.; Martella, V. Outbreak of canine norovirus infection in young dogs. J. Clin. Microbiol. 2010, 48, 2605–2608. [Google Scholar] [CrossRef]
- Bodnar, L.; Lorusso, E.; Di Martino, B.; Catella, C.; Lanave, G.; Elia, G.; Bányai, K.; Buonavoglia, C.; Martella, V. Identification of a novel canine norovirus. Infect. Genet. Evol. 2017, 52, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, B.; Di Profio, F.; Melegari, I.; Sarchese, V.; Cafiero, M.A.; Robetto, S.; Aste, G.; Lanave, G.; Marsilio, F.; Martella, V. A novel feline norovirus in diarrheic cats. Infect. Genet. Evol. 2016, 38, 132–137. [Google Scholar] [CrossRef]
- Bull, R.A.; Hansman, G.S.; Clancy, L.E.; Tanaka, M.M.; Rawlinson, W.D.; White, P.A. Norovirus recombination in ORF1/ORF2 overlap. Emerg. Infect. Dis. 2005, 11, 1079–1085. [Google Scholar] [CrossRef]
- Di Martino, B.; Marsilio, F.; Di Profio, F.; Lorusso, E.; Friedrich, K.G.; Buonavoglia, C.; Martella, V. Detection of antibodies against norovirus genogroup GIV in carnivores. Clin. Vaccine Immunol. 2010, 17, 180–182. [Google Scholar] [CrossRef]
- Soma, T.; Nakagomi, O.; Nakagomi, T.; Mochizuki, M. Detection of norovirus and sapovirus from diarrheic dogs and cats in Japan. Microbiol. Immunol. 2014, 59, 123–128. [Google Scholar] [CrossRef]
- Castro, T.X.; Cubel Garcia Rde, C.; Fumian, T.M.; Costa, E.M.; Mello, R.; White, P.A.; Leite, J.P. Detection and molecular characterization of caliciviruses (vesivirus and norovirus) in an outbreak of acute diarrhea in kittens from Brazil. Vet. J. 2015, 206, 115–117. [Google Scholar] [CrossRef] [PubMed]
- Takano, T.; Hiramatsu, K.; Matsuyama, M.; Mutoh, K.; Matsumoto, Y.; Fukushima, T.; Doki, T.; Kusuhara, H.; Hohdatsu, T. Viral shedding and clinical status of feline-norovirus-infected cats after reinfection with the same strain. Arch. Virol. 2018, 163, 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Huang, P.W.; Zhong, W.M.; Farkas, T.; Cubitt, D.W.; Matson, D.O. Design and evaluation of a primer pair that detects both Norwalk- and Sapporo-like caliciviruses by RT-PCR. J. Virol. Methods 1999, 83, 145–154. [Google Scholar] [CrossRef]
- Vennema, H.; de Bruin, E.; Koopmans, M. Rational optimization of generic primers used for Norwalk-like virus detection by reverse transcriptase polymerase chain reaction. J. Clin. Virol. 2002, 25, 233–235. [Google Scholar] [CrossRef]
- Scotto-Lavino, E.; Du, G.; Frohman, M.A. 3′ end cDNA amplification using classic RACE. Nat. Protoc. 2006, 1, 2742–2745. [Google Scholar] [CrossRef] [PubMed]
- Wobus, C.E.; Karst, S.M.; Thackray, L.B.; Chang, K.O.; Sosnovtsev, S.V.; Belliot, G.; Krug, A.; Mackenzie, J.M.; Green, K.Y.; Virgin, H.W. Replication of norovirus in cell culture reveals a tropism for dendritic cells and macrophages. PLoS Biol. 2004, 2, e4. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.K.; Watanabe, M.; Zhu, S.; Graves, C.L.; Keyes, L.R.; Grau, K.R.; Gonzalez-Hernandez, M.B.; Iovine, N.M.; Wobus, C.E.; Vinje, J.; et al. Enteric bacteria promote human and mouse norovirus infection of b cells. Science 2014, 346, 755–759. [Google Scholar] [CrossRef]
- Ettayebi, K.; Crawford, S.E.; Murakami, K.; Broughman, J.R.; Karandikar, U.; Tenge, V.R.; Neill, F.H.; Blutt, S.E.; Zeng, X.L.; Qu, L.; et al. Replication of human noroviruses in stem cell-derived human enteroids. Science 2016, 353, 1387–1393. [Google Scholar] [CrossRef]
- Bhar, S.; Jones, M.K. In Vitro Replication of Human Norovirus. Viruses 2019, 11, 547. [Google Scholar] [CrossRef]
- Jiang, X.; Zhong, W.M.; Huang, W.; Wilton, N.; Barret, E.; Fulton, D.; Morrow, R.; Matson, D.O. Baculovirus expression and antigenic characterization of the capsid proteins of three Norwalk-like viruses. Arch. Virol. 2002, 147, 119–130. [Google Scholar] [CrossRef]
- Jiang, X.; Wilton, N.; Zhong, W.M.; Farkas, T.; Huang, P.W.; Barrett, E.; Guerrero, M.; Ruiz-Palacios, G.; Green, K.Y.; Green, J.; et al. Diagnosis of human calicivirus by use of enzyme immunoassays. J. Infect. Dis. 2000, 181, S349–S359. [Google Scholar] [CrossRef] [PubMed]
- Farkas, T.; Nakajima, S.; Sugieda, M.; Deng, X.; Zhong, W.M.; Jiang, X. Seroprevalence of noroviruses in swine. J. Clin. Microbiol. 2005, 43, 657–661. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, B.; Di Profio, F.; Ceci, C.; Di Felice, E.; Green, K.Y.; Bok, K.; De Grazia, S.; Giammanco, G.M.; Massirio, I.; Lorusso, E.; et al. Seroprevalence of norovirus genogroup IV antibodies among humans, Italy, 2010–2011. Emerg. Infect. Dis. 2014, 20, 1828–1832. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, B.; Di Profio, F.; Melegari, I.; Sarchese, V.; Massirio, I.; Palermo, G.; Romito, G.; Lorusso, E.; Lanave, G.; Bodnar, L.; et al. Seroprevalence for norovirus genogroup II, IV and VI in dogs. Vet. Microbiol. 2017, 203, 68–72. [Google Scholar] [CrossRef]
- Di Profio, F.; Sarchese, V.; Melegari, I.; Palombieri, A.; Massirio, I.; Bermudez Sanchez, S.; Friedrich, K.G.; Coccia, F.; Marsilio, F.; Martella, V.; et al. Seroprevalence for norovirus genogroups GII and GIV in captive non-human primates. Zoonoses Public Health 2019, 66, 310–315. [Google Scholar] [CrossRef]
- ICTV-International Commettee on Taxonomy of Viruses. Available online: https://talk.ictvonline.org/ictvreports/ictv_online_report/positivesensernaviruses/picornavirales/w/picornaviridae/686/genus-kobuvirus (accessed on 14 August 2019).
- Yamashita, T.; Kobayashi, S.; Sakae, K.; Nakata, S.; Chiba, S.; Ishihara, Y.; Isomura, S. Isolation of cytopathic small round viruses with BS-C-1 cells from patients with gastroenteritis. J. Infect. Dis. 1991, 164, 954–957. [Google Scholar] [CrossRef] [PubMed]
- Ambert-Balay, K.; Lorrot, M.; Bon, F.; Giraudon, H.; Kaplon, J.; Wolfer, M.; Lebon, P.; Gendrel, D.; Pothier, P. Prevalence and genetic diversity of Aichi virus strains in stool samples from community and hospitalized patients. J. Clin. Microbiol. 2008, 46, 1252–1258. [Google Scholar] [CrossRef]
- Reuter, G.; Boldizsar, A.; Papp, G.; Pankovics, P. Detection of Aichi virus shedding in a child with enteric and extraintestinal symptoms in Hungary. Arch. Virol. 2009, 154, 1529–1532. [Google Scholar] [CrossRef]
- Kaikkonen, S.; Räsänen, S.; Rämet, M.; Vesikari, T. Aichi virus infection in children with acute gastroenteritis in Finland. Epidemiol. Infect. 2010, 138, 1166–1171. [Google Scholar] [CrossRef]
- Jonsson, N.; Wahlström, K.; Svensson, L.; Serrander, L.; Lindberg, A.M. Aichi virus infection in elderly people in Sweden. Arch. Virol. 2012, 157, 1365–1369. [Google Scholar] [CrossRef]
- Li, L.; Pesavento, P.A.; Shan, T.; Leutenegger, C.M.; Wang, C.; Delwart, E. Viruses in diarrhoeic dogs include novel kobuviruses and sapoviruses. J. Gen. Virol. 2011, 92, 2534–2541. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, A.; Simmonds, P.; Dubovi, E.J.; Qaisar, N.; Henriquez, J.A.; Medina, J.; Shields, S.; Lipkin, W.I. Characterization of a canine homolog of human Aichivirus. J. Virol. 2011, 85, 11520–11525. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, B.; Di Profio, F.; Melegari, I.; Robetto, S.; Di Felice, E.; Orusa, R.; Marsilio, F. Molecular evidence of kobuviruses in free-ranging red foxes (Vulpes vulpes). Arch. Virol. 2014, 159, 1803–1806. [Google Scholar] [CrossRef] [PubMed]
- Olarte-Castillo, X.A.; Heeger, F.; Mazzoni, C.J.; Greenwood, A.D.; Fyumagwa, R.; Moehlman, P.D.; Hofer, H.; East, M.L. Molecular characterization of canine kobuvirus in wild carnivores and the domestic dog in Africa. Virology 2015, 477, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Melegari, I.; Sarchese, V.; Di Profio, F.; Robetto, S.; Carella, E.; Bermudez Sanchez, S.; Orusa, R.; Martella, V.; Marsilio, F.; Di Martino, B. First molecular identification of kobuviruses in wolves (Canis lupus) in Italy. Arch. Virol. 2018, 163, 509–513. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.J.; King, A.M.; Carstens, E.B. Ratification vote on taxonomic proposals to the International Committee on Taxonomy of Viruses (2013). Arch. Virol. 2013, 158, 2023–2030. [Google Scholar] [CrossRef]
- Adams, M.J.; Lefkowitz, E.J.; King, A.M.Q.; Harrach, B.; Harrison, R.L.; Knowles, N.J.; Kropinski, A.M.; Krupovic, M.; Kuhn, J.H.; Mushegian, A.R.; et al. Changes to taxonomy and the International Code of Virus Classification and Nomenclature ratified by the International Committee on Taxonomy of Viruses. Arch. Virol. 2017, 162, 2505–2538. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, B.; Di Felice, E.; Ceci, C.; Di Profio, F.; Marsilio, F. Canine kobuviruses in diarrhoeic dogs in Italy. Vet. Microbiol. 2013, 166, 246–249. [Google Scholar] [CrossRef]
- Carmona-Vicente, N.; Buesa, J.; Brown, P.A.; Merga, J.Y.; Darby, A.C.; Stavisky, J.; Sadler, L.; Gaskell, R.M.; Dawson, S.; Radford, A.D. Phylogeny and prevalence of kobuviruses in dogs and cats in the UK. Vet. Microbiol. 2013, 164, 246–252. [Google Scholar] [CrossRef]
- Phan, T.G.; Kapusinszky, B.; Wang, C.; Rose, R.K.; Lipton, H.L.; Delwart, E.L. The fecal viral flora of wild rodents. PLoS Pathog. 2011, 7, e1002218. [Google Scholar] [CrossRef]
- Cho, Y.Y.; Lim, S.I.; Kim, Y.K.; Song, J.Y.; Lee, J.B.; An, D.J. Molecular characterization of the full kobuvirus genome in a cat. Genome Announc. 2014, 2, e00420-14. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.W.; Lee, M.H.; Lee, K.K.; Oem, J.K. Genetic characteristics of the complete feline kobuvirus genome. Virus Genes 2014, 50, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, B.; Di Profio, F.; Melegari, I.; Marsilio, F.; Martella, V. Detection of feline kobuviruses in diarrhoeic cats, Italy. Vet. Microbiol. 2015, 176, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Zhang, X.; Luo, J.; Sun, Y.; Xu, H.; Huang, J.; Ou, J.; Li, S. First report and genetic characterization of feline kobuvirus in diarrhoeic cats in China. Transbound. Emerg. Dis. 2018, 65, 1357–1363. [Google Scholar] [CrossRef]
- Niu, T.J.; Yi, S.S.; Wang, X.; Wang, L.H.; Guo, B.Y.; Zhao, L.Y.; Zhang, S.; Dong, H.; Wang, K.; Hu, X.G. Detection and genetic characterization of kobuvirus in cats: The first molecular evidence from Northeast China. Infect. Genet. Evol. 2019, 68, 58–67. [Google Scholar] [CrossRef]
- Han, X.; Zhang, W.; Xue, Y.; Shao, S. Sequence analysis reveals mosaic genome of Aichi virus. Virol. J. 2011, 8, 390. [Google Scholar] [CrossRef]
- Reuter, G.; Boros, A.; Pankovics, P. Kobuviruses—A comprehensive review. Rev. Med. Virol. 2011, 21, 32–41. [Google Scholar] [CrossRef]
- Cho, Y.Y.; Lim, S.I.; Kim, Y.K.; Song, J.Y.; Lee, J.B.; An, D.J. Molecular evolution of kobuviruses in cats. Arch. Virol. 2015, 160, 537–541. [Google Scholar] [CrossRef]
- Yamashita, T.; Ito, M.; Kabashima, Y.; Tsuzuki, H.; Fujiura, A.; Sakae, K. Isolation and characterization of a new species of kobuvirus associated with cattle. J. Gen. Virol. 2003, 84, 3069–3077. [Google Scholar] [CrossRef]
- Reuter, G.; Boldizsár, A.; Pankovics, P. Complete nucleotide and amino acid sequences and genetic organization of porcine kobuvirus, a member of a new species in the genus Kobuvirus, family Picornaviridae. Arch. Virol. 2009, 154, 101–108. [Google Scholar] [CrossRef]
- Cotmore, S.F.; Tattersall, P. Structure and organization of the viral genome. In Parvoviruses; Kerr, J., Cotmore, S.F., Bloom, M.E., Linden, R.M., Parrish, C.R., Eds.; Hodder Arnold: London, UK, 2005; pp. 73–94. [Google Scholar]
- Cotmore, S.F.; Agbandje-McKenna, M.; Chiorini, J.A.; Mukha, D.V.; Pintel, D.J.; Qiu, J.; Soderlund-Venermo, M.; Tattersall, P.; Tijssen, P.; Gatherer, D.; et al. The family Parvoviridae. Arch. Virol. 2014, 159, 1239–1247. [Google Scholar] [CrossRef] [PubMed]
- Csiza, C.K.; Scott, F.W.; De Lahunta, A.; Gillespie, J.H. Pathogenesis of feline panleukopenia virus in susceptible newborn kittens I. Clinical signs, hematology, serology, and virology. Infect. Immun. 1971, 3, 833–837. [Google Scholar] [PubMed]
- Barrs, V.R. Feline Panleukopenia: A Re-Emergent Disease. Vet. Clin. N. Am. Small Anim. Pract. 2019, 49, 651–670. [Google Scholar] [CrossRef] [PubMed]
- Allander, T.; Tammi, M.T.; Eriksson, M.; Bjerkner, A.; Tiveljung-Lindell, A.; Andersson, B. Cloning of a human parvovirus by molecular screening of respiratory tract samples. Proc. Natl. Acad. Sci. USA 2005, 102, 12891–12896. [Google Scholar] [CrossRef] [PubMed]
- Vicente, D.; Cilla, G.; Montes, M.; Perez-Yarza, E.G.; Perez-Trallero, E. Human bocavirus, a respiratory and enteric virus. Emerg. Infect. Dis. 2007, 13, 636–637. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, A.; Mehta, N.; Esper, F.; Poljsak-Prijatelj, M.; Quan, P.L.; Qaisar, N.; Delwart, E.; Lipkin, W.I. Identification and characterization of a new bocavirus species in gorillas. PLoS ONE 2010, 5, e11948. [Google Scholar] [CrossRef]
- Sharp, C.P.; LeBreton, M.; Kantola, K.; Nana, A.; Diffo, J.D.; Djoko, C.F.; Tamoufe, U.; Kiyang, J.A.; Babila, T.G.; Ngole, E.M.; et al. Widespread infection with homologues of human parvoviruses B19, PARV4, and human bocavirus of chimpanzees and gorillas in the wild. J. Virol. 2010, 84, 10289–10296. [Google Scholar] [CrossRef]
- Cheng, W.X.; Li, J.S.; Huang, C.P.; Yao, D.P.; Liu, N.; Cui, S.X.; Jin, Y.; Duan, Z.J. Identification and nearly full-length genome characterization of novel porcine bocaviruses. PLoS ONE 2010, 5, e13583. [Google Scholar] [CrossRef]
- Shan, T.; Lan, D.; Li, L.; Wang, C.; Cui, L.; Zhang, W.; Hua, X.; Zhu, C.; Zhao, W.; Delwart, E. Genomic characterization and high prevalence of bocaviruses in swine. PLoS ONE 2011, 6, e17292. [Google Scholar] [CrossRef]
- Wu, Z.; Ren, X.; Yang, L.; Hu, Y.; Yang, J.; He, G.; Zhang, J.; Dong, J.; Sun, L.; Du, J.; et al. Virome analysis for identification of novel mammalian viruses in bat species from Chinese provinces. J. Virol. 2012, 86, 10999–11012. [Google Scholar] [CrossRef]
- Lanave, G.; Martella, V.; Farkas, S.; Marton, S.; Fehér, E.; Bodnar, L.; Lavazza, A.; Decaro, N.; Buonavoglia, C.; Bányai, K. Novel bocaparvoviruses in rabbits. Vet. J. 2015, 206, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Wang, Y.; Li, W.; Fan, Z.; Jiang, L.; Lin, Y.; Fu, X.; Shen, Q.; Sun, Z.; Wang, X.; et al. A novel bocavirus from domestic mink, China. Virus Genes 2016, 52, 887–890. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.K.; Yeung, H.C.; Li, K.S.; Lam, C.S.; Cai, J.P.; Yuen, M.C.; Wang, M.; Zheng, B.J.; Woo, P.C.; Yuen, K.Y. Identification and genomic characterization of a novel rat bocavirus from brown rats in China. Infect. Genet. Evol. 2017, 47, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Van den Brand, J.M.; van Leeuwen, M.; Schapendonk, C.M.; Simon, J.H.; Haagmans, B.L.; Osterhaus, A.D.; Smits, S.L. Metagenomic analysis of the viral flora of pine marten and European badger feces. J. Virol. 2012, 86, 2360–2365. [Google Scholar] [CrossRef] [PubMed]
- Binn, L.N.; Lazar, E.C.; Eddy, G.A.; Kajima, M. Recovery and characterization of a minute virus of canines. Infect. Immun. 1970, 1, 503–508. [Google Scholar] [PubMed]
- Kapoor, A.; Mehta, N.; Dubovi, E.J.; Simmonds, P.; Govindasamy, L.; Medina, J.L.; Street, C.; Shields, S.; Lipkin, W.I. Characterization of novel canine bocaviruses and their association with respiratory disease. J. Gen. Virol. 2012, 93, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.C.; Shull, B.C.; Moses, E.A.; Lederman, M.; Stout, E.R.; Bates, R.C. Complete nucleotide sequence and genome organization of bovine parvovirus. J. Virol. 1986, 60, 1085–1097. [Google Scholar]
- Sun, Y.; Chen, A.Y.; Cheng, F.; Guan, W.; Johnson, F.B.; Qiu, J. Molecular characterization of infectious clones of the minute virus of canines reveals unique features of bocaviruses. J. Virol. 2009, 83, 3956–3967. [Google Scholar] [CrossRef] [PubMed]
- Zou, W.; Cheng, F.; Shen, W.; Engelhardt, J.F.; Yan, Z.; Qiu, J. Nonstructural protein NP1 of human Bocavirus 1 plays a critical role in the expression of viral capsid proteins. J. Virol. 2016, 90, 4658–4669. [Google Scholar] [CrossRef]
- ICTV-International Commettee on Taxonomy of Viruses. Available online: https://talk.ictvonline.org/ictvreports/ictv_online_report/ssdnaviruses/w/parvoviridae/1041/genus-bocaparvovirus (accessed on 30 August 2019).
- Takano, T.; Takadate, Y.; Doki, T.; Hohdatsu, T. Genetic characterization of feline bocavirus detected in cats in Japan. Arch. Virol. 2016, 161, 2825–2828. [Google Scholar] [CrossRef]
- Liu, C.; Liu, F.; Li, Z.; Qu, L.; Liu, D. First report of feline bocavirus associated with severe enteritis of cat in Northeast China, 2015. J. Vet. Med. Sci. 2018, 80, 731–735. [Google Scholar] [CrossRef] [PubMed]
- Yi, S.; Niu, J.; Wang, H.; Dong, G.; Zhao, Y.; Dong, H.; Guo, Y.; Wang, K.; Hu, G. Detection and genetic characterization of feline bocavirus in Northeast China. Virol. J. 2018, 15, 125. [Google Scholar] [CrossRef] [PubMed]
- Sloots, T.P.; McErlean, P.; Speicher, D.J.; Arden, K.E.; Nissen, M.D.; Mackay, I.M. Evidence of human coronavirus HKU1 and human bocavirus in Australian children. J. Clin. Virol. 2006, 35, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.K.; Yip, C.C.; Que, T.L.; Lee, R.A.; Au-Yeung, R.K.; Zhou, B.P.; So, L.Y.; Lau, Y.L.; Chan, K.H.; Woo, P.C.; et al. Clinical and molecular epidemiology of human bocavirus in respiratory and fecal samples from children in Hong Kong. J. Infect. Dis. 2007, 196, 986–993. [Google Scholar] [CrossRef] [PubMed]
- Sőderlund-Venermo, M.; Lahtinen, A.; Jartti, T.; Hedman, L.; Kemppainen, K.; Lehtinen, P.; Allander, T.; Ruuskanen, O.; Hedman, K. Clinical assessment and improved diagnosis of bocavirus-induced wheezing in children, Finland. Emerg. Infect. Dis. 2009, 15, 1423–1430. [Google Scholar] [CrossRef]
- Arthur, J.L.; Higgins, G.D.; Davidson, G.P.; Givney, R.C.; Ratcliff, R.M. A novel bocavirus associated with acute gastroenteritis in Australian children. PLoS Pathog. 2009, 5, e1000391. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, A.; Slikas, E.; Simmonds, P.; Chieochansin, T.; Naeem, A.; Shaukat, S.; Alam, M.M.; Sharif, S.; Angez, M.; Zaidi, S.; et al. A newly identified bocavirus species in human stool. J. Infect. Dis. 2009, 199, 196–200. [Google Scholar] [CrossRef]
- Hao, R.; Ni, K.; Xia, Q.; Peng, C.; Deng, Y.; Zhao, X.; Fu, Z.; Liu, W.; Liu, E. Correlation between nucleotide mutation and viral loads of human bocavirus 1 in hospitalized children with respiratory tract infection. J. Gen. Virol. 2013, 94, 1079–1185. [Google Scholar] [CrossRef]
- Manteufel, J.; Truyen, U. Animal bocaviruses: A brief review. Intervirology 2008, 51, 328–334. [Google Scholar] [CrossRef]
- Choi, J.W.; Lee, K.H.; Lee, J.I.; Lee, M.H.; Lee, K.K.; Oem, J.K. Genetic characteristics of canine bocaviruses in Korean dogs. Vet. Microbiol. 2015, 179, 177–183. [Google Scholar] [CrossRef]
- Li, L.; Pesavento, P.A.; Leutenegger, C.M.; Estrada, M.; Coffey, L.L.; Naccache, S.N.; Samayoa, E.; Chiu, C.; Qiu, J.; Wang, C.; et al. A novel bocavirus in canine liver. Virol. J. 2013, 10, 54. [Google Scholar] [CrossRef]
- Mochizuki, M.; Hashimoto, M.; Hajima, T.; Takiguchi, M.; Hashimoto, A.; Une, Y.; Roerink, F.; Ohshima, T.; Parrish, C.R.; Carmichael, L.E. Virologic and serologic identification of minute virus of canines (canine parvovirus type 1) from dogs in Japan. J. Clin. Microbiol. 2002, 40, 3993–3998. [Google Scholar] [CrossRef] [PubMed]
- Dijkman, R.; Koekkoek, S.M.; Molenkamp, R.; Schildgen, O.; van der Hoek, L. Human bocavirus can be cultured in differentiated human airway epithelial cells. J. Virol. 2009, 83, 7739–7748. [Google Scholar] [CrossRef] [PubMed]
- Ohshima, T.; Kawakamia, K.; Abeb, T.; Mochizuki, M. A minute virus of canines (MVC: Canine bocavirus) isolated from an elderly dog with severe gastroenteritis, and phylogenetic analysis of MVC strains. Vet. Microbiol. 2010, 145, 334–338. [Google Scholar] [CrossRef] [PubMed]
- McKillen, J.; McNeilly, F.; Duffy, C.; McMenamy, M.; McNair, I.; Hjertner, B.; Millar, A.; McKay, K.; Lagan, P.; Adair, B.; et al. Isolation in cell cultures and initial characterisation of two novel bocavirus species from swine in Northern Ireland. Vet. Microbiol. 2011, 152, 39–45. [Google Scholar] [CrossRef] [PubMed]
- ICTV-International Commettee on Taxonomy of Viruses. Available online: https://talk.ictvonline.org/ictvreports/ictv_online_report/ssdnaviruses/w/parvoviridae/1045/genus-protoparvovirus (accessed on 31 August 2019).
- Johnson, R.H.; Margolis, G.; Kilham, L. Identity of feline Ataxia virus with feline Panleucopenia virus. Nature 1967, 214, 175. [Google Scholar] [CrossRef]
- Truyen, U.; Platzer, G.; Parrish, C.R. Antigenic type distribution among canine parvoviruses in dogs and cats in Germany. Vet. Rec. 1996, 138, 365–366. [Google Scholar] [CrossRef]
- Truyen, U. Evolution of canine parvovirus—A need for new vaccines? Vet. Microbiol. 2006, 117, 9–13. [Google Scholar] [CrossRef]
- Decaro, N.; Buonavoglia, D.; Desario, C.; Amorisco, F.; Colaianni, M.L.; Parisi, A.; Terio, V.; Elia, G.; Lucente, M.S.; Cavalli, A.; et al. Characterisation of canine parvovirus strains isolated from cats with feline panleukopenia. Res. Vet. Sci. 2010, 89, 275–278. [Google Scholar] [CrossRef]
- Appel, M.J.; Scott, F.W.; Carmichael, L.E. Isolation and immunisation studies of a canine parvo-like virus from dogs with haemorrhagic enteritis. Vet. Rec. 1979, 105, 156–159. [Google Scholar] [CrossRef]
- Parrish, C.R.; O’Connell, P.H.; Evermann, J.F.; Carmichael, L.E. Natural variation of canine parvovirus. Science 1985, 230, 1046–1048. [Google Scholar] [CrossRef] [PubMed]
- Parrish, C.R.; Charles, F.; Strassheim, M.L.; Evermann, J.F.; Sgro, J.Y.; Mohammed, H.O. Rapid antigenic-type replacement and DNA sequence evolution of canine parvovirus. J. Virol. 1991, 65, 6544–6552. [Google Scholar] [PubMed]
- Buonavoglia, C.; Martella, V.; Pratella, A.; Tempesta, M.; Cavalli, A.; Buonavoglia, D.; Bozzo, G.; Elia, G.; Decaro, N.; Carmichael, L. Evidence for evolution of canine parvovirus type 2 in Italy. J. Gen. Virol. 2001, 82, 3021–3025. [Google Scholar] [CrossRef] [PubMed]
- Truyen, U.; Evermann, J.F.; Vieler, E.; Parrish, C.R. Evolution of canine parvovirus involved loss and gain of feline host range. Virology 1996, 215, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Hueffer, K.; Parrish, C.R. Parvovirus host range, cell tropism and evolution. Curr. Opin. Microbiol. 2003, 6, 392–398. [Google Scholar] [CrossRef]
- Martella, V.; Lanave, G.; Mihalov-Kovács, E.; Marton, S.; Varga-Kugler, R.; Kaszab, E.; Di Martino, B.; Camero, M.; Decaro, N.; Buonavoglia, C.; et al. Novel Parvovirus Related to Primate Bufaviruses in Dogs. Emerg. Infect. Dis. 2018, 24, 1061–1068. [Google Scholar] [CrossRef] [PubMed]
- Phan, T.G.; Vo, N.P.; Bonkoungou, I.J.; Kapoor, A.; Barro, N.; O’Ryan, M.; Kapusinszky, B.; Wang, C.; Delwart, E. Acute diarrhea in West African children: Diverse enteric viruses and a novel parvovirus genus. J. Virol. 2012, 86, 11024–11030. [Google Scholar] [CrossRef]
- Yahiro, T.; Wangchuk, S.; Tshering, K.; Bandhari, P.; Zangmo, S.; Dorji, T.; Tshering, K.; Matsumoto, T.; Nishizono, A.; Söderlund-Venermo, M.; et al. Novel human bufavirus genotype 3 in children with severe diarrhea, Bhutan. Emerg. Infect. Dis. 2014, 20, 1037–1039. [Google Scholar] [CrossRef]
- Handley, S.A.; Thackray, L.B.; Zhao, G.; Presti, R.; Miller, A.D.; Droit, L.; Abbink, P.; Maxfield, L.F.; Kambal, A.; Duan, E.; et al. Pathogenic simian immunodeficiency virus infection is associated with expansion of the enteric virome. Cell 2012, 151, 253–266. [Google Scholar] [CrossRef]
- Melegari, I.; Di Profio, F.; Palombieri, A.; Sarchese, V.; Diakoudi, G.; Robetto, S.; Orusa, R.; Marsilio, F.; Bányai, K.; Martella, V.; et al. Molecular detection of canine bufaviruses in wild canids. Arch. Virol. 2019, 164, 2315–2320. [Google Scholar] [CrossRef]
- Vaïsänen, E.; Paloniemi, M.; Kuisma, I.; Lithovius, V.; Kumar, A.; Franssila, R.; Ahmed, K.; Delwart, E.; Vesikari, T.; Hedman, K.; et al. Epidemiology of two human protoparvoviruses, bufavirus and tusavirus. Sci. Rep. 2016, 6, 39267. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Zhang, S.; Huang, H.; Wang, W.; Cao, L.; Zheng, M.; Yin, Y.; Zhang, H.; Lu, H.; Jin, N. First identification of a novel parvovirus distantly related to human bufavirus from diarrheal dogs in China. Virus Res. 2019, 19, 30048–30056. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, M.; Orba, Y.; Anindita, P.D.; Ishii, A.; Ueno, K.; Hang’Ombe, B.M.; Mweene, A.S.; Ito, K.; Sawa, H. Distinct lineages of bufavirus in wild shrews and nonhuman primates. Emerg. Infect. Dis. 2015, 21, 1230–1233. [Google Scholar] [CrossRef] [PubMed]
- Siqueira, J.D.; Ng, T.F.; Miller, M.; Li, L.; Deng, X.; Dodd, E.; Batac, F.; Delwart, E. Endemic infection of stranded southern sea otters (Enhydra lutris nereis) with novel parvovirus, poluomavirus, and adenovirus. J. Wildl. Dis. 2017, 53, 532–542. [Google Scholar] [CrossRef]
- Cortez, V.; Meliopoulos, V.A.; Karlsson, E.A.; Hargest, V.; Johnson, C.; Schultz-Cherry, S. Astrovirus biology and pathogenesis. Annu. Rev. Virol. 2017, 4, 327–348. [Google Scholar] [CrossRef] [PubMed]
- ICTV-International Commettee on Taxonomy of Viruses. Available online: https://talk.ictvonline.org/ictv-reports/ictv_9th_report/positive-sense-rna-viruses-2011/w/posrna_viruses/247/astroviridae (accessed on 27 August 2019).
- Burrell, C.J.; Howard, C.; Murphy, R.F.A. Astroviruses. In Fenner and White’s Medical Virology, 5th ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 473–476. [Google Scholar]
- Harbour, D.A.; Ashley, C.R.; Williams, P.D.; Gruffydd-Jones, T.J. Natural and experimental astrovirus infection of cats. Vet. Rec. 1987, 120, 555–557. [Google Scholar] [CrossRef]
- Yi, S.; Niu, J.; Wang, H.; Dong, G.; Guo, Y.; Dong, H.; Wang, K.; Hu, G. Molecular characterization of feline astrovirus in domestic cats from Northeast China. PLoS ONE 2018, 13, e0205441. [Google Scholar] [CrossRef]
- Bodewes, R.; van der Giessen, J.; Haagmans, B.L.; Osterhaus, A.D.M.E.; Smits, S.L. Identification of multiple novel viruses, including a parvovirus and a hepevirus, in feces of red foxes. J. Virol. 2013, 87, 7758–7764. [Google Scholar] [CrossRef]
- Lawler, P.E.; Cook, K.A.; Williams, H.G.; Archer, L.L.; Schaedel, K.E.; Isaza, N.M.; Wellehan, J.F.X., Jr. Determination of the diversity of astroviruses in feces from cats in Florida. J. Vet. Diagn. Investig. 2018, 30, 275–279. [Google Scholar] [CrossRef]
- Moschidou, P.; Martella, V.; Lorusso, E.; Desario, C.; Pinto, P.; Losurdo, M.; Catella, C.; Parisi, A.; Bányai, K.; Buonavoglia, C. Mixed infection by Feline astrovirus and Feline panleukopenia virus in a domestic cat with gastroenteritis and panleukopenia. J. Vet. Diagn. Investig. 2011, 23, 581–584. [Google Scholar] [CrossRef]
- Lau, S.K.; Woo, P.C.; Yip, C.C.; Bai, R.; Wu, Y.; Tse, H.; Yuen, K.Y. Complete genome sequence of a novel feline astrovirus from a domestic cat in Hong Kong. Genome Announc. 2013, 1, e00708-13. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.Y.; Lim, S.I.; Kim, Y.K.; Song, J.Y.; Lee, J.B.; An, D.J. Molecular characterization and phylogenetic analysis of feline astrovirus in Korean cats. J. Feline Med. Surg. 2014, 16, 679–683. [Google Scholar] [CrossRef] [PubMed]
- Sabshin, S.J.; Levy, J.K.; Tupler, T.; Tucker, S.J.; Greiner, E.C.; Leutenegger, C.M. Enteropathogens identified in cats entering a Florida animal shelter with normal feces or diarrhea. J. Am. Vet. Med. Assoc. 2012, 241, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Bishop, R.F.; Davidson, G.P.; Holmes, I.H.; Ruck, B.J. Virus particles in epithelial cells of duodenal mucosa from children with acute non-bacterial gastroenteritis. Lancet 1973, 2, 1281–1283. [Google Scholar] [CrossRef]
- Bányai, K.; Estes, M.K.; Martella, V.; Parashar, U.D. Viral gastroenteritis. Lancet 2018, 392, 175–186. [Google Scholar] [CrossRef]
- ICTV-International Commettee on Taxonomy of Viruses. Available online: https://talk.ictvonline.org/ictvreports/ictv_9th_report/dsrnaviruses2011/w/dsrna_viruses/188/reoviridae (accessed on 27 August 2019).
- Bányai, K.; Kemenesi, G.; Budinski, I.; Földes, F.; Zana, B.; Marton, S.; Varga-Kugler, R.; Oldal, M.; Kurucz, K.; Jakab, F. Candidate new rotavirus species in Schreiber’s bats, Serbia. Infect. Genet. Evol. 2017, 48, 19–26. [Google Scholar] [CrossRef]
- Estes, M.K.; Kapikian, A.Z. Rotaviruses. In Fields Virology, 5th ed.; Knipe, D.M., Howley, P.M., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007; pp. 1918–1974. [Google Scholar]
- Matthijnssens, J.; Ciarlet, M.; McDonald, S.M.; Attoui, H.; Bányai, K.; Brister, J.R.; Buesa, J.; Esona, M.D.; Estes, M.K.; Gentsch, J.R.; et al. Uniformity of rotavirus strain nomenclature proposed by the rotavirus classification working group (RCWG). Arch. Virol. 2011, 156, 1397–1413. [Google Scholar] [CrossRef]
- Yen, C.; Cortese, M.M. Rotaviruses. In Principles and Practice of Pediatric Infectious Diseases; Long, S.S., Prober, C.G., Fischer, M., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 1122–1125. [Google Scholar]
- McNulty, M.S.; Allan, G.M.; Thompson, D.J.; O’Boyle, J.D. Antibody to rotavirus in dogs and cats. Vet. Rec. 1978, 102, 534–535. [Google Scholar] [CrossRef]
- Snodgrass, D.R.; Angus, K.W.; Gray, E.W. A rotavirus from kittens. Vet. Rec. 1979, 104, 222–223. [Google Scholar] [CrossRef]
- Hoshino, Y.; Baldwin, C.A.; Scott, F.W. Isolation and characterization of feline rotavirus. J. Gen. Virol. 1981, 54, 313–323. [Google Scholar] [CrossRef]
- Mochizuki, M.; Nakagomi, T.; Nakagomi, O. Isolation from diarrheal and asymptomatic kittens of three rotavirus strains that belong to the AU-1 genogroup of human rotaviruses. J. Clin. Microbiol. 1997, 35, 1272–1275. [Google Scholar]
- Yamaguchi, N.; Macdonald, D.W.; Passanisi, W.C.; Harbour, D.A.; Hopper, C.D. Parasite prevalence in free-ranging farm cats, Felis silvestris Catus. Epidemiol. Infect. 1996, 116, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Birch, C.J.; Heath, R.L.; Marshall, J.A.; Liut, S.; Gust, I.D. Isolation of feline rotaviruses and their relationship to human and simian isolates by electropherotype and serotype. J. Gen. Virol. 1985, 66, 2731–2735. [Google Scholar] [CrossRef] [PubMed]
- German, A.C.; Iturriza-Gómara, M.; Dove, W.; Sandrasegaram, M.; Nakagomi, T.; Nakagomi, O.; Cunliffe, N.; Radford, A.D.; Morgan, K.L. Molecular epidemiology of rotavirus in cats in the United Kingdom. J. Clin. Microbiol. 2015, 53, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Mihalov-Kovács, E.; Gellért, Á.; Marton, S.; Farkas, S.L.; Fehér, E.; Oldal, M.; Jakab, F.; Martella, V.; Bányai, K. Candidate new rotavirus species in sheltered dogs, Hungary. Emerg. Infect. Dis. 2015, 21, 660–663. [Google Scholar] [CrossRef]
- ICTV-International Commettee on Taxonomy of Viruses. Available online: https://talk.ictvonline.org/ictv-reports/ictv_online_report/ssdna-viruses/w/circoviridae (accessed on 2 September 2019).
- Kapoor, A.; Dubovi, E.J.; Henriquez-Rivera, J.A.; Lipkin, W.I. Complete genome sequence of the first canine circovirus. J. Virol. 2012, 86, 7018. [Google Scholar] [CrossRef]
- Zaccaria, G.; Malatesta, D.; Scipioni, G.; Di Felice, E.; Campolo, M.; Casaccia, C.; Savini, G.; Di Sabatino, D.; Lorusso, A. Circovirus in domestic and wild carnivores: An important opportunistic agent? Virology 2016, 490, 69–74. [Google Scholar] [CrossRef]
- Li, L.; McGraw, S.; Zhu, K.; Leutenegger, C.M.; Marks, S.L.; Kubiski, S.; Gaffney, P.; Dela Cruz, F.N.; Wang, C.; Delwart, E.; et al. Circovirus in tissues of dogs with vasculitis and hemorrhage. Emerg. Infect. Dis. 2013, 19, 534–541. [Google Scholar] [CrossRef]
- Decaro, N.; Martella, V.; Desario, C.; Lanave, G.; Circella, E.; Cavalli, A.; Elia, G.; Camero, M.; Buonavoglia, C. Genomic characterization of a circovirus associated with fatal hemorrhagic enteritis in dog, Italy. PLoS ONE 2014, 22, e105909. [Google Scholar] [CrossRef]
- Johne, R.; Buck, C.B.; Allander, T.; Atwood, W.J.; Garcea, R.L.; Imperiale, M.J.; Major, E.O.; Ramqvist, T.; Norkin, L.C. Taxonomical developments in the family Polyomaviridae. Arch. Virol. 2011, 156, 1627–1634. [Google Scholar] [CrossRef]
- Peretti, A.; FitzGerald, P.C.; Bliskovsky, V.; Pastrana, D.V.; Buck, C.B. Genome Sequence of a Fish-Associated Polyomavirus, Black Sea Bass (Centropristis striata) Polyomavirus 1. Genome Announc. 2015, 3, e01476-14. [Google Scholar] [CrossRef]
- Gheit, T.; Dutta, S.; Oliver, J.; Robitaille, A.; Hampras, S.; Combes, J.D.; McKayChopin, S.; Le Calvez-Kelm, F.; Fenske, N.; Cherpelis, B.; et al. Isolation and characterization of a novel putative human polyomavirus. Virology 2017, 506, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Caddy, S.; Breiman, A.; le Pendu, J.; Goodfellow, I. Genogroup IV and VI canine noroviruses interact with histo-blood group antigens. J. Virol. 2014, 88, 10377–10391. [Google Scholar] [CrossRef] [PubMed]
- De Grazia, S.; Giammanco, G.M.; Martella, V.; Ramirez, S.; Colomba, C.; Cascio, A.; Arista, S. Rare AU-1-like G3P [9] human rotaviruses with a Kun-like NSP4 gene detected in children with diarrhea in Italy. J. Clin. Microbiol. 2008, 46, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Pietsch, P.; Liebert, U.G. Evidence for presumable feline origin of sporadic G6P [9] rotaviruses in humans. Infect. Genet. Evol. 2018, 63, 180–194. [Google Scholar] [CrossRef] [PubMed]
- Mesquita, J.R.; Costantini, V.P.; Cannon, J.L.; Lin, S.C.; Nascimento, M.S.; Vinjé, J. Presence of antibodies against genogroup VI norovirus in humans. Virol. J. 2013, 10, 176. [Google Scholar] [CrossRef] [PubMed]
- Martella, V.; Pinto, P.; Lorusso, E.; Di Martino, B.; Wang, Q.; Larocca, V.; Cavalli, A.; Camero, M.; Decaro, N.; Bányai, K.; et al. Detection and Full-Length Genome Characterization of Novel Canine Vesiviruses. Emerg. Infect. Dis. 2015, 21, 1433–1436. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, B.; Di Profio, F.; Lanave, G.; De Grazia, S.; Giammanco, G.M.; Lavazza, A.; Buonavoglia, C.; Marsilio, F.; Bányai, K.; Martella, V. Antibodies for strain 2117-like vesiviruses (caliciviruses) in humans. Virus Res. 2015, 210, 279–282. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, B.; Di Profio, F.; Melegari, I.; Sarchese, V.; Massirio, I.; Luciani, A.; Lanave, G.; Marsilio, F.; Martella, V. Serological and molecular investigation of 2117-like vesiviruses in cats. Arch. Virol. 2018, 163, 197–201. [Google Scholar] [CrossRef]
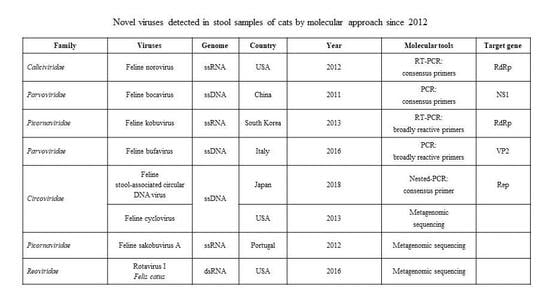
| Family | Viruses | Genome | Country | Year | Molecular Tools | Target Gene | References |
|---|---|---|---|---|---|---|---|
| Caliciviridae | Feline norovirus | ssRNA | USA | 2012 | RT-PCR: consensus primers | RdRp | [11] |
| Parvoviridae | Feline bocavirus | ssDNA | China | 2011 | PCR: consensus primers | NS1 | [12] |
| Picornaviridae | Feline kobuvirus | ssRNA | South Korea | 2013 | RT-PCR: broadly reactive primers | RdRp | [13] |
| Parvoviridae | Feline bufavirus | ssDNA | Italy | 2016 | PCR: broadly reactive primers | VP2 | [14] |
| Circoviridae | Feline stool-associated circular DNA virus | ssDNA | Japan | 2018 | Nested-PCR: consensus primer | Rep | [15] |
| Feline cyclovirus | USA | 2013 | Metagenomic sequencing | [17] | |||
| Picornaviridae | Feline sakobuvirus A | ssRNA | Portugal | 2012 | Metagenomic sequencing | [16] | |
| Reoviridae | Rotavirus I Felis catus | dsRNA | USA | 2016 | Metagenomic sequencing | [18] | |
| Polyomaviridae | Lyon-IARC polyomavirus | dsDNA | Canada | 2019 | Metagenomic sequencing | [19] |
| Primer | Sequence (5′ to 3′) | Sense | Target | References |
|---|---|---|---|---|
| p289 p290 | TGACAATGTAATCATCACCATA GATTACTCCAAGTGGGACTCCAC | + - | RdRp | [54] |
| JV12Y JV13I | ATACCACCTATGATGCAGAYTA TCATCATCACCATAGAAGAG | + - | RdRp | [55] |
| FNoV-F9d FNoV-R15 | GCCCACTGGATWTACACCCTCTC CTGATGGTTGGGTCCTCTGGTCCA | + - | RdRp | [11] |
| FNoV-F9d FNoV-R14d | GCCCACTGGATWTACACCCTCTC CYTGGT RTACCCAAACTCCA C | + - | ORF2 | [11] |
| QT | CCAGTGAGCAGAGTGACGAGGACTCGAGCTCAAGC (T17) | +/- | 3′/5′end | [56] |
| Country | Samples Tested | Positive Samples | References | ||||
|---|---|---|---|---|---|---|---|
| Total | Diarrhoeic | Non Diarrhoeic | Total | Diarrhoeic | Non Diarrhoeic | ||
| Korea | 39 | 39 | 0 | 6 (15.4%) | 6 (15.4%) | 0 | [13] |
| Korea | 71 | 52 | 19 | 17 (23.9%) | 15 (28.8%) | 2 (10.5%) | [90] |
| Italy | 83 | 37 | 46 | 5 (6.0%) | 5 (13.5%) | 0 (0.0%) | [85] |
| China | 81 | 52 | 29 | 8 (9.9%) | 8 (15.4%) | 0 (0.0%) | [86] |
| China | 197 | 105 | 92 | 28 (14.2%) | 20 (19.1%) | 8 (8.7%) | [87] |
| Genus | Species | Common Names Used in Literature | Country | Year | Detection Source | References |
|---|---|---|---|---|---|---|
| Bocaparvovirus | Carnivore bocaparvovirus 3 | Feline bocaparvovirus (FBoV) | China | 2011 | Stools, urine, kidney, blood and respiratory samples | [12] |
| Carnivore bocaparvovirus 4 | FBoV2 | Portugal | 2012 | Stools | [16] | |
| Carnivore bocaparvovirus 5 | FBoV3 | USA | 2013 | Stools | [17] | |
| Protoparvovirus | Carnivore protoparvovirus 2* | Feline bufavirus (FBuV) | Italy | 2017 | Stools and respiratory samples | [14] |
| Genus/Species | Primer | Nucleotide Sequence (5′to 3′) | Lenght (bp) | Target | References |
|---|---|---|---|---|---|
| BoVs | BoVF | GCCAGCACNGGNAARACMAA | 141 | NS1 | [12] |
| BoVR | CATNAGNCAYTCYTCCCACCA | ||||
| FBoV | FBoV1F | TCTACAAGTGGGACATTGGA | 133 | NS1 | [12] |
| FBoV1R | GAGCTTGATTGCATTCACGA | ||||
| FBoV2 | FBoV2F | TCGTTCGTCTTGGAACATAGC | 301 | NS1 | [16] |
| FBoV2R | CAGAGCGTGGATCTGTCTGA | ||||
| FBoV3 | FBD1L1 | TGACTCGTCTGTGGCGGGCT | 546 | VP1 | [17] |
| FBD1R1 | TCGTTCGTGAGACGCTGCCA | ||||
| FBD1L2 | CAAAGGATCGGGAGCGGGCG | 388 | VP1 | [17] | |
| FBD1R2 | TGCCCATGGTGTTGTGATTCCTATCCA |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Martino, B.; Di Profio, F.; Melegari, I.; Marsilio, F. Feline Virome—A Review of Novel Enteric Viruses Detected in Cats. Viruses 2019, 11, 908. https://doi.org/10.3390/v11100908
Di Martino B, Di Profio F, Melegari I, Marsilio F. Feline Virome—A Review of Novel Enteric Viruses Detected in Cats. Viruses. 2019; 11(10):908. https://doi.org/10.3390/v11100908
Chicago/Turabian StyleDi Martino, Barbara, Federica Di Profio, Irene Melegari, and Fulvio Marsilio. 2019. "Feline Virome—A Review of Novel Enteric Viruses Detected in Cats" Viruses 11, no. 10: 908. https://doi.org/10.3390/v11100908
APA StyleDi Martino, B., Di Profio, F., Melegari, I., & Marsilio, F. (2019). Feline Virome—A Review of Novel Enteric Viruses Detected in Cats. Viruses, 11(10), 908. https://doi.org/10.3390/v11100908