The Incorporation of Host Proteins into the External HIV-1 Envelope
Abstract
1. Introduction
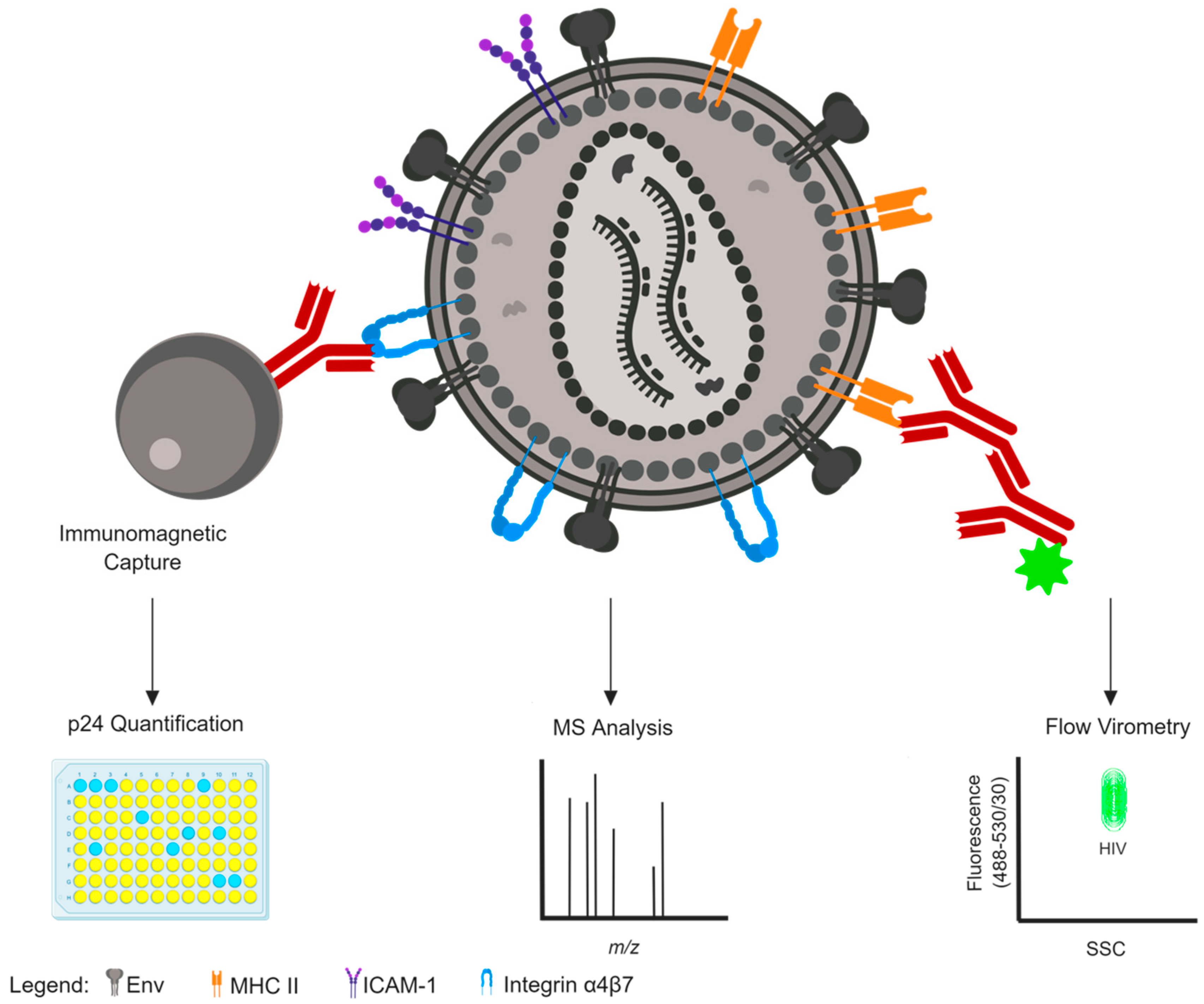
2. Methods to Identify Host Protein Incorporation
2.1. Mass Spectrometry
2.2. Immunomagnetic Capture
2.3. Nanoscale Flow Cytometry and Flow Virometry
3. Host Proteins Incorporated on Virion Surfaces
3.1. Major Histocompatibility Complex (MHC)
3.2. ICAM-1 and LFA-1
3.3. Integrin α4β7
3.4. Complement Proteins
3.5. Other Molecules
4. Mechanisms of Incorporation
4.1. Budding Through Lipid Rafts
4.2. Assembly Within Exosomes
4.3. Mechanisms Involving Cytoskeletal Proteins
4.4. Mechanisms of MHC and ICAM-1 Incorporation
5. Effects of Host Protein Incorporation on Viral Pathogenesis
5.1. ICAM-1
5.2. Other Adhesion Proteins and Integrins
5.3. Major Histocompatibility Complex (MHC)
6. Novel Targets and Treatments
6.1. Therapeutic Targeting of Integrin α4β7
6.2. Therapeutic Targeting of MHC Proteins
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chandran, K. Endosomal Proteolysis of the Ebola Virus Glycoprotein Is Necessary for Infection. Science 2005, 308, 1643–1645. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.J.; Vijaykrishna, D.; Bahl, J.; Lycett, S.J.; Worobey, M.; Pybus, O.G.; Ma, S.K.; Cheung, C.L.; Raghwani, J.; Bhatt, S. Origins and evolutionary genomics of the 2009 swine-origin H1N1 influenza A epidemic. Nature 2009, 459, 1122. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.C.; Fass, D.; Berger, J.M.; Kim, P.S. Core Structure of gp41 from the HIV Envelope Glycoprotein. Cell 1997, 89, 263–273. [Google Scholar] [CrossRef]
- Chen, B.J.; Lamb, R.A. Mechanisms for enveloped virus budding: Can some viruses do without an ESCRT? Virology 2008, 372, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Garoff, H.; Hewson, R.; Opstelten, D.-J.E. Virus maturation by budding. Microbiol. Mol. Biol. Rev. 1998, 62, 1171–1190. [Google Scholar] [PubMed]
- Schols, D.; Pauwels, R.; Desmyter, J.; De Clercq, E. Dextran sulfate and other polyanionic anti-HIV compounds specifically interact with the viral gp120 glycoprotein expressed by T-cells persistently infected with HIV-1. Virology 1990, 175, 556–561. [Google Scholar] [CrossRef]
- Novikoff, A.B.; Beard, D.; Beard, J.W. Electron Microscopic Study of the Atpase Activity of the Bai Strain a (Myeloblastosis) Avian Tumor Virus. J. Cell Biol. 1962, 15, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Aoki, T.; Stephenson, J.R.; Aaronson, S.A. Demonstration of a Cell-Surface Antigen Associated with Murine Sarcoma Virus by Immunoelectron Microscopy. Proc. Natl. Acad. Sci. USA 1973, 70, 742–746. [Google Scholar] [CrossRef]
- De-Thé, G. Cytoplasmic Microtubules in Different Animal Cells. J. Cell Biol. 1964, 23, 265–275. [Google Scholar] [CrossRef]
- Azocar, J.; Essex, M. Incorporation of HLA antigens into the envelope of RNA tumor viruses grown in human cells. Cancer Res. 1979, 39, 3388–3391. [Google Scholar]
- Orentas, R.J.; Hildreth, J.E.K. Association of Host Cell Surface Adhesion Receptors and Other Membrane Proteins with HIV and SIV. AIDS Res. Hum. Retrovir. 1993, 9, 1157–1165. [Google Scholar] [CrossRef] [PubMed]
- Hoxie, J.A.; Fitzharris, T.P.; Youngbar, P.R.; Matthews, D.M.; Rackowski, J.L.; Radka, S.F. Nonrandom association of cellular antigens with HTLV-III virions. Hum. Immunol. 1987, 18, 39–52. [Google Scholar] [CrossRef]
- Meerloo, T.; Parmentier, H.K.; Osterhaus, A.D.M.E.; Goudsmit, J.; Schuurman, H.-J. Modulation of cell surface molecules during HIV-1 infection of H9 cells. An immunoelectron microscopic study. AIDS 1992, 6, 1105–1116. [Google Scholar] [CrossRef] [PubMed]
- Meerloo, T.; Sheikh, M.A.; Bloem, A.C.; de Ronde, A.; Schutten, M.; van Els, C.A.C.; Roholl, P.J.M.; Joling, P.; Goudsmit, J.; Schuurman, H.-J. Host cell membrane proteins on human immunodeficiency virus type 1 after in vitro infection of H9 cells and blood mononuclear cells. An immuno-electron microscopic study. J. Gen. Virol. 1993, 74, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Arthur, L.O.; Bess, J.W.; Sowder, R.C.; Benveniste, R.E.; Mann, D.L.; Chermann, J.-C.; Henderson, L.E. Cellular proteins bound to immunodeficiency viruses: Implications for pathogenesis and vaccines. Science 1992, 258, 1935–1938. [Google Scholar] [CrossRef] [PubMed]
- Ott, D.E. Cellular proteins in HIV virions. Rev. Med. Virol. 1997, 7, 167–180. [Google Scholar] [CrossRef]
- Tremblay, M.J.; Fortin, J.-F.; Cantin, R. The acquisition of host-encoded proteins by nascent HIV-1. Immunol. Today 1998, 19, 346–351. [Google Scholar] [CrossRef]
- Guzzo, C.; Ichikawa, D.; Park, C.; Phillips, D.; Liu, Q.; Zhang, P.; Kwon, A.; Miao, H.; Lu, J.; Rehm, C.; et al. Virion incorporation of integrin α4β7 facilitates HIV-1 infection and intestinal homing. Sci. Immunol. 2017, 2, eaam7341. [Google Scholar] [CrossRef]
- Fortin, J.-F.; Cantin, R.; Lamontagne, G.; Tremblay, M. Host-derived ICAM-1 glycoproteins incorporated on human immunodeficiency virus type 1 are biologically active and enhance viral infectivity. J. Virol. 1997, 71, 3588–3596. [Google Scholar]
- Byrareddy, S.N.; Kallam, B.; Arthos, J.; Cicala, C.; Nawaz, F.; Hiatt, J.; Kersh, E.N.; McNicholl, J.M.; Hanson, D.; Reimann, K.A.; et al. Targeting α4β7 integrin reduces mucosal transmission of SIV and protects GALT from infection. Nat. Med. 2014, 20, 1397–1400. [Google Scholar] [CrossRef]
- Ansari, A.A.; Reimann, K.A.; Mayne, A.E.; Takahashi, Y.; Stephenson, S.T.; Wang, R.; Wang, X.; Li, J.; Price, A.A.; Little, D.M.; et al. Blocking of α4β7 Gut-Homing Integrin during Acute Infection Leads to Decreased Plasma and Gastrointestinal Tissue Viral Loads in Simian Immunodeficiency Virus-Infected Rhesus Macaques. J. Immunol. 2011, 186, 1044–1059. [Google Scholar] [CrossRef] [PubMed]
- Byrareddy, S.N.; Arthos, J.; Cicala, C.; Villinger, F.; Ortiz, K.T.; Little, D.; Sidell, N.; Kane, M.A.; Yu, J.; Jones, J.W.; et al. Sustained virologic control in SIV+ macaques after antiretroviral and α4β7 antibody therapy. Science 2016, 354, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Rizzuto, C.D.; Sodroski, J.G. Contribution of virion ICAM-1 to human immunodeficiency virus infectivity and sensitivity to neutralization. J. Virol. 1997, 71, 4847–4851. [Google Scholar] [PubMed]
- Chung, C.-S.; Chen, C.-H.; Ho, M.-Y.; Huang, C.-Y.; Liao, C.-L.; Chang, W. Vaccinia Virus Proteome: Identification of Proteins in Vaccinia Virus Intracellular Mature Virion Particles. J. Virol. 2006, 80, 2127–2140. [Google Scholar] [CrossRef]
- Resch, W.; Hixson, K.K.; Moore, R.J.; Lipton, M.S.; Moss, B. Protein composition of the vaccinia virus mature virion. Virology 2007, 358, 233–247. [Google Scholar] [CrossRef]
- Yoder, J.D.; Chen, T.S.; Gagnier, C.R.; Vemulapalli, S.; Maier, C.S.; Hruby, D.E. Pox proteomics: Mass spectrometry analysis and identification of Vaccinia virion proteins. Virol. J. 2006, 3, 10. [Google Scholar] [CrossRef]
- Stegen, C.; Yakova, Y.; Henaff, D.; Nadjar, J.; Duron, J.; Lippé, R. Analysis of Virion-Incorporated Host Proteins Required for Herpes Simplex Virus Type 1 Infection through a RNA Interference Screen. PLoS ONE 2013, 8, e53276. [Google Scholar] [CrossRef] [PubMed]
- Chertova, E.; Chertov, O.; Coren, L.V.; Roser, J.D.; Trubey, C.M.; Bess, J.W.; Sowder, R.C.; Barsov, E.; Hood, B.L.; Fisher, R.J. Proteomic and biochemical analysis of purified human immunodeficiency virus type 1 produced from infected monocyte-derived macrophages. J. Virol. 2006, 80, 9039–9052. [Google Scholar] [CrossRef]
- Jalaguier, P.; Cantin, R.; Maaroufi, H.; Tremblay, M.J. Selective Acquisition of Host-Derived ICAM-1 by HIV-1 Is a Matrix-Dependent Process. J. Virol. 2015, 89, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Bos, L. Beijerinck’s work on tobacco mosaic virus: Historical context and legacy. Philos. Trans. R. Soc. B Biol. Sci. 1999, 354, 675. [Google Scholar] [CrossRef] [PubMed]
- Roingeard, P. Viral detection by electron microscopy: Past, present and future. Biol. Cell 2008, 100, 491–501. [Google Scholar] [CrossRef]
- Goldsmith, C.S.; Miller, S.E. Modern Uses of Electron Microscopy for Detection of Viruses. Clin. Microbiol. Rev. 2009, 22, 552–563. [Google Scholar] [CrossRef] [PubMed]
- Morales-Kastresana, A.; Telford, B.; Musich, T.A.; McKinnon, K.; Clayborne, C.; Braig, Z.; Rosner, A.; Demberg, T.; Watson, D.C.; Karpova, T.S. Labeling extracellular vesicles for nanoscale flow cytometry. Sci. Rep. 2017, 7, 1878. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Bartesaghi, A.; Borgnia, M.J.; Sapiro, G.; Subramaniam, S. Molecular architecture of native HIV-1 gp120 trimers. Nature 2008, 455, 109. [Google Scholar] [CrossRef]
- Julien, J.-P.; Cupo, A.; Sok, D.; Stanfield, R.L.; Lyumkis, D.; Deller, M.C.; Klasse, P.-J.; Burton, D.R.; Sanders, R.W.; Moore, J.P.; et al. Crystal structure of a soluble cleaved HIV-1 envelope trimer. Science 2013, 342. [Google Scholar] [CrossRef] [PubMed]
- Shafique, S. Envelope Protein Structure of Zika Virus—A Target For Vaccine Development And Therapeutics. Timely Top. Clin. Immunol. 2017, 1, 1–4. [Google Scholar]
- Ingale, A.G. Epitopes Identification for Vaccine Design and Structural Aspects of Dengue Virus 3 Envelope Protein. Biochem. Physiol. Open Access 2014, 3, 134. [Google Scholar] [CrossRef]
- Pejchal, R.; Wilson, I.A. Structure-based vaccine design in HIV: Blind men and the elephant? Curr. Pharm. Des. 2010, 16, 3744–3753. [Google Scholar] [CrossRef] [PubMed]
- Shirato, K.; Miyoshi, H.; Goto, A.; Ako, Y.; Ueki, T.; Kariwa, H.; Takashima, I. Viral envelope protein glycosylation is a molecular determinant of the neuroinvasiveness of the New York strain of West Nile virus. J. Gen. Virol. 2004, 85, 3637–3645. [Google Scholar] [CrossRef]
- Domon, B.; Aebersold, R. Mass spectrometry and protein analysis. Science 2006, 312, 212–217. [Google Scholar] [CrossRef]
- Aebersold, R.; Mann, M. Mass spectrometry-based proteomics. Nature 2003, 422, 198. [Google Scholar] [CrossRef] [PubMed]
- Link, A.J.; Eng, J.; Schieltz, D.M.; Carmack, E.; Mize, G.J.; Morris, D.R.; Garvik, B.M.; Yates, J.R. Direct analysis of protein complexes using mass spectrometry. Nat. Biotechnol. 1999, 17, 676. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Sugrue, R.J.; Tang, K. Mass spectrometry based proteomic studies on viruses and hosts—A review. Anal. Chim. Acta 2011, 702, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Ott, D.E. Potential roles of cellular proteins in HIV-1. Rev. Med. Virol. 2002, 12, 359–374. [Google Scholar] [CrossRef] [PubMed]
- Cross, T.G.; Hornshaw, M.P. Can LC and LC-MS ever replace immunoassays? J. Appl. Bioanal. 2016, 2, 108–116. [Google Scholar] [CrossRef]
- Peeters, B.; Herijgers, P.; Beuselinck, K.; Peetermans, W.E.; Herregods, M.-C.; Desmet, S.; Lagrou, K. Comparison of PCR-Electrospray Ionization Mass Spectrometry with 16S rRNA PCR and Amplicon Sequencing for Detection of Bacteria in Excised Heart Valves. J. Clin. Microbiol. 2016, 54, 2825–2831. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Holzgreve, W.; Kiefer, V.; Hahn, S. MALDI-TOF Mass Spectrometry Compared with Real-Time PCR for Detection of Fetal Cell-Free DNA in Maternal Plasma. Clin. Chem. 2006, 52, 2311–2312. [Google Scholar] [CrossRef] [PubMed]
- Loret, S.; Guay, G.; Lippe, R. Comprehensive Characterization of Extracellular Herpes Simplex Virus Type 1 Virions. J. Virol. 2008, 82, 8605–8618. [Google Scholar] [CrossRef]
- Shaw, M.L.; Stone, K.L.; Colangelo, C.M.; Gulcicek, E.E.; Palese, P. Cellular Proteins in Influenza Virus Particles. PLoS Pathog. 2008, 4, e1000085. [Google Scholar] [CrossRef]
- Saphire, A.C.S.; Gallay, P.A.; Bark, S.J. Proteomic Analysis of Human Immunodeficiency Virus Using Liquid Chromatography/Tandem Mass Spectrometry Effectively Distinguishes Specific Incorporated Host Proteins. J. Proteome Res. 2006, 5, 530–538. [Google Scholar] [CrossRef]
- Kannagi, M.; Kiyotaki, M.; King, N.W.; Lord, C.I.; Letvin, N.L. Simian Immunodeficiency Virus Induces Expression of Class II Major Histocompatibility Complex Structures on Infected Target Cells In Vitro. J. Virol. 1987, 61, 6. [Google Scholar]
- Frank, I.; Stoiber, H.; Godar, S.; Stockinger, H.; Steindl, F.; Katinger, H.W.D.; Dierich, M.P. Acquisition of host cell-surface-derived molecules by HIV-1. AIDS 1996, 10, 1611–1620. [Google Scholar] [CrossRef] [PubMed]
- Montefiori, D.C.; Cornell, R.J.; Zhou, J.Y.; Zhou, J.T.; Hirsch, V.M.; Johnson, P.R. Complement control proteins, CD46, CD55, and CD59, as common surface constituents of human and simian immunodeficiency viruses and possible targets for vaccine protection. Virology 1994, 205, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Cantin, R.; Fortin, J.-F.; Tremblay, M. The Amount of Host HLA-DR Proteins Acquired by HIV-1 Is Virus Strain- and Cell Type-Specific. Virology 1996, 218, 372–381. [Google Scholar] [CrossRef] [PubMed]
- Capobianchi, M.R.; Fais, S.; Castilletti, C.; Gentile, M.; Ameglio, F.; Dianzani, F. A Simple and Reliable Method to Detect Cell Membrane Proteins on Infectious Human Immunodeficiency Virus Type 1 Particles. J. Infect. Dis. 1994, 169, 886–889. [Google Scholar] [CrossRef] [PubMed]
- Lawn, S.D.; Roberts, B.D.; Griffin, G.E.; Folks, T.M.; Butera, S.T. Cellular Compartments of Human Immunodeficiency Virus Type 1 Replication In Vivo: Determination by Presence of Virion-Associated Host Proteins and Impact of Opportunistic Infection. J. Virol. 2000, 74, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Bounou, S.; Giguère, J.-F.; Cantin, R.; Gilbert, C.; Imbeault, M.; Martin, G.; Tremblay, M.J. The importance of virus-associated host ICAM-1 in human immunodeficiency virus type 1 dissemination depends on the cellular context. FASEB J. 2004, 18, 1294–1296. [Google Scholar] [CrossRef]
- Esser, M.T.; Graham, D.R.; Coren, L.V.; Trubey, C.M.; Bess, J.W.; Arthur, L.O.; Ott, D.E.; Lifson, J.D. Differential Incorporation of CD45, CD80 (B7-1), CD86 (B7-2), and Major Histocompatibility Complex Class I and II Molecules into Human Immunodeficiency Virus Type 1 Virions and Microvesicles: Implications for Viral Pathogenesis and Immune Regulation. J. Virol. 2001, 75, 6173–6182. [Google Scholar] [CrossRef]
- Linde, M.E.; Colquhoun, D.R.; Mohien, C.U.; Kole, T.; Aquino, V.; Cotter, R.; Edwards, N.; Hildreth, J.E.K.; Graham, D.R. The Conserved Set of Host Proteins Incorporated into HIV-1 Virions Suggests a Common Egress Pathway in Multiple Cell Types. J. Proteome Res. 2013, 12, 2045–2054. [Google Scholar] [CrossRef]
- Bess, J.W., Jr.; Gorelick, R.J.; Bosche, W.J.; Henderson, L.E.; Arthur, L.O. Microvesicles are a source of contaminating cellular proteins found in purified HIV-1 preparations. Virology 1997, 230, 134–144. [Google Scholar] [CrossRef]
- Martin, G.; Tremblay, M.J. HLA-DR, ICAM-1, CD40, CD40L, and CD86 are incorporated to a similar degree into clinical human immunodeficiency virus type 1 variants expanded in natural reservoirs such as peripheral blood mononuclear cells and human lymphoid tissue cultured ex vivo. Clin. Immunol. 2004, 111, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Frank, I.; Kacani, L.; Stoiber, H.; Steindl, F.; Romani, N.; Dierich, M.P. Human Immunodeficiency Virus Type 1 Derived from Cocultures of Immature Dendritic Cells with Autologous T Cells Carries T-Cell-Specific Molecules on Its Surface and Is Highly Infectious. J. Virol. 1999, 73, 6. [Google Scholar]
- Thibault, S.; Tardif, M.R.; Gilbert, C.; Tremblay, M.J. Virus-associated host CD62L increases attachment of human immunodeficiency virus type 1 to endothelial cells and enhances trans infection of CD4+ T lymphocytes. J. Gen. Virol. 2007, 88, 2568–2573. [Google Scholar] [CrossRef] [PubMed]
- Bastiani, L.; Laal, S.; Kim, M.; Zolla-Pazner, S. Host Cell-Dependent Alterations in Envelope Components of Human Immunodeficiency Virus Type 1 Virions. J. Virol. 1997, 71, 7. [Google Scholar]
- Macey, M.G.; Macey, M.G. Flow Cytometry; Springer: Berlin, Germany, 2007. [Google Scholar]
- Jaroszeski, M.J.; Radcliff, G. Fundamentals of flow cytometry. Mol. Biotechnol. 1999, 11, 37–53. [Google Scholar] [CrossRef] [PubMed]
- Betters, D.M. Use of flow cytometry in clinical practice. J. Adv. Pract. Oncol. 2015, 6, 435. [Google Scholar]
- Brown, M.; Wittwer, C. Flow cytometry: Principles and clinical applications in hematology. Clin. Chem. 2000, 46, 1221–1229. [Google Scholar]
- Steen, H.B. Flow cytometer for measurement of the light scattering of viral and other submicroscopic particles. Cytom. Part J. Int. Soc. Anal. Cytol. 2004, 57, 94–99. [Google Scholar] [CrossRef]
- Bonar, M.M.; Tilton, J.C. High sensitivity detection and sorting of infectious human immunodeficiency virus (HIV-1) particles by flow virometry. Virology 2017, 505, 80–90. [Google Scholar] [CrossRef]
- Hercher, M.; Mueller, W.; Shapiro, H.M. Detection and discrimination of individual viruses by flow cytometry. J. Histochem. Cytochem. 1979, 27, 350–352. [Google Scholar] [CrossRef]
- Marie, D.; Brussaard, C.P.D.; Thyrhaug, R.; Bratbak, G.; Vaulot, D. Enumeration of Marine Viruses in Culture and Natural Samples by Flow Cytometry. Appl. Environ. Microbiol. 1999, 65, 8. [Google Scholar]
- Brussaard, C.P.D.; Marie, D.; Bratbak, G. Flow cytometric detection of viruses. J. Virol. Methods 2000, 85, 175–182. [Google Scholar] [CrossRef]
- Brussaard, C.P.D. Optimization of Procedures for Counting Viruses by Flow Cytometry. Appl. Environ. Microbiol. 2004, 70, 1506–1513. [Google Scholar] [CrossRef] [PubMed]
- Arakelyan, A.; Fitzgerald, W.; Margolis, L.; Grivel, J.-C. Nanoparticle-based flow virometry for the analysis of individual virions. J. Clin. Investig. 2013, 123, 3716–3727. [Google Scholar] [CrossRef] [PubMed]
- Tang, V.A.; Renner, T.M.; Fritzsche, A.K.; Burger, D.; Langlois, M.-A. Single-Particle Discrimination of Retroviruses from Extracellular Vesicles by Nanoscale Flow Cytometry. Sci. Rep. 2017, 7, 17769. [Google Scholar] [CrossRef] [PubMed]
- Tang, V.A.; Renner, T.M.; Varette, O.; Le Boeuf, F.; Wang, J.; Diallo, J.-S.; Bell, J.C.; Langlois, M.-A. Single-particle characterization of oncolytic vaccinia virus by flow virometry. Vaccine 2016, 34, 5082–5089. [Google Scholar] [CrossRef] [PubMed]
- Reyes, J.L.Z.; Aguilar, H.C. Flow virometry as a tool to study viruses. Methods 2018, 134–135, 87–97. [Google Scholar]
- Lippé, R. Flow virometry: A powerful tool to functionally characterize viruses. J. Virol. 2018, 92, e01765-17. [Google Scholar] [CrossRef]
- Nolte, E.; Cremer, T.; Gallo, R.C.; Margolis, L.B. Extracellular vesicles and viruses: Are they close relatives? Proc. Natl. Acad. Sci. USA 2016, 113, 9155–9161. [Google Scholar] [CrossRef]
- Musich, T.; Jones, J.C.; Keele, B.F.; Jenkins, L.M.M.; Demberg, T.; Uldrick, T.S.; Yarchoan, R.; Robert-Guroff, M. Flow virometric sorting and analysis of HIV quasispecies from plasma. JCI Insight 2017, 2, e90626. [Google Scholar] [CrossRef]
- Gluschankof, P.; Mondor, I.; Gelderblom, H.R.; Sattentau, Q.J. Cell membrane vesicles are a major contaminant of gradient-enriched human immunodeficiency virus type-1 preparations. Virology 1997, 230, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Tang, V.A.; Fritzsche, A.K.; Renner, T.M.; Burger, D.; Brittain, G.C.; Lannigan, J.A.; Ouellet, C.; van der Pol, E.; Langlois, M.-A. Retroviruses as Fluorescent Reference Particles for Nanoscale Flow Cytometry. BioRxiv. (Manuscript in preparation).
- Van der Pol, E.; Coumans, F.A.W.; Sturk, A.; Nieuwland, R.; van Leeuwen, T.G. Refractive Index Determination of Nanoparticles in Suspension Using Nanoparticle Tracking Analysis. Nano Lett. 2014, 14, 6195–6201. [Google Scholar] [CrossRef] [PubMed]
- Chandler, W.L.; Yeung, W.; Tait, J.F. A new microparticle size calibration standard for use in measuring smaller microparticles using a new flow cytometer. J. Thromb. Haemost. 2011, 9, 1216–1224. [Google Scholar] [CrossRef] [PubMed]
- FCM PASS—Joshua A. Welsh. Available online: http://www.joshuawelsh.co.uk/scatter-diameter-software/ (accessed on 21 December 2018).
- Rosetta Calibration. Available online: https://www.exometry.com/products/rosetta-calibration (accessed on 21 December 2018).
- Boily, M.-C.; Baggaley, R.F.; Wang, L.; Masse, B.; White, R.G.; Hayes, R.J.; Alary, M. Heterosexual risk of HIV-1 infection per sexual act: Systematic review and meta-analysis of observational studies. Lancet Infect. Dis. 2009, 9, 118–129. [Google Scholar] [CrossRef]
- Saarloos, M.-N.; Sullivan, B.L.; Czerniewski, M.A.; Parameswar, K.D.; Spear, G.T. Detection of HLA-DR Associated with Monocytotropic, Primary, and Plasma Isolates of Human Immunodeficiency Virus Type. J. Virol. 1997, 71, 4. [Google Scholar]
- Cantin, R.; Martin, G.; Tremblay, M.J. A novel virus capture assay reveals a differential acquisition of host HLA-DR by clinical isolates of human immunodeficiency virus type 1 expanded in primary human cells depending on the nature of producing cells and the donor source. J. Gen. Virol. 2001, 82, 2979–2987. [Google Scholar] [CrossRef] [PubMed]
- Scheppler, J.A.; Nicholson, J.K.; Swan, D.C.; Ahmed-Ansari, A.; McDougal, J.S. Down-modulation of MHC-I in a CD4+ T cell line, CEM-E5, after HIV-1 infection. J. Immunol. 1989, 143, 2858–2866. [Google Scholar]
- Kerkau, T.; Bacik, I.; Bennink, J.R.; Yewdell, J.W.; Hünig, T.; Schimpl, A.; Schubert, U. The Human Immunodeficiency Virus Type 1 (HIV-1) Vpu Protein Interferes with an Early Step in the Biosynthesis of Major Histocompatibility Complex (MHC) Class I Molecules. J. Exp. Med. 1997, 185, 1295–1306. [Google Scholar] [CrossRef]
- Cohen, G.B.; Gandhi, R.T.; Davis, D.M.; Mandelboim, O.; Chen, B.K.; Strominger, J.L.; Baltimore, D. The Selective Downregulation of Class I Major Histocompatibility Complex Proteins by HIV-1 Protects HIV-Infected Cells from NK Cells. Immunity 1999, 10, 661–671. [Google Scholar] [CrossRef]
- Bonaparte, M.I. Killing of human immunodeficiency virus-infected primary T-cell blasts by autologous natural killer cells is dependent on the ability of the virus to alter the expression of major histocompatibility complex class I molecules. Blood 2004, 104, 2087–2094. [Google Scholar] [CrossRef]
- Bengtsson, A.; Joergensen, L.; Barbati, Z.R.; Craig, A.; Hviid, L.; Jensen, A.T.R. Transfected HEK293 Cells Expressing Functional Recombinant Intercellular Adhesion Molecule 1 (ICAM-1)—A Receptor Associated with Severe Plasmodium falciparum Malaria. PLoS ONE 2013, 8, e69999. [Google Scholar] [CrossRef] [PubMed]
- Staunton, D.E.; Merluzzi, V.J.; Rothlein, R.; Barton, R.; Marlin, S.D.; Springer, T.A. A cell adhesion molecule, ICAM-1, is the major surface receptor for rhinoviruses. Cell 1989, 56, 849–853. [Google Scholar] [CrossRef]
- Marlin, S.D.; Springer, T.A. Purified intercellular adhesion molecule-1 (ICAM-1) is a ligand for lymphocyte function-associated antigen 1 (LFA-1). Cell 1987, 51, 813–819. [Google Scholar] [CrossRef]
- Lifson, J.D.; Reyes, G.R.; McGrath, M.S.; Stein, B.S.; Engleman, E.G. AIDS retrovirus induced cytopathology: Giant cell formation and involvement of CD4 antigen. Science 1986, 232, 1123–1127. [Google Scholar] [CrossRef] [PubMed]
- Hildreth, J.E.K.; Orentas, R.J. Involvement of a Leukocyte Adhesion Receptor (LFA-1) in HIV-Induced Syncytium Formation. Science 1989, 244, 1075–1078. [Google Scholar] [CrossRef] [PubMed]
- Butini, L.; De Fougerolles, A.R.; Vaccarezza, M.; Graziosi, C.; Cohen, D.I.; Montroni, M.; Springer, T.A.; Pantaleo, G.; Fauci, A.S. Intercellular adhesion molecules (ICAM)-1 ICAM-2 and ICAM-3 function as counter-receptors for lymphocyte function-associated molecule 1 in human immunodeficiency virus-mediated syncytia formation. Eur. J. Immunol. 1994, 24, 2191–2195. [Google Scholar] [CrossRef]
- Fecondo, J.V.; Pavuk, N.C.; Silburn, K.A.; Read, D.M.Y.; Mansell, A.S.; Boyd, A.W.; McPHEE, D.A. Synthetic Peptide Analogs of Intercellular Adhesion Molecule 1 (ICAM-1) Inhibit HIV-1 Replication in MT-2 Cells. AIDS Res. Hum. Retrovir. 1993, 9, 733–740. [Google Scholar] [CrossRef]
- Sommerfelt, M.A.; Asjo, B. Intercellular adhesion molecule 3, a candidate human immunodeficiency virus type 1 co-receptor on lymphoid and monocytoid cells. J. Gen. Virol. 1995, 76, 1345–1352. [Google Scholar] [CrossRef]
- Briskin, M.; Winsor-Hines, D.; Shyjan, A.; Cochran, N.; Bloom, S.; Wilson, J.; McEvoy, L.M.; Butcher, E.C.; Kassam, N.; Mackay, C.R.; et al. Human mucosal addressin cell adhesion molecule-1 is preferentially expressed in intestinal tract and associated lymphoid tissue. Am. J. Pathol. 1997, 151, 97–110. [Google Scholar]
- Arthos, J.; Cicala, C.; Martinelli, E.; Macleod, K.; Van Ryk, D.; Wei, D.; Xiao, Z.; Veenstra, T.D.; Conrad, T.P.; Lempicki, R.A.; et al. HIV-1 envelope protein binds to and signals through integrin α4β7, the gut mucosal homing receptor for peripheral T cells. Nat. Immunol. 2008, 9, 301–309. [Google Scholar] [CrossRef]
- Wang, X.; Xu, H.; Gill, A.F.; Pahar, B.; Kempf, D.; Rasmussen, T.; Lackner, A.A.; Veazey, R.S. Monitoring α4β7 integrin expression on circulating CD4+ T cells as a surrogate marker for tracking intestinal CD4+ T-cell loss in SIV infection. Mucosal Immunol. 2009, 2, 518. [Google Scholar] [CrossRef] [PubMed]
- Sivro, A.; Schuetz, A.; Sheward, D.; Joag, V.; Yegorov, S.; Liebenberg, L.J.; Yende-Zuma, N.; Stalker, A.; Mwatelah, R.S.; Selhorst, P.; et al. Integrin α4β7 expression on peripheral blood CD4+ T cells predicts HIV acquisition and disease progression outcomes. Sci. Transl. Med. 2018, 10, eaam6354. [Google Scholar] [CrossRef] [PubMed]
- Brenchley, J.M.; Schacker, T.W.; Ruff, L.E.; Price, D.A.; Taylor, J.H.; Beilman, G.J.; Nguyen, P.L.; Khoruts, A.; Larson, M.; Haase, A.T.; et al. CD4+ T Cell Depletion during all Stages of HIV Disease Occurs Predominantly in the Gastrointestinal Tract. J. Exp. Med. 2004, 200, 749–759. [Google Scholar] [CrossRef] [PubMed]
- Cicala, C.; Martinelli, E.; McNally, J.P.; Goode, D.J.; Gopaul, R.; Hiatt, J.; Jelicic, K.; Kottilil, S.; Macleod, K.; O’Shea, A. The integrin α4β7 forms a complex with cell-surface CD4 and defines a T-cell subset that is highly susceptible to infection by HIV-1. Proc. Natl. Acad. Sci. USA 2009, 106, 20877–20882. [Google Scholar] [CrossRef] [PubMed]
- McKinnon, L.R.; Kaul, R. Quality and quantity: Mucosal CD4+ T cells and HIV susceptibility. Curr. Opin. HIV AIDS 2012, 7, 195. [Google Scholar] [CrossRef]
- Charles A Janeway, J.; Travers, P.; Walport, M.; Shlomchik, M.J. The complement system and innate immunity. In Immunobiology: The Immune System in Health and Disease, 5th ed.; Garland Publishing: New York, NY, USA, 2001. [Google Scholar]
- Walport, M.J. Complement. N. Engl. J. Med. 2001, 344, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Weiss, L.; Okada, N.; Haeffner-Cavaillon, N.; Hattori, T.; Faucher, C.; Kazatchkine, M.D.; Okada, H. Decreased Expression of the Membrane Inhibitor of Complement-Mediated Cytolysis CD59 on T-lymphocytes of HIV-Infected Patients. AIDS 1992, 6, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Terpos, E.; Sarantopoulos, A.; Kouramba, A.; Katsarou, O.; Stavropoulos, J.; Masouridi, S.; Karafoulidou, A.; Meletis, J. Reduction of CD55 and/or CD59 in red blood cells of patients with HIV infection. Med. Sci. Monit. 2008, 14, CR276–CR280. [Google Scholar]
- Banapour, B.; Sernatinger, J.; Levy, J.A. The AIDS-associated retrovirus is not sensitive to lysis or inactivation by human serum. Virology 1986, 152, 268–271. [Google Scholar] [CrossRef]
- Saifuddin, M. Role of virion-associated glycosylphosphatidylinositol-linked proteins CD55 and CD59 in complement resistance of cell line-derived and primary isolates of HIV-1. J. Exp. Med. 1995, 182, 501–509. [Google Scholar] [CrossRef]
- Foy, T.M.; Aruffo, A.; Bajorath, J.; Buhlmann, J.E.; Noelle, R.J. Immune Regulation by Cd40 and Its Ligand Gp39. Annu. Rev. Immunol. 1996, 14, 591–617. [Google Scholar] [CrossRef] [PubMed]
- Grewal, I.S.; Flavell, R.A. A central role of CD40 ligand in the regulation of CD4+ T-cell responses. Immunol. Today 1996, 17, 410–414. [Google Scholar] [CrossRef]
- Maurais, É.; Cantin, R.; Tremblay, M.J. Human immunodeficiency virus type 1-anchored CD40 ligand induces secretion of the chemokine interleukin-8 by human primary macrophages. Virology 2009, 385, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Martin, G.; Roy, J.; Barat, C.; Ouellet, M.; Gilbert, C.; Tremblay, M.J. Human Immunodeficiency Virus Type 1-Associated CD40 Ligand Transactivates B Lymphocytes and Promotes Infection of CD4+ T Cells. J. Virol. 2007, 81, 5872–5881. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.G.; Booth, A.; Gould, S.J.; Hildreth, J.E.K. Evidence That HIV Budding in Primary Macrophages Occurs through the Exosome Release Pathway. J. Biol. Chem. 2003, 278, 52347–52354. [Google Scholar] [CrossRef] [PubMed]
- Roberts, B.D.; Butera, S.T. Host protein incorporation is conserved among diverse HIV-1 subtypes. AIDS 1999, 13, 425. [Google Scholar] [CrossRef] [PubMed]
- Gelderblom, H.R. Fine Structure of HIV and SIV; Los Alamos National Laboratory: Los Alamos, NM, USA, 1997.
- Nguyen, D.H.; Hildreth, J.E.K. Evidence for Budding of Human Immunodeficiency Virus Type 1 Selectively from Glycolipid-Enriched Membrane Lipid Rafts. J. Virol. 2000, 74, 3264–3272. [Google Scholar] [CrossRef] [PubMed]
- Ono, A.; Freed, E.O. Plasma membrane rafts play a critical role in HIV-1 assembly and release. Proc. Natl. Acad. Sci. USA 2001, 98, 13925–13930. [Google Scholar] [CrossRef]
- Brown, D.A.; London, E. Functions of Lipid Rafts in Biological Membranes. Annu. Rev. Cell Dev. Biol. 1998, 14, 111–136. [Google Scholar] [CrossRef]
- Simons, K.; Ikonen, E. Functional rafts in cell membranes. Nature 1997, 387, 4. [Google Scholar] [CrossRef]
- Liao, Z.; Cimakasky, L.M.; Hampton, R.; Nguyen, D.H.; Hildreth, J.E.K. Lipid Rafts and HIV Pathogenesis: Host Membrane Cholesterol Is Required for Infection by HIV Type 1. AIDS Res. Hum. Retrovir. 2001, 17, 1009–1019. [Google Scholar] [CrossRef] [PubMed]
- Chazal, N.; Gerlier, D. Virus Entry, Assembly, Budding, and Membrane Rafts. Microbiol. Mol. Biol. Rev. 2003, 67, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Aloia, R.C.; Tian, H.; Jensen, F.C. Lipid composition and fluidity of the human immunodeficiency virus envelope and host cell plasma membranes. Proc. Natl. Acad. Sci. USA 1993, 90, 5181–5185. [Google Scholar] [CrossRef] [PubMed]
- Guyader, M.; Kiyokawa, E.; Abrami, L.; Turelli, P.; Trono, D. Role for Human Immunodeficiency Virus Type 1 Membrane Cholesterol in Viral Internalization. J. Virol. 2002, 76, 10356–10364. [Google Scholar] [CrossRef] [PubMed]
- Liao, Z.; Graham, D.R.; Hildreth, J.E.K. Lipid Rafts and HIV Pathogenesis: Virion-Associated Cholesterol Is Required for Fusion and Infection of Susceptible Cells. AIDS Res. Hum. Retrovir. 2003, 19, 675–687. [Google Scholar] [CrossRef] [PubMed]
- Graham, D.R.M.; Chertova, E.; Hilburn, J.M.; Arthur, L.O.; Hildreth, J.E.K. Cholesterol Depletion of Human Immunodeficiency Virus Type 1 and Simian Immunodeficiency Virus with -Cyclodextrin Inactivates and Permeabilizes the Virions: Evidence for Virion-Associated Lipid Rafts. J. Virol. 2003, 77, 8237–8248. [Google Scholar] [CrossRef] [PubMed]
- Popik, W.; Alce, T.M.; Au, W.-C. Human Immunodeficiency Virus Type 1 Uses Lipid Raft-Colocalized CD4 and Chemokine Receptors for Productive Entry into CD4+ T Cells. J. Virol. 2002, 76, 4709–4722. [Google Scholar] [CrossRef] [PubMed]
- Aloia, R.C.; Jensen, F.C.; Curtain, C.C.; Mobley, P.W.; Gordon, L.M. Lipid composition and fluidity of the human immunodeficiency virus. Proc. Natl. Acad. Sci. USA 1988, 85, 900–904. [Google Scholar] [CrossRef]
- Jury, E.C.; Flores-Borja, F.; Kabouridis, P.S. Lipid rafts in T cell signalling and disease. Semin. Cell Dev. Biol. 2007, 18, 608–615. [Google Scholar] [CrossRef]
- Zhang, M.; Moran, M.; Round, J.; Low, T.A.; Patel, V.P.; Tomassian, T.; Hernandez, J.D.; Miceli, M.C. CD45 Signals outside of Lipid Rafts to Promote ERK Activation, Synaptic Raft Clustering, and IL-2 Production. J. Immunol. 2005, 174, 1479–1490. [Google Scholar] [CrossRef]
- Nakano, A.; Harada, T.; Morikawa, S.; Kato, Y. Expression of leukocyte common antigen (CD45) on various human leukemia/lymphoma cell lines. Pathol. Int. 1990, 40, 107–115. [Google Scholar] [CrossRef]
- Ono, A.; Freed, E.O. Role of Lipid Rafts in Virus Replication**This chapter was written by Akira Ono and Eric O. Freed in their personal capacity. The views expressed in this chapter do not necessarily reflect the views of the NIH, DHHS, or the United States. In Advances in Virus Research; Virus Structure and Assembly; Academic Press: New York, NY, USA, 2005; Volume 64, pp. 311–358. [Google Scholar]
- Janes, P.W.; Ley, S.C.; Magee, A.I. Aggregation of Lipid Rafts Accompanies Signaling via the T Cell Antigen Receptor. J. Cell Biol. 1999, 147, 447–461. [Google Scholar] [CrossRef] [PubMed]
- Parolini, I.; Topa, S.; Sorice, M.; Pace, A.; Ceddia, P.; Montesoro, E.; Pavan, A.; Lisanti, M.P.; Peschle, C.; Sargiacomo, M. Phorbol Ester-induced Disruption of the CD4-Lck Complex Occurs within a Detergent-resistant Microdomain of the Plasma Membrane: Involvement of the Translocation of Activated Protein Kinase C Isoforms. J. Biol. Chem. 1999, 274, 14176–14187. [Google Scholar] [CrossRef] [PubMed]
- Cinek, T.; Horejsi, V. The nature of large noncovalent complexes containing glycosyl-phosphatidylinositol-anchored membrane glycoproteins and protein tyrosine kinases. J. Immunol. 1992, 149, 2262–2270. [Google Scholar] [PubMed]
- Millán, J.; Cerny, J.; Horejsi, V.; Alonso, M.A. CD4 segregates into specific detergent-resistant T-cell membrane microdomains. Tissue Antigens 1999, 53, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Aiken, C. Nef induces CD4 endocytosis: Requirement for a critical dileucine motif in the membrane-proximal CD4 cytoplasmic domain. Cell 1994, 76, 853–864. [Google Scholar] [CrossRef]
- Willey, R.L.; Maldarelli, F.; Martin, M.A.; Strebel, K. Human immunodeficiency virus type 1 Vpu protein induces rapid degradation of CD4. J. Virol. 1992, 66, 7193–7200. [Google Scholar] [PubMed]
- Lu, Y.E.; Kielian, M. Semliki Forest Virus Budding: Assay, Mechanisms, and Cholesterol Requirement. J. Virol. 2000, 74, 7708–7719. [Google Scholar] [CrossRef] [PubMed]
- Scheiffele, P.; Rietveld, A.; Wilk, T.; Simons, K. Influenza Viruses Select Ordered Lipid Domains during Budding from the Plasma Membrane. J. Biol. Chem. 1999, 274, 2038–2044. [Google Scholar] [CrossRef] [PubMed]
- Orenstein, J.M.; Meltzer, M.S.; Phipps, T.; Gendelman, H.E. Cytoplasmic assembly and accumulation of human immunodeficiency virus types 1 and 2 in recombinant human colony-stimulating factor-1-treated human monocytes: An ultrastructural study. J. Virol. 1988, 62, 2578–2586. [Google Scholar]
- Venzke, S.; Keppler, O.T. Role of macrophages in HIV infection and persistence. Expert Rev. Clin. Immunol. 2006, 2, 613–626. [Google Scholar] [CrossRef] [PubMed]
- Cantin, R.; Methot, S.; Tremblay, M.J. Plunder and Stowaways: Incorporation of Cellular Proteins by Enveloped Viruses. J. Virol. 2005, 79, 6577–6587. [Google Scholar] [CrossRef] [PubMed]
- Gould, S.J.; Booth, A.M.; Hildreth, J.E. The Trojan exosome hypothesis. Proc. Natl. Acad. Sci. USA 2003, 100, 10592–10597. [Google Scholar] [CrossRef] [PubMed]
- Wubbolts, R.; Leckie, R.S.; Veenhuizen, P.T.M.; Schwarzmann, G.; Möbius, W.; Hoernschemeyer, J.; Slot, J.-W.; Geuze, H.J.; Stoorvogel, W. Proteomic and Biochemical Analyses of Human B Cell-derived Exosomes: Potential Implications for Their Function and Multivesicular Body Formation. J. Biol. Chem. 2003, 278, 10963–10972. [Google Scholar] [CrossRef] [PubMed]
- Raposo, G.; Moore, M.; Innes, D.; Leijendekker, R.; Leigh-Brown, A.; Benaroch, P.; Geuze, H. Human macrophages accumulate HIV-1 particles in MHC II compartments. Traffic 2002, 3, 718–729. [Google Scholar] [CrossRef] [PubMed]
- Pelchen-Matthews, A.; Kramer, B.; Marsh, M. Infectious HIV-1 assembles in late endosomes in primary macrophages. J. Cell Biol. 2003, 162, 443–455. [Google Scholar] [CrossRef] [PubMed]
- Pelchen-Matthews, A.; Raposo, G.; Marsh, M. Endosomes, exosomes and Trojan viruses. Trends Microbiol. 2004, 12, 310–316. [Google Scholar] [CrossRef]
- Cassol, E.; Alfano, M.; Biswas, P.; Poli, G. Monocyte-derived macrophages and myeloid cell lines as targets of HIV-1 replication and persistence. J. Leukoc. Biol. 2006, 80, 1018–1030. [Google Scholar] [CrossRef]
- Stolp, B.; Fackler, O.T. How HIV Takes Advantage of the Cytoskeleton in Entry and Replication. Viruses 2011, 3, 293–311. [Google Scholar] [CrossRef]
- Iyengar, S.; Hildreth, J.E.K.; Schwartz, D.H. Actin-Dependent Receptor Colocalization Required for Human Immunodeficiency Virus Entry into Host Cells. J. Virol. 1998, 72, 5251–5255. [Google Scholar]
- Gladnikoff, M.; Shimoni, E.; Gov, N.S.; Rousso, I. Retroviral Assembly and Budding Occur through an Actin-Driven Mechanism. Biophys. J. 2009, 97, 2419–2428. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Baranda, S.; Gómez-Moutón, C.; Rojas, A.; Martínez-Prats, L.; Mira, E.; Lacalle, R.A.; Valencia, A.; Dimitrov, D.S.; Viola, A.; Delgado, R. Filamin-A regulates actin-dependent clustering of HIV receptors. Nat. Cell Biol. 2007, 9, 838. [Google Scholar] [CrossRef] [PubMed]
- Audoly, G.; Popoff, M.R.; Gluschankof, P. Involvement of a small GTP binding protein in HIV-1 release. Retrovirology 2005, 2, 48. [Google Scholar] [CrossRef]
- Sasaki, H.; Nakamura, M.; Ohno, T.; Matsuda, Y.; Yuda, Y.; Nonomura, Y. Myosin-actin interaction plays an important role in human immunodeficiency virus type 1 release from host cells. Proc. Natl. Acad. Sci. USA 1995, 92, 2026–2030. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Dai, R.; Tian, C.-J.; Dawson, L.; Gorelick, R.; Yu, X.-F. Interaction of the human immunodeficiency virus type 1 nucleocapsid with actin. J. Virol. 1999, 73, 2901–2908. [Google Scholar]
- Rey, O.; Canon, J.; Krogstad, P. HIV-1 Gag protein associates with F-actin present in microfilaments. Virology 1996, 220, 530–534. [Google Scholar] [CrossRef]
- Martinez, N.W.; Xue, X.; Berro, R.G.; Kreitzer, G.; Resh, M.D. Kinesin KIF4 Regulates Intracellular Trafficking and Stability of the Human Immunodeficiency Virus Type 1 Gag Polyprotein. J. Virol. 2008, 82, 9937–9950. [Google Scholar] [CrossRef]
- Ospina Stella, A.; Turville, S. All-round manipulation of the actin cytoskeleton by HIV. Viruses 2018, 10, 63. [Google Scholar] [CrossRef]
- Dai, L.; Lidie, K.B.; Chen, Q.; Adelsberger, J.W.; Zheng, X.; Huang, D.; Yang, J.; Lempicki, R.A.; Rehman, T.; Dewar, R.L.; et al. IL-27 inhibits HIV-1 infection in human macrophages by down-regulating host factor SPTBN1 during monocyte to macrophage differentiation. J. Exp. Med. 2013, 210, 517–534. [Google Scholar] [CrossRef]
- Swaminathan, S.; Dai, L.; Lane, H.C.; Imamichi, T. Evaluating the potential of IL-27 as a novel therapeutic agent in HIV-1 infection. Cytokine Growth Factor Rev. 2013, 24, 571–577. [Google Scholar] [CrossRef]
- Del Río-Iñiguez, I.; Vázquez-Chávez, E.; Cuche, C.; Di Bartolo, V.; Bouchet, J.; Alcover, A. HIV-1 Nef Hijacks Lck and Rac1 Endosomal Traffic to Dually Modulate Signaling-Mediated and Actin Cytoskeleton–Mediated T Cell Functions. J. Immunol. 2018, 201, 2624–2640. [Google Scholar] [CrossRef] [PubMed]
- Vérollet, C.; Le Cabec, V.; Maridonneau-Parini, I. HIV-1 infection of T lymphocytes and macrophages affects their migration via Nef. Front. Immunol. 2015, 6, 514. [Google Scholar] [CrossRef] [PubMed]
- Martin, G.; Beauséjour, Y.; Thibodeau, J.; Tremblay, M.J. Envelope glycoproteins are dispensable for insertion of host HLA-DR molecules within nascent human immunodeficiency virus type 1 particles. Virology 2005, 335, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Beausejour, Y.; Tremblay, M.J. Interaction between the Cytoplasmic Domain of ICAM-1 and Pr55Gag Leads to Acquisition of Host ICAM-1 by Human Immunodeficiency Virus Type 1. J. Virol. 2004, 78, 11916–11925. [Google Scholar] [CrossRef] [PubMed]
- Poon, D.T.K.; Coren, L.V.; Ott, D.E. Efficient Incorporation of HLA Class II onto Human Immunodeficiency Virus Type 1 Requires Envelope Glycoprotein Packaging. J. Virol. 2000, 74, 3918–3923. [Google Scholar] [CrossRef] [PubMed]
- Fortin, J.-F.O.; Cantin, R.J.; Tremblay, M.J. T Cells Expressing Activated LFA-1 Are More Susceptible to Infection with Human Immunodeficiency Virus Type 1 Particles Bearing Host-Encoded ICAM-. J. Virol. 1998, 72, 8. [Google Scholar]
- Fortin, J.-F.; Cantin, R.; Bergeron, M.G.; Tremblay, M.J. Interaction between Virion-Bound Host Intercellular Adhesion Molecule-1 and the High-Affinity State of Lymphocyte Function-Associated Antigen-1 on Target Cells Renders R5 and X4 Isolates of Human Immunodeficiency Virus Type 1 More Refractory to Neutralization. Virology 2000, 268, 493–503. [Google Scholar]
- Cosma, A.; Blanc, D.; Braun, J.; Quillent, C.; Barassi, C.; Moog, C.; Klasen, S.; Spire, B.; Scarlatti, G.; Pesenti, E.; et al. Enhanced HIV infectivity and changes in GP120 conformation associated with viral incorporation of human leucocyte antigen class I molecules. AIDS 1999, 13, 2033. [Google Scholar] [CrossRef]
- Bounou, S.; Leclerc, J.E.; Tremblay, M.J. Presence of Host ICAM-1 in Laboratory and Clinical Strains of Human Immunodeficiency Virus Type 1 Increases Virus Infectivity and CD4+-T-Cell Depletion in Human Lymphoid Tissue, a Major Site of Replication In Vivo. J. Virol. 2002, 76, 1004–1014. [Google Scholar] [CrossRef]
- Fujiwara, M.; Tsunoda, R.; Shigeta, S.; Yokota, T.; Baba, M. Human Follicular Dendritic Cells Remain Uninfected and Capture Human Immunodeficiency Virus Type 1 through CD54-CD11a Interaction. J. Virol. 1999, 73, 5. [Google Scholar]
- Heath, S.L.; Tew, J.G.; Tew, J.G.; Szakal, A.K.; Burton, G.F. Follicular dendritic cells and human immunodeficiency virus infectivity. Nature 1995, 377, 740. [Google Scholar] [CrossRef] [PubMed]
- Hioe, C.E.; Chien, P.C.; Lu, C.; Springer, T.A.; Wang, X.-H.; Bandres, J.; Tuen, M. LFA-1 Expression on Target Cells Promotes Human Immunodeficiency Virus Type 1 Infection and Transmission. J. Virol. 2001, 75, 1077–1082. [Google Scholar] [CrossRef] [PubMed]
- Cantin, R.; Fortin, J.-F.; Lamontagne, G.; Tremblay, M. The presence of host-derived HLA-DR1 on human immunodeficiency virus type 1 increases viral infectivity. J. Virol. 1997, 71, 1922–1930. [Google Scholar] [PubMed]
- Cantin, R.; Fortin, J.-F.; Lamontagne, G.; Tremblay, M. The acquisition of host-derived major histocompatibility complex class II glycoproteins by human immunodeficiency virus type 1 accelerates the process of virus entry and infection in human T-lymphoid cells. Blood 1997, 90, 1091–1100. [Google Scholar] [PubMed]
- Rossio, J.L.; Bess, J., Jr.; Henderson, L.E.; Cresswell, P.; Arthur, L.O. HLA class II on HIV particles is functional in superantigen presentation to human T cells: Implications for HIV pathogenesis. AIDS Res. Hum. Retrovir. 1995, 11, 1433–1439. [Google Scholar] [CrossRef] [PubMed]
- Euler, Z.; Alter, G. Exploring the Potential of Monoclonal Antibody Therapeutics for HIV-1 Eradication. AIDS Res. Hum. Retrovir. 2014, 31, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Jin, W.; Du, T.; Wu, B.; Liu, Y.; Shattock, R.J.; Hu, Q. Binding of HIV-1 virions to α4β7 expressing cells and impact of antagonizing α4β7 on HIV-1 infection of primary CD4+ T cells. Virol. Sin. 2014, 29, 381–392. [Google Scholar] [CrossRef]
- Perez, L.G.; Chen, H.; Liao, H.-X.; Montefiori, D.C. Envelope Glycoprotein Binding to the Integrin 4 7 Is Not a General Property of Most HIV-1 Strains. J. Virol. 2014, 88, 10767–10777. [Google Scholar] [CrossRef]
- Santangelo, P.J.; Cicala, C.; Byrareddy, S.N.; Ortiz, K.T.; Little, D.; Lindsay, K.E.; Gumber, S.; Hong, J.J.; Jelicic, K.; Rogers, K.A. Early treatment of SIV+ macaques with an α4 β7 mAb alters virus distribution and preserves CD4+ T cells in later stages of infection. Mucosal Immunol. 2018, 11, 932. [Google Scholar] [CrossRef]
- Uzzan, M.; Tokuyama, M.; Rosenstein, A.K.; Tomescu, C.; SahBandar, I.N.; Ko, H.M.; Leyre, L.; Chokola, A.; Kaplan-Lewis, E.; Rodriguez, G.; et al. Anti-α4β7 therapy targets lymphoid aggregates in the gastrointestinal tract of HIV-1 infected individuals. Sci. Transl. Med. 2018, 10, eaau4711. [Google Scholar] [CrossRef]
- Fauci, A.S. An HIV Vaccine Is Essential for Ending the HIV/AIDS Pandemic. JAMA 2017, 318, 1535–1536. [Google Scholar] [CrossRef] [PubMed]
- Das, K.; Arnold, E. HIV-1 Reverse Transcriptase and Antiviral Drug Resistance (Part 1 of 2). Curr. Opin. Virol. 2013, 3, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Lehner, T.; Shearer, G.M.; Hackett, C.J.; Schultz, A.; Sharma, O.K. Alloimmunization as a Strategy for Vaccine Design against HIV/AIDS. AIDS Res. Hum. Retrovir. 2000, 16, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Shearer, G.M.; Clerici, M.; Dalgleish, A. Alloimmunization as an AIDS Vaccine? Science 1993, 262, 161–162. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y. Development of a human leukocyte antigen-based HIV vaccine. F1000Research 2018, 7, 874. [Google Scholar] [CrossRef] [PubMed]
- Mörner, A.; Jansson, M.; Bunnik, E.M.; Schøller, J.; Vaughan, R.; Wang, Y.; Montefiori, D.C.; Otting, N.; Bontrop, R.; Bergmeier, L.A.; et al. Immunization with Recombinant HLA Classes I and II, HIV-1 gp140, and SIV p27 Elicits Protection against Heterologous SHIV Infection in Rhesus Macaques. J. Virol. 2011, 85, 6442–6452. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.-B.; Wang, Y.; Babaahmady, K.; Schøller, J.; Rahman, D.; Bunnik, E.; Spallek, R.; Zong, C.-M.; Duan, J.-Z.; Qin, C.; et al. Immunization with recombinant macaque major histocompatibility complex class I and II and human immunodeficiency virus gp140 inhibits simian–human immunodeficiency virus infection in macaques. J. Gen. Virol. 2012, 93, 1506–1518. [Google Scholar] [CrossRef]
- Wang, Y.; Tao, L.; Mitchell, E.; Bravery, C.; Berlingieri, P.; Armstrong, P.; Vaughan, R.; Underwood, J.; Lehner, T. Allo-immunization elicits CD8+ T cell-derived chemokines, HIV suppressor factors and resistance to HIV infection in women. Nat. Med. 1999, 5, 1004–1009. [Google Scholar] [CrossRef]
- Jeffrey, P.-L.; Wang, Y.; Seidl, T.; Babaahmady, K.; Vaughan, R.; Lehner, T. The effect of allogeneic in vitro stimulation and in vivo immunization on memory CD4+ T-cell APOBEC3G expression and HIV-1 infectivity: Clinical immunology. Eur. J. Immunol. 2009, 39, 1956–1965. [Google Scholar] [CrossRef]
- Peters, B.; Whittall, T.; Babaahmady, K.; Gray, K.; Vaughan, R.; Lehner, T. Effect of heterosexual intercourse on mucosal alloimmunisation and resistance to HIV-1 infection. Lancet 2004, 363, 518–524. [Google Scholar] [CrossRef]
- Zamani, C.; Elzey, J.D.; Hildreth, J.E. Greater ethnic diversity correlates with lower HIV prevalence in Africa: Justification for an alloimmunity vaccine. HIVAIDS 2013, 5, 75–80. [Google Scholar]
- Murphey-Corb, M.; Martin, L.N.; Davison-Fairburn, B.; Montelaro, R.C.; Miller, M.; West, M.; Ohkawa, S.; Baskin, G.B.; Zhang, J.Y.; Putney, S.D.; et al. A formalin-inactivated whole SIV vaccine confers protection in macaques. Science 1989, 246, 1293–1297. [Google Scholar] [CrossRef] [PubMed]
- Carlson, J.R.; McGraw, T.P.; Keddie, E.; Yee, J.L.; Rosenthal, A.; Langlois, A.J.; Dickover, R.; Donovan, R.; Luciw, P.A.; Jennings, M.B.; et al. Vaccine Protection of Rhesus Macaques Against Simian Immunodeficiency Virus Infection. AIDS Res. Hum. Retrovir. 1990, 6, 1239–1246. [Google Scholar] [CrossRef] [PubMed]
- The European Concerted Action on ‘Macaque Models for AIDS Research’; Dormont, D.; Le Grand, R.; Cranage, M.; Greenaway, P.; Hunsmann, G.; Stahl-Hennig, C.; Rossi, G.; Verani, P.; Stott, J.; et al. Protection of macaques against simian immunodeficiency virus infection with inactivated vaccines: Comparison of adjuvants, doses and challenge viruses. Vaccine 1995, 13, 295–300. [Google Scholar]
- Stott, E.J.; Taffs, F.; Kitchin, P.; Chan, W.L.; Mills, K.; Page, M.; Cranage, M.; Greenaway, P. Preliminary report: Protection of cynomolgus macaques against simian immunodeficiency virus by fixed infected-cell vaccine. Lancet 1990, 336, 1538–1541. [Google Scholar] [CrossRef]
- Tardif, M.R.; Tremblay, M.J. LFA-1 Is a Key Determinant for Preferential Infection of Memory CD4+ T Cells by Human Immunodeficiency Virus Type 1. J. Virol. 2005, 79, 13714–13724. [Google Scholar] [CrossRef]
- Cheng, L.; Wang, Q.; Li, G.; Banga, R.; Ma, J.; Yu, H.; Yasui, F.; Zhang, Z.; Pantaleo, G.; Perreau, M.; et al. TLR3 agonist and CD40-targeting vaccination induces immune responses and reduces HIV-1 reservoirs. J. Clin. Investig. 2018, 128, 4387–4396. [Google Scholar] [CrossRef]
| Host Protein | Host Cell | HIV Isolate | Method of Detection | Reference |
|---|---|---|---|---|
| CD3 | H9, CD4+ T cells, Monocytes, PBMC | Ba-L, f/s.8, IIIB | EM, PB-cap, IM-cap | [13,52,56] |
| CD5 | H9, M8166, PBMC | IIIB | EM, PB-cap | [13,52] |
| CD6 | H9, M8166 | IIIB | PB-cap | [52] |
| CD11a/CD18 (LFA-1) | PBMC, SupT1, CEMX174, Jurkat, M8166, U937, H9, C8166 | IIIB, SF162, HTLV-IIIRF, LAI | PB-cap, IM-cap | [11,14,18,52,55] |
| CD11b/CD18 (Mac1) | U937, M8166 | IIIB | PB-cap | [52] |
| CD11c (integrin αX) | U937, M8166, PBMC | IIIB | PB-cap | [52] |
| CD25 | CD4+ T cells, Monocytes | Ba-L, f/s.8 | EM, IM-cap | [13,14,56] |
| CD26 | CD4+ T cells, Monocytes | Ba-L, f/s.8 | IM-cap | [56] |
| CD27 | PBMC | SF162 | IM-cap | [18] |
| CD29 (integrin β1) | PBMC, H9, U937 | IIIB | IM-cap, PB-cap | [18,52] |
| CD30 | H9 | IIIB | EM | [13] |
| CD36 | CD4+ T cells, Monocytes | Ba-L, f/s.8 | IM-cap | [56] |
| CD40/CD40L | DC, PBMC | Ba-L, CI | IM-cap, PB-cap | [61,62] |
| CD43 | PBMC, U937, SupT1, CEM.NKr, Jurkat, H9 | IIIB, SF162, HTLV-IIIRF | IM-cap, PB-cap | [11,18,52] |
| CD44 | PBMC, CD4+ T cells, Monocytes, SupT1, CEM.NKr, CEMX174 | Ba-L, f/s.8 | IM-cap, PB-cap | [11,56] |
| CD45 | PBMC, Jurkat | SF162, HTLV-IIIRF | IM-cap, PB-cap | [11,18] |
| CD46 | PBMC, CEMX174 | SF162, IIIB | IM-cap, IP | [18,53] |
| CD48 | PBMC, U937, H9, M8166 | IIIB | PB-cap | [52] |
| CD49d (integrin α4) | PBMC | SF162 | IM-cap | [18] |
| CD54 (ICAM-1) | PBMC, U937, H9, M8166, C8166 | IIIB, SF162, LAI, CI | EM, IM-cap, PB-cap | [14,18,52,54,55] |
| CD55 | CEMX174, PBMC, U937, H9, M8166 | IIIB | IP, PB-cap | [52,53] |
| CD59 | CEMX174, PBMC, H9, M8166 | IIIB | IP, PB-cap | [52,53] |
| CD62L (L-selectin) | CD4+ T cells, CEM-SS, PBMC | NL4-3, IIIB | IM-cap, PB-cap | [63,64] |
| CD63 (tetraspanin) | SupT1, CEM. NKr, Jurkat, H9, PBMC | IIIB, HTLV-IIIRF | PB-cap, EM | [11,13,14] |
| CD64 | CD4+ T cells, Monocytes | Ba-L, f/s.8 | IM-cap | [56] |
| CD71 (transferrin receptor) | H9, SupT1, CEMX174, PBMC, U937 | IIIB, HTLV-IIIRF | PB-cap | [11,52] |
| CD102 (ICAM-2) | PBMC | SF162 | IM-cap | [18] |
| CDw108 (semaphorin 7A) | H9, U937, M8166 | IIIB | PB-cap | [52] |
| MHC Class I: | ||||
| HLA-ABC | H9, PBMC, U937, M8166 | IIIB, SF162, HTLVIII, CI | EM, IM-cap, PB-cap | [12,14,18,52,54] |
| β-2 microglobulin | H9, C8166 | IIIB, HTLVIII, CI | PB-cap | [12,55] |
| MHC Class II: | ||||
| HLA-DR, -DP, -DQ | PBMC, H9, U937, M8166, H9, HUT 78, Molt 4 clone 8, C8166 | HTLV-IIIRF, SF162, IIIB, LAI, NL4-3, CI | EM, IM-cap, PB-cap, WB | [11,12,13,18,52,54,55] |
| Integrin α4β7 | PBMC | Ba-L, IIIB, SF162 | IM-cap | [18] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burnie, J.; Guzzo, C. The Incorporation of Host Proteins into the External HIV-1 Envelope. Viruses 2019, 11, 85. https://doi.org/10.3390/v11010085
Burnie J, Guzzo C. The Incorporation of Host Proteins into the External HIV-1 Envelope. Viruses. 2019; 11(1):85. https://doi.org/10.3390/v11010085
Chicago/Turabian StyleBurnie, Jonathan, and Christina Guzzo. 2019. "The Incorporation of Host Proteins into the External HIV-1 Envelope" Viruses 11, no. 1: 85. https://doi.org/10.3390/v11010085
APA StyleBurnie, J., & Guzzo, C. (2019). The Incorporation of Host Proteins into the External HIV-1 Envelope. Viruses, 11(1), 85. https://doi.org/10.3390/v11010085




