The Magistral Phage
Abstract
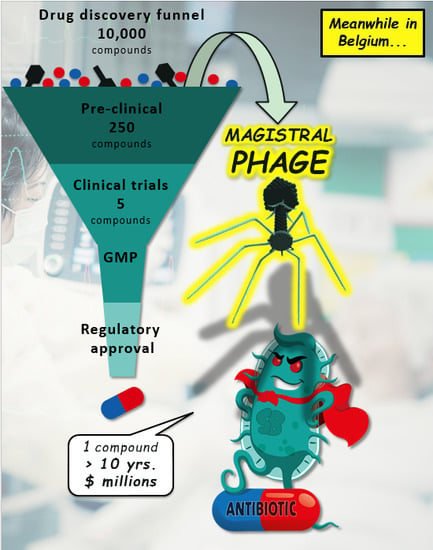
:1. The Age of the Superbug
2. Phage Therapy
The Promise of the Phage Therapy Medicinal Product
3. Enter the Magistral Phage
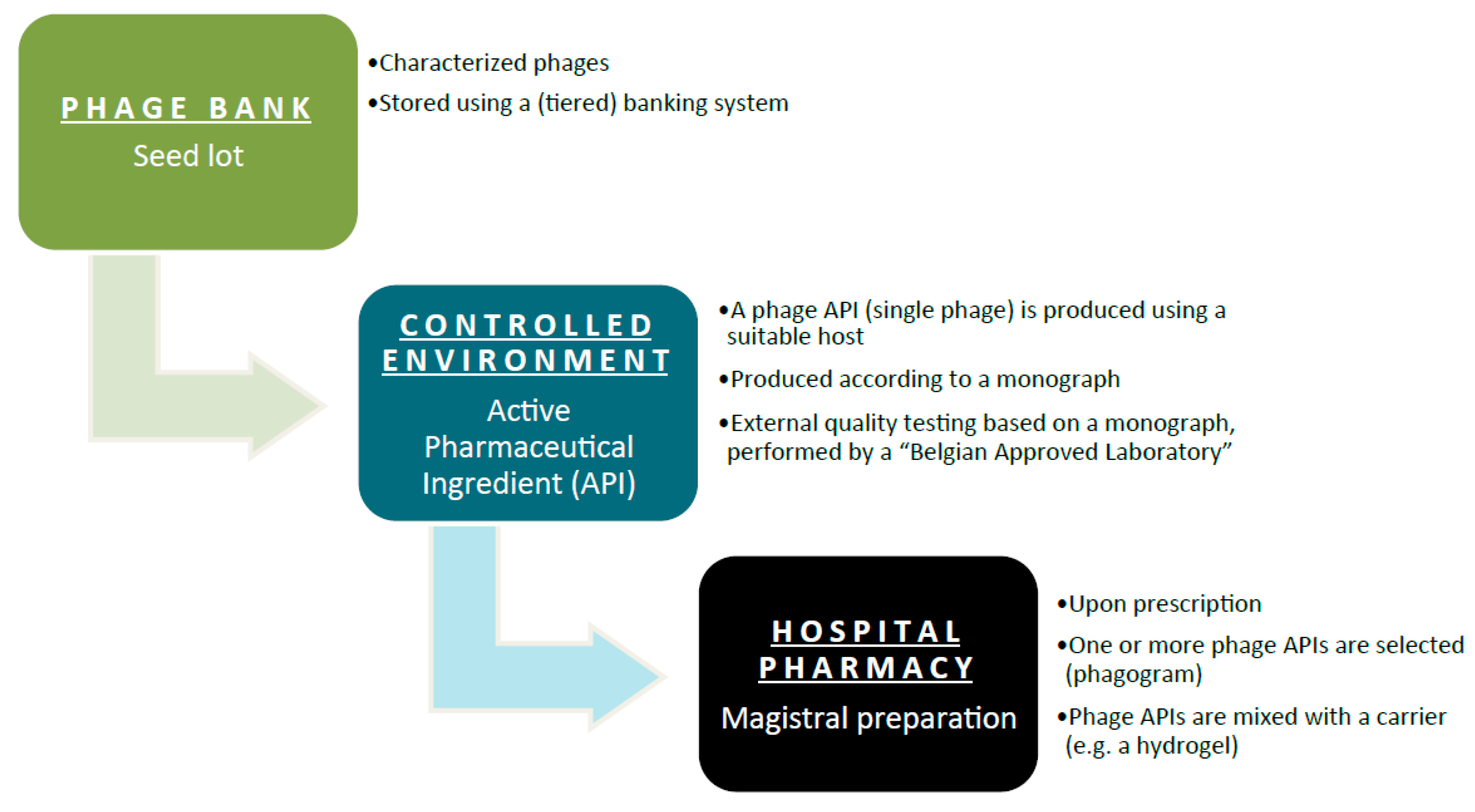
3.1. Magistral Preparations
3.2. The Belgian Magistral Phage Medicine Strategy
- Phages should be delivered in the form of a magistral preparation to a specific (nominal) patient.
- Magistral preparations should always be delivered under the direct responsibility of a medical doctor and a pharmacist.
- The relevant characteristics and qualities of the phage APIs should be defined in an internal monograph (prepared by the supplier).
- Before the pharmacist can use the unlicensed material, he/she must ascertain—based on certificates of analysis issued by a Belgian Approved Laboratory—that the raw materials conform to the provisions of the internal monograph.
- Even if not legally required, it is recommended that the supplier submits the monograph for assessment by the FAMHP.
3.3. Phage API Monograph
3.4. Pricing and Reimbursement
4. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Tackling Drug-Resistant Infections Globally: Final Report and Recommendations. The Review on Antimicrobial Resistance. 2016 Release. Available online: https://amr-review.org/sites/default/files/160525_Final%20paper_with%20cover.pdf (accessed on 4 February 2018).
- United Nations. Draft Political Declaration of the High-Level Meeting of the General Assembly on Antimicrobial Resistance (16-16108 (E)). 2016 Release. Available online: http://www.un.org/pga/71/wp-content/uploads/sites/40/2016/09/DGACM_GAEAD_ESCAB-AMR-Draft-Political-Declaration-1616108E.pdf (accessed on 17 December 2017).
- Thiel, K. Old dogma, new tricks—21st Century phage therapy. Nat. Biotechnol. 2004, 22, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.R.; De Vos, D.; Friman, V.P.; Pirnay, J.P.; Buckling, A. Effects of sequential and simultaneous applications of bacteriophages on populations of Pseudomonas aeruginosa in vitro and in wax moth larvae. Appl. Environ. Microbiol. 2012, 78, 5646–5652. [Google Scholar] [CrossRef] [PubMed]
- Jennes, S.; Merabishvili, M.; Soentjens, P.; Pang, K.W.; Rose, T.; Keersebilck, E.; Soete, O.; François, P.M.; Teodorescu, S.; Verween, G.; et al. Use of bacteriophages in the treatment of colistin-only-sensitive Pseudomonas aeruginosa septicaemia in a patient with acute kidney injury—A case report. Crit. Care 2017, 21, 129. [Google Scholar] [CrossRef] [PubMed]
- Schooley, R.T.; Biswas, B.; Gill, J.J.; Hernandez-Morales, A.; Lancaster, J.; Lessor, L.; Barr, J.J.; Reed, S.L.; Rohwer, F.; Benler, S.; et al. Development and use of personalized bacteriophage-based therapeutic cocktails to treat a patient with a disseminated resistant Acinetobacter baumannii Infection. Antimicrob. Agents Chemother. 2017, 61, e00954-17. [Google Scholar] [CrossRef] [PubMed]
- Pirnay, J.P.; De Vos, D.; Verbeken, G.; Merabishvili, M.; Chanishvili, N.; Vaneechoutte, M.; Zizi, M.; Laire, G.; Lavigne, R.; Huys, I.; et al. The phage therapy paradigm: Prêt-à-porter or sur-mesure? Pharm. Res. 2011, 28, 934–937. [Google Scholar] [CrossRef] [PubMed]
- Servick, K. Beleaguered phage therapy trial presses on. Science 2016, 352, 1506. [Google Scholar] [CrossRef] [PubMed]
- Commission De La Santé Publique, De L’environnement Et Du Renouveau De La Société. Questions Jointes De Mme Muriel Gerkens Et M. Philippe Blanchart À La Ministre Des Affaires Sociales Et De La Santé Publique Sur “La Phagothérapie” À La Ministre Des Affaires Sociales Et De La Santé Publique” (N° 11955 and N° 12911). 2016 Release. Available online: https://www.dekamer.be/doc/CCRA/pdf/54/ac464.pdf (accessed on 17 December 2017).
- Fauconnier, A. Guidelines for Bacteriophage Product Certification. Methods Mol. Biol. 2018, 1693, 253–268. [Google Scholar] [PubMed]
- Fauconnier, A. Regulating phage therapy: The biological master file concept could help to overcome regulatory challenge of personalized medicines. EMBO Rep. 2017, 18, 198–200. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pirnay, J.-P.; Verbeken, G.; Ceyssens, P.-J.; Huys, I.; De Vos, D.; Ameloot, C.; Fauconnier, A. The Magistral Phage. Viruses 2018, 10, 64. https://doi.org/10.3390/v10020064
Pirnay J-P, Verbeken G, Ceyssens P-J, Huys I, De Vos D, Ameloot C, Fauconnier A. The Magistral Phage. Viruses. 2018; 10(2):64. https://doi.org/10.3390/v10020064
Chicago/Turabian StylePirnay, Jean-Paul, Gilbert Verbeken, Pieter-Jan Ceyssens, Isabelle Huys, Daniel De Vos, Charlotte Ameloot, and Alan Fauconnier. 2018. "The Magistral Phage" Viruses 10, no. 2: 64. https://doi.org/10.3390/v10020064
APA StylePirnay, J.-P., Verbeken, G., Ceyssens, P.-J., Huys, I., De Vos, D., Ameloot, C., & Fauconnier, A. (2018). The Magistral Phage. Viruses, 10(2), 64. https://doi.org/10.3390/v10020064