Small Animal Models of Respiratory Viral Infection Related to Asthma
Abstract
1. Introduction
2. Human Rhinovirus (HRV)
2.1. Virology and HRV-Induced Airways Disease
2.2. Animal Models of HRV Infection
2.3. Models of HRV-Induced Asthma Exacerbation
2.4. Models of HRV-Induced Asthma Development in Immature Mice
3. Enterovirus D68 (EV-D68)
3.1. Virology and EV-D68 Disease
3.2. Animal Models of EV-D68
Enterovirus-D68 Acute Flaccid Myelitis (AFM) Model
3.3. Animal Models of EV-D68 Induced Asthma Exacerbation
4. Human Respiratory Syncytial Virus (RSV)
4.1. Virology and RSV Disease
4.2. Experimental Animal Models of RSV
4.3. Human RSV Mouse Model
4.4. Models of RSV-Induced Asthma Exacerbation in Mice with Allergic Airways Disease
4.5. Models of Human RSV-Induced Asthma Development in Immature Mice
4.6. PVM Model of Asthma Development
5. Human Metapneumovirus (hMPV)
5.1. hMPV Virology and Disease
5.2. Animal Models of hMPV Infection
6. Human Parainfluenza Virus (hPIV)
6.1. hPIV Virology and Disease
6.2. Animal Models of hPIV Infection
6.3. SeV Model of Asthma Development
7. Human Coronavirus (HCoV) and Human Bocaviruses (HBoV)
8. Closing Remarks
Funding
Conflicts of Interest
References
- Wenzel, S. Severe asthma in adults. Am. J. Respir. Crit. Care Med. 2005, 172, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.S.; Hamid, Q.; Ying, S.; Tsicopoulos, A.; Barkans, J.; Bentley, A.M.; Corrigan, C.; Durham, S.R.; Kay, A.B. Predominant TH2-like Bronchoalveolar T-Lymphocyte Population in Atopic Asthma. N. Engl. J. Med. 1992, 326, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, P.G.; Modrek, B.; Choy, D.F.; Jia, G.; Abbas, A.R.; Ellwanger, A.; Arron, J.R.; Koth, L.L.; Fahy, J.V. T-helper Type 2–driven Inflammation Defines Major Subphenotypes of Asthma. Am. J. Respir. Crit. Care Med. 2009, 180, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, N.W.; Walton, R.P.; Edwards, M.R.; Aniscenko, J.; Caramori, G.; Zhu, J.; Glanville, N.; Choy, K.J.; Jourdan, P.; Burnet, J.; et al. Mouse models of rhinovirus-induced disease and exacerbation of allergic airway inflammation. Nat. Med. 2008, 14, 199–204. [Google Scholar] [CrossRef]
- Newcomb, D.C.; Sajjan, U.S.; Nagarkar, D.R.; Wang, Q.; Nanua, S.; Zhou, Y.; McHenry, C.L.; Hennrick, K.T.; Tsai, W.C.; Bentley, J.K.; et al. Human rhinovirus 1B exposure induces phosphatidylinositol 3-kinase-dependent airway inflammation in mice. Am. J. Respir. Crit. Care Med. 2008, 177, 1111–1121. [Google Scholar] [CrossRef] [PubMed]
- Blanco, J.C.; Core, S.; Pletneva, L.M.; March, T.H.; Boukhvalova, M.S.; Kajon, A.E. Prophylactic Antibody Treatment and Intramuscular Immunization Reduce Infectious Human Rhinovirus 16 Load in the Lower Respiratory Tract of Challenged Cotton Rats. Trials Vaccinol. 2014, 3, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.C.; Pletneva, L.M.; Boukhvalova, M.S.; Vogel, S.N.; Kajon, A.E.; Blanco, J.C.G. Immunization with Live Human Rhinovirus (HRV) 16 Induces Protection in Cotton Rats against HRV14 Infection. Front. Microbiol. 2017, 8, 1646. [Google Scholar] [CrossRef]
- Wei, W.; Guo, H.R.; Chang, J.L.; Yu, Y.Z.; Liu, G.C.; Zhang, N.N.; Willard, S.H.; Zheng, S.; Yu, X.F. ICAM-5/Telencephalin Is a Functional Entry Receptor for Enterovirus D68. Cell Host Microbe 2016, 20, 631–641. [Google Scholar] [CrossRef]
- Rajput, C.; Han, M.; Bentley, J.K.; Lei, J.; Ishikawa, T.; Wu, Q.; Hinde, J.L.; Callear, A.P.; Stillwell, T.L.; Jackson, W.T.; et al. Enterovirus D68 infection induces IL-17-dependent neutrophilic airway inflammation and hyperresponsiveness. JCI Insight 2018, 3, 121882. [Google Scholar] [CrossRef]
- Hixon, A.M.; Yu, G.; Leser, J.S.; Yagi, S.; Clarke, P.; Chiu, C.Y.; Tyler, K.L. A mouse model of paralytic myelitis caused by enterovirus D68. PLoS Pathog. 2017, 13, e1006199. [Google Scholar] [CrossRef]
- Patel, M.C.; Wang, W.; Pletneva, L.M.; Rajagopala, S.V.; Tan, Y.; Hartert, T.V.; Boukhvalova, M.S.; Vogel, S.N.; Das, S.R.; Blanco, J.C. Enterovirus D-68 Infection, Prophylaxis, and Vaccination in a Novel Permissive Animal Model, the Cotton Rat (Sigmodon hispidus). PLoS ONE 2016, 11, e0166336. [Google Scholar] [CrossRef] [PubMed]
- Zheng, H.W.; Sun, M.; Guo, L.; Wang, J.J.; Song, J.; Li, J.Q.; Li, H.Z.; Ning, R.T.; Yang, Z.N.; Fan, H.T.; et al. Nasal Infection of Enterovirus D68 Leading to Lower Respiratory Tract Pathogenesis in Ferrets (Mustela putorius furo). Viruses 2017, 9. [Google Scholar]
- Prince, G.A.; Horswood, R.L.; Berndt, J.; Suffin, S.C.; Chanock, R.M. Respiratory syncytial virus infection in inbred mice. Infect. Immun. 1979, 26, 764–766. [Google Scholar] [PubMed]
- Prince, G.A.; Jenson, A.B.; Horswood, R.L.; Camargo, E.; Chanock, R.M. The pathogenesis of respiratory syncytial virus infection in cotton rats. Am. J. Pathol. 1978, 93, 771–791. [Google Scholar]
- Hegele, R.G.; Robinson, P.J.; Gonzalez, S.; Hogg, J.C. Production of acute bronchiolitis in guinea-pigs by human respiratory syncytial virus. Eur. Respir. J. 1993, 6, 1324–1331. [Google Scholar] [PubMed]
- Liang, B.; Surman, S.; Amaro-Carambot, E.; Kabatova, B.; Mackow, N.; Lingemann, M.; Yang, L.; McLellan, J.S.; Graham, B.S.; Kwong, P.D.; et al. Enhanced Neutralizing Antibody Response Induced by Respiratory Syncytial Virus Prefusion F Protein Expressed by a Vaccine Candidate. J. Virol. 2015, 89, 9499–9510. [Google Scholar] [CrossRef]
- Prince, G.A.; Porter, D.D. The pathogenesis of respiratory syncytial virus infection in infant ferrets. Am. J. Pathol. 1976, 82, 339–352. [Google Scholar]
- Dyer, K.D.; Garcia-Crespo, K.E.; Glineur, S.; Domachowske, J.B.; Rosenberg, H.F. The Pneumonia Virus of Mice (PVM) model of acute respiratory infection. Viruses 2012, 4, 3494–3510. [Google Scholar] [CrossRef]
- Alvarez, R.; Harrod, K.S.; Shieh, W.J.; Zaki, S.; Tripp, R.A. Human metapneumovirus persists in BALB/c mice despite the presence of neutralizing antibodies. J. Virol. 2004, 78, 14003–14011. [Google Scholar] [CrossRef]
- Wyde, P.R.; Chetty, S.N.; Jewell, A.M.; Schoonover, S.L.; Piedra, P.A. Development of a cotton rat-human metapneumovirus (hMPV) model for identifying and evaluating potential hMPV antivirals and vaccines. Antivir. Res. 2005, 66, 57–66. [Google Scholar] [CrossRef]
- Williams, J.V.; Tollefson, S.J.; Johnson, J.E.; Crowe, J.E. The cotton rat (Sigmodon hispidus) is a permissive small animal model of human metapneumovirus infection, pathogenesis, and protective immunity. J. Virol. 2005, 79, 10944–10951. [Google Scholar] [CrossRef] [PubMed]
- MacPhail, M.; Schickli, J.H.; Tang, R.S.; Kaur, J.; Robinson, C.; Fouchier, R.A.; Osterhaus, A.D.; Spaete, R.R.; Haller, A.A. Identification of small-animal and primate models for evaluation of vaccine candidates for human metapneumovirus (hMPV) and implications for hMPV vaccine design. J. Gen. Virol. 2004, 85, 1655–1663. [Google Scholar] [CrossRef] [PubMed]
- Hussell, T.; Openshaw, P.J. Intracellular IFN-gamma expression in natural killer cells precedes lung CD8+ T cell recruitment during respiratory syncytial virus infection. J. Gen. Virol. 1998, 79, 2593–2601. [Google Scholar] [CrossRef] [PubMed]
- Porter, D.D.; Prince, G.A.; Hemming, V.G.; Porter, H.G. Pathogenesis of human parainfluenza virus 3 infection in two species of cotton rats: Sigmodon hispidus develops bronchiolitis, while Sigmodon fulviventer develops interstitial pneumonia. J. Virol. 1991, 65, 103–111. [Google Scholar] [PubMed]
- Buckner, C.K.; Clayton, D.E.; Ain-Shoka, A.A.; Busse, W.W.; Dick, E.C.; Shult, P. Parainfluenza 3 infection blocks the ability of a beta adrenergic receptor agonist to inhibit antigen-induced contraction of guinea pig isolated airway smooth muscle. J. Clin. Investig. 1981, 67, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Buthala, D.A.; Soret, M.G. Parainfluenza Type 3 Virus Infection in Hamsters: Virologic, Serologic, and Pathologic Studies. J. Infect. Dis. 1964, 114, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Metzgar, D.P.; Gower, T.A.; Larson, E.J.; Fuscaldo, A.A.; Mascoli, C.C. Effect of Parainfluenza Virus Type-3 on Newborn Ferrets. J. Biol. Stand. 1974, 2, 273–282. [Google Scholar] [CrossRef]
- Walter, M.J.; Morton, J.D.; Kajiwara, N.; Agapov, E.; Holtzman, M.J. Viral induction of a chronic asthma phenotype and genetic segregation from the acute response. J. Clin. Investig. 2002, 110, 165–175. [Google Scholar] [CrossRef]
- Uhl, E.W.; Castleman, W.L.; Sorkness, R.L.; Busse, W.W.; Lemanske, R.F., Jr.; McAllister, P.K. Parainfluenza virus-induced persistence of airway inflammation, fibrosis, and dysfunction associated with TGF-beta 1 expression in brown Norway rats. Am. J. Respir. Crit. Care Med. 1996, 154, 1834–1842. [Google Scholar] [CrossRef]
- Carr, M.J.; Hunter, D.D.; Jacoby, D.B.; Undem, B.J. Expression of tachykinins in nonnociceptive vagal afferent neurons during respiratory viral infection in guinea pigs. Am. J. Respir. Crit. Care Med. 2002, 165, 1071–1075. [Google Scholar] [CrossRef]
- Percy, D.H.; Palmer, D.J. Pathogenesis of Sendai virus infection in the Syrian hamster. Lab. Anim. Sci. 1997, 47, 132–137. [Google Scholar] [PubMed]
- Teo, S.M.; Mok, D.; Pham, K.; Kusel, M.; Serralha, M.; Troy, N.; Holt, B.J.; Hales, B.J.; Walker, M.L.; Hollams, E.; et al. The Infant Nasopharyngeal Microbiome Impacts Severity of Lower Respiratory Infection and Risk of Asthma Development. Cell Host Microbe 2015, 17, 704–715. [Google Scholar] [CrossRef] [PubMed]
- Jackson, D.J.; Evans, M.D.; Gangnon, R.E.; Tisler, C.J.; Pappas, T.E.; Lee, W.M.; Gern, J.E.; Lemanske, R.F. Evidence for a Causal Relationship between Allergic Sensitization and Rhinovirus Wheezing in Early Life. Am. J. Respir. Crit. Care Med. 2012, 185, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Caliskan, M.; Bochkov, Y.A.; Kreiner-Moller, E.; Bonnelykke, K.; Stein, M.M.; Du, G.X.; Bisgaard, H.; Jackson, D.J.; Gern, J.E.; Lemanske, R.F.; et al. Rhinovirus Wheezing Illness and Genetic Risk of Childhood-Onset Asthma. N. Engl. J. Med. 2013, 368, 1398–1407. [Google Scholar] [CrossRef] [PubMed]
- Johnston, S.L.; Pattemore, P.K.; Sanderson, G.; Smith, S.; Lampe, F.; Josephs, L.; Symington, P.; O’Toole, S.; Myint, S.H.; Tyrrell, D.A.; et al. Community study of role of viral infections in exacerbations of asthma in 9–11 year old children. BMJ 1995, 310, 1225–1229. [Google Scholar] [CrossRef] [PubMed]
- Wark, P.A.; Johnston, S.L.; Moric, I.; Simpson, J.L.; Hensley, M.J.; Gibson, P.G. Neutrophil degranulation and cell lysis is associated with clinical severity in virus-induced asthma. Eur. Respir. J. 2002, 19, 68–75. [Google Scholar] [CrossRef]
- Heymann, P.W.; Carper, H.T.; Murphy, D.D.; Platts-Mills, T.A.E.; Patrie, J.; McLaughlin, A.P.; Erwin, E.A.; Shaker, M.S.; Hellems, M.; Peerzada, J.; et al. Viral infections in relation to age, atopy, and season of admission among children hospitalized for wheezing. J. Allergy Clin. Immunol. 2004, 114, 239–247. [Google Scholar] [CrossRef]
- Roberts, N.; Al Mubarak, R.; Francisco, D.; Kraft, M.; Chu, H.W. Comparison of paired human nasal and bronchial airway epithelial cell responses to rhinovirus infection and IL-13 treatment. Clin. Transl. Med. 2018, 7, 13. [Google Scholar] [CrossRef]
- Terajima, M.; Yamaya, M.; Sekizawa, K.; Okinaga, S.; Suzuki, T.; Yamada, N.; Nakayama, K.; Ohrui, T.; Oshima, T.; Numazaki, Y.; et al. Rhinovirus infection of primary cultures of human tracheal epithelium: Role of ICAM-1 and IL-1beta. Am. J. Physiol. 1997, 273, 749–759. [Google Scholar] [CrossRef]
- Proud, D.; Sanders, S.P.; Wiehler, S. Human rhinovirus infection induces airway epithelial cell production of human beta-defensin 2 both in vitro and in vivo. J. Immunol. 2004, 172, 4637–4645. [Google Scholar] [CrossRef]
- Gern, J.E.; Dick, E.C.; Lee, W.M.; Murray, S.; Meyer, K.; Handzel, Z.T.; Busse, W.W. Rhinovirus enters but does not replicate inside monocytes and airway macrophages. J. Immunol. 1996, 156, 621–627. [Google Scholar]
- Akoto, C.; Davies, D.E.; Swindle, E.J. Mast cells are permissive for rhinovirus replication: Potential implications for asthma exacerbations. Clin. Exp. Allergy 2017, 47, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Villenave, R.; Thavagnanam, S.; Sarlang, S.; Parker, J.; Douglas, I.; Skibinski, G.; Heaney, L.G.; McKaigue, J.P.; Coyle, P.V.; Shields, M.D.; et al. In vitro modeling of respiratory syncytial virus infection of pediatric bronchial epithelium, the primary target of infection in vivo. Proc. Natl. Acad. Sci. USA 2012, 109, 5040–5045. [Google Scholar] [CrossRef] [PubMed]
- Proud, D.; Turner, R.B.; Winther, B.; Wiehler, S.; Tiesman, J.P.; Reichling, T.D.; Juhlin, K.D.; Fulmer, A.W.; Ho, B.Y.; Walanski, A.A.; et al. Gene Expression Profiles during In Vivo Human Rhinovirus Infection: Insights into the Host Response. Am. J. Respir. Crit. Care Med. 2008, 178, 962–968. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.J.; Zhu, Z.; Gwaltney, J.M.; Elias, J.A. Rhinovirus Regulation of IL-1 Receptor Antagonist In Vivo and In Vitro: A Potential Mechanism of Symptom Resolution. J. Immunol. 1999, 162, 7461–7469. [Google Scholar]
- Schneider, D.; Hong, J.Y.; Bowman, E.R.; Chung, Y.; Nagarkar, D.R.; McHenry, C.L.; Goldsmith, A.M.; Bentley, J.K.; Lewis, T.C.; Hershenson, M.B. Macrophage/epithelial cell CCL2 contributes to rhinovirus-induced hyperresponsiveness and inflammation in a mouse model of allergic airways disease. Am. J. Physiol. Lung Cell. Mol. Physiol. 2013, 304, L162–L169. [Google Scholar] [CrossRef] [PubMed]
- Nagarkar, D.R.; Bowman, E.R.; Schneider, D.; Wang, Q.; Shim, J.; Zhao, Y.; Linn, M.J.; McHenry, C.L.; Gosangi, B.; Bentley, J.K.; et al. Rhinovirus infection of allergen-sensitized and -challenged mice induces eotaxin release from functionally polarized macrophages. J. Immunol. 2010, 185, 2525–2535. [Google Scholar] [CrossRef]
- Toussaint, M.; Jackson, D.J.; Swieboda, D.; Guedan, A.; Tsourouktsoglou, T.D.; Ching, Y.M.; Radermecker, C.; Makrinioti, H.; Aniscenko, J.; Bartlett, N.W.; et al. Host DNA released by NETosis promotes rhinovirus-induced type-2 allergic asthma exacerbation. Nat. Med. 2017, 23, 681–691. [Google Scholar] [CrossRef]
- Collison, A.; Hatchwell, L.; Verrills, N.; Wark, P.A.; de Siqueira, A.P.; Tooze, M.; Carpenter, H.; Don, A.S.; Morris, J.C.; Zimmermann, N.; et al. The E3 ubiquitin ligase midline 1 promotes allergen and rhinovirus-induced asthma by inhibiting protein phosphatase 2A activity. Nat. Med. 2013, 19, 232–237. [Google Scholar] [CrossRef]
- Girkin, J.L.; Hatchwell, L.M.; Collison, A.M.; Starkey, M.R.; Hansbro, P.M.; Yagita, H.; Foster, P.S.; Mattes, J. TRAIL signaling is proinflammatory and proviral in a murine model of rhinovirus 1B infection. Am. J. Physiol. Lung Cell. Mol. Physiol. 2017, 312, L89–L99. [Google Scholar] [CrossRef]
- Mukherjee, S.; Rasky, A.J.; Lundy, P.A.; Kittan, N.A.; Kunkel, S.L.; Maillard, I.P.; Kowalski, P.E.; Kousis, P.C.; Guidos, C.J.; Lukacs, N.W. STAT5-induced lunatic fringe during Th2 development alters delta-like 4-mediated Th2 cytokine production in respiratory syncytial virus-exacerbated airway allergic disease. J. Immunol. 2014, 192, 996–1003. [Google Scholar] [CrossRef] [PubMed]
- John, A.E.; Berlin, A.A.; Lukacs, N.W. Respiratory syncytial virus-induced CCL5/RANTES contributes to exacerbation of allergic airway inflammation. Eur. J. Immunol. 2003, 33, 1677–1685. [Google Scholar] [CrossRef] [PubMed]
- Schneider, D.; Hong, J.Y.; Popova, A.P.; Bowman, E.R.; Linn, M.J.; McLean, A.M.; Zhao, Y.; Sonstein, J.; Bentley, J.K.; Weinberg, J.B.; et al. Neonatal rhinovirus infection induces mucous metaplasia and airways hyperresponsiveness. J. Immunol. 2012, 188, 2894–2904. [Google Scholar] [CrossRef]
- You, D.; Siefker, D.T.; Shrestha, B.; Saravia, J.; Cormier, S.A. Building a better neonatal mouse model to understand infant respiratory syncytial virus disease. Respir. Res. 2015, 16, 91. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Rajput, C.; Hong, J.Y.; Lei, J.; Hinde, J.L.; Wu, Q.; Bentley, J.K.; Hershenson, M.B. The Innate Cytokines IL-25, IL-33, and TSLP Cooperate in the Induction of Type 2 Innate Lymphoid Cell Expansion and Mucous Metaplasia in Rhinovirus-Infected Immature Mice. J. Immunol. 2017, 199, 1308–1318. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.Y.; Bentley, J.K.; Chung, Y.T.; Lei, J.; Steenrod, J.M.; Chen, Q.; Sajjan, U.S.; Hershenson, M.B. Neonatal rhinovirus induces mucous metaplasia and airways hyperresponsiveness through IL-25 and type 2 innate lymphoid cells. J. Allergy Clin. Immunol. 2014, 134, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Stark, J.M.; McDowell, S.A.; Koenigsknecht, V.; Prows, D.R.; Leikauf, J.E.; Le Vine, A.M.; Leikauf, G.D. Genetic susceptibility to respiratory syncytial virus infection in inbred mice. J. Med. Virol. 2002, 67, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G.; Stott, E.J.; Hughes, M.; Collins, A.P. Respiratory syncytial virus infection in mice. Infect. Immun. 1984, 43, 649–655. [Google Scholar] [PubMed]
- Anderson, J.J.; Norden, J.; Saunders, D.; Toms, G.L.; Scott, R. Analysis of the local and systemic immune responses induced in BALB/c mice by experimental respiratory syncytial virus infection. J. Gen. Virol. 1990, 71, 1561–1570. [Google Scholar] [CrossRef]
- Graham, B.S.; Perkins, M.D.; Wright, P.F.; Karzon, D.T. Primary respiratory syncytial virus infection in mice. J. Med. Virol. 1988, 26, 153–162. [Google Scholar] [CrossRef]
- Culley, F.J.; Pollott, J.; Openshaw, P.J.M. Age at first viral infection determines the pattern of T cell-mediated disease during reinfection in adulthood. J. Exp. Med. 2002, 196, 1381–1386. [Google Scholar] [CrossRef] [PubMed]
- Hussell, T.; Baldwin, C.J.; O’Garra, A.; Openshaw, P.J.M. CD8(+) T cells control Th2-driven pathology during pulmonary respiratory syncytial virus infection. Eur. J. Immunol. 1997, 27, 3341–3349. [Google Scholar] [CrossRef] [PubMed]
- Openshaw, P.J.M.; Clarke, S.L.; Record, F.M. Pulmonary Eosinophilic Response to Respiratory Syncytial Virus-Infection in Mice Sensitized to the Major Surface Glycoprotein-G. Int. Immunol. 1992, 4, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Connors, M.; Giese, N.A.; Kulkarni, A.B.; Firestone, C.Y.; Morse, H.C.; Murphy, B.R. Enhanced Pulmonary Histopathology Induced by Respiratory Syncytial Virus (Rsv) Challenge of Formalin-Inactivated Rsv-Immunized Balb/C Mice Is Abrogated by Depletion of Interleukin-4 (Il-4) and Il-10. J. Virol. 1994, 68, 5321–5325. [Google Scholar] [PubMed]
- Stokes, K.L.; Chi, M.H.; Sakamoto, K.; Newcomb, D.C.; Currier, M.G.; Huckabee, M.M.; Lee, S.; Goleniewska, K.; Pretto, C.; Williams, J.V.; et al. Differential pathogenesis of respiratory syncytial virus clinical isolates in BALB/c mice. J. Virol. 2011, 85, 5782–5793. [Google Scholar] [CrossRef] [PubMed]
- Stier, M.T.; Bloodworth, M.H.; Toki, S.; Newcomb, D.C.; Goleniewska, K.; Boyd, K.L.; Quitalig, M.; Hotard, A.L.; Moore, M.L.; Hartert, T.V.; et al. Respiratory syncytial virus infection activates IL-13-producing group 2 innate lymphoid cells through thymic stromal lymphopoietin. J. Allergy Clin. Immunol. 2016, 138, 814–824. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.R.; Haas, D.L.; Poore, S.; Isakovic, S.; Gahan, M.; Mahalingam, S.; Fu, Z.F.; Tripp, R.A. Human Metapneumovirus Establishes Persistent Infection in the Lungs of Mice and Is Reactivated by Glucocorticoid Treatment. J. Virol. 2009, 83, 6837–6848. [Google Scholar] [CrossRef]
- Hamelin, M.E.; Yim, K.; Kuhn, K.H.; Cragin, R.P.; Boukhvalova, M.; Blanco, J.C.G.; Prince, G.A.; Boivin, G. Pathogenesis of human metapneumovirus lung infection in BALB/c mice and cotton rats. J. Virol. 2005, 79, 8894–8903. [Google Scholar] [CrossRef]
- Hamelin, M.E.; Prince, G.A.; Gomez, A.M.; Kinkead, R.; Boivin, G. Human metapneumovirus infection induces long-term pulmonary inflammation associated with airway obstruction and hyperresponsiveness in mice. J. Infect. Dis. 2006, 193, 1634–1642. [Google Scholar] [CrossRef]
- Walter, M.J.; Kajiwara, N.; Karanja, P.; Castro, M.; Holtzman, M.J. Interleukin 12 p40 production by barrier epithelial cells during airway inflammation. J. Exp. Med. 2001, 193, 339–351. [Google Scholar] [CrossRef]
- Tyner, J.W.; Uchida, O.; Kajiwara, N.; Kim, E.Y.; Patel, A.C.; O’Sullivan, M.P.; Walter, M.J.; Schwendener, R.A.; Cook, D.N.; Danoff, T.M.; et al. CCL5-CCR5 interaction provides antiapoptotic signals for macrophage survival during viral infection. Nat. Med. 2005, 11, 1180–1187. [Google Scholar] [CrossRef]
- Stephens, R.; Randolph, D.A.; Huang, G.M.; Holtzman, M.J.; Chaplin, D.D. Antigen-nonspecific recruitment of Th2 cells to the lung as a mechanism for viral infection-induced allergic asthma. J. Immunol. 2002, 169, 5458–5467. [Google Scholar] [CrossRef] [PubMed]
- Hegele, R.G.; Hayashi, S.; Bramley, A.M.; Hogg, J.C. Persistence of respiratory syncytial virus genome and protein after acute bronchiolitis in guinea pigs. Chest 1994, 105, 1848–1854. [Google Scholar] [CrossRef] [PubMed]
- Castleman, W.L.; Brundage-Anguish, L.J.; Kreitzer, L.; Neuenschwander, S.B. Pathogenesis of bronchiolitis and pneumonia induced in neonatal and weanling rats by parainfluenza (Sendai) virus. Am. J. Pathol. 1987, 129, 277–286. [Google Scholar] [PubMed]
- Castleman, W.L.; Owens, S.B.; Brundage-Anguish, L.J. Acute and persistent alterations in pulmonary inflammatory cells and airway mast cells induced by Sendai virus infection in neonatal rats. Vet. Pathol. 1989, 26, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Sorkness, R.; Clough, J.J.; Castleman, W.L.; Lemanske, R.F., Jr. Virus-induced airway obstruction and parasympathetic hyperresponsiveness in adult rats. Am. J. Respir. Crit. Care Med. 1994, 150, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Sorkness, R.; Lemanske, R.F.; Castleman, W.L. Persistent Airway Hyperresponsiveness after Neonatal Viral Bronchiolitis in Rats. J. Appl. Physiol. 1991, 70, 375–383. [Google Scholar] [CrossRef]
- Hadfield, A.T.; Lee, W.M.; Zhao, R.; Oliveira, M.A.; Minor, I.; Rueckert, R.R.; Rossmann, M.G. The refined structure of human rhinovirus 16 at 2.15 angstrom resolution: Implications for the viral life cycle. Structure 1997, 5, 427–441. [Google Scholar] [CrossRef]
- Stanway, G.; Hughes, P.J.; Mountford, R.C.; Minor, P.D.; Almond, J.W. The Complete Nucleotide-Sequence of a Common Cold Virus—Human Rhinovirus 14. Nucleic Acids Res. 1984, 12, 7859–7875. [Google Scholar] [CrossRef]
- Palmenberg, A.C.; Gern, J.E. Classification and Evolution of Human Rhinoviruses. In Rhinoviruses: Methods and Protocols; Jans, D.A., Ghildyal, R., Eds.; Springer: New York, NY, USA, 2015; pp. 1–10. [Google Scholar]
- Greve, J.M.; Davis, G.; Meyer, A.M.; Forte, C.P.; Yost, S.C.; Marior, C.W.; Kamarck, M.E.; Mcclelland, A. The Major Human Rhinovirus Receptor Is Icam-1. Cell 1989, 56, 839–847. [Google Scholar] [CrossRef]
- Staunton, D.E.; Merluzzi, V.J.; Rothlein, R.; Barton, R.; Marlin, S.D.; Springer, T.A. A Cell-Adhesion Molecule, Icam-1, Is the Major Surface-Receptor for Rhinoviruses. Cell 1989, 56, 849–853. [Google Scholar] [CrossRef]
- Tomassini, J.E.; Graham, D.; Dewitt, C.M.; Lineberger, D.W.; Rodkey, J.A.; Colonno, R.J. Cdna Cloning Reveals That the Major Group Rhinovirus Receptor on Hela-Cells Is Intercellular-Adhesion Molecule-1. Proc. Natl. Acad. Sci. USA 1989, 86, 4907–4911. [Google Scholar] [CrossRef] [PubMed]
- Vlasak, M.; Roivainen, M.; Reithmayer, M.; Goesler, I.; Laine, P.; Snyers, L.; Hovi, T.; Blaas, D. The minor receptor group of human rhinovirus (HRV) includes HRV23 and HRV25, but the presence of a lysine in the VP1 HI loop is not sufficient for receptor binding. J. Virol. 2005, 79, 7389–7395. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.K.; Yip, C.C.; Woo, P.C.; Yuen, K.Y. Human rhinovirus C: A newly discovered human rhinovirus species. Emerg. Health Threats J. 2010, 3, e2. [Google Scholar] [CrossRef] [PubMed]
- Bochkov, Y.A.; Watters, K.; Ashraf, S.; Griggs, T.F.; Devries, M.K.; Jackson, D.J.; Palmenberg, A.C.; Gern, J.E. Cadherin-related family member 3, a childhood asthma susceptibility gene product, mediates rhinovirus C binding and replication. Proc. Natl. Acad. Sci. USA 2015, 112, 5485–5490. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, S.E.; Lamson, D.M.; St George, K.; Walsh, T.J. Human rhinoviruses. Clin. Microbiol. Rev. 2013, 26, 135–162. [Google Scholar] [CrossRef] [PubMed]
- Arruda, E.; Pitkaranta, A.; Witek, T.J.; Doyle, C.A.; Hayden, F.G. Frequency and natural history of rhinovirus infections in adults during autumn. J. Clin. Microbiol. 1997, 35, 2864–2868. [Google Scholar] [PubMed]
- Arruda, E.; Boyle, T.R.; Winther, B.; Pevear, D.C.; Gwaltney, J.M.; Hayden, F.G. Localization of Human Rhinovirus Replication in the Upper Respiratory Tract by In Situ Hybridization. J. Infect. Dis. 1995, 171, 1329–1333. [Google Scholar] [CrossRef] [PubMed]
- Gern, J.E.; Galagan, D.M.; Jarjour, N.N.; Dick, E.C.; Busse, W.W. Detection of rhinovirus RNA in lower airway cells during experimentally induced infection. Am. J. Respir. Crit. Care Med. 1997, 155, 1159–1161. [Google Scholar] [CrossRef] [PubMed]
- Gern, J.E.; Calhoun, W.; Swenson, C.; Shen, G.; Busse, W.W. Rhinovirus infection preferentially increases lower airway responsiveness in allergic subjects. Am. J. Respir. Crit. Care Med. 1997, 155, 1872–1876. [Google Scholar] [CrossRef]
- Mosser, A.G.; Vrtis, R.; Burchell, L.; Lee, W.-M.; Dick, C.R.; Weisshaar, E.; Bock, D.; Swenson, C.A.; Cornwell, R.D.; Meyer, K.C.; et al. Quantitative and Qualitative Analysis of Rhinovirus Infection in Bronchial Tissues. Am. J. Respir. Crit. Care Med. 2005, 171, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, N.G.; Bates, P.J.; Bardin, P.G.; Papi, A.; Leir, S.H.; Fraenkel, D.J.; Meyer, J.; Lackie, P.M.; Sanderson, G.; Holgate, S.T.; et al. Rhinoviruses infect the lower airways. J. Infect. Dis. 2000, 181, 1875–1884. [Google Scholar] [CrossRef] [PubMed]
- Wos, M.; Sanak, M.; Soja, J.; Olechnowicz, H.; Busse, W.W.; Szczeklik, A. The Presence of Rhinovirus in Lower Airways of Patients with Bronchial Asthma. Am. J. Respir. Crit. Care Med. 2008, 177, 1082–1089. [Google Scholar] [CrossRef]
- Mosser, A.G.; Brockman-Schneider, R.; Amineva, S.; Burchell, L.; Sedgwick, J.B.; Busse, W.W.; Gern, J.E. Similar frequency of rhinovirus-infectible cells in upper and lower airway epithelium. J. Infect. Dis. 2002, 185, 734–743. [Google Scholar] [CrossRef] [PubMed]
- DeMore, J.P.; Weisshaar, E.H.; Vrtis, R.F.; Swenson, C.A.; Evans, M.D.; Morin, A.; Hazel, E.; Bork, J.A.; Kakumanu, S.; Sorkness, R.; et al. Similar colds in subjects with allergic asthma and nonatopic subjects after inoculation with rhinovirus-16. J. Allergy Clin. Immunol. 2009, 124, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Johnston, N.W.; Johnston, S.L.; Duncan, J.M.; Greene, J.M.; Kebadze, T.; Keith, P.K.; Roy, M.; Waserman, S.; Sears, M.R. The September epidemic of asthma exacerbations in children: A search for etiology. J. Allergy Clin. Immunol. 2005, 115, 132–138. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, K.G.; Kent, J.; Ireland, D.C. Respiratory viruses and exacerbations of asthma in adults. BMJ 1993, 307, 982–986. [Google Scholar] [CrossRef]
- Tan, W.C.; Xiang, X.; Qiu, D.; Ng, T.P.; Lam, S.F.; Hegele, R.G. Epidemiology of respiratory viruses in patients hospitalized with near-fatal asthma, acute exacerbations of asthma, or chronic obstructive pulmonary disease*1. Am. J. Med. 2003, 115, 272–277. [Google Scholar] [CrossRef]
- Green, R.H.; Brightling, C.E.; McKenna, S.; Hargadon, B.; Parker, D.; Bradding, P.; Wardlaw, A.J.; Pavord, I.D. Asthma exacerbations and sputum eosinophil counts: A randomised controlled trial. Lancet 2002, 360, 1715–1721. [Google Scholar] [CrossRef]
- Kistler, A.; Avila, P.C.; Rouskin, S.; Wang, D.; Ward, T.; Yagi, S.; Schnurr, D.; Ganem, D.; DeRisi, J.L.; Boushey, H.A. Pan-Viral Screening of Respiratory Tract Infections in Adults with and without Asthma Reveals Unexpected Human Coronavirus and Human Rhinovirus Diversity. J. Infect. Dis. 2007, 196, 817–825. [Google Scholar] [CrossRef]
- Harju, T.H.; Leinonen, M.; Nokso-Koivisto, J.; Korhonen, T.; Räty, R.; He, Q.; Hovi, T.; Mertsola, J.; Bloigu, A.; Rytilä, P.; et al. Pathogenic bacteria and viruses in induced sputum or pharyngeal secretions of adults with stable asthma. Thorax 2006, 61, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Atmar, R.L.; Guy, E.; Guntupalli, K.K.; Zimmerman, J.L.; Bandi, V.D.; Baxter, B.D.; Greenberg, S.B. Respiratory tract viral infections in inner-city asthmatic adults. Arch. Intern. Med. 1998, 158, 2453–2459. [Google Scholar] [CrossRef] [PubMed]
- Khetsuriani, N.; Kazerouni, N.N.; Erdman, D.D.; Lu, X.; Redd, S.C.; Anderson, L.J.; Teague, W.G. Prevalence of viral respiratory tract infections in children with asthma. J. Allergy Clin. Immunol. 2007, 119, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, S.B.; Allen, M.; Wilson, J.; Atmar, R.L. Respiratory viral infections in adults with and without chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2000, 162, 167–173. [Google Scholar] [CrossRef] [PubMed]
- Seemungal, T.; Harper-Owen, R.; Bhowmik, A.; Moric, I.; Sanderson, G.; Message, S.; Maccallum, P.; Meade, T.W.; Jeffries, D.J.; Johnston, S.L.; et al. Respiratory Viruses, Symptoms, and Inflammatory Markers in Acute Exacerbations and Stable Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2001, 164, 1618–1623. [Google Scholar] [CrossRef] [PubMed]
- Rohde, G.; Wiethege, A.; Borg, I.; Kauth, M.; Bauer, T.T.; Gillissen, A.; Bufe, A.; Schultze-Werninghaus, G. Respiratory viruses in exacerbations of chronic obstructive pulmonary disease requiring hospitalisation: A case-control study. Thorax 2003, 58, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Ko, F.W.S.; Ip, M.; Chan, P.K.S.; Fok, J.P.C.; Chan, M.C.H.; Ngai, J.C.; Chan, D.P.S.; Hui, D.S.C. A 1-Year Prospective Study of the Infectious Etiology in Patients Hospitalized with Acute Exacerbations of COPD. Chest 2007, 131, 44–52. [Google Scholar] [CrossRef]
- McManus, T.E.; Marley, A.-M.; Baxter, N.; Christie, S.N.; O’Neill, H.J.; Elborn, J.S.; Coyle, P.V.; Kidney, J.C. Respiratory viral infection in exacerbations of COPD. Respir. Med. 2008, 102, 1575–1580. [Google Scholar] [CrossRef]
- Kling, S.; Donninger, H.; Williams, Z.; Vermeulen, J.; Weinberg, E.; Latiff, K.; Ghildyal, R.; Bardin, P. Persistence of rhinovirus RNA after asthma exacerbation in children. Clin. Exp. Allergy 2005, 35, 672–678. [Google Scholar] [CrossRef]
- Corne, J.M.; Marshall, C.; Smith, S.; Schreiber, J.; Sanderson, G.; Holgate, S.T.; Johnston, S.L. Frequency, severity, and duration of rhinovirus infections in asthmatic and non-asthmatic individuals: A longitudinal cohort study. Lancet 2002, 359, 831–834. [Google Scholar] [CrossRef]
- Kotaniemi-Syrjanen, A.; Vainionpaa, R.; Reijonen, T.M.; Waris, M.; Korhonen, K.; Korppi, M. Rhinovirus-induced wheezing in infancy--the first sign of childhood asthma? J. Allergy Clin. Immunol. 2003, 111, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Lemanske, R.F., Jr.; Jackson, D.J.; Gangnon, R.E.; Evans, M.D.; Li, Z.; Shult, P.A.; Kirk, C.J.; Reisdorf, E.; Roberg, K.A.; Anderson, E.L.; et al. Rhinovirus illnesses during infancy predict subsequent childhood wheezing. J. Allergy Clin. Immunol. 2005, 116, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Jackson, D.J.; Gangnon, R.E.; Evans, M.D.; Roberg, K.A.; Anderson, E.L.; Pappas, T.E.; Printz, M.C.; Lee, W.M.; Shult, P.A.; Reisdorf, E.; et al. Wheezing rhinovirus illnesses in early life predict asthma development in high-risk children. Am. J. Respir. Crit. Care Med. 2008, 178, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Rubner, F.J.; Jackson, D.J.; Evans, M.D.; Gangnon, R.E.; Tisler, C.J.; Pappas, T.E.; Gern, J.E.; Lemanske, R.F. Early life rhinovirus wheezing, allergic sensitization, and asthma risk at adolescence. J. Allergy Clin. Immunol. 2017, 139, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Harris, J.R.; Racaniello, V.R. Changes in rhinovirus protein 2C allow efficient replication in mouse cells. J. Virol. 2003, 77, 4773–4780. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tuthill, T.J.; Papadopoulos, N.G.; Jourdan, P.; Challinor, L.J.; Sharp, N.A.; Plumpton, C.; Shah, K.; Barnard, S.; Dash, L.; Burnet, J.; et al. Mouse respiratory epithelial cells support efficient replication of human rhinovirus. J. Gen. Virol. 2003, 84, 2829–2836. [Google Scholar] [CrossRef] [PubMed]
- Reithmayer, M.; Reischl, A.; Snyers, L.; Blaas, D. Species-specific receptor recognition by a minor-group human rhinovirus (HRV): HRV serotype 1A distinguishes between the murine and the human low-density lipoprotein receptor. J. Virol. 2002, 76, 6957–6965. [Google Scholar] [CrossRef]
- Rasmussen, A.L.; Racaniello, V.R. Selection of rhinovirus 1A variants adapted for growth in mouse lung epithelial cells. Virology 2011, 420, 82–88. [Google Scholar] [CrossRef][Green Version]
- Foxman, E.F.; Storer, J.A.; Fitzgerald, M.E.; Wasik, B.R.; Hou, L.; Zhao, H.Y.; Turner, P.E.; Pyle, A.M.; Iwasaki, A. Temperature-dependent innate defense against the common cold virus limits viral replication at warm temperature in mouse airway cells. Proc. Natl. Acad. Sci. USA 2015, 112, 827–832. [Google Scholar] [CrossRef]
- Nagarkar, D.R.; Wang, Q.; Shim, J.; Zhao, Y.; Tsai, W.C.; Lukacs, N.W.; Sajjan, U.; Hershenson, M.B. CXCR2 Is Required for Neutrophilic Airway Inflammation and Hyperresponsiveness in a Mouse Model of Human Rhinovirus Infection. J. Immunol. 2009, 183, 6698–6707. [Google Scholar] [CrossRef]
- Wang, Q.; Miller, D.J.; Bowman, E.R.; Nagarkar, D.R.; Schneider, D.; Zhao, Y.; Linn, M.J.; Goldsmith, A.M.; Bentley, J.K.; Sajjan, U.S.; et al. MDA5 and TLR3 Initiate Pro-Inflammatory Signaling Pathways Leading to Rhinovirus-Induced Airways Inflammation and Hyperresponsiveness. PLoS Pathog. 2011, 7, e1002070. [Google Scholar] [CrossRef] [PubMed]
- Girkin, J.; Hatchwell, L.; Foster, P.; Johnston, S.L.; Bartlett, N.; Collison, A.; Mattes, J. CCL7 and IRF-7 Mediate Hallmark Inflammatory and IFN Responses following Rhinovirus 1B Infection. J. Immunol. 2015, 194, 4924–4930. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Chung, Y.; Young Hong, J.; Rajput, C.; Lei, J.; Hinde, J.L.; Chen, Q.; Weng, S.P.; Bentley, J.K.; Hershenson, M.B. Toll-like receptor 2-expressing macrophages are required and sufficient for rhinovirus-induced airway inflammation. J. Allergy Clin. Immunol. 2016, 138, 1619–1630. [Google Scholar] [CrossRef] [PubMed]
- Glanville, N.; Peel, T.J.; Schroder, A.; Aniscenko, J.; Walton, R.P.; Finotto, S.; Johnston, S.L. Tbet Deficiency Causes T Helper Cell Dependent Airways Eosinophilia and Mucus Hypersecretion in Response to Rhinovirus Infection. PLoS Pathog. 2016, 12, e1005913. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.; Hong, J.Y.; Lei, J.; Chen, Q.; Bentley, J.K.; Hershenson, M.B. Rhinovirus Infection Induces IL-13 Production from CD11b-positive, Exudative M2-polarized Exudative Macrophages. Am. J. Respir. Cell Mol. Biol. 2015, 52, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Bentley, J.K.; Sajjan, U.S.; Dzaman, M.B.; Jarjour, N.N.; Lee, W.M.; Gern, J.E.; Hershenson, M.B. Rhinovirus colocalizes with CD68- and CD11b-positive macrophages following experimental infection in humans. J. Allergy Clin. Immunol. 2013, 132, 758–761. [Google Scholar] [CrossRef] [PubMed]
- Mehta, A.K.; Duan, W.; Doerner, A.M.; Traves, S.L.; Broide, D.H.; Proud, D.; Zuraw, B.L.; Croft, M. Rhinovirus infection interferes with induction of tolerance to aeroantigens through OX40 ligand, thymic stromal lymphopoietin, and IL-33. J. Allergy Clin. Immunol. 2016, 137, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Chairakaki, A.-D.; Saridaki, M.-I.; Pyrillou, K.; Mouratis, M.-A.; Koltsida, O.; Walton, R.P.; Bartlett, N.W.; Stavropoulos, A.; Boon, L.; Rovina, N.; et al. Plasmacytoid dendritic cells drive acute asthma exacerbations. J. Allergy Clin. Immunol. 2018, 142, 542–556. [Google Scholar] [CrossRef]
- Phan, J.A.; Kicic, A.; Berry, L.J.; Fernandes, L.B.; Zosky, G.R.; Sly, P.D.; Larcombe, A.N. Rhinovirus exacerbates house-dust-mite induced lung disease in adult mice. PLoS ONE 2014, 9, e92163. [Google Scholar] [CrossRef]
- Hatchwell, L.; Collison, A.; Girkin, J.; Parsons, K.; Li, J.Y.; Zhang, J.; Phipps, S.; Knight, D.; Bartlett, N.W.; Johnston, S.L.; et al. Toll-like receptor 7 governs interferon and inflammatory responses to rhinovirus and is suppressed by IL-5-induced lung eosinophilia. Thorax 2015, 70, 854–861. [Google Scholar] [CrossRef]
- Han, M.; Hong, J.Y.; Jaipalli, S.; Rajput, C.; Lei, J.; Hinde, J.L.; Chen, Q.; Hershenson, N.M.; Bentley, J.K.; Hershenson, M.B. IFN-gamma Blocks Development of an Asthma Phenotype in Rhinovirus-Infected Baby Mice by Inhibiting Type 2 Innate Lymphoid Cells. Am. J. Respir. Cell Mol. Biol. 2017, 56, 242–251. [Google Scholar] [PubMed]
- Lachowicz-Scroggins, M.E.; Boushey, H.A.; Finkbeiner, W.E.; Widdicombe, J.H. Interleukin-13-Induced Mucous Metaplasia Increases Susceptibility of Human Airway Epithelium to Rhinovirus Infection. Am. J. Respir. Cell Mol. Biol. 2010, 43, 652–661. [Google Scholar] [CrossRef] [PubMed]
- Rajput, C.; Cui, T.; Han, M.Y.; Lei, J.; Hinde, J.L.; Wu, Q.; Bentley, J.K.; Hershenson, M.B. ROR alpha-dependent type 2 innate lymphoid cells are required and sufficient for mucous metaplasia in immature mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 2017, 312, L983–L993. [Google Scholar] [CrossRef] [PubMed]
- Schieble, J.H.; Fox, V.L.; Lennette, E.H. A probable new human picornavirus associated with respiratory diseases. Am. J. Epidemiol. 1967, 85, 297–310. [Google Scholar] [CrossRef] [PubMed]
- Tokarz, R.; Firth, C.; Madhi, S.A.; Howie, S.R.C.; Wu, W.; Sall, A.A.; Haq, S.; Briese, T.; Lipkin, W.I. Worldwide emergence of multiple clades of enterovirus 68. J. Gen. Virol. 2012, 93, 1952–1958. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, T.; Mizuta, K.; Abiko, C.; Aoki, Y.; Itagaki, T.; Katsushima, F.; Katsushima, Y.; Matsuzaki, Y.; Fuji, N.; Imamura, T.; et al. Acute respiratory infections due to enterovirus 68 in Yamagata, Japan between 2005 and 2010. Microbiol. Immunol. 2012, 56, 139–143. [Google Scholar] [CrossRef]
- Lu, Q.B.; Wo, Y.; Wang, H.Y.; Wei, M.T.; Zhang, L.; Yang, H.; Liu, E.M.; Li, T.Y.; Zhao, Z.T.; Liu, W.; et al. Detection of enterovirus 68 as one of the commonest types of enterovirus found in patients with acute respiratory tract infection in China. J. Med. Microbiol. 2014, 63, 408–414. [Google Scholar] [CrossRef]
- Meijer, A.; Benschop, K.S.; Donker, G.A.; van der Avoort, H.G. Continued seasonal circulation of enterovirus D68 in the Netherlands, 2011–2014. Eurosurveillance 2014, 19, 2–7. [Google Scholar] [CrossRef]
- Meijer, A.; van der Sanden, S.; Snijders, B.E.P.; Jaramillo-Gutierrez, G.; Bont, L.; van der Ent, C.K.; Overduin, P.; Jenny, S.L.; Jusic, E.; van der Avoort, H.G.A.M.; et al. Emergence and epidemic occurrence of enterovirus 68 respiratory infections in The Netherlands in 2010. Virology 2012, 423, 49–57. [Google Scholar] [CrossRef]
- Imamura, T.; Suzuki, A.; Meijer, A.; Niesters, H.G.M.; Rahamat-Langendoen, J.C.; Lojo, J.; Hodinka, R.L.; Coffin, S.; Ostroff, S.M.; Kraft, C.S.; et al. Clusters of Acute Respiratory Illness Associated with Human Enterovirus 68-Asia, Europe, and United States, 2008–2010 (Reprinted from MMWR, vol 60, pg 1301–1304, 2011). JAMA J. Am. Med. Assoc. 2011, 306, 1971–1973. [Google Scholar]
- Midgley, C.M.; Watson, J.T.; Nix, W.A.; Curns, A.T.; Rogers, S.L.; Brown, B.A.; Conover, C.; Dominguez, S.R.; Feikin, D.R.; Gray, S.; et al. Severe respiratory illness associated with a nationwide outbreak of enterovirus D68 in the USA (2014): A descriptive epidemiological investigation. Lancet Respir. Med. 2015, 3, 879–887. [Google Scholar] [CrossRef]
- Messacar, K.; Abzug, M.J.; Dominguez, S.R. 2014 outbreak of enterovirus D68 in North America. J. Med. Virol. 2016, 88, 739–745. [Google Scholar] [CrossRef]
- Rao, S.; Messacar, K.; Torok, M.R.; Rick, A.M.; Holzberg, J.; Montano, A.; Bagdure, D.; Curtis, D.J.; Oberste, M.S.; Nix, W.A.; et al. Enterovirus D68 in Critically Ill Children: A Comparison with Pandemic H1N1 Influenza. Pediatr. Crit. Care Med. 2016, 17, 1023–1031. [Google Scholar] [CrossRef]
- Abedi, G.R.; Watson, J.T.; Nix, W.A.; Oberste, M.S.; Gerber, S.I. Enterovirus and Parechovirus Surveillance—United States, 2014–2016. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 515–518. [Google Scholar] [CrossRef] [PubMed]
- Leshem, E. Acute Flaccid Myelitis among Persons Aged <= 21 Years—United States, August 1–November 13, 2014. MMWR Morb. Mortal. Wkly. Rep. 2015, 63, 1243–1244. [Google Scholar]
- Greninger, A.L.; Naccache, S.N.; Messacar, K.; Clayton, A.; Yu, G.X.; Somasekar, S.; Federman, S.; Stryke, D.; Anderson, C.; Yagi, S.; et al. A novel outbreak enterovirus D68 strain associated with acute flaccid myelitis cases in the USA (2012–14): A retrospective cohort study. Lancet Infect. Dis. 2015, 15, 671–682. [Google Scholar] [CrossRef]
- Aliabadi, N.; Messacar, K.; Pastula, D.M.; Robinson, C.C.; Leshem, E.; Sejvar, J.J.; Nix, W.A.; Oberste, M.S.; Feikin, D.R.; Dominguez, S.R. Enterovirus D68 Infection in Children with Acute Flaccid Myelitis, Colorado, USA, 2014. Emerg. Infect. Dis. 2016, 22, 1387–1394. [Google Scholar] [CrossRef]
- Du, J.; Zheng, B.; Zheng, W.; Li, P.; Kang, J.; Hou, J.; Markham, R.; Zhao, K.; Yu, X.F. Analysis of Enterovirus 68 Strains from the 2014 North American Outbreak Reveals a New Clade, Indicating Viral Evolution. PLoS ONE 2015, 10, e0144208. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Sheng, J.; Fokine, A.; Meng, G.; Shin, W.H.; Long, F.; Kuhn, R.J.; Kihara, D.; Rossmann, M.G. Structure and inhibition of EV-D68, a virus that causes respiratory illness in children. Science 2015, 347, 71–74. [Google Scholar] [CrossRef]
- Oberste, M.S.; Maher, K.; Schnurr, D.; Flemister, M.R.; Lovchik, J.C.; Peters, H.; Sessions, W.; Kirk, C.; Chatterjee, N.; Fuller, S.; et al. Enterovirus 68 is associated with respiratory illness and shares biological features with both the enteroviruses and the rhinoviruses. J. Gen. Virol. 2004, 85, 2577–2584. [Google Scholar] [CrossRef]
- Savolainen, C.; Blomqvist, S.; Mulders, M.N.; Hovi, T. Genetic clustering of all 102 human rhinovirus prototype strains: Serotype 87 is close to human enterovirus 70. J. Gen. Virol. 2002, 83, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Ishiko, H.; Miura, R.; Shimada, Y.; Hayashi, A.; Nakajima, H.; Yamazaki, S.; Takeda, N. Human Rhinovirus 87 Identified as Human Enterovirus 68 by VP4-Based Molecular Diagnosis. Intervirology 2002, 45, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Blomqvist, S.; Savolainen, C.; Raman, L.; Roivainen, M.; Hovi, T. Human rhinovirus 87 and enterovirus 68 represent a unique serotype with rhinovirus and enterovirus features. J. Clin. Microbiol. 2002, 40, 4218–4223. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Sheng, J.; Baggen, J.; Meng, G.; Xiao, C.; Thibaut, H.J.; van Kuppeveld, F.J.; Rossmann, M.G. Sialic acid-dependent cell entry of human enterovirus D68. Nat. Commun. 2015, 6, 8865. [Google Scholar] [CrossRef] [PubMed]
- Baggen, J.; Thibaut, H.J.; Staring, J.; Jae, L.T.; Liu, Y.; Guo, H.; Slager, J.J.; de Bruin, J.W.; van Vliet, A.L.; Blomen, V.A.; et al. Enterovirus D68 receptor requirements unveiled by haploid genetics. Proc. Natl. Acad. Sci. USA 2016, 113, 1399–1404. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, X.; Dai, W.; Liu, Q.; Xiong, P.; Wang, S.; Geng, L.; Gong, S.; Huang, Z. A Mouse Model of Enterovirus D68 Infection for Assessment of the Efficacy of Inactivated Vaccine. Viruses 2018, 10, 58. [Google Scholar] [CrossRef] [PubMed]
- Morrey, J.D.; Wang, H.; Hurst, B.L.; Zukor, K.; Siddharthan, V.; Van Wettere, A.J.; Sinex, D.G.; Tarbet, E.B. Causation of Acute Flaccid Paralysis by Myelitis and Myositis in Enterovirus-D68 Infected Mice Deficient in Interferon/Receptor Deficient Mice. Viruses 2018, 10, 33. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, K.; Ellis, E.F.; Hoffman, L.S.; Lybass, T.G.; Eller, J.J.; Fulginiti, V.A. The association of viral and bacterial respiratory infections with exacerbations of wheezing in young asthmatic children. J. Pediatr. 1973, 82, 578–590. [Google Scholar] [CrossRef]
- Glezen, P.; Denny, F.W. Epidemiology of acute lower respiratory disease in children. N. Engl. J. Med. 1973, 288, 498–505. [Google Scholar] [CrossRef]
- Leader, S.; Kohlhase, K. Recent trends in severe respiratory syncytial virus (RSV) among US infants, 1997 to 2000. J. Pediatr. 2003, 143, S127–S132. [Google Scholar] [CrossRef]
- Glezen, W.P.; Taber, L.H.; Frank, A.L.; Kasel, J.A. Risk of Primary Infection and Reinfection with Respiratory Syncytial Virus. Am. J. Dis. Child. 1986, 140, 543–546. [Google Scholar] [CrossRef] [PubMed]
- Stein, R.; Sherrill, D.; Morgan, W.J.; Holberg, C.J.; Halonen, M.; Taussig, L.M.; Wright, A.L.; Martinez, F.D. Respiratory syncytial virus in early life and risk of wheeze and allergy by age 13 years. Lancet 1999, 354, 541–545. [Google Scholar] [CrossRef]
- Sigurs, N.; Bjarnason, R.; Sigurbergsson, F.; Kjellman, B. Respiratory syncytial virus bronchiolitis in infancy is an important risk factor for asthma and allergy at age 7. Am. J. Respir. Crit. Care Med. 2000, 161, 1501–1507. [Google Scholar] [CrossRef] [PubMed]
- Sigurs, N.; Gustafsson, P.M.; Bjarnason, R.; Lundberg, F.; Schmidt, S.; Sigurbergsson, F.; Kjellman, B. Severe respiratory syncytial virus bronchiolitis in infancy and asthma and allergy at age 13. Am. J. Respir. Crit. Care Med. 2005, 171, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Rima, B.; Collins, P.; Easton, A.; Fouchier, R.; Kurath, G.; Lamb, R.A.; Lee, B.; Maisner, A.; Rota, P.; Wang, L.F.; et al. ICTV Virus Taxonomy Profile: Pneumoviridae. J. Gen. Virol. 2017, 98, 2912–2913. [Google Scholar] [CrossRef] [PubMed]
- Tayyari, F.; Marchant, D.; Moraes, T.J.; Duan, W.M.; Mastrangelo, P.; Hegele, R.G. Identification of nucleolin as a cellular receptor for human respiratory syncytial virus. Nat. Med. 2011, 17, 1132–1135. [Google Scholar] [CrossRef] [PubMed]
- Cirino, N.M.; Panuska, J.R.; Villani, A.; Taraf, H.; Rebert, N.A.; Merolla, R.; Tsivitse, P.; Gilbert, I.A. Restricted Replication of Respiratory Syncytial Virus in Human Alveolar Macrophages. J. Gen. Virol. 1993, 74, 1527–1537. [Google Scholar] [CrossRef]
- Blount, R.E., Jr.; Morris, J.A.; Savage, R.E. Recovery of cytopathogenic agent from chimpanzees with coryza. Proc. Soc. Exp. Biol. Med. 1956, 92, 544–549. [Google Scholar]
- Belshe, R.B.; Richardson, L.S.; London, W.T.; Sly, D.L.; Lorfeld, J.H.; Camargo, E.; Prevar, D.A.; Chanock, R.M. Experimental respiratory syncytial virus infection of four species of primates. J. Med. Virol. 1977, 1, 157–162. [Google Scholar] [CrossRef]
- Richardson, L.S.; Belshe, R.B.; Sly, D.L.; London, W.T.; Prevar, D.A.; Camargo, E.; Chanock, R.M. Experimental respiratory syncytial virus pneumonia in cebus monkeys. J. Med. Virol. 1978, 2, 45–59. [Google Scholar] [CrossRef]
- Richardson, L.S.; Belshe, R.B.; London, W.T.; Sly, D.L.; Prevar, D.A.; Camargo, E.; Chanock, R.M. Evaluation of five temperature-sensitive mutants of respiratory syncytial virus in primates: I. Viral shedding, immunologic response, and associated illness. J. Med. Virol. 1978, 3, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Taylor, G. Animal models of respiratory syncytial virus infection. Vaccine 2017, 35, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Dakhama, A.; Vitalis, T.Z.; Hegele, R.G. Persistence of respiratory syncytial virus (RSV) infection and development of RSV-specific IgG1 response in a guinea-pig model of acute bronchiolitis. Eur. Respir. J. 1997, 10, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Bramley, A.M.; Khan, M.A.; Manson, H.E.; Hegele, R.G. Development of respiratory syncytial virus “bronchiolitis” in guinea pigs does not reflect an allergic predisposition in the host. Chest 2003, 124, 671–681. [Google Scholar] [CrossRef] [PubMed]
- Robinson, P.J.; Hegele, R.G.; Schellenberg, R.R. Allergic sensitization increases airway reactivity in guinea pigs with respiratory syncytial virus bronchiolitis. J. Allergy Clin. Immunol. 1997, 100, 492–498. [Google Scholar] [CrossRef]
- Chan, K.F.; Carolan, L.A.; Druce, J.; Chappell, K.; Watterson, D.; Young, P.; Korenkov, D.; Subbarao, K.; Barr, I.G.; Laurie, K.L.; et al. Pathogenesis, Humoral Immune Responses, and Transmission between Cohoused Animals in a Ferret Model of Human Respiratory Syncytial Virus Infection. J. Virol. 2018, 92, e01322-17. [Google Scholar] [CrossRef]
- Prince, G.A.; Hemming, V.G.; Horswood, R.L.; Baron, P.A.; Chanock, R.M. Effectiveness of topically administered neutralizing antibodies in experimental immunotherapy of respiratory syncytial virus infection in cotton rats. J. Virol. 1987, 61, 1851–1854. [Google Scholar]
- Cullen, L.M.; Blanco, J.C.G.; Morrison, T.G. Cotton rat immune responses to virus-like particles containing the pre-fusion form of respiratory syncytial virus fusion protein. J. Transl. Med. 2015, 13, 350. [Google Scholar] [CrossRef]
- Stobart, C.C.; Rostad, C.A.; Ke, Z.L.; Dillard, R.S.; Hampton, C.M.; Strauss, J.D.; Yi, H.; Hotard, A.L.; Meng, J.; Pickles, R.J.; et al. A live RSV vaccine with engineered thermostability is immunogenic in cotton rats despite high attenuation. Nat. Commun. 2016, 7, 13916. [Google Scholar] [CrossRef]
- Horsfall, F.L.; Hahn, R.G. A Latent Virus in Normal Mice Capable of Producing Pneumonia in Its Natural Host. J. Exp. Med. 1940, 71, 391–408. [Google Scholar] [CrossRef]
- Taylor, G.; Thomas, L.H.; Wyld, S.G.; Furze, J.; Sopp, P.; Howard, C.J. Role of T-lymphocyte subsets in recovery from respiratory syncytial virus infection in calves. J. Virol. 1995, 69, 6658–6664. [Google Scholar] [PubMed]
- Sacco, R.E.; Durbin, R.K.; Durbin, J.E. Animal models of respiratory syncytial virus infection and disease. Curr. Opin. Virol. 2015, 13, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Bem, R.A.; Domachowske, J.B.; Rosenberg, H.F. Animal models of human respiratory syncytial virus disease. Am. J. Physiol. Lung Cell. Mol. Physiol. 2011, 301, L148–L156. [Google Scholar] [CrossRef] [PubMed]
- Rameix-Welti, M.A.; Le Goffic, R.; Herve, P.L.; Sourimant, J.; Remot, A.; Riffault, S.; Yu, Q.; Galloux, M.; Gault, E.; Eleouet, J.F. Visualizing the replication of respiratory syncytial virus in cells and in living mice. Nat. Commun. 2014, 5, 5104. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, S.; Arenas, D.; Moore, M.M.; Golding, H.; Khurana, S. Development of bioluminescence imaging of respiratory syncytial virus (RSV) in virus-infected live mice and its use for evaluation of therapeutics and vaccines. Vaccine 2017, 35, 694–702. [Google Scholar] [CrossRef] [PubMed]
- High, M.; Cho, H.Y.; Marzec, J.; Wiltshire, T.; Verhein, K.C.; Caballero, M.T.; Acosta, P.L.; Ciencewicki, J.; Mccaw, Z.R.; Kobzik, L.; et al. Determinants of host susceptibility to murine respiratory syncytial virus (RSV) disease identify a role for the innate immunity scavenger receptor MARCO gene in human infants. Ebiomedicine 2016, 11, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Ruckwardt, T.J.; Malloy, A.M.W.; Gostick, E.; Price, D.A.; Dash, P.; McClaren, J.L.; Thomas, P.G.; Graham, B.S. Neonatal CD8 T-cell Hierarchy Is Distinct from Adults and Is Influenced by Intrinsic T cell Properties in Respiratory Syncytial Virus Infected Mice. PLoS Pathog. 2011, 7, e1002377. [Google Scholar] [CrossRef]
- Cormier, S.A.; You, D.H.; Honnegowda, S. The use of a neonatal mouse model to study respiratory syncytial virus infections. Expert Rev. Anti-Infect. Ther. 2010, 8, 1371–1380. [Google Scholar] [CrossRef]
- Empey, K.M.; Orend, J.G.; Peebles, R.S.; Egana, L.; Norris, K.A.; Oury, T.D.; Kolls, J.K. Stimulation of Immature Lung Macrophages with Intranasal Interferon Gamma in a Novel Neonatal Mouse Model of Respiratory Syncytial Virus Infection. PLoS ONE 2012, 7, e40499. [Google Scholar] [CrossRef]
- Li, F.A.; Zhu, H.Q.; Sun, R.; Wei, H.M.; Tian, Z.G. Natural Killer Cells Are Involved in Acute Lung Immune Injury Caused by Respiratory Syncytial Virus Infection. J. Virol. 2012, 86, 2251–2258. [Google Scholar] [CrossRef]
- Liu, J.; Haddad, E.K.; Marceau, J.; Morabito, K.M.; Rao, S.S.; Filali-Mouhim, A.; Sekaly, R.P.; Graham, B.S. A Numerically Subdominant CD8 T Cell Response to Matrix Protein of Respiratory Syncytial Virus Controls Infection with Limited Immunopathology. PLoS Pathog. 2016, 12, e1005486. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.E.; Knudson, C.J.; Hartwig, S.M.; Pewe, L.L.; Meyerholz, D.K.; Langlois, R.A.; Harty, J.T.; Varga, S.M. Memory CD8 T cells mediate severe immunopathology following respiratory syncytial virus infection. PLoS Pathog. 2018, 14, e1006810. [Google Scholar] [CrossRef] [PubMed]
- Kinnear, E.; Lambert, L.; McDonald, J.U.; Cheeseman, H.M.; Caproni, L.J.; Tregoning, J.S. Airway T cells protect against RSV infection in the absence of antibody. Mucosal Immunol. 2018, 11, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Jozwik, A.; Habibi, M.S.; Paras, A.; Zhu, J.; Guvenel, A.; Dhariwal, J.; Almond, M.; Wong, E.H.C.; Sykes, A.; Maybeno, M.; et al. RSV-specific airway resident memory CD8+T cells and differential disease severity after experimental human infection. Nat. Commun. 2015, 6, 10224. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, H.Y.; Hai, Y.; Yin, W.; Li, W.J.; Zheng, B.Y.; Du, X.M.; Li, N.; Zhang, Z.Z.; Deng, Y.Q.; et al. CpG in Combination with an Inhibitor of Notch Signaling Suppresses Formalin-Inactivated Respiratory Syncytial Virus-Enhanced Airway Hyperresponsiveness and Inflammation by Inhibiting Th17 Memory Responses and Promoting Tissue-Resident Memory Cells in Lungs. J. Virol. 2017, 91, e02111-16. [Google Scholar] [CrossRef] [PubMed]
- Fulton, R.B.; Meyerholz, D.K.; Varga, S.M. Foxp3(+) CD4 Regulatory T Cells Limit Pulmonary Immunopathology by Modulating the CD8 T Cell Response during Respiratory Syncytial Virus Infection. J. Immunol. 2010, 185, 2382–2392. [Google Scholar] [CrossRef] [PubMed]
- Ruckwardt, T.J.; Bonaparte, K.L.; Nason, M.C.; Graham, B.S. Regulatory T Cells Promote Early Influx of CD8(+) T Cells in the Lungs of Respiratory Syncytial Virus-Infected Mice and Diminish Immunodominance Disparities. J. Virol. 2009, 83, 3019–3028. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.C.P.; Harker, J.A.E.; Tregoning, J.S.; Atabani, S.F.; Johansson, C.; Schwarze, J.; Openshaw, P.J.M. CD25(+) Natural Regulatory T Cells Are Critical in Limiting Innate and Adaptive Immunity and Resolving Disease following Respiratory Syncytial Virus Infection. J. Virol. 2010, 84, 8790–8798. [Google Scholar] [CrossRef]
- Durant, L.R.; Makris, S.; Voorburg, C.M.; Loebbermann, J.; Johansson, C.; Openshaw, P.J. Regulatory T cells prevent Th2 immune responses and pulmonary eosinophilia during respiratory syncytial virus infection in mice. J. Virol. 2013, 87, 10946–10954. [Google Scholar] [CrossRef]
- Ruckwardt, T.J.; Malloy, A.M.W.; Morabito, K.M.; Graham, B.S. Quantitative and Qualitative Deficits in Neonatal Lung-Migratory Dendritic Cells Impact the Generation of the CD8+T Cell Response. PLoS Pathog. 2014, 10, e1003934. [Google Scholar] [CrossRef]
- Lynch, J.P.; Werder, R.B.; Loh, Z.X.; Sikder, M.A.; Curren, B.; Zhang, V.V.; Rogers, M.J.; Lane, K.; Simpson, J.; Mazzone, S.B.; et al. Plasmacytoid dendritic cells protect from viral bronchiolitis and asthma through semaphorin 4a-mediated T reg expansion. J. Exp. Med. 2018, 215, 537–557. [Google Scholar] [CrossRef] [PubMed]
- Roman, M.; Calhoun, W.J.; Hinton, K.L.; Avendano, L.F.; Simon, V.; Escobar, A.M.; Gaggero, A.; Diaz, P.V. Respiratory syncytial virus infection in infants is associated with predominant Th-2-like response. Am. J. Respir. Crit. Care Med. 1997, 156, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Pala, P.; Bjarnason, R.; Sigurbergsson, F.; Metcalfe, C.; Sigurs, N.; Openshaw, P.J.M. Enhanced IL-4 responses in children with a history of respiratory syncytial virus bronchiolitis in infancy. Eur. Respir. J. 2002, 20, 376–382. [Google Scholar] [CrossRef] [PubMed]
- Srikiatkhachorn, A.; Braciale, T.J. Virus-specific CD8+ T lymphocytes downregulate T helper cell type 2 cytokine secretion and pulmonary eosinophilia during experimental murine respiratory syncytial virus infection. J. Exp. Med. 1997, 186, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Hussell, T.; Openshaw, P.J. IL-12-activated NK cells reduce lung eosinophilia to the attachment protein of respiratory syncytial virus but do not enhance the severity of illness in CD8 T cell-immunodeficient conditions. J. Immunol. 2000, 165, 7109–7115. [Google Scholar] [CrossRef] [PubMed]
- Herlocher, M.L.; Ewasyshyn, M.; Sambhara, S.; Gharaee-Kermani, M.; Cho, D.; Lai, J.; Klein, M.; Maassab, H.F. Immunological properties of plaque purified strains of live attenuated respiratory syncytial virus (RSV) for human vaccine. Vaccine 1999, 17, 172–181. [Google Scholar] [CrossRef]
- Lukacs, N.W.; Moore, M.L.; Rudd, B.D.; Berlin, A.A.; Collins, R.D.; Olson, S.J.; Ho, S.B.; Peebles, R.S., Jr. Differential immune responses and pulmonary pathophysiology are induced by two different strains of respiratory syncytial virus. Am. J. Pathol. 2006, 169, 977–986. [Google Scholar] [CrossRef]
- Moore, M.L.; Chi, M.H.; Luongo, C.; Lukacs, N.W.; Polosukhin, V.V.; Huckabee, M.M.; Newcomb, D.C.; Buchholz, U.J.; Crowe, J.E., Jr.; Goleniewska, K.; et al. A chimeric A2 strain of respiratory syncytial virus (RSV) with the fusion protein of RSV strain line 19 exhibits enhanced viral load, mucus, and airway dysfunction. J. Virol. 2009, 83, 4185–4194. [Google Scholar] [CrossRef]
- Fujimura, K.E.; Demoor, T.; Rauch, M.; Faruqi, A.A.; Jang, S.; Johnson, C.C.; Boushey, H.A.; Zoratti, E.; Ownby, D.; Lukacs, N.W.; et al. House dust exposure mediates gut microbiome Lactobacillus enrichment and airway immune defense against allergens and virus infection. Proc. Natl. Acad. Sci. USA 2014, 111, 805–810. [Google Scholar] [CrossRef]
- Petersen, B.C.; Dolgachev, V.; Rasky, A.; Lukacs, N.W. IL-17E (IL-25) and IL-17RB promote respiratory syncytial virus-induced pulmonary disease. J. Leukoc. Biol. 2014, 95, 809–815. [Google Scholar] [CrossRef]
- Lee, H.C.; Headley, M.B.; Loo, Y.M.; Berlin, A.; Gale, M.; Debley, J.S.; Lukacs, N.W.; Ziegler, S.F. Thymic stromal lymphopoietin is induced by respiratory syncytial virus-infected airway epithelial cells and promotes a type 2 response to infection. J. Allergy Clin. Immunol. 2012, 130, 1187–1196. [Google Scholar] [CrossRef]
- Stier, M.T.; Goleniewska, K.; Cephus, J.Y.; Newcomb, D.C.; Sherrill, T.P.; Boyd, K.L.; Bloodworth, M.H.; Moore, M.L.; Chen, K.; Kolls, J.K.; et al. STAT1 Represses Cytokine-Producing Group 2 and Group 3 Innate Lymphoid Cells during Viral Infection. J. Immunol. 2017, 199, 510–519. [Google Scholar] [CrossRef]
- Peebles, R.S.; Sheller, J.R.; Johnson, J.E.; Mitchell, D.B.; Graham, B.S. Respiratory syncytial virus infection prolongs methacholine-induced airway hyperresponsiveness in ovalbumin-sensitized mice. J. Med. Virol. 1999, 57, 186–192. [Google Scholar] [CrossRef]
- Hashimoto, K.; Graham, B.S.; Ho, S.B.; Adler, K.B.; Collins, R.D.; Olson, S.J.; Zhou, W.; Suzutani, T.; Jones, P.W.; Goleniewska, K.; et al. Respiratory syncytial virus in allergic lung inflammation increases Muc5ac and gob-5. Am. J. Respir. Crit. Care Med. 2004, 170, 306–312. [Google Scholar] [CrossRef]
- Newcomb, D.C.; Boswell, M.G.; Reiss, S.; Zhou, W.; Goleniewska, K.; Toki, S.; Harintho, M.T.; Lukacs, N.W.; Kolls, J.K.; Peebles, R.S., Jr. IL-17A inhibits airway reactivity induced by respiratory syncytial virus infection during allergic airway inflammation. Thorax 2013, 68, 717–723. [Google Scholar] [CrossRef] [PubMed]
- Mori, H.; Parker, N.S.; Rodrigues, D.; Hulland, K.; Chappell, D.; Hincks, J.S.; Bright, H.; Evans, S.M.; Lamb, D.J. Differences in respiratory syncytial virus and influenza infection in a house-dust-mite-induced asthma mouse model: Consequences for steroid sensitivity. Clin. Sci. 2013, 125, 565–574. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lukacs, N.W.; Tekkanat, K.K.; Berlin, A.; Hogaboam, C.M.; Miller, A.; Evanoff, H.; Lincoln, P.; Maassab, H. Respiratory syncytial virus predisposes mice to augmented allergic airway responses via IL-13-mediated mechanisms. J. Immunol. 2001, 167, 1060–1065. [Google Scholar] [CrossRef]
- Knudson, C.J.; Weiss, K.A.; Hartwig, S.M.; Varga, S.M. The Pulmonary Localization of Virus-Specific T Lymphocytes Is Governed by the Tissue Tropism of Infection. J. Virol. 2014, 88, 9010–9016. [Google Scholar] [CrossRef]
- Eichinger, K.M.; Kosanovich, J.L.; Empey, K.M. Localization of the T-cell response to RSV infection is altered in infant mice. Pediatr. Pulmonol. 2018, 53, 145–153. [Google Scholar] [CrossRef]
- Fulton, R.B.; Weiss, K.A.; Pewe, L.L.; Harty, J.T.; Varga, S.M. Aged Mice Exhibit a Severely Diminished CD8 T Cell Response following Respiratory Syncytial Virus Infection. J. Virol. 2013, 87, 12694–12700. [Google Scholar] [CrossRef]
- Malloy, A.M.; Ruckwardt, T.J.; Morabito, K.M.; Lau-Kilby, A.W.; Graham, B.S. Pulmonary Dendritic Cell Subsets Shape the Respiratory Syncytial Virus-Specific CD8+ T Cell Immunodominance Hierarchy in Neonates. J. Immunol. 2017, 198, 394–403. [Google Scholar] [CrossRef] [PubMed]
- Ruckwardt, T.J.; Morabito, K.M.; Bar-Haim, E.; Nair, D.; Graham, B.S. Neonatal mice possess two phenotypically and functionally distinct lung-migratory CD103(+) dendritic cell populations following respiratory infection. Mucosal Immunol. 2018, 11, 186–198. [Google Scholar] [CrossRef] [PubMed]
- Dakhama, A.; Park, J.W.; Taube, C.; Joetham, A.; Balhorn, A.; Miyahara, N.; Takeda, K.; Gelfand, E.W. The enhancement or prevention of airway hyperresponsiveness during reinfection with respiratory syncytial virus is critically dependent on the age at first infection and IL-13 production. J. Immunol. 2005, 175, 1876–1883. [Google Scholar] [CrossRef] [PubMed]
- Saravia, J.; You, D.; Shrestha, B.; Jaligama, S.; Siefker, D.; Lee, G.I.; Harding, J.N.; Jones, T.L.; Rovnaghi, C.; Bagga, B.; et al. Respiratory Syncytial Virus Disease Is Mediated by Age-Variable IL-33. PLoS Pathog. 2015, 11, e1005217. [Google Scholar] [CrossRef] [PubMed]
- Harker, J.A.; Yamaguchi, Y.; Culley, F.J.; Tregoning, J.S.; Openshaw, P.J.M. Delayed Sequelae of Neonatal Respiratory Syncytial Virus Infection Are Dependent on Cells of the Innate Immune System. J. Virol. 2014, 88, 604–611. [Google Scholar] [CrossRef]
- Tregoning, J.S.; Yamaguchi, Y.; Harker, J.; Wang, B.; Openshaw, P.J.M. The role of T cells in the enhancement of respiratory syncytial virus infection severity during adult reinfection of neonatally sensitized mice. J. Virol. 2008, 82, 4115–4124. [Google Scholar] [CrossRef]
- Pribul, P.K.; Harker, J.; Wang, B.; Wang, H.; Tregoning, J.S.; Schwarze, J.; Openshaw, P.J. Alveolar macrophages are a major determinant of early responses to viral lung infection but do not influence subsequent disease development. J. Virol. 2008, 82, 4441–4448. [Google Scholar] [CrossRef]
- Tregoning, J.S.; Yamaguchi, Y.; Wang, B.; Mihm, D.; Harker, J.A.; Bushell, E.S.C.; Zheng, M.; Liao, G.C.; Peltz, G.; Openshaw, P.J.M. Genetic Susceptibility to the Delayed Sequelae of Neonatal Respiratory Syncytial Virus Infection Is MHC Dependent. J. Immunol. 2010, 185, 5384–5391. [Google Scholar] [CrossRef]
- Cormier, S.A.; Shrestha, B.; Saravia, J.; Lee, G.I.; Shen, L.; DeVincenzo, J.P.; Kim, Y.I.; You, D.H. Limited Type I Interferons and Plasmacytoid Dendritic Cells during Neonatal Respiratory Syncytial Virus Infection Permit Immunopathogenesis upon Reinfection. J. Virol. 2014, 88, 9350–9360. [Google Scholar] [CrossRef]
- Srinivasa, B.T.; Restori, K.H.; Shan, J.C.; Cyr, L.; Xing, L.; Lee, S.; Ward, B.J.; Fixman, E.D. STAT6 inhibitory peptide given during RSV infection of neonatal mice reduces exacerbated airway responses upon adult reinfection. J. Leukoc. Biol. 2017, 101, 519–529. [Google Scholar] [CrossRef]
- You, D.H.; Becnel, D.; Wang, K.; Ripple, M.; Daly, M.; Cormier, S.A. Exposure of neonates to Respiratory Syncytial Virus is critical in determining subsequent airway response in adults. Respir. Res. 2006, 7, 107. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Bai, S.; Cui, Y.; Zhao, N.; Qi, F.; Liu, J.; Zeng, S.; Xu, L.; Hu, H.; Liu, B. Respiratory syncytial virus prevents the subsequent development of ovalbumin-induced allergic responses by inhibiting ILC2 via the IL-33/ST2 pathway. Immunotherapy 2018, 10, 1065–1076. [Google Scholar] [CrossRef] [PubMed]
- Krishnamoorthy, N.; Khare, A.; Oriss, T.B.; Raundhal, M.; Morse, C.; Yarlagadda, M.; Wenzel, S.E.; Moore, M.L.; Peebles, R.S.; Ray, A.; et al. Early infection with respiratory syncytial virus impairs regulatory T cell function and increases susceptibility to allergic asthma. Nat. Med. 2012, 18, 1525–1530. [Google Scholar] [CrossRef] [PubMed]
- Davidson, S.; Kaiko, G.; Loh, Z.; Lalwani, A.; Zhang, V.; Spann, K.; Foo, S.Y.; Hansbro, N.; Uematsu, S.; Akira, S.; et al. Plasmacytoid dendritic cells promote host defense against acute pneumovirus infection via the TLR7-MyD88-dependent signaling pathway. J. Immunol. 2011, 186, 5938–5948. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.P.; Werder, R.B.; Simpson, J.; Loh, Z.; Zhang, V.; Haque, A.; Spann, K.; Sly, P.D.; Mazzone, S.B.; Upham, J.W.; et al. Aeroallergen-induced IL-33 predisposes to respiratory virus-induced asthma by dampening antiviral immunity. J. Allergy Clin. Immunol. 2016, 138, 1326–1337. [Google Scholar] [CrossRef] [PubMed]
- Van den Hoogen, B.G.; de Jong, J.C.; Groen, J.; Kuiken, T.; de Groot, R.; Fouchier, R.A.; Osterhaus, A.D. A newly discovered human pneumovirus isolated from young children with respiratory tract disease. Nat. Med. 2001, 7, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Jartti, T.; van den Hoogen, B.; Garofalo, R.P.; Osterhaus, A.D.; Ruuskanen, O. Metapneumovirus and acute wheezing in children. Lancet 2002, 360, 1393–1394. [Google Scholar] [CrossRef]
- Boivin, G.; Abed, Y.; Pelletier, G.; Ruel, L.; Moisan, D.; Cote, S.; Peret, T.C.T.; Erdman, D.D.; Anderson, L.J. Virological features and clinical manifestations associated with human metapneumovirus: A new paramyxovirus responsible for acute respiratory-tract infections in all age groups. J. Infect. Dis. 2002, 186, 1330–1334. [Google Scholar] [CrossRef] [PubMed]
- Van den Hoogen, B.G.; van Doornum, G.J.J.; Fockens, J.C.; Cornelissen, J.J.; Beyer, W.E.P.; de Groot, R.; Osterhaus, A.D.M.E.; Fouchier, R.A.M. Prevalence and clinical symptoms of human metapneumovirus infection in hospitalized patients. J. Infect. Dis. 2003, 188, 1571–1577. [Google Scholar] [CrossRef] [PubMed]
- Falsey, A.R.; Erdman, D.; Anderson, L.J.; Walsh, E.E. Human metapneumovirus infections in young and elderly adults. J. Infect. Dis. 2003, 187, 785–790. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.V.; Crowe, J.E., Jr.; Enriquez, R.; Minton, P.; Peebles, R.S., Jr.; Hamilton, R.G.; Higgins, S.; Griffin, M.; Hartert, T.V. Human metapneumovirus infection plays an etiologic role in acute asthma exacerbations requiring hospitalization in adults. J. Infect. Dis. 2005, 192, 1149–1153. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.V.; Harris, P.A.; Tollefson, S.J.; Halburnt-Rush, L.L.; Pingsterhaus, J.M.; Edwards, K.M.; Wright, P.F.; Crowe, J.E. Human metapneumovirus and lower respiratory tract disease in otherwise healthy infants and children. N. Engl. J. Med. 2004, 350, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Van den Hoogen, B.G.; Bestebroer, T.M.; Osterhaus, A.D.M.E.; Fouchier, R.A.M. Analysis of the genomic sequence of a human metapneumovirus. Virology 2002, 295, 119–132. [Google Scholar] [CrossRef]
- Kuiken, T.; van den Hoogen, B.G.; van Riel, D.A.; Laman, J.D.; van Amerongen, G.; Sprong, L.; Fouchier, R.A.; Osterhaus, A.D. Experimental human metapneumovirus infection of cynomolgus macaques (Macaca fascicularis) results in virus replication in ciliated epithelial cells and pneumocytes with associated lesions throughout the respiratory tract. Am. J. Pathol. 2004, 164, 1893–1900. [Google Scholar] [CrossRef]
- Alvarez, R.; Tripp, R.A. The immune response to human metapneumovirus is associated with aberrant immunity and impaired virus clearance in BALB/c mice. J. Virol. 2005, 79, 5971–5978. [Google Scholar] [CrossRef] [PubMed]
- Darniot, M.; Petrella, T.; Aho, S.; Pothier, P.; Manoha, C. Immune response and alteration of pulmonary function after primary human metapneumovirus (hMPV) infection of BALB/c mice. Vaccine 2005, 23, 4473–4480. [Google Scholar] [CrossRef]
- Herd, K.A.; Nelson, M.; Mahalingam, S.; Tindle, R.W. Pulmonary infection of mice with human metapneumovirus induces local cytotoxic T-cell and immunoregulatory cytokine responses similar to those seen with human respiratory syncytial virus. J. Gen. Virol. 2010, 91, 1302–1310. [Google Scholar] [CrossRef]
- Cheemarla, N.R.; Banos-Lara, M.D.; Naidu, S.; Guerrero-Plata, A. Neutrophils regulate the lung inflammatory response via gamma delta T cell infiltration in an experimental mouse model of human metapneumovirus infection. J. Leukoc. Biol. 2017, 101, 1383–1392. [Google Scholar] [CrossRef]
- Kolli, D.; Bataki, E.L.; Spetch, L.; Guerrero-Plata, A.; Jewell, A.M.; Piedra, P.A.; Milligan, G.N.; Garofalo, R.P.; Casola, A. T lymphocytes contribute to antiviral immunity and pathogenesis in experimental human metapneumovirus infection. J. Virol. 2008, 82, 8560–8569. [Google Scholar] [CrossRef]
- Darniot, M.; Pitoiset, C.; Petrella, T.; Aho, S.; Pothier, P.; Manoha, C. Age-Associated Aggravation of Clinical Disease after Primary Metapneumovirus Infection of BALB/c Mice. J. Virol. 2009, 83, 3323–3332. [Google Scholar] [CrossRef]
- Huck, B.; Neumann-Haefelin, D.; Schmitt-Graeff, A.; Weckmann, M.; Mattes, J.; Ehl, S.; Falcone, V. Human metapneumovirus induces more severe disease and stronger innate immune response in BALB/c mice as compared with respiratory syncytial virus. Respir. Res. 2007, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Hamelin, M.E.; Couture, C.; Sackett, M.K.; Boivin, G. Enhanced lung disease and Th2 response following human metapneumovirus infection in mice immunized with the inactivated virus. J. Gen. Virol. 2007, 88, 3391–3400. [Google Scholar] [CrossRef] [PubMed]
- Lay, M.K.; Céspedes, P.F.; Palavecino, C.E.; León, M.A.; Díaz, R.A.; Salazar, F.J.; Méndez, G.P.; Bueno, S.M.; Kalergis, A.M. Human metapneumovirus infection activates the TSLP pathway that drives excessive pulmonary inflammation and viral replication in mice. Eur. J. Immunol. 2015, 45, 1680–1695. [Google Scholar] [CrossRef] [PubMed]
- Amarasinghe, G.K.; Bao, Y.; Basler, C.F.; Bavari, S.; Beer, M.; Bejerman, N.; Blasdell, K.R.; Bochnowski, A.; Briese, T.; Bukreyev, A.; et al. Taxonomy of the order Mononegavirales: Update 2017. Arch. Virol. 2017, 162, 2493–2504. [Google Scholar] [CrossRef] [PubMed]
- Chanock, R.M.; Parrott, R.H.; Cook, K.; Andrews, B.E.; Bell, J.A.; Reichelderfer, T.; Kapikian, A.Z.; Mastrota, F.M.; Huebner, R.J. Newly recognized myxoviruses from children with respiratory disease. N. Engl. J. Med. 1958, 258, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Canchola, J.; Christmann, E.; Kim, H.W.; Parrott, R.H.; Chanock, R.M.; Vargosko, A.J.; Jeffries, B. Antigenic Variation among Newly Isolated Strains of Parainfluenza Type 4 Virus. Am. J. Hyg. 1964, 79, 357–364. [Google Scholar] [PubMed]
- Henrickson, K.J.; Savatski, L.L. Two distinct human parainfluenza virus type 1 genotypes detected during the 1991 Milwaukee epidemic. J. Clin. Microbiol. 1996, 34, 695–700. [Google Scholar]
- Prinoski, K.; Cote, M.J.; Kang, C.Y.; Dimock, K. Evolution of the fusion protein gene of human parainfluenza virus 3. Virus Res. 1992, 22, 55–69. [Google Scholar] [CrossRef]
- Henrickson, K.J.; Kuhn, S.M.; Savatski, L.L. Epidemiology and cost of infection with human parainfluenza virus types 1 and 2 in young children. Clin. Infect. Dis. 1994, 18, 770–779. [Google Scholar] [CrossRef]
- Fiore, A.E.; Iverson, C.; Messmer, T.; Erdman, D.; Lett, S.M.; Talkington, D.F.; Anderson, L.J.; Fields, B.; Carlone, G.M.; Breiman, R.F.; et al. Outbreak of pneumonia in a long-term care facility: Antecedent human parainfluenza virus 1 infection may predispose to bacterial pneumonia. J. Am. Geriatr. Soc. 1998, 46, 1112–1117. [Google Scholar] [CrossRef]
- Prince, G.A.; Porter, D.D. Treatment of parainfluenza virus type 3 bronchiolitis and pneumonia in a cotton rat model using topical antibody and glucocorticosteroid. J. Infect. Dis. 1996, 173, 598–608. [Google Scholar] [CrossRef] [PubMed]
- Downham, M.A.P.; Mcquillin, J.; Gardner, P.S. Diagnosis and Clinical Significance of Parainfluenza Virus-Infections in Children. Arch. Dis. Child. 1974, 49, 8–15. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Weinberg, G.A.; Hall, C.B.; Iwane, M.K.; Poehling, K.A.; Edwards, K.M.; Griffin, M.R.; Staat, M.A.; Curns, A.T.; Erdman, D.D.; Szilagyi, P.G.; et al. Parainfluenza Virus Infection of Young Children: Estimates of the Population-Based Burden of Hospitalization. J. Pediatr. 2009, 154, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Mao, N.Y.; Ji, Y.X.; Xie, Z.D.; Wang, H.H.; Wang, H.L.; An, J.J.; Zhang, X.X.; Zhang, Y.; Zhu, Z.; Cui, A.L.; et al. Human Parainfluenza Virus-Associated Respiratory Tract Infection among Children and Genetic Analysis of HPIV-3 Strains in Beijing, China. PLoS ONE 2012, 7, e0043893. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, A.; Munoz, J.; Montecalvo, M.; Clones, B.; LaGamma, E.F. Outbreak of Parainfluenza Virus Type 3 in a Neonatal Intensive Care Unit. Am. J. Perinat. 2009, 26, 361–364. [Google Scholar] [CrossRef] [PubMed]
- Costa, L.D.C.; Camargos, P.A.M.; Brand, P.L.P.; Fiaccadori, F.S.; Souza, M.B.D.D.E.; Cardoso, D.D.D.; Castro, I.D.; Minamisavas, R.; da Costa, P.S.S. Asthma exacerbations in a subtropical area and the role of respiratory viruses: A cross-sectional study. BMC Pulm. Med. 2018, 18, 109. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Zhao, L.Q.; Zhu, R.N.; Deng, J.; Sun, Y.; Ding, Y.X.; Tian, R.; Qian, Y. Parainfluenza Virus Types 1, 2, and 3 in Pediatric Patients with Acute Respiratory Infections in Beijing During 2004 to 2012. Chin. Med. J. 2015, 128, 2726–2730. [Google Scholar]
- Matsuse, H.; Kondo, Y.; Saeki, S.; Nakata, H.; Fukushima, C.; Mizuta, Y.; Kohno, S. Naturally occurring parainfluenza virus 3 infection in adults induces mild exacerbation of asthma associated with increased sputum concentrations of cysteinyl leukotrienes. Int. Arch. Allergy Immunol. 2005, 138, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Ottolini, M.G.; Porter, D.D.; Blanco, J.C.; Prince, G.A. A cotton rat model of human parainfluenza 3 laryngotracheitis: Virus growth, pathology, and therapy. J. Infect. Dis. 2002, 186, 1713–1717. [Google Scholar] [CrossRef] [PubMed]
- Mascoli, C.C.; Gower, T.A.; Capilupo, F.A.; Metzgar, D.P. Further studies on the neonatal ferret model of infection and immunity to and attenuation of human parainfluenza viruses. Dev. Biol. Stand. 1976, 33, 384–390. [Google Scholar] [PubMed]
- Chang, P.W.; Hsiung, G.D. Experimental infection of parainfluenza virus type 5 in mice, hamsters and monkeys. J. Immunol. 1965, 95, 591–601. [Google Scholar] [PubMed]
- Burke, C.W.; Bridges, O.; Brown, S.; Rahija, R.; Russell, C.J. Mode of parainfluenza virus transmission determines the dynamics of primary infection and protection from reinfection. PLoS Pathog. 2013, 9, e1003786. [Google Scholar] [CrossRef] [PubMed]
- Akk, A.M.; Simmons, P.M.; Chan, H.W.; Agapov, E.; Holtzman, M.J.; Grayson, M.H.; Pham, C.T. Dipeptidyl peptidase I-dependent neutrophil recruitment modulates the inflammatory response to Sendai virus infection. J. Immunol. 2008, 180, 3535–3542. [Google Scholar] [CrossRef] [PubMed]
- McWilliam, A.S.; Marsh, A.M.; Holt, P.G. Inflammatory infiltration of the upper airway epithelium during Sendai virus infection: Involvement of epithelial dendritic cells. J. Virol. 1997, 71, 226–236. [Google Scholar] [PubMed]
- Grayson, M.H.; Ramos, M.S.; Rohlfing, M.M.; Kitchens, R.; Wang, H.D.; Gould, A.; Agapov, E.; Holtzman, M.J. Controls for lung dendritic cell maturation and migration during respiratory viral infection. J. Immunol. 2007, 179, 1438–1448. [Google Scholar] [CrossRef]
- Kim, E.Y.; Battaile, J.T.; Patel, A.C.; You, Y.; Agapov, E.; Grayson, M.H.; Benoit, L.A.; Byers, D.E.; Alevy, Y.; Tucker, J.; et al. Persistent activation of an innate immune response translates respiratory viral infection into chronic lung disease. Nat. Med. 2008, 14, 633–640. [Google Scholar] [CrossRef]
- Grayson, M.H.; Cheung, D.; Rohlfing, M.M.; Kitchens, R.; Spiegel, D.E.; Tucker, J.; Battaile, J.T.; Alevy, Y.; Yan, L.; Agapov, E.; et al. Induction of high-affinity IgE receptor on lung dendritic cells during viral infection leads to mucous cell metaplasia. J. Exp. Med. 2007, 204, 2759–2769. [Google Scholar] [CrossRef]
- Cheung, D.S.; Ehlenbach, S.J.; Kitchens, R.T.; Riley, D.A.; Thomas, L.L.; Holtzman, M.J.; Grayson, M.H. Cutting Edge: CD49d+ Neutrophils Induce FcεRI Expression on Lung Dendritic Cells in a Mouse Model of Postviral Asthma. J. Immunol. 2010, 185, 4983–4987. [Google Scholar] [CrossRef]
- Thomas, M.A.; Buelow, B.J.; Nevins, A.M.; Jones, S.E.; Peterson, F.C.; Gundry, R.L.; Grayson, M.H.; Volkman, B.F. Structure-function analysis of CCL28 in the development of post-viral asthma. J. Biol. Chem. 2015, 290, 4528–4536. [Google Scholar] [CrossRef]
- Cheung, D.S.; Sigua, J.A.; Simpson, P.M.; Yan, K.; Hussain, S.R.A.; Santoro, J.L.; Buell, E.J.; Hunter, D.A.; Rohlfing, M.; Patadia, D.; et al. Cysteinyl leukotriene receptor 1 expression identifies a subset of neutrophils during the antiviral response that contributes to postviral atopic airway disease. J. Allergy Clin. Immunol. 2018, 142, 1206–1217. [Google Scholar] [CrossRef]
- Mori, I.; Komatsu, T.; Takeuchi, K.; Nakakuki, K.; Sudo, M.; Kimura, Y. Parainfluenza virus type 1 infects olfactory neurons and establishes long-term persistence in the nerve tissue. J. Gen. Virol. 1995, 76 Pt 5, 1251–1254. [Google Scholar] [CrossRef]
- Lee, A.M.; Fryer, A.D.; van Rooijen, N.; Jacoby, D.B. Role of macrophages in virus-induced airway hyperresponsiveness and neuronal M2 muscarinic receptor dysfunction. Am. J. Physiol. Lung Cell Mol. Physiol. 2004, 286, L1255–L1259. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Castleman, W.L.; Sorkness, R.L.; Lemanske, R.F.; Grasee, G.; Suyemoto, M.M. Neonatal Viral Bronchiolitis and Pneumonia Induces Bronchiolar Hypoplasia and Alveolar Dysplasia in Rats. Lab. Investig. 1988, 59, 387–396. [Google Scholar] [PubMed]
- Castleman, W.L.; Sorkness, R.L.; Lemanske, R.F., Jr.; McAllister, P.K. Viral bronchiolitis during early life induces increased numbers of bronchiolar mast cells and airway hyperresponsiveness. Am. J. Pathol. 1990, 137, 821–831. [Google Scholar] [PubMed]
- Kumar, A.; Sorkness, R.L.; Kaplan, M.R.; Lemanske, R.F., Jr. Chronic, episodic, reversible airway obstruction after viral bronchiolitis in rats. Am. J. Respir. Crit. Care Med. 1997, 155, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Sorkness, R.L.; Gern, J.E.; Grindle, K.A.; Mosser, A.G.; Rosenthal, L.A.; Mikus, L.D.; Lemanske, R.F., Jr. Persistence of viral RNA in 2 rat strains differing in susceptibility to postbronchiolitis airway dysfunction. J. Allergy Clin. Immunol. 2002, 110, 607–609. [Google Scholar] [CrossRef] [PubMed]
- Mikus, L.D.; Rosenthal, L.A.; Sorkness, R.L.; Lemanske, R.F., Jr. Reduced interferon-gamma secretion by natural killer cells from rats susceptible to postviral chronic airway dysfunction. Am. J. Respir. Cell. Mol. Biol. 2001, 24, 74–82. [Google Scholar] [CrossRef]
- Rosenthal, L.A.; Mikus, L.D.; Tuffaha, A.; Mosser, A.G.; Sorkness, R.L.; Lemanske, R.F., Jr. Attenuated innate mechanisms of interferon-gamma production in rats susceptible to postviral airway dysfunction. Am. J. Respir. Cell. Mol. Biol. 2004, 30, 702–709. [Google Scholar] [CrossRef]
- Uhl, E.W.; Clarke, T.J.; Hogan, R.J. Differential expression of nuclear factor-kappaB mediates increased pulmonary expression of tumor necrosis factor-alpha and virus-induced asthma. Viral. Immunol. 2009, 22, 79–89. [Google Scholar] [CrossRef]
- Uhl, E.W.; Clarke, T.J.; Lester, C.; Hogan, R.J. Rats Susceptible to Virus-Induced Asthma Have a Persistent Virus-Induced Change in the Predominant Pulmonary Form of the NF-kappa B Inhibitor I kappa B alpha. Vet. Pathol. 2010, 47, 1021–1027. [Google Scholar] [CrossRef]
- Allander, T.; Tammi, M.T.; Eriksson, M.; Bjerkner, A.; Tiveljung-Lindell, A.; Andersson, B. Cloning of a human parvovirus by molecular screening of respiratory tract samples. Proc. Natl. Acad. Sci. USA 2005, 102, 12891–12896. [Google Scholar] [CrossRef] [PubMed]
- Bastien, N.; Brandt, K.; Dust, K.; Ward, D.; Li, Y. Human Bocavirus infection, Canada. Emerg. Infect. Dis. 2006, 12, 848–850. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.K.; Yip, C.C.; Que, T.L.; Lee, R.A.; Au-Yeung, R.K.; Zhou, B.; So, L.Y.; Lau, Y.L.; Chan, K.H.; Woo, P.C.; et al. Clinical and molecular epidemiology of human bocavirus in respiratory and fecal samples from children in Hong Kong. J. Infect. Dis. 2007, 196, 986–993. [Google Scholar] [CrossRef] [PubMed]
- Allander, T.; Jartti, T.; Gupta, S.; Niesters, H.G.; Lehtinen, P.; Osterback, R.; Vuorinen, T.; Waris, M.; Bjerkner, A.; Tiveljung-Lindell, A.; et al. Human bocavirus and acute wheezing in children. Clin. Infect. Dis. 2007, 44, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Kesebir, D.; Vazquez, M.; Weibel, C.; Shapiro, E.D.; Ferguson, D.; Landry, M.L.; Kahn, J.S. Human bocavirus infection in young children in the United States: Molecular epidemiological profile and clinical characteristics of a newly emerging respiratory virus. J. Infect. Dis. 2006, 194, 1276–1282. [Google Scholar] [CrossRef] [PubMed]
- Vallet, C.; Pons-Catalano, C.; Mandelcwajg, A.; Wang, A.; Raymond, J.; Lebon, P.; Gendrel, D. Human bocavirus: A cause of severe asthma exacerbation in children. J. Pediatr. 2009, 155, 286–288. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.; Feng, Z.; Sun, X.; Zhang, Y.; Zou, W.; Wang, Z.; Jensen-Cody, C.; Liang, B.; Park, S.Y.; Qiu, J.; et al. Human Bocavirus Type-1 Capsid Facilitates the Transduction of Ferret Airways by Adeno-Associated Virus Genomes. Hum. Gene Ther. 2017, 28, 612–625. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Chang, Y.J.; Subramanian, S.; Lee, H.H.; Albacker, L.A.; Matangkasombut, P.; Savage, P.B.; McKenzie, A.N.; Smith, D.E.; Rottman, J.B.; et al. Innate lymphoid cells responding to IL-33 mediate airway hyperreactivity independently of adaptive immunity. J. Allergy Clin. Immunol. 2012, 129, 216–227. [Google Scholar] [CrossRef]
- Juno, J.A.; Keynan, Y.; Fowke, K.R. Invariant NKT Cells: Regulation and Function during Viral Infection. PLoS Pathog. 2012, 8, e1002838. [Google Scholar] [CrossRef]
- Morita, H.; Moro, K.; Koyasu, S. Innate lymphoid cells in allergic and nonallergic inflammation. J. Allergy Clin. Immunol. 2016, 138, 1253–1264. [Google Scholar] [CrossRef]
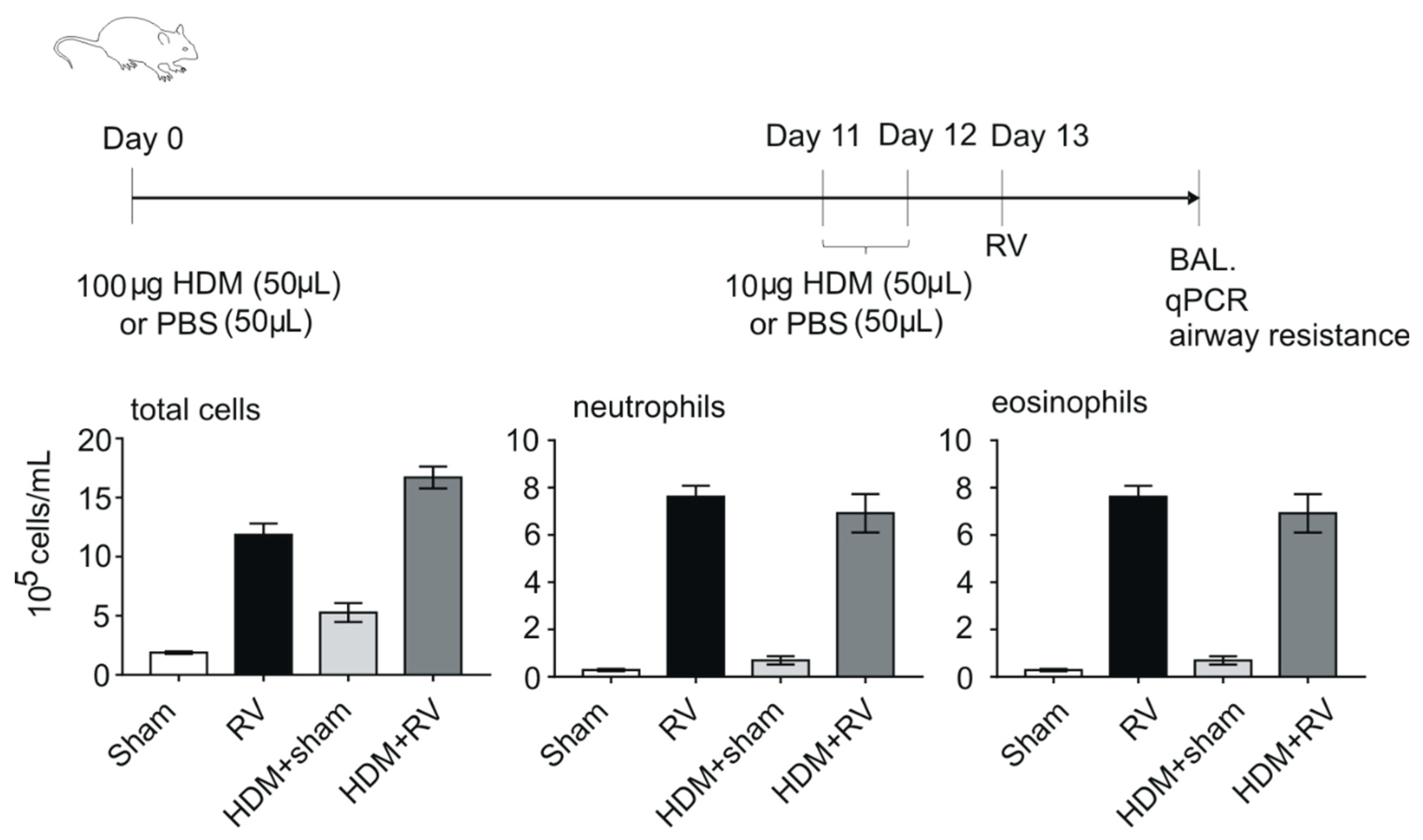
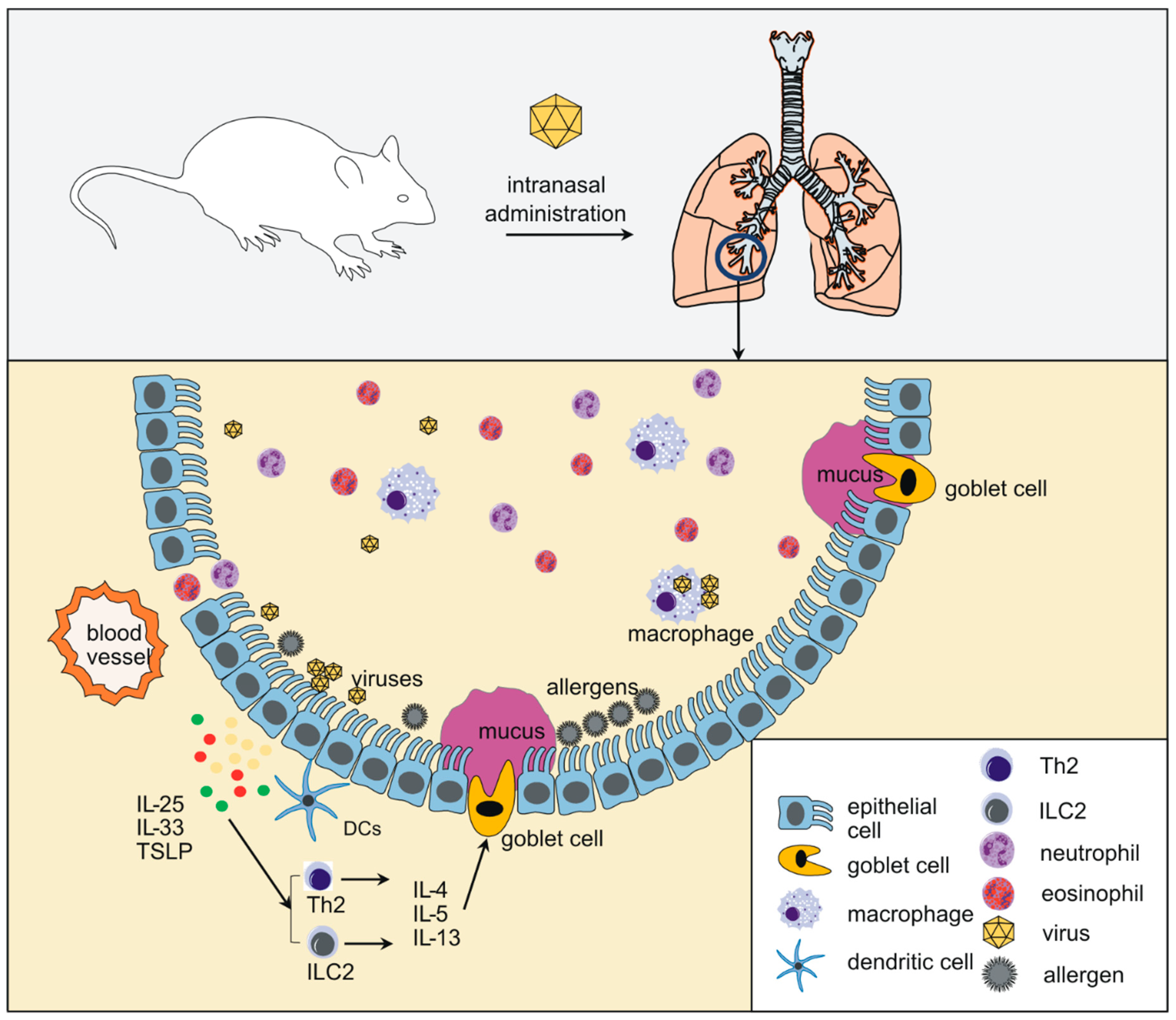
| Taxonomy | Species | |||||||
|---|---|---|---|---|---|---|---|---|
| Order | Family | Subfamily and Genus | Species | Mice | Rats | Guinea Pigs | Hamsters | Ferrets |
| Picornavirales | Picornaviridae | Enterovirus | Rhinovirus A-C | HRVA-1B [4,5] HRV-A16 [4] | HRVA-16 [6] HRVB-14 [7] | |||
| Enterovirus D | EV-D68 [8,9,10] Fermon and epidemic strains | EV-D68 [11] Fermon and epidemic strains | EV-D68 [12] Fermon strain | |||||
| Mononegavirales | Pneumoviridae | Orthopneumovirus | Human orthopneumovirus | human RSV-A [13] | human RSV-A [14] | human RSV-A [15] | human RSV-A [16] | human RSV-A [17] |
| Murine orthopneumovirus | PVM [18] | |||||||
| Metapneumovirus | hMPV [19] | hMPV [20] | hMPV [21] | hMPV [21,22] | hMPV [23] | |||
| Paramyxoviridae | Respirovirus | Human Respirovirus 1,3 | hPIV3 [24] | hPIV3 [25] | hPIV3 [26] | hPIV3 [27] | ||
| Murine Respirovirus | SeV [28] | SeV [29] | SeV [30] | SeV [31] | ||||
| Rubulavirus | Human Respirovirus 2,4 | |||||||
| Viruses | Species a | Age | Gender b | Route c | Virus Strains/Isolates | Inoculum d | Dura-Tion e | Detection Method | Applications and Major Observations | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| HRV | BALB/c mice | 6 weeks | F | i.n. | HRVA-1B | 5 × 106 TCID50 | 72–96 h | Viral titer/Viral RNA/ v(-) RNA | HRV induced airway inflammation with inflammatory infiltrates and increased expression of cytokines, chemokines and IFNs as well as mucus-related proteins | [4] |
| huICAM BALB/c mice | N/A | N/A | i.n. | HRVA-16 | 5 × 106 TCID50 | N/A | Viral RNA/ | |||
| C57BL/6 mice | 6–8 weeks | F | i.n. | HRVA-1B | 5 × 106 TCID50 | 96 h | Viral RNA/ v(-) RNA | HRV induced phosphatidylinositol 3-kinase dependent airway inflammation and airway responsiveness | [5] | |
| BALB/c mice | 6 days | N/A | i.n. | HRVA-1B | 2 × 106 TCID50 | 7 d * | Viral RNA/ | Neonatal model of HRV infection. Early-life HRV infection induced the development of asthma like phenotype which is IL-13 dependent and ILC2 expansion | [53,56] | |
| EV-D68 | BALB/c mice | 8–12 weeks | F | i.n. | US/MO/14-18947 | 5 × 106 epfu | 96 h | Viral RNA | EV-D68 infection induces IL-17-dependent airway inflammation and hyperresponsiveness which is greater than HRV in naïve mice | [9] |
| Human RSV | 20 strains | 3 days | N/A | i.n. | Long strain | 103.3–103.7 pfu | N/A | Viral titer | The susceptibility of HRSV infection is different among 20 strains of inbred 3-day old mice. DBA/2N is the most permissive strain. | [13] |
| 8 strains | 8–10 weeks | F | i.t. | A2 strain | 4 × 107 pfu | N/A | Viral titer | AKR/J is the most permissive among eight strains of adult mice for human RSV infection, C57BL/6 is the most resistant | [57] | |
| BALB/c mice | 1 day-32 weeks | N/A | i.n. | A2 strain Long strain | 104–107 pfu | 8–12 d * | Viral titer | Human RSV causes weight loss, bronchiolitis, pneumonia and increased CD8+ T cell and NK cell responses. RSV reinfection after early primary infection causes more severe disease. RSV persists in the lung. | [58,59,60,61,62,63,64] | |
| BALB/c mice | 6–8 weeks | N/A | i.n. | 6 clinical isolates | 105 pfu | N/A | N/A | The clinical RSV isolate A2001/2-20 caused more severe lung dysfunction, airway responsiveness, IL-13 and mucus expression | [65] | |
| BALB/c mice | 8 weeks | N/A | i.n. | A2001/2-20 strain | 3 × 106 pfu | N/A | N/A | In adult mice, RSV A2001/2-20 strain triggers expansion of IL-13 expressing ILC2s which is TSLP dependent. | [66] | |
| BALB/c mice | 5 days | N/A | i.n. | A2 strain rA2-19F strain | 104.68 TCID50/g | 6 d * | Viral RNA Viral titer | Neonatal infection with recombinant human RSV rA2-19F causes increased Th2 responses at primary infection and augmented airway hyperreactivity, mucus hyperproduction and eosinophilia during reinfection compared to the A2 strain. | [54] | |
| HMPV | BALB/c mice | 4–8 weeks | N/A | i.n. | NL/100 strain C-85473 strain CAN98-75 strain CAN98-83 strain | 5 × 105–108 pfu | 21–60 d | Virus titer | hMPV replicates with a biphasic growth kinetic and persists in the lung. hMPV induces both Th1 and Th2 responses. hMPV infection causes clinical symptoms (dyspnea and weight loss) as well as mucus production, airway hyperresponsiveness and obstruction. | [19,22,67,68,69] |
| 9 strains | 5–6 weeks | N/A | i.n. | TN/96-12 strain | 105 pfu | N/A | Virus titer | DBA/2 mice is the most permissive strain for hMPV. | [21] | |
| HPIV/ SeV | C57BL/6 mice | 6–12 weeks | N/A | i.n. | Fushimi strain | 2 × 105 pfu/ 5000 EID50 | N/A | N/A | Acute SeV infection leads to chronic airway hyperresponsiveness and mucus metaplasia, along with increased and maintained IL-13 expression by CD4+ T cells and macrophages. | [28,70,71] |
| BALB/c mice | 6–8 weeks | F | i.n. | N/A | 500 EIU | N/A | N/A | [72] |
| Viruses | Species | Age | Gender | Routes | Virus Strain/isolates | Inoculum | Duration | Detection Method | Reference |
|---|---|---|---|---|---|---|---|---|---|
| HRV | Cotton rat (Sigmodon hispidus) | 8 weeks | F/M | i.n. | HRVA-16 | 107 pfu | 48 h | Viral titer/v(-) RNA | [6] |
| Cotton rat (Sigmodon hispidus) | 4–6 weeks | N/A | i.n. | HRVB-14 | 3.97 × 106 pfu | 48 h | Viral titer/v(-) RNA | [7] | |
| EV-D68 | Cotton Rat (Sigmodon hispidus) | 6–8 weeks | F | i.n. | Fermon | 106 pfu | N/A | Viral titer | [11] |
| VANBT/1 | 106 pfu | 24 h | Viral titer/v(-) RNA | ||||||
| US/MO/14/18949 | 106 pfu | N/A | Viral titer | ||||||
| Ferret (Mustela putorius furo) | N/A | M | aerosol spray | Fermon | 104.5 CCID50 | 15 d * | Viral RNA | [12] | |
| Human RSV | Cotton rat (Sigmodon hispidus) | 1–28 days | N/A | i.n. | Long | 104 pfu | 7 d | Virus titer | [14] |
| Ferret (Mustela putorius furo) | 1–28 days | N/A | i.n. | Long | 3.6 × 103 pfu | 9 d | Virus titer | [17] | |
| guinea pigs (Cam Hartley) | N/A | Female | i.n. | Long | 3.9 × 103 pfu | 14 d * | Viral titer | [15,73] | |
| hMPV | Cotton rat (Sigmodon hispidus) | 5 weeks | N/A | i.n. | NL/100 | 106 pfu | N/A | N/A | [22] |
| 5–6 weeks | N/A | i.n. | TN/96-12 | 105 pfu | 8 d | Virus titer | [21] | ||
| N/A | N/A | i.n. | 26583(subtype A) 26575(subtype B) | 9 × 105 TCID50 | 14 d | N/A | [20] | ||
| Ferrets (Mustela putorius) | 5 weeks | N/A | i.n. | NL/100 | 106 pfu | N/A | N/A | [22] | |
| Hamster (Mesocricetus auratus) | 5 weeks | N/A | i.n. | NL/100 | 106 pfu | N/A | N/A | [22] | |
| 5–6 weeks | N/A | i.n. | TN/96-12 | 105 pfu | N/A | Virus titer | [21] | ||
| guinea pigs (Cavia porcellus) | 5–6 weeks | N/A | i.n. | TN/96-12 | 105 pfu | N/A | Virus titer | [21] | |
| hPIV | Cotton rat (Sigmodon hispidus) | N/A | N/A | i.n. | hPIV3/F518 | 105.8 pfu | 8 d | Virus titer | [24] |
| Cotton rat (Sigmodon fulviventer) | N/A | N/A | i.n. | hPIV3/F518 | 105.8 pfu | 8 d | Virus titer | [24] | |
| Ferret | 1 day | N/A | Aerosolization | HPIV3/224466 HPIV3/C243 | N/A | N/A | N/A | [27] | |
| Hamster (Mesocricetus auratus) | N/A | N/A | i.n. | hPIV3 strain C243 | 100–6000 pfu | 7–8 d | Virus titer | [26] | |
| SeV | Crl:CD(SD) rat | 5–25 days | N/A | i.n. | N/A | 102.4 TCID50 | 7–10 d | Virus titer | [74] |
| Crl:CD(SD) rat | 5–25 days | N/A | Aerosol exposure | N/A | 1.34 pfu/mL gas | N/A | N/A | [75] | |
| Crl:CD(SD) rat | 10 weeks | Male | Aerosol exposure | SeV P3193 | 1–3 pfu/mL gas | N/A | N/A | [76] | |
| Crl:CD(SD) rat | 5 days | N/A | Aerosol exposure | SeV P3193 | 1–2 pfu/mL gas | N/A | N/A | [77] | |
| Brown Norway rat | 5–25 days | N/A | aerosol exposure | SeV P3193 | 1–3 pfu/mL gas | N/A | N/A | [29] | |
| Fischer 334 rat | 5–25 days | N/A | aerosol exposure | SeV P3193 | 1–3 pfu/mL gas | N/A | N/A | ||
| Guinea pigs | N/A | Male | i.n. | SeV52 | 5 × 105 TCID50 | N/A | N/A | [30] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, M.; Rajput, C.; Ishikawa, T.; Jarman, C.R.; Lee, J.; Hershenson, M.B. Small Animal Models of Respiratory Viral Infection Related to Asthma. Viruses 2018, 10, 682. https://doi.org/10.3390/v10120682
Han M, Rajput C, Ishikawa T, Jarman CR, Lee J, Hershenson MB. Small Animal Models of Respiratory Viral Infection Related to Asthma. Viruses. 2018; 10(12):682. https://doi.org/10.3390/v10120682
Chicago/Turabian StyleHan, Mingyuan, Charu Rajput, Tomoko Ishikawa, Caitlin R. Jarman, Julie Lee, and Marc B. Hershenson. 2018. "Small Animal Models of Respiratory Viral Infection Related to Asthma" Viruses 10, no. 12: 682. https://doi.org/10.3390/v10120682
APA StyleHan, M., Rajput, C., Ishikawa, T., Jarman, C. R., Lee, J., & Hershenson, M. B. (2018). Small Animal Models of Respiratory Viral Infection Related to Asthma. Viruses, 10(12), 682. https://doi.org/10.3390/v10120682






