Effects of Staphylococcus aureus Bacteriophage K on Expression of Cytokines and Activation Markers by Human Dendritic Cells In Vitro
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Media
2.2. Bacteriophage Propagation and Purification
2.3. Phage Efficiency of Plating (EOP) Tests
2.4. Monocyte-Derived Dendritic Cells
2.5. Flow Cytometry
2.6. Bead-Based Immunoassays
2.7. Statistics
3. Results
3.1. Phage K Host Ranges on Military Clinical Isolates
3.2. Phage K Stability in PBS
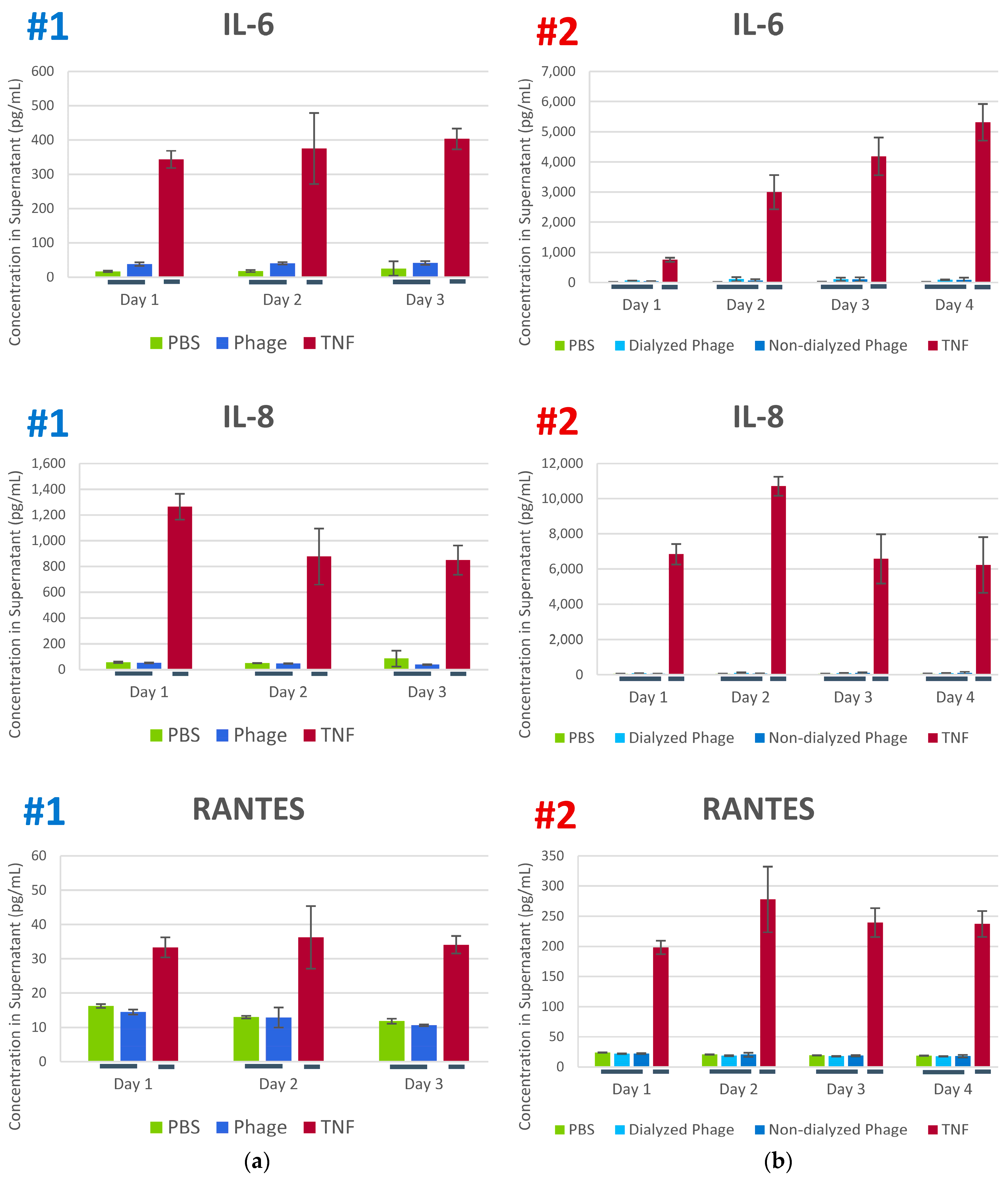
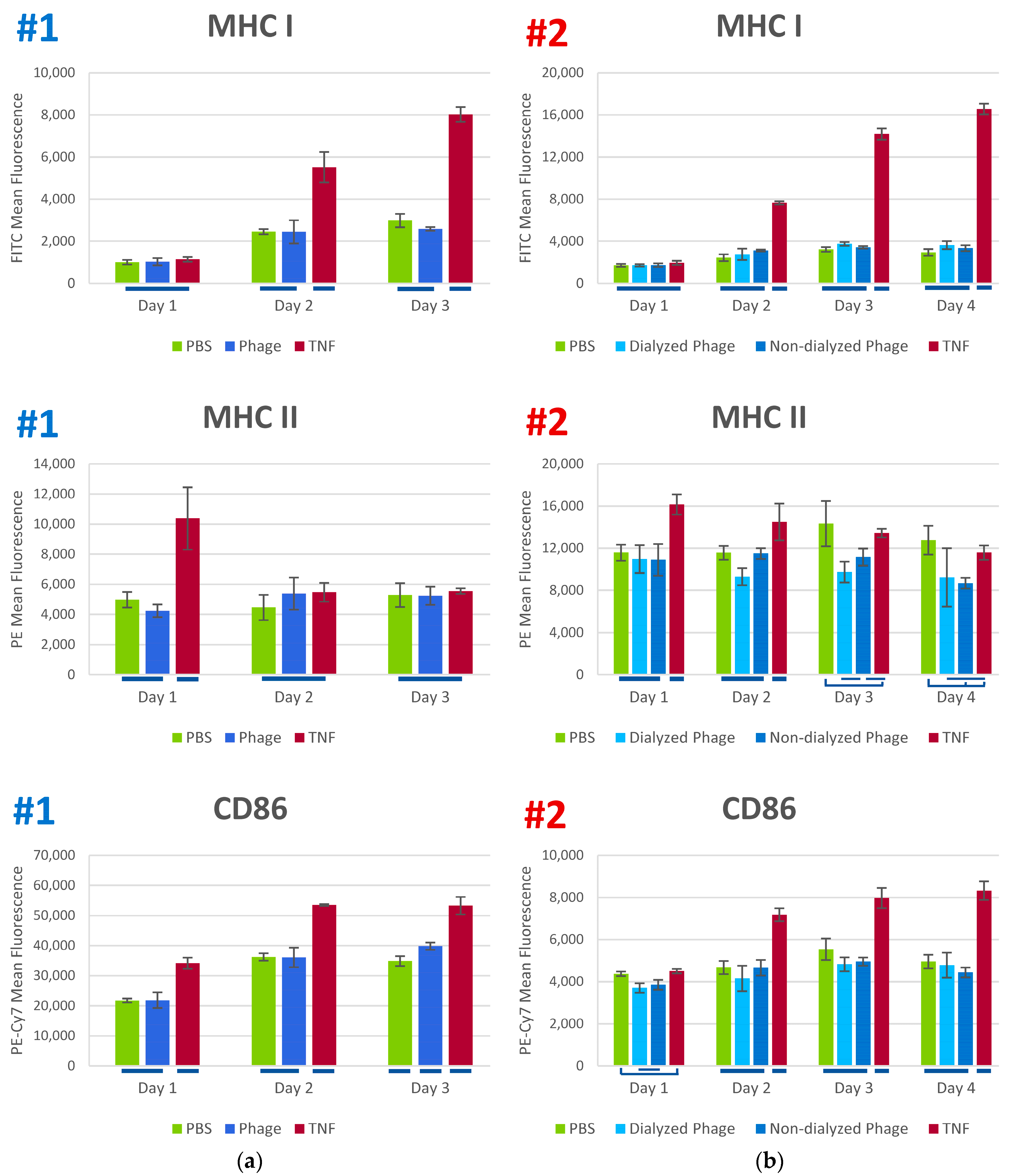
3.3. Phage K Effects on Human Dendritic Cells
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tong, S.Y.; Davis, J.S.; Eichenberger, E.; Holland, T.L.; Fowler, V.G., Jr. Staphylococcus aureus infections: Epidemiology, pathophysiology, clinical manifestations, and management. Clin. Microbiol. Rev. 2015, 28, 603–661. [Google Scholar] [CrossRef] [PubMed]
- Hiramatsu, K.; Katayama, Y.; Matsuo, M.; Sasaki, T.; Morimoto, Y.; Sekiguchi, A.; Baba, T. Multi-drug-resistant Staphylococcus aureus and future chemotherapy. J. Infect. Chemother. 2014, 20, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Lister, J.L.; Horswill, A.R. Staphylococcus aureus biofilms: Recent developments in biofilm dispersal. Front. Cell. Infect. Microbiol. 2014, 4, 178. [Google Scholar] [CrossRef] [PubMed]
- Kaźmierczak, Z.; Górski, A.; Dąbrowska, K. Facing antibiotic resistance: Staphylococcus aureus phages as a medical tool. Viruses 2014, 6, 2551–2570. [Google Scholar] [CrossRef] [PubMed]
- Nobrega, F.L.; Costa, A.R.; Kluskens, L.D.; Azeredo, J. Revisiting phage therapy: New applications for old resources. Trends Microbiol. 2015, 23, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Weber-Dąbrowska, B.; Jończyk-Matysiak, E.; Żaczek, M.; Łobocka, M.; Łusiak-Szelachowska, M.; Górski, A. Bacteriophage procurement for therapeutic purposes. Front. Microbiol. 2016, 7, 1177. [Google Scholar] [CrossRef] [PubMed]
- Górski, A.; Międzybrodzki, R.; Weber-Dąbrowska, B.; Fortuna, W.; Letkiewicz, S.; Rogóż, P.; Jończyk-Matysiak, E.; Dąbrowska, K.; Majewska, J.; Borysowski, J. Phage therapy: Combating infections with potential for evolving from merely a treatment for complications to targeting diseases. Front. Microbiol. 2016, 7, 1515. [Google Scholar] [CrossRef] [PubMed]
- Górski, A.; Międzybrodzki, R.; Borysowski, J.; Dąbrowska, K.; Wierzbicki, P.; Ohams, M.; Korczak-Kowalska, G.; Olszowska-Zaremba, N.; Łusiak-Szelachowska, M.; Kłak, M.; et al. Phage as a modulator of immune responses: Practical implications for phage therapy. Adv. Virus Res. 2012, 83, 41–71. [Google Scholar] [CrossRef] [PubMed]
- Jończyk-Matysiak, E.; Weber-Dąbrowska, B.; Owczarek, B.; Międzybrodzki, R.; Łusiak-Szelachowska, M.; Łodej, N.; Górski, A. Phage-phagocyte interactions and their implications for phage application as therapeutics. Viruses 2017, 9, 150. [Google Scholar] [CrossRef] [PubMed]
- Górski, A.; Dąbrowska, K.; Międzybrodzki, R.; Weber-Dąbrowska, B.; Łusiak-Szelachowska, M.; Jończyk-Matysiak, E.; Borysowski, J. Phages and immunomodulation. Future Microbiol. 2017, 12, 905–914. [Google Scholar] [CrossRef] [PubMed]
- Mirzaei, M.K.; Haileselassie, Y.; Navis, M.; Cooper, C.; Sverremark-Ekström, E.; Nilsson, A.S. Morphologically distinct Escherichia coli bacteriophages differ in their efficacy and ability to stimulate cytokine release in vitro. Front. Microbiol. 2016, 7, 437. [Google Scholar] [CrossRef]
- Dufour, N.; Henry, M.; Ricard, J.-D.; Debarbieux, L. Commentary: Morphologically distinct Escherichia coli bacteriophages differ in their efficacy and ability to stimulate cytokine release in vitro. Front. Microbiol. 2016, 7, 1029. [Google Scholar] [CrossRef] [PubMed]
- Miernikiewicz, P.; Dąbrowska, K.; Piotrowicz, A.; Owczarek, B.; Wojas-Turek, J.; Kicielińska, J.; Rossowska, J.; Pajtasz-Piasecka, E.; Hodyra, K.; Macegoniuk, K.; et al. T4 phage and its head surface proteins do not stimulate inflammatory mediator production. PLoS ONE 2013, 8, e71036. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Thimmapuram, J.; Zhang, J.; Collings, C.K.; Bhide, K.; Schmidt, K.; Ebner, P.D. The impact of orally administered phages on host immune response and surrounding microbial communities. Bacteriophage 2016, 6, e1211066. [Google Scholar] [CrossRef] [PubMed]
- Bocian, K.; Borysowski, J.; Zarzycki, M.; Pacek, M.; Weber-Dąbrowska, B.; Machcińska, M.; Korczak-Kowalska, G.; Górski, A. The effects of T4 and A3/R bacteriophages on differentiation of human myeloid dendritic cells. Front. Microbiol. 2016, 7, 1267. [Google Scholar] [CrossRef] [PubMed]
- Pajtasz-Piasecka, E.; Rossowska, J.; Duś, D.; Weber-Dabrowska, B.; Zabłocka, A.; Górski, A. Bacteriophages support anti-tumor response initiated by DC-based vaccine against murine transplantable colon carcinoma. Immunol. Lett. 2008, 116, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Miedzybrodzki, R.; Fortuna, W.; Weber-Dabrowska, B.; Górski, A. A retrospective analysis of changes in inflammatory markers in patients treated with bacterial viruses. Clin. Exp. Med. 2009, 9, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Miernikiewicz, P.; Kłopot, A.; Soluch, R.; Szkuta, P.; Kęska, W.; Hodyra-Stefaniak, K.; Konopka, A.; Nowak, M.; Lecion, D.; Kaźmierczak, Z.; et al. T4 phage tail adhesin gp12 counteracts LPS-induced inflammation in vivo. Front. Microbiol. 2016, 7, 1112. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zheng, P.; Ji, W.; Fu, Q.; Wang, H.; Yan, Y.; Sun, J. SLPW: A virulent bacteriophage targeting methicillin-resistant Staphylococcus aureus in vitro and in vivo. Front. Microbiol. 2016, 7, 934. [Google Scholar] [CrossRef] [PubMed]
- Shiley, J.R.; Comfort, K.K.; Robinson, J.B. Immunogenicity and antimicrobial effectiveness of Pseudomonas aeruginosa specific bacteriophage in a human lung in vitro model. Appl. Microbiol. Biotechnol. 2017, 101, 7977–7985. [Google Scholar] [CrossRef] [PubMed]
- Pincus, N.B.; Reckhow, J.D.; Saleem, D.; Jammeh, M.L.; Datta, S.K.; Myles, I.A. Strain specific phage treatment for Staphylococcus aureus infection is influenced by host immunity and site of infection. PLoS ONE 2015, 10, e0124280. [Google Scholar] [CrossRef] [PubMed]
- Barfoot, R.; Denham, S.; Gyure, L.A.; Hall, J.G.; Hobbs, S.M.; Jackson, L.E.; Robertson, D. Some properties of dendritic macrophages from peripheral lymph. Immunology 1989, 68, 233–239. [Google Scholar] [PubMed]
- Kaźmierczak, Z.; Piotrowicz, A.; Owczarek, B.; Hodyra, K.; Miernikiewicz, P.; Lecion, D.; Harhala, M.; Górski, A.; Dąbrowska, K. Molecular imaging of T4 phage in mammalian tissues and cells. Bacteriophage 2014, 4, e28364. [Google Scholar] [CrossRef] [PubMed]
- Jończyk-Matysiak, E.; Łusiak-Szelachowska, M.; Kłak, M.; Bubak, B.; Międzybrodzki, R.; Weber-Dąbrowska, B.; Żaczek, M.; Fortuna, W.; Rogóż, P.; Letkiewicz, S.; et al. The effect of bacteriophage preparations on intracellular killing of bacteria by phagocytes. J. Immunol. Res. 2015, 2015, 482863. [Google Scholar] [CrossRef] [PubMed]
- Weber-Dabrowska, B.; Zimecki, M.; Mulczyk, M.; Górski, A. Effect of phage therapy on the turnover and function of peripheral neutrophils. FEMS Immunol. Med. Microbiol. 2002, 34, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Roach, D.R.; Leung, C.Y.; Henry, M.; Morello, E.; Singh, D.; Di Santo, J.P.; Weitz, J.S.; Debarbieux, L. Synergy between the host immune system and bacteriophage is essential for successful phage therapy against an acute respiratory pathogen. Cell Host Microbe. 2017, 22, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Capparelli, R.; Parlato, M.; Borriello, G.; Salvatore, P.; Iannelli, D. Experimental phage therapy against Staphylococcus aureus in mice. Antimicrob. Agents Chemother. 2007, 51, 2765–2773. [Google Scholar] [CrossRef] [PubMed]
- Broxmeyer, L.; Sosnowska, D.; Miltner, E.; Chacón, O.; Wagner, D.; McGarvey, J.; Barletta, R.G.; Bermudez, L.E. Killing of Mycobacterium avium and Mycobacterium tuberculosis by a mycobacteriophage delivered by a nonvirulent mycobacterium: A model for phage therapy of intracellular bacterial pathogens. J. Infect. Dis. 2002, 186, 1155–1160. [Google Scholar] [CrossRef] [PubMed]
- Filippov, A.A.; Sergueev, K.V.; He, Y.; Huang, X.Z.; Gnade, B.T.; Mueller, A.J.; Fernandez-Prada, C.M.; Nikolich, M.P. Bacteriophage therapy of experimental bubonic plague in mice. Adv. Exp. Med. Biol. 2012, 954, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Dąbrowska, K.; Miernikiewicz, P.; Piotrowicz, A.; Hodyra, K.; Owczarek, B.; Lecion, D.; Kaźmierczak, Z.; Letarov, A.; Górski, A. Immunogenicity studies of proteins forming the T4 phage head surface. J. Virol. 2014, 88, 12551–12557. [Google Scholar] [CrossRef] [PubMed]
- Bruttin, A.; Brüssow, H. Human volunteers receiving Escherichia coli phage T4 orally: A safety test of phage therapy. Antimicrob. Agents Chemother. 2005, 49, 2874–2878. [Google Scholar] [CrossRef] [PubMed]
- Łusiak-Szelachowska, M.; Zaczek, M.; Weber-Dąbrowska, B.; Międzybrodzki, R.; Kłak, M.; Fortuna, W.; Letkiewicz, S.; Rogóż, P.; Szufnarowski, K.; Jończyk-Matysiak, E.; et al. Phage neutralization by sera of patients receiving phage therapy. Viral Immunol. 2014, 27, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Żaczek, M.; Łusiak-Szelachowska, M.; Jończyk-Matysiak, E.; Weber-Dąbrowska, B.; Międzybrodzki, R.; Owczarek, B.; Kopciuch, A.; Fortuna, W.; Rogóż, P.; Górski, A. Antibody production in response to staphylococcal MS-1 phage cocktail in patients undergoing phage therapy. Front. Microbiol. 2016, 7, 1681. [Google Scholar] [CrossRef]
- Łusiak-Szelachowska, M.; Żaczek, M.; Weber-Dąbrowska, B.; Międzybrodzki, R.; Letkiewicz, S.; Fortuna, W.; Rogóż, P.; Szufnarowski, K.; Jończyk-Matysiak, E.; Olchawa, E.; et al. Antiphage activity of sera during phage therapy in relation to its outcome. Future Microbiol. 2017, 12, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Krueger, A.P. A method for the quantitative determination of bacteriophage. J. Gen. Physiol. 1930, 13, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Deghorain, M.; Van Melderen, L. The staphylococci phages family: An overview. Viruses 2012, 4, 3316–3335. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, S.E.; Lo, H.H.; Chen, S.T.; Lee, M.C.; Tseng, Y.H. Wide host range and strong lytic activity of Staphylococcus aureus lytic phage Stau2. Appl. Environ. Microbiol. 2011, 77, 756–761. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.; McAuliffe, O.; Ross, R.P.; O’Mahony, J.; Coffey, A. Development of a broad-host-range phage cocktail for biocontrol. Bioeng. Bugs 2011, 2, 31–37. [Google Scholar] [CrossRef] [PubMed]
- O’Flaherty, S.; Ross, R.P.; Meaney, W.; Fitzgerald, G.F.; Elbreki, M.F.; Coffey, A. Potential of the polyvalent anti-Staphylococcus bacteriophage K for control of antibiotic-resistant staphylococci from hospitals. Appl. Environ. Microbiol. 2005, 71, 1836–1842. [Google Scholar] [CrossRef] [PubMed]
- Lungren, M.P.; Christensen, D.; Kankotia, R.; Falk, I.; Paxton, B.E.; Kim, C.Y. Bacteriophage K for reduction of Staphylococcus aureus biofilm on central venous catheter material. Bacteriophage 2013, 3, e26825. [Google Scholar] [CrossRef] [PubMed]
- Alves, D.R.; Gaudion, A.; Bean, J.E.; Perez Esteban, P.; Arnot, T.C.; Harper, D.R.; Kot, W.; Hansen, L.H.; Enright, M.C.; Jenkins, A.T. Combined use of bacteriophage K and a novel bacteriophage to reduce Staphylococcus aureus biofilm formation. Appl. Environ. Microbiol. 2014, 80, 6694–6703. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.; McAuliffe, O.; Ross, R.P.; Coffey, A. Prevention of Staphylococcus aureus biofilm formation and reduction in established biofilm density using a combination of phage K and modified derivatives. Lett. Appl. Microbiol. 2012, 54, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Lungren, M.P.; Donlan, R.M.; Kankotia, R.; Paxton, B.E.; Falk, I.; Christensen, D.; Kim, C.Y. Bacteriophage K antimicrobial-lock technique for treatment of Staphylococcus aureus central venous catheter-related infection: A leporine model efficacy analysis. J. Vasc. Interv. Radiol. 2014, 25, 1627–1632. [Google Scholar] [CrossRef] [PubMed]
- Wills, Q.F.; Kerrigan, C.; Soothill, J.S. Experimental bacteriophage protection against Staphylococcus aureus abscesses in a rabbit model. Antimicrob. Agents Chemother. 2005, 49, 1220–1221. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Xu, F. Dendritic cells during Staphylococcus aureus infection: Subsets and roles. J. Transl. Med. 2014, 12, 358. [Google Scholar] [CrossRef] [PubMed]
| Strain | Source | Tissue/Organ of Origin | MSSA or MRSA 1 | Phage K Susceptibility 2 | Phage K Titer (PFU/mL) | Phage K EOP 3 |
|---|---|---|---|---|---|---|
| NSCO308 | WID 4 | UNK 5 | MSSA | S | 6.0 × 1011 | 1.00 |
| MRSN18 | MRSN 6 | Wound | MRSA | S | 3.0 × 1011 | 0.50 |
| MRSN30 | MRSN | Wound | MRSA | S | 8.0 × 1010 | 0.13 |
| MRSN42 | MRSN | Wound | MRSA | S | 2.5 × 1011 | 0.42 |
| MRSN214 | MRSN | Wound | MRSA | S | 2.5 × 1010 | 0.04 |
| MRSN219 | MRSN | Urine | MRSA | S | 2.0 × 1011 | 0.33 |
| MRSN250 | MRSN | Urine | MRSA | S | 1.5 × 1011 | 0.25 |
| MRSN352 | MRSN | Wound | MRSA | S | 2.5 × 1011 | 0.42 |
| MRSN549 | MRSN | Wound | MRSA | S | 2.5 × 1011 | 0.42 |
| MRSN563 | MRSN | Wound | MRSA | S | 2.2 × 1011 | 0.37 |
| MRSN1722 | MRSN | Sputum | MRSA | S | 1.3 × 1010 | 0.02 |
| MRSN1732 | MRSN | Tissue | MRSA | S | 4.5 × 1011 | 0.75 |
| MRSN1952 | MRSN | Wound | MRSA | S | 1.0 × 1011 | 0.17 |
| MRSN2339 | MRSN | Wound | MRSA | S | 2.5 × 1010 | 0.04 |
| MRSN2763 | MRSN | Wound | MRSA | S | 9.5 × 1010 | 0.16 |
| MRSN3573 | MRSN | Blood | MRSA | R | 0 | 0 |
| MRSN3643 | MRSN | Tissue | MRSA | S | 1.3 × 1011 | 0.22 |
| MRSN3710 | MRSN | Wound | MRSA | S | 3.0 × 1011 | 0.50 |
| MRSN3966 | MRSN | Blood | MRSA | R | 0 | 0 |
| MRSN4109 | MRSN | Sputum | MRSA | S | 6.0 × 1010 | 0.10 |
| MRSN4344 | MRSN | Wound | MSSA | S | 2.0 × 1011 | 0.33 |
| MRSN4531 | MRSN | Wound | MRSA | S | 4.0 × 1011 | 0.67 |
| MRSN4535 | MRSN | Wound | MRSA | S | 8.0 × 1010 | 0.13 |
| MRSN5079 | MRSN | Wound | MSSA | S | 2.0 × 1011 | 0.33 |
| MRSN6168 | MRSN | Blood | MSSA | R | 0 | 0 |
| MRSN7983 | MRSN | Wound | MSSA | S | 1.5 × 1011 | 0.25 |
| MRSN8383 | MRSN | Wound | MSSA | S | 2.0 × 1011 | 0.33 |
| MRSN9127 | MRSN | Sputum | MSSA | S | 1.5 × 1011 | 0.25 |
| MRSN9287 | MRSN | Wound | MSSA | R | 0 | 0 |
| MRSN9832 | MRSN | Nasal swab | MRSA | R | 0 | 0 |
| MRSN9834 | MRSN | Nasal swab | MRSA | S | 4.0 × 1010 | 0.07 |
| MRSN10110 | MRSN | Wound | MSSA | S | 1.1 × 1011 | 0.18 |
| MRSN10185 | MRSN | Blood | MSSA | S | 7.5 × 1010 | 0.12 |
| MRSN12239 | MRSN | Eye | MSSA | S | 9.5 × 1011 | 1.58 |
| NAJAF22 | MRSN | UNK | MRSA | S | 1.2 × 1011 | 0.20 |
| NAJAF33 | MRSN | UNK | MRSA | R | 0 | 0 |
| Phage K Stock | Phage titer (PFU/mL) on: | ||||
|---|---|---|---|---|---|
| Day 1 | Day 9 | Day 15 | Day 23 | Day 31 | |
| Non-dialyzed | 5 × 1011 | 7 × 1011 | 6 × 1011 | 3 × 1011 | 7 × 1011 |
| Dialyzed | 7 × 1011 | 5 × 1011 | 6 × 1011 | 4 × 1011 | 3 × 1011 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freyberger, H.R.; He, Y.; Roth, A.L.; Nikolich, M.P.; Filippov, A.A. Effects of Staphylococcus aureus Bacteriophage K on Expression of Cytokines and Activation Markers by Human Dendritic Cells In Vitro. Viruses 2018, 10, 617. https://doi.org/10.3390/v10110617
Freyberger HR, He Y, Roth AL, Nikolich MP, Filippov AA. Effects of Staphylococcus aureus Bacteriophage K on Expression of Cytokines and Activation Markers by Human Dendritic Cells In Vitro. Viruses. 2018; 10(11):617. https://doi.org/10.3390/v10110617
Chicago/Turabian StyleFreyberger, Helen R., Yunxiu He, Amanda L. Roth, Mikeljon P. Nikolich, and Andrey A. Filippov. 2018. "Effects of Staphylococcus aureus Bacteriophage K on Expression of Cytokines and Activation Markers by Human Dendritic Cells In Vitro" Viruses 10, no. 11: 617. https://doi.org/10.3390/v10110617
APA StyleFreyberger, H. R., He, Y., Roth, A. L., Nikolich, M. P., & Filippov, A. A. (2018). Effects of Staphylococcus aureus Bacteriophage K on Expression of Cytokines and Activation Markers by Human Dendritic Cells In Vitro. Viruses, 10(11), 617. https://doi.org/10.3390/v10110617






