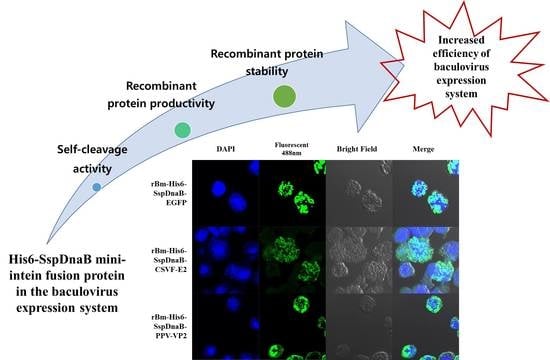
Enhanced Production of Recombinant Protein by Fusion Expression with Ssp DnaB Mini-Intein in the Baculovirus Expression System
Abstract
1. Introduction
2. Materials and Methods
2.1. Cells and Viruses
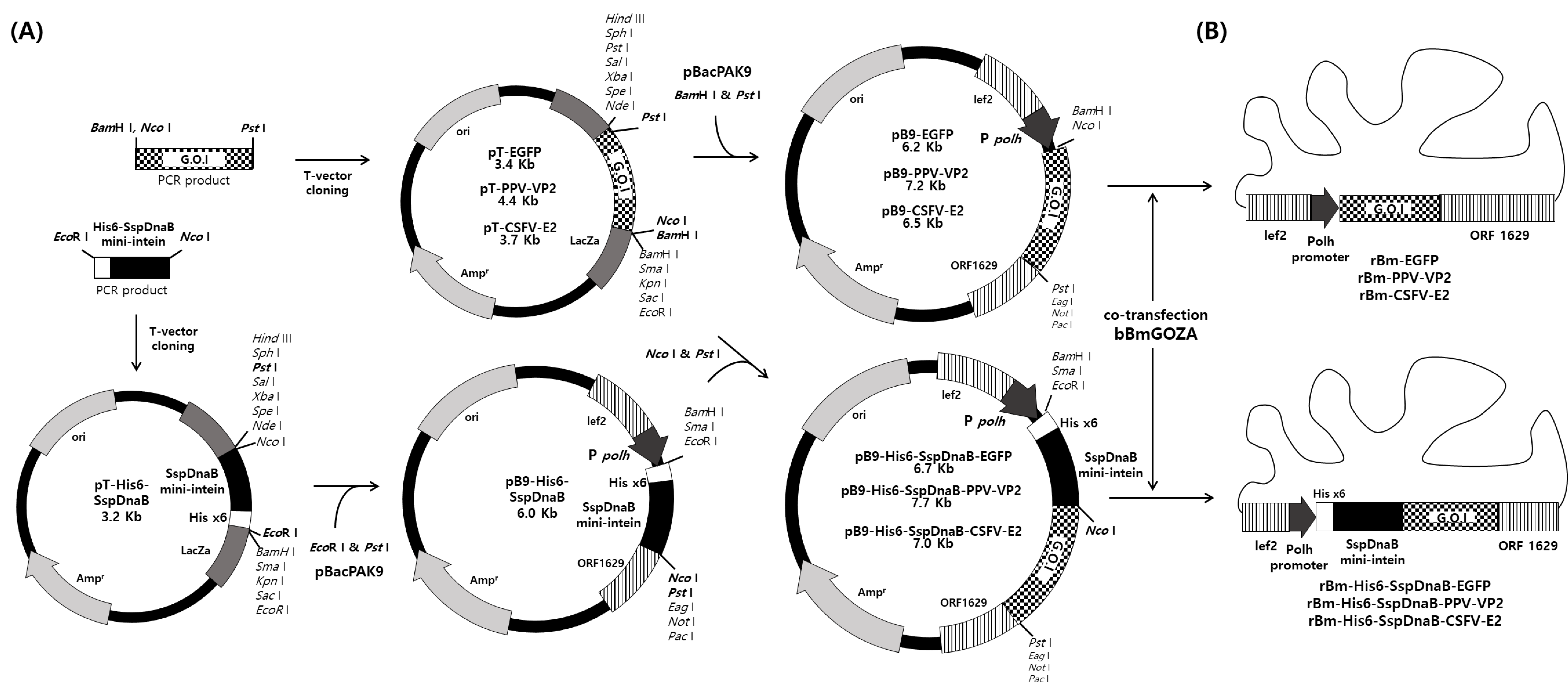
2.2. Construction of Transfer Vector
2.3. Generation of Recombinant Virus
2.4. Measurement of Fluorescence
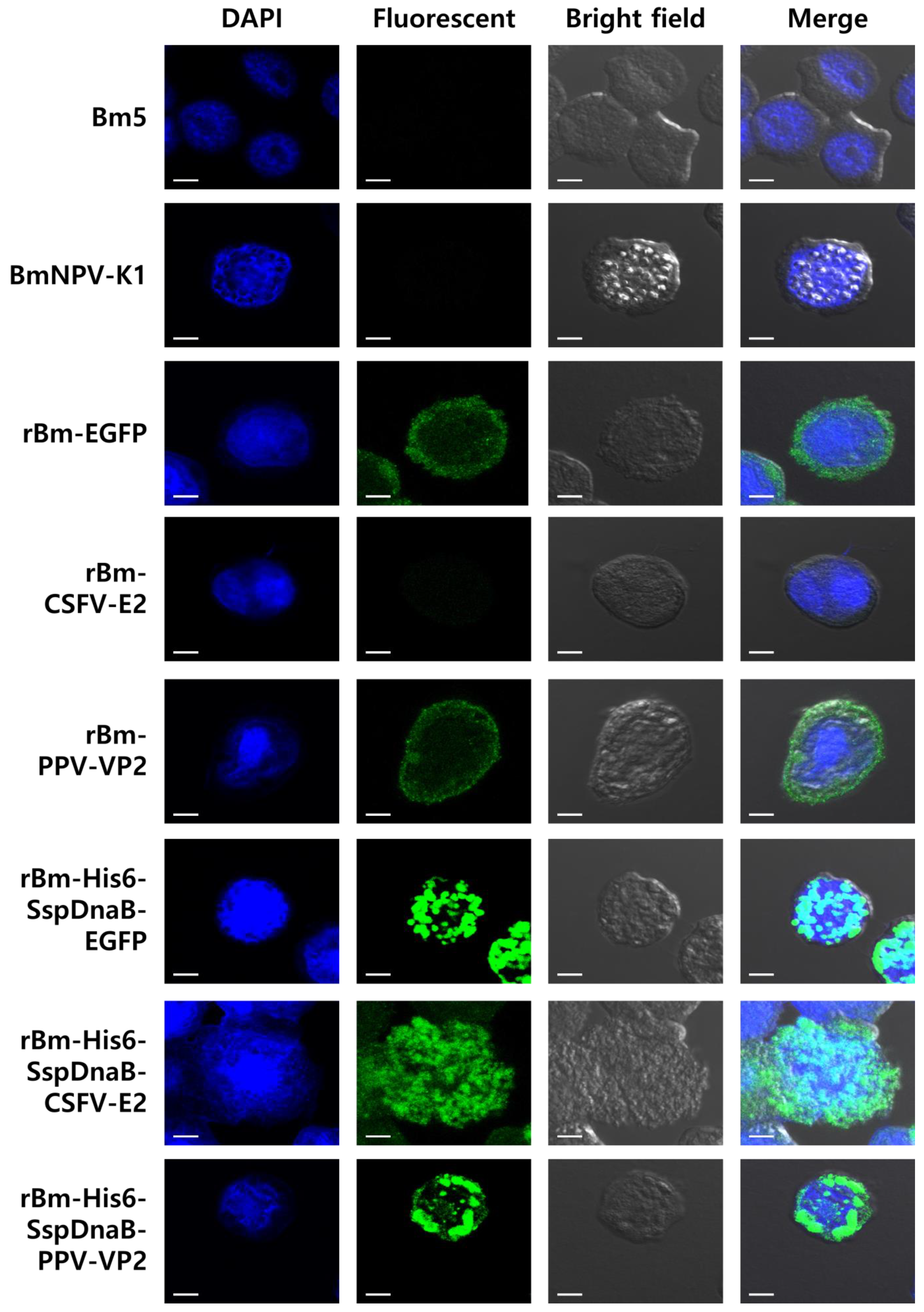
2.5. Immuno-Fluorescence Assay
2.6. SDS-PAGE and Western Blot Analysis
2.7. Temperature and pH Inducible Cleavage
3. Results and Discussion
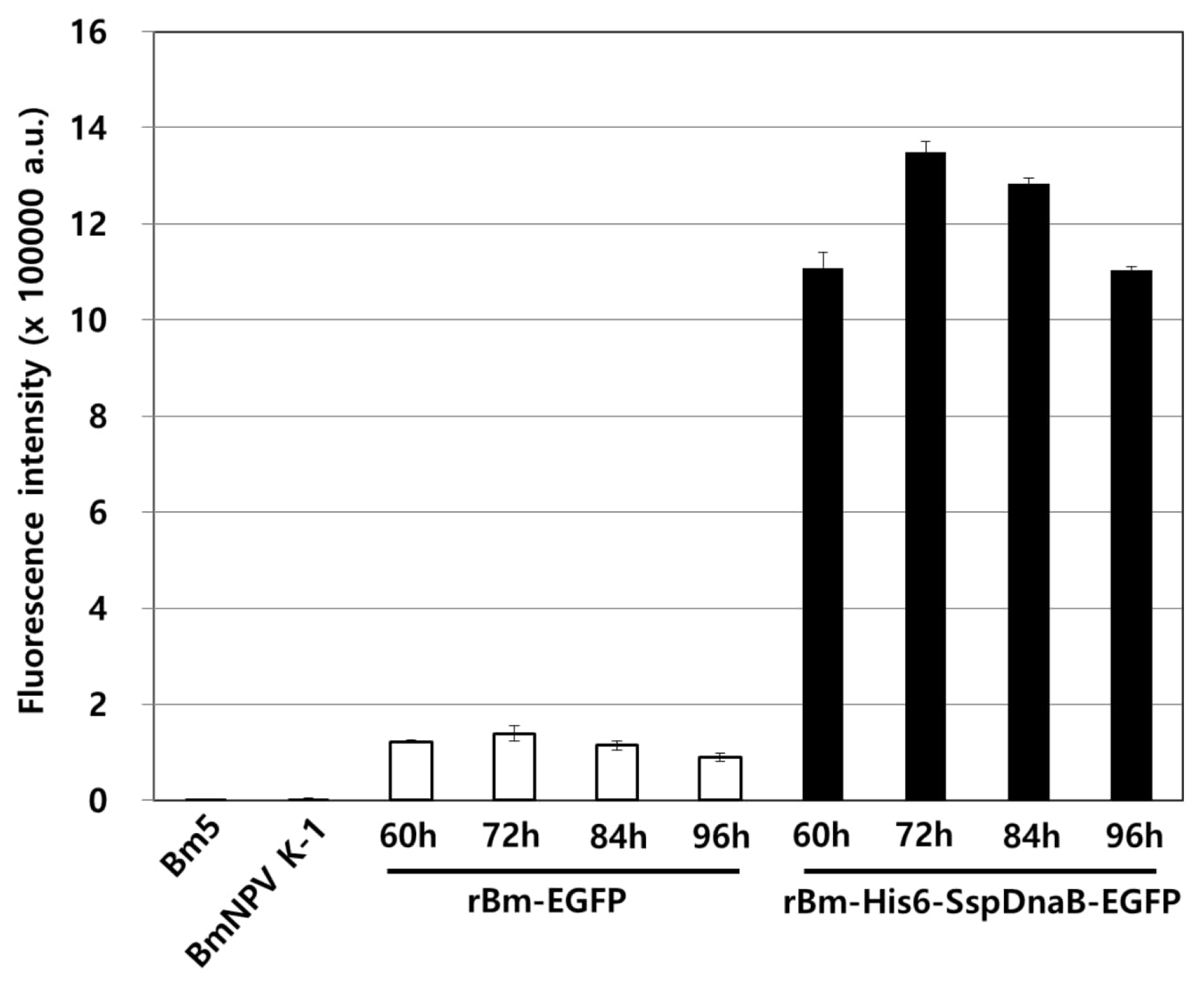
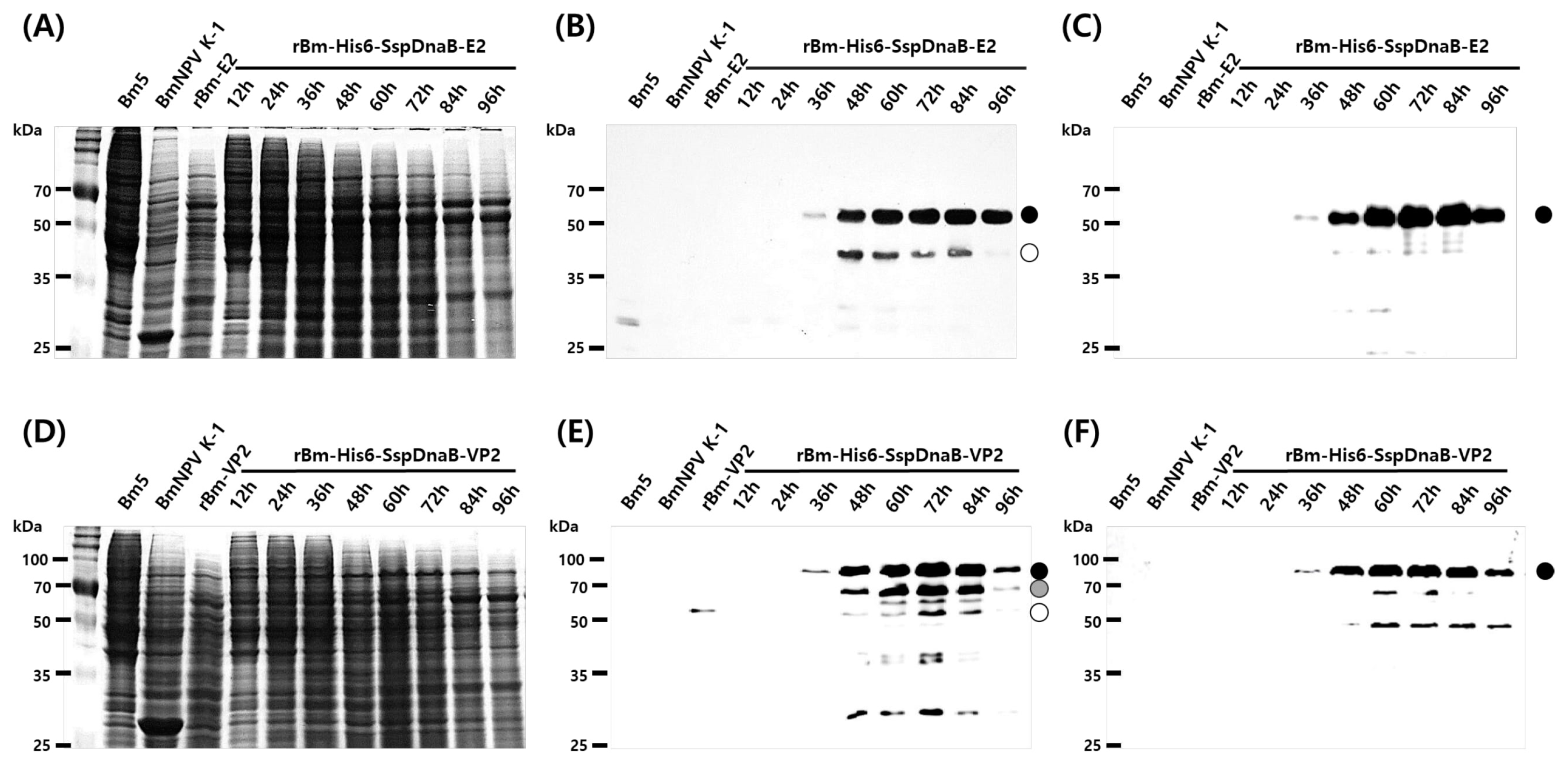
3.1. Effect of Mini-Intein Fusion Expression
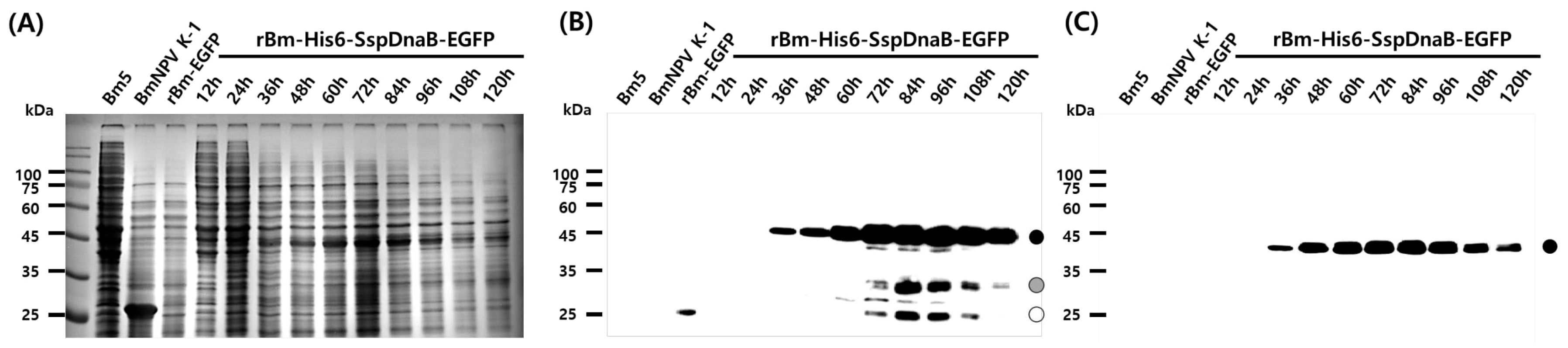
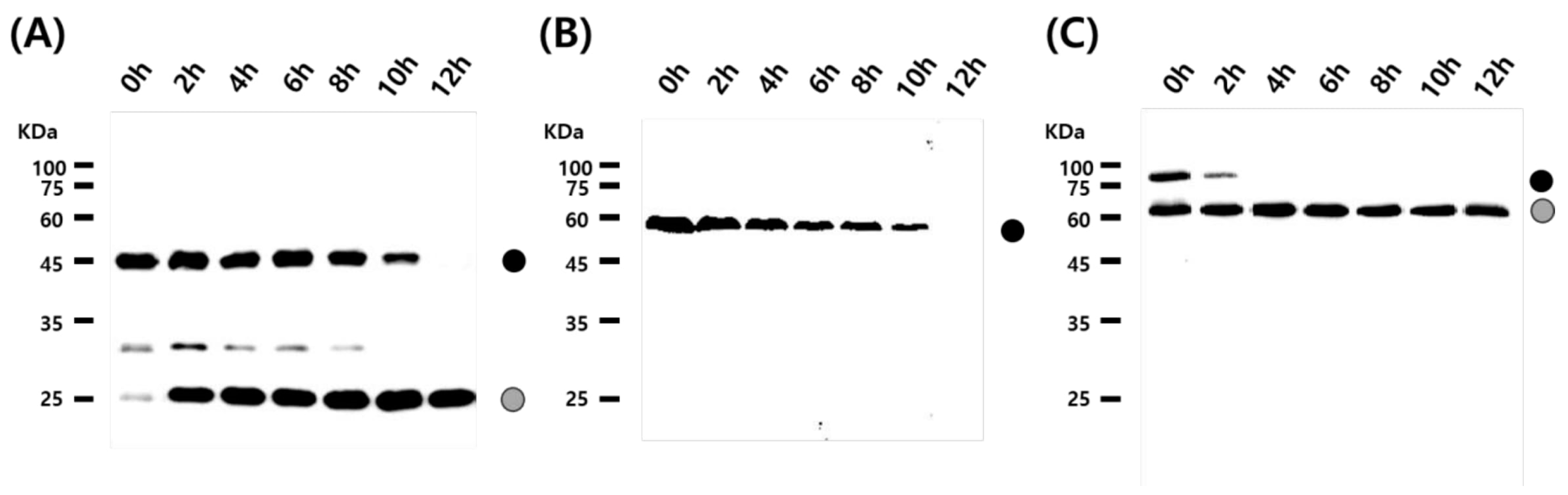
3.2. Cleavage Activity of the Mini-Intein
3.3. Status of the Mini-Intein Fusion Protein
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Radner, S.; Celie, P.H.N.; Fuchs, K.; Sieghart, W.; Sixma, T.K.; Stornaiuolo, M. Transient transfection coupled to baculovirus infection for rapid protein expression screening in insect cells. J. Struct. Biol. 2012, 179, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, L.; Kawar, S.; Hollister, R. Engineering N-glycosylation pathways in the baculovirus-insect cell system. Curr. Opin. Biotechnol. 1998, 9, 528–533. [Google Scholar] [CrossRef]
- Kato, T.; Kajikawa, M.; Maenaka, K.; Park, E.Y. Silkworm expression system as a platform technology in life science. Appl. Microbiol. 2010, 85, 459–470. [Google Scholar] [CrossRef] [PubMed]
- Hitchman, R.; Possee, R.; King, L. Baculovirus expression systems for recombinant protein production in insect cells. Recent Pat. Biotechnol. 2009, 3, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Carter, J.; Zhang, J.; Dang, T.L.; Hasegawa, H.; Cheng, J.D.; Gianan, I.; O’Neill, J.W.; Wolfson, M.; Siu, S.; Qu, S.; et al. Fusion partners can increase the expression of recombinant interleukins via transient transfection in 2936E cells. Protein Sci. 2010, 19, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Waugh, D.S. An overview of enzymatic reagents for the removal of affinity tags. Protein Expr. Purif. 2011, 80, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Li, Y. Self-cleaving fusion tags for recombinant protein production. Biotechnol. Lett. 2011, 33, 869–881. [Google Scholar] [CrossRef]
- Phan, J.; Zdanov, A.; Evdokimov, A.G.; Tropea, J.E.; Peters, H.K., 3rd; Kapust, R.B.; Li, M.; Wlodawer, A.; Waugh, D.S. Structural basis for the substrate specificity of tobacco etch virus protease. J. Biol. Chem. 2002, 277, 50564–50572. [Google Scholar] [CrossRef]
- Pedersen, J.; Lauritzen, C.; Madsen, M.T.; Weis Dahl, S. Removal of N-terminal polyhistidine tags from recombinant proteins using engineered aminopeptidases. Protein Expr. Purif. 1999, 15, 389–400. [Google Scholar] [CrossRef]
- Tan, H.; Wang, J.; Zhao, Z. Purification and refolding optimization of recombinant bovine enterokinase light chain overexpressed in Escherichia coli. Protein Expr. Purif. 2007, 56, 40–47. [Google Scholar] [CrossRef]
- Pietrokovski, S. A new intein in cyanobacteria and its significance for the spread of inteins. Trends Genet. 1996, 12, 287–288. [Google Scholar] [CrossRef]
- Wu, H.; Xu, M.Q.; Liu, X.Q. Protein trans-splicing and functional mini-inteins of a cyanobacterial DnaB Intein. Biochim. Biophys. Acta 1998, 1387, 422–432. [Google Scholar] [CrossRef]
- Mathys, S.; Evans, T.C.; Chute, I.C.; Wu, H.; Chong, S.; Benner, J.; Liu, X.-Q.; Xu, M.-Q. Characterization of a self-splicing mini-intein and its conversion into autocatalytic N- and C-terminal cleavage elements: Facile production of protein building blocks for protein ligation. Gene 1999, 231, 1–13. [Google Scholar] [CrossRef]
- Volkmann, G.; Mootz, H.D. Recent progress in intein research: From mechanism to directed evolution and applications. Cell. Mol. Life Sci. 2013, 70, 1185–1206. [Google Scholar] [CrossRef]
- Cooper, M.A.; Taris, J.E.; Shi, C.; Wood, D.W. A convenient split-intein tag method for the purification of tagless target proteins. Curr. Protoc. Protein Sci. 2018, 91, 5.29.1–5.29.23. [Google Scholar] [CrossRef]
- Ji, Y.; Lu, Y.; Yan, Y.; Liu, X.; Su, N.; Zhang, C.; Bi, S.; Xing, X.-H. Design of fusion proteins for efficient and soluble production of immunogenic Ebola virus glycoprotein in Escherichia coli. Biotechnol. J. 2018, 13, e1700627. [Google Scholar] [CrossRef]
- Cui, C.; Zhao, W.; Chen, J.; Wang, J.; Li, Q. Elimination of in vivo cleavage between target protein and intein in the intein-mediated protein purification systems. Protein Expr. Purif. 2006, 50, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Gui, Z.Z.; Lee, K.S.; Kim, B.Y.; Choi, Y.S.; Wei, Y.D.; Choo, Y.M.; Kang, P.D.; Yoon, H.J.; Kim, I.; Je, Y.H.; et al. Functional role of aspartic proteinase cathepsin D in insect metamorphosis. BMC Dev. Biol. 2006, 6, 49. [Google Scholar] [CrossRef] [PubMed]
- Hitchman, R.B.; Locanto, E.; Possee, R.D.; King, L.A. Optimizing the baculovirus expression vector system. Methods 2011, 55, 52–57. [Google Scholar] [CrossRef]
- Je, Y.H.; Chang, J.H.; Kim, M.H.; Roh, J.Y.; Jin, B.R.; O’reilly, D.R. The use of defective Bombyx mori nucleopolyhedrovirus genomes maintained in Escherichia coli for the rapid generation of occlusion-positive and occlusion-negative expression vectors. Biotechnol. Lett. 2001, 23, 1809–1817. [Google Scholar] [CrossRef]
- Bae, S.M.; Kim, H.J.; Lee, J.B.; Choi, J.B.; Shin, T.Y.; Koo, H.N.; Choi, J.Y.; Lee, K.S.; Je, Y.H.; Jin, B.R.; et al. Hyper-enhanced production of foreign recombinant protein by fusion with the partial polyhedrin of nucleopolyhedrovirus. PLoS ONE 2013, 8, e60835. [Google Scholar] [CrossRef]
- Ramirez, M.; Guan, D.; Ugaz, V.; Chen, Z. Intein-triggered artificial protein hydrogels that support the immobilization of bioactive proteins. J. Am. Chem. Soc. 2013, 135, 5290–5293. [Google Scholar] [CrossRef]
- Guan, D.; Ramirez, M.; Chen, Z. Split intein mediated ultra-rapid purification of tag less protein (SIRP): Rapid purification of tagless protein using SIRP. Biotechnol. Bioeng. 2013, 110, 2471–2481. [Google Scholar] [CrossRef] [PubMed]
- Topilina, N.I.; Mills, K.V. Recent advances in in vivo applications of intein-mediated protein splicing. Mob. DNA 2014, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.Y.; Tsai, T.Y.; Liu, T.T.; Lin, C.C.; Chen, J.H.; Yang, S.C.; Shieh, C.J.; Liu, Y.C. Production of recombinant EGFP via surface display of ice nucleation protein and self-cleavage intein. Biochem. Eng. J. 2011, 54, 158–163. [Google Scholar] [CrossRef]
- Banki, M.R.; Feng, L.; Wood, D.W. Simple bioseparations using self-cleaving elastin-like polypeptide Tags. Nat. Methods 2005, 2, 659–662. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Chen, J.; Yao, H.; Liu, L.; Wang, J.; Zhang, J.; Liu, J.-N. Use of Ssp DnaB derived mini-intein as a fusion partner for production of recombinant human brain natriuretic peptide in Escherichia coli. Protein Expr. Purif. 2005, 43, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.M.; Hsuan, S.L.; Chen, Z.W.; Jinn, T.R.; Huang, C.; Liao, J.W.; Chen, T.H.; Liao, C.M.; Lee, W.C.; Wu, T.Y.; et al. Expression and immunological studies of classical swine fever virus glycoprotein E2 in the bi-cistronic baculovirus/larvae expression system. Biosci. Biotechnol. Biochem. 2010, 74, 1343–1349. [Google Scholar] [CrossRef] [PubMed]
- Tong, Y.; Gao, J.; Zhou, J.; Yuan, L. Optimizing the method for cleavage of Ssp DnaB mini-intein mediated precursor proteins. J. Anim. Vet. Adv. 2013, 12, 1537–1540. [Google Scholar] [CrossRef]
- Seo, J.H.; Li, L.; Yeo, J.S.; Cha, H.J. Baculoviral polyhedrin as a novel fusion partner for formation of inclusion body in Escherichia coli. Biotechnol. Bioeng. 2003, 84, 467–473. [Google Scholar] [CrossRef]
- Park, J.S.; Ahn, J.Y.; Lee, S.H.; Lee, H.; Han, K.Y.; Seo, H.S.; Ahn, K.Y.; Min, B.H.; Sim, S.J.; Choi, I.S.; et al. Enhanced stability of heterologous proteins by supramolecular self-assembly. Appl. Microbiol. Biotechnol. 2007, 75, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Kim, Y.S.; Hwang, D.S.; Seo, J.H.; Jung, H.J.; Du, J.; Cha, H.J. High and compact formation of baculoviral polyhedrin-induced inclusion body by co-expression of baculoviral FP25 in Escherichia coli. Biotechnol. Bioeng. 2007, 96, 1183–1190. [Google Scholar] [CrossRef]
- Ahn, J.Y. Heterologous gene expression using self-assembled supra-molecules with high affinity for hsp70 chaperone. Nucleic Acids Res. 2005, 33, 3751–3762. [Google Scholar] [CrossRef] [PubMed]
| Name of Primer | Primer Sequence * |
|---|---|
| His6-SspDnaB-F | 5′-GAATTCGCCACCATGCACCACCACCACCACCACATCTCTGGCGATAG-3′ |
| SspDnaB-R | 5′-CCATGGCTCTTCCGTTGTGTAC-3′ |
| EGFP-F | 5′-GAATTCCCATGGTGAGCAAGGGCGAGGAGCTG-3′ |
| EGFP-R | 5′-CTGCAGTTACTTGTACAGCTCGTCCATGCCG-3′ |
| PPV-VP2-F | 5′-GAATTCCCATGGCAATGAGTGAAAATGTGGAACAACA-3′ |
| PPV-VP2-R | 5′-CTGCAGCTAGTATAATTTTCTTGGTATAAGTTGTG-3′ |
| CSFV-E2-F | 5′-GAACCTCCATGGCAATGCGGCTAGCCTGCA-3′ |
| CSFV-E2-R | 5′-CTGCAGTCAGTCAGTCACGTCCAGG-3′ |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gwak, W.S.; Choi, J.B.; Han, B.K.; Bae, S.M.; Woo, S.D. Enhanced Production of Recombinant Protein by Fusion Expression with Ssp DnaB Mini-Intein in the Baculovirus Expression System. Viruses 2018, 10, 523. https://doi.org/10.3390/v10100523
Gwak WS, Choi JB, Han BK, Bae SM, Woo SD. Enhanced Production of Recombinant Protein by Fusion Expression with Ssp DnaB Mini-Intein in the Baculovirus Expression System. Viruses. 2018; 10(10):523. https://doi.org/10.3390/v10100523
Chicago/Turabian StyleGwak, Won Seok, Jae Bang Choi, Beom Ku Han, Sung Min Bae, and Soo Dong Woo. 2018. "Enhanced Production of Recombinant Protein by Fusion Expression with Ssp DnaB Mini-Intein in the Baculovirus Expression System" Viruses 10, no. 10: 523. https://doi.org/10.3390/v10100523
APA StyleGwak, W. S., Choi, J. B., Han, B. K., Bae, S. M., & Woo, S. D. (2018). Enhanced Production of Recombinant Protein by Fusion Expression with Ssp DnaB Mini-Intein in the Baculovirus Expression System. Viruses, 10(10), 523. https://doi.org/10.3390/v10100523