Bacteriophage GC1, a Novel Tectivirus Infecting Gluconobacter Cerinus, an Acetic Acid Bacterium Associated with Wine-Making
Abstract
:1. Introduction
2. Material and Methods
2.1. Strains and Media
2.2. Isolation of Acetic Acid Bacteria from Grapes
2.3. AAB Identification
2.4. Isolation and Purification of Phages Infecting AAB
2.5. Production of High-Titres Lysates
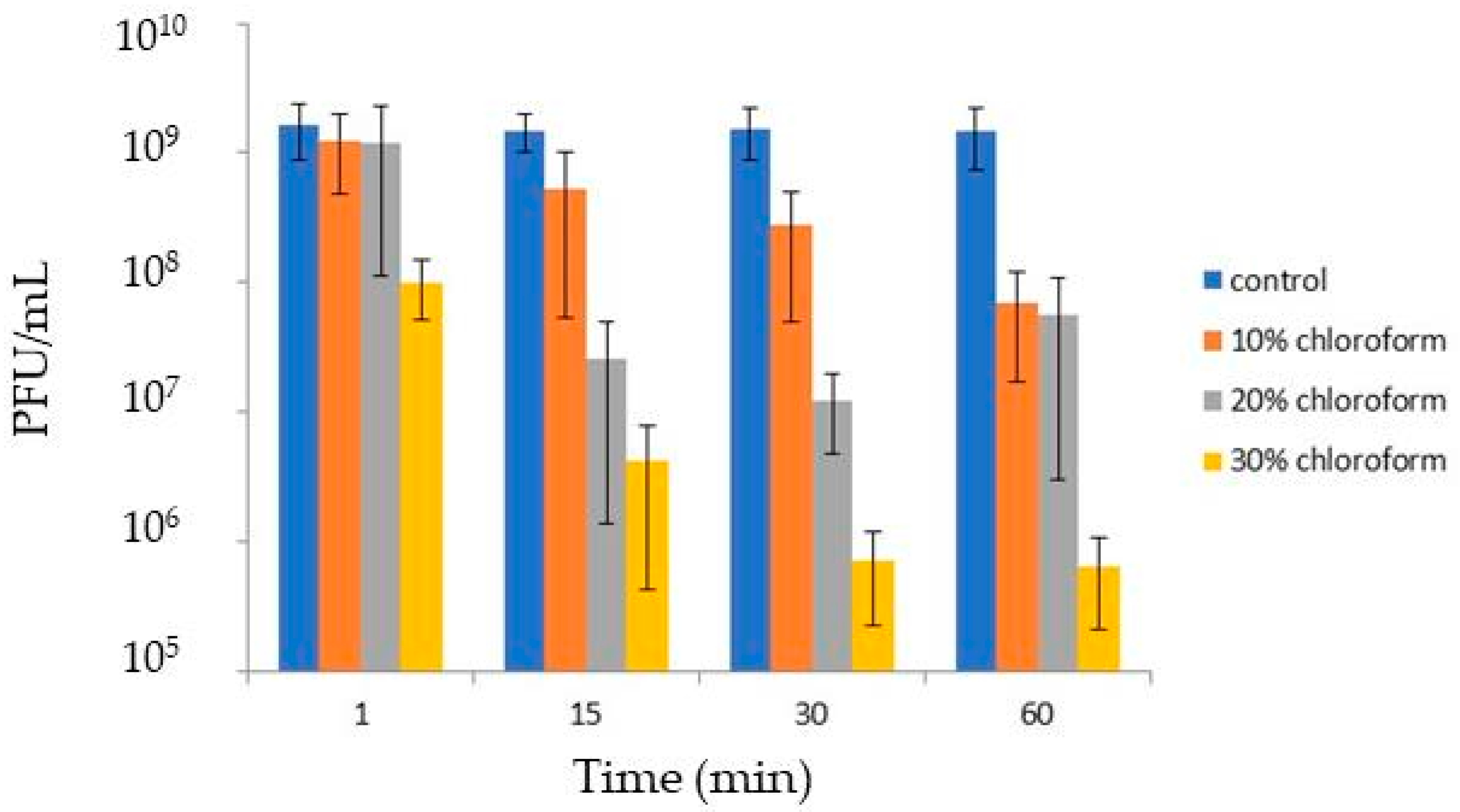
2.6. Chloroform Sensitivity Assay
2.7. Concentration of Phage
2.8. Electron Microscopy
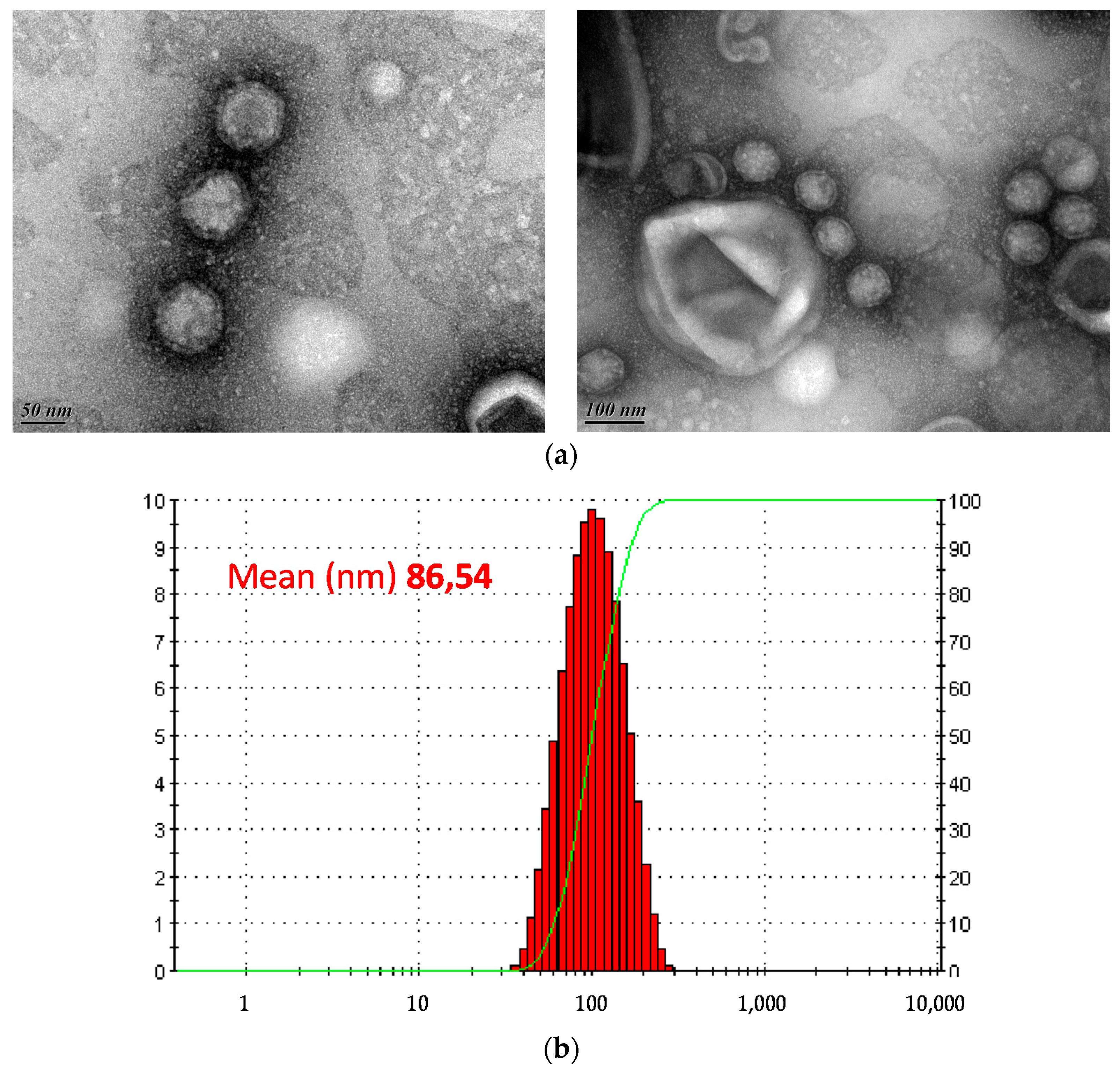
2.9. Dynamic Light Scattering (DLS)
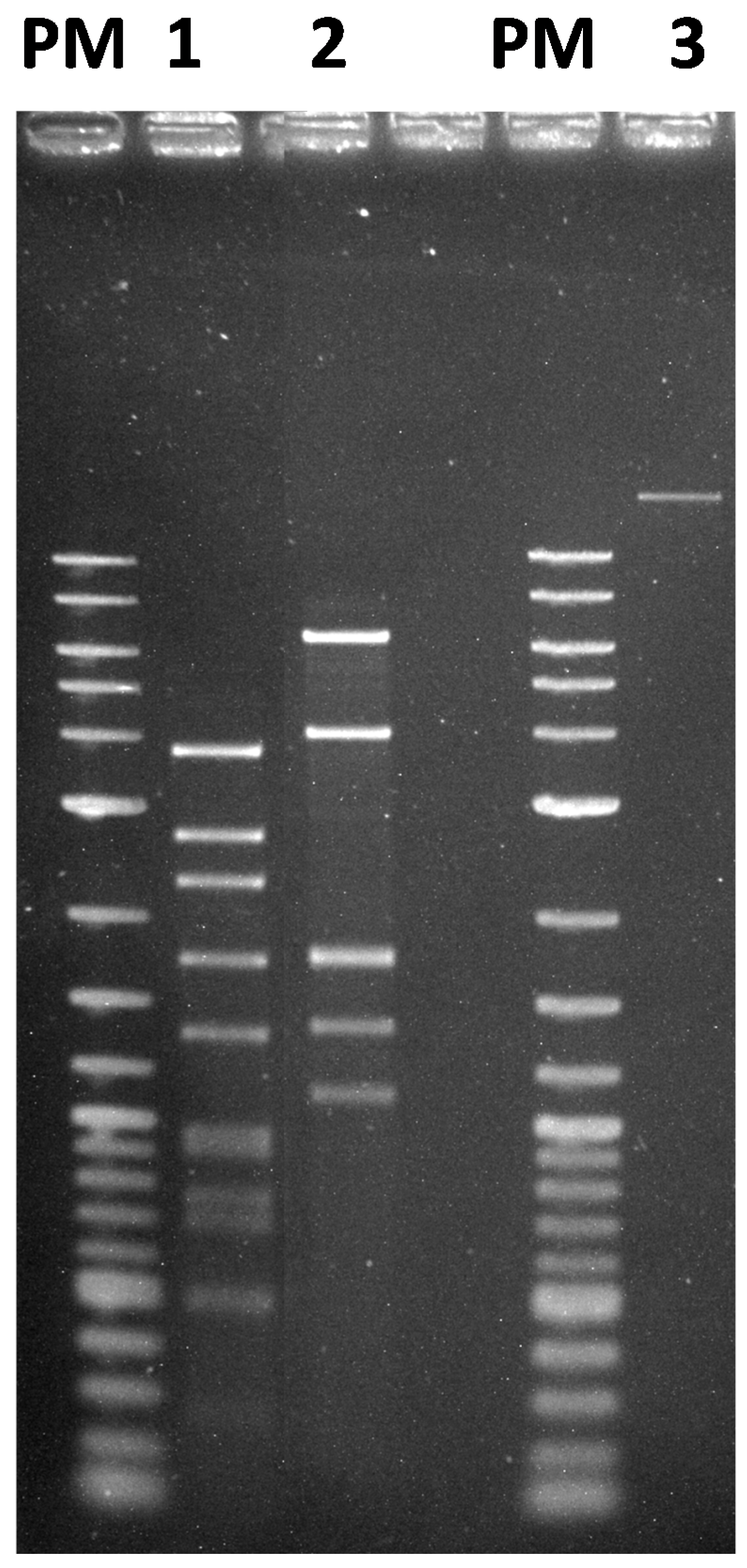
2.10. Extraction of Phage DNA and Restriction Digestion
2.11. DNA Sequencing
2.12. Bacterial Lysogen Generation
2.13. Homology Searches and Phylogenetic Analyses
3. Results and Discussion
3.1. Isolation of GC1 from Enological Samples
3.2. Host Spectrum
3.3. Morphology and Genome Analysis
3.4. Virion Stability
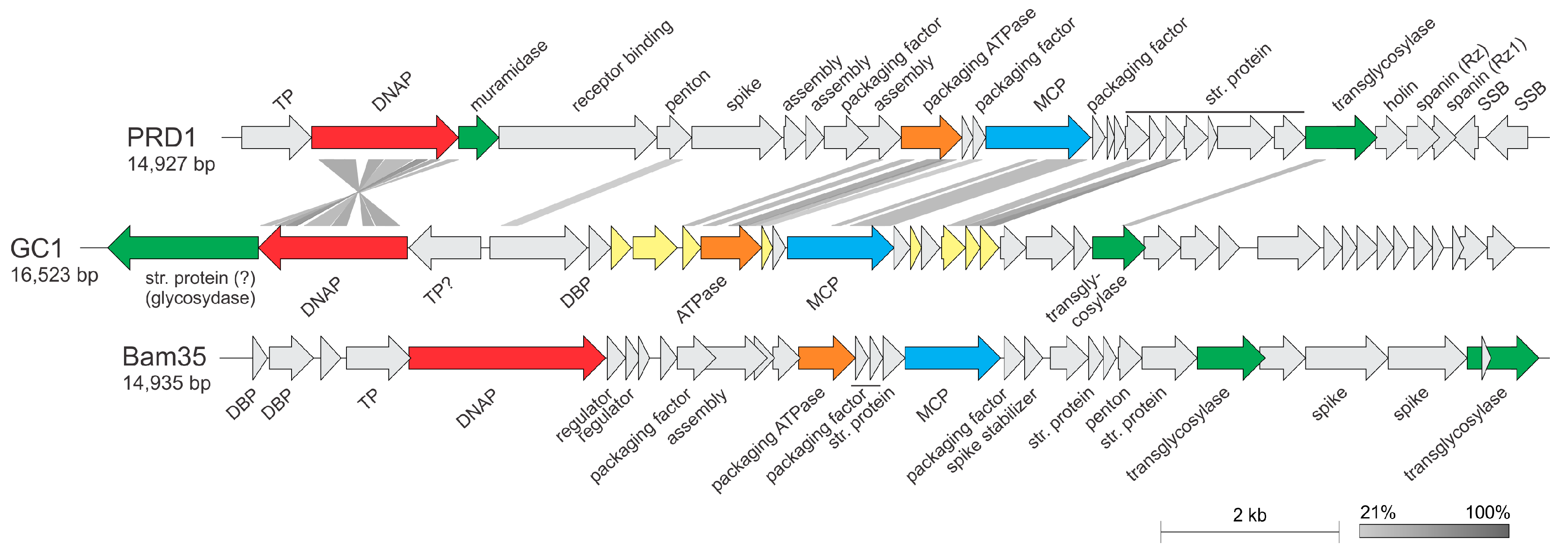
3.5. ORF Function Assignment and Genomic Organization
3.6. GC1 Is a Temperate Phage
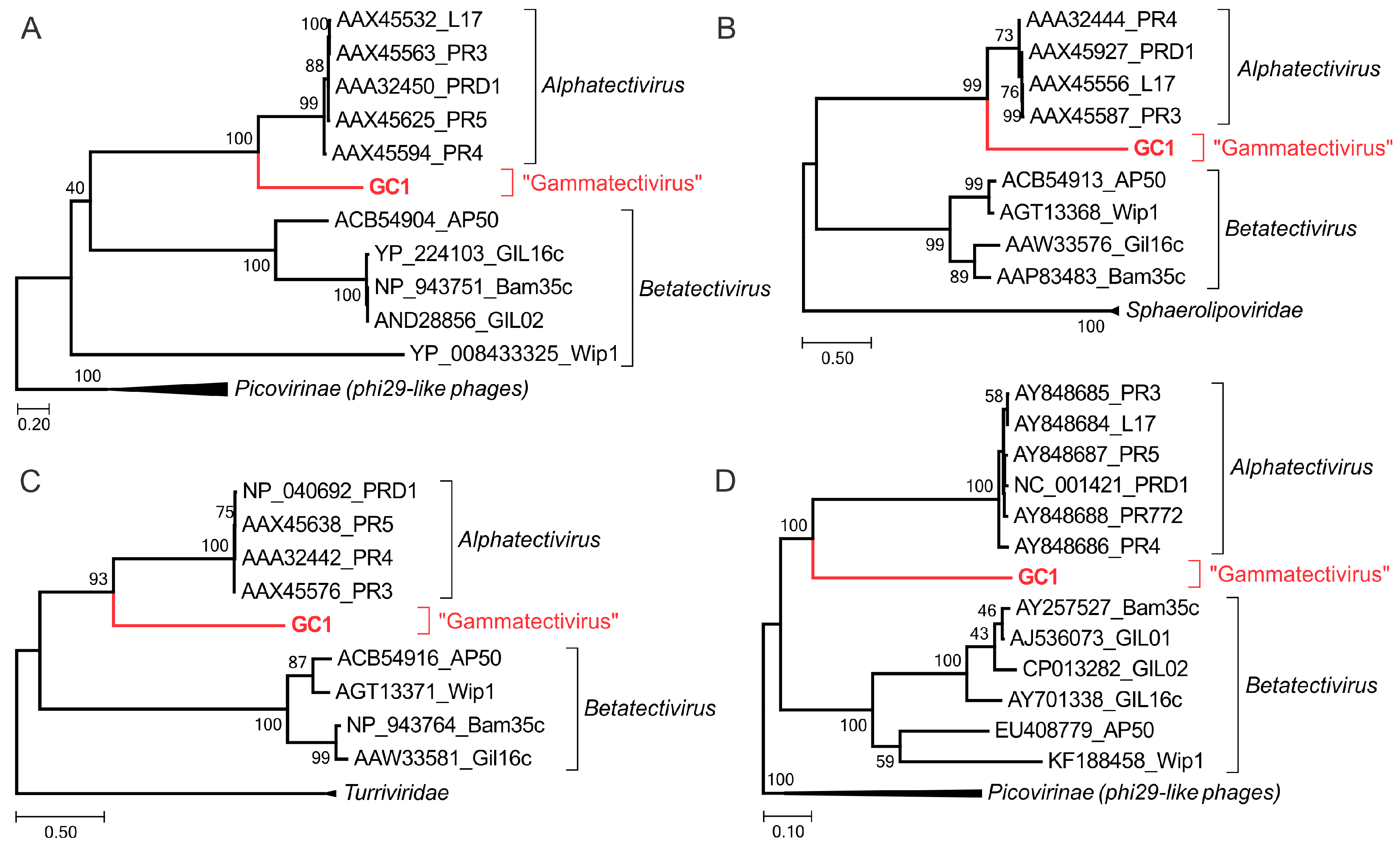
3.7. Phylogenetic Position of GC1 within the TECTIVIRIDAE
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Raspor, P.; Goranovic, D. Biotechnological applications of acetic acid bacteria. Crit. Rev. Biotechnol. 2008, 28, 101–124. [Google Scholar] [CrossRef] [PubMed]
- Jakob, F.; Pfaff, A.; Novoa-Carballal, R.; Rübsam, H.; Becker, T.; Vogel, R.F. Structural analysis of fructans produced by acetic acid bacteria reveals a relation to hydrocolloid function. Carbohydr. Polym. 2013, 92, 1234–1242. [Google Scholar] [CrossRef] [PubMed]
- Ua-Arak, T.; Jakob, F.; Vogel, R.F. Influence of levan-producing acetic acid bacteria on buckwheat-sourdough breads. Food Microbiol. 2017, 65, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Mas, A.; Torija, M.J.; García-Parrilla Mdel, C.; Troncoso, A.M. Acetic acid bacteria and the production and quality of wine vinegar. Sci. World J. 2014, 2014, 394671. [Google Scholar] [CrossRef] [PubMed]
- De Vuyst, L.; Weckxx, S. The cocoa bean fermentation process: From ecosystem analysis to starter culture development. J. Appl. Microbiol. 2016, 121, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Pothakos, V.; Illeghems, K.; Laureys, D.; Spitaels, F.; Vandamme, P.; de Vuyst, L. Acetic Acid Bacteria in Fermented Food and Beverage Ecosystems. In Acetic Acid Bacteria: Ecology and Physiology; Matsushita, K., Toyama, H., Tonouchi, N., Okamoto-Kainuma, A., Eds.; Springer: New York, NY, USA, 2016; pp. 73–99. ISBN 978-4-4-431-55931-3. [Google Scholar]
- Bartowsky, E.J.; Henschke, P.A. Acetic acid bacteria spoilage of bottled red wine—A review. Int. J. Food Microbiol. 2008, 125, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Joyeux, A.; Lafon-Lafourcade, S.; Ribéreau-Gayon, P. Evolution of acetic Acid bacteria during fermentation and storage of wine. Appl. Environ. Microbiol. 1984, 48, 153–156. [Google Scholar] [PubMed]
- Navarro, D.; Mateo, E.; Torija, M.J.; Mas, A. Acetic acid bacteria in grape must. Acetic Acid Bacteria 2013, 2, e419–e423. [Google Scholar] [CrossRef]
- Barata, A.; Malfeito-Ferreira, M.; Loureiro, V. Changes in sour rotten grape berry microbiota during ripening and wine fermentation. Int. J. Food Microbiol. 2012, 154, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Mateo, E.; Torija, M.J.; Mas, A.; Bartowsky, E.J. Acetic acid bacteria isolated from grapes of South Australian vineyards. Int. J. Food Microbiol. 2014, 178, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Bokulich, N.A.; Joseph, C.M.; Allen, G.; Benson, A.K.; Mills, D.A. Next-Generation Sequencing Reveals Significant Bacterial Diversity of Botrytized Wine. PLoS ONE 2012, 7, e36357. [Google Scholar] [CrossRef] [PubMed]
- Pinto, C.; Pinho, D.; Cardoso, R.; Custódio, V.; Fernandes, J.; Sousa, S.; Pinheiro, M.; Egas, C.; Gomes, A.C. Wine fermentation microbiome: A landscape from different Portuguese wine appellations. Front. Microbiol. 2015, 6, 905. [Google Scholar] [CrossRef] [PubMed]
- Portillo, M.; Mas, A. Analysis of microbial diversity and dynamics during wine fermentation of Grenache grape variety by high-throughput barcoding sequencing. LWT Food Sci. Technol. 2016, 72, 317–321. [Google Scholar] [CrossRef]
- Kiesel, B.; Wünsche, L.J. Phage Acm1-mediated transduction in the facultatively methanol-utilizing Acetobacter methanolicus MB 58/4. J. Gen. Virol. 1993, 74, 1741–1745. [Google Scholar] [CrossRef] [PubMed]
- Schocher, A.J.; Kuhn, H.; Schindler, B.; Palleroni, N.J.; Despreaux, C.W.; Boublik, M.; Miller, P.A. Acetobacter bacteriophage A-1. Arch. Microbiol. 1979, 121, 193–197. [Google Scholar] [CrossRef]
- Stamm, W.W.; Kittelmann, M.; Follmann, H.; Trüper, H.G. The occurrence of bacteriophages in spirit vinegar fermentation. Appl. Microbiol. Biotechnol. 1989, 30, 41–46. [Google Scholar]
- Bamford, D.H.; Caldentey, J.; Bamford, J.K. Bacteriophage PRD1: A broad host range DSDNA tectivirus with an internal membrane. Adv. Virus Res. 1995, 45, 281–319. [Google Scholar] [PubMed]
- Abrescia, N.G.; Cockburn, J.J.; Grimes, J.M.; Sutton, G.C.; Diprose, J.M.; Butcher, S.J.; Fuller, S.D.; San Martín, C.; Burnett, R.M.; Stuart, D.I.; et al. Insights into assembly from structural analysis of bacteriophage PRD1. Nature 2004, 432, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Butcher, S.J.; Manole, V.; Karhu, N.J. Lipid-containing viruses: Bacteriophage PRD1 assembly. Adv. Exp. Med. Biol. 2012, 726, 365–377. [Google Scholar] [PubMed]
- Redrejo-Rodríguez, M.; Salas, M. Multiple roles of genome-attached bacteriophage terminal proteins. Virology 2014, 468, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Berjón-Otero, M.; Villar, L.; de Vega, M.; Salas, M.; Redrejo-Rodríguez, M. DNA polymerase from temperate phage Bam35 is endowed with processive polymerization and abasic sites translesion synthesis capacity. Proc. Natl. Acad. Sci. USA 2015, 112, E3476–E3484. [Google Scholar] [CrossRef] [PubMed]
- Gillis, A.; Bin Jang, H.; Mahillon, J.; Kuhn, J.H.; Kropinski, A.M.; Adriaenssens, E.; Lavigne, R. ICTV Taxonomic Proposal 2017. 013B.U.v1. In the Family Tectiviridae, Change the Name of Genus Tectivirus to Alphatectivirus and Create a New Genus, Betatectivirus. International Committee on Taxonomy in Viruses (ICTV). 2017. Available online: https://talk.ictvonline.org/files/proposals/taxonomy_proposals_prokaryote1/m/bact02/69222017 (assessed on 20 October 2017).
- Saren, A.M.; Ravantti, J.J.; Benson, S.D.; Burnett, R.M.; Paulin, L.; Bamford, D.H.; Bamford, J.K. A snapshot of viral evolution from genome analysis of the tectiviridae family. J. Mol. Biol. 2005, 350, 427–440. [Google Scholar] [CrossRef] [PubMed]
- Gillis, A.; Mahillon, J. Prevalence, genetic diversity, and host range of tectiviruses among members of the Bacillus cereus group. Appl. Environ. Microbiol. 2014, 80, 4138–4152. [Google Scholar] [CrossRef] [PubMed]
- Jalasvuori, M.; Palmu, S.; Gillis, A.; Kokko, H.; Mahillon, J.; Bamford, J.K.; Fornelos, N. Identification of five novel tectiviruses in Bacillus strains: Analysis of a highly variable region generating genetic diversity. Res. Microbiol. 2013, 164, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Ravantti, J.J.; Gaidelyte, A.; Bamford, D.H.; Bamford, J.K. Comparative analysis of bacterial viruses Bam35, infecting a gram-positive host, and PRD1, infecting gram-negative hosts, demonstrates a viral lineage. Virology 2003, 313, 401–414. [Google Scholar] [CrossRef]
- Berjón-Otero, M.; Lechuga, A.; Mehla, J.; Uetz, P.; Salas, M.; Redrejo-Rodríguez, M. Bam35 tectivirus intraviral interaction map unveils new function and localization of phage ORFan proteins. J. Virol. 2017, 26. [Google Scholar] [CrossRef] [PubMed]
- Krupovic, M.; Cvirkaite-Krupovic, V.; Bamford, D.H. Identification and functional analysis of the Rz/Rz1-like accessory lysis genes in the membrane-containing bacteriophage PRD1. Mol. Microbiol. 2008, 68, 492–503. [Google Scholar] [CrossRef] [PubMed]
- Gaidelyte, A.; Jaatinen, S.T.; Daugelavicius, R.; Bamford, J.K.; Bamford, D.H. The linear double-stranded DNA of phage Bam35 enters lysogenic host cells, but the late phage functions are suppressed. J. Bacteriol. 2005, 187, 3521–3527. [Google Scholar] [CrossRef] [PubMed]
- Gaidelyte, A.; Cvirkaite-Krupovic, V.; Daugelavicius, R.; Bamford, J.K.; Bamford, D.H. The entry mechanism of membrane-containing phage Bam35 infecting Bacillus thuringiensis. J. Bacteriol. 2006, 188, 5925–5934. [Google Scholar] [CrossRef] [PubMed]
- Fornelos, N.; Butala, M.; Hodnik, V.; Anderluh, G.; Bamford, J.K.; Salas, M. Bacteriophage GIL01 gp7 interacts with host LexA repressor to enhance DNA binding and inhibit RecA-mediated auto-cleavage. Nucleic Acids Res. 2015, 43, 7315–7329. [Google Scholar] [CrossRef] [PubMed]
- Laurinmäki, P.A.; Huiskonen, J.T.; Bamford, D.H.; Butcher, S.J. Membrane proteins modulate the bilayer curvature in the bacterial virus Bam35. Structure 2005, 13, 1819–1828. [Google Scholar] [CrossRef] [PubMed]
- Krupovic, M.; Bamford, D.H. Virus evolution: How far does the double beta-barrel viral lineage extend? Nat. Rev. Microbiol. 2008, 6, 941–948. [Google Scholar] [CrossRef] [PubMed]
- Krupovic, M.; Koonin, E.V. Polintons: A hotbed of eukaryotic virus, transposon and plasmid evolution. Nat. Rev. Microbiol. 2015, 13, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Koonin, E.V.; Krupovic, M. Polintons, virophages and transpovirons: A tangled web linking viruses, transposons and immunity. Curr. Opin. Virol. 2017, 25, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Qian, P.Y. Conservative fragments in bacterial 16S rRNA genes and primer design for 16S ribosomal DNA amplicons in metagenomic studies. PLoS ONE 2009, 4, e7401. [Google Scholar] [CrossRef] [PubMed]
- Philippe, C.; Jaomanjaka, F.; Claisse, O.; Laforgue, R.; Maupeu, J.; Petrel, M.; Le Marrec, C. A survey of oenophages during wine making reveals a novel group with unusual genomic characteristics. Int. J. Food Microbiol. 2017, 257, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Jaomanjaka, F.; Claisse, O.; Blanche-Barbat, M.; Petrel, M.; Ballestra, P.; Le Marrec, C. Characterization of a new virulent phage infecting the lactic acid bacterium Oenococcus oeni. Food Microbiol. 2016, 54, 167–177. [Google Scholar] [CrossRef]
- Sambrook, J.; Russell, D.W. Molecular Cloning: A Laboratory Manual; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2001. [Google Scholar]
- Zinno, P.; Janzen, T.; Bennedsen, M.; Ercolini, D.; Mauriello, G. Characterization of Streptococcus thermophilus lytic bacteriophages from mozzarella cheese plants. Int. J. Food Microbiol. 2010, 138, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Bankevich, A.; Nurk, S.; Antipov, D.; Gurevich, A.A.; Dvorkin, M.; Kulikov, A.S.; Lesin, V.M.; Nikolenko, S.I.; Pham, S.; Prjibelski, A.D.; et al. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 2012, 19, 455–477. [Google Scholar] [CrossRef] [PubMed]
- Seemann, T. Prokka: Rapid Prokaryotic Genome Annotation. Bioinformatics 2014, 30, 2068–2069. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, M.J.; Petty, N.K.; Beatson, S.A. Easyfig: A genome comparison visualizer. Bioinformatics 2011, 27, 1009–1010. [Google Scholar] [CrossRef] [PubMed]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef] [PubMed]
- Söding, J.; Biegert, A.; Lupas, A.N. The HHpred interactive server for protein homology detection and structure prediction. Nucleic Acids Res. 2005, 33, W244–W248. [Google Scholar] [CrossRef] [PubMed]
- Marchler-Bauer, A.; Bryant, S.H. CD-Search: Protein domain annotations on the fly. Nucleic Acids Res. 2004, 32, W327–W331. [Google Scholar] [CrossRef] [PubMed]
- Pei, J.; Grishin, N.V. PROMALS3D: Multiple protein sequence alignment enhanced with evolutionary and three-dimensional structural information. Methods Mol. Biol. 2014, 1079, 263–271. [Google Scholar] [PubMed]
- Capella-Gutiérrez, S.; Silla-Martínez, J.M.; Gabaldón, T. trimAl: A tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics 2009, 25, 1972–1973. [Google Scholar] [CrossRef] [PubMed]
- Guindon, S.; Dufayard, J.F.; Lefort, V.; Anisimova, M.; Hordijk, W.; Gascuel, O. New algorithms and methods to estimate maximum-likelihood phylogenies: Assessing the performance of PhyML 3.0. Syst. Biol. 2010, 59, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Meier-Kolthoff, J.P.; Göker, M. VICTOR: Genome-based Phylogeny and Classification of Prokaryotic Viruses. Bioinformatics 2017, 33, 3396–3404. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, H.W.; Prangishvili, D. Prokaryote viruses studied by electron microscopy. Arch. Virol. 2012, 157, 1843–1849. [Google Scholar] [CrossRef] [PubMed]
- Oksanen, H.M. ICTV Report Consortium. ICTV Virus Taxonomy Profile: Corticoviridae. J. Gen. Virol. 2017, 98, 888–889. [Google Scholar] [PubMed]
- Pawlowski, A.; Rissanen, I.; Bamford, J.K.; Krupovic, M.; Jalasvuori, M. Gammasphaerolipovirus, a newly proposed bacteriophage genus, unifies viruses of halophilic archaea and thermophilic bacteria within the novel family Sphaerolipoviridae. Arch. Virol. 2014, 159, 1541–1554. [Google Scholar] [CrossRef] [PubMed]
- Laurinavicius, S.; Käkelä, R.; Somerharju, P.; Bamford, D.H. Phospholipid molecular species profiles of tectiviruses infecting Gram-negative and Gram-positive hosts. Virology 2004, 322, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Grahn, A.M.; Daugelavicius, R.; Bamford, D.H. Sequential model of phage PRD1 DNA delivery: Active involvement of the viral membrane. Mol. Microbiol. 2002, 45, 1199–1209. [Google Scholar] [CrossRef]
- Peralta, B.; Gil-Carton, D.; Castaño-Díez, D.; Bertin, A.; Boulogne, C.; Oksanen, H.M.; Bamford, D.H.; Abrescia, N.G. Mechanism of membranous tunnelling nanotube formation in viral genome delivery. PLoS Biol. 2013, 11, e1001667. [Google Scholar] [CrossRef] [PubMed]
- Sainz, F.; Mas, A.; Torija, M.J. Draft genome sequences of Gluconobacter cerinus CECT 9110 and Gluconobacter japonicus CECT 8443, acetic acid bacteria Isolated from grape must. Genome Announc. 2016, 4. [Google Scholar] [CrossRef] [PubMed]
- Redrejo-Rodríguez, M.; Muñoz-Espín, D.; Holguera, I.; Mencía, M.; Salas, M. Functional eukaryotic nuclear localization signals are widespread in terminal proteins of bacteriophages. Proc. Natl. Acad. Sci. USA 2012, 109, 18482–18487. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.E.; Pourhossein, M.; Waterhouse, A.; Hudson, T.; Goldrick, M.; Derrick, J.P.; Roberts, I.S. The K5 lyase KflA combines a viral tail spike structure with a bacterial polysaccharide lyase mechanism. J. Biol. Chem. 2010, 285, 23963–23969. [Google Scholar] [CrossRef] [PubMed]
- Rydman, P.S.; Bamford, D.H. The lytic enzyme of bacteriophage PRD1 is associated with the viral membrane. J. Bacteriol. 2002, 184, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Krupovic, M.; Vilen, H.; Bamford, J.K.; Kivelä, H.M.; Aalto, J.M.; Savilahti, H.; Bamford, D.H. Genome characterization of lipid-containing marine bacteriophage PM2 by transposon insertion mutagenesis. J. Virol. 2006, 80, 9270–9278. [Google Scholar] [CrossRef] [PubMed]
- Gillis, A.; Mahillon, J. Influence of lysogeny of Tectiviruses GIL01 and GIL16 on Bacillus thuringiensis growth, biofilm formation, and swarming motility. Appl. Environ. Microbiol. 2014, 80, 7620–7630. [Google Scholar] [CrossRef] [PubMed]
- Roh, S.W.; Nam, Y.D.; Chang, H.W.; Kim, K.H.; Kim, M.S.; Ryu, J.H.; Kim, S.H.; Lee, W.J.; Bae, J.W. Phylogenetic characterization of two novel commensal bacteria involved with innate immune homeostasis in Drosophila melanogaster. Appl. Environ. Microbiol. 2008, 74, 6171–6177. [Google Scholar] [CrossRef] [PubMed]
- Buchon, N.; Broderick, N.A.; Lemaitre, B. Gut homeostasis in a microbial world: Insights from Drosophila melanogaster. Nat. Rev. Microbiol. 2013, 11, 615–626. [Google Scholar] [CrossRef] [PubMed]
- Fischer, C.N.; Trautman, E.P.; Crawford, J.M.; Stabb, E.V.; Handelsman, J.; Broderick, N.A. Metabolite exchange between microbiome members produces compounds that influence Drosophila behavior. eLife 2017, 9, e18855. [Google Scholar] [CrossRef] [PubMed]

| Strains | Name | Isolation | |
|---|---|---|---|
| Year | Source | ||
| Acetobacter aceti | CRBO08ba14 | 2008 | Grapes, Aquitaine |
| CIP103111 * | na | Alcohol vinegar | |
| ATCC15973 * | na | Wine vinegar | |
| Acetobacter pasteurianus | CRBO03ba01, CRBO03ba02 | 2003 | na |
| CRBO08ba01, CRBO08ba03 | 2008 | Grapes, Aquitaine | |
| Gluconobacter cerinus | CRBO11174, CRBO11176, CRBO11178, CRBO11179 | 2011 | Matured grapes, Grande Ferrade, INRA Bordeaux |
| CRBO96ba02 | 1996 | Grapes, Pessac-Leognan | |
| CRBO96ba39 | 1997 | Matured grapes, Bordeaux (La Tour Blanche) | |
| Gluconobacter oxydans | CRBO11187 | 2011 | Matured grapes, Bordeaux |
| CRBO11203, CRBO11204, CRBO11205 | 2011 | Matured grapes, Léoville | |
| Number | Samples Characteristics a | Presence of Phages b | ||
|---|---|---|---|---|
| Wine | Appellation/Grape Variety | Step | ||
| 67, 68, 69, 70 | Sweet white | Sauternes/Sémillon | Must | − |
| 14 | PDC | − | ||
| 12 | Dry white | Bordeaux/Sémillon | PDC | − |
| 1 | Bordeaux/SauvBlanc | Must | − | |
| 2 | Juice for PDC | + | ||
| 26 | PDC | − | ||
| 27, 29 | Early AF | − | ||
| 31 | Lussac Saint Emilion/SauvBlanc | PDC | − | |
| 125 | Red | Saint-Emilion/CabFranc | − | |
| 18 | Saint Emilion/Merlot | Must | − | |
| 62 | − | |||
| 3 | Lussac Saint Emilion/Merlot | Must | − | |
| 30 | − | |||
| 22, 23, 24 | PDC | − | ||
| 60, 63 | Margaux/Merlot | Must | − | |
| 61 | Entre-Deux-Mers/Merlot | Must | − | |
| 143 | Pomerol/CabSauv | MLF | − | |
| 144, 147 | Pomerol/Merlot | − | ||
| 148 | Gers-Gamay/Merlot | − | ||
| 149 | Gers/Merlot | − | ||
| 151, 153 | Medoc/CabSauv | − | ||
| Name | Sequence (5′-3′) | Position on GC1-Genome |
|---|---|---|
| ITR-F | CTCTTCCACGGCAACAATCC | 271–252 16,253–16,272 |
| ITR-R | ACAAGTACTACAGGGAGGGG | 52–71 16,472–16,453 |
| gp36-F | TAAGCGCGGATGGTTTAAGC | 15,957–15,976 |
| gp1-F | TACAATCGTGACGGCGGATA | 886–867 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Philippe, C.; Krupovic, M.; Jaomanjaka, F.; Claisse, O.; Petrel, M.; Le Marrec, C. Bacteriophage GC1, a Novel Tectivirus Infecting Gluconobacter Cerinus, an Acetic Acid Bacterium Associated with Wine-Making. Viruses 2018, 10, 39. https://doi.org/10.3390/v10010039
Philippe C, Krupovic M, Jaomanjaka F, Claisse O, Petrel M, Le Marrec C. Bacteriophage GC1, a Novel Tectivirus Infecting Gluconobacter Cerinus, an Acetic Acid Bacterium Associated with Wine-Making. Viruses. 2018; 10(1):39. https://doi.org/10.3390/v10010039
Chicago/Turabian StylePhilippe, Cécile, Mart Krupovic, Fety Jaomanjaka, Olivier Claisse, Melina Petrel, and Claire Le Marrec. 2018. "Bacteriophage GC1, a Novel Tectivirus Infecting Gluconobacter Cerinus, an Acetic Acid Bacterium Associated with Wine-Making" Viruses 10, no. 1: 39. https://doi.org/10.3390/v10010039
APA StylePhilippe, C., Krupovic, M., Jaomanjaka, F., Claisse, O., Petrel, M., & Le Marrec, C. (2018). Bacteriophage GC1, a Novel Tectivirus Infecting Gluconobacter Cerinus, an Acetic Acid Bacterium Associated with Wine-Making. Viruses, 10(1), 39. https://doi.org/10.3390/v10010039





