Climate-Induced Northerly Expansion of Siberian Silkmoth Range
Abstract
:1. Introduction
2. Materials and Methods
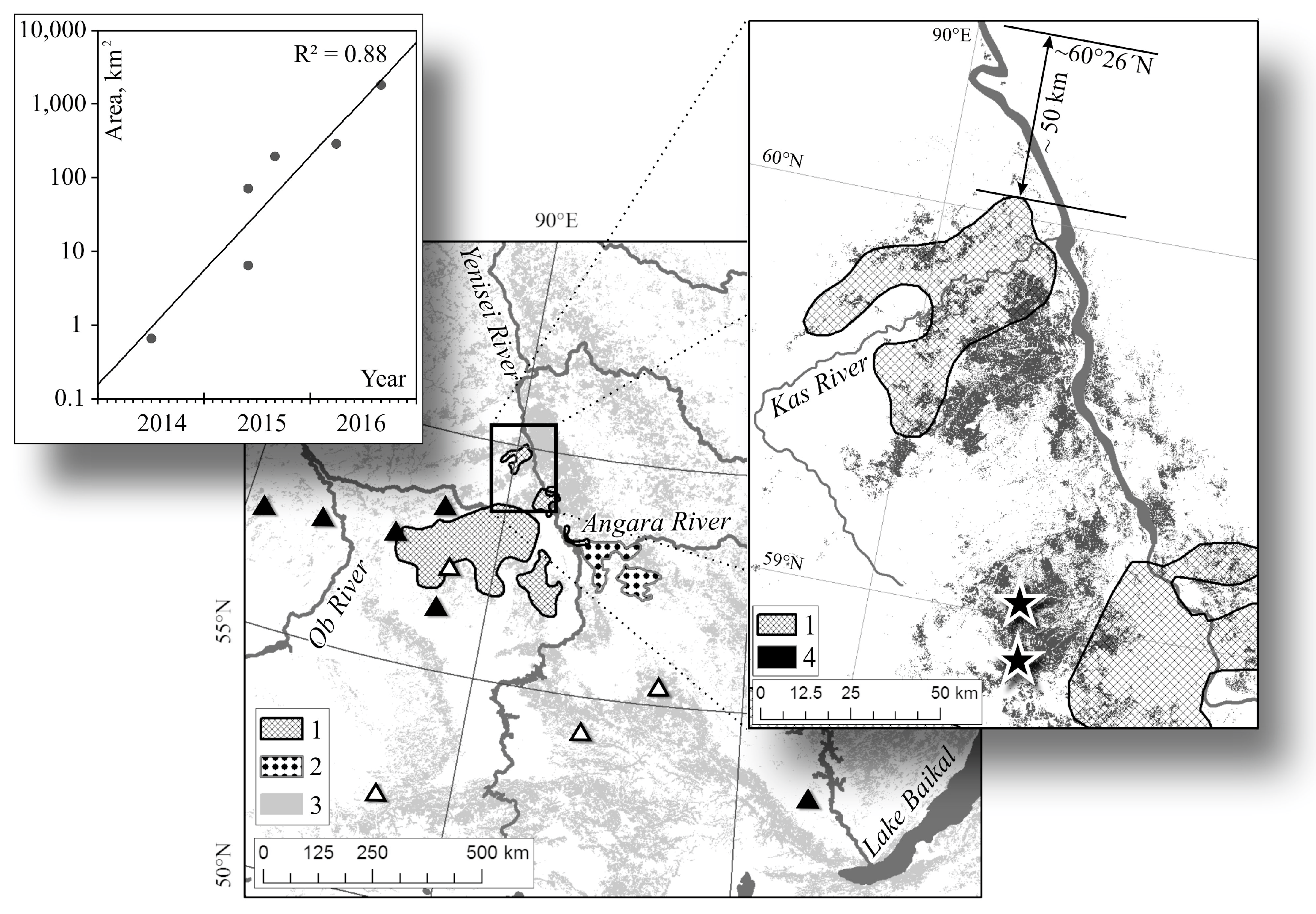
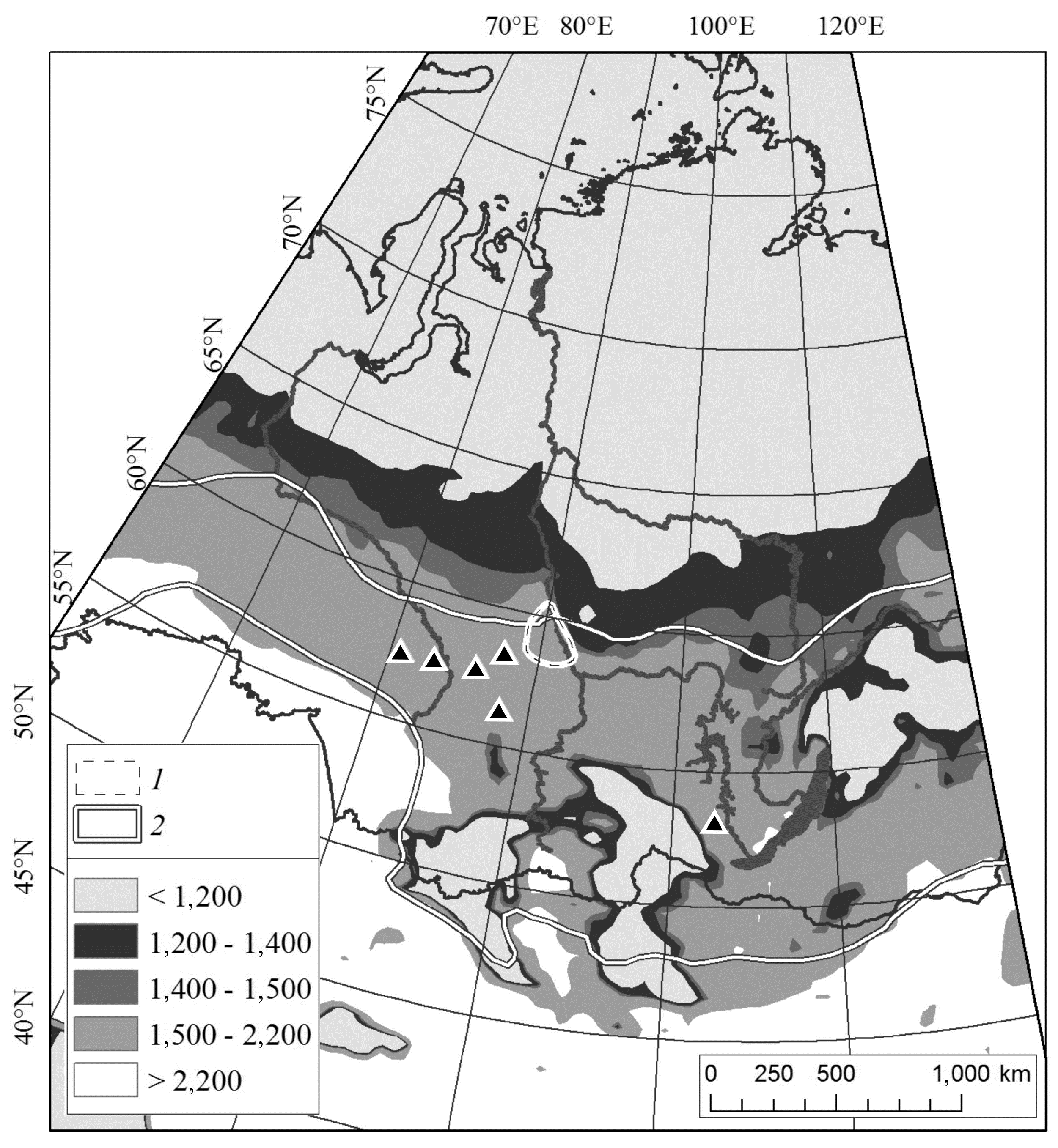
2.1. Study Area
2.2. Data
2.3. Image Analysis
3. Results and Discussion
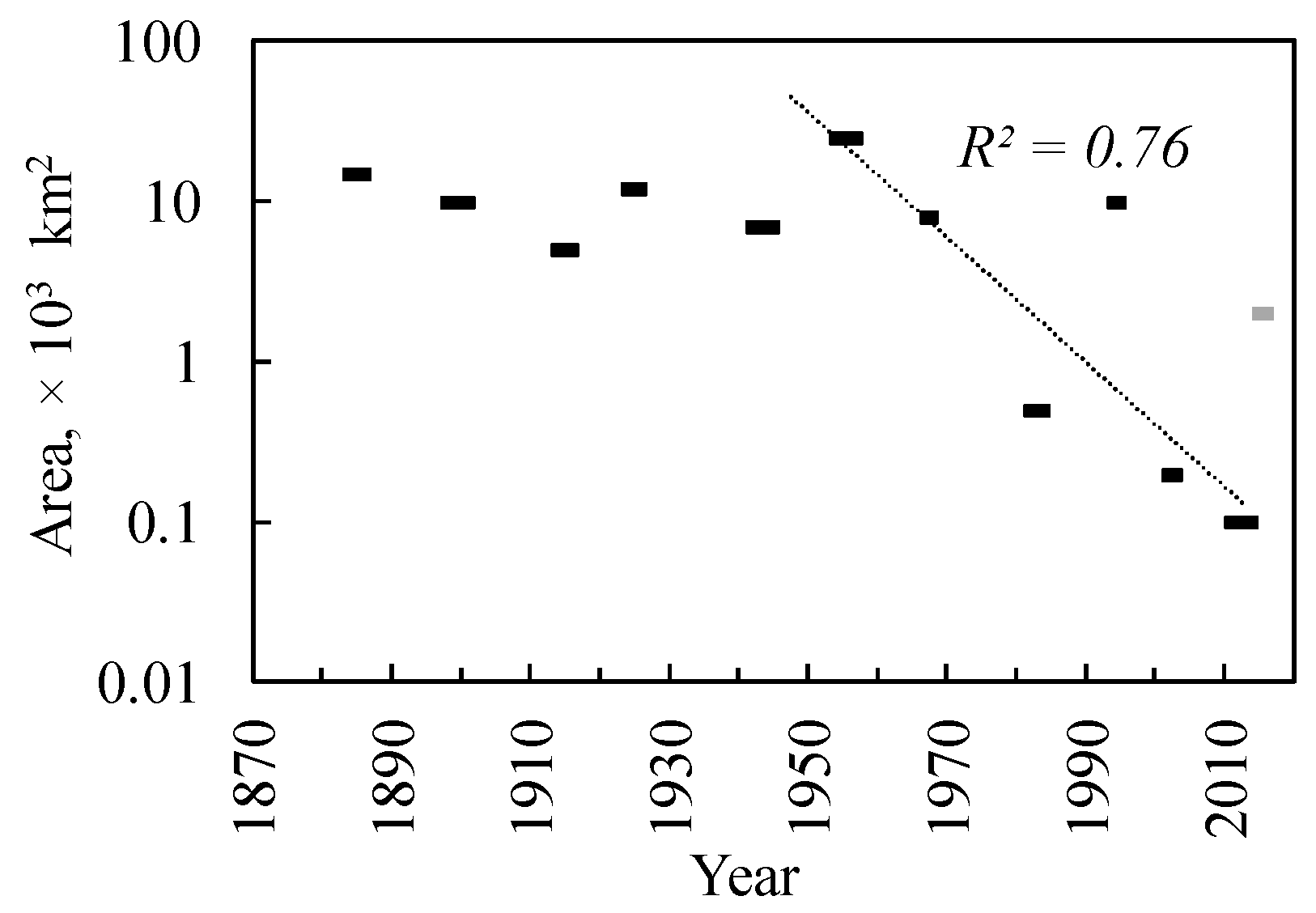
3.1. Siberian Silkmoth Outbreak Chronology in Central Siberia
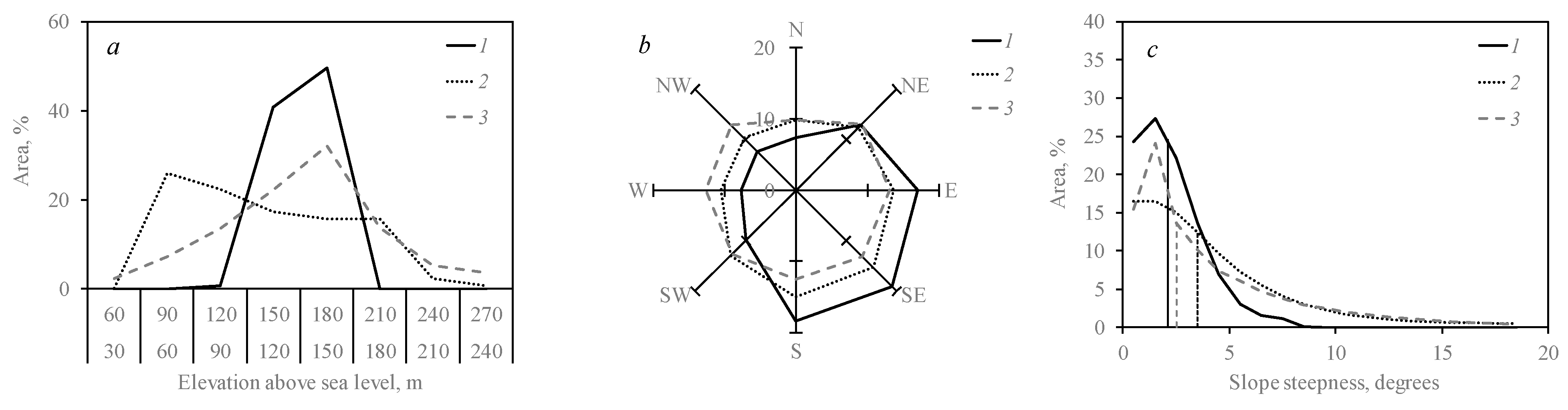
3.2. Outbreak Relevance to Relief Features
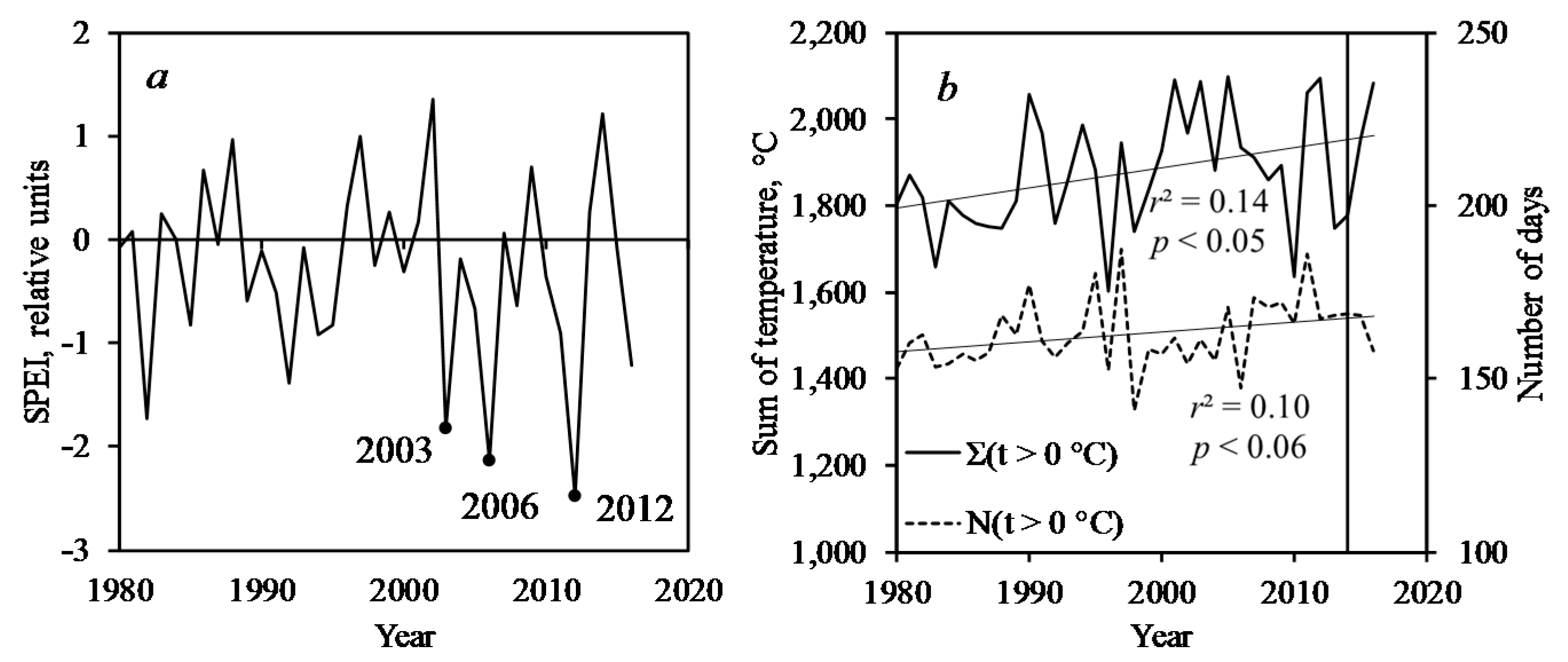
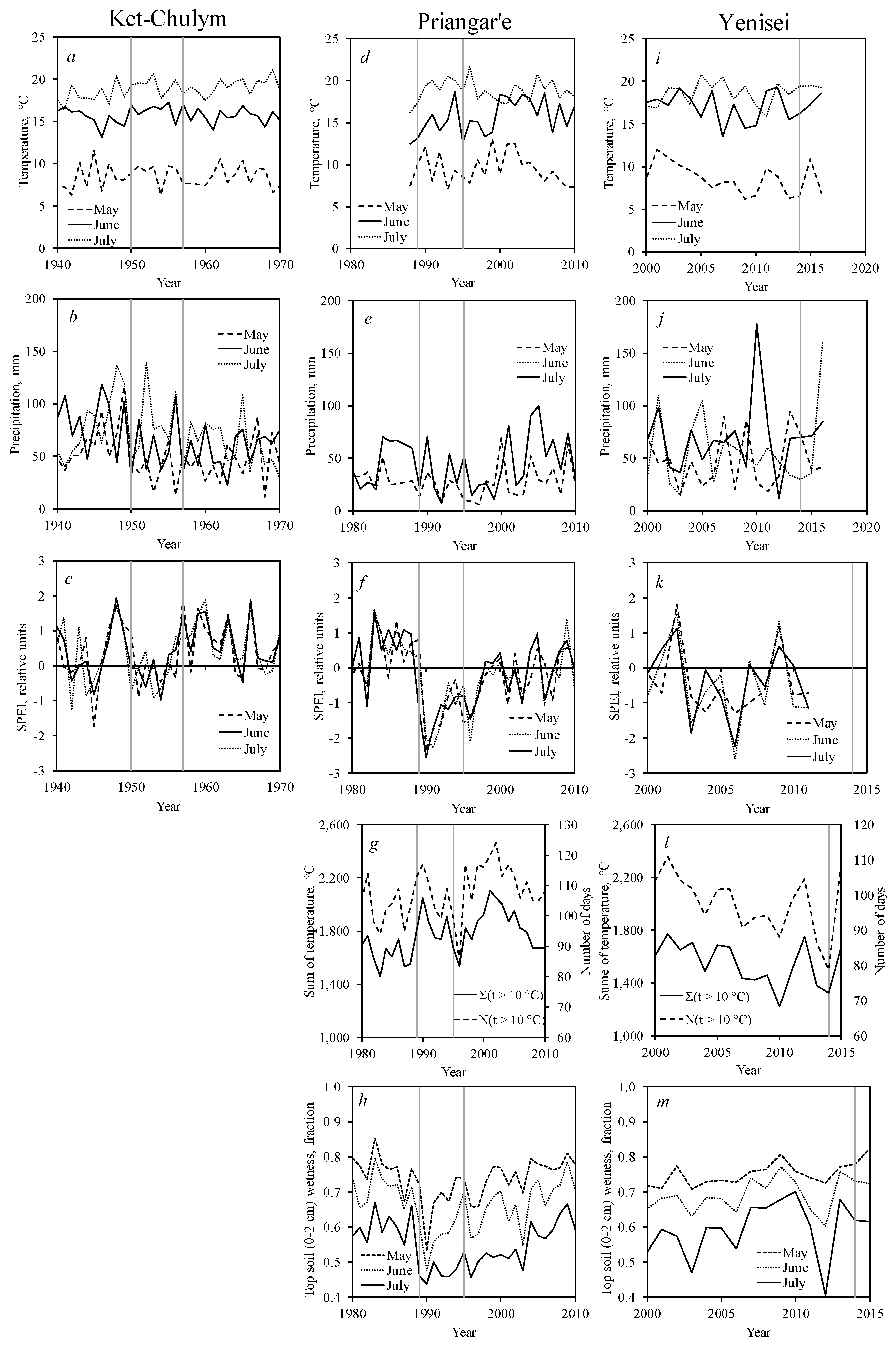
3.3. Pest Outbreak and Climate Variables
3.4. Northern Boundary of Siberian Silkmoth Range
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kolomiecz, N.G.; Mayer, E.I. The Most Important Forest Pests in Tomskaya Oblast and Pest Control; Tomskoye: Tomsk, Russia, 1963; p. 36. (In Russian) [Google Scholar]
- Kharuk, V.I.; Ranson, K.J.; Kozuhovskaya, A.G.; Kondakov, Y.P.; Pestunov, I.A. NOAA/AVHRR satellite detection of Siberian silkmoth outbreaks in eastern Siberia. Int. J. Remote Sens. 2004, 25, 5543–5556. [Google Scholar] [CrossRef]
- Kondakov, Y.P. Siberian silkmoth outbreaks in Krasnoyarskii krai. In Entomology Researches in Siberia; KF REO: Krasnoyarsk, Russia, 2002; Volume 2, pp. 25–74. (In Russian) [Google Scholar]
- Rojkov, A.S. Siberian Silkmoth Outbreak and Pest Control; Nauka: Moskow, Russia, 1965; p. 180. (In Russian) [Google Scholar]
- Kondakov, Y.P. Patterns of Siberian silkmoth outbreaks. In Ecology of Forest Animal Population in Siberia; Nauka: Novosibirsk, Russia, 1974; pp. 206–264. (In Russian) [Google Scholar]
- De la Giroday, H.C.; Carroll, A.L.; Aukema, B.H. Breach of the northern Rocky Mountain geoclimatic barrier: Initiation of range expansion by the mountain pine beetle. J. Biogeogr. 2012, 39, 1112–1123. [Google Scholar] [CrossRef]
- Seiter, S.; Kingsolver, J. Environmental determinants of population divergence in life-history traits for an invasive species: Climate, seasonality and natural enemies. J. Evol. Biol. 2013, 26, 1634–1645. [Google Scholar] [CrossRef] [PubMed]
- Kolb, T.E.; Fettig, C.J.; Ayres, M.P.; Bentz, B.J.; Hicke, J.A.; Mathiasen, R.; Stewart, J.E.; Weed, A.S. Observed and anticipated impacts of drought on forests insects and diseases in the United States. For. Ecol. Manag. 2016, 380, 321–334. [Google Scholar] [CrossRef]
- Pureswaran, D.S.; De Grandpré, L.; Paré, D.; Taylor, A.; Barrette, M.; Morin, H.; Régnière, J.; Kneeshaw, D.D. Climate-induced changes in host tree–insect phenology may drive ecological state-shift in boreal forests. Ecology 2015, 96, 1480–1491. [Google Scholar] [CrossRef]
- Coleman, T.W.; Jones, M.I.; Courtial, B.; Graves, A.D.; Woods, M.; Roques, A.; Seybold, S.J. Impact of the first recorded outbreak of the Douglas-fir tussock moth, Orgyia pseudotsugata, in southern California and the extent of its distribution in the Pacific Southwest region. For. Ecol. Manag. 2014, 329, 295–305. [Google Scholar] [CrossRef]
- Régnière, J.; Nealis, V.; Porter, K. Climate suitability and management of the gypsy moth invasion into Canada. Biol. Invasions 2009, 11, 135–148. [Google Scholar] [CrossRef]
- Millar, C.I.; Stephenson, N.L. Temperate forest health in an era of emerging megadisturbance. Science 2015, 349, 823–826. [Google Scholar] [CrossRef] [PubMed]
- Haynes, K.J.; Allstadt, A.; Klimetzek, D. Forest defoliator outbreaks under climate change: Effects on the frequency and severity of outbreaks of five pine insect pests. Glob. Chang. Biol. 2014, 20, 2004–2018. [Google Scholar] [CrossRef] [PubMed]
- Kharuk, V.I.; Demidko, D.A.; Fedotova, E.V.; Dvinskaya, M.L.; Budnik, U.A. Spatial and temporal dynamics of Siberian silk moth large-scale outbreak in dark-needle coniferous tree stands in Altai. Contemp. Probl. Ecol. 2016, 9, 711–720. [Google Scholar] [CrossRef]
- Kharuk, V.I.; Im, S.T.; Yagunov, M.N. Migration of the northern limit of Siberian silkmoth outbreaks. Contemp. Probl. Ecol. submitted. (In Russian).
- IPCC. 2014: Climate Change 2014: Impacts, Adaptation, and Vulnerability. A Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Field, C.B., Barros, V.R., Dokken, D.J., Mach, K.J., Mastrandrea, M.D., Bilir, T.E., Chatterjee, M., Ebi, K.L., Estrada, Y.O., Genova, R.C., et al., Eds.; World Meteorological Organization: Geneva, Switzerland, 2014; p. 190. Available online: http://www.ipcc.ch/report/ar5/wg2/ (accessed on 19 May 2017).
- Bartalev, S.A.; Egorov, V.A.; Ershov, D.V.; Isaev, A.S.; Loupian, E.A.; Plotnikov, D.E.; Uvarov, I.A. Sputnikovoe kartografirovanie rastitel’nogo pokrova Rossii po dannym spektroradiometra MODIS (Maping of Russia’s vegetation cover using MODIS satellite data). Sovrem. Probl. Distantsionnogo Zondirovaniya Zemli Kosmosa 2011, 8, 285–302. (In Russian) [Google Scholar]
- Riano, D.; Chuvieco, E.; Salas, J.; Aguado, I. Assessment of Different Topographic Corrections in Landsat-TM Data for Mapping Vegetation Types. IEEE Trans. Geosci. Remote Sens. 2003, 41, 1056–1061. [Google Scholar] [CrossRef]
- Vicente-Serrano, S.M.; Beguería, S.; López-Moreno, J.I. A Multi-scalar drought index sensitive to global warming: The Standardized Precipitation Evapotranspiration Index—SPEI. J. Clim. 2010, 23, 1696–1711. [Google Scholar] [CrossRef]
- Kharuk, V.I.; Ranson, K.J.; Kuz’michev, V.V.; Im, S.T. Landsat-based analysis of insect outbreaks in southern Siberia. Can. J. Remote Sens. 2003, 29, 286–297. [Google Scholar] [CrossRef]
- Kharuk, V.I.; Ranson, K.J.; Fedotova, E.V. Spatial pattern of Siberian silkmoth outbreak and taiga mortality. Scand. J. For. Res. 2007, 22, 531–536. [Google Scholar] [CrossRef]
- Kharuk, V.I.; Ranson, K.J.; Im, S.T. Siberian silkmoth outbreak pattern analysis based on SPOT VEGETATION data. Int. J. Remote Sens. 2009, 30, 2377–2388. [Google Scholar] [CrossRef]
- Kharuk, V.I.; Im, S.T.; Petrov, I.A.; Yagunov, M.N. Decline of Dark Coniferous Stands in Baikal Region. Contemp. Probl. Ecol. 2016, 9, 617–625. [Google Scholar] [CrossRef]
- Kharuk, V.I.; Im, S.T.; Dvinskaya, M.L.; Ranson, K.J.; Petrov, I.A. Tree wave migration across an elevation gradient in the Altai Mountains, Siberia. J. Mt. Sci. 2017, 14, 442–452. [Google Scholar] [CrossRef]
- Kharuk, V.I.; Antamoskina, O.A. Silkmoth outbreak impact on the taiga wildfires. Contemp. Probl. Ecol. submitted. (In Russian).
| N | Dataset Name | Data Source | Spatial Resolution | Time Coverage | Variables Used |
|---|---|---|---|---|---|
| 1 | Taseevo meteo station | http://meteo.ru | Point | 1988–2016 | Temperature, precipitation (distance from outbreak ~50 km) |
| 2 | Yeniseisk meteo station | http://climexp.knmi.nl | Point | 1871–2016 | Temperature, precipitation (distance from outbreak ~90 km) |
| 3 | CRU TS 3.23 | https://crudata.uea.ac.uk/cru/data/hrg/cru_ts_3.23 | 0.5° × 0.5° | 1901–2014 | Temperature, precipitation |
| 4 | GHCN CAMS | https://www.esrl.noaa.gov/psd/data/gridded/data.ghcncams.html | 0.5° × 0.5° | 1948–2016 | Temperature |
| 5 | GPCC | https://www.esrl.noaa.gov/psd/data/gridded/data.gpcc.html | 0.5° × 0.5° | 2013–2016 | Precipitation |
| 6 | MERRA2 M2SDNXSLV.5.12.4 | ftp://goldsmr4.sci.gsfc.nasa.gov//data/s4pa/MERRA2_MONTHLY | 0.5° × 0.625° | 1980–2016 | Daily temperature Top soil (0–2 cm) moisture |
| 7 | SPEI | http://sac.csic.es/spei | 0.5° × 0.5° | 1950–2016 | Drought index |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kharuk, V.I.; Im, S.T.; Ranson, K.J.; Yagunov, M.N. Climate-Induced Northerly Expansion of Siberian Silkmoth Range. Forests 2017, 8, 301. https://doi.org/10.3390/f8080301
Kharuk VI, Im ST, Ranson KJ, Yagunov MN. Climate-Induced Northerly Expansion of Siberian Silkmoth Range. Forests. 2017; 8(8):301. https://doi.org/10.3390/f8080301
Chicago/Turabian StyleKharuk, Viacheslav I., Sergei T. Im, Kenneth J. Ranson, and Mikhail N. Yagunov. 2017. "Climate-Induced Northerly Expansion of Siberian Silkmoth Range" Forests 8, no. 8: 301. https://doi.org/10.3390/f8080301







