Stem Biomass Production of Paulownia elongata × P. fortunei under Low Irrigation in a Semi-Arid Environment
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Area
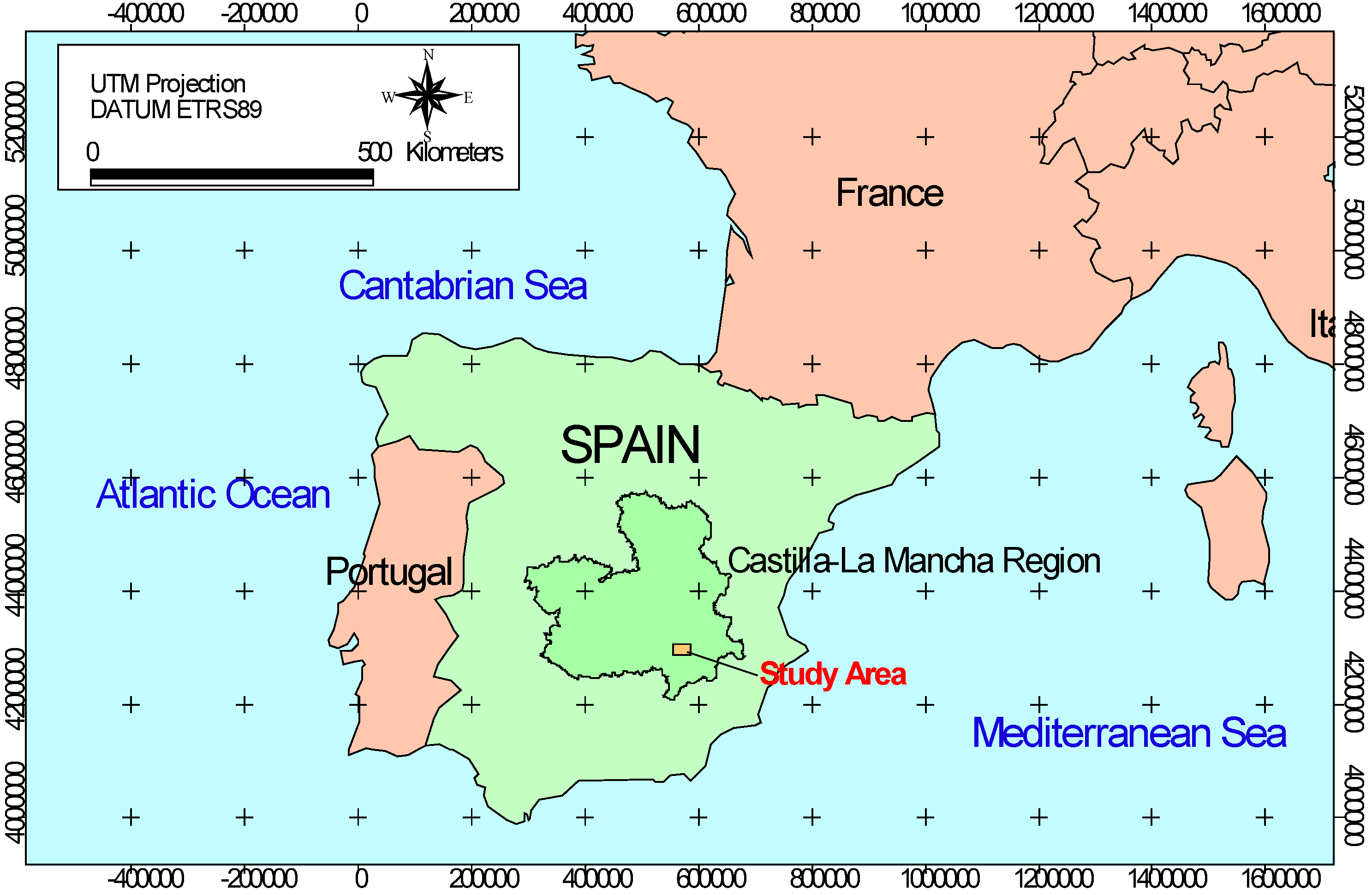
| Soil parameter | Upper horizon (A: 0–10 cm) | Lower horizon (B: 10–50 cm) | Methods |
|---|---|---|---|
| Sand (%) | 17 ± 2 | 19 ± 1 | Bouyoucos |
| Silt (%) | 48 ± 2 | 43 ± 1 | Bouyoucos |
| Clay (%) | 36 ± 2 | 38 ± 2 | Bouyoucos |
| pH (1:2.5) | 8.5 ± 0.1 | 8.5 ± 0.1 | 1;2.5 water |
| Electric conductivity (mmhos/cm) | 0.37 ± 0.05 | 0.33 ± 0.04 | 1:5 saturated paste |
| Chlorides (ppm) | 11 ± 1 | 8 ± 1 | Ion Chromatography |
| Sulfates (mg/100 g) | 39 ± 3 | 32 ± 2 | Ion Chromatography |
| Organic matter (%) | 5.68 ± 0.16 | 4.28 ± 0.21 | Walkley and Black |
| C/N | 13 ± 2 | 10 ± 3 | - |
| Total carbonates (%) | 55.7 ± 7.5 | 57.4 ± 5.3 | Calcimeter Bernard |
| Active limestone (%) | 16.0 ± 2.3 | 15.1 ± 3.4 | Ammonium Oxalate |
| Total nitrogen (%) | 0.26 ± 0.21 | 0.24 ± 0.18 | Kjeldahl |
| Nitric nitrogen (mg/g) | 0.004 ± 0.001 | 0.004 ± 0.001 | Calcium Sulfate extraction |
| Available phosphorus (mg/g) | 0.018 ± 0.0003 | 0.011 ± 0.0002 | Olsen |
| Available potassium (mg/g) | 0.27 ± 0.08 | 0.02 ± 0.06 | Atomic Absorption Spectrometry |
| Available calcium (mg/g) | 3.50 ± 0.45 | 3.16 ± 0.23 | Atomic Absorption Spectrometry |
| Available magnesium (mg/g) | 1.20 ± 0.06 | 1.22 ± 0.07 | Atomic Absorption Spectrometry |
| Available sodium (mg/g) | 0.35 ± 0.03 | 0.23 ± 0.02 | Atomic Absorption Spectrometry |
| CEC (meq/100 g) | 29.24 ± 2.32 | 22.68 ± 1.98 | Sodium Acetate |
2.2. Experimental Design

2.3. Stem Biomass Estimation
2.4. Effects of Irrigation and Fertilization on Stem Biomass Production
3. Results
3.1. Allometric Model for Stem Biomass Estimation
| Variables | Parameters | Estimate | Standard Error | T Statistic | P-Value |
|---|---|---|---|---|---|
| Intercept | β0 | −4.343 | 0.237 | −18.33 | <0.001 |
| ln(Db) | β1 | 2.694 | 0.140 | 19.22 | <0.001 |
| I1 | β2 | −0.733 | 0.383 | −1.91 | 0.059 |
| I1·ln(Db) | β3 | 0.396 | 0.216 | 1.84 | 0.069 |
| I2 | β4 | 1.125 | 0.557 | 0.52 | 0.600 |
| I2·ln(Db) | β5 | −0.519 | 0.295 | −1.76 | 0.081 |
| I3 | β6 | 0.859 | 0.657 | 1.31 | 0.194 |
| I3·ln(Db) | β7 | −0.402 | 0.337 | −1.19 | 0.236 |
| Variables | Parameters | Estimate | Standard Error | T Statistic | P-Value |
|---|---|---|---|---|---|
| Intercept | β0 | −4.462 | 0.153 | −29.1 | <0.001 |
| ln(Db) | β1 | 2.784 | 0.0824 | 33.8 | <0.001 |

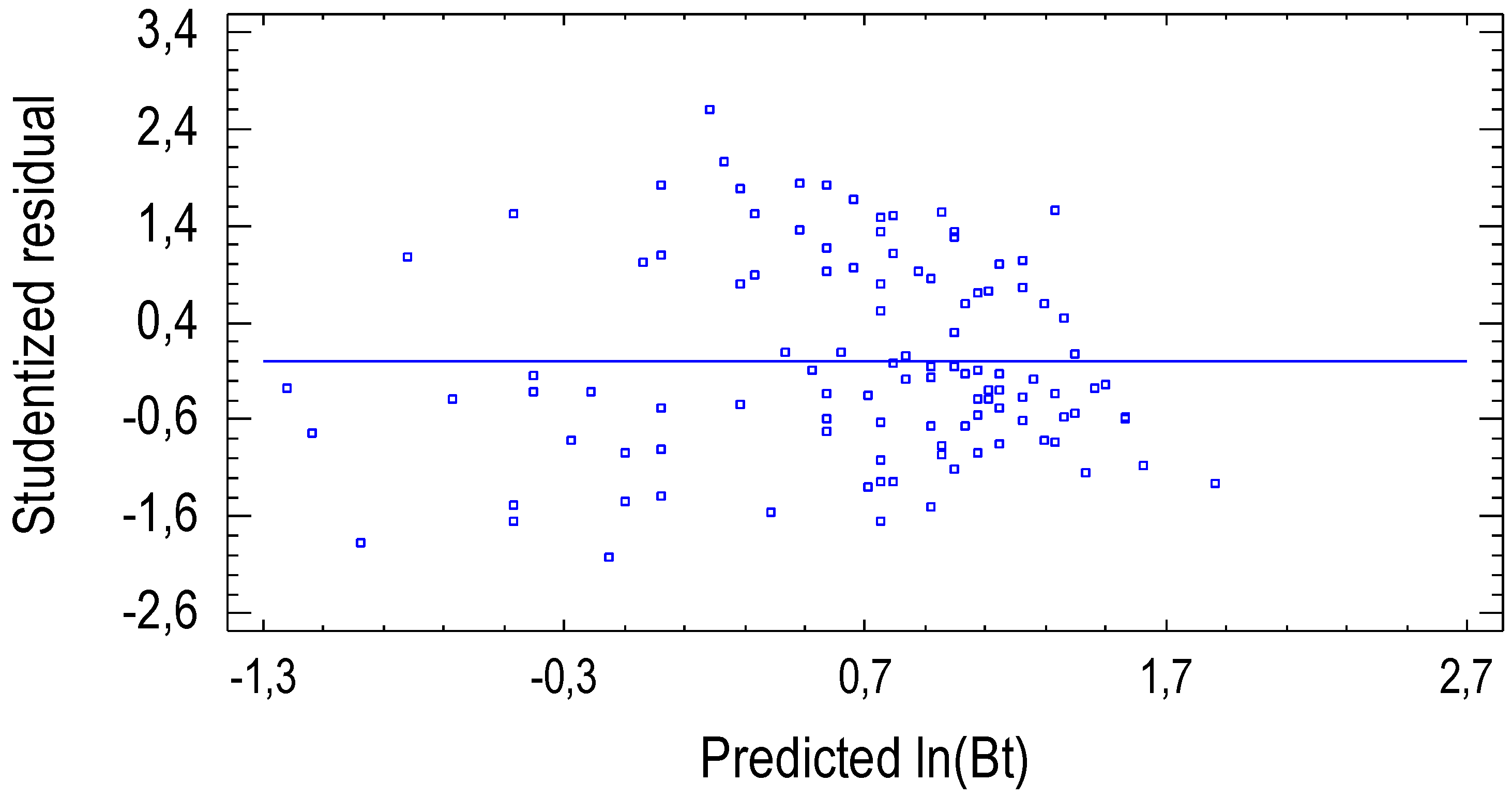
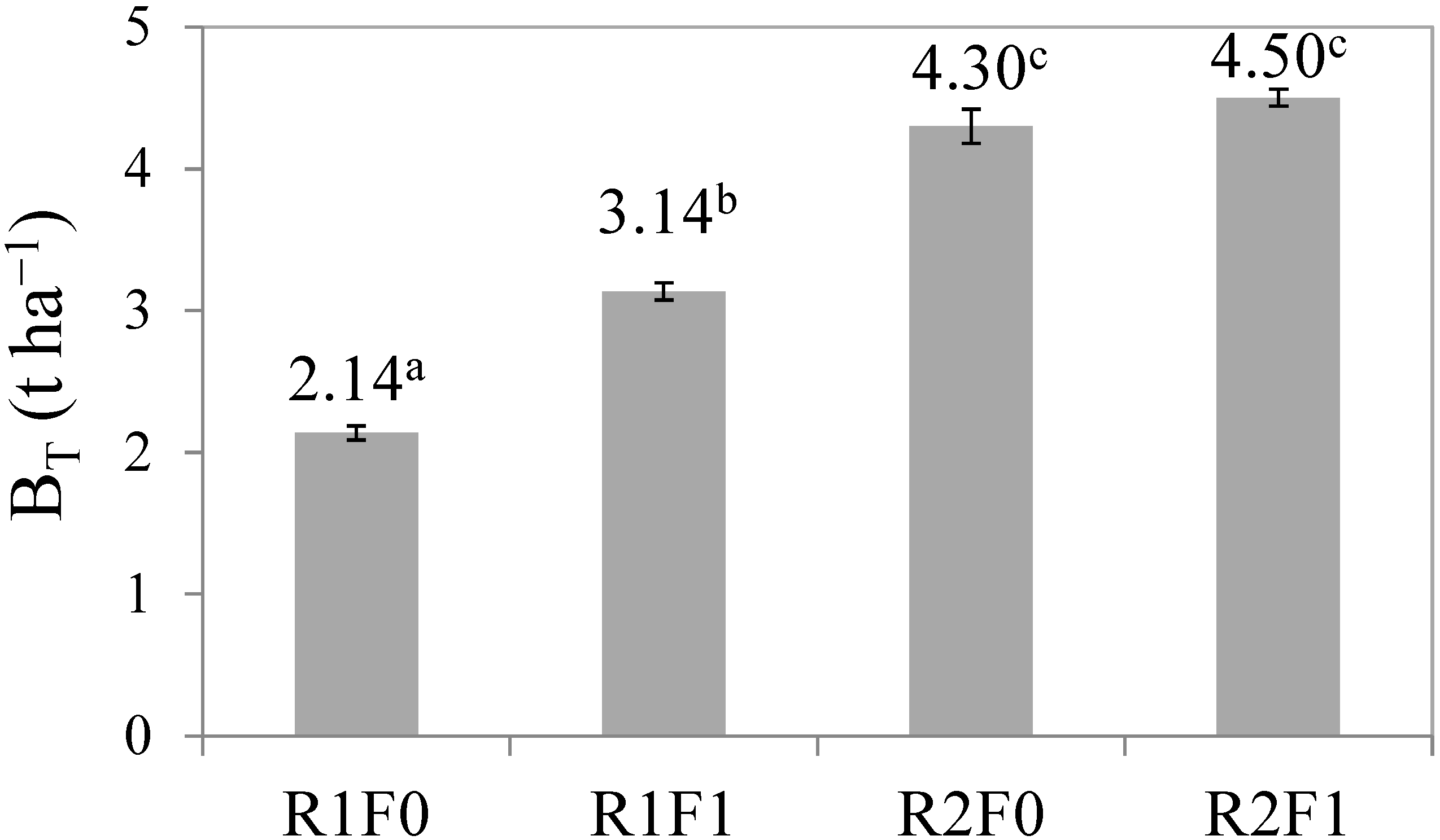
3.2. Effects of Irrigation and Fertilization on Stem Biomass Production
| Source | Sum of Squares | D.f. | Mean Square | F-Ratio | P-Value |
|---|---|---|---|---|---|
| MAIN EFFECTS | |||||
| Block | 0.093 | 3 | 0.031 | 1.34 | 0.321 |
| Irrigation | 12.43 | 1 | 12.43 | 534.88 | <0.001 |
| Fertilization | 1.452 | 1 | 1.452 | 62.50 | <0.001 |
| INTERACTIONS | |||||
| Irrigation x fertilization | 0.632 | 1 | 0.632 | 27.21 | <0.001 |
| RESIDUAL | 0.209 | 9 | 0.023 | ||
| TOTAL (corrected) | 14.81 | 15 |
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Pidwirny, M.; Jones, S. Climate Classification and Climatic Regions of the World. Fundamentals of Physical Geography; University of British Columbia: Columbia, Canada, 2006, 2nd ed. Available online: http://www.physicalgeography.net/fundamentals/7v.html (accessed on 12 May 2014).
- Sanz, D.; Castaño, S.; Cassiraga, E.; Sahuquillo, A.; Gómez-Alday, J.J.; Peña, S.; Calera, A. Modeling aquifer–river interactions under the influence of groundwater abstraction in the Mancha Oriental System (SE Spain). Hydrogeol. J. 2011, 19, 475–487. [Google Scholar] [CrossRef]
- Geeson, N.A.; Brandt, C.J.; Thornes, J.B. Mediterranean Desertification: A Mosaic of Processes and Responses; John Wiley & Sons Ltd: Chichester, UK, 2002; p. 444. [Google Scholar]
- FAO. Lecture Notes on the Major Soils of the World. World Soil Resources Reports 94; Food and Agriculture Organization of the United Nations: Rome, Italy, 2001; p. 337. [Google Scholar]
- Özsoy, G.; Aksoy, E. Characterization, Classification and Agricultural Usage of Vertisols Developed on Neogen Aged Calcareous Marl Parent Materials. J. Biol. Environ. Sci. 2007, 1, 5–10. [Google Scholar]
- Castellanos, A.; Esqueda, M.C. Biodiversity for bioenergy and biofuels in arid lands (BIO3). J. Arid Environ. 2013, 98, 133–134. [Google Scholar] [CrossRef]
- Madeira, M.V.; Fabiao, A.; Pereira, J.S.; Araújo, M.C.; Ribeiro, C. Changes in carbon stocks in Eucalyptus globulus Labill. plantations induced by different water and nutrient availability. For. Ecol. Manag. 2002, 171, 75–85. [Google Scholar] [CrossRef]
- Richardson, J.; Björheden, R.; Hakkila, P.; Lowe, A.T.; Smith, C.T. Bioenergy from Sustainable Forestry. Guiding Principles and Practice; Kluwer Academic Publishers: New York, NY, USA, 2002; p. 335. [Google Scholar]
- Ayan, S.; Sivacioglu, A.; Bilir, N. Growth variation of Paulownia Sieb. and Zucc. species and origins at the nursery stage in Kastamonu-Turkey. J. Environ. Biol. 2006, 27, 499–504. [Google Scholar]
- Joshee, N. Paulownia. In Handbook of Bioenergy Crop Plants; Kole, C., Joshi, C.P., Shonnard, D.R., Eds.; CRC Press: New York, NY, USA, 2012; pp. 671–686. [Google Scholar]
- Muthuri, C.W.; Ong, C.K.; Black, C.R.; Ngumi, V.W.; Mati, B.M. Tree and crop productivity in Grevillea, Alnus and Paulownia-based agroforestry systems in semi-arid Kenya. For. Ecol. Manag. 2005, 212, 23–39. [Google Scholar] [CrossRef]
- Yadav, N.K.; Vaidya, B.N.; Henderson, K.; Lee, J.F.; Stewart, W.M.; Dhekney, S.A.; Joshee, N. A Review of Paulownia Biotechnology: A Short Rotation, Fast Growing Multipurpose Bioenergy Tree. Am. J. Plant Sci. 2013, 4, 2070–2082. [Google Scholar] [CrossRef]
- Lucas-Borja, M.E.; Wic-Baena, C.; Moreno, J.L.; Dadi, T.; García, C.; Andrés-Abellán, M. Microbial activity in soils under fast-growing Paulownia (Paulownia elongata × fortunei) plantations in Mediterranean areas. Appl. Soil Ecol. 2011, 51, 42–51. [Google Scholar] [CrossRef]
- López, F.; Pérez, A.; Zamudio, M.; De Alva, H.E.; García, J.C. Paulownia as raw material for solid biofuel and cellulose pulp. Biomass Bioenergy 2012, 45, 77–86. [Google Scholar] [CrossRef]
- Durán, V.H.; Jiménez, J.A.; Perea, F.; Rodríguez, C.R.; Francia, J.R. Biomass Yield Potential of Paulownia Trees in a Semi-Arid Mediterranean Environment (S Spain). Int. J. Renew. Energy Res. 2013, 3, 789–793. [Google Scholar]
- Zhu, Z.H.; Chao, C.J.; Lu, X.Y.; Xiong, Y.G. Paulownia in China: Cultivation and Utilization; International Development Research Centre: Ottawa, Canada, 1986; p. 65. [Google Scholar]
- Barton, I.; Nicholas, I.; Ecroyd, C. Paulownia; Forest Research Bulletin 231; New Zealand Forest Research Institute: Rotorua, New Zealand, 2007; p. 71. [Google Scholar]
- Campoe, O.C.; Stape, J.L.; Albaugh, T.; Lee Allen, H.; Fox, T.R.; Rubilar, R.; Binkley, D. Fertilization and irrigation effects on tree level aboveground net primary production, light interception and light use efficiency in a loblolly pine plantation. For. Ecol. Manag. 2013, 288, 43–48. [Google Scholar] [CrossRef]
- Nagy, J. Effect of Irrigation on Maize Yield (Zea mays L.). In Acta Agraria Debreceniensis; University of Debrecen, Centre for Agricultural Sciences, Faculty of Agricultural Sciences, Department of Land Use and Rural Development: Debrecen, Hungary, 2003; pp. 1–6. [Google Scholar]
- FAO. Soil Map of the World. Revised Legend. Technical Paper 20; ISRIC: Wageningen, The Netherlands, 1997; p. 140. [Google Scholar]
- University of Tennessee. Tree Crops for Marginal Farmland-Paulownia; The University of Tennessee, Agricultural Extension Service: Tennessee, TN, USA, 2004; p. 31. [Google Scholar]
- García Morote, F.A.; López Serrano, F.R.; Andrés, M.; Rubio, E.; González Jiménez, J.L.; de las Heras, J. Allometries, biomass stocks and biomass allocation in the thermophilic Spanish juniper woodlands of Southern Spain. For. Ecol. Manag. 2012, 270, 85–93. [Google Scholar] [CrossRef]
- Parresol, B. Assessing tree and stand biomass: A review with examples and critical comparisons. For. Sci. 1999, 45, 573–593. [Google Scholar]
- Neter, J.; Kutner, M.; Wasserman, W.; Nachtsheim, C. Applied Linear Statistical Models, 4th ed.; McGraw-Hill: Chicago, IL, USA, 1996; p. 1408. [Google Scholar]
- Baskerville, G. Use of logarithmic regression in the estimation of plant biomass. Can. J. For. Res. 1972, 2, 49–53. [Google Scholar] [CrossRef]
- Evans, J. The Forests Handbook, Volume 2, Applying Forest Science for Sustainable Management; Wiley-Blackwell: New Jersey, NJ, USA, 2001; p. 816. [Google Scholar]
- Aerts, R.; Chapin, F.S. The Mineral Nutrition of Wild Plants Revisited: A Re-evaluation of Processes and Patterns. Adv. Ecol. Res. 2000, 30, 1–67. [Google Scholar]
- Coyle, D.R.; Coleman, M.D. Forest production responses to irrigation and fertilization are not explained by shifts in allocation. For. Ecol. Manag. 2005, 208, 137–152. [Google Scholar] [CrossRef]
- Landsberg, J.J.; Gower, S.T. Applications of Physiological Ecology to Forest Management; Academic Press: New York, NY, USA, 1997; p. 354. [Google Scholar]
- Chapin, F.S. The mineral nutrition of wild plants. Annu. Rev. Ecol. Sist. 1980, 11, 233–260. [Google Scholar] [CrossRef]
- Hangs, R.D.; Schoenau, J.J.; van Rees, K.C.J.; Knight, J.D. The effect of irrigation on nitrogen uptake and use efficiency of two willow (Salix spp.) biomass energy varieties. Can. J. Plant Sci. 2012, 92, 563–575. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Morote, F.A.; López-Serrano, F.R.; Martínez-García, E.; Andrés-Abellán, M.; Dadi, T.; Candel, D.; Rubio, E.; Lucas-Borja, M.E. Stem Biomass Production of Paulownia elongata × P. fortunei under Low Irrigation in a Semi-Arid Environment. Forests 2014, 5, 2505-2520. https://doi.org/10.3390/f5102505
García-Morote FA, López-Serrano FR, Martínez-García E, Andrés-Abellán M, Dadi T, Candel D, Rubio E, Lucas-Borja ME. Stem Biomass Production of Paulownia elongata × P. fortunei under Low Irrigation in a Semi-Arid Environment. Forests. 2014; 5(10):2505-2520. https://doi.org/10.3390/f5102505
Chicago/Turabian StyleGarcía-Morote, Francisco Antonio, Francisco Ramón López-Serrano, Eduardo Martínez-García, Manuela Andrés-Abellán, Tarek Dadi, David Candel, Eva Rubio, and Manuel Esteban Lucas-Borja. 2014. "Stem Biomass Production of Paulownia elongata × P. fortunei under Low Irrigation in a Semi-Arid Environment" Forests 5, no. 10: 2505-2520. https://doi.org/10.3390/f5102505
APA StyleGarcía-Morote, F. A., López-Serrano, F. R., Martínez-García, E., Andrés-Abellán, M., Dadi, T., Candel, D., Rubio, E., & Lucas-Borja, M. E. (2014). Stem Biomass Production of Paulownia elongata × P. fortunei under Low Irrigation in a Semi-Arid Environment. Forests, 5(10), 2505-2520. https://doi.org/10.3390/f5102505







