Edaphic Determinants of Biomass Hyperdominance in Large Trees of the Amazon
Abstract
1. Introduction
2. Materials and Methods
2.1. Description and Location of the Study Area
2.2. Data Collection
2.2.1. Floristic Inventory
2.2.2. Soil Sampling and Analysis
2.2.3. Biomass Calculation
2.2.4. Data Analysis
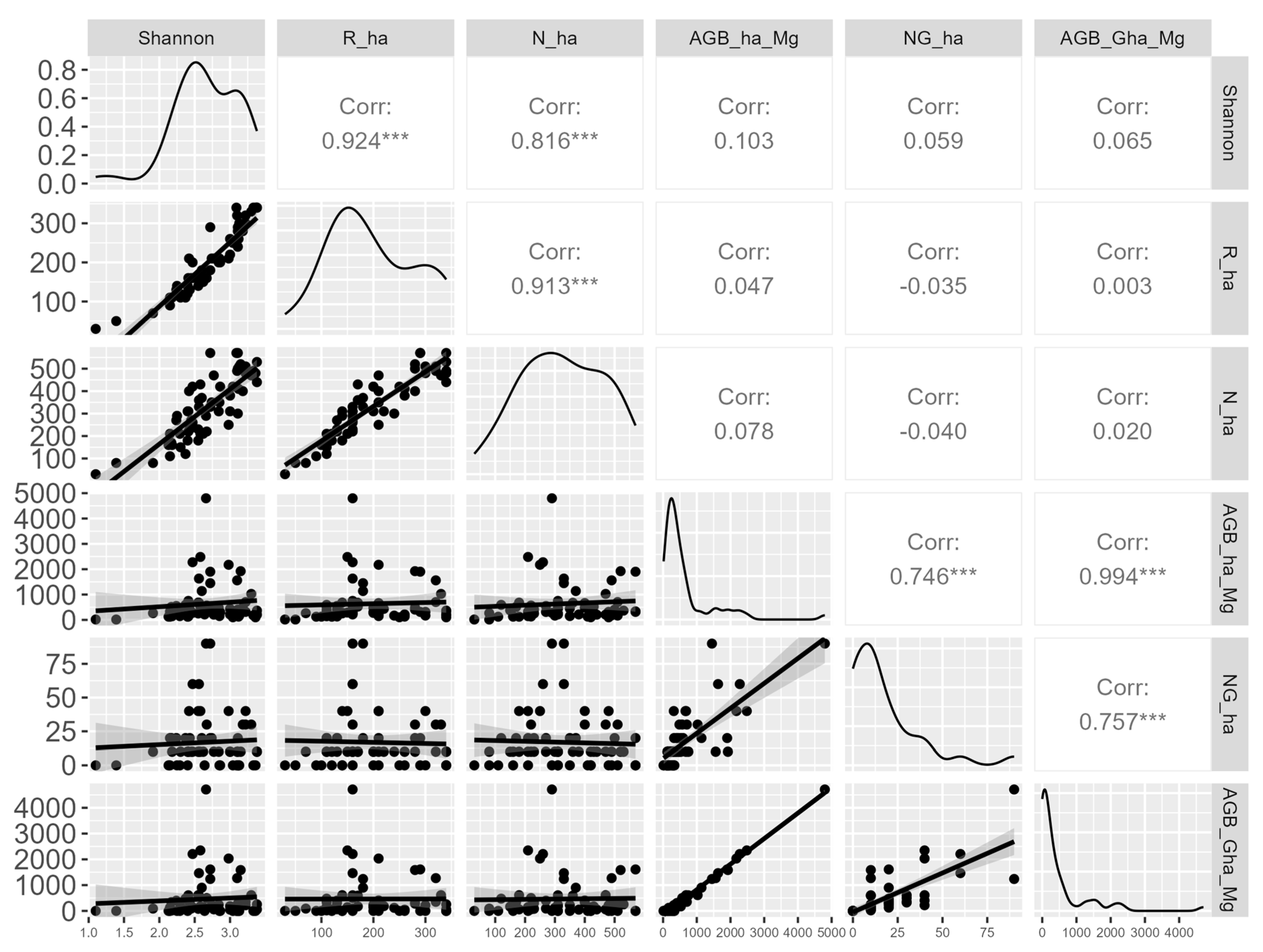
3. Results
4. Discussion
4.1. Edaphic Controls on Large-Tree Biomass
4.2. Limitations and Future Research
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A

| Species | Family | N (Individuals) | Density (ind. ha−1) | DBH (cm) | Height (m) | AGB (Mg ha−1) |
|---|---|---|---|---|---|---|
| Andira sp. | Fabaceae | 1 | 0.167 | 79.6 | 33 | 0.001 |
| Aspidosperma carapanauba | Apocynaceae | 1 | 0.167 | 79.6 | 47 | 0.002 |
| Aspidosperma paraensis | Apocynaceae | 1 | 0.167 | 88.5 | 34 | 0.002 |
| Bertholletia excelsa | Lecythidaceae | 6 | 1 | 164.4 ± 65.3 | 48 ± 11.3 | 0.009 ± 0.009 |
| Bowdichia nitida | Fabaceae | 3 | 0.5 | 85.6 ± 17.4 | 20.3 ± 15 | 0.001 ± 0.001 |
| Bowdichia virgilioides | Fabaceae | 1 | 0.167 | 95.5 | 35 | 0.002 |
| Brosimum parinarioides | Moraceae | 2 | 0.333 | 94.7 ± 27.7 | 35.5 ± 3.5 | 0.002 ± 0.001 |
| Caryocar villosum | Caryocaraceae | 3 | 0.5 | 131.7 ± 54.1 | 37.7 ± 16.3 | 0.005 ± 0.005 |
| Cedrelinga cateniformis | Meliaceae | 2 | 0.333 | 101.1 ± 41.6 | 59.5 ± 7.8 | 0.003 ± 0.002 |
| Chrysophyllum lucentifolium | Sapotaceae | 1 | 0.167 | 79.6 | 33 | 0.001 |
| Corythophora rimosa | Lecythidaceae | 1 | 0.167 | 90.7 | 18 | 0.001 |
| Coutarea hexandra | Rubiaceae | 1 | 0.167 | 79.6 | 47 | 0.001 |
| Dinizia excelsa | Fabaceae | 4 | 0.667 | 197.8 ± 34.5 | 59.2 ± 16.4 | 0.017 ± 0.007 |
| Dipteryx odorata | Fabaceae | 2 | 0.333 | 130.8 ± 27.5 | 46.5 ± 7.8 | 0.006 ± 0.001 |
| Enterolobium schomburgkii | Fabaceae | 3 | 0.5 | 88.1 ± 18.1 | 37.7 ± 16.2 | 0.002 ± 0.000 |
| Eschweilera apiculata | Lecythidaceae | 2 | 0.333 | 85.9 ± 9.0 | 48.5 ± 2.1 | 0.002 ± 0.001 |
| Ficus sp. | Moraceae | 1 | 0.167 | 111.4 | 52 | 0.002 |
| Geissospermum sericeum | Apocynaceae | 1 | 0.167 | 71.6 | 45 | 0.002 |
| Goupia glabra | Goupiaceae | 6 | 1 | 90.2 ± 13.3 | 39.5 ± 9 | 0.002 ± 0.001 |
| Guarea carinata | Meliaceae | 1 | 0.167 | 75.4 | 46 | 0.001 |
| Inga auristellae | Fabaceae | 2 | 0.333 | 189.9 ± 160.5 | 43.5 ± 13.4 | 0.012 ± 0.015 |
| Inga sp. | Fabaceae | 4 | 0.667 | 176.6 ± 99.6 | 42.8 ± 9.1 | 0.009 ± 0.009 |
| Inga striata | Fabaceae | 1 | 0.167 | 73.2 | 48 | 0.001 |
| Laetia procera | Salicaceae | 1 | 0.167 | 109.8 | 62 | 0.004 |
| Lecythis lurida | Lecythidaceae | 1 | 0.167 | 79.6 | 47 | 0.002 |
| Manilkara paraensis | Sapotaceae | 2 | 0.333 | 84.4 ± 15.8 | 48 ± 2.8 | 0.003 ± 0.001 |
| Maquira sclerophylla | Moraceae | 1 | 0.167 | 144.8 | 41 | 0.004 |
| Minquartia guianensis | Coulaceae | 1 | 0.167 | 95.5 | 60 | 0.004 |
| Myrciaria floribunda | Myrtaceae | 5 | 0.833 | 141.2 ± 21.2 | 40.6 ± 2.3 | 0.005 ± 0.002 |
| N.I | N.I | 2 | 0.333 | 75.3 ± 6.1 | 39.5 ± 10.6 | 0.001 ± 0.001 |
| Nadenanthera peregrina | Fabaceae | 1 | 0.167 | 124.1 | 54 | 0.004 |
| Naucleopsis sp. | Moraceae | 1 | 0.167 | 157.6 | 42 | 0.006 |
| Ocotea sp. | Lauraceae | 1 | 0.167 | 89.1 | 59 | 0.002 |
| Parkia multijuga | Fabaceae | 1 | 0.167 | 102.5 | 36 | 0.001 |
| Pouteria sp. | Sapotaceae | 2 | 0.333 | 175.1 ± 78.8 | 35 ± 18.4 | 0.008 ± 0.009 |
| Pouteria vernicosa | Sapotaceae | 1 | 0.167 | 79.7 | 47 | 0.002 |
| Protium altsonii | Burseraceae | 4 | 0.667 | 101.7 ± 24.1 | 39.5 ± 5.6 | 0.002 ± 0.001 |
| Protium decandrum | Burseraceae | 2 | 0.333 | 140.5 ± 78.1 | 37.5 ± 10.6 | 0.004 ± 0.004 |
| Protium sp. | Burseraceae | 1 | 0.167 | 141.6 | 41 | 0.004 |
| Pseudopiptadenia suaveolens | Fabaceae | 4 | 0.667 | 87.3 ± 9.4 | 50 ± 11.9 | 0.002 ± 0.001 |
| Qualea paraensis | Vochysiaceae | 2 | 0.333 | 105.2 ± 18.2 | 44 ± 8.5 | 0.003 ± 0.000 |
| Simarouba amara | Simaroubaceae | 2 | 0.333 | 95.5 ± 22.5 | 49.5 ± 3.5 | 0.002 ± 0.001 |
| Swartzia polyphylla | Fabaceae | 1 | 0.167 | 321.5 | 63 | 0.035 |
| Tachigali myrmecophila | Fabaceae | 3 | 0.5 | 114.8 ± 42.6 | 37.7 ± 4.6 | 0.002 ± 0.002 |
| Terminalia amazonia | Combretaceae | 3 | 0.5 | 155.1 ± 110.0 | 53.3 ± 8.7 | 0.009 ± 0.011 |
| Tetragastris panamensis | Burseraceae | 1 | 0.167 | 70.0 | 11 | 0.000 |
| Theobroma subincanum | Malvaceae | 2 | 0.333 | 149.6 ± 56.3 | 41 ± 5.7 | 0.004 ± 0.003 |
| Toulicia acutifolia | Sapindaceae | 1 | 0.167 | 73.2 | 32 | 0.001 |
| Trattinnickia rhoifolia | Burseraceae | 1 | 0.167 | 80.1 | 33 | 0.001 |
| Virola sp. | Myristicaceae | 2 | 0.333 | 128.1 ± 21.4 | 47 ± 12.7 | 0.003 ± 0.002 |
| Virola surinamensis | Myristicaceae | 1 | 0.167 | 144.8 | 41 | 0.003 |
| Vochysia guianensis | Vochysiaceae | 1 | 0.167 | 86.9 | 49 | 0.002 |
| Variable | AG | Cupixi | Ipitinga | Iratapuru | Urucupatá | Statistic | p-Value |
|---|---|---|---|---|---|---|---|
| pH | 4.37 ± 0.21 a | 4.05 ± 0.08 b | 4.36 ± 0.47 ab | 4.33 ± 0.21 a | 4.75 ± 1.00 a | χ2 = 18.559 | <0.001 |
| SOM | 30.11 ± 8.19 a | 28.92 ± 3.58 a | 26.69 ± 4.30 a | 16.51 ± 2.53 b | 27.50 ± 4.95 a | F = 13.716 | <0.001 |
| P | 2.50 ± 1.15 a | 3.17 ± 4.09 ab | 1.37 ± 0.59 bc | 2.08 ± 0.90 ab | 1.00 ± 0.23 c | χ2 = 18.980 | <0.001 |
| K | 0.04 ± 0.01 a | 0.02 ± 0.00 b | 29.06 ± 9.09 c | 0.02 ± 0.01 b | 17.40 ± 5.91 d | χ2 = 50.764 | <0.001 |
| Ca_Mg | 0.43 ± 0.15 a | 0.08 ± 0.08 b | 0.52 ± 0.53 ac | 0.18 ± 0.07 cd | 0.17 ± 0.15 bd | χ2 = 27.973 | <0.001 |
| Al | 1.30 ± 0.25 ab | 1.30 ± 0.19 ab | 1.05 ± 0.57 b | 1.33 ± 0.31 ab | 1.60 ± 0.32 a | F = 3.790 | 0.009 |
| H_Al | 6.72 ± 1.44 a | 7.62 ± 1.04 a | 4.03 ± 0.68 b | 3.93 ± 1.29 b | 4.61 ± 0.61 b | F = 30.041 | <0.001 |
| CTC_pH7 | 7.17 ± 1.47 a | 7.71 ± 1.02 a | 10.97 ± 12.31 ab | 4.12 ± 1.27 b | 9.68 ± 8.95 ab | χ2 = 23.145 | <0.001 |
| BS | 6.58 ± 2.35 ab | 1.08 ± 1.16 c | 28.15 ± 31.33 a | 5.00 ± 2.63 b | 24.34 ± 35.02 ab | χ2 = 28.831 | <0.001 |
| AS | 74.08 ± 7.70 a | 94.42 ± 5.20 b | 52.04 ± 38.52 a | 87.58 ± 5.37 c | 65.50 ± 39.43 ac | χ2 = 25.839 | <0.001 |
| Clay | 309.99 ± 51.33 a | 460.00 ± 40.47 b | 309.17 ± 140.55 a | 300.08 ± 93.05 a | 480.75 ± 98.37 b | χ2 = 29.459 | <0.001 |
| Coarse_Sand | 337.33 ± 49.76 a | 290.83 ± 30.92 a | 298.58 ± 117.44 a | 313.75 ± 73.10 a | 255.83 ± 131.54 a | F = 1.361 | 0.259 |
| Fine_Sand | 124.83 ± 16.27 a | 51.83 ± 15.40 b | 240.08 ± 122.92 c | 105.50 ± 15.80 d | 174.50 ± 124.29 acd | χ2 = 33.879 | <0.001 |
| Total_Sand | 462.17 ± 59.42 ab | 342.67 ± 25.18 c | 538.67 ± 102.64 a | 419.25 ± 80.61 bc | 430.33 ± 102.44 bc | F = 9.568 | <0.001 |
| Silt | 227.84 ± 40.42 a | 197.33 ± 33.62 a | 152.17 ± 92.00 b | 280.67 ± 51.09 c | 88.92 ± 32.60 b | χ2 = 38.387 | <0.001 |
| Abbreviation | Full Name | Unit |
|---|---|---|
| pH | Soil pH (H2O) | – |
| SOM | Soil organic matter | g kg−1 |
| P | Available phosphorus | mg kg−1 |
| K | Exchangeable potassium | cmolc kg−1 |
| Ca_Mg | Calcium + magnesium | cmolc kg−1 |
| Al | Exchangeable aluminum | cmolc kg−1 |
| H_Al | Potential acidity | cmolc kg−1 |
| CTC_pH 7 | Cation exchange capacity (pH 7) | cmolc kg−1 |
| BS | Base saturation | % |
| AS | Aluminum saturation | % |
| Clay | Clay content | % |
| Silt | Silt content | % |
| Coarse_Sand | Coarse sand | % |
| Fine_Sand | Fine sand | % |
| Total_Sand | Total sand | % |
| Variable | edf | F_Value | p_Value | Deviance_Explained (%) |
|---|---|---|---|---|
| pH | 5.02 | 5.33 | 0.001 | 74.6 |
| SOM | 1.00 | 6.82 | 0.014 | |
| P | 2.66 | 5.43 | 0.004 | |
| Al | 4.43 | 4.75 | 0.002 |
| Edaphic Variable | Functional Threshold | Unit |
|---|---|---|
| pH | 3.52–6.17 | – |
| P | 1.60–6.50 | mg kg−1 |
| Al | 0.73–1.42 | cmolc kg−1 |
| SOM | No defined threshold | g kg−1 |
References
- Marengo, J.A.; Souza, C.M.; Thonicke, K.; Burton, C.; Halladay, K.; Betts, R.A.; Alves, L.M.; Soares, W.R. Changes in Climate and Land Use Over the Amazon Region: Current and Future Variability and Trends. Front. Earth Sci. 2018, 6, 228. [Google Scholar] [CrossRef]
- Quesada, C.A.; Paz, C.; Oblitas Mendoza, E.; Phillips, O.L.; Saiz, G.; Lloyd, J. Variations in Soil Chemical and Physical Properties Explain Basin-Wide Amazon Forest Soil Carbon Concentrations. SOIL 2020, 6, 53–88. [Google Scholar] [CrossRef]
- Pinho, B.X.; Peres, C.A.; Leal, I.R.; Tabarelli, M. Chapter Seven—Critical Role and Collapse of Tropical Mega-Trees: A Key Global Resource. In Advances in Ecological Research; Dumbrell, A.J., Turner, E.C., Fayle, T.M., Eds.; Tropical Ecosystems in the 21st Century; Academic Press: Cambridge, MA, USA, 2020; Volume 62, pp. 253–294. [Google Scholar]
- Enquist, B.J.; Abraham, A.J.; Harfoot, M.B.J.; Malhi, Y.; Doughty, C.E. The Megabiota Are Disproportionately Important for Biosphere Functioning. Nat. Commun. 2020, 11, 699. [Google Scholar] [CrossRef] [PubMed]
- de Lima, R.B.; Görgens, E.B.; da Silva, D.A.S.; de Oliveira, C.P.; Batista, A.P.B.; Caraciolo Ferreira, R.L.; Costa, F.R.C.; Ferreira de Lima, R.A.; da Silva Aparício, P.; de Abreu, J.C.; et al. Giants of the Amazon: How Does Environmental Variation Drive the Diversity Patterns of Large Trees? Glob. Change Biol. 2023, 29, 4861–4879. [Google Scholar] [CrossRef]
- de Lima, R.B.; da Silva, D.A.S.; Nunes, M.H.; de Lima Bittencourt, P.R.; Groenendijk, P.; de Oliveira, C.P.; de Souza, D.G.; Ferreira, R.L.C.; da Silva, J.A.A.; Aguirre-Gutiérrez, J.; et al. Mapping Giant-Tree Density in the Amazon. bioRxiv 2025. [Google Scholar] [CrossRef]
- Harris, D.J.; Ndolo Ebika, S.T.; Sanz, C.M.; Madingou, M.P.N.; Morgan, D.B. Large Trees in Tropical Rain Forests Require Big Plots. Plants People Planet 2021, 3, 282–294. [Google Scholar] [CrossRef]
- Slik, J.W.F.; Paoli, G.; McGuire, K.; Amaral, I.; Barroso, J.; Bastian, M.; Blanc, L.; Bongers, F.; Boundja, P.; Clark, C.; et al. Large Trees Drive Forest Aboveground Biomass Variation in Moist Lowland Forests across the Tropics. Glob. Ecol. Biogeogr. 2013, 22, 1261–1271. [Google Scholar] [CrossRef]
- Bradford, M.; Murphy, H.T. The importance of large-diameter trees in the wet tropical rainforests of Australia. PLoS ONE 2019, 14, e0208377. [Google Scholar] [CrossRef]
- Gorgens, E.B.; Motta, A.Z.; Assis, M.; Nunes, M.H.; Jackson, T.; Coomes, D.; Rosette, J.; Aragão, L.E.O.E.C.; Ometto, J.P. The Giant Trees of the Amazon Basin. Front. Ecol. Environ. 2019, 17, 373–374. [Google Scholar] [CrossRef]
- Quesada, C.A.; Phillips, O.L.; Schwarz, M.; Czimczik, C.I.; Baker, T.R.; Patiño, S.; Fyllas, N.M.; Hodnett, M.G.; Herrera, R.; Almeida, S.; et al. Basin-Wide Variations in Amazon Forest Structure and Function Are Mediated by Both Soils and Climate. Biogeosciences 2012, 9, 2203–2246. [Google Scholar] [CrossRef]
- Draper, F.C.; Costa, F.R.C.; Arellano, G.; Phillips, O.L.; Duque, A.; Macía, M.J.; ter Steege, H.; Asner, G.P.; Berenguer, E.; Schietti, J.; et al. Amazon Tree Dominance across Forest Strata. Nat. Ecol. Evol. 2021, 5, 757–767. [Google Scholar] [CrossRef]
- Fauset, S.; Johnson, M.O.; Gloor, M.; Baker, T.R.; Monteagudo, M.A.; Brienen, R.J.W.; Feldpausch, T.R.; Lopez-Gonzalez, G.; Malhi, Y. Hyperdominance in Amazonian Forest Carbon Cycling. Nat. Commun. 2015, 6, 6857. [Google Scholar] [CrossRef] [PubMed]
- Lutz, J.A.; Furniss, T.J.; Johnson, D.J.; Davies, S.J.; Allen, D.; Alonso, A.; Anderson-Teixeira, K.J.; Andrade, A.; Baltzer, J.; Becker, K.M.L.; et al. Global Importance of Large-Diameter Trees. Glob. Ecol. Biogeogr. 2018, 27, 849–864. [Google Scholar] [CrossRef]
- Carvalho, R.L.; Resende, A.F.; Barlow, J.; França, F.M.; Moura, M.R.; Maciel, R.; Alves-Martins, F.; Shutt, J.; Nunes, C.A.; Elias, F.; et al. Pervasive Gaps in Amazonian Ecological Research. Curr. Biol. 2023, 33, 3495–3504. [Google Scholar] [CrossRef]
- Alvares, C.A.; Stape, J.L.; Sentelhas, P.C.; Leonardo, J.; Gonçalves, M.; Sparovek, G. Köppen’s Climate Classification Map for Brazil. Meteorol. Z. 2013, 26, 711–728. [Google Scholar] [CrossRef]
- IFN—Metodologia—Serviço Florestal Brasileiro. Available online: https://www.gov.br/florestal/pt-br/assuntos/ifn/metodologia (accessed on 31 December 2025).
- Coelho, A.M. Amostragem de Solos: A base para aplicação de corretivos e fertilizantes. In Embrapa Milho e Sorgo. Comunicado Técnico; Embrapa Milho e Sorgo: Sete Lagoas, Brazil, 2003; p. 73. [Google Scholar]
- Guarçoni, A.; Alvarez V, V.H.; Sobreira, F.M.; Guarçoni, A.; Alvarez V, V.H.; Sobreira, F.M. Fundamentação teórica dos sistemas de amostragem de solo de acordo com a variabilidade de características químicas. Terra Latinoam. 2017, 35, 343–352. [Google Scholar] [CrossRef]
- Teixeira, P.C.; Donagemma, G.K.; Fontana, A.; Teixeira, W.G. Manual de Métodos de Análise de Solo; Embrapa Solos: Rio de Janeiro, Brazil, 2017. [Google Scholar]
- Réjou-Méchain, M.; Tanguy, A.; Piponiot, C.; Chave, J.; Hérault, B. Biomass: An r Package for Estimating above-Ground Biomass and Its Uncertainty in Tropical Forests. Methods Ecol. Evol. 2017, 8, 1163–1167. [Google Scholar] [CrossRef]
- Chave, J.; Réjou-Méchain, M.; Búrquez, A.; Chidumayo, E.; Colgan, M.S.; Delitti, W.B.C.; Duque, A.; Eid, T.; Fearnside, P.M.; Goodman, R.C.; et al. Improved Allometric Models to Estimate the Aboveground Biomass of Tropical Trees. Glob. Change Biol. 2014, 20, 3177–3190. [Google Scholar] [CrossRef]
- Legendre, P.; Legendre, L. Numerical Ecology; Elsevier: Amsterdam, The Netherlands, 2012; ISBN 978-0-444-53869-7. [Google Scholar]
- Legendre, P.; Gallagher, E.D. Ecologically Meaningful Transformations for Ordination of Species Data. Oecologia 2001, 129, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Dray, S.; David, B.; Guillaume, B.; Daniel, B.; Sylvie, C.; Guillaume, G. Adespatial: Multivariate Multiscale Spatial Analysis 0.3-28; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Oksanen, J.; Blanchet, F.; Friendly, M.; Kindt, R. Vegan: Community Ecology Package (R Package Version 2.5–5), 2019.
- Burnham, K.P.; Anderson, D.R. (Eds.) Advanced Issues and Deeper Insights. In Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach; Springer: New York, NY, USA, 2002; pp. 267–351. ISBN 978-0-387-22456-5. [Google Scholar]
- Team R Core R. The R Project for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2024. [Google Scholar]
- Wood, S. Generalized Additive Models: An Introduction With R; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Guisan, A.; Zimmermann, N.E. Predictive Habitat Distribution Models in Ecology. Ecol. Model. 2000, 135, 147–186. [Google Scholar] [CrossRef]
- Stephenson, N.L.; Das, A.J.; Condit, R.; Russo, S.E.; Baker, P.J.; Beckman, N.G.; Coomes, D.A.; Lines, E.R.; Morris, W.K.; Rüger, N.; et al. Rate of Tree Carbon Accumulation Increases Continuously with Tree Size. Nature 2014, 507, 90–93. [Google Scholar] [CrossRef]
- Luizão, F.J.; Fearnside, P.M.; Cerri, C.E.P.; Lehmann, J. The Maintenance of Soil Fertility in Amazonian Managed Systems. In Geophysical Monograph Series; Keller, M., Bustamante, M., Gash, J., Silva Dias, P., Eds.; American Geophysical Union: Washington, DC, USA, 2009; Volume 186, pp. 311–336. ISBN 978-0-87590-476-4. [Google Scholar]
- Quesada, C.; Lloyd, J.; Schwarz, M.; Patiño, S.; Baker, T.R.; Czimczik, C.; Paiva, R. Variations in Chemical and Physical Properties of Amazon Forest Soils in Relation to Their Genesis. Biogeosciences 2010, 7, 1515–1541. [Google Scholar] [CrossRef]
- Sayer, E.J.; Banin, L.F. Tree Nutrient Status and Nutrient Cycling in Tropical Forest—Lessons from Fertilization Experiments. Trop. Tree Physiol. 2016, 6, 275–297. [Google Scholar] [CrossRef]
- McCauley, A.; Jones, C.; Jacobsen, J. Soil pH and Organic Matter. Nutr. Manag. Modul. 2009, 8, 1–12. [Google Scholar]
- Poggio, L.; de Sousa, L.M.; Batjes, N.H.; Heuvelink, G.B.M.; Kempen, B.; Ribeiro, E.; Rossiter, D. SoilGrids 2.0: Producing Soil Information for the Globe with Quantified Spatial Uncertainty. SOIL 2021, 7, 217–240. [Google Scholar] [CrossRef]
- van der Sande, M.T.; Arets, E.J.M.M.; Peña-Claros, M.; Hoosbeek, M.R.; Cáceres-Siani, Y.; van der Hout, P.; Poorter, L. Soil Fertility and Species Traits, but Not Diversity, Drive Productivity and Biomass Stocks in a Guyanese Tropical Rainforest. Funct. Ecol. 2018, 32, 461–474. [Google Scholar] [CrossRef]
- Hornink, B.; Zuidema, P.A.; van der Sleen, P.; Zanne, A.E.; Assis-Pereira, G.; Ortega Rodriguez, D.R.; Fontana, C.; Portal-Cahuana, L.A.; Requena-Rojas, E.J.; Barbosa, A.C.M.C.; et al. Biomass Production of Tropical Trees across Space and Time: The Shifting Roles of Diameter Growth and Wood Density. J. Ecol. 2025, 113, 3141–3158. [Google Scholar] [CrossRef]
- Isa, N.; Razak, S.A.; Abdullah, R.; Khan, M.N.; Hamzah, S.N.; Kaplan, A.; Dossou-Yovo, H.O.; Ali, B.; Razzaq, A.; Wahab, S.; et al. Relationship between the Floristic Composition and Soil Characteristics of a Tropical Rainforest (TRF). Forests 2023, 14, 306. [Google Scholar] [CrossRef]
- Brazilian Institute of Geography and Statistics—IBGE Amapá—Mapa Exploratório de Solos Em Escala 1:5.000.000. Available online: https://geoftp.ibge.gov.br/informacoes_ambientais/pedologia/mapas/unidades_da_federacao/ap_pedologia.pdf (accessed on 14 November 2025).
- Brazilian Institute of Geography and Statistics—IBG Pará—Mapa Exploratório de Solos Em Escala 1:5.000.000. Available online: https://geoftp.ibge.gov.br/informacoes_ambientais/pedologia/mapas/ (accessed on 14 November 2025).
- Kochian, L.V.; Piñeros, M.A.; Liu, J.; Magalhaes, J.V. Plant Adaptation to Acid Soils: The Molecular Basis for Crop Aluminum Resistance. Annu. Rev. Plant Biol. 2015, 66, 571–598. [Google Scholar] [CrossRef]
- Reategui-Betancourt, J.L.; de Freitas, L.J.M.; Nascimento, R.G.M.; Briceño, G.; Figueiredo, A.E.S.; Rezende, A.V.; Alder, D. Species Grouping and Diameter Growth of Trees in the Eastern Amazon: Influence of Environmental Factors after Reduced-Impact Logging. For. Ecol. Manag. 2025, 578, 122465. [Google Scholar] [CrossRef]
- Lange, D.F.; Schröter, S.A.; da Luz, F.M.; Pires, E.; Santos, Y.R.; da Silva, J.S.; Hildmann, S.; Hoffmann, T.; Ferreira, S.J.F.; Schäfer, T.; et al. Cycling of Dissolved Organic Nutrients and Indications for Nutrient Limitations in Contrasting Amazon Rainforest Ecosystems. Biogeochemistry 2024, 167, 1567–1588. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Pereira, M.; Reategui-Betancourt, J.L.; Lima, R.d.; Bittencourt, P.; Gorgens, E.; Abreu, G.; Guedes, M.; Silva, J.; Sousa, C.d.; Silva, J.P.d.; et al. Edaphic Determinants of Biomass Hyperdominance in Large Trees of the Amazon. Forests 2026, 17, 367. https://doi.org/10.3390/f17030367
Pereira M, Reategui-Betancourt JL, Lima Rd, Bittencourt P, Gorgens E, Abreu G, Guedes M, Silva J, Sousa Cd, Silva JPd, et al. Edaphic Determinants of Biomass Hyperdominance in Large Trees of the Amazon. Forests. 2026; 17(3):367. https://doi.org/10.3390/f17030367
Chicago/Turabian StylePereira, Manuelle, Jorge Luis Reategui-Betancourt, Robson de Lima, Paulo Bittencourt, Eric Gorgens, Gustavo Abreu, Marcelino Guedes, José Silva, Carla de Sousa, Joselane Priscila da Silva, and et al. 2026. "Edaphic Determinants of Biomass Hyperdominance in Large Trees of the Amazon" Forests 17, no. 3: 367. https://doi.org/10.3390/f17030367
APA StylePereira, M., Reategui-Betancourt, J. L., Lima, R. d., Bittencourt, P., Gorgens, E., Abreu, G., Guedes, M., Silva, J., Sousa, C. d., Silva, J. P. d., Souza, E. d., & Silva, D. A. (2026). Edaphic Determinants of Biomass Hyperdominance in Large Trees of the Amazon. Forests, 17(3), 367. https://doi.org/10.3390/f17030367






