Influence of Edaphic Properties in Determining Forest Community Patterns of the Zabarwan Mountain Range in the Kashmir Himalayas
Abstract
:1. Introduction
2. Materials and Methods
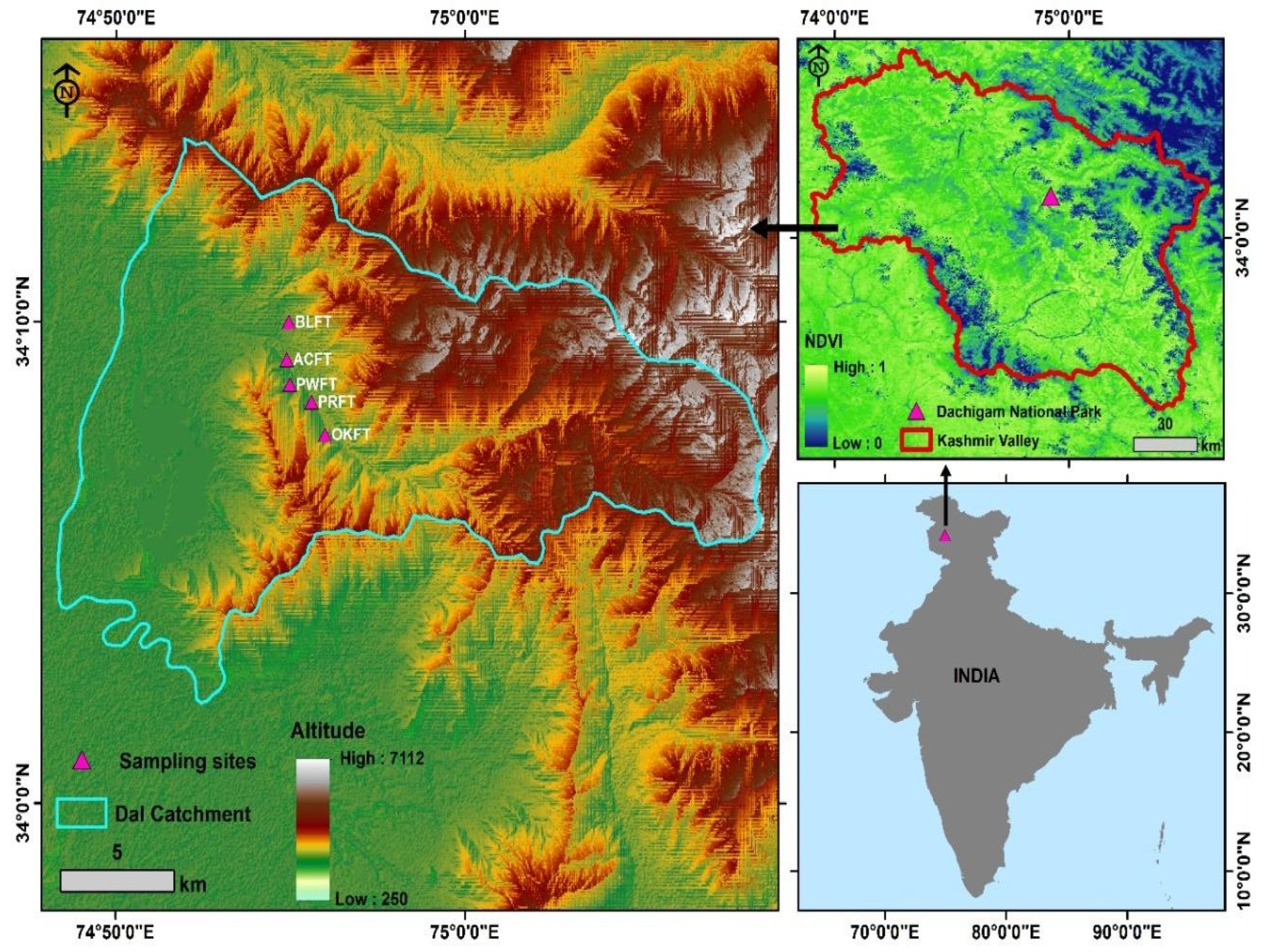
2.1. Study Area
2.2. Sampling and Data Analysis
2.3. Data Analysis
3. Results
3.1. Plant Composition and Distribution
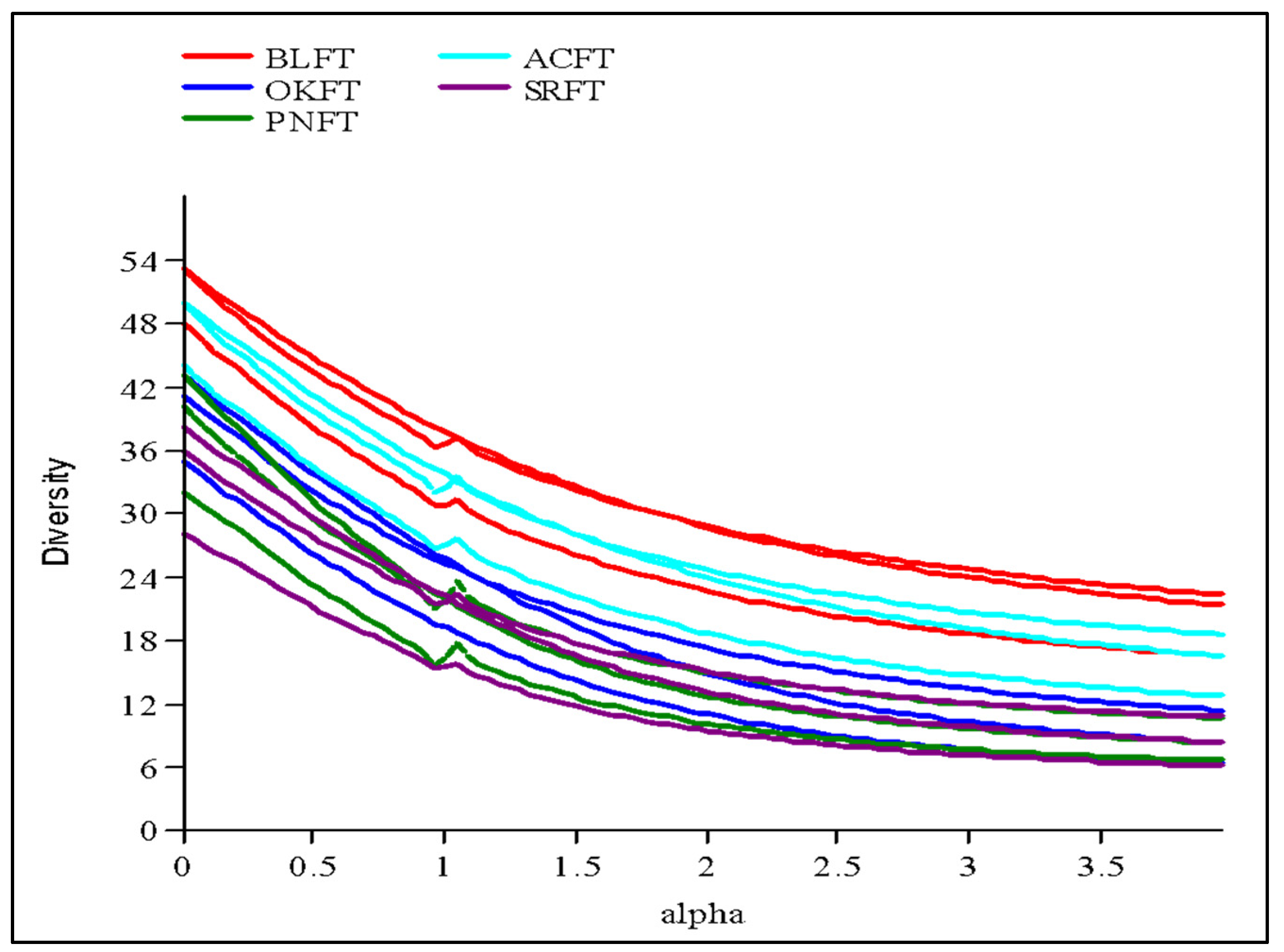
3.2. Diversity and Phytosociological Attributes
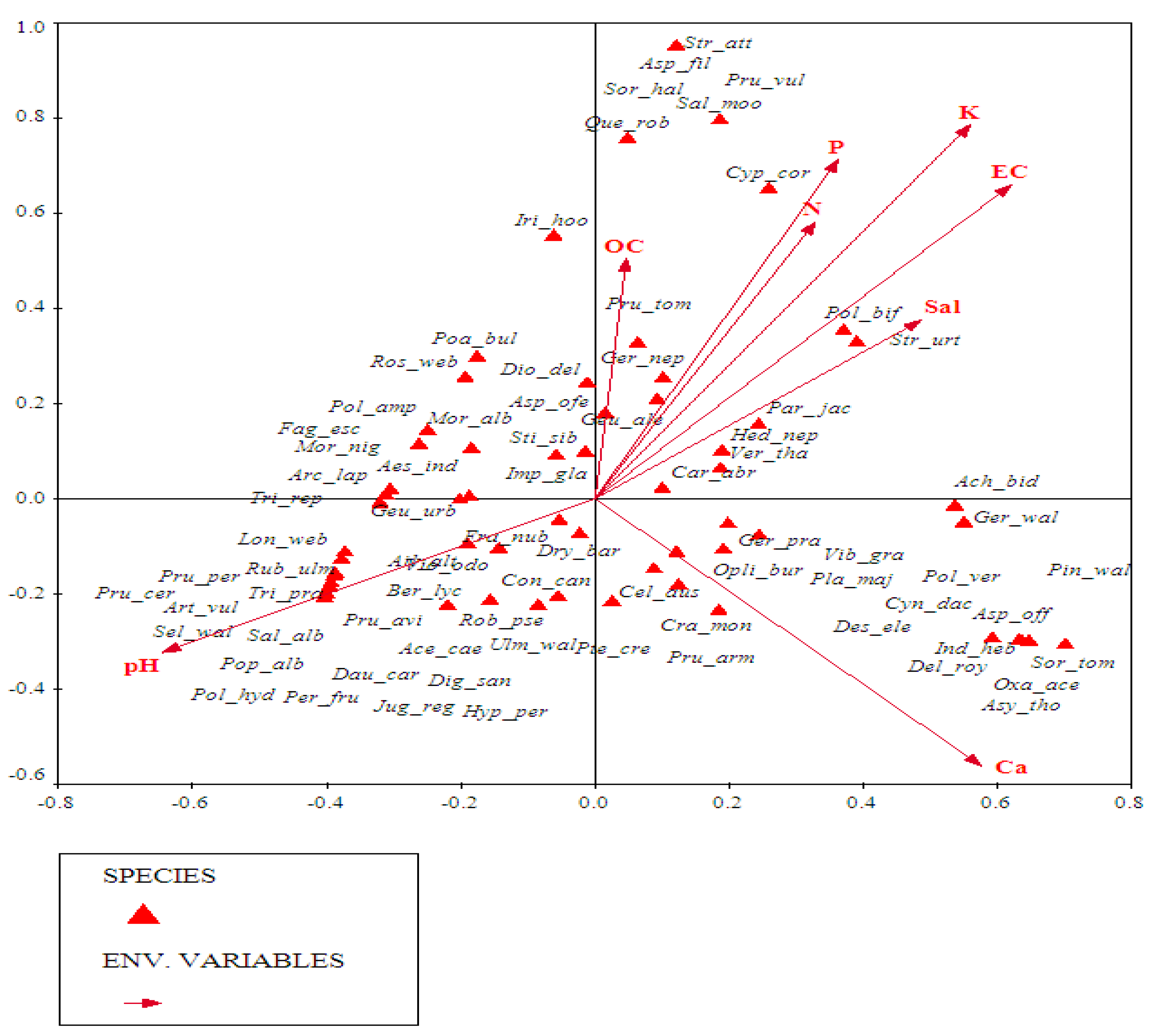
3.3. Vegetation Ordination Approaches
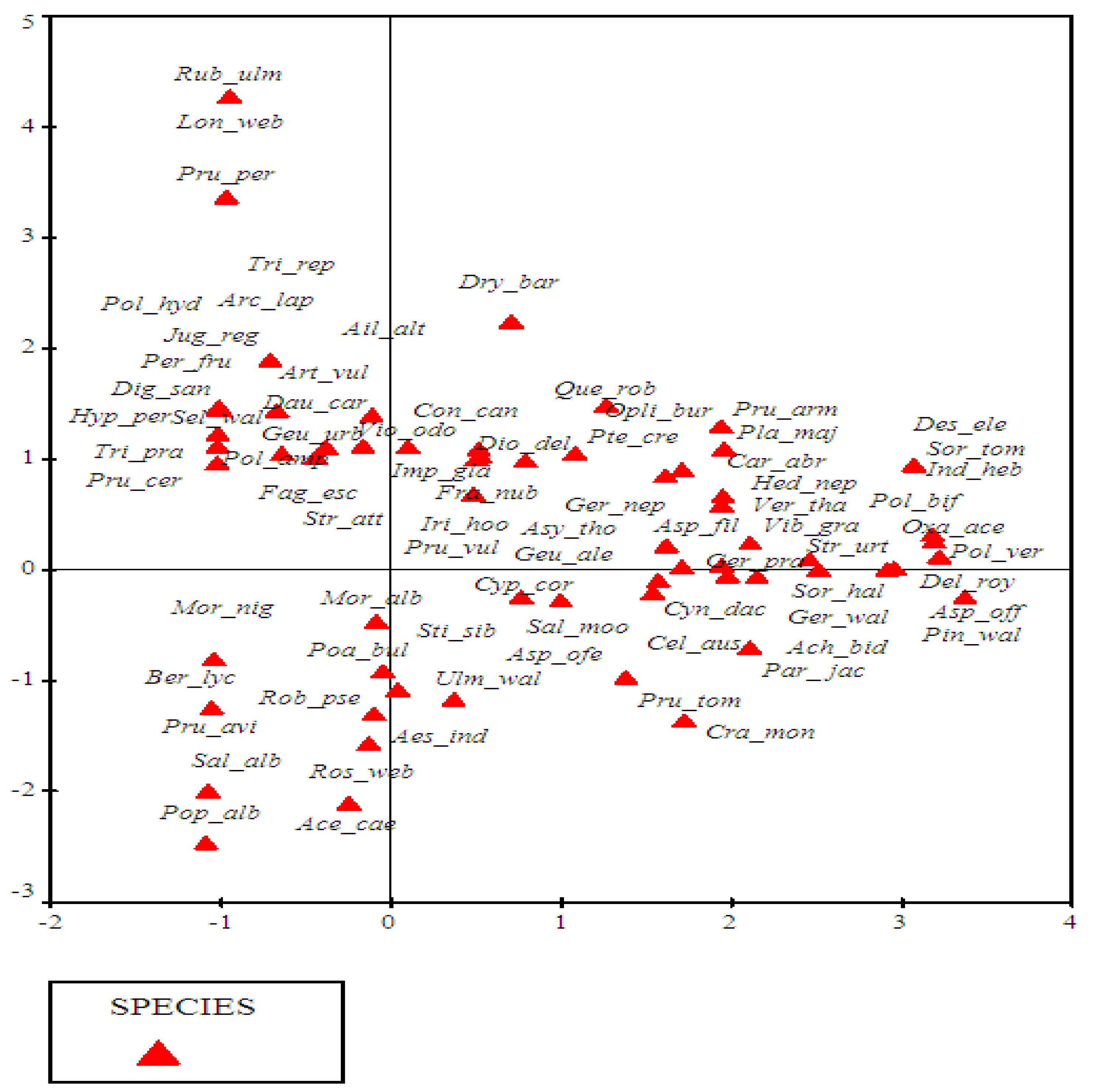
3.3.1. DCA Ordination
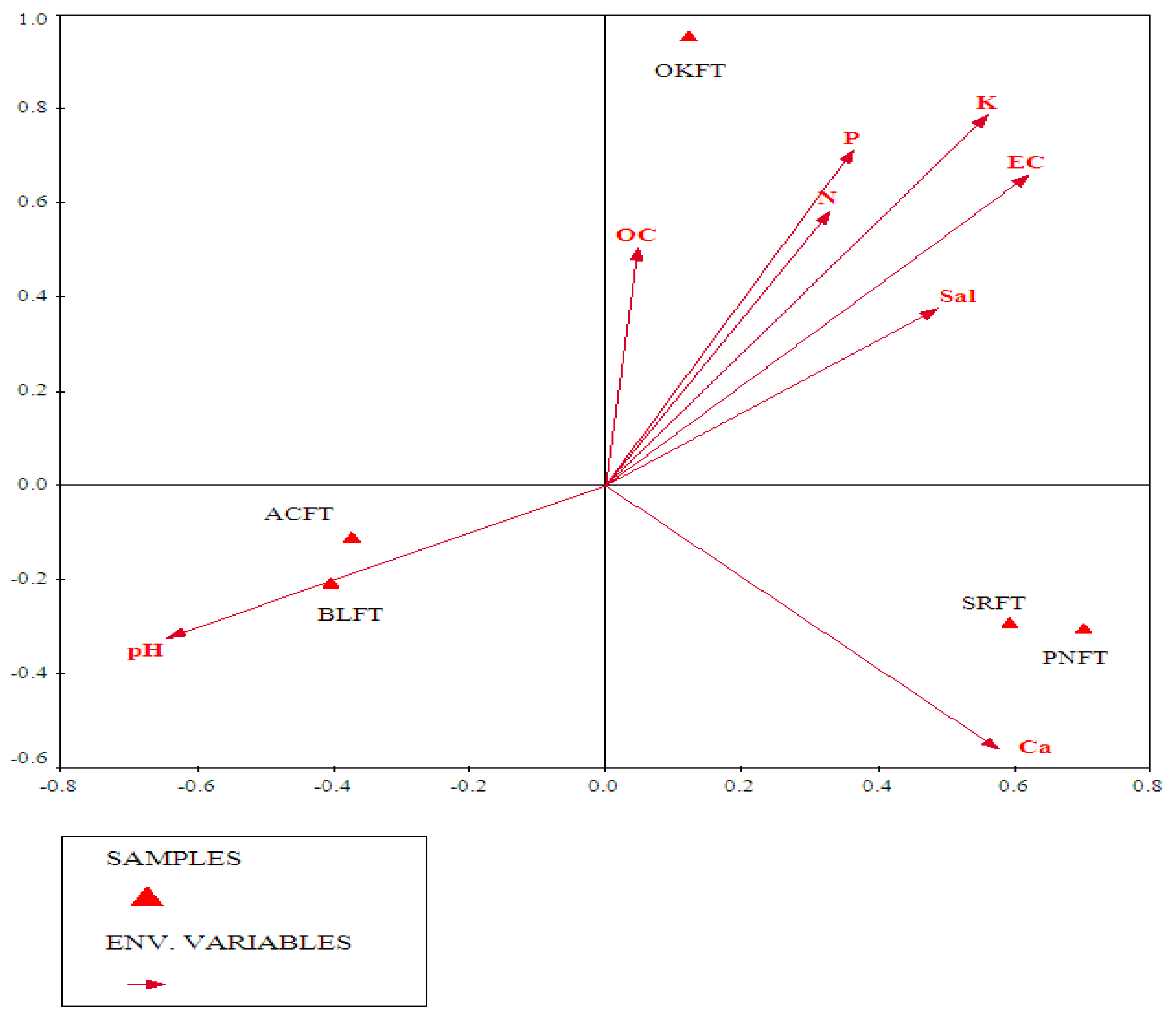
3.3.2. Role of Soil Parameters in Vegetation Patterns
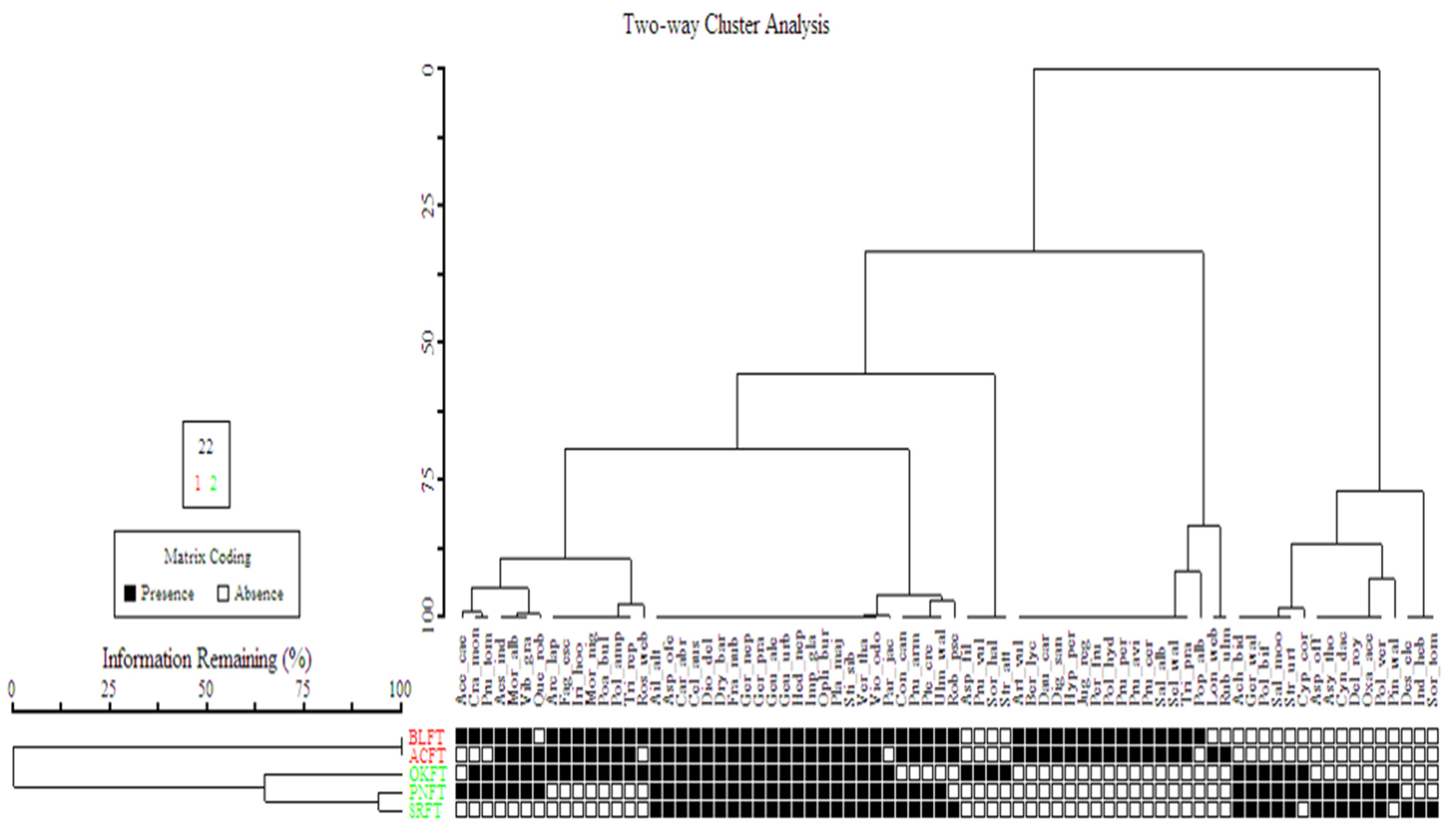
3.3.3. Vegetation Classification
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Perrigo, A.; Hoorn, C.; Antonelli, A. Why mountains matter for biodiversity. J. Biogeogr. 2020, 47, 315–325. [Google Scholar] [CrossRef] [Green Version]
- Haq, S.M.; Khuroo, A.A.; Malik, A.H.; Rashid, I.; Ahmad, R.; Hamid, M.; Dar, G.H. Forest ecosystems of Jammu and Kashmir State. In Biodiversity of the Himalaya; Springer: Singapore, 2020. [Google Scholar]
- Bargali, K.; Beena, J.; Bargali, S.S.; Singh, S.P. Diversity within Oaks. Inter. Oaks 2014, 25, 57–70. [Google Scholar]
- Gosain, B.G.; Negi, G.C.S.; Dhyani, P.P.; Bargali, S.S.; Saxena, R. Ecosystem services of forests: Carbon Stock in vegetation and soil components in a watershed of Kumaun Himalaya, India. Int. J. Ecol. Environ. Sci. 2015, 41, 177–188. [Google Scholar]
- Rana, S.; Bargali, K.; Bargali, S.S. Assessment of plant diversity, regeneration status, biomass and carbon stock in a Central Himalayan cypress forest. Int. J. Biodiv. Conserv. 2015, 7, 321–329. [Google Scholar]
- Manandhar, P.; Rasul, G. The role of the Hindu Kush–Himalayan (HKH) mountain system in the context of a changing climate: A panel discussion. Mt. Res. Dev. 2009, 29, 184–187. [Google Scholar] [CrossRef]
- Bargali, S.S.; Awasthi, P.; Bargali, K. Composition, structure and productivity of the herbaceous vegetation of five forest stands varying in soil moisture and nitrogen in Central Himalaya, India. Euras. J. For. Sci. 2021, 9, 29–42. [Google Scholar] [CrossRef]
- Rasul, G. The role of the Himalayan Mountain systems in food security and agricultural sustainability in South Asia. Int. J. Rural Mgt. 2010, 6, 95–116. [Google Scholar] [CrossRef]
- Hester, A.; Brooker, R. Threatened habitats: Marginal vegetation in upland areas. Issues Environ. Sci. Technol. 2007, 25, 107–134. [Google Scholar]
- Mondoni, A.; Rossi, G.; Orsenigo, S.; Probert, R.J. Climate warming could shift the timing of seed germination in alpine plants. Ann. Bot. 2012, 110, 155–164. [Google Scholar] [CrossRef]
- Foxcroft, L.C.; Richardson, D.M.; Pyšek, P.; Genovesi, P. Invasive alien plants in protected areas threats, opportunities, and the way forward. In Plant Invas Protect Areas; Springer: Dordrecht, The Netherlands, 2013. [Google Scholar]
- Khan, M.; Khan, M.S.; Ilyas, M.; Alqarawi, A.A.; Ahmad, Z.; Abd-Allah, F.E. Plant species and community’s assessment in interaction with edaphic and topographic factors; an ecological study of the mount Eelum District Swat, Pakistan. SJBS 2017, 24, 778–786. [Google Scholar]
- Saima, S.; Altaf, A.; Faiz, M.H.; Shahnaz, F.; Wu, G. Vegetation patterns and composition of mixed coniferous forests along an altitudinal gradient in the Western Himalayas of Pakistan. Austr. J. Forens. Sci. 2018, 135, 159–180. [Google Scholar]
- Arruda, D.M.; Schaefer, C.E.; Corrêa, G.R.; Rodrigues, P.M.; Duque-Brasil, R.; Ferreira-JR, W.G.; Oliveira-Filho, A.T. Landforms and soil attributes determine the vegetation structure in the Brazilian semiarid. Folia Geobot. 2015, 50, 175–184. [Google Scholar] [CrossRef]
- Ehrlich, P.R. Conservation in temperate forests: What do we need to know and do? For. Ecol. Mgt. 1996, 85, 9–19. [Google Scholar] [CrossRef]
- Becknell, J.M.; Powers, J.S. Stand age and soils as drivers of plant functional traits and aboveground biomass in secondary tropical dry forest. Can. J. Forest Res. 2014, 44, 604–613. [Google Scholar] [CrossRef]
- Dybzinski, R.; Fargione, J.E.; Zak, D.R.; Fornara, D.; Tilman, D. Soil fertility increases with plant species diversity in a long-term biodiversity experiment. Oecologia 2008, 158, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Neri, A.V.; Schaefer, C.E.G.R.; Silva, A.F.; Souza, A.L.; Ferreira-Junior, W.G.; Meira-Neto, J.A.A. The influence of soils on the floristic composition and community structure of an area of Brazilian Cerrado vegetation. Edinb. J. Bot. 2012, 69, 1–27. [Google Scholar] [CrossRef]
- Fischer, C.; Leimer, S.; Roscher, C.; Ravenek, J.; de Kroon, H.; Kreutziger, Y.; Baade, J.; Beßler, H.; Eisenhauer, N.; Weigelt, A.; et al. Plant species richness and functional groups have different effects on soil water content in a decade-long grassland experiment. J. Ecol. 2019, 107, 127–141. [Google Scholar] [CrossRef] [Green Version]
- Nadeau, M.B.; Sullivan, T.P. Relationships between plant biodiversity and soil fertility in a mature tropical forest, Costa Rica. Int. J. For. Res. 2015, 2015, 732946. [Google Scholar] [CrossRef]
- Champion, H.G.; Seth, S.K. A Revised Survey of the Forest Types of India; Manager of Publications: Delhi, India, 1968. [Google Scholar]
- Haq, S.M.; Hamid, M.; Lone, F.A.; Singh, B. Himalayan hotspot with alien weeds: A Case study of biological spectrum, phenology, and diversity of weedy plants of high-altitude mountains in district kupwara of J&K Himalaya, India. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2021, 91, 139–152. [Google Scholar]
- Haq, S.M.; Malik, A.H.; Khuroo, A.A.; Rashid, I. Floristic composition and biological spectrum of Keran-a remote valley of northwestern Himalaya. AES 2019, 39, 372–379. [Google Scholar] [CrossRef]
- Haq, S.M.; Rashid, I.; Khuroo, A.A.; Malik, Z.A.; Malik, A.H. Anthropogenic disturbances alter community structure in the forests of Kashmir Himalaya. Trop. Ecol. 2019, 60, 6–15. [Google Scholar] [CrossRef]
- Gupta, P.K. Soil Plant Water and Fertilizer Analysis; Agrobios: Jodhpur, India, 2017. [Google Scholar]
- TerBraak, C.J.F.; Smilauer, P. CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (Version 4.5); Microcomputer Power: Ithaca, NY, USA, 2002; Available online: http://www.canoco.com (accessed on 17 May 2021).
- Leps, J.; Smilauer, P. Multivariate Analysis of Ecological Data Using CANOC; Cambridge University Press: Cambridge, UK, 2003. [Google Scholar]
- Altaf, A.; Haq, S.M.; Shabnum, N.; Jan, H.A. Comparative assessment of Phyto diversity in Tangmarg Forest division in Kashmir Himalaya, India. AES, 2021, in press.
- Sajad, S.; Haq, S.M.; Yaqoob, U.; Calixto, E.S.; Hassan, M. Tree composition and standing biomass in forests of the northern part of Kashmir Himalaya. Vegetos 2021, 34, 857–866. [Google Scholar] [CrossRef]
- Shannon, C.E. A mathematical theory of communication. Bell. Syst. Tech. J. 1948, 27, 379–423. [Google Scholar] [CrossRef] [Green Version]
- Simpson, E.H. Measurement of diversity. Nature 1949, 163, 688. [Google Scholar] [CrossRef]
- Pielou, E.C. An Introduction to Mathematical Ecology; Wiley-Interscience: New York, NY, USA, 1969. [Google Scholar]
- Shaheen, H.; Qureshi, R.A.; Shinwari, Z.K. Structural diversity, vegetation dynamics and anthropogenic impact on lesser Himalayan subtropical forests of Bagh district, Kashmir. Pak. J. Bot. 2011, 43, 1861–1866. [Google Scholar]
- Bokhari, T.Z.; Ahmed, M.; Siddiqui, M.F.; Khan, Z. Forest communities of Azad Kashmir, Pakistan. Fuuast J. Biol. 2013, 3, 137–145. [Google Scholar]
- Deka, J.; Tripathi, O.P.; Khan, M.L. High dominance of Shorea robusta Gaertn. in alluvial plain Kamrupsal forest of Assam, NE India. Int. J. Ecosyst. 2012, 2, 67–73. [Google Scholar] [CrossRef] [Green Version]
- Borah, N.; Athokpam, F.D.; Garkoti, S.C.; Das, A.K.; Hore, D.K. Structural and compositional variations in undisturbed and disturbed tropical forests of Bhuban hills in south Assam, India. Int. J. Biodiv. Sci. Ecosyst. Ser. Mgt. 2014, 10, 9–19. [Google Scholar] [CrossRef]
- Sharma, N.; Kant, S. Vegetation structure, floristic composition and species diversity of woody plant communities in sub-tropical KandiSiwaliks of Jammu, J & K, India. Int. J. Basic Appl. Sci. 2014, 3, 382. [Google Scholar]
- Sharma, J.; Raina, A.K. Quantitative analysis, distributional pattern and species diversity of woody plant species of Lamberi Forest Range, Rajouri, J&K, India. J. Appl. Nat. Sci. 2018, 10, 522–527. [Google Scholar]
- Haq, S.M.; Malik, A.H.; Khuroo, A.A.; Rashid, I. Contribution to the Flora of Keran Valley in Kashmir Himalaya, India. In Ethnobotany and Biodiversity Conservation; Indus Book Services Prvt. Ltd.: New Delhi, India, 2020; ISBN-10-938675657. [Google Scholar]
- Tariq, A.; Shu, H.; Siddiqui, S.; Imran, M.; Farhan, M. Monitoring Land use and land cover changes using geospatial techniques, a case study of Fateh Jang, Attock, Pakistan. Geogr. Environ. Sustain. 2021, 14, 41–52. [Google Scholar] [CrossRef]
- Haq, S.M.; Calixto, E.S.; Kumar, M. Assessing biodiversity and productivity over a small-scale gradient in the protected forests of Indian Western Himalayas. J. Sust. For. 2021, 40, 675–694. [Google Scholar] [CrossRef]
- Gairola, S.; Sharma, C.M.; Ghildiyal, S.K.; Suyal, S. Live tree biomass and carbon variation along an altitudinal gradient in moist temperate valley slopes of the Garhwal Himalaya (India). Curr. Sci. 2011, 100, 1862–1870. [Google Scholar]
- Ummara, U.; Bokhari, T.Z.; Siddiqui, M.F. Quantitative description of understory vegetation of Shogran valley, Pakistan. Fuuast. Biol. 2015, 5, 63–70. [Google Scholar]
- Tariq, A.; Riaz, I.; Ahmad, Z. Land surface temperature relation with normalized satellite indices for the estimation of spatio-temporal trends in temperature among various land use land cover classes of an arid Potohar region using Landsat data. Environ. Earth Sci. 2020, 79, 40. [Google Scholar] [CrossRef]
- Dar, J.A.; Sundarapandian, S. Patterns of plant diversity in seven temperate forest types of Western Himalaya, India. J. Asia Pac. Biodiv. 2016, 9, 280–292. [Google Scholar] [CrossRef] [Green Version]
- Tariq, A.; Shu, H. CA-Markov chain analysis of seasonal land surface temperature and land use land cover change using optical multi-temporal satellite data of Faisalabad, Pakistan. Remote Sens. 2020, 12, 3402. [Google Scholar] [CrossRef]
- Haq, S.M.; Calixto, E.S.; Rashid, I.; Srivastava, G.; Khuroo, A.A. Tree diversity, distribution and regeneration in major forest types along an extensive elevational gradient in Indian Himalaya: Implications for sustainable forest management. For. Ecol. Mgt. 2022, 506, 119968. [Google Scholar] [CrossRef]
- Ahmed, M.; Husain, T.; Sheikh, A.H.; Hussain, S.S.; Siddiqui, M.F. Phytosociology and structure of Himalayan forests from different climatic zones of Pakistan. Pak. J. Bot. 2006, 38, 361–383. [Google Scholar]
- Sreejith, K.A.; Chandrashekara, V.M.; Nirmesh, T.K.; Sreekumar, V.B. Tree species composition and distribution pattern in a Myristica swamp of Northern Kerala, India. Curr. World Environ. 2016, 11, 743–750. [Google Scholar] [CrossRef] [Green Version]
- Tariq, A.; Shu, H.; Saddiqui, S.; Mousa, B.G.; Munir, I.; Nasri, A.; Waqas, H.; Baqa, M.F.; Lu, L. Forest fire Monitoring using spatial-statistical and Geo-spatial analysis of factors determining Forest fire in Margalla Hills, Islamabad, Pakistan. Geomat. Nat. Hazards Risk 2021, 12, 1212–1233. [Google Scholar] [CrossRef]
- Siddiqui, M.F.; Moinuddin, A.; Nasrullah, K.; Khan, I.A. A quantitative description of moist temperate conifer forests of Himalayan region of Pakistan and Azad Kashmir. Int. J. Biol. Biotech. 2010, 7, 175–185. [Google Scholar]
- Tariq, A.; Shu, H.; Siddiqui, S.; Munir, I.; Sharifi, A.; Li, Q.; Lu, L. Spatio-temporal analysis of forest fire events in the Margalla Hills, Islamabad, Pakistan using socio-economic and environmental variable data with machine learning methods. J. For. Res. 2021, 13, 12. [Google Scholar] [CrossRef]
- Bano, S.; Khan, S.M.; Alam, J.; Alqarawi, A.A.; Abd_Allah, E.F.; Ahmad, Z.; Hashem, A. Eco-Floristic studies of native plants of the Beer Hills along the Indus River in the districts Haripur and Abbottabad, Pakistan. Saud. J. Biol. Sci. 2018, 25, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Shahid, M.; Joshi, S.P. Pytosociological assessement and distribution pattern of tree species in forest of doon valley Shivalik Hills of lower Himalaya. Trop. Pant Res. 2016, 3, 263–271. [Google Scholar]
- Tariq, A.; Shu, H.; Li, Q.; Altan, O.; Khan, M.R.; Baqa, M.F.; Lu, L. Quantitative analysis of forest fires in South- eastern Australia using SAR data. Remote Sens. 2021, 13, 2386. [Google Scholar] [CrossRef]
- Wang, J.; Chen, C.; Li, J.; Feng, Y.; Lu, Q. Different ecological processes determined the alpha and beta components of taxonomic, functional, and phylogenetic diversity for plant communities in dry land regions of Northwest China. Peer J. 2019, 6, 6220. [Google Scholar] [CrossRef]
- Tariq, A.; Shu, H.; Gagnon, A.S.; Li, Q.; Mumtaz, F.; Hysa, A.; Siddique, M.A.; Munir, I. Assessing burned areas in wildfires and prescribed fires with spectral indices and SAR images in the margalla hills of Pakistan. Forests 2021, 12, 1371. [Google Scholar] [CrossRef]
- Sainge, M.N.; Lyonga, N.M.; Mbatchou, G.P.; Kenfack, D.; Nchu, F.; Peterson, A.T. Vegetation, floristic composition and structure of a tropical montane forest in Cameroon. Bothalia. Afr. Biodiv. Conser. 2019, 49, 1–12. [Google Scholar] [CrossRef]
- Paudel, S.; Sah, J.P. Physico chemical characters of soil in tropical soil (Shorearobusta Gaertn.) forests in eastern Nepal. Himal. J. Sci. 2003, 1, 107–110. [Google Scholar]
- Bargali, K.; Manral, V.; Padalia, K.; Bargali, S.S.; Upadhyay, V.P. Effect of vegetation type and season on microbial biomass carbon in Central Himalayan forest soils, India. Catena 2018, 171, 125–135. [Google Scholar] [CrossRef]
- Bargali, S.S.; Joshi, M.; Bargali, K. Seasonal pattern of total soil respiration in age series of eucalypt plantation and mixed broad-leaved forest in Tarai belt of Kumaun Himalaya. Oecol. Mont. 1992, 2, 7–11. [Google Scholar]
- Bargali, S.S.; Singh, R.P.; Joshi, M. Changes in soil characteristics in eucalypt plantations replacing natural broad-leaved forests. J. Veg. Sci. 1993, 4, 25–28. [Google Scholar] [CrossRef]
- Hussain, S.; Lu, L.; Mubeen, M.; Nasim, W.; Karuppannan, S.; Fahad, S.; Tariq, A.; Mousa, B.G.; Mumtaz, F.; Aslam, M. Spatiotemporal variation in land use land cover in the response to local climate change using multispectral remote sensing data. Land 2022, 11, 595. [Google Scholar] [CrossRef]
- Khan, A.M.; Li, Q.; Saqib, Z.; Khan, N.; Habib, T.; Khalid, N.; Majeed, M.; Tariq, A. MaxEnt Modelling and impact of climate change on habitat suitability variations of economically important chilgoza pine (Pinus gerardiana Wall.) in South Asia. Forests 2022, 13, 715. [Google Scholar] [CrossRef]
- Bargali, S.S.; Padalia, K.; Bargali, K. Effects of tree fostering on soil health and microbial biomass under different land use systems in Central Himalaya. Land. Degrad. Dev. 2019, 30, 1984–1998. [Google Scholar] [CrossRef]
- Manral, V.; Bargali, K.; Bargali, S.S.; Shahi, C. Changes in soil biochemical properties following replacement of Banj oak forest with Chir pine in Central Himalaya, India. Ecol. Proc. 2020, 9, 30. [Google Scholar] [CrossRef]
- Padalia, K.; Bargali, S.S.; Bargali, K.; Manral, V. Soil microbial biomass phosphorus under different land use systems. Trop. Ecol. 2021, 63, 30–48. [Google Scholar] [CrossRef]
- Bargali, K.; Joshi, B.; Bargali, S.S.; Singh, S.P. Oaks and the biodiversity they sustain. Int. Oaks 2015, 26, 65–76. [Google Scholar]
- Hegazy, A.K.; El-Demerdash, M.A.; Hosni, H.A. Vegetation, species diversity and floristic relations along an altitudinal gradient in south-west Saudi Arabia. J. Arid. Environ. 1998, 38, 3–13. [Google Scholar] [CrossRef]
- Wang, Y.; Singh, M.P. Trust representation and aggregation in a distributed agent system. In Proceedings of the Twenty-First National Conference on Artificial Intelligence and the Eighteenth Innovative Applications of Artificial Intelligence Conference, Boston, MA, USA, 16–20 July 2006; AAAI: Menlo Park, CA, USA, 2006; Volume 6, pp. 1425–1430. [Google Scholar]
- Davies, R.G.; Barbosa, O.; Fuller, R.A.; Tratalos, J.; Burke, N.; Lewis, D.; Gaston, K.J. City-wide relationships between green spaces, urban land use and topography. Urban Ecosyst. 2008, 11, 269–287. [Google Scholar] [CrossRef]
- Khan, S.M.; Page, S.; Ahmad, H.A.; Shaheen, H.; Harper, D.M. Vegetation dynamics in the Western Himalayas, diversity indices and climate change. Sci. Tech. Dev. 2012, 31, 232–243. [Google Scholar]
- Tariq, A.; Siddiqui, S.; Sharifi, A.; Hassan, S.; Ahmad, I. Impact of spatio-temporal land surface temperature on cropping pattern and land use and land cover changes using satellite imagery, Hafizabad District, Punjab, Province of Pakistan. Arab. J. Geosci. 2022, 15, 1045. [Google Scholar] [CrossRef]
- Haq, S.M.; Calixto, E.S.; Malik, A.A.; Rashid, I.A.H.; Kumar, M.; Khuroo, A. Anthropogenic pressure and tree carbon loss in the temperate forests of Kashmir Himalaya. Bot. Lett. 2022, 12, 1–13. [Google Scholar]
- Khan, N.; Shaukat, S.S.; Ahmed, M.; Siddiqui, M.F. Vegetation-environment relationships in the forests of Chitral district Hindukush range. Pak. J. For. Res. 2013, 24, 205–216. [Google Scholar] [CrossRef]
- Tariq, A.; Mumtaz, F.; Zeng, X.; Baloch, M.Y.J.; Moazzam, M.F.U. Spatio-temporal variation of seasonal heat islands mapping of Pakistan during 2000–2019, using day-time and night-time land surface temperatures MODIS and meteorological stations data. Remote Sens. Appl. Soc. Environ. 2022, 27, 100779. [Google Scholar] [CrossRef]
- Hussain, M.; Khan, S.M.; ABD_ALLAH, E.F.; UlHaq, Z.; Alshahrani, T.S. Assessment of plant communities and identification of indicator species of an ecotonal forest zone at Durand line, district Kurram, Pakistan. Appl. Ecol. Environ. Res. 2019, 17, 6375–6396. [Google Scholar] [CrossRef]
- Majeed, M.; Tariq, A.; Haq, S.M.; Waheed, M.; Anwar, M.M.; Li, Q.; Aslam, M.; Abbasi, S.; Mousa, B.G.; Jamil, A. A detailed ecological exploration of the distribution patterns of wild Poaceae from the Jhelum District (Punjab), Pakistan. Sustainability 2022, 14, 3786. [Google Scholar] [CrossRef]
- Rahman, I.U.; Hart, R.E.; Ijaz, F.; Afzal, A.; Iqbal, Z.; Calixto, E.S.; Abd_Allah, E.F.; Alqarawi, A.A.; Hashem, A.; Al-Arjani, A.B.F.; et al. Environmental variables drive plant species composition and distribution in the moist temperate forests of Northwestern Himalaya, Pakistan. PLoS ONE 2022, 17, e0260687. [Google Scholar] [CrossRef]
- Malik, Z.A.; Haq, S.M.; Bussmann, R.W.; Bhat, J.A.; Bhatt, A.B. Altitudinal variation in soil properties with reference to forest structure and composition in Western Himalaya. Indian For. 2021, 147, 288–301. [Google Scholar] [CrossRef]
| Family | Botanical Name | Abbreviation | Life Span | Growth Form | ACFT | BLFT | OKFT | PWFT | SRFT |
|---|---|---|---|---|---|---|---|---|---|
| Acanthaceae | Strobilanthes attenuate Nees (SMH 186) | Str_att | Perennial | Herb | 0 | 0 | 6.03 | 0 | 0 |
| Strobilanthes wallichii Nees (SMH 188) | Str_wal | Perennial | Herb | 0 | 0 | 10.61 | 5.98 | 4.32 | |
| Adoxaceae | Viburnum grandiflorum Wall. ex DC. (SMH 492) | Vib_gra | Perennial | Shrub | 77.89 | 30.62 | 53.51 | 83.57 | 0 |
| Amaranthaceae | Achyranthes bidentata Blume (SMH 264) | Ach_bid | Perennial | Herb | 0 | 0 | 22.42 | 18.83 | 33.14 |
| Asteraceae | Arctium lappa L. (SMH 185) | Arc_lap | Biennial | Herb | 6.39 | 6.27 | 2.98 | 0 | 0 |
| Artemisia vulgaris L. (SMH 190) | Art_vul | Perennial | Herb | 4.85 | 4.47 | 0 | 0 | 0 | |
| Carpesiumabrotanoides L. (SMH 194) | Car_abr | Perennial | Herb | 4.43 | 3.99 | 2.98 | 3.59 | 4.85 | |
| Erigeron canadensis L. (SMH 192) | Eri_can | Annual | Herb | 5.17 | 5.54 | 0 | 4.54 | 3.53 | |
| Asparagaceae | Asparagus filicinus Buch.-Ham. ex D.Don (SMH 210) | Asp_fil | Perennial | Herb | 0 | 0 | 2.99 | 0 | 0 |
| Asparagus officinalis L. (SMH 211) | Asp_off | Perennial | Herb | 0 | 0 | 0 | 4.37 | 3.53 | |
| Polygonatum biflorum (Walter) Elliott (SMH 442) | Pol_bif | Perennial | Herb | 0 | 0 | 5.97 | 4.27 | 5.15 | |
| Polygonatum verticillatum (L.) All. (SMH 445) | Pol_ver | Perennial | Herb | 0 | 0 | 0 | 2.35 | 3.21 | |
| Aspleniaceae | Asplenium ofeliae Salgado, A.E. (SMH 435) | Asp_ofe | Perennial | Herb | 3.75 | 3.27 | 8.05 | 2.19 | 2.801 |
| Apiaceae | Daucus carota L. (SMH 198) | Dau_car | Biennial | Herb | 5.78 | 5.62 | 0 | 0 | 0 |
| Selinum wallichianum (DC.) Raizada& H.O. Saxena (SMH 212) | Sel-wal | Perennial | Herb | 2.25 | 2.14 | 0 | 0 | 0 | |
| Araliaceae | Hedera nepalensis K.Koch (SMH 216) | Hed_nep | Perennial | Climber | 11.24 | 11.71 | 13.12 | 14.09 | 16.51 |
| Balsaminaceae | Impatiens glandulifera Royle (SMH 305) | Imp_gla | Annual | Herb | 14.31 | 13.05 | 16.06 | 9.15 | 10.91 |
| Berberidaceae | Berberis lyceum Royle (SMH 306) | Ber_lyc | Perennial | Shrub | 59.07 | 76.29 | 0 | 0 | 0 |
| Caprifoliaceae | Lonicera webbiana Wall. ex DC. (SMH 326) | Lon_web | Perennial | Shrub | 69.29 | 0 | 0 | 0 | 0 |
| Campanulaceae | Asyneuma thomsonii (C.B.Clarke) Bornm. (SMH 328) | Asy_tho | Perennial | Herb | 0 | 0 | 0 | 2.35 | 2.36 |
| Cannabaceae | Celtis australis L. (SMH 440) | Cel_aus | Perennial | Tree | 33.71 | 42.68 | 15.28 | 45.75 | 46.75 |
| Dioscoreaceae | Dioscorea deltoidea Wall. ex Griseb (SMH 441) | Dio_del | Perennial | Climber | 10.91 | 10.46 | 9.86 | 5.21 | 6.16 |
| Dryopteridaceae | Dryopteris barbigera (T.Moore ex Hook.) Kuntze SMH (443) | Dry_bar | Perennial | Herb | 6.08 | 6.31 | 6.87 | 12.19 | 12.26 |
| Fabaceae | Robiniapseudoacacia L. (SMH 219) | Rob_pse | Perennial | Tree | 87.89 | 14.91 | 0 | 0 | 0 |
| Desmodium elegans DC. (SMH 449) | Des_ele | Perennial | Shrub | 0 | 0 | 0 | 0 | 63.62 | |
| Indigofera hebepetala Baker (SMH 243) | Ind_heb | Perennial | Shrub | 0 | 0 | 0 | 0 | 97.27 | |
| Trifolium pratense L. (SMH 507) | Tri_pra | Perennial | Herb | 14.73 | 16.59 | 0 | 0 | 0 | |
| Trifolium repens L. (SMH 508) | Tri_rep | Perennial | Herb | 11.66 | 10.64 | 5.62 | 0 | 0 | |
| Fagaceae | Quercus robur L. (SMH 516) | Que_rob | Perennial | Tree | 16.46 | 0 | 200.97 | 5.92 | 0 |
| Geraniaceae | Geranium nepalense Sweet (SMH 517) | Ger_nep | Perennial | Herb | 9.97 | 9.43 | 10.49 | 4.62 | 5.48 |
| Geranium pratense L. (SMH 356) | Ger_pra | Perennial | Herb | 3.53 | 4.34 | 2.98 | 4.17 | 3.07 | |
| Geranium wallichianum D.Don ex Sweet (SMH 357) | Ger_wal | Perennial | Herb | 0 | 0 | 5.01 | 1.69 | 2.29 | |
| Hamamelidaceae | Parrotiopsis jacquemontiana (Decne.) Rehder (SMH 138) | Par_jac | Perennial | Shrub | 0 | 65.93 | 116.73 | 170.91 | 154.44 |
| Hypericaceae | Hypericum perforatum L. (SMH 175) | Hyp_per | Perennial | Herb | 2.75 | 2.63 | 0 | 0 | 0 |
| Iridaceae | Iris hookeriana Foster (SMH 382) | Iri_hoo | Perennial | Herb | 3.51 | 3.27 | 9.57 | 0 | 0 |
| Jugalandaceae | Juglans regia L. (SMH 326) | Jug_reg | Perennial | Tree | 25.24 | 10.15 | 0 | 0 | 0 |
| Lamiaceae | Perilla frutescens (L.) Britton (SMH 320) | Per_fru | Annual | Herb | 7.71 | 7.67 | 0 | 0 | 0 |
| Salvia moorcroftiana Wall. ex Benth. (SMH 321) | Sal_moo | Perennial | Herb | 0 | 0 | 2.23 | 2.35 | 3.21 | |
| Prunella vulgaris L. (SMH 322) | Pru_vul | Perennial | Herb | 0 | 0 | 2.02 | 0 | 0 | |
| Moraceae | Morus alba L. (SMH 334) | Mor_alb | Perennial | Tree | 18.01 | 21.94 | 25.46 | 5.99 | 0 |
| Morus nigra L. (SMH 335) | Mor_nig | Perennial | Tree | 26.44 | 21.95 | 6.95 | 0 | 0 | |
| Orchidaceae | Cypripedumcordigerum D.Don (SMH 328) | Cyp_cor | Perennial | Herb | 0 | 0 | 3.87 | 3.2 | 2.81 |
| Oxalidaceae | Oxalis acetosella L. (SMH 329) | Oxa_ace | Perennial | Herb | 0 | 0 | 0 | 6.55 | 10.06 |
| Pinaceae | Pinus wallichiana A.B.Jacks. (SMH 330) | Pin_wal | Perennial | Tree | 0 | 0 | 0 | 184.44 | 0 |
| Poaceae | Cynodon dactylon (L.) Pers. (SMH 331) | Cyn_dac | Perennial | Herb | 0 | 0 | 0 | 38.96 | 21.17 |
| Oplismenus burmannii f. cristata (J. Presl) Hier. ex Peter (SMH 332) | Opl_bur | Annual | Herb | 54.19 | 52.36 | 30.4 | 89.07 | 83.31 | |
| Poa bulbosa L. (SMH 339) | Poa_bul | Annual | Herb | 23.28 | 22.66 | 14.33 | 0 | 0 | |
| Sorghum halepense (L.) Pers. (SMH 480) | Sor_hal | Perennial | Herb | 0 | 0 | 6.86 | 0 | 0 | |
| Stipa sibirica (L.) Lam. (SMH 481) | Sti_sib | Perennial | Herb | 35.48 | 39.14 | 59.11 | 8.3 | 10.02 | |
| Polygonaceae | Fagopyrum esculentum Moench (SMH 483) | Fag_esc | Annual | Herb | 5.81 | 6.99 | 3.39 | 0 | 0 |
| Polygonum amplexicaule D.Don (SMH 482) | Pol_amp | Perennial | Herb | 1.74 | 1.64 | 3.19 | 0 | 0 | |
| Polygonum hydropiper L. (SMH 489) | Pol_hyd | Annual | Herb | 3.51 | 3.27 | 0 | 0 | 0 | |
| Plantaginaceae | Digitalis purpurea L. (SMH 486) | Dig_pur | Perennial | Herb | 6.08 | 5.57 | 0 | 0 | 0 |
| Plantago major L. (SMH 479) | Pla_maj | Perennial | Herb | 5.35 | 6.26 | 3.39 | 4.17 | 4.99 | |
| Pteridaceae | Pteris cretica L. (SMH 460) | Pte_cre | Perennial | Herb | 4.19 | 3.84 | 0 | 3.82 | 4.18 |
| Rosaceae | Crataegus monogyna Jacq. (SMH 462) | Cra_mon | Perennial | Tree | 0 | 4.7 | 6.3 | 25.1 | 0 |
| Prunus persica (L.) Batsch (SMH 463) | Pru_per | Perennial | Tree | 0 | 13.48 | 0 | 0 | 0 | |
| Prunus armeniaca L. (SMH 340) | Pru_arm | Perennial | Tree | 11.65 | 3.12 | 0 | 6.35 | 14.1 | |
| Prunus avium (L.) L. (SMH 370) | Pru_avi | Perennial | Tree | 21.26 | 16.72 | 0 | 0 | 0 | |
| Prunus cerasus L. (SMH 371) | Pru_cer | Perennial | Tree | 5.18 | 13.71 | 0 | 0 | 0 | |
| Rosa webbiana Wall. ex Royle (SMH 204) | Ros_web | Perennial | Shrub | 0 | 52.12 | 40.1 | 0 | 0 | |
| Rubus ulmifolius Schott (SMH 350) | Rub_ulm | Perennial | Shrub | 93.73 | 0 | 0 | 0 | 0 | |
| Sorbaria tomentosa (Lindl.) Rehder (SMH 385) | Sor_tom | Perennial | Shrub | 0 | 0 | 0 | 0 | 69.24 | |
| Fragaria nubicola (Lindl. ex Hook.f.) Lacaita(SMH 450) | Fra_nub | Perennial | Herb | 6.17 | 5.74 | 3.48 | 6.25 | 5.83 | |
| Geum aleppicum Jacq. (SMH 472) | Geu_ale | Perennial | Herb | 2.64 | 3.63 | 9.5 | 4.88 | 4.18 | |
| Geum urbanum L. (SMH 473) | Geu_urb | Perennial | Herb | 10.21 | 9.77 | 6.22 | 4.63 | 5.12 | |
| Prunus tomentosa Thunb. (SMH 474) | Pru_tom | Perennial | Shrub | 0 | 75.03 | 89.63 | 45.51 | 0 | |
| Ranunculaceae | Delphinium roylei Munz (SMH 475) | Del_roy | Perennial | Herb | 0 | 0 | 0 | 2.42 | 4.83 |
| Salicaceae | Populus alba L. (SMH 476) | Pop_alb | Perennial | Tree | 0 | 47.26 | 0 | 0 | 0 |
| Salix alba L. (SMH 477) | Sal_alb | Perennial | Tree | 4.11 | 30.31 | 0 | 0 | 0 | |
| Sapindaceae | Acer caesium Wall. ex Brandis (SMH 478) | Ace_cae | Perennial | Tree | 3.36 | 5.88 | 0 | 6.26 | 0 |
| Aesculus indica (Wall. ex Cambess.) Hook. (SMH 110) | Aes_ind | Perennial | Tree | 3.76 | 15.33 | 30.55 | 6.57 | 0 | |
| Simaraubaceae | Ailanthus altissima (Mill.) Swingle (SMH 135) | Ail_alt | Perennial | Tree | 37.04 | 19.41 | 14.46 | 5.9 | 46.84 |
| Scrophulariaceae | Verbascum thapsus L. (SMH 511) | Ver_tha | Biennial | Herb | 3.88 | 3.47 | 2.98 | 4.44 | 3.53 |
| Ulmaceae | Ulmus wallichiana Planch. (SMH 525) | Ulm_wal | Perennial | Tree | 5.82 | 18.37 | 0 | 7.68 | 37.86 |
| Violaceae | Viola odorata L. (SMH 512) | Vio_odo | Perennial | Herb | 8.30 | 8.11 | 7.26 | 20.25 | 17.06 |
| Forest Types | ACFT | BLFT | OKFT | PNFT | SRFT |
|---|---|---|---|---|---|
| Species Richness | 51 | 55 | 44 | 46 | 38 |
| Dominance | 0.041 | 0.033 | 0.066 | 0.075 | 0.076 |
| Shannon | 3.524 | 3.664 | 3.26 | 3.14 | 3.092 |
| Simpson | 0.958 | 0.966 | 0.933 | 0.924 | 0.923 |
| Evenness | 0.664 | 0.709 | 0.592 | 0.502 | 0.579 |
| Fisher Alpha | 16.33 | 16.63 | 14.89 | 16.62 | 15.03 |
| pH | 6.56 | 6.5 | 5.9 | 5.51 | 6.66 |
| Electrical Conductivity (µS/cm) | 384 | 370 | 660 | 610 | 396 |
| Organic Carbon (%) | 4.14 | 5.18 | 5.44 | 4.5 | 5.04 |
| Available Nitrogen(kg/ha) | 0.08 | 0.13 | 0.16 | 0.12 | 0.13 |
| Phosphorus (μg/g) | 18.6 | 8.2 | 15.2 | 8.2 | 8.1 |
| Potassium (μg/g) | 147 | 129 | 463 | 230 | 339 |
| Calcium (μg/g) | 4.05 | 3.7 | 3.6 | 4.5 | 4.1 |
| Salinity (ppm) | 23.7 | 24.6 | 83.1 | 21.3 | 13.3 |
| Density (mean ± SD; trees/ha−1) | 850 ± 204.61 | 1057.5 ± 367.28 | 640 ± 140.95 | 707.5 ± 148.18 | 1197.5 ± 199.56 |
| Basal Area (mean ± SD; m2ha−1) | 46.82 ± 14.73 | 58.63 ± 21.57 | 41.41 ± 3.81 | 74.49 ± 12.09 | 15.40 ± 6.20 |
| Statistic | Axis 1 | Axis 2 | Axis 3 | Axis 4 |
|---|---|---|---|---|
| Eigenvalues | 0.81 | 0.42 | 0.28 | 0.04 |
| Accumulative explained variation | 17.6 | 26.8 | 32.9 | 33.9 |
| Gradient length | 6.84 | 3.04 | 2.44 | 2.07 |
| Total inertia | 4.603 | |||
| Statistic | Axis 1 | Axis 2 | Axis 3 | Axis 4 |
|---|---|---|---|---|
| Eigenvalues | 0.736 | 0.446 | 0.388 | 0.347 |
| Explained variation | 17.9 | 28.7 | 38.2 | 46.6 |
| Pseudo-canonical correlation | 0.984 | 0.958 | 0.980 | 0.962 |
| Explained fitted variation | 31.5 | 50.6 | 67.2 | 82.7 |
| Total inertia | 4.109 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haq, S.M.; Tariq, A.; Li, Q.; Yaqoob, U.; Majeed, M.; Hassan, M.; Fatima, S.; Kumar, M.; Bussmann, R.W.; Moazzam, M.F.U.; et al. Influence of Edaphic Properties in Determining Forest Community Patterns of the Zabarwan Mountain Range in the Kashmir Himalayas. Forests 2022, 13, 1214. https://doi.org/10.3390/f13081214
Haq SM, Tariq A, Li Q, Yaqoob U, Majeed M, Hassan M, Fatima S, Kumar M, Bussmann RW, Moazzam MFU, et al. Influence of Edaphic Properties in Determining Forest Community Patterns of the Zabarwan Mountain Range in the Kashmir Himalayas. Forests. 2022; 13(8):1214. https://doi.org/10.3390/f13081214
Chicago/Turabian StyleHaq, Shiekh Marifatul, Aqil Tariq, Qingting Li, Umer Yaqoob, Muhammad Majeed, Musheerul Hassan, Sammer Fatima, Manoj Kumar, Rainer W. Bussmann, Muhammad Farhan Ul Moazzam, and et al. 2022. "Influence of Edaphic Properties in Determining Forest Community Patterns of the Zabarwan Mountain Range in the Kashmir Himalayas" Forests 13, no. 8: 1214. https://doi.org/10.3390/f13081214
APA StyleHaq, S. M., Tariq, A., Li, Q., Yaqoob, U., Majeed, M., Hassan, M., Fatima, S., Kumar, M., Bussmann, R. W., Moazzam, M. F. U., & Aslam, M. (2022). Influence of Edaphic Properties in Determining Forest Community Patterns of the Zabarwan Mountain Range in the Kashmir Himalayas. Forests, 13(8), 1214. https://doi.org/10.3390/f13081214











