The Impact of Drought Stress on the Height Growth of Young Norway Spruce Full-Sib and Half-Sib Clonal Trials in Sweden and Finland
Abstract
1. Introduction
2. Materials and Methods
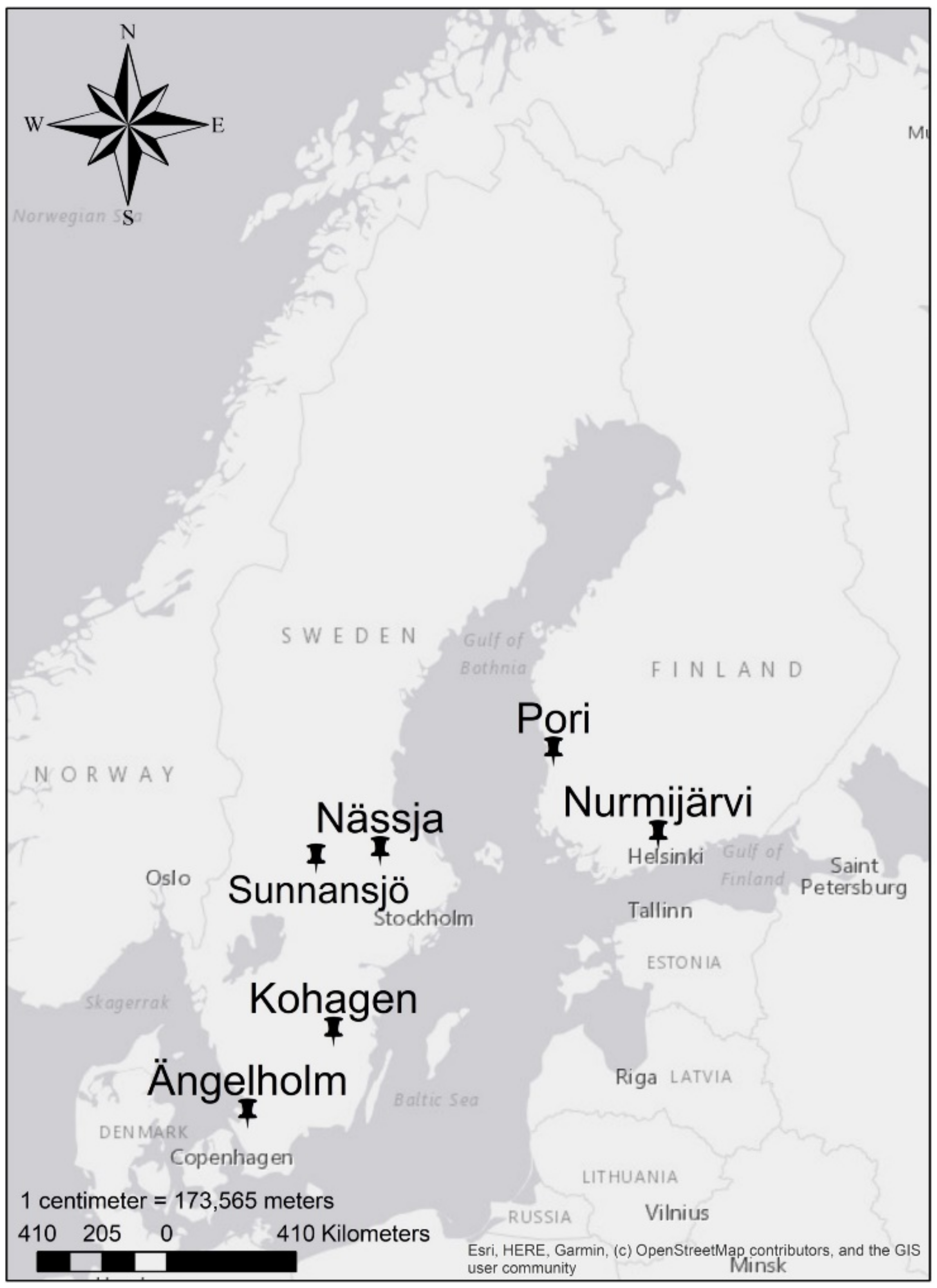
2.1. Field Data
2.1.1. Full-Sib Clonal Trials
2.1.2. Half-Sib Clonal Trials
2.2. Measurements
2.3. Climate Data and the SPEI
2.4. Statistical Data Analysis
2.4.1. Single-Site Analysis
2.4.2. Multi-Site Analysis
3. Results
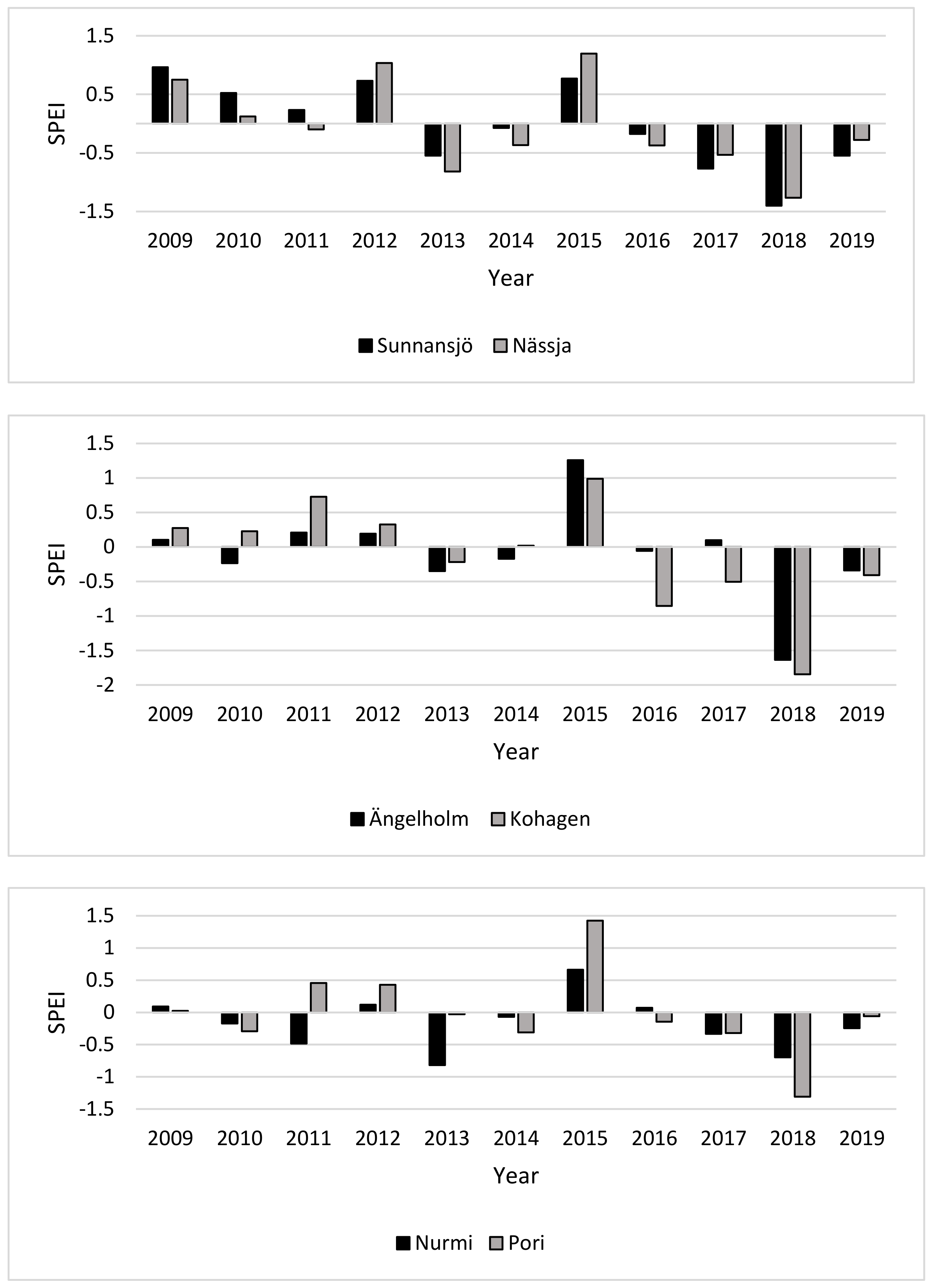
3.1. Drought Index (SPEI)
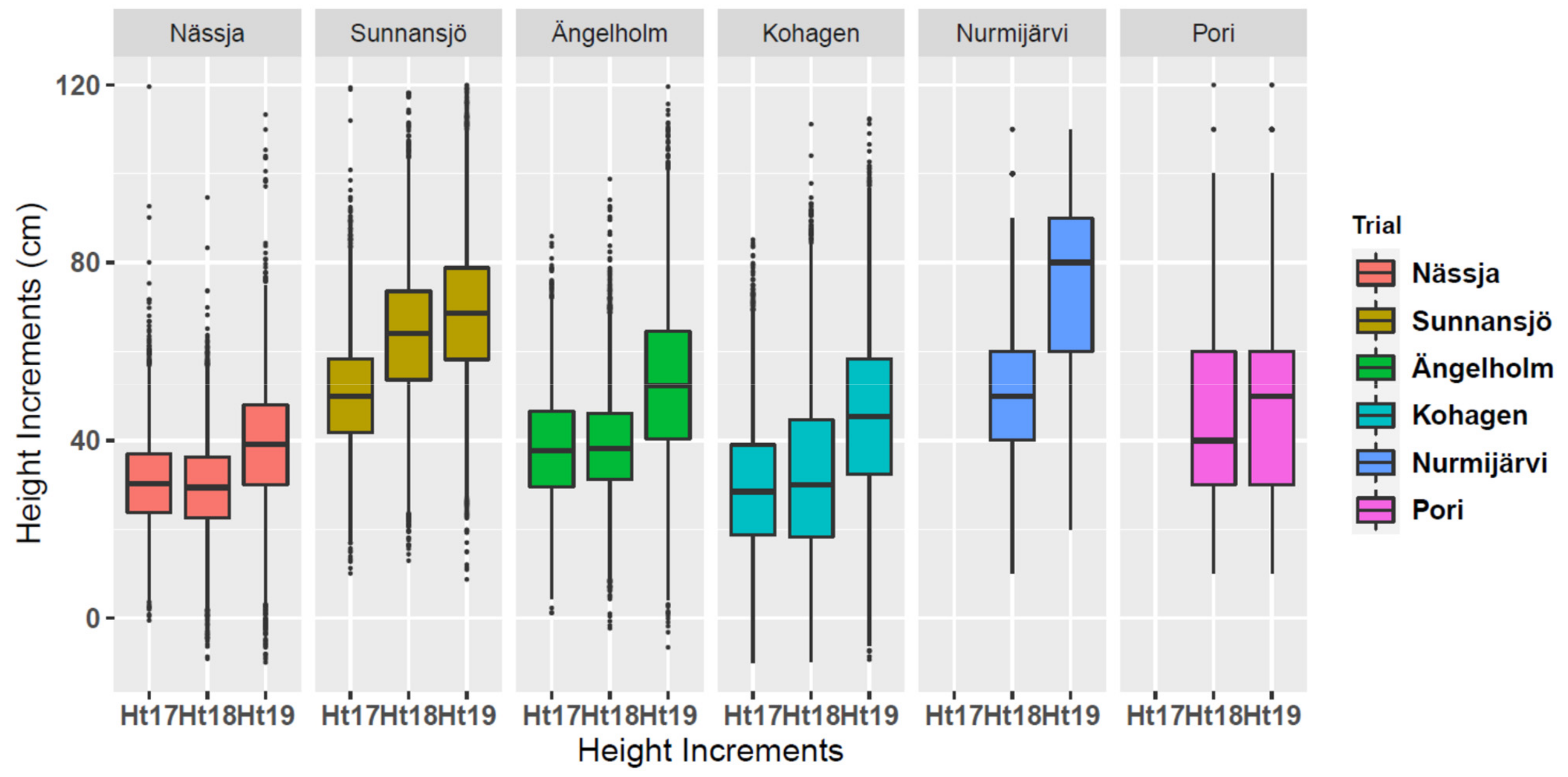
3.2. Annual Height Increments by Trials
3.3. Genetic Variance Components and Heritability Estimates
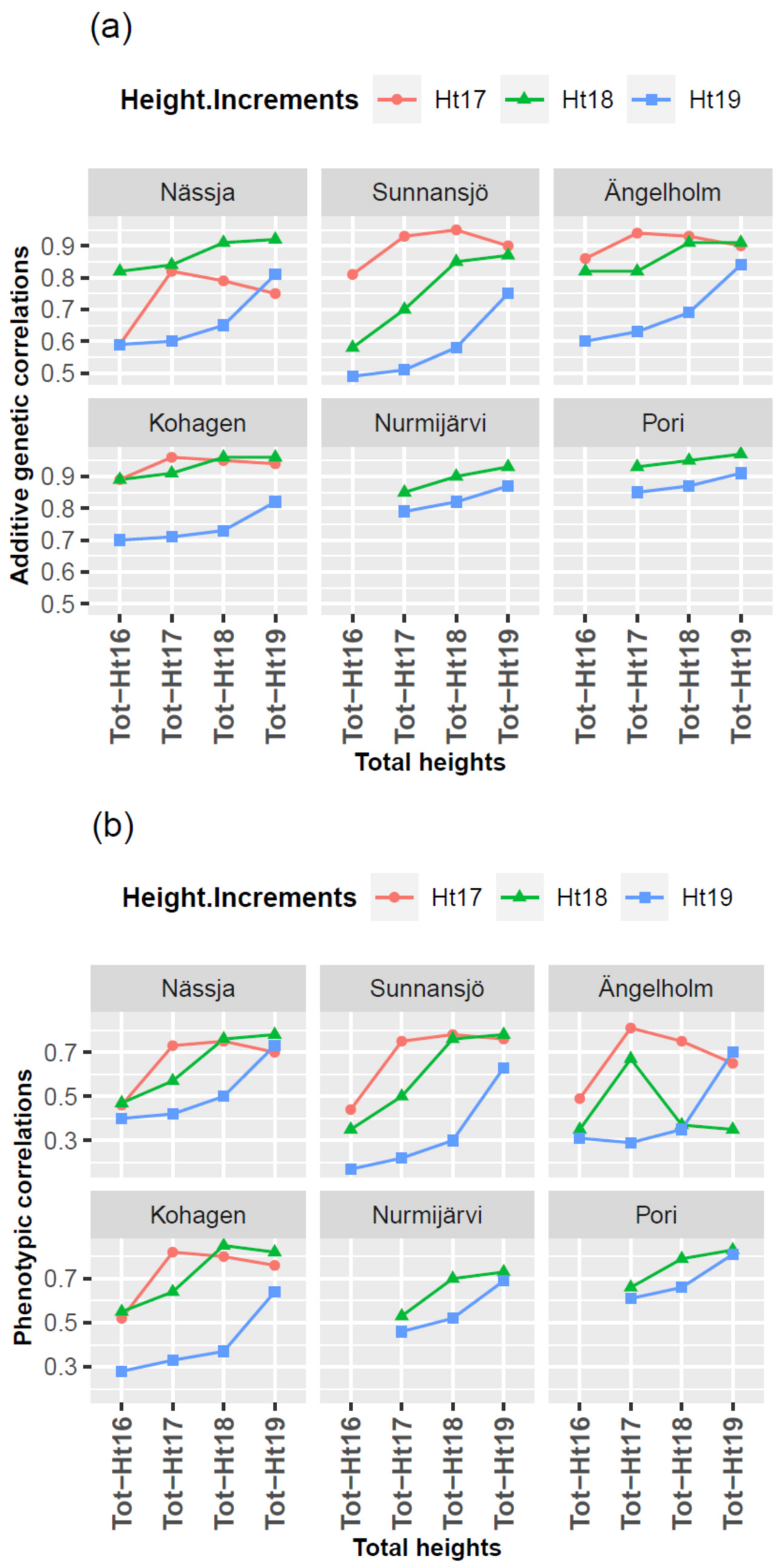
3.4. Genetic and Phenotypic Correlations
3.5. G × E and Type-B Genetic Correlations
4. Discussion
4.1. Drought Index and Annual Height Increments
4.2. Genetic Variance Components and Heritability Estimates
4.3. G × E and Type-B Genetic Correlations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ryan, M.G. Tree responses to drought. Tree Physiol. 2011, 31, 237–239. [Google Scholar] [CrossRef]
- Lindner, M.; Maroschek, M.; Netherer, S.; Kremer, A.; Barbati, A.; Garcia-Gonzalo, J.; Seidl, R.; Delzon, S.; Corona, P.; Kolström, M.; et al. Climate change impacts, adaptive capacity, and vulnerability of European forest ecosystems. For. Ecol. Manag. 2010, 259, 698–709. [Google Scholar] [CrossRef]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.T.; et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Huang, M.; Wang, X.; Keenan, T.F.; Piao, S. Drought timing influences the legacy of tree growth recovery. Glob. Chang. Biol. 2018, 24, 3546–3559. [Google Scholar] [CrossRef] [PubMed]
- Bréda, N.; Huc, R.; Granier, A.; Dreyer, E. Temperate forest trees and stands under severe drought: A review of ecophysiological responses, adaptation processes and long-term consequences. Ann. For. Sci. 2006, 63, 625–644. [Google Scholar] [CrossRef]
- Moran, E.; Lauder, J.; Musser, C.; Stathos, A.; Shu, M. The genetics of drought tolerance in conifers. New Phytol. 2017, 216, 1034–1048. [Google Scholar] [CrossRef] [PubMed]
- Trujillo-Moya, C.; George, J.-P.; Fluch, S.; Geburek, T.; Grabner, M.; Karanitsch-Ackerl, S.; Konrad, H.; Mayer, K.; Sehr, E.M.; Wischnitzki, E.; et al. Drought sensitivity of Norway spruce at the species’ warmest fringe: Quantitative and molecular analysis reveals high genetic variation among and within provenances. G3 Genes Genomes Genet. 2018, 8, 1225–1245. [Google Scholar] [CrossRef]
- Peña-Gallardo, M.; Vicente-Serrano, S.M.; Camarero, J.J.; Gazol, A.; Sánchez-Salguero, R.; Domínguez-Castro, F.; El Kenawy, A.; Beguería-Portugés, S.; Gutiérrez, E.; De Luis, M. Drought sensitiveness on forest growth in peninsular Spain and the Balearic Islands. Forests 2018, 9, 524. [Google Scholar] [CrossRef]
- Hannrup, B.; Cahalan, C.; Chantre, G.; Grabner, M.; Karlsson, B.; Bayon, I.L.; Jones, G.L.; Müller, U.; Pereira, H.; Rodrigues, J.C. Genetic parameters of growth and wood quality traits in Picea abies. Scand. J. For. Res. 2004, 19, 14–29. [Google Scholar] [CrossRef]
- Cermák, P.; Kolář, T.; Žid, T.; Trnka, M.; Rybníček, M. Norway spruce responses to drought forcing in areas affected by forest decline. For. Syst. 2019, 28, 4. [Google Scholar]
- Van der Maaten-Theunissen, M.; Kahle, H.-P.; van der Maaten, E. Drought sensitivity of Norway spruce is higher than that of silver fir along an altitudinal gradient in southwestern Germany. Ann. For. Sci. 2013, 70, 185–193. [Google Scholar] [CrossRef]
- Rosner, S.; Gierlinger, N.; Klepsch, M.; Karlsson, B.; Evans, R.; Lundqvist, S.-O.; Světlík, J.; Børja, I.; Dalsgaard, L.; Andreassen, K. Hydraulic and mechanical dysfunction of Norway spruce sapwood due to extreme summer drought in Scandinavia. For. Ecol. Manag. 2018, 409, 527–540. [Google Scholar] [CrossRef]
- Rybníček, M.; Čermák, P.; Žid, T.; Kolář, T. Radial growth and health condition of Norway spruce (Picea abies (L.) Karst.) stands in relation to climate (Silesian Beskids, Czech Republic). Geochronometria 2010, 36, 9–16. [Google Scholar] [CrossRef]
- Nilsson, U.; Örlander, G. Effects of regeneration methods on drought damage to newly planted Norway spruce seedlings. Can. J. For. Res. 1995, 25, 790–802. [Google Scholar] [CrossRef]
- Solberg, S. Summer drought: A driver for crown condition and mortality of Norway spruce in Norway. For. Pathol. 2004, 34, 93–104. [Google Scholar] [CrossRef]
- Mäkinen, H.; Nöjd, P.; Mielikäinen, K. Climatic signal in annual growth variation in damaged and healthy stands of Norway spruce [Picea abies (L.) Karst.] in southern Finland. Trees 2001, 15, 177–185. [Google Scholar] [CrossRef]
- Sonesson, J.; Eriksson, G. Genetic variation in drought tolerance in Picea abies seedlings and its relationship to growth in controlled and field environments. Scand. J. For. Res. 2003, 18, 7–18. [Google Scholar] [CrossRef]
- Savolainen, O.A.; Hurme, P.K. Conifers from the cold. In Environmental Stress, Adaptation and Evolution; Springer: Berlin/Heidelberg, Germany, 1997; pp. 43–62. [Google Scholar]
- Zhou, L.; Chen, Z.; Lundqvist, S.-O.; Olsson, L.; Grahn, T.; Karlsson, B.; Wu, H.X.; García-Gil, M.R. Genetic analysis of wood quality traits in Norway spruce open-pollinated progenies and their parent plus trees at clonal archives and the evaluation of phenotypic selection of plus trees. Can. J. For. Res. 2019, 49, 810–818. [Google Scholar] [CrossRef]
- Chen, Z.-Q.; Gil, M.R.G.; Karlsson, B.; Lundqvist, S.-O.; Olsson, L.; Wu, H.X. Inheritance of growth and solid wood quality traits in a large Norway spruce population tested at two locations in southern Sweden. Tree Genet. Genomes 2014, 10, 1291–1303. [Google Scholar] [CrossRef]
- Yang, M.; Yan, D.; Yu, Y.; Yang, Z. SPEI-based spatiotemporal analysis of drought in Haihe River Basin from 1961 to 2010. Adv. Meteorol. 2016, 2016, 7658015. [Google Scholar] [CrossRef]
- Vicente-Serrano, S.M.; Beguería, S.; López-Moreno, J.I. A multiscalar drought index sensitive to global warming: The standardized precipitation evapotranspiration index. J. Clim. 2010, 23, 1696–1718. [Google Scholar] [CrossRef]
- Bhuyan, U.; Zang, C.; Menzel, A. Different responses of multispecies tree ring growth to various drought indices across Europe. Dendrochronologia 2017, 44, 1–8. [Google Scholar] [CrossRef]
- Li, B.; Zhou, W.; Zhao, Y.; Ju, Q.; Yu, Z.; Liang, Z.; Acharya, K. Using the SPEI to assess recent climate change in the Yarlung Zangbo River Basin, South Tibet. Water 2015, 7, 5474–5486. [Google Scholar] [CrossRef]
- Schuldt, B.; Buras, A.; Arend, M.; Vitasse, Y.; Beierkuhnlein, C.; Damm, A.; Gharun, M.; Grams, T.E.; Hauck, M.; Hajek, P.; et al. A first assessment of the impact of the extreme 2018 summer drought on Central European forests. Basic Appl. Ecol. 2020, 45, 86–103. [Google Scholar] [CrossRef]
- Wulff, S.; Roberge, C. Nationell Riktad Skogssadeinventering (NRS)—Inventering av Granbarkborreangrepp i Götaland och Svealand 2020; Institutionen för Skoglig Resurshushållning: Umeå, Sweden, 2021. [Google Scholar]
- Perttu, K.; Morén, A.-S. Regional Temperature and Radiation Indices and Their Adjustment to Horizontal and Inclined Forest Land; Faculty of Forestry, Swedish University of Agricultural Sciences: Uppsala, Sweden, 1994. [Google Scholar]
- Bärring, L.; Berlin, M.; Andersson Gull, B. Tailored climate indices for climate-proofing operational forestry applications in Sweden and Finland. Int. J. Climatol. 2017, 37, 123–142. [Google Scholar] [CrossRef]
- Jin, X.; Qiang, H.; Zhao, L.; Jiang, S.; Cui, N.; Cao, Y.; Feng, Y. SPEI-based analysis of spatio-temporal variation characteristics for annual and seasonal drought in the Zoige Wetland, Southwest China from 1961 to 2016. Theor. Appl. Climatol. 2020, 139, 711–725. [Google Scholar] [CrossRef]
- Team, R.C. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016; Available online: www.R-project.org/ (accessed on 19 October 2019).
- Vicente-Serrano, S.M.; Camarero, J.J.; Azorin-Molina, C. Diverse responses of forest growth to drought time-scales in the N orthern H emisphere. Glob. Ecol. Biogeogr. 2014, 23, 1019–1030. [Google Scholar] [CrossRef]
- Dutkowski, G.W.; Silva, J.C.E.; Gilmour, A.R.; Lopez, G.A. Spatial analysis methods for forest genetic trials. Can. J. For. Res. 2002, 32, 2201–2214. [Google Scholar] [CrossRef]
- Berlin, M.; Jansson, G.; Högberg, K.-A.; Helmersson, A. Analysis of non-additive genetic effects in Norway spruce. Tree Genet. Genomes 2019, 15, 42. [Google Scholar] [CrossRef]
- Burdon, R. Genetic correlation as a concept for studying genotype-environment interaction in forest tree breeding. Silvae Genet 1977, 26, 168–175. [Google Scholar]
- Gilmour, A.; Gogel, B.; Cullis, B.; Thompson, R. ASReml User Guide Release 4.0; VSN International Ltd.: Hemel Hempstead, UK, 2014. [Google Scholar]
- Malosetti, M.; Ribaut, J.-M.; van Eeuwijk, F.A. The statistical analysis of multi-environment data: Modeling genotype-by-environment interaction and its genetic basis. Front. Physiol. 2013, 4, 44. [Google Scholar] [CrossRef]
- Hägglund, B.; Lundmark, J.-E. Site Index Estimation by Means of Site Properties; Skogshögskolanc: Stockholm, Sweden, 1977. [Google Scholar]
- Sharma, R.P.; Brunner, A.; Eid, T. Site index prediction from site and climate variables for Norway spruce and Scots pine in Norway. Scand. J. For. Res. 2012, 27, 619–636. [Google Scholar] [CrossRef]
- Zhao, H.; Gao, G.; An, W.; Zou, X.; Li, H.; Hou, M. Timescale differences between SC-PDSI and SPEI for drought monitoring in China. Phys. Chem. Earthparts A/B/C 2015, 102, 48–58. [Google Scholar] [CrossRef]
- Vicente-Serrano, S.M.; Beguería, S.; Lorenzo-Lacruz, J.; Camarero, J.J.; López-Moreno, J.I.; Azorin-Molina, C.; Revuelto, J.; Morán-Tejeda, E.; Sanchez-Lorenzo, A. Performance of drought indices for ecological, agricultural, and hydrological applications. Earth Interact. 2012, 16, 1–27. [Google Scholar] [CrossRef]
- Kroon, J.; Ericsson, T.; Jansson, G.; Andersson, B. Patterns of genetic parameters for height in field genetic tests of Picea abies and Pinus sylvestris in Sweden. Tree Genet. Genomes 2011, 7, 1099–1111. [Google Scholar] [CrossRef]
- Chen, Z.-Q.; Hai, H.N.T.; Helmersson, A.; Liziniewicz, M.; Hallingbäck, H.R.; Fries, A.; Berlin, M.; Wu, H.X. Advantage of clonal deployment in Norway spruce (Picea abies (L.) H. Karst). Ann. For. Sci. 2020, 77, 1–15. [Google Scholar] [CrossRef]
- Richards, T.J.; Karacic, A.; Apuli, R.-P.; Weih, M.; Ingvarsson, P.K.; Rönnberg-Wästljung, A.C. Quantitative genetic architecture of adaptive phenology traits in the deciduous tree, Populus trichocarpa (Torr. and Gray). Heredity 2020, 125, 449–458. [Google Scholar] [CrossRef]
- Rubio-Cuadrado, Á.; Bravo-Oviedo, A.; Mutke, S.; Del Río, M. Climate effects on growth differ according to height and diameter along the stem in Pinus pinaster Ait. iForest-Biogeosci. For. 2018, 11, 237–242. [Google Scholar] [CrossRef]
- Gyllenstrand, N.; Clapham, D.; Källman, T.; Lagercrantz, U.; Richardt, S.; Lang, D.; Reski, R.; Frank, W.; Rensing, S.A. A Norway spruce FLOWERING LOCUS T homolog is implicated in control of growth rhythm in conifers. Plant Physiol. 2007, 144, 248–257. [Google Scholar] [CrossRef] [PubMed]
- White, T.L.; Adams, W.T.; Neale, D.B. Forest Genetics; CABI: Wallingford, UK, 2007. [Google Scholar]
- Chen, Z.-Q.; Karlsson, B.; Wu, H.X. Patterns of additive genotype-by-environment interaction in tree height of Norway spruce in southern and central Sweden. Tree Genet. Genomes 2017, 13, 25. [Google Scholar] [CrossRef]
- Karlsson, B.; Högberg, K. Genotypic parameters and clone x site interaction in clone tests of Norway spruce (Picea abies (L.) Karst.). For. Genet. 1998, 5, 21–30. [Google Scholar]
- Berlin, M.; Jansson, G.; Högberg, K.-A. Genotype by environment interaction in the southern Swedish breeding population of Picea abies using new climatic indices. Scand. J. For. Res. 2014, 30, 112–121. [Google Scholar] [CrossRef]
| Details | Swedish Trials | Finnish Trials | ||||
|---|---|---|---|---|---|---|
| Trial | Nässja | Sunnansjö | Ängelholm | Kohagen | Nurmijärvi | Pori |
| Latitude (N) | 60.27 | 60.17 | 56.35 | 57.62 | 60.50 | 61.65 |
| Longitude (E) | 16.79 | 14.97 | 13.02 | 15.47 | 24.71 | 21.73 |
| Altitude (m) | 81 | 175 | 170 | 165 | 100 | 20 |
| Soil type | Podzol, morain | Podzol, sandy morain | Podzol, morain | Podzol, morain | Clayey fine silt | Fine sand |
| a Tsum (°C) | 1337.3 | 1216.7 | 1414.6 | 1407.7 | 1298.3 | 1264.4 |
| b MAP (mm) | 648.3 | 757.2 | 1189.7 | 643.4 | 579.7 | 538.2 |
| No. of clones | 2121 | 2120 | 2600 | 2601 | 1062 | 931 |
| No. of families | 57 | 57 | 84 | 85 | 96 | 96 |
| Average No. of ramets/clone | 3.99 | 3.95 | 3.31 | 3.32 | 5.99 | 7.98 |
| Average No. of clones/family | 37.2 | 37.2 | 30.9 | 30.6 | 10 | 10 |
| Type of material | Full-sib | Full-sib | Full-sib | Full-sib | Half-sib | Half-sib |
| Traits | ||||
|---|---|---|---|---|
| Nässja | ||||
| Ht17 | 9.75 (2.70) | 16.90 (2.10) | 0.10 (0.03) | 0.26 (0.02) |
| Ht18 | 4.4 (3.30) | 20.60 (2.40) | 0.04 (0.03) | 0.23 (0.01) |
| Ht19 | 20.2 (5.50) | 18.20 (3.90) | 0.10 (0.03) | 0.20 (0.02) |
| Tot-Ht16 | 22.76 (11.30) | 78.42 (8.40) | 0.06 (0.03) | 0.26 (0.02) |
| Tot-Ht17 | 50.3 (21.50) | 147.55 (15.40) | 0.07 (0.03) | 0.29 (0.02) |
| Tot-Ht18 | 77.51 (38.40) | 252.28 (26.30) | 0.07 (0.03) | 0.30 (0.02) |
| Tot-Ht19 | 136.9 (65.50) | 370.30 (43.20) | 0.07 (0.03) | 0.29 (0.02) |
| Sunnansjö | ||||
| Ht17 | 8.3 (5.60) | 20.90 (3.40) | 0.05 (0.04) | 0.22 (0.02) |
| Ht18 | 15.0 (6.60) | 28.73 (4.40) | 0.07 (0.03) | 0.20 (0.01) |
| Ht19 | * 0.00 | 43.4 (3.30) | * 0.00 | 0.20 (0.01) |
| Tot-Ht16 | 26.50 (12.50) | 75.60 (8.80) | 0.06 (0.03) | 0.23 (0.01) |
| Tot-Ht17 | 57.05 (30.0) | 152.00 (18.90) | 0.07 (0.03) | 0.27 (0.02) |
| Tot-Ht18 | 102.10 (55.50) | 270.90 (34.50) | 0.07 (0.04) | 0.27 (0.02) |
| Tot-Ht19 | 185.20 (87.70) | 462.80 (55.50) | 0.07 (0.03) | 0.27 (0.01) |
| Ängelholm | ||||
| Ht17 | 12.73 (4.48) | 18.87 (2.89) | 0.09 (0.03) | 0.32 (0.02) |
| Ht18 | 20.15 (5.29) | 18.52 (3.21) | 0.15 (0.04) | 0.29 (0.02) |
| Ht19 | 36.89 (11.55) | 58.79 (7.25) | 0.12 (0.04) | 0.23 (0.01) |
| Tot-Ht16 | 61.95 (15.68) | 31.32 (8.72) | 0.21 (0.05) | 0.33 (0.02) |
| Tot-Ht17 | 123.73 (34.2) | 88.27 (19.14) | 0.19 (0.05) | 0.35 (0.02) |
| Tot-Ht18 | 236.33 (63.0) | 171.3 (34.67) | 0.23 (0.05) | 0.42(0.02) |
| Tot-Ht19 | 400.54 (112.19) | 389.02 (62.04) | 0.23 (0.06) | 0.49 (0.02) |
| Kohagen | ||||
| Ht17 | 22.46 (7.20) | 21.45 (4.51) | 0.10 (0.03) | 0.22 (0.02) |
| Ht18 | 110.39 (23.39) | 22.52 (12.65) | 0.29 (0.05) | 0.35 (0.02) |
| Ht19 | 36.75 (10.59) | 46.55 (7.06) | 0.10 (0.03) | 0.24 (0.02) |
| Tot-Ht16 | 131.85 (31.24) | 19.43 (16.61) | 0.29 (0.06) | 0.34 (0.02) |
| Tot-Ht17 | 283.47 (72.68) | 78.56 (38.70) | 0.27 (0.06) | 0.37 (0.02) |
| Tot-Ht18 | 729.72 (168.50) | 159.03 (89.30) | 0.32 (0.06) | 0.41 (0.02) |
| Tot-Ht19 | 1040.83(241.49) | 344.57 (129.05) | 0.31 (0.06) | 0.43 (0.02) |
| Traits | ||||
|---|---|---|---|---|
| Nurmijärvi | ||||
| Ht18 | 48.57 (11.01) | 20.12 (7.00) | 0.17 (0.04) | 0.24 (0.02) |
| Ht19 | 66.78 (13.46) | 1.39 (7.75) | 0.21 (0.04) | 0.21 (0.02) |
| Tot-Ht17 | 815.95 (177.58) | 563.00 (111.04) | 0.26 (0.05) | 0.44 (0.02) |
| Tot-Ht18 | 1197.30 (257.50) | 763.46 (159.40) | 0.27 (0.05) | 0.44 (0.02) |
| Tot-Ht19 | 1796.11 (378.12) | 951.01 (229.16) | 0.28 (0.05) | 0.43 (0.02) |
| Pori | ||||
| Ht18 | 42.11 (10.85) | 23.80 (7.34) | 0.13 (0.03) | 0.20 (0.02) |
| Ht19 | 76.19 (17.35) | 23.24 (10.80) | 0.18 (0.04) | 0.24 (0.02) |
| Tot-Ht17 | 642.20 (166.80) | 652.23 (113.40) | 0.18 (0.04) | 0.36 (0.02) |
| Tot-Ht18 | 983.60 (252.20) | 943.90 (170.10) | 0.18 (0.04) | 0.36 (0.02) |
| Tot-Ht19 | 1634.96 (434.83) | 2047.83 (303.38) | 0.17 (0.04) | 0.39 (0.02) |
| Trait | Trial Pairs | |||||
|---|---|---|---|---|---|---|
| Nässja-Sunnansjö | Ängelholm-Kohagen | Nurmijärvi-Pori | ||||
| Ht17 | 0.69 (0.11) | 9.7, 4.32 | 0.59 (0.11) | 13.6, 9.4 | - | - |
| Ht18 | 0.71 (0.11) | 11.3, 4.6 | 0.23 (0.13) | 14.6, 54.2 | 0.48 (0.11) | 22.4, 23.8 |
| Ht19 | 0.79 (0.09) | 16.5, 4.3 | 0.35 (0.15) | 15.4, 28.7 | 0.44 (0.10) | 25.8, 35.7 |
| Tot-Ht16 | 0.72 (0.11) | 26.3, 10.1 | 0.70 (0.08) | 79.7, 33.8 | - | - |
| Tot-Ht17 | 0.78 (0.09) | 65.4, 18.4 | 0.67 (0.08) | 168.9, 83.3 | 0.66 (0.09) | 506, 256.10 |
| Tot-Ht18 | 0.82 (0.08) | 127.2, 27.7 | 0.54 (0.10) | 296.5, 262.7 | 0.63 (0.10) | 726.8, 421.1 |
| Tot-Ht19 | 0.86 (0.06) | 212.1, 35.8 | 0.52 (0.62) | 437.3, 404.5 | 0.62 (0.10) | 1099.2, 690 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hayatgheibi, H.; Haapanen, M.; Lundströmer, J.; Berlin, M.; Kärkkäinen, K.; Helmersson, A. The Impact of Drought Stress on the Height Growth of Young Norway Spruce Full-Sib and Half-Sib Clonal Trials in Sweden and Finland. Forests 2021, 12, 498. https://doi.org/10.3390/f12040498
Hayatgheibi H, Haapanen M, Lundströmer J, Berlin M, Kärkkäinen K, Helmersson A. The Impact of Drought Stress on the Height Growth of Young Norway Spruce Full-Sib and Half-Sib Clonal Trials in Sweden and Finland. Forests. 2021; 12(4):498. https://doi.org/10.3390/f12040498
Chicago/Turabian StyleHayatgheibi, Haleh, Matti Haapanen, Jenny Lundströmer, Mats Berlin, Katri Kärkkäinen, and Andreas Helmersson. 2021. "The Impact of Drought Stress on the Height Growth of Young Norway Spruce Full-Sib and Half-Sib Clonal Trials in Sweden and Finland" Forests 12, no. 4: 498. https://doi.org/10.3390/f12040498
APA StyleHayatgheibi, H., Haapanen, M., Lundströmer, J., Berlin, M., Kärkkäinen, K., & Helmersson, A. (2021). The Impact of Drought Stress on the Height Growth of Young Norway Spruce Full-Sib and Half-Sib Clonal Trials in Sweden and Finland. Forests, 12(4), 498. https://doi.org/10.3390/f12040498






