IoT Monitoring of Urban Tree Ecosystem Services: Possibilities and Challenges
Abstract
1. Introduction
2. Materials and Methods
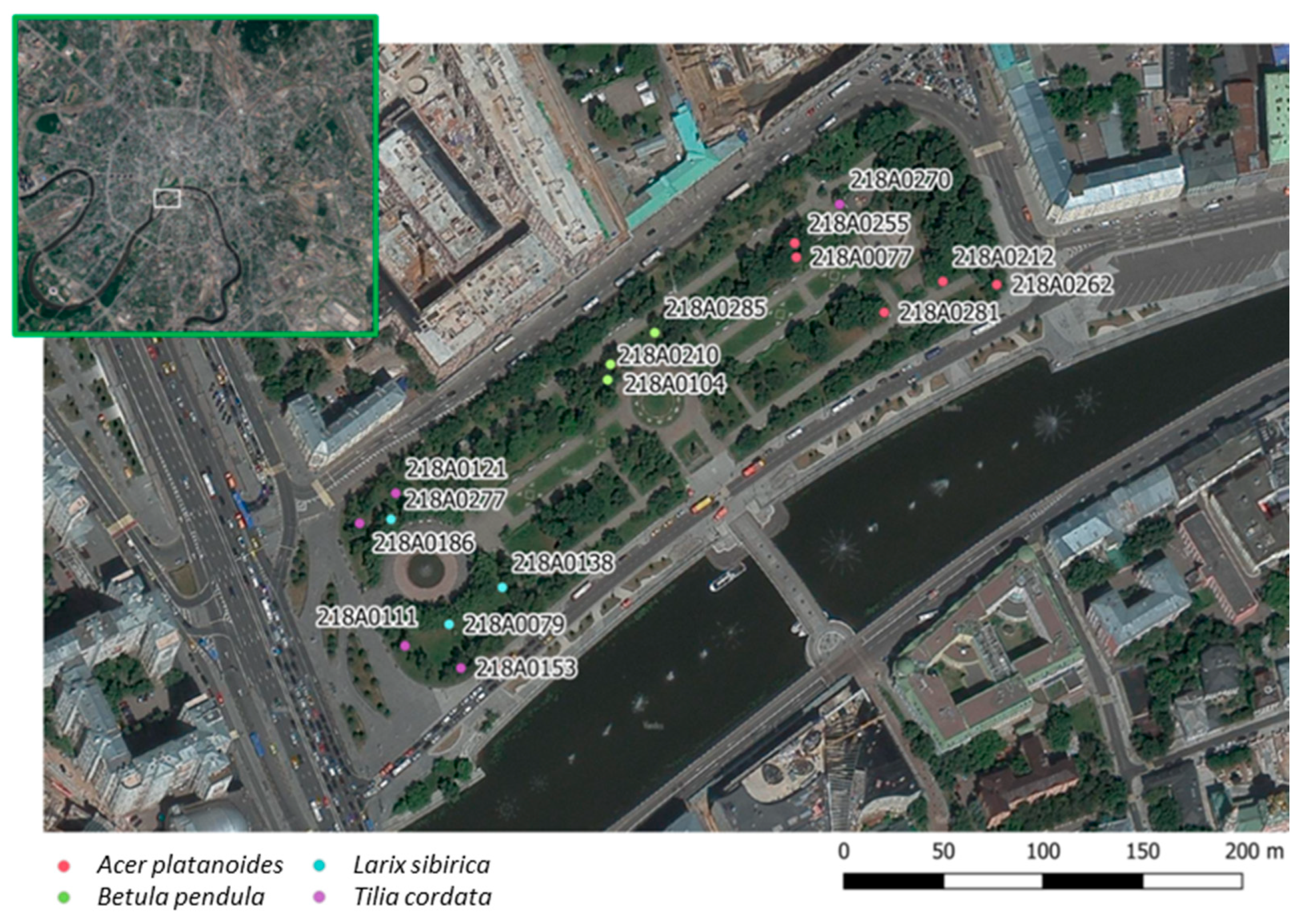
2.1. Study Site and Network Setup
2.2. Choice of ES Indicators
2.2.1. Carbon Sequestration
2.2.2. Climate Regulation via Air Temperature Control
2.2.3. Water Fluxes and Energy Consumption through Transpiration
2.2.4. LAI
2.2.5. Particulate Adsorption
2.3. Data Processing
3. Results and Discussion
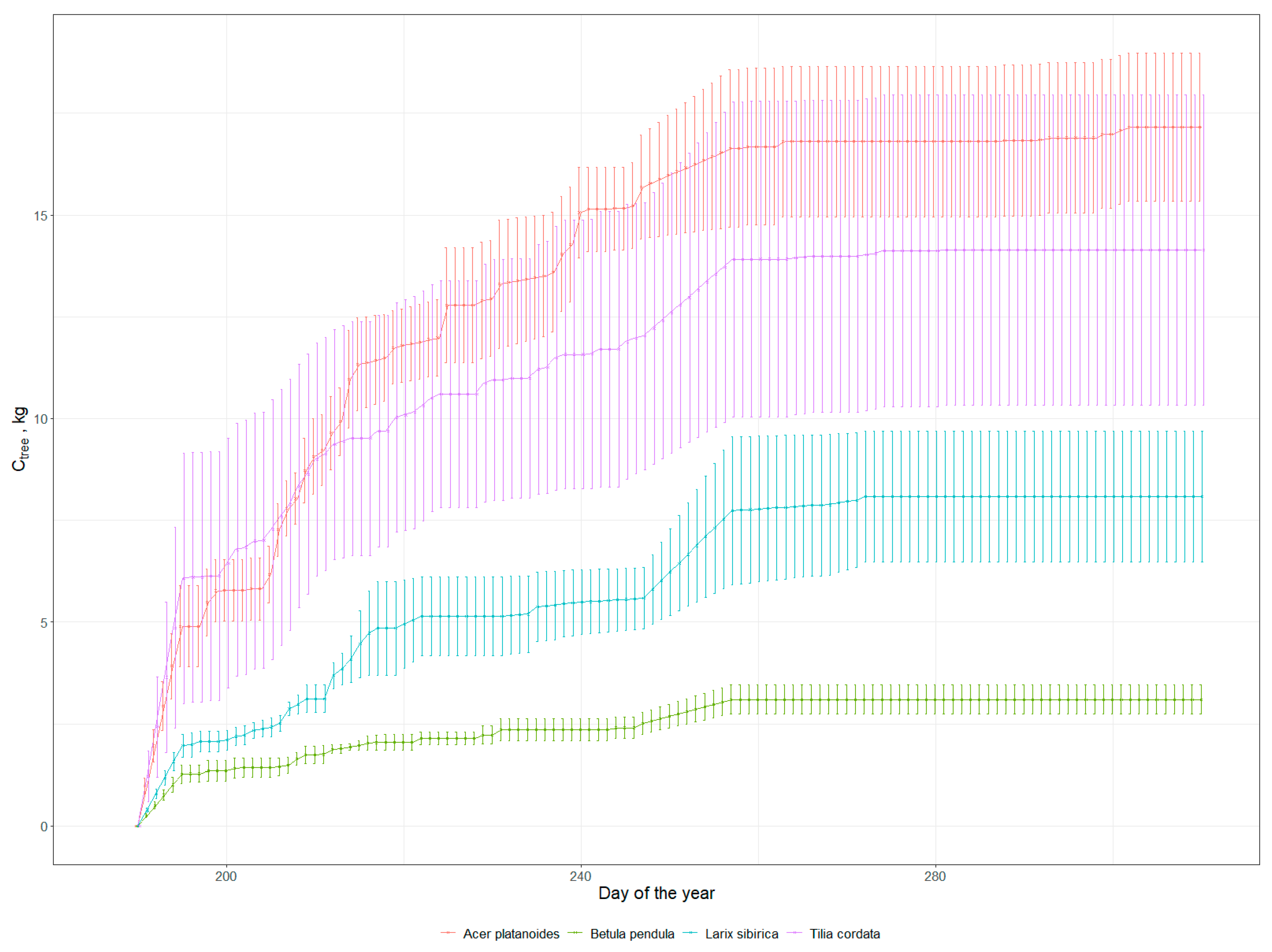
3.1. Carbon Sequestration
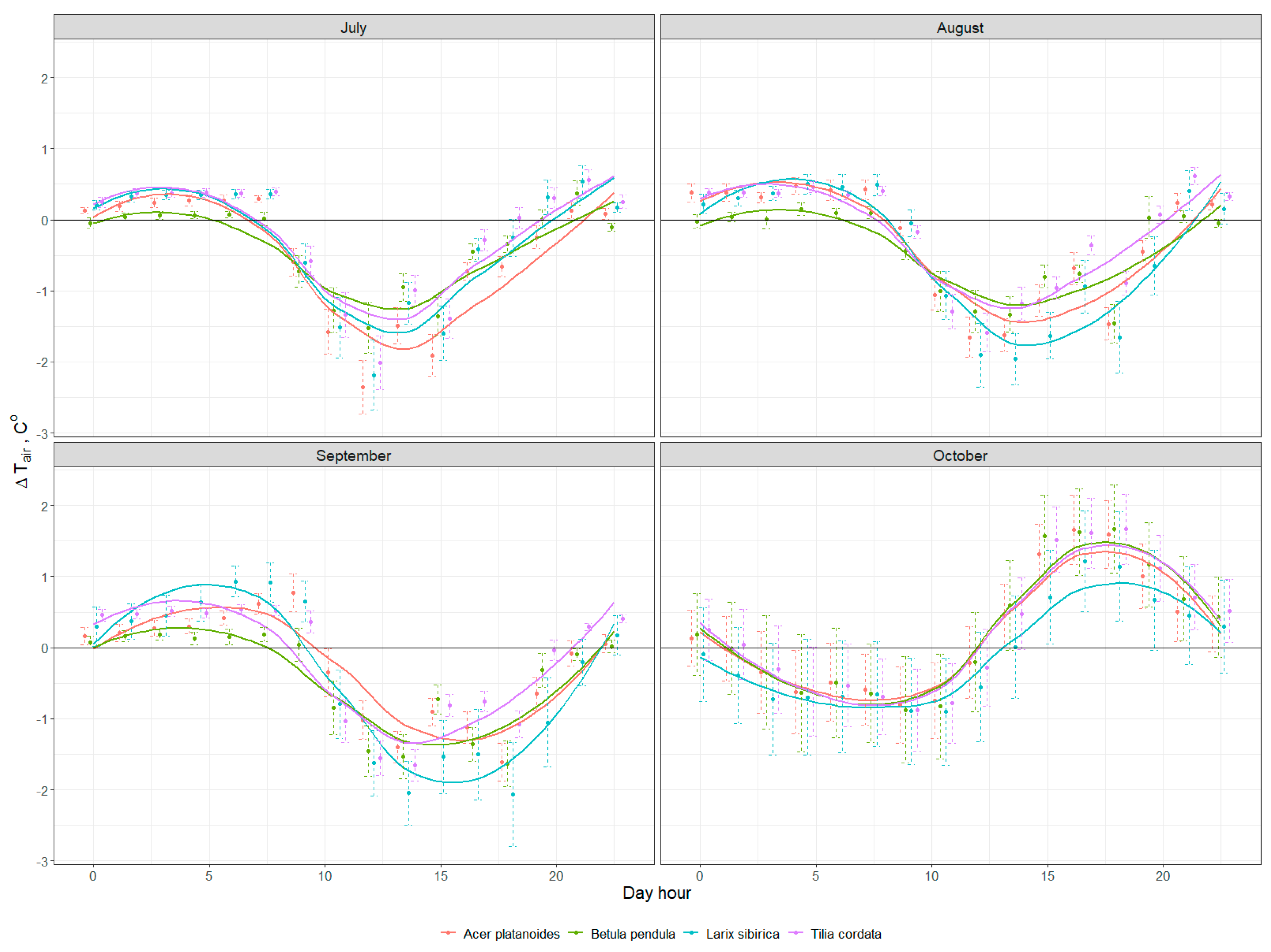
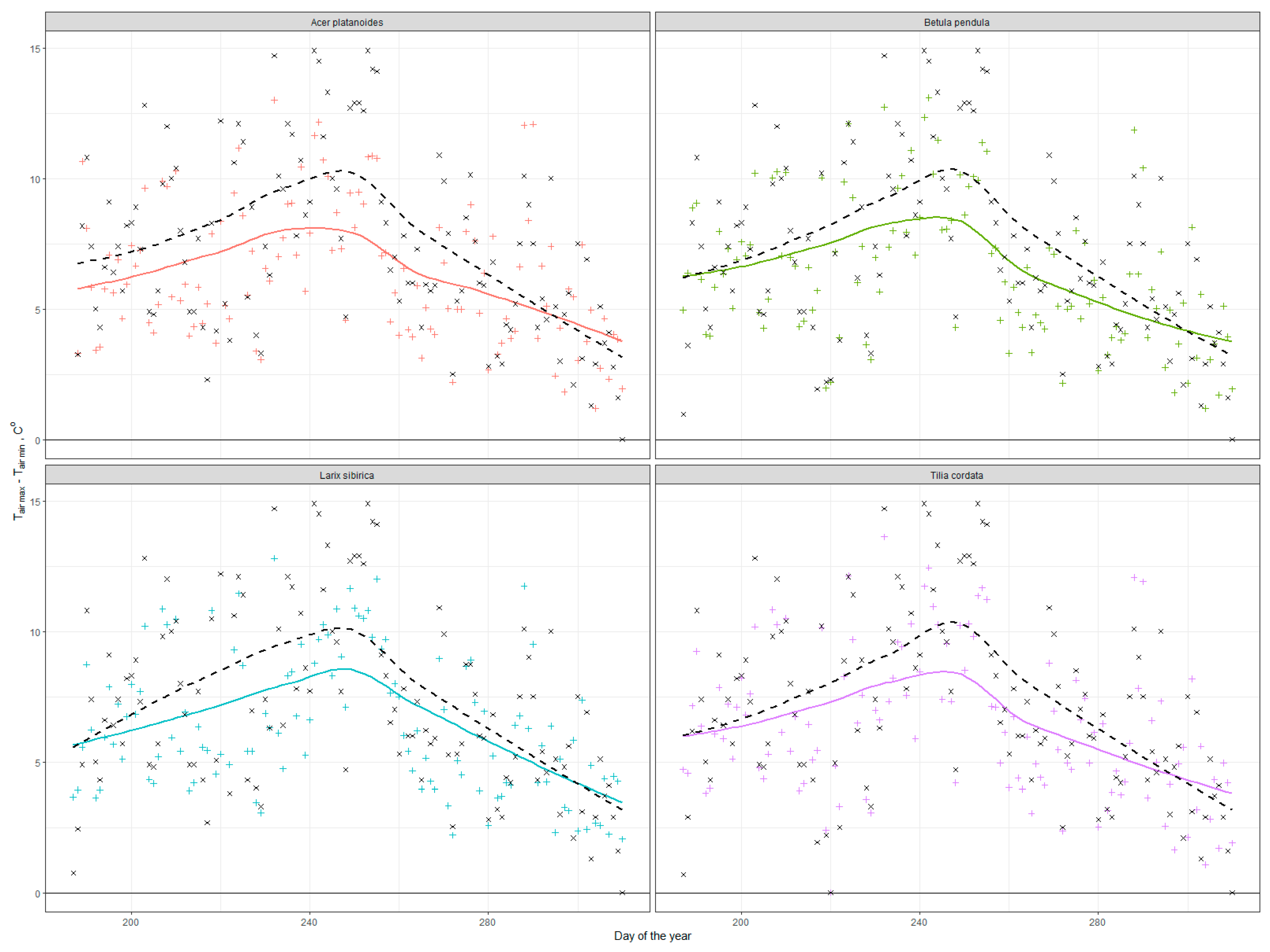
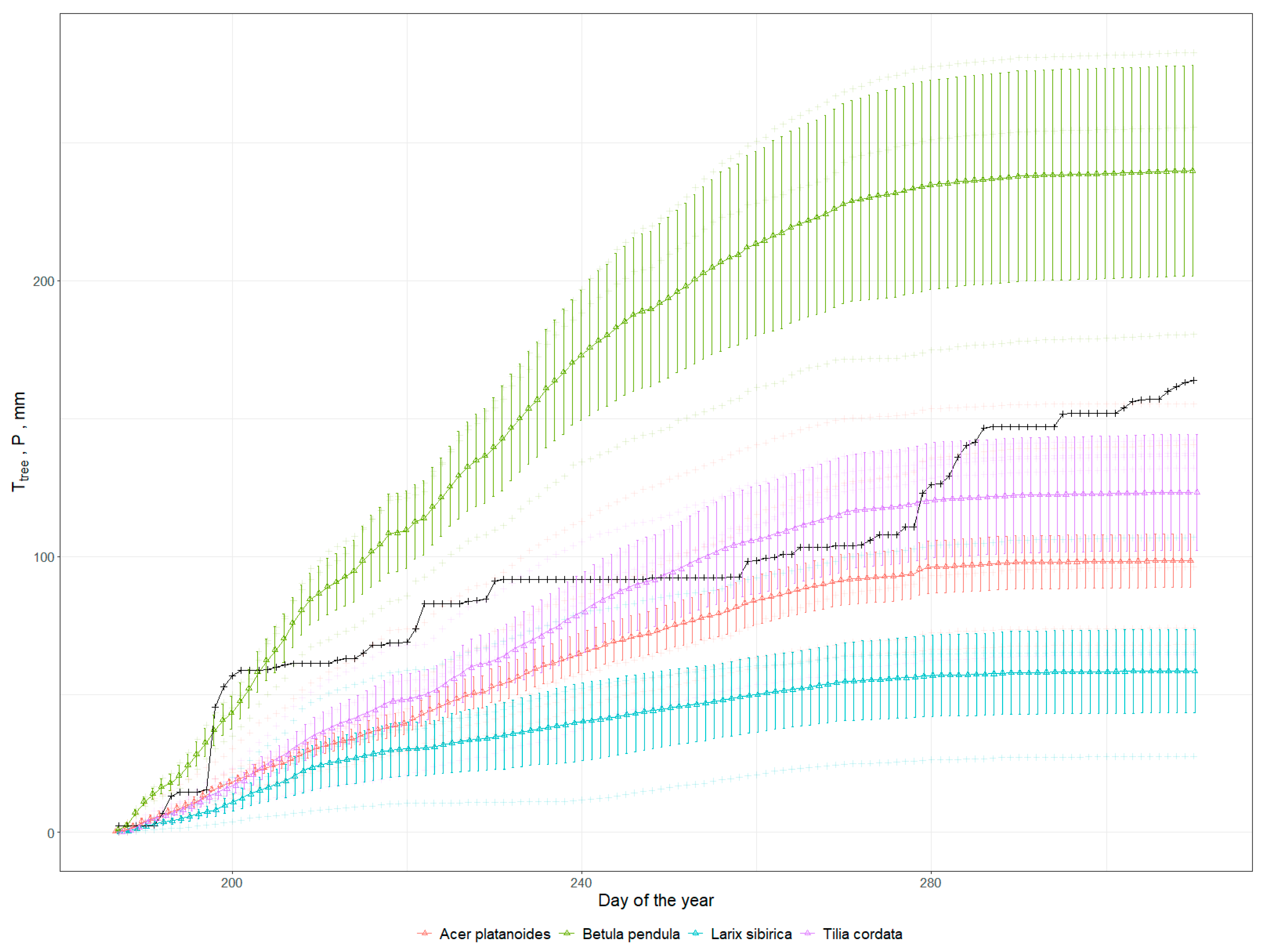
3.2. Cooling Effect
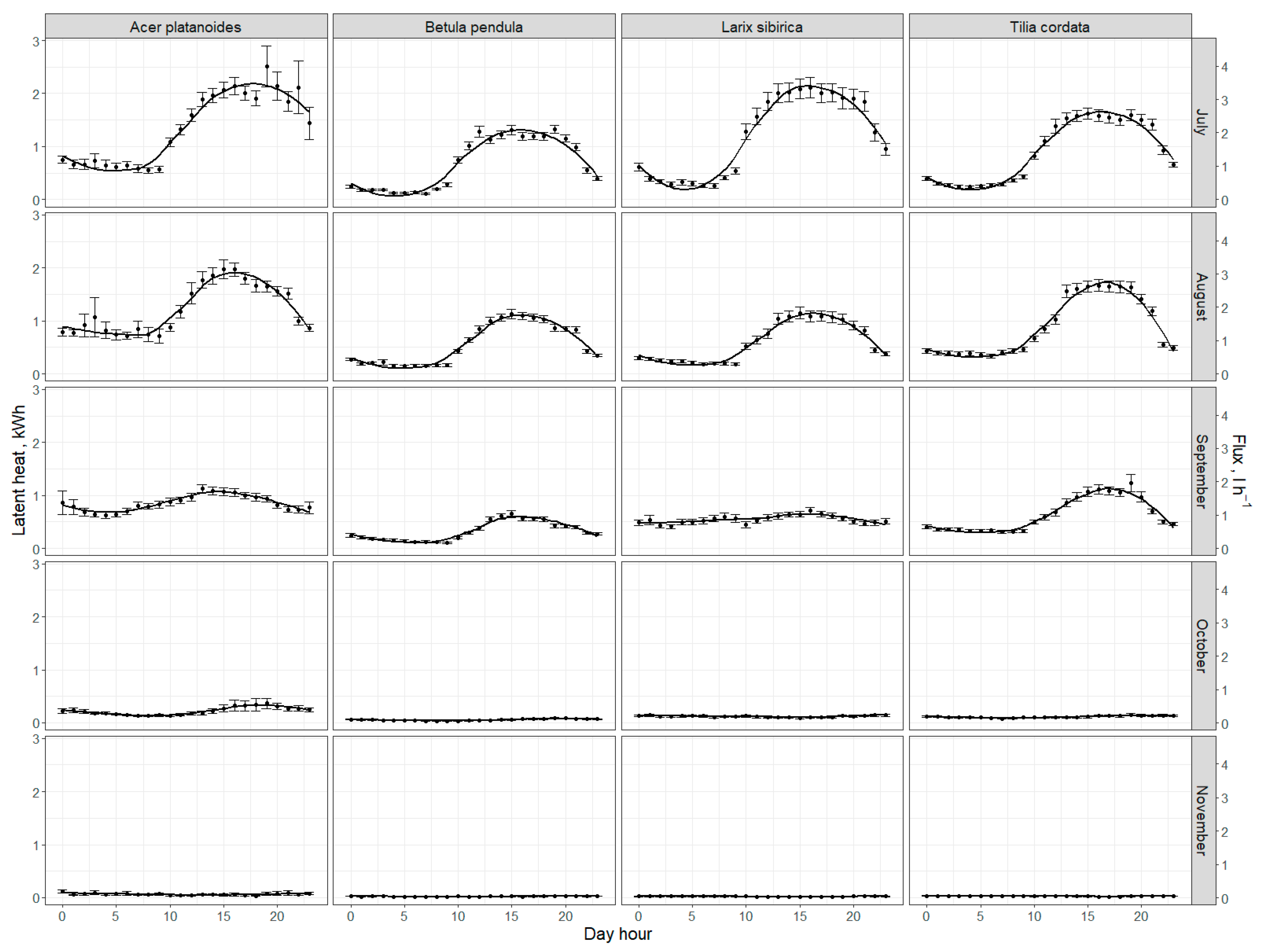
3.3. Run-Off Mitigation and Energy Consumption by Trees via Transpiration
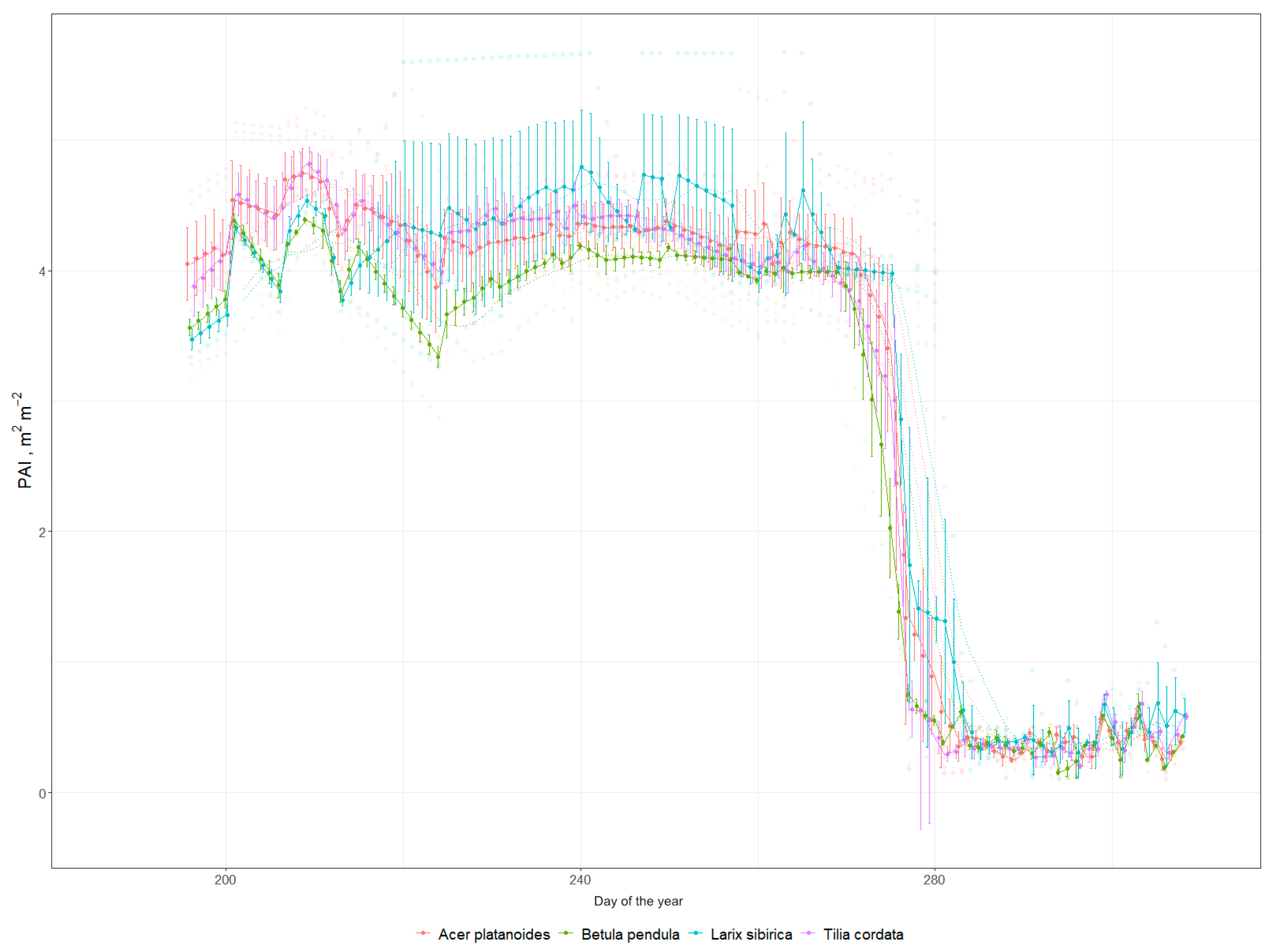
3.4. LAI as a Proxy for ES Provision
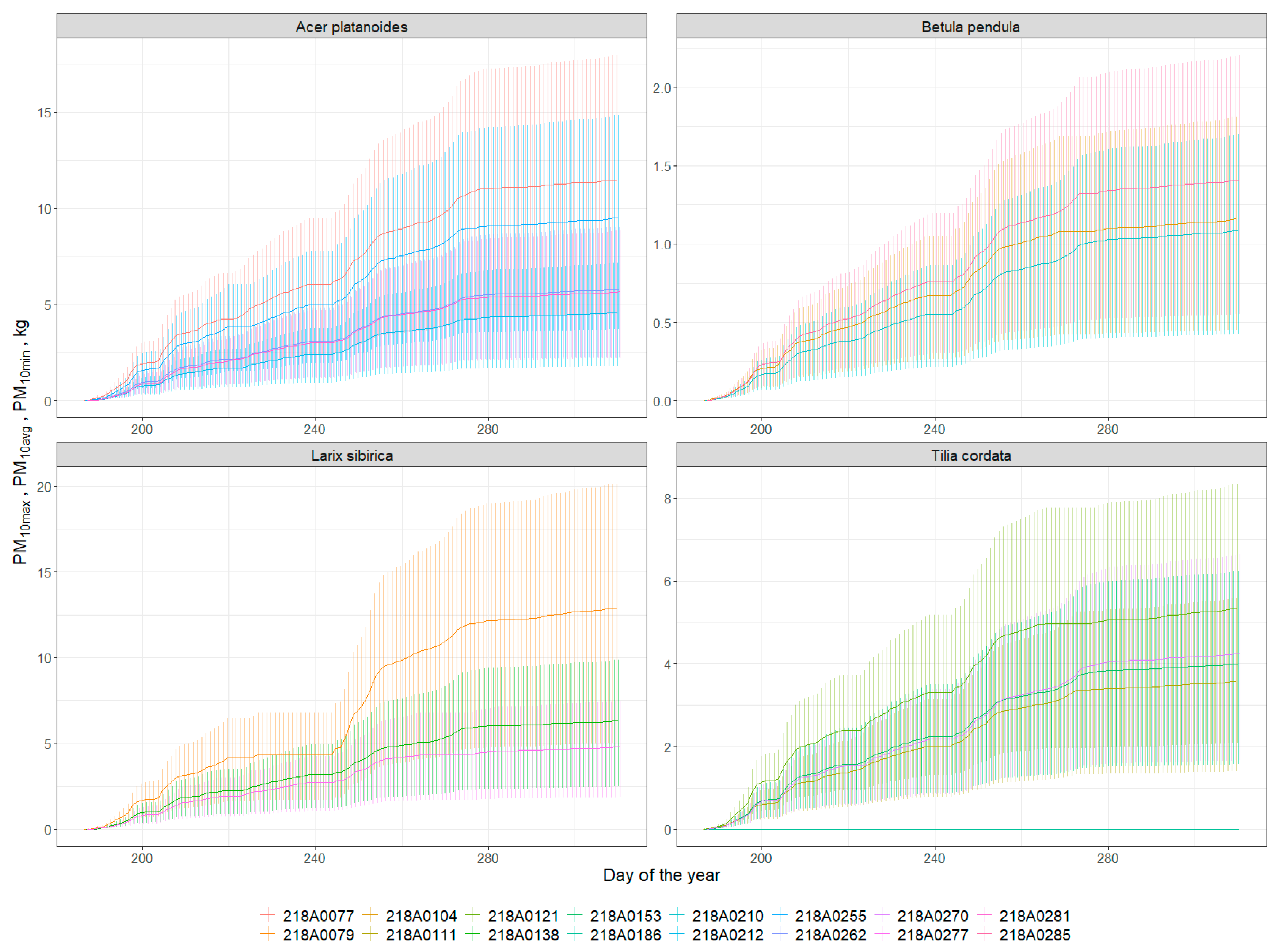
3.5. Particulate Adsorption
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Trees Description | Biomass Carbon | Transpiration and Precipitation | Energy Absorbed - L, kWh | PM10 Particles Absorbed, kg | Leaf and Wood Indexes | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| id | Age Group | Tree Height, m | Stem Diameter, cm | Stem Radial Increment | Canopy Area, m2 | VTA | BEF | BCEF | R/S | Total Tree-carbon Stock, kg | Average Annual Carbon Increment | Current Annual Increment kg | Carbon Stored per Canopy Area, kg m−2 | Transpiration, mm | Precipitation, mm | Ratio of Precipitation Evaporated, mm | PM10max | PM10avg | PM10min | PAI, m2m−2 | WAI, m2m−2 | LAI, m2m−2 | |
| Acer platanoides | |||||||||||||||||||||||
| 218A0077 | 50–60 | 20 | 35.7 | 3.4 | 55.7 | 2 | 1.3 | 1.1 | 0.3 | 580.0 | 10.6 | 13.0 | 0.2 | 68.0 | 183.5 | 0.4 | 2454 | 18.0 | 11.5 | 4.5 | 4.8 | 0.5 | 4.3 |
| 218A0212 | 50–60 | 15 | 33.7 | 3.2 | 27.6 | 3 | 1.3 | 1.1 | 0.3 | 393.6 | 7.2 | 8.8 | 0.3 | 96.2 | 183.5 | 0.5 | 1615 | 7.3 | 4.6 | 1.8 | 4.0 | 0.5 | 3.5 |
| 218A0255 | 50–60 | 20 | 34.4 | 3.2 | 55.3 | 2 | 1.3 | 1.1 | 0.3 | 559.8 | 10.2 | 12.5 | 0.2 | 74.0 | 183.5 | 0.4 | 2506 | 15.1 | 9.6 | 3.8 | 4.3 | 0.4 | 3.8 |
| 218A0262 | 50–60 | 13 | 34.7 | 3.3 | 28.5 | 1 | 1.3 | 1.1 | 0.3 | 373.7 | 6.8 | 8.4 | 0.3 | 155.2 | 183.5 | 0.8 | 2739 | 7.9 | 5.0 | 2.0 | 4.8 | 0.6 | 4.2 |
| 218A0281 | 50–60 | 14 | 45.8 | 4.3 | 35.8 | 4 | 1.3 | 1.1 | 0.3 | 685.0 | 12.5 | 15.3 | 0.4 | 140.4 | 183.5 | 0.8 | 3042 | 11.9 | 7.6 | 3.0 | 3.6 | 0.4 | 3.2 |
| Mean | 16.8 | 36.9 | 3.5 | 40.6 | 518.4 | 9.4 | 11.6 | 0.3 | 106.8 | 183.5 | 0.6 | 2471.2 | 12.0 | 7.7 | 3.0 | 4.3 | 0.5 | 3.8 | |||||
| SE | 1.7 | 2.5 | 0.2 | 7.0 | 66.0 | 1.2 | 1.5 | 0.0 | 39.3 | 0.0 | 0.2 | 266.0 | 2.3 | 1.5 | 0.6 | 0.2 | 0.0 | 0.2 | |||||
| Betula Pendula | |||||||||||||||||||||||
| 218A0104 | 50–60 | 11 | 21.7 | 2.7 | 7.6 | 1 | 1.2 | 0.8 | 0.2 | 80.4 | 1.5 | 2.4 | 0.3 | 180.5 | 183.5 | 1.0 | 1157 | 2.0 | 1.3 | 0.5 | 4.0 | 0.4 | 3.5 |
| 218A0210 | 30–40 | 11 | 21.0 | 2.6 | 6.4 | 1 | 1.2 | 0.8 | 0.2 | 73.7 | 1.3 | 2.2 | 0.3 | 255.5 | 183.5 | 1.4 | 1226 | 2.0 | 1.3 | 0.5 | 4.0 | 0.4 | 3.6 |
| 218A0285 | 30–40 | 11 | 23.9 | 3.0 | 8.2 | 1 | 1.2 | 0.8 | 0.2 | 93.9 | 1.7 | 2.8 | 0.3 | 282.7 | 183.5 | 1.5 | 1756 | 2.5 | 1.6 | 0.6 | 4.1 | 0.4 | 3.7 |
| Mean | 11.0 | 22.2 | 2.8 | 7.4 | 82.7 | 1.5 | 2.5 | 0.3 | 239.6 | 183.5 | 1.3 | 1379.4 | 2.2 | 1.4 | 0.5 | 4.0 | 0.4 | 3.6 | |||||
| SE | 0.0 | 7.0 | 0.9 | 0.6 | 8.3 | 0.2 | 0.4 | 0.1 | 52.9 | 0.0 | 0.3 | 436.7 | 0.1 | 0.1 | 0.0 | 0.1 | 0.0 | 0.1 | |||||
| Larix Sibirica | |||||||||||||||||||||||
| 218A0079 | 80–100 | 25 | 32.2 | 2.0 | 65.9 | 3 | 1.1 | 0.8 | 0.3 | 421.1 | 7.7 | 6.3 | 0.1 | 27.5 | 183.5 | 0.1 | 1701 | 17.5 | 11.2 | 4.4 | 4.7 | 0.8 | 3.9 |
| 218A0138 | 80–100 | 19 | 40.7 | 2.6 | 37.4 | 2 | 1.1 | 0.8 | 0.3 | 519.5 | 9.5 | 7.8 | 0.2 | 106.9 | 183.5 | 0.6 | 3238 | 9.8 | 6.3 | 2.5 | 4.1 | 0.5 | 3.6 |
| 218A0277 | 80–100 | 24 | 26.1 | 1.6 | 32.3 | 2 | 1.1 | 0.8 | 0.3 | 272.5 | 5.0 | 4.1 | 0.1 | 65.3 | 183.5 | 0.4 | 1481 | 8.8 | 5.6 | 2.2 | 4.0 | 0.4 | 3.6 |
| Mean | 22.7 | 33.0 | 2.1 | 45.2 | 404.4 | 7.4 | 6.0 | 0.1 | 66.6 | 183.5 | 0.4 | 2140.0 | 12.1 | 7.7 | 3.0 | 4.3 | 0.5 | 3.7 | |||||
| SE | 2.2 | 5.2 | 0.3 | 12.8 | 87.9 | 1.6 | 1.3 | 0.0 | 39.7 | 0.0 | 0.2 | 676.9 | 3.4 | 2.2 | 0.8 | 0.2 | 0.1 | 0.1 | |||||
| Tilia Cordata | |||||||||||||||||||||||
| 218A0111 | 50–60 | 12 | 28.0 | 5.3 | 20.0 | 3 | 1.2 | 0.7 | 0.3 | 137.6 | 2.5 | 6.1 | 0.3 | 132.1 | 183.5 | 0.7 | 2195 | 6.5 | 4.1 | 1.6 | 4.3 | 0.5 | 3.8 |
| 218A0121 | 50–60 | 17 | 37.9 | 7.1 | 31.3 | 1 | 1.2 | 0.7 | 0.3 | 345.0 | 6.3 | 15.3 | 0.5 | 142.1 | 183.5 | 0.8 | 3370 | 6.7 | 4.3 | 1.7 | 4.6 | 0.6 | 4.0 |
| 218A0153 | 40–50 | 14 | 35.3 | 6.7 | 21.1 | 2 | 1.2 | 0.7 | 0.3 | 245.6 | 4.5 | 10.9 | 0.5 | 137.3 | 183.5 | 0.7 | 2196 | 5.4 | 3.5 | 1.4 | 4.4 | 0.4 | 4.0 |
| 218A0186 | 40–50 | 17 | 40.4 | 7.6 | 19.5 | 3 | 1.2 | 0.7 | 0.3 | 400.1 | 7.3 | 17.8 | 0.9 | 136.5 | 183.5 | 0.7 | 2152 | 4.9 | 3.1 | 1.2 | 3.8 | 0.4 | 3.4 |
| 218A0270 | 30–40 | 11 | 25.2 | 4.7 | 22.4 | 3 | 1.2 | 0.7 | 0.3 | 96.8 | 1.8 | 4.3 | 0.2 | 64.4 | 183.5 | 0.4 | 1196 | 6.5 | 4.1 | 1.6 | 4.4 | 0.5 | 4.0 |
| Mean | 14.1 | 33.4 | 6.3 | 22.9 | 245.0 | 4.5 | 10.9 | 0.5 | 122.5 | 183.5 | 0.7 | 2221.7 | 6.0 | 3.8 | 1.5 | 4.3 | 0.5 | 3.8 | |||||
| SE | 1.3 | 3.3 | 0.6 | 2.4 | 65.0 | 1.2 | 2.9 | 0.1 | 32.7 | 0.0 | 0.2 | 385.4 | 0.4 | 0.3 | 0.1 | 0.1 | 0.0 | 0.1 | |||||
Appendix B
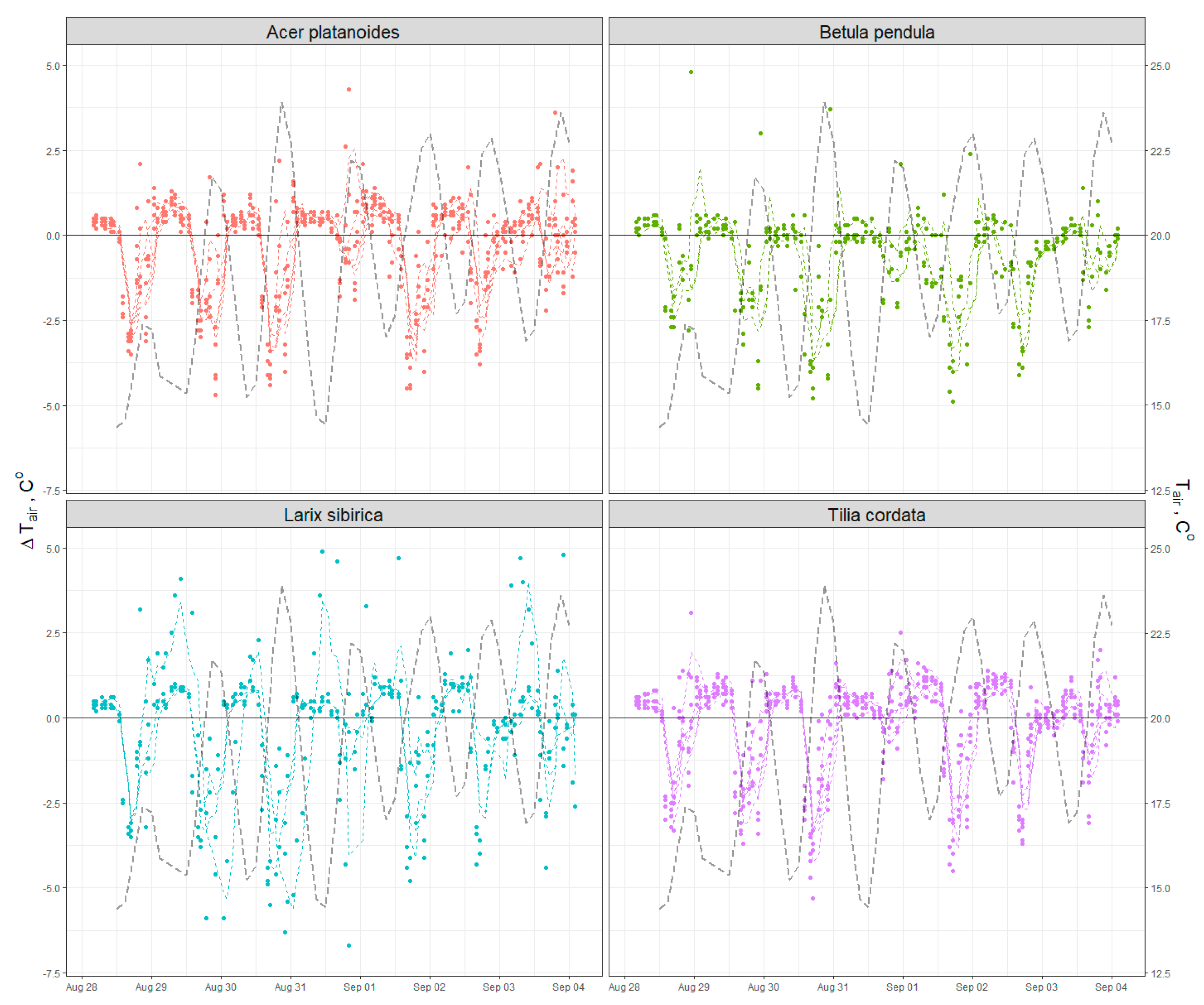
Appendix C
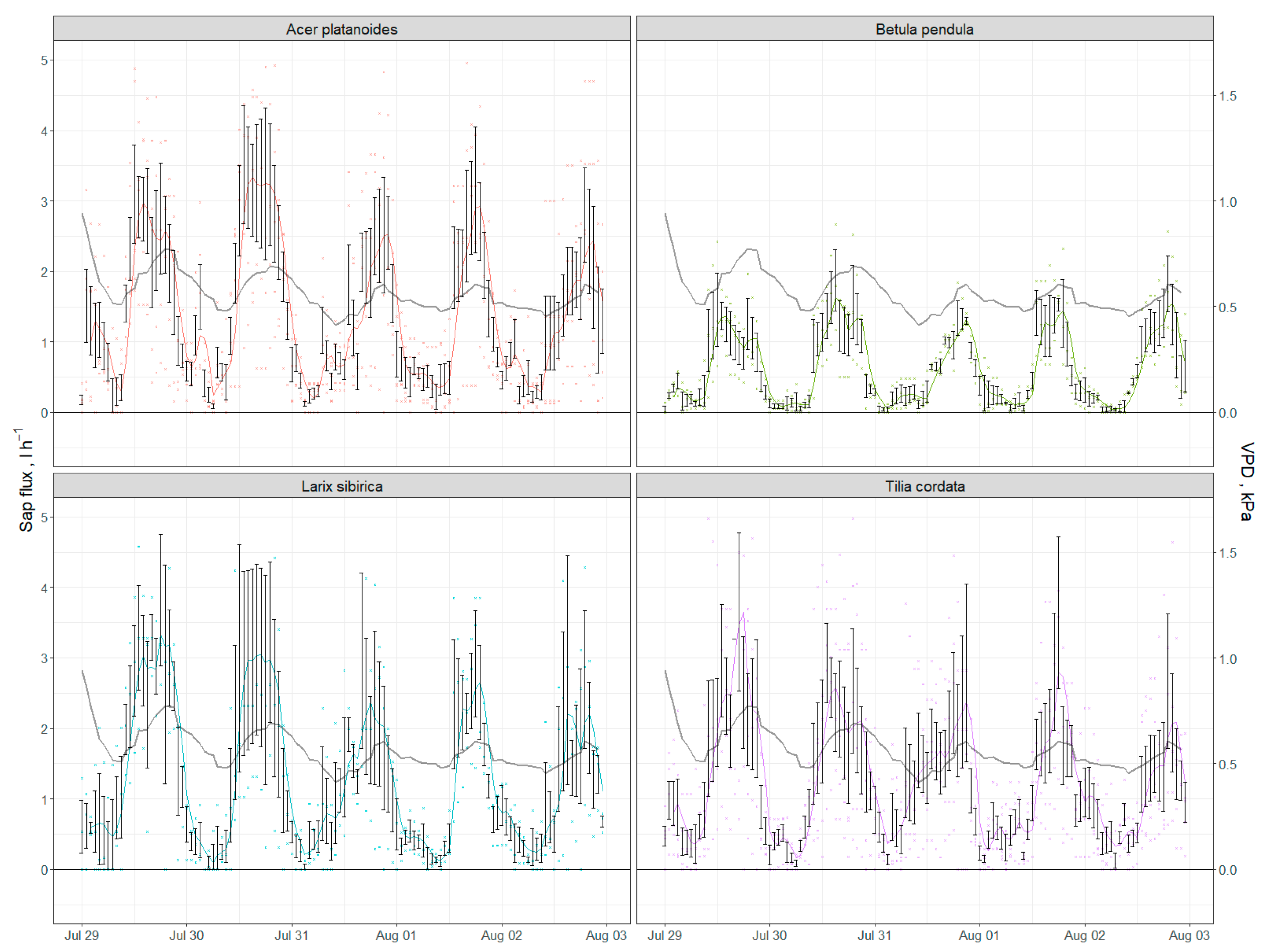
Appendix D

Appendix E
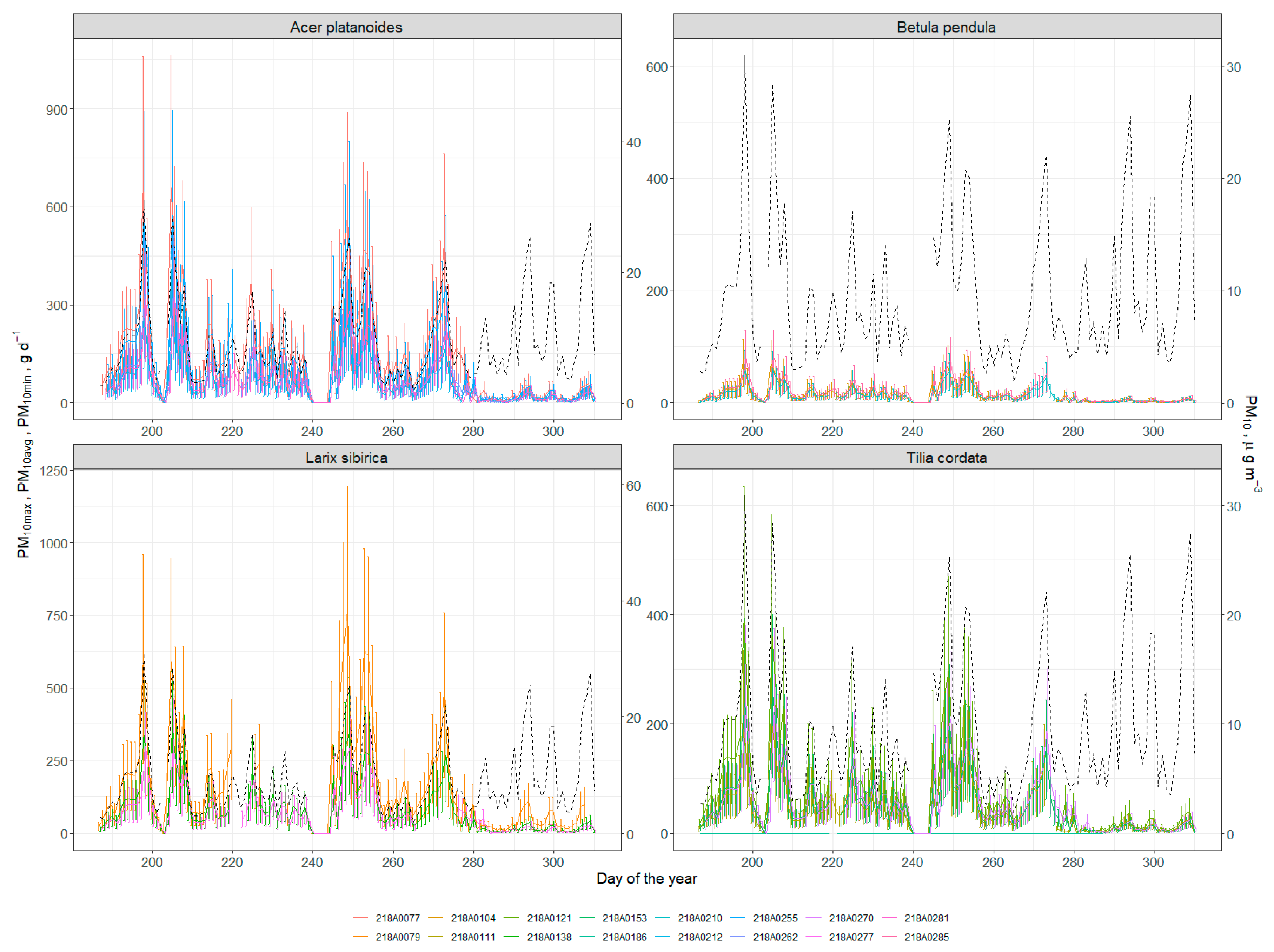
References
- Dye, C. Health and Urban Living. Science 2008, 319, 766. [Google Scholar] [CrossRef] [PubMed]
- Bettencourt, L.M.A.; Lobo, J.; Helbing, D.; Kuhnert, C.; West, G.B. Growth, innovation, scaling, and the pace of life in cities. Proc. Natl. Acad. Sci. USA 2007, 104, 7301–7306. [Google Scholar] [CrossRef] [PubMed]
- Frumkin, H. Healthy Places: Exploring the Evidence. Am. J. Public Health 2003, 93, 1451–1456. [Google Scholar] [CrossRef] [PubMed]
- Lederbogen, F.; Kirsch, P.; Haddad, L.; Streit, F.; Tost, H.; Schuch, P.; Wüst, S.; Pruessner, J.C.; Rietschel, M.; Deuschle, M.; et al. City living and urban upbringing affect neural social stress processing in humans. Nature 2011, 474, 498–501. [Google Scholar] [CrossRef] [PubMed]
- Grimm, N.B.; Faeth, S.H.; Golubiewski, N.E.; Redman, C.L.; Wu, J.; Bai, X.; Briggs, J.M. Global Change and the Ecology of Cities. Science 2008, 319, 756. [Google Scholar] [CrossRef] [PubMed]
- Seto, K.C.; Guneralp, B.; Hutyra, L.R. Global forecasts of urban expansion to 2030 and direct impacts on biodiversity and carbon pools. Proc. Natl. Acad. Sci. USA 2012, 109, 16083–16088. [Google Scholar] [CrossRef] [PubMed]
- Bolund, P.; Hunhammar, S. Ecosystem services in urban areas. Ecol. Econ. 1999, 29, 293–301. [Google Scholar] [CrossRef]
- Guo, Z.; Zhang, L.; Li, Y. Increased Dependence of Humans on Ecosystem Services and Biodiversity. PLoS ONE 2010, 5, e13113. [Google Scholar] [CrossRef]
- Krausmann, F.; Lauk, C.; Haas, W.; Wiedenhofer, D. From resource extraction to outflows of wastes and emissions: The socioeconomic metabolism of the global economy, 1900–2015. Glob. Environ. Change 2018, 52, 131–140. [Google Scholar] [CrossRef]
- Blanusa, T.; Garratt, M.; Cathcart-James, M.; Hunt, L.; Cameron, R.W.F. Urban hedges: A review of plant species and cultivars for ecosystem service delivery in north-west Europe. Urban For. Urban Green. 2019, 44, 126391. [Google Scholar] [CrossRef]
- Gómez-Baggethun, E.; Barton, D.N. Classifying and valuing ecosystem services for urban planning. Ecol. Econ. 2013, 86, 235–245. [Google Scholar] [CrossRef]
- Lovell, S.T.; Taylor, J.R. Supplying urban ecosystem services through multifunctional green infrastructure in the United States. Landsc. Ecol. 2013, 28, 1447–1463. [Google Scholar] [CrossRef]
- Neugarten, R.A.; Langhammer, P.F.; Osipova, E.; Bagstad, K.J.; Bhagabati, N.; Butchart, S.H.M.; Dudley, N.; Elliott, V.; Gerber, L.R.; Gutierrez Arrellano, C.; et al. Tools for Measuring, Modelling, and Valuing Ecosystem Services: Guidance for Key Biodiversity Areas, Natural World Heritage Sites, and Protected Areas, 1st ed.; Groves, C., Ed.; IUCN, International Union for Conservation of Nature: Gland, Switzerland, 2018; ISBN 978-2-8317-1917-7. [Google Scholar]
- Zhao, C.; Sander, H.A. Assessing the sensitivity of urban ecosystem service maps to input spatial data resolution and method choice. Landsc. Urban Plan. 2018, 175, 11–22. [Google Scholar] [CrossRef]
- Andersson, E.; Barthel, S.; Borgström, S.; Colding, J.; Elmqvist, T.; Folke, C.; Gren, Å. Reconnecting Cities to the Biosphere: Stewardship of Green Infrastructure and Urban Ecosystem Services. AMBIO 2014, 43, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Burkhard, B.; Maes, J.; Potschin-Young, M.; Santos-Martín, F.; Geneletti, D.; Stoev, P.; Kopperoinen, L.; Adamescu, C.; Adem Esmail, B.; Arany, I.; et al. Mapping and assessing ecosystem services in the EU—Lessons learned from the ESMERALDA approach of integration. One Ecosyst. 2018, 3, e29153. [Google Scholar] [CrossRef]
- Mexia, T.; Vieira, J.; Príncipe, A.; Anjos, A.; Silva, P.; Lopes, N.; Freitas, C.; Santos-Reis, M.; Correia, O.; Branquinho, C.; et al. Ecosystem services: Urban parks under a magnifying glass. Environ. Res. 2018, 160, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Nowak, D.J.; Hirabayashi, S.; Doyle, M.; McGovern, M.; Pasher, J. Air pollution removal by urban forests in Canada and its effect on air quality and human health. Urban For. Urban Green. 2018, 29, 40–48. [Google Scholar] [CrossRef]
- Van Oudenhoven, A.P.E.; Petz, K.; Alkemade, R.; Hein, L.; de Groot, R.S. Framework for systematic indicator selection to assess effects of land management on ecosystem services. Ecol. Indic. 2012, 21, 110–122. [Google Scholar] [CrossRef]
- Van Oudenhoven, A.P.E.; Schröter, M.; Drakou, E.G.; Geijzendorffer, I.R.; Jacobs, S.; van Bodegom, P.M.; Chazee, L.; Czúcz, B.; Grunewald, K.; Lillebø, A.I.; et al. Key criteria for developing ecosystem service indicators to inform decision making. Ecol. Indic. 2018, 95, 417–426. [Google Scholar] [CrossRef]
- La Rosa, D.; Spyra, M.; Inostroza, L. Indicators of Cultural Ecosystem Services for urban planning: A review. Ecol. Indic. 2016, 61, 74–89. [Google Scholar] [CrossRef]
- Wissen Hayek, U.; Teich, M.; Klein, T.M.; Grêt-Regamey, A. Bringing ecosystem services indicators into spatial planning practice: Lessons from collaborative development of a web-based visualization platform. Ecol. Indic. 2016, 61, 90–99. [Google Scholar] [CrossRef]
- Andrea, F.; Bini, C.; Amaducci, S. Soil and ecosystem services: Current knowledge and evidences from Italian case studies. Appl. Soil Ecol. 2018, 123, 693–698. [Google Scholar] [CrossRef]
- Drobnik, T.; Greiner, L.; Keller, A.; Grêt-Regamey, A. Soil quality indicators—From soil functions to ecosystem services. Ecol. Indic. 2018, 94, 151–169. [Google Scholar] [CrossRef]
- Norton, L.; Greene, S.; Scholefield, P.; Dunbar, M. The importance of scale in the development of ecosystem service indicators? Ecol. Indic. 2016, 61, 130–140. [Google Scholar] [CrossRef]
- Aalders, I.; Stanik, N. Spatial units and scales for cultural ecosystem services: A comparison illustrated by cultural heritage and entertainment services in Scotland. Landsc. Ecol. 2019, 34, 1635–1651. [Google Scholar] [CrossRef]
- Willcock, S.; Hooftman, D.; Sitas, N.; O’Farrell, P.; Hudson, M.D.; Reyers, B.; Eigenbrod, F.; Bullock, J.M. Do ecosystem service maps and models meet stakeholders’ needs? A preliminary survey across sub-Saharan Africa. Ecosyst. Serv. 2016, 18, 110–117. [Google Scholar] [CrossRef]
- Czúcz, B.; Kalóczkai, Á.; Arany, I.; Kelemen, K.; Papp, J.; Havadtői, K.; Campbell, K.; Kelemen, M.; Vári, Á. How to design a transdisciplinary regional ecosystem service assessment: A case study from Romania, Eastern Europe. One Ecosyst. 2018, 3, e26363. [Google Scholar] [CrossRef]
- Van Reeth, W. Ecosystem Service Indicators. In Ecosystem Services; Elsevier: Amsterdam, The Netherlands, 2013; pp. 41–61. ISBN 978-0-12-419964-4. [Google Scholar]
- Alonzo, M.; Bookhagen, B.; Roberts, D.A. Urban tree species mapping using hyperspectral and lidar data fusion. Remote Sens. Environ. 2014, 148, 70–83. [Google Scholar] [CrossRef]
- Elliott, S. The potential for automating assisted natural regeneration of tropical forest ecosystems. Biotropica 2016, 48, 825–833. [Google Scholar] [CrossRef]
- Bauer, J.; Jarmer, T.; Schittenhelm, S.; Siegmann, B.; Aschenbruck, N. Processing and filtering of leaf area index time series assessed by in-situ wireless sensor networks. Comput. Electron. Agric. 2019, 165, 104867. [Google Scholar] [CrossRef]
- Mesas-Carrascosa, F.J.; Verdú Santano, D.; Meroño, J.E.; Sánchez de la Orden, M.; García-Ferrer, A. Open source hardware to monitor environmental parameters in precision agriculture. Biosyst. Eng. 2015, 137, 73–83. [Google Scholar] [CrossRef]
- Farina, A.; James, P.; Bobryk, C.; Pieretti, N.; Lattanzi, E.; McWilliam, J. Low cost (audio) recording (LCR) for advancing soundscape ecology towards the conservation of sonic complexity and biodiversity in natural and urban landscapes. Urban Ecosyst. 2014, 17, 923–944. [Google Scholar] [CrossRef]
- Mydlarz, C.; Sharma, M.; Lockerman, Y.; Steers, B.; Silva, C.; Bello, J. The Life of a New York City Noise Sensor Network. Sensors 2019, 19, 1415. [Google Scholar] [CrossRef] [PubMed]
- Valentini, R.; Belelli Marchesini, L.; Gianelle, D.; Sala, G.; Yarovslavtsev, A.; Vasenev, V.; Castaldi, S. New tree monitoring systems: From Industry 4.0 to Nature 4.0. Ann. Silvic. Res. 2019, 43, 84–88. [Google Scholar] [CrossRef]
- Vasenev, V.I.; Yaroslavtsev, A.M.; Vasenev, I.I.; Demina, S.A.; Dovltetyarova, E.A. Land-Use Change in New Moscow: First Outcomes after Five Years of Urbanization. Geogr. Environ. Sustain. 2019, 12, 24–34. [Google Scholar] [CrossRef]
- Serebryanny, L. Mixed and deciduous forests. In The Physical Geography of Northern Eurasia; Shahgedanova, M., Ed.; Oxford University: Oxford, UK, 2002; pp. 234–247. [Google Scholar]
- Lokoshchenko, M.A. Urban ‘heat island’ in Moscow. Urban Clim. 2014, 10, 550–562. [Google Scholar] [CrossRef]
- Varentsov, M.; Wouters, H.; Platonov, V.; Konstantinov, P. Megacity-Induced Mesoclimatic Effects in the Lower Atmosphere: A Modeling Study for Multiple Summers over Moscow, Russia. Atmosphere 2018, 9, 50. [Google Scholar] [CrossRef]
- Belelli Marchesini, L.; Valentini, R.; Frizzera, L.; Cavagna, M.; Chini, I.; Zampedri, R.; Gianelle, D. Impact of climate anomalies on the functionality of beech trees in a mixed forest in the Italian south-eastern Alps. In Proceedings of the EGU General Assembly, Online. 4–8 May 2020. [Google Scholar] [CrossRef]
- Valentini, R. New approaches in tree phenomics using IoT technologies and AI machine learning: The TreeTalker network. In Proceedings of the EGU General Assembly, Online. 4–8 May 2020. [Google Scholar] [CrossRef]
- Do, F.C.; Puangjumpa, N.; Rocheteau, A.; Duthoit, M.; Nhean, S.; Isarangkool Na Ayutthaya, S. Towards reduced heating duration in the transient thermal dissipation system of sap flow measurements. Acta Hortic. 2018, 149–154. [Google Scholar] [CrossRef]
- Fink, S. Hazard tree identification by visual tree assessment (VTA): Scientifically solid and practically approved. Arboric. J. 2009, 32, 139–155. [Google Scholar] [CrossRef]
- Andersson-Sköld, Y.; Klingberg, J.; Gunnarsson, B.; Cullinane, K.; Gustafsson, I.; Hedblom, M.; Knez, I.; Lindberg, F.; Ode Sang, Å.; Pleijel, H.; et al. A framework for assessing urban greenery’s effects and valuing its ecosystem services. J. Environ. Manag. 2018, 205, 274–285. [Google Scholar] [CrossRef]
- Hirabayashi, S.; Kroll, C.N.; Nowak, D.J. i-Tree Eco Dry Deposition Model Descriptions. Citeseer 2012, 36. [Google Scholar]
- Nowak, D.J.; Crane, D.E. Carbon storage and sequestration by urban trees in the USA. Environ. Pollut. 2002, 116, 381–389. [Google Scholar] [CrossRef]
- Gratani, L.; Varone, L. Carbon sequestration by Quercus ilex L. and Quercus pubescens Willd. and their contribution to decreasing air temperature in Rome. Urban Ecosyst. 2006, 9, 27–37. [Google Scholar] [CrossRef]
- Lindén, L.; Riikonen, A.; Setälä, H.; Yli-Pelkonen, V. Quantifying carbon stocks in urban parks under cold climate conditions. Urban For. Urban Green. 2020, 49, 126633. [Google Scholar] [CrossRef]
- Marando, F.; Salvatori, E.; Sebastiani, A.; Fusaro, L.; Manes, F. Regulating Ecosystem Services and Green Infrastructure: Assessment of Urban Heat Island effect mitigation in the municipality of Rome, Italy. Ecol. Model. 2019, 392, 92–102. [Google Scholar] [CrossRef]
- Krayenhoff, E.S.; Jiang, T.; Christen, A.; Martilli, A.; Oke, T.R.; Bailey, B.N.; Nazarian, N.; Voogt, J.A.; Giometto, M.G.; Stastny, A.; et al. A multi-layer urban canopy meteorological model with trees (BEP-Tree): Street tree impacts on pedestrian-level climate. Urban Clim. 2020, 32, 100590. [Google Scholar] [CrossRef]
- Morakinyo, T.E.; Ouyang, W.; Lau, K.K.-L.; Ren, C.; Ng, E. Right tree, right place (urban canyon): Tree species selection approach for optimum urban heat mitigation—Development and evaluation. Sci. Total Environ. 2020, 719, 137461. [Google Scholar] [CrossRef]
- Lee, K.H.; Ehsani, R.; Castle, W.S. A laser scanning system for estimating wind velocity reduction through tree windbreaks. Comput. Electron. Agric. 2010, 73, 1–6. [Google Scholar] [CrossRef]
- Hefny Salim, M.; Heinke Schlünzen, K.; Grawe, D. Including trees in the numerical simulations of the wind flow in urban areas: Should we care? J. Wind Eng. Ind. Aerodyn. 2015, 144, 84–95. [Google Scholar] [CrossRef]
- Kang, G.; Kim, J.-J.; Choi, W. Computational fluid dynamics simulation of tree effects on pedestrian wind comfort in an urban area. Sustain. Cities Soc. 2020, 56, 102086. [Google Scholar] [CrossRef]
- Puzachenko, Y.; Sandlersky, R.; Sankovski, A. Methods of Evaluating Thermodynamic Properties of Landscape Cover Using Multispectral Reflected Radiation Measurements by the Landsat Satellite. Entropy 2013, 15, 3970–3982. [Google Scholar] [CrossRef]
- Rana, G.; Ferrara, R.M. Air cooling by tree transpiration: A case study of Olea europaea, Citrus sinensis and Pinus pinea in Mediterranean town. Urban Clim. 2019, 29, 100507. [Google Scholar] [CrossRef]
- Deng, J.; Pickles, B.J.; Kavakopoulos, A.; Blanusa, T.; Halios, C.H.; Smith, S.T.; Shao, L. Concept and methodology of characterising infrared radiative performance of urban trees using tree crown spectroscopy. Build. Environ. 2019, 157, 380–390. [Google Scholar] [CrossRef]
- Su, Y.; Liu, L.; Wu, J.; Chen, X.; Shang, J.; Ciais, P.; Zhou, G.; Lafortezza, R.; Wang, Y.; Yuan, W.; et al. Quantifying the biophysical effects of forests on local air temperature using a novel three-layered land surface energy balance model. Environ. Int. 2019, 132, 105080. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhao, P.; Hu, Y.; Ouyang, L.; Zhu, L.; Ni, G. Canopy transpiration and its cooling effect of three urban tree species in a subtropical city- Guangzhou, China. Urban For. Urban Green. 2019, 43, 126368. [Google Scholar] [CrossRef]
- Marchionni, V.; Guyot, A.; Tapper, N.; Walker, J.P.; Daly, E. Water balance and tree water use dynamics in remnant urban reserves. J. Hydrol. 2019, 575, 343–353. [Google Scholar] [CrossRef]
- Urban, J.; Rubtsov, A.V.; Urban, A.V.; Shashkin, A.V.; Benkova, V.E. Canopy transpiration of a Larix sibirica and Pinus sylvestris forest in Central Siberia. Agric. For. Meteorol. 2019, 271, 64–72. [Google Scholar] [CrossRef]
- Zölch, T.; Henze, L.; Keilholz, P.; Pauleit, S. Regulating urban surface runoff through nature-based solutions—An assessment at the micro-scale. Environ. Res. 2017, 157, 135–144. [Google Scholar] [CrossRef]
- Pereira, F.L.; Gash, J.H.C.; David, J.S.; Valente, F. Evaporation of intercepted rainfall from isolated evergreen oak trees: Do the crowns behave as wet bulbs? Agric. For. Meteorol. 2009, 149, 667–679. [Google Scholar] [CrossRef]
- Smets, V.; Wirion, C.; Bauwens, W.; Hermy, M.; Somers, B.; Verbeiren, B. The importance of city trees for reducing net rainfall: Comparing measurements and simulations. Hydrol. Earth Syst. Sci. 2019, 23, 3865–3884. [Google Scholar] [CrossRef]
- Valente, F.; Gash, J.H.; Nóbrega, C.; David, J.S.; Pereira, F.L. Modelling rainfall interception by an olive-grove/pasture system with a sparse tree canopy. J. Hydrol. 2020, 581, 124417. [Google Scholar] [CrossRef]
- Nowak, D.J.; Crane, D.E.; Stevens, J.C. Air pollution removal by urban trees and shrubs in the United States. Urban For. Urban Green. 2006, 4, 115–123. [Google Scholar] [CrossRef]
- Sæbø, A.; Popek, R.; Nawrot, B.; Hanslin, H.M.; Gawronska, H.; Gawronski, S.W. Plant species differences in particulate matter accumulation on leaf surfaces. Sci. Total Environ. 2012, 427–428, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Intergovernmental Panel on Climate Change. Good Practice Guidance for Land Use, Land-Use Change and Forestry; IPCC, Penman, J., IPPC National Greenhouse Gas Inventories Programme, Eds.; Intergovernmental Panel on Climate Change: Kanagawa, Japan, 2003; ISBN 978-4-88788-003-0. [Google Scholar]
- Schepaschenko, D.; Moltchanova, E.; Shvidenko, A.; Blyshchyk, V.; Dmitriev, E.; Martynenko, O.; See, L.; Kraxner, F. Improved Estimates of Biomass Expansion Factors for Russian Forests. Forests 2018, 9, 312. [Google Scholar] [CrossRef]
- LeBlanc, D.C.; Foster, J.R. Predicting effects of global warming on growth and mortality of upland oak species in the midwestern United States: A physiologically based dendroecological approach. Can. J. For. Res. 1992, 22, 1739–1752. [Google Scholar] [CrossRef]
- Biondi, F.; Qeadan, F. A Theory-Driven Approach to Tree-Ring Standardization: Defining the Biological Trend from Expected Basal Area Increment. Tree-Ring Res. 2008, 64, 81–96. [Google Scholar] [CrossRef]
- Ucar, I.; Pebesma, E.; Azcorra, A. Measurement Errors in R. R J. 2018, 10, 549–557. [Google Scholar] [CrossRef]
- Granier, A. Une nouvelle méthode pour la mesure du flux de sève brute dans le tronc des arbres. Ann. Sci. For. 1985, 42, 193–200. [Google Scholar] [CrossRef]
- Do, F.C.; Isarangkool Na Ayutthaya, S.; Rocheteau, A. Transient thermal dissipation method for xylem sap flow measurement: Implementation with a single probe. Tree Physiol. 2011, 31, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Daley, M.J.; Phillips, N.G.; Pettijohn, C.; Hadley, J.L. Water use by eastern hemlock (Tsuga canadensis) and black birch (Betula lenta): Implications of effects of the hemlock woolly adelgid. Can. J. For. Res. 2007, 37, 2031–2040. [Google Scholar] [CrossRef]
- Gebauer, T.; Horna, V.; Leuschner, C. Variability in radial sap flux density patterns and sapwood area among seven co-occurring temperate broad-leaved tree species. Tree Physiol. 2008, 28, 1821–1830. [Google Scholar] [CrossRef] [PubMed]
- Thurner, M.; Beer, C.; Crowther, T.; Falster, D.; Manzoni, S.; Prokushkin, A.; Schulze, E. Sapwood biomass carbon in northern boreal and temperate forests. Glob. Ecol. Biogeogr. 2019, 28, 640–660. [Google Scholar] [CrossRef]
- Wang, X.; Liu, F.; Wang, C. Towards a standardized protocol for measuring leaf area index in deciduous forests with litterfall collection. For. Ecol. Manag. 2019, 447, 87–94. [Google Scholar] [CrossRef]
- Yan, G.; Hu, R.; Luo, J.; Weiss, M.; Jiang, H.; Mu, X.; Xie, D.; Zhang, W. Review of indirect optical measurements of leaf area index: Recent advances, challenges, and perspectives. Agric. For. Meteorol. 2019, 265, 390–411. [Google Scholar] [CrossRef]
- Monsi, M. On the Factor Light in Plant Communities and its Importance for Matter Production. Ann. Bot. 2004, 95, 549–567. [Google Scholar] [CrossRef] [PubMed]
- Neinavaz, E.; Skidmore, A.K.; Darvishzadeh, R.; Groen, T.A. Retrieval of leaf area index in different plant species using thermal hyperspectral data. ISPRS J. Photogramm. Remote Sens. 2016, 119, 390–401. [Google Scholar] [CrossRef]
- Thimonier, A.; Sedivy, I.; Schleppi, P. Estimating leaf area index in different types of mature forest stands in Switzerland: A comparison of methods. Eur. J. For. Res. 2010, 129, 543–562. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2014. [Google Scholar]
- Moser-Reischl, A.; Rahman, M.A.; Pauleit, S.; Pretzsch, H.; Rötzer, T. Growth patterns and effects of urban micro-climate on two physiologically contrasting urban tree species. Landsc. Urban Plan. 2019, 183, 88–99. [Google Scholar] [CrossRef]
- Wilkes, P.; Disney, M.; Vicari, M.B.; Calders, K.; Burt, A. Estimating urban above ground biomass with multi-scale LiDAR. Carbon Balance Manag. 2018, 13, 10. [Google Scholar] [CrossRef]
- Pretzsch, H.; Zenner, E.K. Toward managing mixed-species stands: From parametrization to prescription. For. Ecosyst. 2017, 4, 19. [Google Scholar] [CrossRef]
- Moser, A.; Rötzer, T.; Pauleit, S.; Pretzsch, H. Structure and ecosystem services of small-leaved lime (Tilia cordata Mill.) and black locust (Robinia pseudoacacia L.) in urban environments. Urban For. Urban Green. 2015, 14, 1110–1121. [Google Scholar] [CrossRef]
- Deslauriers, A.; Rossi, S.; Anfodillo, T. Dendrometer and intra-annual tree growth: What kind of information can be inferred? Dendrochronologia 2007, 25, 113–124. [Google Scholar] [CrossRef]
- Deslauriers, A.; Anfodillo, T.; Rossi, S.; Carraro, V. Using simple causal modeling to understand how water and temperature affect daily stem radial variation in trees. Tree Physiol. 2007, 27, 1125–1136. [Google Scholar] [CrossRef] [PubMed]
- Repola, J.; Hökkä, H.; Salminen, H. Models for diameter and height growth of Scots pine, Norway spruce and pubescent birch in drained peatland sites in Finland. Silva Fenn. 2018, 52, 23. [Google Scholar] [CrossRef]
- Rahman, M.A.; Stratopoulos, L.M.F.; Moser-Reischl, A.; Zölch, T.; Häberle, K.-H.; Rötzer, T.; Pretzsch, H.; Pauleit, S. Traits of trees for cooling urban heat islands: A meta-analysis. Build. Environ. 2020, 170, 106606. [Google Scholar] [CrossRef]
- Buccolieri, R.; Santiago, J.-L.; Rivas, E.; Sáanchez, B. Reprint of: Review on urban tree modelling in CFD simulations: Aerodynamic, deposition and thermal effects. Urban For. Urban Green. 2019, 37, 56–64. [Google Scholar] [CrossRef]
- Kremer, P.; Hamstead, Z.A.; McPhearson, T. The value of urban ecosystem services in New York City: A spatially explicit multicriteria analysis of landscape scale valuation scenarios. Environ. Sci. Policy 2016, 62, 57–68. [Google Scholar] [CrossRef]
- Tonyaloğlu, E.E. Spatiotemporal dynamics of urban ecosystem services in Turkey: The case of Bornova, Izmir. Urban For. Urban Green. 2020, 49, 126631. [Google Scholar] [CrossRef]
- Riikonen, A.; Järvi, L.; Nikinmaa, E. Environmental and crown related factors affecting street tree transpiration in Helsinki, Finland. Urban Ecosyst. 2016, 19, 1693–1715. [Google Scholar] [CrossRef]
- Livesley, S.J.; McPherson, E.G.; Calfapietra, C. The Urban Forest and Ecosystem Services: Impacts on Urban Water, Heat, and Pollution Cycles at the Tree, Street, and City Scale. J. Environ. Qual. 2016, 45, 119–124. [Google Scholar] [CrossRef]
- Scharenbroch, B.C.; Morgenroth, J.; Maule, B. Tree Species Suitability to Bioswales and Impact on the Urban Water Budget. J. Environ. Qual. 2016, 45, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Q.; McPherson, E.G. Surface Water Storage Capacity of Twenty Tree Species in Davis, California. J. Environ. Qual. 2016, 45, 188–198. [Google Scholar] [CrossRef] [PubMed]
- Prasad Ghimire, C.; Adrian Bruijnzeel, L.; Lubczynski, M.W.; Ravelona, M.; Zwartendijk, B.W.; van Meerveld, H.J. (Ilja) Measurement and modeling of rainfall interception by two differently aged secondary forests in upland eastern Madagascar. J. Hydrol. 2017, 545, 212–225. [Google Scholar] [CrossRef]
- Syrbe, R.-U.; Schorcht, M.; Grunewald, K.; Meinel, G. Indicators for a nationwide monitoring of ecosystem services in Germany exemplified by the mitigation of soil erosion by water. Ecol. Indic. 2018, 94, 46–54. [Google Scholar] [CrossRef]
- Bremer, M.; Wichmann, V.; Rutzinger, M. Calibration and Validation of a Detailed Architectural Canopy Model Reconstruction for the Simulation of Synthetic Hemispherical Images and Airborne LiDAR Data. Remote Sens. 2017, 9, 220. [Google Scholar] [CrossRef]
- Taheriazad, L.; Moghadas, H.; Sanchez-Azofeifa, A. Calculation of leaf area index in a Canadian boreal forest using adaptive voxelization and terrestrial LiDAR. Int. J. Appl. Earth Obs. Geoinf. 2019, 83, 101923. [Google Scholar] [CrossRef]
- Zhu, X.; Skidmore, A.K.; Wang, T.; Liu, J.; Darvishzadeh, R.; Shi, Y.; Premier, J.; Heurich, M. Improving leaf area index (LAI) estimation by correcting for clumping and woody effects using terrestrial laser scanning. Agric. For. Meteorol. 2018, 263, 276–286. [Google Scholar] [CrossRef]
- Klingberg, J.; Konarska, J.; Lindberg, F.; Johansson, L.; Thorsson, S. Mapping leaf area of urban greenery using aerial LiDAR and ground-based measurements in Gothenburg, Sweden. Urban For. Urban Green. 2017, 26, 31–40. [Google Scholar] [CrossRef]
- Wang, R.; Chen, J.M.; Luo, X.; Black, A.; Arain, A. Seasonality of leaf area index and photosynthetic capacity for better estimation of carbon and water fluxes in evergreen conifer forests. Agric. For. Meteorol. 2019, 279, 107708. [Google Scholar] [CrossRef]
- Muhammad, S.; Wuyts, K.; Samson, R. Atmospheric net particle accumulation on 96 plant species with contrasting morphological and anatomical leaf characteristics in a common garden experiment. Atmos. Environ. 2019, 202, 328–344. [Google Scholar] [CrossRef]
- Bottalico, F.; Chirici, G.; Giannetti, F.; De Marco, A.; Nocentini, S.; Paoletti, E.; Salbitano, F.; Sanesi, G.; Serenelli, C.; Travaglini, D. Air Pollution Removal by Green Infrastructures and Urban Forests in the City of Florence. Agric. Agric. Sci. Procedia 2016, 8, 243–251. [Google Scholar] [CrossRef]
- Selmi, W.; Weber, C.; Rivière, E.; Blond, N.; Mehdi, L.; Nowak, D. Air pollution removal by trees in public green spaces in Strasbourg city, France. Urban For. Urban Green. 2016, 17, 192–201. [Google Scholar] [CrossRef]
- Song, X.P.; Tan, P.Y.; Edwards, P.; Richards, D. The economic benefits and costs of trees in urban forest stewardship: A systematic review. Urban For. Urban Green. 2018, 29, 162–170. [Google Scholar] [CrossRef]
- Müller, F.; Burkhard, B. The indicator side of ecosystem services. Ecosyst. Serv. 2012, 1, 26–30. [Google Scholar] [CrossRef]
- Doser, J.W.; Finley, A.O.; Kasten, E.P.; Gage, S.H. Assessing soundscape disturbance through hierarchical models and acoustic indices: A case study on a shelterwood logged northern Michigan forest. Ecol. Indic. 2020, 113, 106244. [Google Scholar] [CrossRef]
- Margaritis, E.; Kang, J.; Filipan, K.; Botteldooren, D. The influence of vegetation and surrounding traffic noise parameters on the sound environment of urban parks. Appl. Geogr. 2018, 94, 199–212. [Google Scholar] [CrossRef]
- Nitoslawski, S.A.; Galle, N.J.; Van Den Bosch, C.K.; Steenberg, J.W.N. Smarter ecosystems for smarter cities? A review of trends, technologies, and turning points for smart urban forestry. Sustain. Cities Soc. 2019, 51, 101770. [Google Scholar] [CrossRef]
- Schröter, M.; Kraemer, R.; Mantel, M.; Kabisch, N.; Hecker, S.; Richter, A.; Neumeier, V.; Bonn, A. Citizen science for assessing ecosystem services: Status, challenges and opportunities. Ecosyst. Serv. 2017, 28, 80–94. [Google Scholar] [CrossRef]
- Cortinovis, C.; Geneletti, D. A framework to explore the effects of urban planning decisions on regulating ecosystem services in cities. Ecosyst. Serv. 2019, 38, 100946. [Google Scholar] [CrossRef]
- Bodnaruk, E.W.; Kroll, C.N.; Yang, Y.; Hirabayashi, S.; Nowak, D.J.; Endreny, T.A. Where to plant urban trees? A spatially explicit methodology to explore ecosystem service tradeoffs. Landsc. Urban Plan. 2017, 157, 457–467. [Google Scholar] [CrossRef]
- Speak, A.; Escobedo, F.J.; Russo, A.; Zerbe, S. An ecosystem service-disservice ratio: Using composite indicators to assess the net benefits of urban trees. Ecol. Indic. 2018, 95, 544–553. [Google Scholar] [CrossRef]
- Teixeira, F.Z.; Bachi, L.; Blanco, J.; Zimmermann, I.; Welle, I.; Carvalho-Ribeiro, S.M. Perceived ecosystem services (ES) and ecosystem disservices (EDS) from trees: Insights from three case studies in Brazil and France. Landsc. Ecol. 2019, 34, 1583–1600. [Google Scholar] [CrossRef]
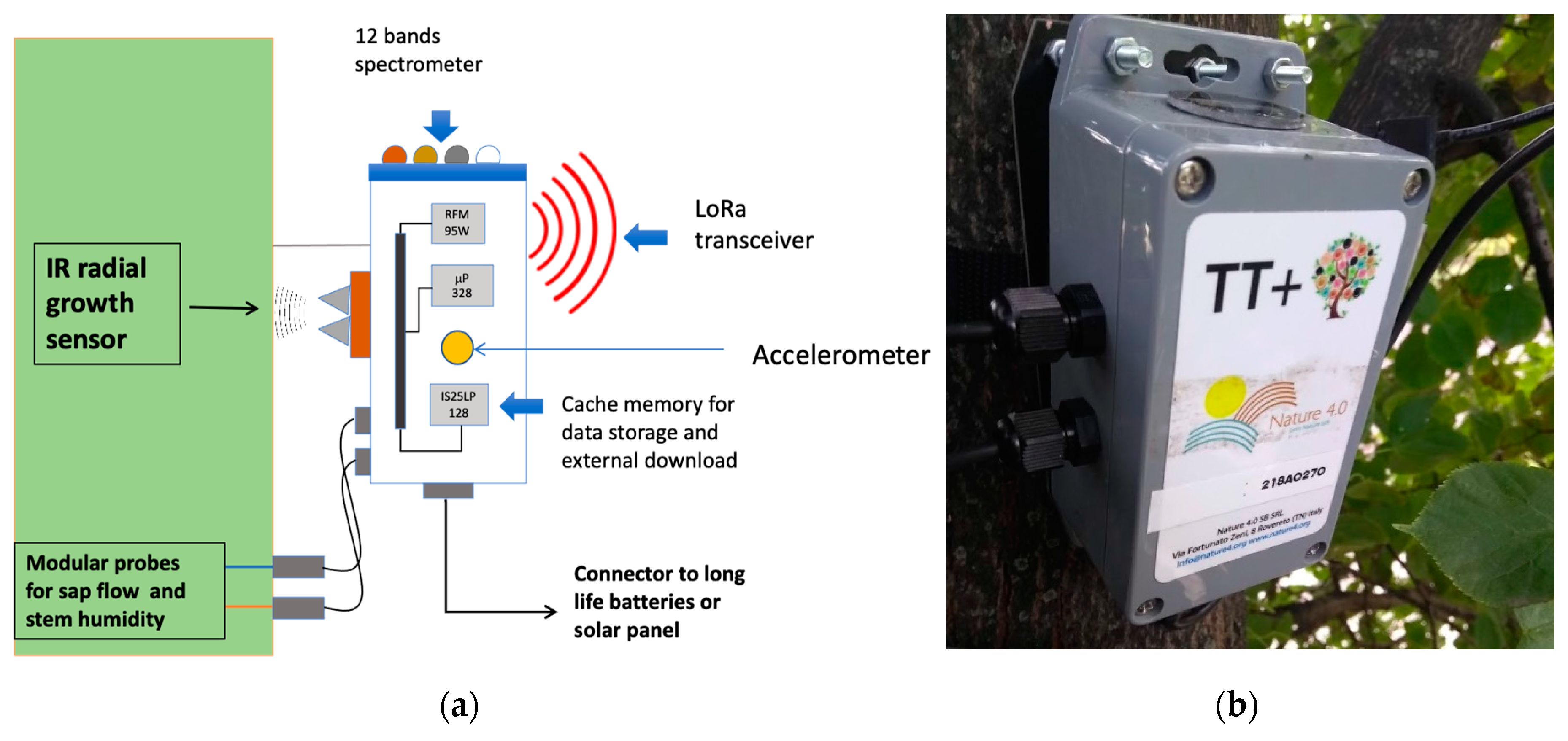
| Sensor | Range | Accuracy |
|---|---|---|
| Accelerometer | 0–360° (0–8g) | ±0.01° |
| Diameter growth sensor | 0–1 cm | ±200 µm |
| Temperature probes | −40–+40 °C | ±0.1 °C |
| Stem humidity probe | 0–100% | ±2% v/v (resolution, accuracy under investigation) |
| Visible spectrometer | 400–700 nm | ±5 nm peak ±20 nm half bandwidth (450, 500, 550, 570, 600, 650 nm) |
| Near-infrared spectrometer | 700–900 nm | ±5 nm peak ±10 nm Half Bandwidth (HBW) (610, 680, 730, 760, 810, 860 nm) |
| Air and humidity sensor | −10–+85 0–100% | ±1 °C ±5% |
| ES Group | Type of ES | Indicator | Sensor | Type of Equation | Units | Key References |
|---|---|---|---|---|---|---|
| Global climate regulation | Carbon sequestration | Tree growth rate | IR growth sensor | Indirect Biomass expansion factors | kg C | [47,48,49] |
| Local climate regulation | Climate comfort regulation | Air temperature | Thermo-hygrometer sensor | Direct | C degrees | [50,51,52] |
| Wind velocity | Spectrometer | Indirect LAI | m s−1 | [53,54,55] | ||
| Energy balance regulation | Latent energy via transpiration | Sap-flow sensors | Direct | W m−2 | [56,57,58,59], | |
| Water regulation | Run-off mitigation | Transpiration | Sap-flow sensors | Direct | l hr−1 or mm | [60,61,62,63] |
| Rain buffer | Spectrometer | Indirect LAI | % | [64,65,66] | ||
| Air quality regulation | Particulate adsorption | PM removal | Spectrometer | Indirect LAI | g m−2 | [18,46,67,68] |
| Gas regulation | Gaseous pollutants removal | Spectrometer | Indirect LAI | g m−2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matasov, V.; Belelli Marchesini, L.; Yaroslavtsev, A.; Sala, G.; Fareeva, O.; Seregin, I.; Castaldi, S.; Vasenev, V.; Valentini, R. IoT Monitoring of Urban Tree Ecosystem Services: Possibilities and Challenges. Forests 2020, 11, 775. https://doi.org/10.3390/f11070775
Matasov V, Belelli Marchesini L, Yaroslavtsev A, Sala G, Fareeva O, Seregin I, Castaldi S, Vasenev V, Valentini R. IoT Monitoring of Urban Tree Ecosystem Services: Possibilities and Challenges. Forests. 2020; 11(7):775. https://doi.org/10.3390/f11070775
Chicago/Turabian StyleMatasov, Victor, Luca Belelli Marchesini, Alexey Yaroslavtsev, Giovanna Sala, Olga Fareeva, Ivan Seregin, Simona Castaldi, Viacheslav Vasenev, and Riccardo Valentini. 2020. "IoT Monitoring of Urban Tree Ecosystem Services: Possibilities and Challenges" Forests 11, no. 7: 775. https://doi.org/10.3390/f11070775
APA StyleMatasov, V., Belelli Marchesini, L., Yaroslavtsev, A., Sala, G., Fareeva, O., Seregin, I., Castaldi, S., Vasenev, V., & Valentini, R. (2020). IoT Monitoring of Urban Tree Ecosystem Services: Possibilities and Challenges. Forests, 11(7), 775. https://doi.org/10.3390/f11070775







