Vitex agnus-castus L.: Main Features and Nutraceutical Perspectives
Abstract
1. Introduction
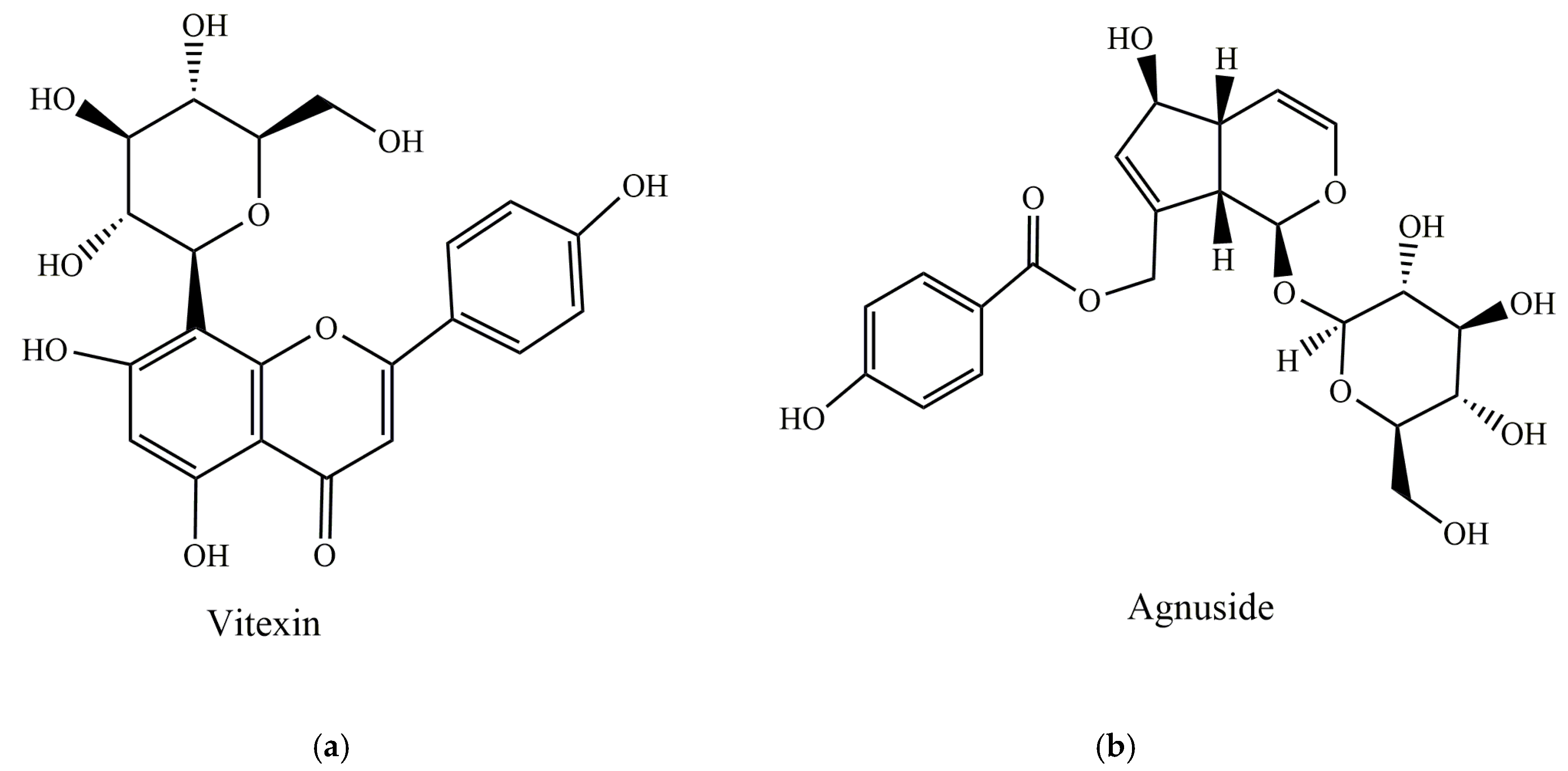
2. Main Substances of Nutraceutical Interest in Vitex agnus-castus
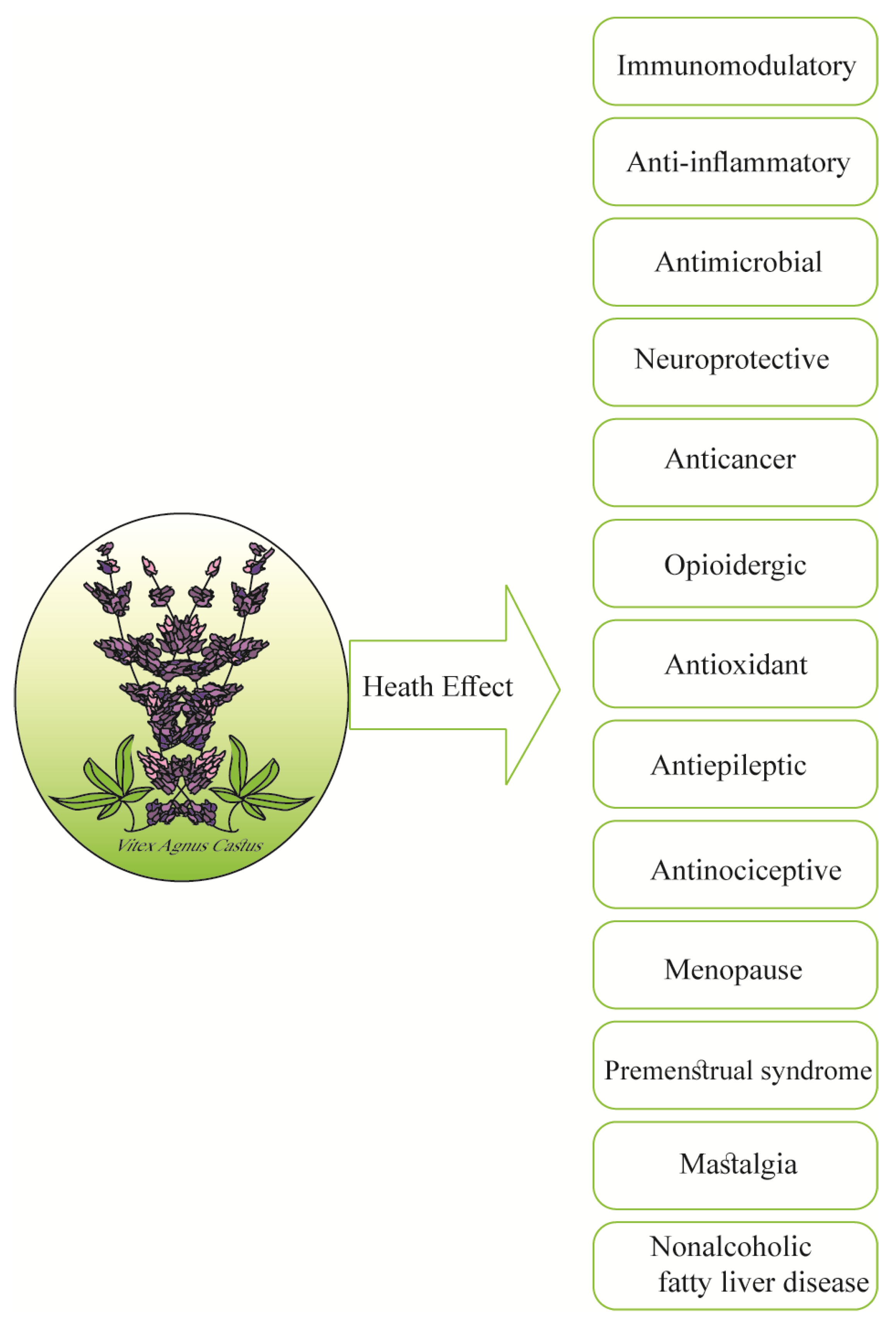
3. An Updated Snapshot of In Vitro and In Vivo Studies on Vitex agnus-castus
3.1. Health-Promoting Activities of Vitex agnus-castus L. In Vitro
3.2. Health-Promoting Activities of Vitex agnus-castus L. in Animals
3.3. Health-Promoting Activities of Vitex agnus-castus L. in Humans, with Particular Regard to Clinical Trials
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Yeung, A.W.K.; Heinrich, M.; Kijjoa, A.; Tzvetkov, N.T.; Atanasov, A.G. The ethnopharmacological literature: An analysis of the scientific landscape. J. Ethnopharmacol. 2020, 250, 112414. [Google Scholar] [CrossRef] [PubMed]
- Santini, A.; Novellino, E. Nutraceuticals: Beyond the diet before the drugs. Curr. Bioact. Compd. 2014, 10, 1–12. [Google Scholar] [CrossRef]
- Durazzo, A. Extractable and Non-extractable Polyphenols: An Overview. In Non-Extractable Polyphenols and Carotenoids: Importance in Human Nutrition and Health; Saura-Calixto, F., Pérez-Jiménez, J., Eds.; Royal Society of Chemistry: London, UK, 2018. [Google Scholar]
- Durazzo, A.; Lucarini, M. A current shot and re-thinking of antioxidant research strategy. Braz. J. Anal. Chem. 2018, 5, 9–11. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M. Extractable and non-extractable antioxidants. Molecules 2019, 24, 1993. [Google Scholar] [CrossRef] [PubMed]
- Durazzo, A.; Lucarini, M.; Kiefer, J.; Mahesar, S.A. State-of-the-Art Infrared Applications in Drugs, Dietary Supplements, and Nutraceuticals. J. Spectrosc. 2020, 2020, 1397275. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M. The State of Science and Innovation of Bioactive Research and Applications, Health and Diseases. Front. Nutr. 2019, 6, 178. [Google Scholar] [CrossRef]
- Santini, A.; Novellino, E.; Armini, V.; Ritieni, A. State of the art of Ready-to-Use Therapeutic Food: A tool for nutraceuticals addition to foodstuff. Food Chem. 2013, 140, 843–849. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M.; Novellino, E.; Souto, E.B.; Daliu, P.; Santini, A. Abelmoschus esculentus (L.): Bioactive Components’ Beneficial Properties—Focused on Antidiabetic Role—For Sustainable Health Applications. Molecules 2019, 24, 38. [Google Scholar] [CrossRef]
- Lucarini, M.; Durazzo, A.; Kiefer, J.; Santini, A.; Lombardi-Boccia, G.; Souto, E.B.; Romani, A.; Lampe, A.; Ferrari Nicoli, S.; Gabrielli, P. Grape Seeds: Chromatographic Profile of Fatty Acids and Phenolic Compounds and Qualitative Analysis by FTIR-ATR Spectroscopy. Foods 2020, 9, 10. [Google Scholar] [CrossRef]
- Salehi, B.; Venditti, A.; Sharifi-Rad, M.; Kręgiel, D.; Sharifi-Rad, J.; Durazzo, A.; Lucarini, M.; Santini, A.; Souto, E.B.; Novellino, E. The therapeutic potential of apigenin. Int. J. Mol. Sci. 2019, 20, 1305. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M.; Souto, E.B.; Cicala, C.; Caiazzo, E.; Izzo, A.A.; Novellino, E.; Santini, A. Polyphenols: A concise overview on the chemistry, occurrence, and human health. Phytother. Res. 2019, 33, 2221–2243. [Google Scholar] [CrossRef]
- Abenavoli, L.; Izzo, A.A.; Milić, N.; Cicala, C.; Santini, A.; Capasso, R. Milk thistle (Silybum marianum): A concise overview on its chemistry, pharmacological, and nutraceutical uses in liver diseases. Phytother. Res. 2018, 32, 2202–2213. [Google Scholar] [CrossRef] [PubMed]
- Santini, A.; Tenore, G.C.; Novellino, E. Nutraceuticals: A paradigm of proactive medicine. Eur. J. Pharm. Sci. 2017, 96, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Daliu, P.; Santini, A.; Novellino, E. A decade of nutraceutical patents: Where are we now in 2018? Expert Opin. Ther. Pat. 2018, 28, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Santini, A.; Novellino, E. Nutraceuticals-shedding light on the grey area between pharmaceuticals and food. Expert Rev. Clin. Pharmacol. 2018, 11, 545–547. [Google Scholar] [CrossRef]
- Bircher, J.; Hahn, E.G. Understanding the nature of health: New perspectives for medicine and public health. Improved wellbeing at lower costs: New Perspectives for Medicine and Public Health: Improved Wellbeing at lower Cost. F1000Research 2016, 5, 167. [Google Scholar] [CrossRef]
- Santini, A.; Cammarata, S.M.; Capone, G.; Ianaro, A.; Tenore, G.C.; Pani, L.; Novellino, E. Nutraceuticals: Opening the debate for a regulatory framework. Br. J. Clin. Pharmacol. 2018, 84, 659–672. [Google Scholar] [CrossRef]
- Daliu, P.; Santini, A.; Novellino, E. From pharmaceuticals to nutraceuticals: Bridging disease prevention and management. Expert Rev. Clin. Pharmacol. 2019, 12, 1–7. [Google Scholar] [CrossRef]
- Durazzo, A.; D’Addezio, L.; Camilli, E.; Piccinelli, R.; Turrini, A.; Marletta, L.; Marconi, S.; Lucarini, M.; Lisciani, S.; Gabrielli, P. From plant compounds to botanicals and back: A current snapshot. Molecules 2018, 23, 1844. [Google Scholar] [CrossRef]
- Durazzo, A.; Camilli, E.; D’Addezio, L.; Piccinelli, R.; Mantur-Vierendeel, A.; Marletta, L.; Finglas, P.; Turrini, A.; Sette, S. Development of Dietary Supplement Label Database in Italy: Focus of FoodEx2 Coding. Nutrients 2020, 12, 89. [Google Scholar] [CrossRef]
- Khasim, S.; Long, C.; Thammasiri, K.; Lutken, H. Medicinal Plants: Biodiversity, Sustainable Utilization and Conservation; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar] [CrossRef]
- Orbe, I.; Paz, D.; Pejenaute, L.; Puente, A.; de Alda, L.D.; Yague, S.; Lete, I. Medicinal Herbs: Its Therapeutic Use in Obstetrics and Gynaecology. In Approaching Complex Diseases: Network-Based Pharmacology and Systems Approach in Bio-Medicine; Bizzarri, M., Ed.; Springer International Publishing: Berlin/Heidelberg, Germany, 2020; pp. 437–455. [Google Scholar] [CrossRef]
- Rani, A.; Sharma, A. The genus Vitex: A review. Pharmacogn. Rev. 2013, 7, 188. [Google Scholar] [CrossRef] [PubMed]
- Köngül, E. Agnus Castus. In Nonvitamin and Nonmineral Nutritional Supplements, 1st ed.; Nabavi, S., Silva, A.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 139–143. [Google Scholar]
- Roemheld-Hamm, B. Chasteberry. Am. Fam. Physician 2005, 72, 821–824. [Google Scholar] [PubMed]
- King, S.R. Medicinal Plants of the World Volume 2: Chemical Constituents, Traditional and Modern Medicinal Uses by Ivan, A. Ross (U.S. Food and Drug Administration). Humana Press, Inc., Totowa, NJ. J. Nat. Prod. 2002, 65, 1085. [Google Scholar] [CrossRef]
- Niroumand, M.C.; Heydarpour, F.; Farzaei, M.H. Pharmacological and Therapeutic Effects of Vitex agnus-castus L.: A Review. Pharmacogn. Rev. 2018, 12, 103–114. [Google Scholar]
- Girman, A.; Lee, R.; Kligler, B. An integrative medicine approach to premenstrual syndrome. Am. J. Obstet. Gynecol. 2003, 188, S56–S65. [Google Scholar] [CrossRef]
- Chan, E.W.C.; Wong, S.K.; Chan, H.T. Casticin from Vitex species: A short review on its anticancer and anti-inflammatory properties. J. Integr. Med. 2018, 16, 147–152. [Google Scholar] [CrossRef]
- Mari, A.; Montoro, P.; D’Urso, G.; Macchia, M.; Pizza, C.; Piacente, S. Metabolic profiling of Vitex agnus castus leaves, fruits and sprouts: Analysis by LC/ESI/(QqQ) MS and (HR) LC/ESI/(Orbitrap)/MSn. J. Pharm. Biomed. Anal. 2015, 102, 215–221. [Google Scholar] [CrossRef]
- Nigam, M.; Saklani, S.; Plygun, S.; Mishra, A.P. Antineoplastic potential of the Vitex species: An overview. Bol. Latinoam. Caribe Plantas Med. Aromát. 2018, 17, 492–502. [Google Scholar]
- Al Saka, F.; Daghestani, M.; Karabet, F. Composition and Antioxidant Activity of Vitex agnus-castus L. and Rosmarinus Officinalis, L. Leaves Essential Oils Cultivated in Syria. SM Anal Bioanal Tech. 2017, 2, 1010. [Google Scholar] [CrossRef]
- Heskes, A.M.; Sundram, T.C.; Boughton, B.A.; Jensen, N.B.; Hansen, N.L.; Crocoll, C.; Cozzi, F.; Rasmussen, S.; Hamberger, B.; Hamberger, B. Biosynthesis of bioactive diterpenoids in the medicinal plant Vitex agnus-castus. Plant J. 2018, 93, 943–958. [Google Scholar] [CrossRef]
- Kırmızıbekmez, H.; Demir, D. Iridoid Glycosides and Phenolic Compounds from the Flowers of Vitex agnus-castus. Helv. Chim. Acta 2016, 99, 518–522. [Google Scholar] [CrossRef]
- Rajić, M.; Molnar, M.; Bilić, M.; Jokić, S. The impact of extraction methods on isolation of pharmacologically active compounds from Vitex agnus-castus-a review. Int. J. Pharm. Res. Allied Sci. 2016, 5, 15–21. [Google Scholar]
- Zahid, H.; Rizwani, G.H.; Ishaqe, S. Phytopharmacological review on Vitex agnus-castus: A potential medicinal plant. Chin. Herb. Med. 2016, 8, 24–29. [Google Scholar] [CrossRef]
- Shah, S.; Dhanani, T.; Kumar, S. Validated HPLC method for identification and quantification of p-hydroxy benzoic acid and agnuside in Vitex negundo and Vitex trifolia. J. Pharm. Anal. 2013, 3, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Gökbulut, A.; Özhan, O.; Karacaoğlu, M.; Şarer, E. Radical scavenging activity and vitexin content of Vitex agnus-castus leaves and fruits. FABAD J. Pharm. Sci. 2010, 35, 85–91. [Google Scholar]
- Sogame, M.; Naraki, Y.; Sasaki, T.; Seki, M.; Yokota, K.; Masada, S.; Hakamatsuka, T. Quality Assessment of Medicinal Product and Dietary Supplements Containing Vitex agnus-castus by HPLC Fingerprint and Quantitative Analyses. Chem. Pharm. Bull. 2019, 67, 527–533. [Google Scholar] [CrossRef]
- Yahagi, T.; Masada, S.; Oshima, N.; Suzuki, R.; Matsufuji, H.; Takahashi, Y.; Watanabe, M.; Yahara, S.; Iida, O.; Kawahara, N. Determination and identification of a specific marker compound for discriminating Shrub Chaste Tree Fruit from Agnus Castus Fruit based on LC/MS metabolic analysis. Chem. Pharm. Bull. 2016, 64, 305–310. [Google Scholar] [CrossRef]
- Högner, C.; Sturm, S.; Seger, C.; Stuppner, H. Development and validation of a rapid ultra-high performance liquid chromatography diode array detector method for Vitex agnus-castus. J. Chromatogr. B 2013, 927, 181–190. [Google Scholar] [CrossRef]
- Mari, A.; Montoro, P.; Pizza, C.; Piacente, S. Liquid chromatography tandem mass spectrometry determination of chemical markers and principal component analysis of Vitex agnus-castus L. fruits (Verbenaceae) and derived food supplements. J. Pharm. Biomed. Anal. 2012, 70, 224–230. [Google Scholar] [CrossRef]
- Şarer, E.; Gökbulut, A. Determination of caffeic and chlorogenic acids in the leaves and fruits of Vitex agnus-castus. Turk J. Pharm. Sci. 2008, 5, 167–174. [Google Scholar]
- Li, S.; Qiu, S.; Yao, P.; Sun, H.; Fong, H.H.; Zhang, H. Compounds from the fruits of the popular European medicinal plant Vitex agnus-castus in chemoprevention via NADP (H): Quinone oxidoreductase type 1 induction. Evid.-Based Complement. Altern. Med. 2013, 2013, 432829. [Google Scholar] [CrossRef]
- Eryigit, T.; Çig, A.; Okut, N.; Yildirim, B.; Ekici, K. Evaluation of chemical composition and antimicrobial activity of Vitex agnus castus L. fruits’ essential oils from west Anatolia, Turkey. J. Essent. 2015, 18, 208–214. [Google Scholar]
- Ono, M.; Eguchi, K.; Konoshita, M.; Furusawa, C.; Sakamoto, J.; Yasuda, S.; Ikeda, T.; Okawa, M.; Kinjo, J.; Yoshimitsu, H. A new diterpenoid glucoside and two new diterpenoids from the fruit of Vitex agnus-castus. Chem. Pharm. Bull. 2011, 59, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Onaran, A. In Vitro antifungal activities of some plant extracts against plant pathogenic fungi in Turkey. Egypt. J. Biol. Pest Control 2016, 26, 111. [Google Scholar]
- Kikuchi, H.; Yuan, B.; Yuhara, E.; Imai, M.; Furutani, R.; Fukushima, S.; Hazama, S.; Hirobe, C.; Ohyama, K.; Takagi, N. Involvement of histone H3 phosphorylation via the activation of p38 MAPK pathway and intracellular redox status in cytotoxicity of HL-60 cells induced by Vitex agnus-castus fruit extract. Int. J. Oncol. 2014, 45, 843–852. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Abdel-Lateef, E.E.-S.; Hammam, O.A.; Mahmoud, F.S.; Atta, S.A.; El-Sayed, M.M.; Hassenein, H.I. Induction of apoptosis in HepG2 by Vitex agnus-castus L. leaves extracts and identification of their active chemical constituents by LC-ESI-MS. Asian Pac. J. Trop. Dis. 2016, 6, 539–548. [Google Scholar] [CrossRef]
- Sa, A.; Kb, P.; Pa, V. Free radical scavenging and in vitro cytotoxicity activity of agnuside from Vitex agnus castus (Verbenacae). J. Pharm. Res. 2012, 5, 2548–2552. [Google Scholar]
- Imai, M.; Yuan, B.; Kikuchi, H.; Saito, M.; Ohyama, K.; Hirobe, C.; Oshima, T.; Hosoya, T.; Morita, H.; Toyoda, H. Growth inhibition of a human colon carcinoma cell, COLO 201, by a natural product, Vitex agnus-castus fruits extract, in vivo and in vitro. Adv. Biol. Chem. 2012, 2, 20–28. [Google Scholar] [CrossRef]
- Ribat, Z.W.; Sahib, H.B.; Al-Shammari, A.M. Effect of vitex agnus castus fruits methanol extract against murine mammary adenocarcinoma cell line (amn3) and rat embryonic fibroblast normal cell line (ref). Int. J. Res. Pharm. Sci. 2019, 10, 1910–1913. [Google Scholar]
- Imai, M.; Kikuchi, H.; Yuan, B.; Aihara, Y.; Mizokuchi, A.; Ohya-Ma, K.; Hirobe, C.; Toyoda, H. Enhanced growth inhibitory effect of 5-fluorouracil in combination with Vitex agnus-castus fruits extract against a human colon adenocarcinoma cell line, COLO 201. J. Chin. Clin. Med. 2011, 6, 14–19. [Google Scholar]
- Mesaik, M.A.; Murad, S.; Khan, K.M.; Tareen, R.B.; Ahmed, A.; Choudhary, M.I. Isolation and immunomodulatory properties of a flavonoid, casticin from Vitex agnus-castus. Phytother. Res. 2009, 23, 1516–1520. [Google Scholar] [CrossRef] [PubMed]
- Sarac, N.; Ugur, A.; Sen, B. In vitro antimutagenic activity of Vitex agnus-castus L. essential oils and ethanolic extracts. Ind. Crop. Prod. 2015, 63, 100–103. [Google Scholar] [CrossRef]
- Hajdú, Z.; Hohmann, J.; Forgo, P.; Martinek, T.; Dervarics, M.; Zupkó, I.; Falkay, G.; Cossuta, D.; Máthé, I. Diterpenoids and flavonoids from the fruits of Vitex agnus-castus and antioxidant activity of the fruit extracts and their constituents. Phytother. Res. 2007, 21, 391–394. [Google Scholar] [CrossRef] [PubMed]
- Marongiu, B.; Piras, A.; Porcedda, S.; Falconieri, D.; Gonçalves, M.J.; Salgueiro, L.; Maxia, A.; Lai, R. Extraction, separation and isolation of volatiles from Vitex agnus-castus L.(Verbenaceae) wild species of Sardinia, Italy, by supercritical CO2. Nat. Prod. Res. 2010, 24, 569–579. [Google Scholar] [CrossRef]
- Ahmad, B.; Hafeez, N.; Ara, G.; Azam, S.; Bashir, S.; Khan, I. Antibacterial activity of crude methanolic extract and various fractions of Vitex agnus castus and Myrsine africana against clinical isolates of Methicillin Resistant Staphylococcus aureus. Pak. J. Pharm. Sci. 2016, 29, 1977–1983. [Google Scholar] [PubMed]
- Asdadi, A.; Idrissi Hassani, L.; Chebli, B.; Moutaj, R.; Gharby, S.; Harhar, H.; Salghi, R.; El Hadek, M. Chemical composition and antifungal activity of vitex agnus-castus L. seeds oil growing in Morocco. J. Mater. Environ. Sci. 2014, 5, 823–830. [Google Scholar]
- Ağalar, H.G.; Çiftçi, G.A.; Gögera, F.; Kιrιmera, N. The LC/ESI-MSMS Profiles and Biological Potentials of Vitex agnus castus Extracts. Nat. Prod. Commun. 2016, 11, 1655–1660. [Google Scholar] [CrossRef]
- Kikuchi, H.; Yuan, B.; Nishimura, Y.; Imai, M.; Furutani, R.; Kamoi, S.; Seno, M.; Fukushima, S.; Hazama, S.; Hirobe, C. Cytotoxicity of Vitex agnus-castus fruit extract and its major component, casticin, correlates with differentiation status in leukemia cell lines. Int. J. Oncol. 2013, 43, 1976–1984. [Google Scholar] [CrossRef] [PubMed]
- Sahib, H.B.; Al-Zubaidy, A.A.; Hussein, S.M.; Dahham, S.S.; Al-Suede, F.S.; Shah, A.M. The Anti-proliferative Activity of Vitex agnus-castus Leaves Methanol Extract against Breast and Prostate Cancer Cell Line. Am. J. Phyto Clin. Ther. 2015, 3, 2321–2748. [Google Scholar]
- Habbab, A.; Sekkoum, K.; Belboukhari, N.; Cheriti, A.Y.; Aboul-Enein, H. Essential oil chemical composition of Vitex agnus-castus L. from Southern-West Algeria and its antimicrobial activity. Curr. Bioact. Compd. 2016, 12, 51–60. [Google Scholar] [CrossRef]
- Ghannadi, A.; Bagherinejad, M.; Abedi, D.; Jalali, M.; Absalan, B.; Sadeghi, N. Antibacterial activity and composition of essential oils from Pelargonium graveolens L’Her and Vitex agnus-castus L. Iran. J. Microbiol. 2012, 4, 171. [Google Scholar] [PubMed]
- Afarin, H.; Dakhili, M.; Zolfaghari, M.R. Comparison of antimicrobioal effect of essential oil of Vitex agnus-castus with common antibiotics invitro. Qom Uni. Med. Sci. J. 2015, 9, 12–19. [Google Scholar]
- Katiraee, F.; Mahmoudi, R.; Tahapour, K.; Hamidian, G.; Emami, S.J. Biological properties of Vitex agnus-castus essential oil (Phytochemical component, antioxidant and antifungal activity). Biotechnol. Health Sci. 2015, 2, e26797. [Google Scholar] [CrossRef]
- Yilar, M.; Bayan, Y.; Onaran, A. Chemical composition and antifungal effects of Vitex agnus-castus L. and Myrtus communis L. Plants. Not. Bot. Horti Agrobot. Cluj-Napoca 2016, 44, 466–471. [Google Scholar] [CrossRef]
- Stojković, D.; Soković, M.; Glamočlija, J.; Džamić, A.; Ćirić, A.; Ristić, M.; Grubišić, D. Chemical composition and antimicrobial activity of Vitex agnus-castus L. fruits and leaves essential oils. Food Chem. 2011, 128, 1017–1022. [Google Scholar] [CrossRef]
- Arokiyaraj, S.; Perinbam, K.; Agastian, P.; Kumar, R.M. Phytochemical analysis and antibacterial activity of Vitex agnus-castus. Int. J. Green Pharm. (IJGP) 2009, 3, 162–164. [Google Scholar] [CrossRef]
- Goncalves, R.; Ayres, V.F.; Carvalho, C.E.; Souza, M.G.; Guimaraes, A.C.; Correa, G.M.; Martins, C.H.; Takeara, R.; Silva, E.O.; Crotti, A.E. Chemical composition and antibacterial activity of the essential oil of Vitex agnus-castus L.(Lamiaceae). An. Acad. Bras. Ciências 2017, 89, 2825–2832. [Google Scholar] [CrossRef]
- Keikha, N.; Shafaghat, M.; Mousavia, S.M.; Moudi, M.; Keshavarzi, F. Antifungal effects of ethanolic and aqueous extracts of Vitex agnus-castus against vaginal isolates of Candida albicans. Curr. Med. Mycol. 2018, 4, 1. [Google Scholar] [CrossRef]
- Asdadi, A.; Hamdouch, A.; Oukacha, A.; Moutaj, R.; Gharby, S.; Harhar, H.; El Hadek, M.; Chebli, B.; Hassani, L.I. Study on chemical analysis, antioxidant and in vitro antifungal activities of essential oil from wild Vitex agnus-castus L. seeds growing in area of Argan Tree of Morocco against clinical strains of Candida responsible for nosocomial infections. J. Mycol. Med. 2015, 25, e118–e127. [Google Scholar] [CrossRef]
- Sultan Aslantürk, Ö.; Aşkın Çelik, T. Antioxidant activity and anticancer effect of Vitex agnus-castus L.(Verbenaceae) seed extracts on MCF–7 breast cancer cells. J. Biomol. Struct. 2013, 66, 257–267. [Google Scholar]
- Rashed, K.N. Antioxidant activity of different extracts of Vitex agnus-castus (L.) and phytochemical profile. Res. Pharm. 2013, 3, 1–5. [Google Scholar]
- Sağlam, H.; Pabuçcuoğlu, A.; Kıvçak, B. Antioxidant activity of Vitex agnus-castus L. extracts. Phytother. Res. 2007, 21, 1059–1060. [Google Scholar]
- Maltaş, E.; Uysal, A.; Yildiz, S.; Durak, Y. Evaluation of antioxidant and antimicrobial activity of Vitex agnus-castus L. Fresenius Environ. Bull. 2010, 19, 3094–3099. [Google Scholar]
- Dugoua, J.-J.; Seely, D.; Perri, D.; Koren, G.; Mills, E. Safety and efficacy of chastetree (Vitex agnus-castus) during pregnancy and lactation. J. Popul. Ther. Clin. Pharmacol. 2008, 15, 74–79. [Google Scholar]
- Ahmad, B.; Azam, S.; Bashir, S.; Adhikari, A.; Choudhary, M.I. Biological activities of a new compound isolated from the aerial parts of Vitex agnus castus L. Afr. J. Biotechnol. 2010, 9, 9063–9069. [Google Scholar]
- Choudhary, M.I.; Jalil, S.; Nawaz, S.A.; Khan, K.M.; Tareen, R.B. Antiinflammatory and lipoxygenase inhibitory compounds from vitex agnus-castus. Phytother. Res. 2009, 23, 1336–1339. [Google Scholar] [CrossRef]
- Certo, G.; Costa, R.; D’Angelo, V.; Russo, M.; Albergamo, A.; Dugo, G.; Germanò, M.P. Anti-angiogenic activity and phytochemical screening of fruit fractions from Vitex agnus castus. Nat. Prod. Res. 2017, 31, 2850–2856. [Google Scholar] [CrossRef]
- Kour, M.; Madom Anantharaya, V.N.; Bhat, K.M.R.; Chakraborti, S.; Kodavanji, B. Effect of Vitex agnus extract on MNU induced mammary tumor of Sprague Dawley rats. J. Young Pharm. 2017, 9, 367. [Google Scholar] [CrossRef]
- Khalilzadeh, E.; Saiah, G.V.; Hasannejad, H.; Ghaderi, A.; Ghaderi, S.; Hamidian, G.; Mahmoudi, R.; Eshgi, D.; Zangisheh, M. Antinociceptive effects, acute toxicity and chemical composition of Vitex agnus-castus essential oil. Avicenna J. Phytomedicine 2015, 5, 218. [Google Scholar]
- Hamza, A.H.; AlBishri, W.M.; Alfaris, M.H. Effect of Vitex agnus-castus plant extract on polycystic ovary syndrome complications in experimental rat model. Asian Pac. J. Reprod. 2019, 8, 63. [Google Scholar]
- Abu-Raghif, A.R.; Sahib, H.B.; Abbas, S.N. Anti-hyperlipidemic effect of Vitex agnus castus Extracts in Mice. Int. J. Pharm. Sci. Rev. Res. 2015, 35, 120–125. [Google Scholar]
- Moreno, F.N.; Campos-Shimada, L.B.; Costa, S.C.D.; Garcia, R.F.; Cecchini, A.L.; Natali, M.R.M.; Vitoriano, A.d.S.; Ishii-Iwamoto, E.L.; Salgueiro-Pagadigorria, C.L. Vitex agnus-castus L.(Verbenaceae) improves the liver lipid metabolism and redox state of ovariectomized rats. Evid.-Based Complement. Alternat. Med. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Röhrl, J.; Werz, O.; Ammendola, A.; Künstle, G. Vitex agnus-castus dry extract BNO 1095 (Agnucaston®) inhibits uterine hyper-contractions and inflammation in experimental models for primary dysmenorrhea. Clin. Phytoscience 2017, 2, 1–12. [Google Scholar]
- Sahib, H.B.; Al-Zubaidy, A.A.; Hussain, S.M.; Jassim, G.A. The anti angiogenic activity of vitex agnus castus leaves extracts. Int. J. Pharm. Pharm. Sci. 2014, 6, 863–869. [Google Scholar]
- Ahangarpour, A.; Najimi, S.A.; Farbood, Y. Effects of Vitex agnus-castus fruit on sex hormones and antioxidant indices in a d-galactose-induced aging female mouse model. J. Chin. Med. Assoc. 2016, 79, 589–596. [Google Scholar] [CrossRef]
- Saberi, M.; Rezvanizadeh, A.; Bakhtiarian, A. The antiepileptic activity of Vitex agnus castus extract on amygdala kindled seizures in male rats. Neurosci. Lett. 2008, 441, 193–196. [Google Scholar] [CrossRef] [PubMed]
- Sehmisch, S.; Boeckhoff, J.; Wille, J.; Seidlova-Wuttke, D.; Rack, T.; Tezval, M.; Wuttke, W.; Stuermer, K.; Stuermer, E. Vitex agnus castus as prophylaxis for osteopenia after orchidectomy in rats compared with estradiol and testosterone supplementation. Phytother. Res. 2009, 23, 851–858. [Google Scholar] [CrossRef]
- Chhabra, G.; Kulkarni, S. Evaluation of anti-inflammatory activity of Vitex agnus castus leaves. Quantitative analysis of flavonoids as possible active constituents. J. Pharmacogn. Phytochem. 2014, 3, 183–189. [Google Scholar]
- Deniz, G.Y.; Laloglu, E.; Altun, S.; Yiğit, N.; Gezer, A. Antioxidant and anti-apoptotic effects of vitexilactone on cisplatin-induced nephrotoxicity in rats. Biotech. Histochem. 2020, 1–8. [Google Scholar] [CrossRef]
- Lee, H.; Jung, K.-H.; Lee, H.; Park, S.; Choi, W.; Bae, H. Casticin, an active compound isolated from Vitex Fructus, ameliorates the cigarette smoke-induced acute lung inflammatory response in a murine model. Int. Immunopharmacol. 2015, 28, 1097–1101. [Google Scholar] [CrossRef]
- Ibrahim, A.Y.; El-Newary, S.A.; Youness, E.R.; Ibrahim, A.M.; El Kashak, W.A. Protective and therapeutic effect of Vitex agnus-castus against prostate cancer in rat. J. Appl. Pharm. Sci. 2017, 7, 133–143. [Google Scholar]
- Webster, D.E.; He, Y.; Chen, S.-N.; Pauli, G.F.; Farnsworth, N.R.; Wang, Z.J. Opioidergic mechanisms underlying the actions of Vitex agnus-castus L. Biochem. Pharmacol. 2011, 81, 170–177. [Google Scholar] [CrossRef]
- Ibrahim, S.R.M.; Ahmed, N.; Almalki, S.; Alharbi, N.; El-Agamy, D.S.; Alahmadi, L.A.; Saubr, M.K.; Elkablawy, M.; Elshafie, R.M.; Mohamed, G.A. Vitex agnus-castus safeguards the lung against lipopolysaccharide-induced toxicity in mice. J. Food Biochem. 2019, 43, e12750. [Google Scholar] [CrossRef] [PubMed]
- Allahtavakoli, M.; Honari, N.; Pourabolli, I.; Arababadi, M.K.; Ghafarian, H.; Roohbakhsh, A.; Nadimi, A.E.; Shamsizadeh, A. Vitex agnus castus extract improves learning and memory and increases the transcription of estrogen receptor α in hippocampus of ovariectomized rats. Basic Clin. Neurosci. 2015, 6, 185. [Google Scholar] [PubMed]
- Oroojan, A.; Ahangarpour, A.; Khorsandi, L.; Najimi, S. Effects of hydro-alcoholic extract of Vitex agnus-castus fruit on kidney of d-galactose-induced aging model in female mice. Iran. J. Vet. Res. 2016, 17, 203. [Google Scholar]
- Alimohamadi, R.; Fatemi, I.; Naderi, S.; Hakimizadeh, E.; Rahmani, M.-R.; Allahtavakoli, M. Protective effects of Vitex agnus-castus in ovariectomy mice following permanent middle cerebral artery occlusion. Iran. J. Basic Med. Sci. 2019, 22, 1097. [Google Scholar]
- Csupor, D.; Lantős, T.; Hegyi, P.; Benko, R.; Viola, R.; Gyöngyi, Z.; Csécsei, P.; Tóth, B.; Vasas, A.; Márta, K. Vitex agnus-castus in premenstrual syndrome: A meta-analysis of double-blind randomised controlled trials. Complement. Ther. Med. 2019, 47, 102190. [Google Scholar] [CrossRef]
- Cerqueira, R.O.; Frey, B.N.; Leclerc, E.; Brietzke, E. Vitex agnus castus for premenstrual syndrome and premenstrual dysphoric disorder: A systematic review. Arch. Women′s. Ment. Health 2017, 20, 713–719. [Google Scholar] [CrossRef]
- Seidlova-Wuttke, D.; Wuttke, W. The premenstrual syndrome, premenstrual mastodynia, fibrocystic mastopathy and infertility have often common roots: Effects of extracts of chasteberry (Vitex agnus castus) as a solution. Clin. Phytoscience 2017, 3, 6. [Google Scholar] [CrossRef]
- Rafieian-Kopaei, M.; Movahedi, M. Systematic review of premenstrual, postmenstrual and infertility disorders of Vitex agnus castus. Electron. Physician 2017, 9, 3685. [Google Scholar] [CrossRef]
- Aksoy, A.N.; Gözükara, I.; Kabil Kucur, S. Evaluation of the efficacy of F ructus agni casti in women with severe primary dysmenorrhea: A prospective comparative D oppler study. J. Obstet. Gynaecol. Res. 2014, 40, 779–784. [Google Scholar] [CrossRef]
- Eftekhari, M.H.; Rostami, Z.H.; Emami, M.J.; Tabatabaee, H.R. Effects of “vitex agnus castus” extract and magnesium supplementation, alone and in combination, on osteogenic and angiogenic factors and fracture healing in women with long bone fracture. J. Res. Med. Sci. 2014, 19, 1. [Google Scholar]
- Eltbogen, R.; Litschgi, M.; Gasser, U.; Nebel, S.; Zahner, C. Vitex agnus-castus extract (Ze 440) improves symptoms in women with menstrual cycle irregularities. Planta Medica 2014, 80, SL19. [Google Scholar] [CrossRef]
- De Franciscis, P.; Colacurci, N.; Riemma, G.; Conte, A.; Pittana, E.; Guida, M.; Schiattarella, A. A nutraceutical approach to menopausal complaints. Medicina 2019, 55, 544. [Google Scholar] [CrossRef] [PubMed]
- Naseri, R.; Farnia, V.; Yazdchi, K.; Alikhani, M.; Basanj, B.; Salemi, S. Comparison of Vitex agnus-castus Extracts with Placebo in Reducing Menopausal Symptoms: A Randomized Double-Blind Study. J. Korean Acad. Fam. Med. 2019, 40, 362–367. [Google Scholar] [CrossRef] [PubMed]
- Greene, J. Methods for assessing climacteric symptoms. J. Br. Menopause Soc. 1999, 5, 173–176. [Google Scholar] [CrossRef]
- Yavarikia, P.; Shahnazi, M.; Mirzaie, S.H.; Javadzadeh, Y.; Lutfi, R. Comparing the effect of mefenamic acid and vitex agnus on intrauterine device induced bleeding. J. Caring Sci. 2013, 2, 245. [Google Scholar]
- Higham, J.M.; O′brien, P.; Shaw, R. Assessment of menstrual blood loss using a pictorial chart. BJOG 1990, 97, 734–739. [Google Scholar] [CrossRef]
- He, Z.; Chen, R.; Zhou, Y.; Geng, L.; Zhang, Z.; Chen, S.; Yao, Y.; Lu, J.; Lin, S. Treatment for premenstrual syndrome with Vitex agnus castus: A prospective, randomized, multi-center placebo controlled study in China. Maturitas 2009, 63, 99–103. [Google Scholar] [CrossRef]
- Ma, L.; Lin, S.; Chen, R.; Wang, X. Treatment of moderate to severe premenstrual syndrome with Vitex agnus castus (BNO 1095) in Chinese women. Gynecol. Endocrinol. 2010, 26, 612–616. [Google Scholar] [CrossRef]
- Zamani, M.; Neghab, N.; Torabian, S. Therapeutic effect of Vitex agnus castus in patients with premenstrual syndrome. Acta Med. Iran. 2012, 50, 101–106. [Google Scholar] [PubMed]
- Shahnazi, M.; Khalili, A.F.; Hamdi, K.; Ghahremaninasab, P. The effects of combined low-dose oral contraceptives and Vitex agnus on the improvement of clinical and paraclinical parameters of polycystic ovarian syndrome: A triple-blind, randomized, controlled clinical trial. Iran. Red Crescent Med. J. 2016, 18. [Google Scholar] [CrossRef]
- Molaie, M.; Darvishi, B.; Jafari Azar, Z.; Shirazi, M.; Amin, G.; Afshar, S. Effects of a combination of Nigella sativa and Vitex agnus-castus with citalopram on healthy menopausal women with hot flashes: Results from a subpopulation analysis. Gynecol. Endocrinol. 2019, 35, 58–61. [Google Scholar] [CrossRef] [PubMed]
- De Franciscis, P.; Grauso, F.; Luisi, A.; Schettino, M.T.; Torella, M.; Colacurci, N. Adding Agnus Castus and magnolia to soy isoflavones relieves sleep disturbances besides postmenopausal vasomotor symptoms-long term safety and effectiveness. Nutrients 2017, 9, 129. [Google Scholar] [CrossRef]
- Aydin, İ.; Baltaci, D.; Trkyilmaz, S. Comparison of Vitex Agnus Castus with Meloxicam and Placebo in Treatment of Patients with Cyclical Mastalgia. Duzce Med. J. 2012, 14, 1–5. [Google Scholar]
- Momoeda, M.; Sasaki, H.; Tagashira, E.; Ogishima, M.; Takano, Y.; Ochiai, K. Efficacy and safety of Vitex agnus-castus extract for treatment of premenstrual syndrome in Japanese patients: A prospective, open-label study. Adv. Ther. 2014, 31, 362–373. [Google Scholar] [CrossRef] [PubMed]
- Ambrosini, A.; Di Lorenzo, C.; Coppola, G.; Pierelli, F. Use of Vitex agnus-castus in migrainous women with premenstrual syndrome: An open-label clinical observation. Acta Neurol. Belg. 2013, 113, 25–29. [Google Scholar] [CrossRef]
- Schellenberg, R.; Zimmermann, C.; Drewe, J.; Hoexter, G.; Zahner, C. Dose-dependent efficacy of the Vitex agnus castus extract Ze 440 in patients suffering from premenstrual syndrome. Phytomedicine 2012, 19, 1325–1331. [Google Scholar] [CrossRef]
- Abbaspoor, Z.; Hajikhani, N.A.; Afshari, P. Effect of Vitex agnus-castus on menopausal early symptoms in postmenopausal women: A randomized, double blind, placebo–controlled study. J. Adv. Med. Med. Res. 2011, 1, 132–140. [Google Scholar] [CrossRef]
- van Die, M.D.; Bone, K.M.; Burger, H.G.; Reece, J.E.; Teede, H.J. Effects of a combination of Hypericum perforatum and Vitex agnus-castus on PMS-like symptoms in late-perimenopausal women: Findings from a subpopulation analysis. J. Altern. Complement. Med. 2009, 15, 1045–1048. [Google Scholar] [CrossRef]
- Hossein-Rashidi, B.; Nemati, M. Effects of Vitex agnus-castus extract on the secretory function of pituitary-gonadal axis and pregnancy rate in patients with premature ovarian aging (POA). J. Herb. Med. 2017, 10, 24–30. [Google Scholar] [CrossRef]
- Merz, P.-G.; Gorkow, C.; Schrödter, A.; Rietbrock, S.; Sieder, C.; Loew, D.; Dericks-Tan, J.; Taubert, H. The effects of a special Agnus castus extract (BP1095E1) on prolactin secretion in healthy male subjects. Exp. Clin. Endocrinol. Diabetes 1996, 104, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Owolabi, M.A.; Abass, M.M.; Emeka, P.M.; Jaja, S.I.; Nnoli, M.; Dosa, B.O. Biochemical and histologic changes in rats after prolonged administration of the crude aqueous extract of the leaves of Vitex grandifolia. Pharmacogn. Res. 2010, 2, 273–278. [Google Scholar] [CrossRef]
| Condition | Plant Part | Extract | Activity | Effect | Reference |
|---|---|---|---|---|---|
| In vitro | Fruits | Ethyl acetate | Antioxidant activity | Lipid peroxidation was inhibited by casticin with an IC50 value of 0.049 mM. | [57] |
| In vitro | Leaves | Supercritical CO2 | Antifungal activity | The antifungal potential of essential oil with an MIC value of 0.64 µL/mL. | [58] |
| In vitro | Leaves | Hydrodistillation | Antifungal activity | The antimutagenic activity of V. agnus-castus leaf extract against Salmonella typhimurium. | [56] |
| In vitro | Aerial parts | Methanolic extract (Cr. MeOH Ext.) | Antimicrobial activity | The V. agnus-castus leaf essential oil showed an antibacterial effect against Staphylococcus aureus with an MIC value of 0.31% v/v. | [59] |
| In vitro | Seed | n-hexane | Antifungal activity | Essential oil was effective against Candida species with an MIC50 value of 1.75 mg/mL. | [60] |
| In vitro | Ripened fruits and fruitless aerial parts | Ethanol | Anticancer activity | Cytotoxic effect of V. agnus-castus fruit extract against MCF-7 cancer cells with IC50 = 88 μg/mL. | [61] |
| In vitro | Fruits | Chloroform–methanol (250:1) | Anticancer activity | The cytotoxicity was due to differentiation of the hematopoietic cell line. | [62] |
| In vitro | Fruits | Ethanol | Anticancer activity | Enhanced apoptosis and decreased intracellular ROS levels. | [49] |
| In vitro | Leaves | Methanol | Antioxidant effect | The free radical scavenging effect of V. agnus-castus methanol extract with an IC50 value of 126.79 mg/mL. | [63] |
| Condition | Plant Part | Extract | Activity | Effect | Reference |
|---|---|---|---|---|---|
| Animal model | Leaves | Hydrodistillation | Antinociceptive activity and analgesic effect | The analgesic activity was due to the activation of muscarinic receptors of the cholinergic system and endogenous opioidergic system. | [83] |
| Animal model | Stems and leaves | Ethanol | Treatment of polycystic ovary syndrome | V. agnus-castus exhibited hypoglycemic, antioxidant, and antihyperlipidemic activities in rats. | [84] |
| Animal model | Fruits | Chloroform, methanol, and water | Antihyperlipidemic activity | The V. agnus-castus extract (500 mg/kg for 28 days) decreased the levels of VLDL, LDL, TG, and TC. | [85] |
| Animal model | Fruits | Hexane, ethyl ether, and n-butanol | Protected against nonalcoholic fat liver disease | Prevented oxidative stress and treated nonalcoholic fat liver disease. | [86] |
| Animal model | Berries | Ethanol | Anti-inflammatory activity | Inhibited the production of reactive oxygen species, the release of cytokines, and the formation of leukotriene. | [87] |
| Animal model | Leaves | Chloroform | Antiangiogenic activity | Prevented growth of psoriasis, cataract, and tumor. | [88] |
| Animal model | Fruits | Ethanol | Antiaging effects | Improved d-galactose-induced aging symptoms, including enhanced serum LH and FSH levels, follicle degeneration, and endometrial atrophy. | [89] |
| Animal model | Fruits | Methanol | Antiepileptic activity | Reduced stage 5 duration and after-discharge duration. | [90] |
| Animal model | Fruits | Ethanol | Osteoprotective effects | Enhanced biomechanical stability of bone via connectivity density in the orchidectomized rats and improved the trabecular microarchitecture. | [91] |
| Animal model | Leaves | Methanol | Anti-inflammatory effect | The V. agnus-castus methanol extract (400 mg/kg) reduced IL-6 and TNF-α levels. | [92] |
| Animal model | - | Methanol, n-hexane, and Ethyl acetate | Antioxidant and antiapoptotic effects | Vitexilactone extracted from V. agnus-castus reduced caspase-3 and apoptosis marker expression in Sprague–Dawley rats. | [93] |
| Condition | Activity | Administration | Effect | Reference |
|---|---|---|---|---|
| Clinical trial | Treatment of vasomotor symptoms. | The administration of V. agnus-castus (40 mg) once a day for a month in women with postmenopausal symptoms. | Improvement of sleep satisfaction. | [118] |
| Clinical trial | Treatment of mastalgia. | The administration of V. agnus-castus in patients with mastalgia. | Reduction in prolactin level after three months. | [119] |
| Clinical trial | Treatment of premenstrual syndrome. | Administration of V. agnus-castus extract (20 mg) once a day for three menstrual cycles in Japanese women. | The symptoms of premenstrual syndrome were improved. | [120] |
| Clinical trial | Treatment of premenstrual syndrome. | The administration of V. agnus-castus (40 mg) once a day for three months in migrainous women with premenstrual syndrome. | The symptoms of premenstrual syndrome were reduced in 66 women. | [121] |
| Clinical trial | Treatment of premenstrual syndrome. | The administration of V. agnus-castus extract Ze 440 (20 mg) once a day. | The symptoms of premenstrual syndrome were relieved in women. | [122] |
| Clinical trial | Treatment of menopausal syndrome. | The administration of V. agnus-castus extract (40 drops) once a day for 8 weeks in women. | Hot flashes were positively influenced by Vitex in women. | [123] |
| Clinical trial | Treatment of premenstrual syndrome. | The co-administration of V. agnus-castus with Hypericum perforatum twice a day for 16 weeks in women with premenstrual syndrome. | Symptoms such as hydration clusters and anxiety were alleviated. | [124] |
| Clinical trial | Treatment of premenstrual syndrome. | The administration of V. agnus-castus extract (40 drops) for 4 months. | The pregnancy rate, endometrial thickness, ovulation, and fertility were increased in women. | [125] |
| Clinical trial | Prolactin-inhibiting activity. | The daily administration of Agnus-castus extract (BP1O95E1) at a concentration of 480 mg for two weeks in healthy male subjects. | Decreased prolactin profile levels. | [126] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Souto, E.B.; Durazzo, A.; Nazhand, A.; Lucarini, M.; Zaccardelli, M.; Souto, S.B.; Silva, A.M.; Severino, P.; Novellino, E.; Santini, A. Vitex agnus-castus L.: Main Features and Nutraceutical Perspectives. Forests 2020, 11, 761. https://doi.org/10.3390/f11070761
Souto EB, Durazzo A, Nazhand A, Lucarini M, Zaccardelli M, Souto SB, Silva AM, Severino P, Novellino E, Santini A. Vitex agnus-castus L.: Main Features and Nutraceutical Perspectives. Forests. 2020; 11(7):761. https://doi.org/10.3390/f11070761
Chicago/Turabian StyleSouto, Eliana B., Alessandra Durazzo, Amirhossein Nazhand, Massimo Lucarini, Massimo Zaccardelli, Selma B. Souto, Amelia M. Silva, Patricia Severino, Ettore Novellino, and Antonello Santini. 2020. "Vitex agnus-castus L.: Main Features and Nutraceutical Perspectives" Forests 11, no. 7: 761. https://doi.org/10.3390/f11070761
APA StyleSouto, E. B., Durazzo, A., Nazhand, A., Lucarini, M., Zaccardelli, M., Souto, S. B., Silva, A. M., Severino, P., Novellino, E., & Santini, A. (2020). Vitex agnus-castus L.: Main Features and Nutraceutical Perspectives. Forests, 11(7), 761. https://doi.org/10.3390/f11070761










