Recent Consequences of Climate Change Have Affected Tree Growth in Distinct Nothofagus macrocarpa (DC.) FM Vaz & Rodr Age Classes in Central Chile
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and N. macrocarpa Populations
2.2. Characteristics of N. macrocarpa Populations and Tree-Ring Analysis
2.3. Tree-Growth Patterns
2.4. Avoiding Ontogenetic Problem
2.5. Effect of Climate and CO2 on Tree Growth
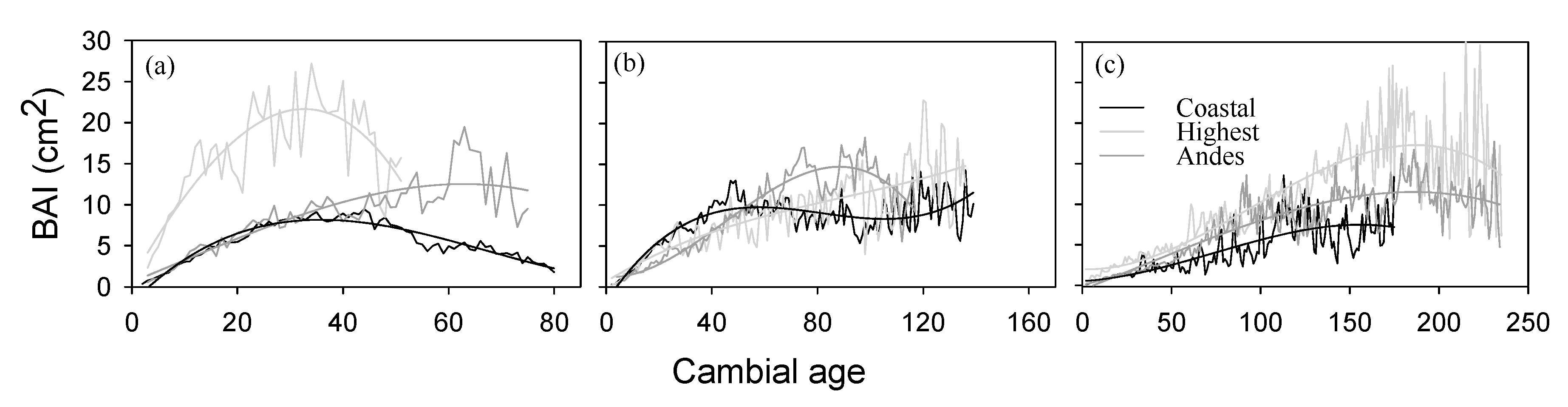
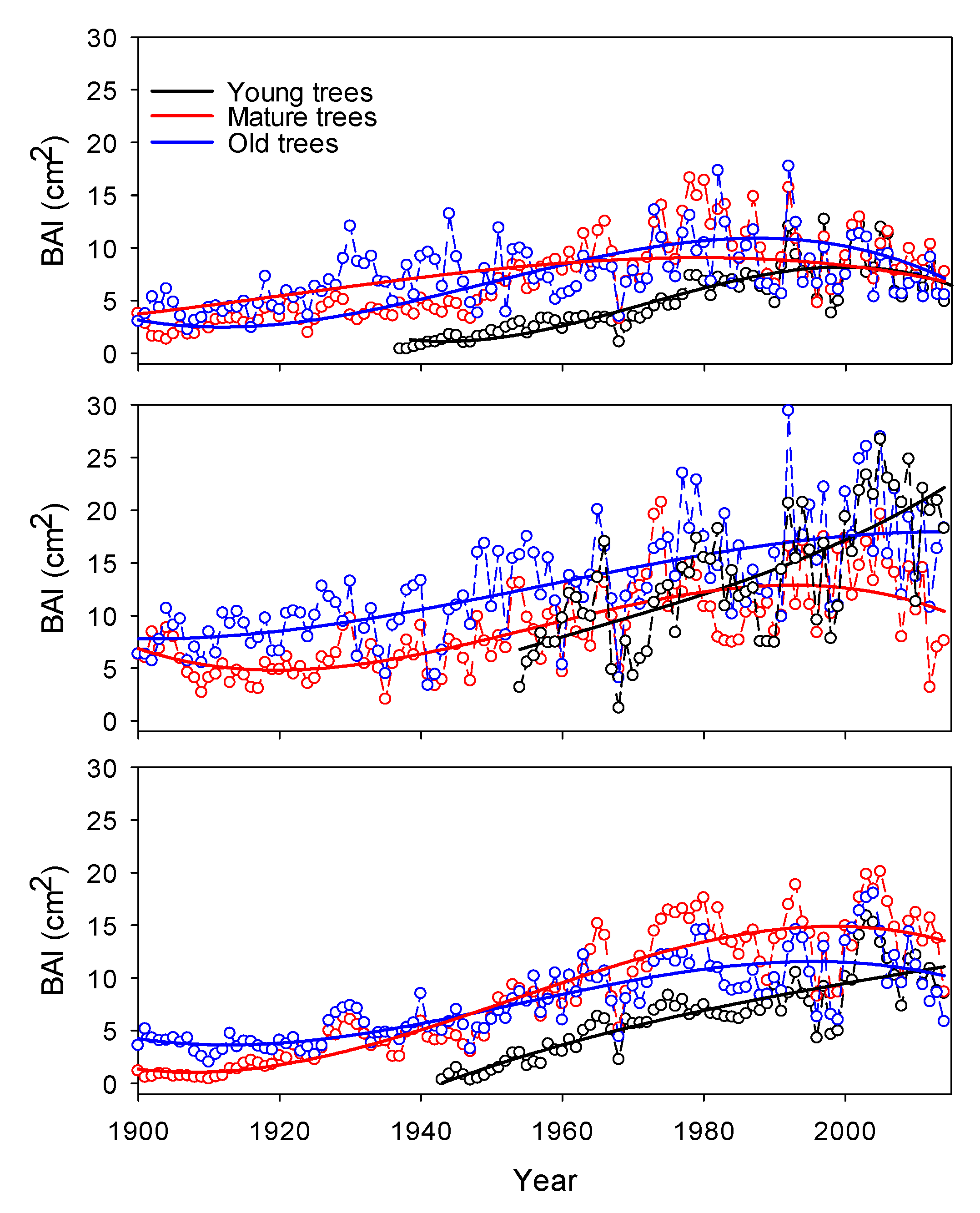
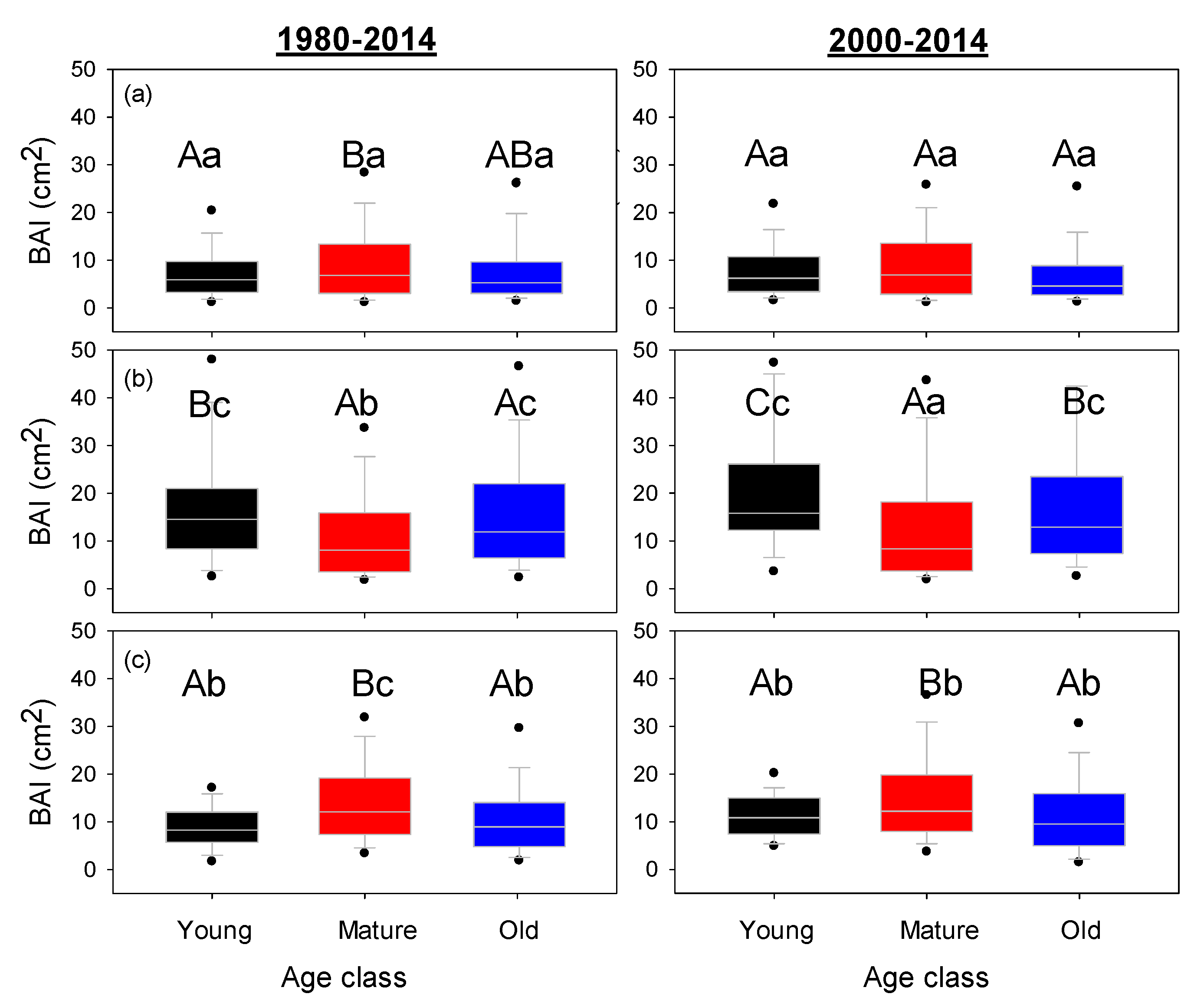
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stocker, T.F.; Qin, D.; Plattner, G.K.; Tignor, M.; Allen, S.K.; Boschung, J.; Nauels, A.; Xia, Y.; Bex, B.; Midgley, B.M. Contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change. In IPCC, 2013: Climate Change 2013: The Physical Science Basis; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Wullschleger, S.D.; Tschaplinski, T.J.; Norby, R.J. Plant water relations at elevated CO2–Implications for water-limited environments. Plant Cell Environ. 2002, 25, 319–331. [Google Scholar] [CrossRef] [PubMed]
- Allen, C.D.; Breshears, D.D.; McDowell, N.G. On underestimation of global vulnerability to tree mortality and forest die-off from hotter drought in the Anthropocene. Ecosphere 2015, 6, 1–55. [Google Scholar] [CrossRef]
- Bert, D.; Leavitt, S.W.; Dupouey, J.L. Variations of wood δ13C and water-use efficiency of Abies alba during the last century. Ecology 1997, 78, 1588–1596. [Google Scholar]
- Battipaglia, G.; Saurer, M.; Cherubini, P.; Calfapietra, C.; McCarthy, H.R.; Norby, R.J.; Francesca Cotrufo, M. Elevated CO2 increases tree-level intrinsic water use efficiency: Insights from carbon and oxygen isotope analyses in tree rings across three forest FACE sites. New Phytol. 2013, 197, 544–554. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Zhang, X.; He, X.; Davi, N.K.; Li, L.; Bai, X. Response of radial growth to warming and CO2 enrichment in southern Northeast China: A case of Pinus tabulaeformis. Clim. Chang. 2015, 130, 559–571. [Google Scholar] [CrossRef]
- Srur, A.M.; Villalba, R.; Villagra, P.E.; Hertel, D. Influencias de las variaciones en el clima y en la concentración de C0 (2) sobre el crecimiento de Nothofagus pumilio en la Patagonia. Rev. Chil. Hist. Nat. 2008, 81, 239–256. [Google Scholar] [CrossRef]
- Camarero, J.J.; Gazol, A.; Galván, J.D.; Sangüesa-Barreda, G.; Gutiérrez, E. Disparate effects of global-change drivers on mountain conifer forests: Warming-induced growth enhancement in young trees vs. CO2 fertilization in old trees from wet sites. Glob. Chang. Biol. 2015, 21, 738–749. [Google Scholar] [CrossRef]
- Konter, O.; Büntgen, U.; Carrer, M.; Timonen, M.; Esper, J. Climate signal age effects in boreal tree-rings: Lessons to be learned for paleoclimatic reconstructions. Quat. Sci. Rev. 2016, 142, 164–172. [Google Scholar] [CrossRef]
- Carrer, M.; Urbinati, C. Long-term change in the sensitivity of tree-ring growth to climate forcing in Larix decidua. New Phytol. 2006, 170, 861–872. [Google Scholar] [CrossRef]
- Pompa-García, M.; Hadad, M.A. Sensitivity of pines in Mexico to temperature varies with age. Atmósfera 2016, 29, 209–219. [Google Scholar] [CrossRef]
- Hadad, M.A.; Roig Juñent, F.A.; Boninsegna, J.A.; Patón, D. Age effects on the climatic signal in Araucaria araucana from xeric sites in Patagonia, Argentina. Plant Ecol. Divers. 2015, 8, 343–351. [Google Scholar] [CrossRef]
- Arco Molina, J.G.; Helle, G.; Hadad, M.A.; Roig, F.A. Variations in the intrinsic water-use efficiency of north Patagonian forests under a present climate change scenario: Tree age, site conditions and long-term environmental effects. Tree Physiol. 2019, 39, 661–678. [Google Scholar] [CrossRef] [PubMed]
- Brienen, R.J.W.; Gloor, E.; Zuidema, P.A. Detecting evidence for CO2 fertilization from tree ring studies: The potential role of sampling biases. Glob. Biogeochem. Cycles 2012, 26. [Google Scholar] [CrossRef]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, D.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.H.T. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; da Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853. [Google Scholar] [CrossRef] [PubMed]
- Le Quesne, C.; Stahle, D.W.; Cleaveland, M.K.; Therrell, M.D.; Aravena, J.C.; Barichivich, J. Ancient Austrocedrus tree-ring chronologies used to reconstruct central Chile precipitation variability from AD 1200 to 2000. J. Clim. 2006, 19, 5731–5744. [Google Scholar] [CrossRef]
- Luebert, F.; Pliscoff, P. Sinopsis Bioclimática y Vegetacional de Chile. Editorial Universitaria: Santiago, Chile, 2006. [Google Scholar]
- Donoso, S.R.; Pena-Rojas, K.; Delgado-Flores, C.; Riquelme, A.; Paratori, M. Above-ground biomass accumulation and growth in a marginal Nothofagus macrocarpa forest in central Chile. Interciencia 2010, 35, 65–69. [Google Scholar]
- Donoso, C. Reseña ecológica de los bosques mediterráneos de Chile. Bosque 1982, 4, 117–146. [Google Scholar] [CrossRef]
- Christie, D.A.; Boninsegna, J.A.; Cleaveland, M.K.; Lara, A.; Le Quesne, C.; Morales, M.S.; Mudelsee, M.; Stahle, D.W.; Villalba, R. Aridity changes in the Temperate-Mediterranean transition of the Andes since AD 1346 reconstructed from tree-rings. Clim. Dyn. 2011, 36, 1505–1521. [Google Scholar] [CrossRef]
- Garreaud, R.D.; Vuille, M.; Compagnucci, R.; Marengo, J. Present-day South American climate. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2009, 281, 180–195. [Google Scholar] [CrossRef]
- Carrasco, J.F.; Casassa, G.; Quintana, J. Changes of the 0 °C isotherm and the equilibrium line altitude in central Chile during the last quarter of the 20th century/Changements de l’isotherme 0 °C et de la ligne d’équilibre des neiges dans le Chili central durant le dernier quart du 20ème siècle. Hydrol. Sci. J. 2005, 50. [Google Scholar] [CrossRef]
- Le Quesne, C.; Acuña, C.; Boninsegna, J.A.; Rivera, A.; Barichivich, J. Long-term glacier variations in the Central Andes of Argentina and Chile, inferred from historical records and tree-ring reconstructed precipitation. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2009, 281, 334–344. [Google Scholar] [CrossRef]
- Venegas-González, A.; Juñent, F.R.; Gutiérrez, A.G.; Filho, M.T. Recent radial growth decline in response to increased drought conditions in the northernmost Nothofagus populations from South America. For. Ecol. Manag. 2018, 409, 94–104. [Google Scholar] [CrossRef]
- Vicuña, S.; Garreaud, R.D.; McPhee, J. Climate change impacts on the hydrology of a snowmelt driven basin in semiarid Chile. Clim. Chang. 2011, 105, 469–488. [Google Scholar] [CrossRef]
- Schulz, J.J.; Cayuela, L.; Echeverria, C.; Salas, J.; Benayas, J.M.R. Monitoring land cover change of the dryland forest landscape of Central Chile (1975–2008). Appl. Geogr. 2010, 30, 436–447. [Google Scholar] [CrossRef]
- Villagrán, C.; Armesto, J.J. Fitogeografía histórica de la Cordillera de la Costa de Chile. In Historia, Biodiversidad y Ecologia de Los Bosques Costeros de Chile; Editorial Universitaria: Santiago, Chile, 2005; pp. 99–116. [Google Scholar]
- Roig, F.A.; Villalba, R. Understanding climate from Patagonian tree rings. Dev. Quat. Sci. 2008, 11, 411–435. [Google Scholar]
- Boninsegna, J.A.; Argollo, J.; Aravena, J.C.; Barichivich, J.; Christie, D.; Ferrero, M.E.; Lara, A.; Le Quesne, C.; Luckman, B.H.; Masiokas, M.; et al. Dendroclimatological reconstructions in South America: A review. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2009, 281, 210–228. [Google Scholar] [CrossRef]
- Huang, J.G.; Bergeron, Y.; Denneler, B.; Berninger, F.; Tardif, J. Response of forest trees to increased atmospheric CO2. CRC Crit. Rev. Plant Sci. 2007, 26, 265–283. [Google Scholar] [CrossRef]
- Körner, C. Significance of temperature in plant life. In Plant Growth and Climate Change; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2006; pp. 48–69. [Google Scholar]
- Rossi, S.; Deslauriers, A.; Anfodillo, T.; Carrer, M. Age-dependent xylogenesis in timberline conifers. New Phytol. 2008, 177, 199–208. [Google Scholar] [CrossRef]
- Camarero, J.J.; Gazol, A.; Tardif, J.C.; Conciatori, F. Attributing forest responses to global-change drivers: Limited evidence of a CO2—Fertilization effect in Iberian pine growth. J. Biogeogr. 2015, 42, 2220–2233. [Google Scholar] [CrossRef]
- Venegas-González, A.; Roig, F.; Gutiérrez, A.G.; Peña-Rojas, K.; Tomazello Filho, M. Efecto de la variabilidad climática sobre los patrones de crecimiento y establecimiento de Nothofagus macrocarpa en Chile central. Bosque 2018, 39, 81–93. [Google Scholar] [CrossRef]
- Montecinos, A.; Aceituno, P. Seasonality of the ENSO-related rainfall variability in central Chile and associated circulation anomalies. J. Clim. 2003, 16, 281–296. [Google Scholar] [CrossRef]
- Villagrán, C.M. Quaternary history of the Mediterranean vegetation of Chile. In Ecology and Biogeography of Mediterranean Ecosystems in Chile, California, and Australia; Springer: Berlin/Heidelberg, Germany, 1995; pp. 3–20. [Google Scholar]
- Heenan, P.B.; Smissen, R.D. Revised circumscription of Nothofagus and recognition of the segregate genera Fuscospora, Lophozonia, and Trisyngyne (Nothofagaceae). Phytotaxa 2013, 146, 1–31. [Google Scholar] [CrossRef]
- Amigo, J.; Rodriguez-Guitian, M. Bioclimatic and phytosociological diagnosis of the species of the Nothofagus genus (Nothofagaceae) in South America. Int. J. Geobot. Res. 2011, 1, 1–20. [Google Scholar] [CrossRef]
- Gajardo, R. Antecedentes sobre el “roble de Santiago” o “roble blanco”(Nothofagus macrocarpa) y sus problemas de conservación. Rev. Bosque Nativ. 2001, 28, 3–7. [Google Scholar]
- Vazquez, F.M.; Rodríguez, R.A. A new subspecies and two new combinations of Nothofagus Blume (Nothofagaceae) from Chile. Bot. J. Linn. Soc. 1999, 129, 75–83. [Google Scholar] [CrossRef]
- Nehrbass-Ahles, C.; Babst, F.; Klesse, S.; Nötzli, M.; Bouriaud, O.; Neukom, R.; Dobbertin, M.; Frank, D. The influence of sampling design on tree-ring-based quantification of forest growth. Glob. Chang. Biol. 2014, 20, 2867–2885. [Google Scholar] [CrossRef]
- Holmes, R.L.; Adams, R.K.; Fritts, H.C. Tree-Ring Chronologies of Western North America: California, Eastern Oregon and Northern Great Basin with Procedures Used in the Chronology Development Work Including Users Manuals for Computer Programs COFECHA and ARSTAN; Laboratory of Tree-Ring Research, University of Arizona: Tucson, AZ, USA, 1986. [Google Scholar]
- Husson, F.; Josse, J.; Le, S.; Mazet, J.; Husson, M.F. Package ‘FactoMineR’. 2019. Available online: http://factominer.free.fr (accessed on 3 July 2019).
- Biondi, F.; Qeadan, F. A theory-driven approach to tree-ring standardization: Defining the biological trend from expected basal area increment. Tree Ring Res. 2008, 64, 81–96. [Google Scholar] [CrossRef]
- Girardin, M.P.; Bouriaud, O.; Hogg, E.H.; Kurz, W.; Zimmermann, N.E.; Metsaranta, J.M.; de Jong, R.; Frank, D.C.; Esper, J.; Büntgen, U. No growth stimulation of Canada’s boreal forest under half-century of combined warming and CO2 fertilization. Proc. Natl. Acad. Sci. USA 2016, 113, E8406–E8414. [Google Scholar] [CrossRef]
- Bunn, A.G. A dendrochronology program library in R (dplR). Dendrochronologia 2008, 26, 115–124. [Google Scholar] [CrossRef]
- Duncan, R.P. An evaluation of errors in tree age estimates based on increment cores in kahikatea (Dacrycarpus dacrydioides). N. Zeal. Nat. Sci. 1989, 16, 1–37. [Google Scholar]
- Muggeo, V.M.R. Segmented: An R package to fit regression models with broken-line relationships. R News 2008, 8, 20–25. [Google Scholar]
- Harris, I.; Jones, P.D.; Osborn, T.J.; Lister, D.H. Updated high resolution grids of monthly climatic observations–the CRU TS3. 10 Dataset. Int. J. Clim. 2013, 34, 623–642. [Google Scholar] [CrossRef]
- Tans, P. Trends in Atmospheric Carbon Dioxide. Available online: www.esrl.noaa.gov/gmd/ccgg/trends/ (accessed on 14 December 2016).
- Barton, K. MUMIn: Multi-model inference. R. Package Version 1.40.4. 2018. Available online: https: //cran.r-project.org/web/packages/MuMIn/index.html (accessed on 20 November 2018).
- Burnham, K.P.; Anderson, D.R. Information and likelihood theory: A basis for model selection and inference. In Model Selection Multimodel Inference a Practical Information-Theoretic Approach; Springer: Berlin/Heidelberg, Germany, 2002; pp. 49–97. [Google Scholar]
- Barichivich, J.; Sauchyn, D.J.; Lara, A. Climate signals in high elevation tree-rings from the semiarid Andes of north-central Chile: Responses to regional and large-scale variability. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2009, 281, 320–333. [Google Scholar] [CrossRef]
- Rozas, V.; DeSoto, L.; Olano, J.M. Sex-specific, age-dependent sensitivity of tree-ring growth to climate in the dioecious tree Juniperus thurifera. New Phytol. 2009, 182, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Bond, B.J. Age-related changes in photosynthesis of woody plants. Trends Plant Sci. 2000, 5, 349–353. [Google Scholar] [CrossRef]
- Gea-Izquierdo, G.; Cañellas, I. Local climate forces instability in long-term productivity of a Mediterranean oak along climatic gradients. Ecosystems 2014, 17, 228–241. [Google Scholar] [CrossRef]
- Körner, C.; Asshoff, R.; Bignucolo, O.; Hättenschwiler, S.; Keel, S.G.; Peláez-Riedl, S.; Pepin, S.; Siegwolf, R.T.W.; Zotz, G. Carbon flux and growth in mature deciduous forest trees exposed to elevated CO2. Science 2005, 309, 1360–1362. [Google Scholar] [CrossRef] [PubMed]
- Voelker, S.L.; Muzika, R.M.; Guyette, R.P.; Stambaugh, M.C. Historical CO2 growth enhancement declines with age in Quercus and Pinus. Ecol. Monogr. 2006, 76, 549–564. [Google Scholar] [CrossRef]
- Linares, J.C.; Camarero, J.J. From pattern to process: Linking intrinsic water-use efficiency to drought-induced forest decline. Glob. Chang. Biol. 2012, 18, 1000–1015. [Google Scholar] [CrossRef]
- Azcon, J.; Fleck, I.; Aranda, X.; Gómez, N. Fotosíntesis: Factores ambientales y cambio climático. In Fundamentos de Fisiología Vegetal; McGraw-Hill Interam: Madrid, Spain, 2008; pp. 247–263. [Google Scholar]
- Tognetti, R.; Lombardi, F.; Lasserre, B.; Cherubini, P.; Marchetti, M. Tree-ring stable isotopes reveal twentieth-century increases in water-use efficiency of Fagus sylvatica and Nothofagus spp. in Italian and Chilean mountains. PLoS ONE 2014, 9, e113136. [Google Scholar] [CrossRef]
- Handa, I.T.; Koerner, C.; Hättenschwiler, S. Conifer stem growth at the altitudinal treeline in response to four years of CO2 enrichment. Glob. Chang. Biol. 2006, 12, 2417–2430. [Google Scholar] [CrossRef]
- Andreu-Hayles, L.; Planells, O.; Gutierrez, E.; Muntan, E.; Helle, G.; Anchukaitis, K.J.; Schleser, G.H. Long tree-ring chronologies reveal 20th century increases in water-use efficiency but no enhancement of tree growth at five Iberian pine forests. Glob. Chang. Biol. 2011, 17, 2095–2112. [Google Scholar] [CrossRef]
- Penuelas, J.; Hunt, J.M.; Ogaya, R.; Jump, A.S. Twentieth century changes of tree-ring δ13C at the southern range-edge of Fagus sylvatica: Increasing water-use efficiency does not avoid the growth decline induced by warming at low altitudes. Glob. Chang. Biol. 2008, 14, 1076–1088. [Google Scholar] [CrossRef]
- Perez-Quezada, J.F.; Celis-Diez, J.L.; Brito, C.E.; Gaxiola, A.; Nuñez-Avila, M.; Pugnaire, F.I.; Armesto, J.J. Carbon fluxes from a temperate rainforest site in southern South America reveal a very sensitive sink. Ecosphere 2018, 9, e02193. [Google Scholar] [CrossRef]
- Simon, J.; Machar, I.; Brus, J.; Pechanec, V. Combining a growth-simulation model with acoustic-wood tomography as a decision-support tool for adaptive management and conservation of forest ecosystems. Ecol. Inform. 2015, 30, 309–312. [Google Scholar] [CrossRef]
- Boisier, J.P.; Alvarez-Garretón, C.; Cordero, R.R.; Damiani, A.; Gallardo, L.; Garreaud, R.D.; Lambert, F.; Ramallo, C.; Rojas, M.; Rondanelli, R. Anthropogenic drying in central-southern Chile evidenced by long-term observations and climate model simulations. Elem. Sci. Anthr. 2018, 6. [Google Scholar] [CrossRef]
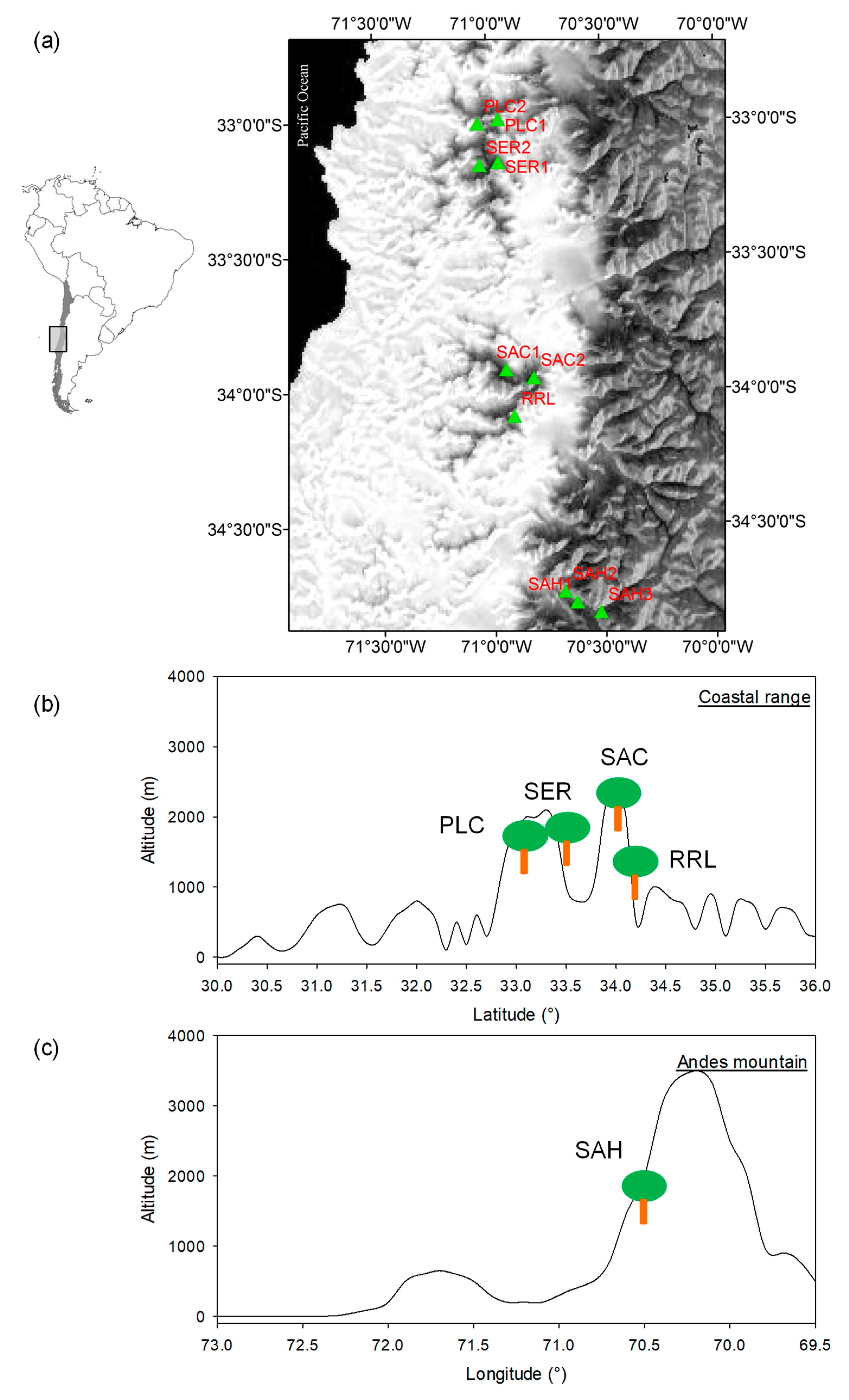

| Range | TPWA a | YSP b | Site | Code | Elev. (masl) | No. Trees c (chron.) | DBH ± SE d | DPR e | TGC f | Inter. g |
|---|---|---|---|---|---|---|---|---|---|---|
| Coastal | National park | 1967 | La Campana 1 | PLC1 | 1350 | 15(15) | 23.5 ± 0.7 | 10 | 1 | High |
| La Campana 2 | PLC2 | 1210 | 15(14) | 32.1 ± 3.1 | 7 | 1 | High | |||
| Nature Sanctuary | 2000 | El Roble 1 | SER1 | 1590 | 15(15) | 41.6 ± 3.63 | 6 | 1 | Medium | |
| El Roble 2 | SER2 | 1600 | 15(14) | 29.6 ± 1.2 | 5 | 1 | High | |||
| Nature Sanctuary | 2010 | Altos Cantillana 1 | SAC1 | 1810 | 15(13) | 43.7 ± 5.4 | * | 9 | Low | |
| Altos Cantillana 2 | SAC2 | 1850 | 12(11) | 51.4 ± 4.5 | * | 9 | Low | |||
| National reserve | 1996 | Robleria Loncha | RRL | 1090 | 24(23) | 35.4 ± 2.0 | 28 | 2 | Medium | |
| Andes | Nature Sanctuary | 1996 | Alto Huemul 1 | SAH1 | 1500 | 20(18) | 51.8 ± 10.5 | 50 | 4 | Low |
| Alto Huemul 2 | SAH2 | 1600 | 14(13) | 50.0 ± 4.2 | 53 | 4 | Low | |||
| Alto Huemul 3 | SAH3 | 1550 | 10(10) | 37.7 ± 4.7 | 47 | 4 | Medium |
| Cluster | Age Class | Trend Sign | p May–Nov (1) | T Oct–Dec (2) | CO2 (3) | Best Two Models | Delta AIC | Relative Weight |
|---|---|---|---|---|---|---|---|---|
| Coastal | <80 | (+) | 0.00267 *** | −12.0 *** | 0.0260 *** | 1,2,3 | 0 | 0.87 |
| 80–160 | (−) * | 0.00221 *** | −0.112 * | −0.0115 * | 1,2,3–1,2 | 0–0.16 | 0.31–0.29 | |
| >160 | (−) * | 0.00155 ** | - | −0.0117 | 2–1,3 | 0–1.20 | 0.34–0.19 | |
| Highest | <80 | (+) * | 0.00286 | - | 0.175 *** | 3–1,3 | 0–0.53 | 0.39–0.30 |
| 80–160 | (−) | 0.00220 | - | 0.0630 | 1–1,3 | 0–0.27 | 0.20–0.17 | |
| >160 | (-) | 0.00121 | - | 0.0585 | 1–1,3 | 0–1.9 | 0.26–0.24 | |
| Andes | <80 | (+) * | 0.00148 ** | −10.8 | 0.0985 *** | 1,3–1,2,3 | 0–0.22 | 0.48–0.43 |
| 80–160 | (−) | 0.00116 | −22.4 * | - | 2–1,2 | 0–0.43 | 0.23–0.18 | |
| >160 | (−) | 0.00156 * | −15.4 * | - | 1–1,2 | 0–0.75 | 0.23–0.16 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Venegas-González, A.; Roig, F.A.; Peña-Rojas, K.; Hadad, M.A.; Aguilera-Betti, I.; Muñoz, A.A. Recent Consequences of Climate Change Have Affected Tree Growth in Distinct Nothofagus macrocarpa (DC.) FM Vaz & Rodr Age Classes in Central Chile. Forests 2019, 10, 653. https://doi.org/10.3390/f10080653
Venegas-González A, Roig FA, Peña-Rojas K, Hadad MA, Aguilera-Betti I, Muñoz AA. Recent Consequences of Climate Change Have Affected Tree Growth in Distinct Nothofagus macrocarpa (DC.) FM Vaz & Rodr Age Classes in Central Chile. Forests. 2019; 10(8):653. https://doi.org/10.3390/f10080653
Chicago/Turabian StyleVenegas-González, Alejandro, Fidel A. Roig, Karen Peña-Rojas, Martín A. Hadad, Isabella Aguilera-Betti, and Ariel A. Muñoz. 2019. "Recent Consequences of Climate Change Have Affected Tree Growth in Distinct Nothofagus macrocarpa (DC.) FM Vaz & Rodr Age Classes in Central Chile" Forests 10, no. 8: 653. https://doi.org/10.3390/f10080653
APA StyleVenegas-González, A., Roig, F. A., Peña-Rojas, K., Hadad, M. A., Aguilera-Betti, I., & Muñoz, A. A. (2019). Recent Consequences of Climate Change Have Affected Tree Growth in Distinct Nothofagus macrocarpa (DC.) FM Vaz & Rodr Age Classes in Central Chile. Forests, 10(8), 653. https://doi.org/10.3390/f10080653





