Review of Japanese Pine Bast Scale, Matsucoccus matsumurae (Kuwana) (Coccomorpha: Matsucoccidae), Occurring on Japanese Black Pine (Pinus thunbergii Parl.) and Japanese Red Pine (P. densiflora Siebold & Zucc.) from Korea
Abstract
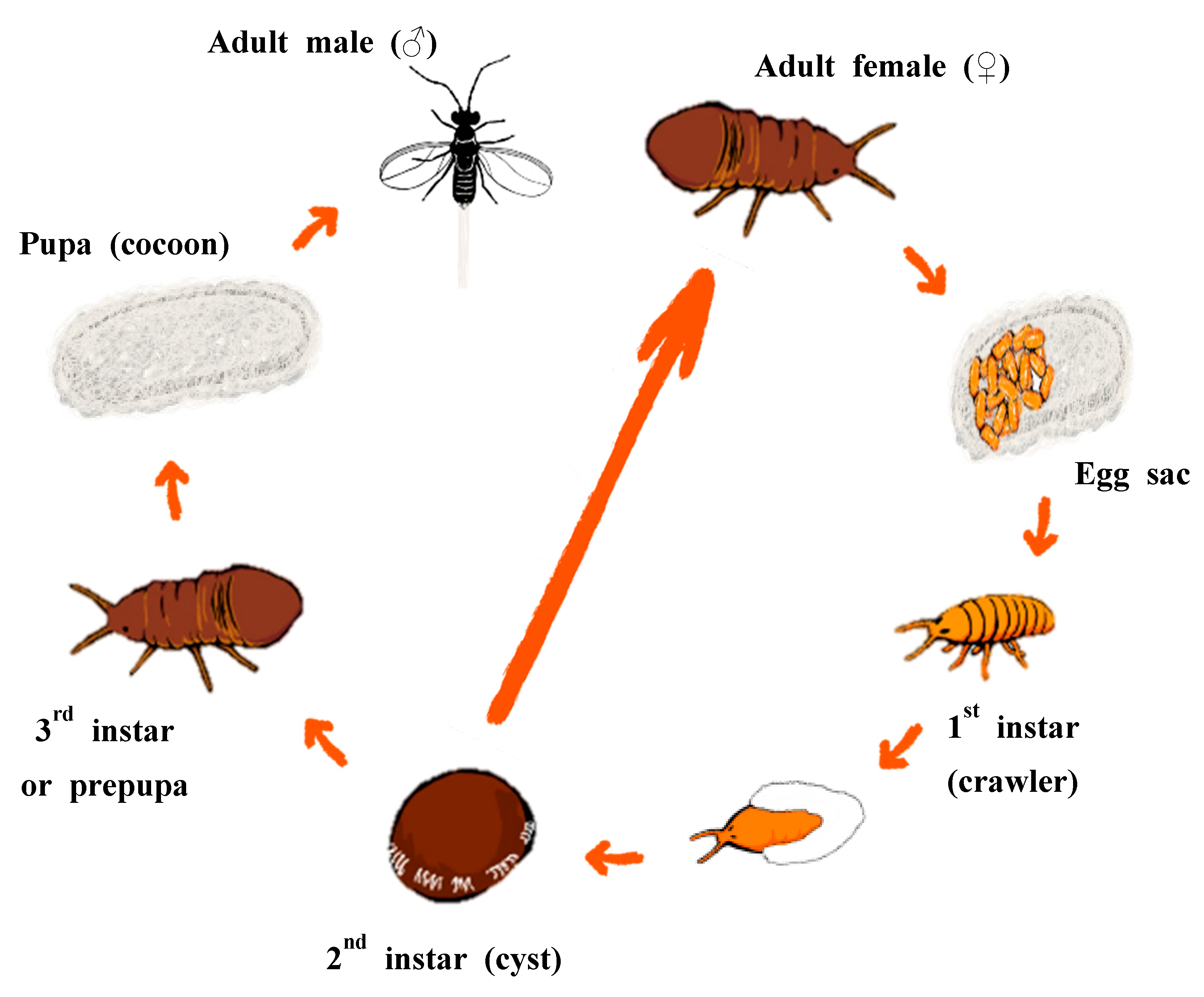
1. Introduction
2. Materials and Methods
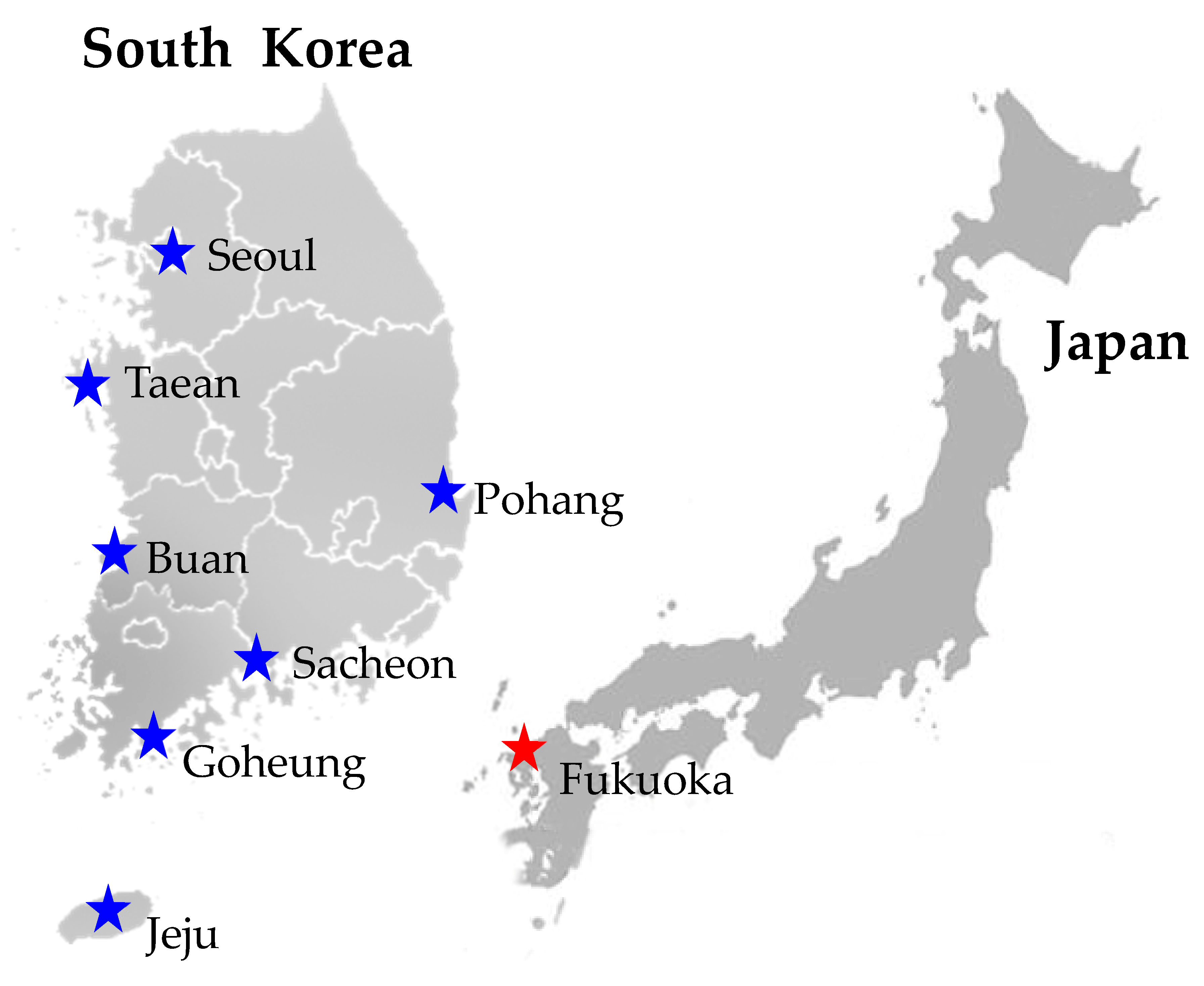
2.1. Sample Collection
2.2. Morphological Identification
2.3. Molecular Analyses
3. Results
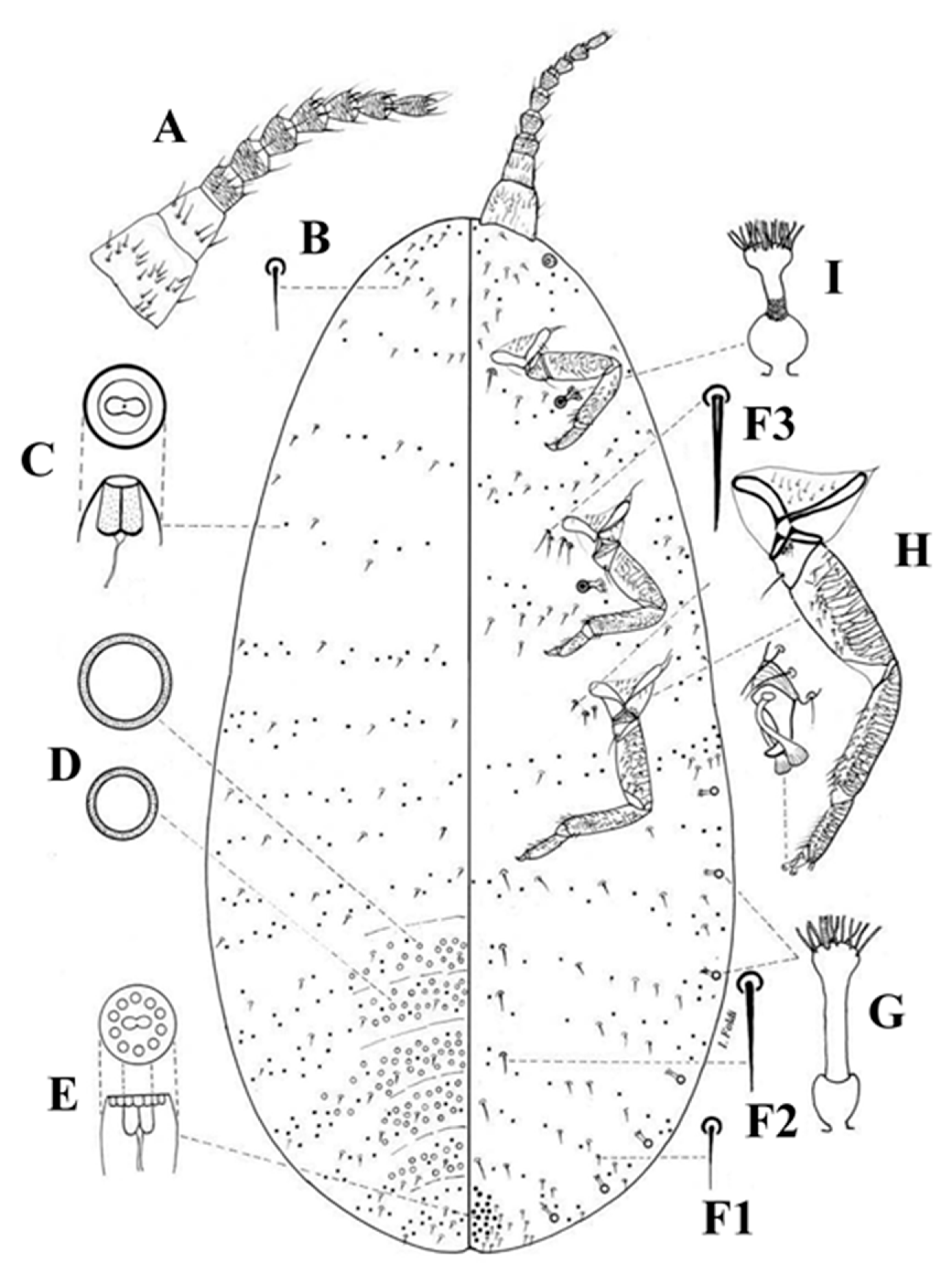
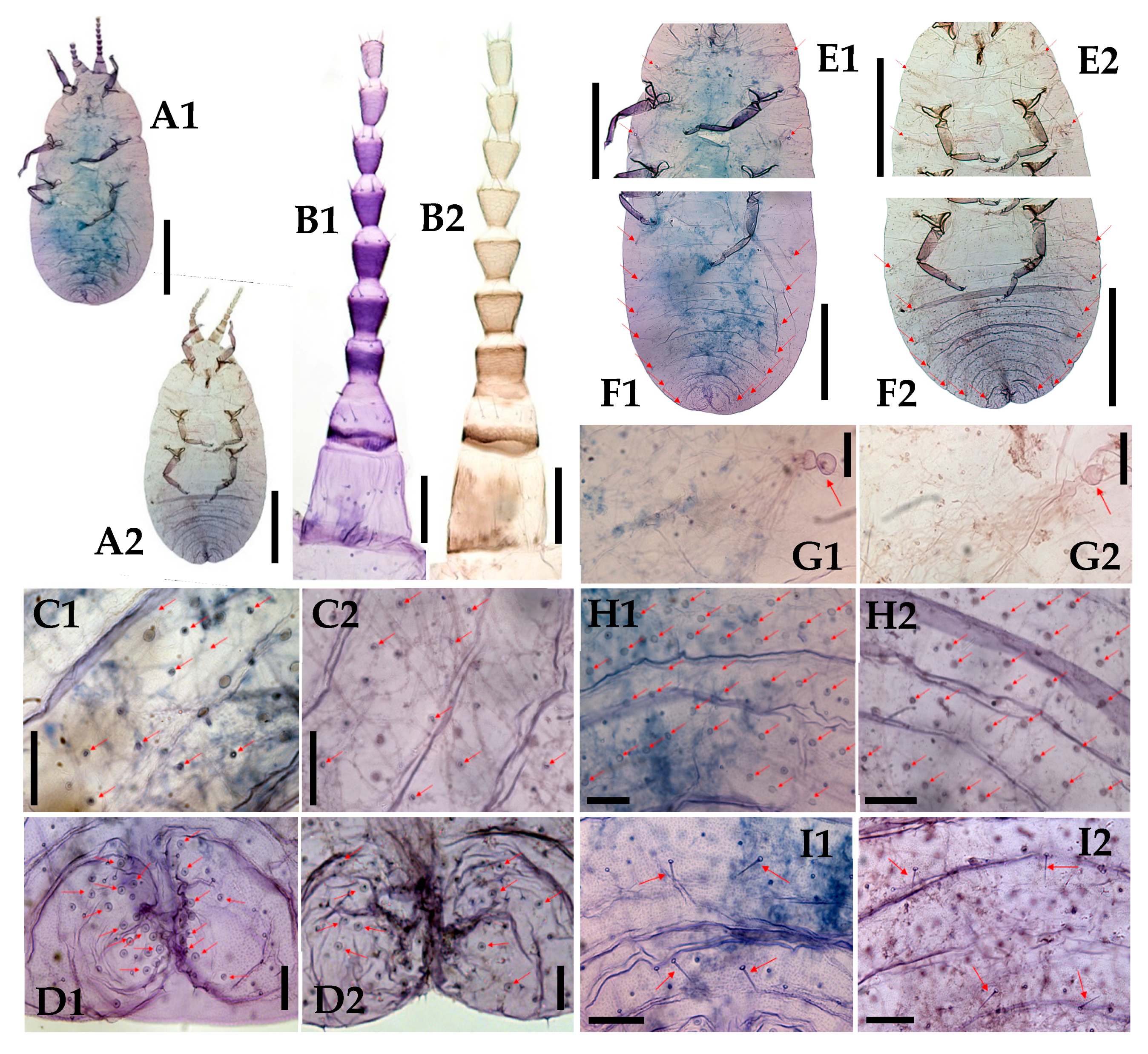
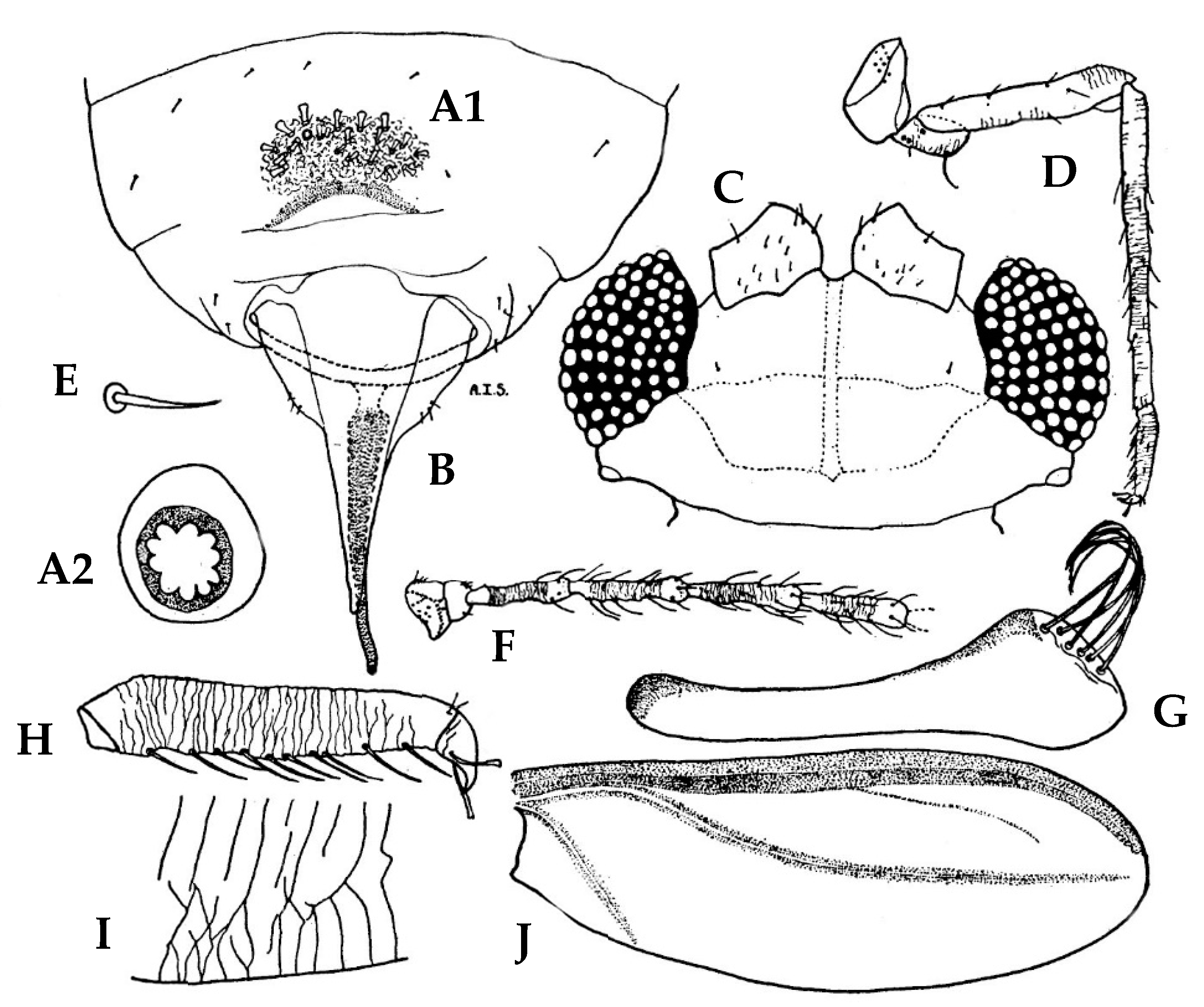
3.1. Morphological Comparison
3.1.1. Adult Females
3.1.2. Adult Males
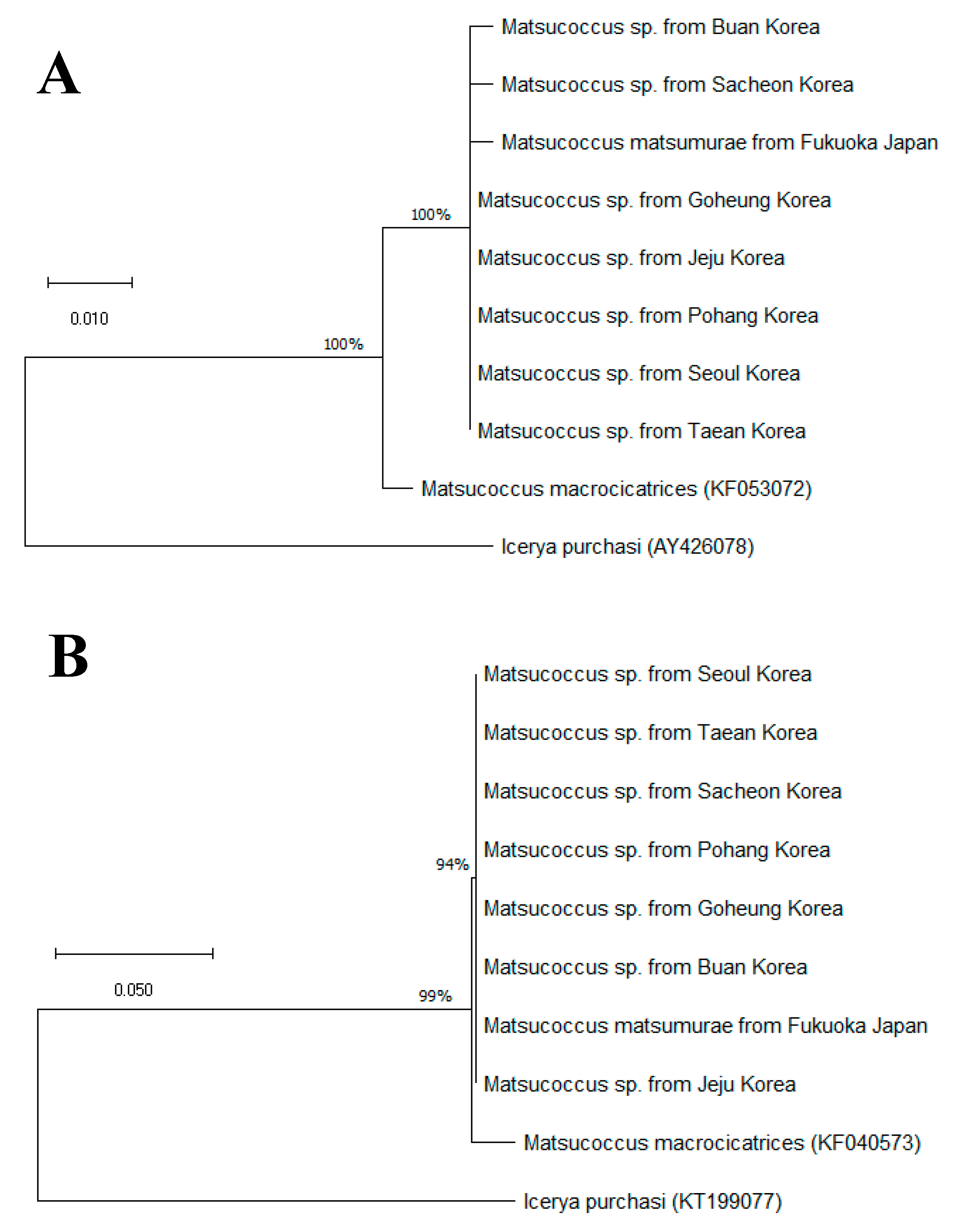
3.2. Molecular Comparison
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cockerell, T.D.A. The Japanese Coccidae. Can. Entomol. 1909, 41, 55–56. [Google Scholar] [CrossRef]
- García Morales, M.; Denno, B.D.; Miller, D.R.; Miller, G.L.; Ben-Dov, Y.; Hardy, N.B. ScaleNet: A literature-Based Model of Scale Insect Biology and Systematics. Database 2016. Available online: http://scalenet.info (accessed on 1 October 2018).
- McKenzie, H.L.; Gill, L.S.; Ellis, D.E. The prescott scale (Matsucoccus vexillorum) and associated organisms that cause flagging injury to ponderosa pine in the southwest. J. Agric. Res. 1948, 76, 33–51. [Google Scholar]
- Furniss, R.L.; Carolin, V.M. Western Forest Insects; US Department of Agriculture, Forest Service: Washington, DC, USA, 1977; Volume 1339.
- Foldi, I. The Matsucoccidae in the Mediterranean basin with a world list of species (Hemiptera: Sternorrhyncha: Coccoidea). Annales de la Société Entomologique de France 2004, 40, 145–168. [Google Scholar] [CrossRef]
- Lim, E.; Kim, D.S.; Lee, S.M.; Choi, K.S.; Lee, D.W.; Chung, Y.J.; Park, C.G. Effect of fenitrothion on different life stages of black pine bast scale, Matsucoccus thunbergianae. J. Asia Pac. Entomol. 2013, 16, 55–59. [Google Scholar] [CrossRef]
- Liu, W.; Xie, Y.; Yang, Q.; Xue, J.; Tian, F. New Research on Matsucoccus matsumurae (Kuwana) (Hemiptera: Matsucoccidae) in China. Acta Zool. Bulg. 2014, 6, 95–102. [Google Scholar]
- Kuwana, S.I. A new Xylococcus in Japan. Insect World 1905, 9, 91–95. [Google Scholar]
- Liu, W.; Xie, Y.; Xue, J.; Zhang, Y.; Tian, F.; Yang, Q.; Wu, J.; Tang, X.; Geng, Y.; Zhang, Y.; et al. Morphology, behavior and natural enemies of Matsucoccus matsumurae (Homoptera: Matsucoccidae) during development. Sci. Silvae Sin. 2015, 51, 69–83. [Google Scholar]
- Kanda, S. Studies on Coccidae from Corea. Insect World 1941, 45, 296–303. [Google Scholar]
- Miller, D.R.; Park, S.C. A new species of Matsucoccus (Homoptera: Coccoidea: Margarodidae) from Korea. Korean J. Plant Prot. 1987, 26, 49–62. [Google Scholar]
- Young, B.L.; Miller, D.R.; McClure, M.S. Attractivity of the female sex pheromone of Chinese Matsucoccus matsumurae (Kuwana) to males of M. matsumurae in Japan and to males of M. resinosae Bean and Godwin in the United States (Margarodidae, Coccoidea, Homoptera). Contrib. Shanghai Inst. Entomol. 1984, 1984, 1–20. [Google Scholar]
- Park, S.C.; West, J.R.; Abrahamson, L.P.; Lanier, G.N.; Silverstein, R.M. Cross-attraction between two species of Matsucoccus. J. Chem. Ecol. 1986, 12, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Lanier, G.N.; Qi, Y.T.; West, J.R.; Park, S.C.; Webster, F.X.; Silverstein, R.M. Identification of the sex pheromone of three Matsucoccus pine bast scales. J. Chem. Ecol. 1989, 15, 1645–1659. [Google Scholar] [CrossRef] [PubMed]
- Hibbard, B.E.; Lanier, G.N.; Parks, S.C.; Qi, Y.T.; Webster, F.X.; Silverstein, R.M. Laboratory and field tests with the synthetic sex pheromone of three Matsucoccus pine bast scales. J. Chem. Ecol. 1991, 17, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Booth, J.M.; Gullan, P.J. Synonymy of three pestiferous Matsucoccus scale insects (Hemiptera: Coccoidea: Matsucoccidae) based on morphological and molecular evidence. Proc. Entomol. Soc. Wash. 2006, 108, 749–760. [Google Scholar]
- Lim, E.G.; Lee, S.M.; Kim, D.S.; Kim, J.B.; Lee, S.H.; Choi, K.S.; Park, C.G.; Lee, D.W. The spread of black pine bast scale, Matsucoccus thunbergianae (Hemiptera: Margarodidae) in Korea. Korean J. Appl. Entomol. 2012, 51, 59–65. [Google Scholar] [CrossRef]
- Kim, J.; Kim, D.S.; Matsuyama, S.; Lee, S.M.; Lee, S.C.; Park, I.K. Development of a pheromone trap for monitor black pine bast scale, Matsucoccus thunbergianae (Hemiptera: Margarodidae). J. Asia Pac. Entomol. 2016, 19, 899–902. [Google Scholar] [CrossRef]
- Lee, C.J.; Kim, D.S.; Chung, Y.H.; Lee, S.M.; Lee, S.J.; Lee, D.W. Monitoring of black pine bast scale, Matsucoccus thunbergianae (Homoptera: Margarodidae) using yellow sticky trap. Korean J. Appl. Entomol. 2018, 57, 143–149. [Google Scholar]
- Danzig, E.M.; Gavrilov-Zimin, I.A. Palaearctic Mealybugs (Homoptera: Coccinea: Pseudococcidae), Part 1: Subfamily Phenacoccinae; Russian Academy of Sciences, Zoological Institute: St. Petersburg, Russia, 2014. [Google Scholar]
- Morrison, H. A Classification of the Higher Groups and Genera of the Coccid Family Margarodidae (No. 52); US Department of Agriculture Technical Bulletin: Washington, DC, USA, 1928.
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Kimura, M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef]
- Boratynski, K.L. Matsucoccus pini (Green, 1925) (Homoptera, Coccoidea: Margarodidae): Bionomics and external anatomy with reference to the variability of some taxonomic characters. Trans. R. Entomol. Soc. Lond. 1952, 103, 285–326. [Google Scholar] [CrossRef]
- Rieux, R. Matsucoccus pini Green (1925) (Homoptera, Margarodidae) dans le Sud-Est de la France. Variations intraspécifiques. Comparaison avec des espèces les plus proches. Annales de Zoologie Ecologie Animale 1976, 8, 231–263. [Google Scholar]
- Ben-Dov, Y. Redescription of Matsucoccus josephi Bodenheimer and Harpaz (Homoptera: Coccoidea: Margarodidae). Isr. J. Entomol. 1981, 15, 35–51. [Google Scholar]
- McClure, M.S. Temperature and host availability affect the distribution of Matsucoccus matsumurae (Kuwana) (Homoptera: Margarodidae) in Asia and North America. Ann. Entomol. Soc. Am. 1983, 76, 761–765. [Google Scholar] [CrossRef]
- Wang, X.; Xie, Y.; Zhang, Y.; Liu, W.; Wu, J. The structure and morphogenic changes of antennae of Matsucoccus matsumurae (Hemiptera: Coccoidea: Matsucoccidae) in different instars. Arthropod Struct. Dev. 2016, 45, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Tian, F.; Liu, W.; Zhang, Y.; Xue, J.; Zhao, Y.; Wu, J. The wax glands and wax secretion of Matsucoccus matsumurae at different development stages. Arthropod Struct. Dev. 2014, 43, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Paik, W.H. Illustrated Flora and Fauna of Korea. Insecta (VIl); No. 22; Min. Education: Seoul, Korea, 1978.
- Kong, W.S. Species composition and distribution of native Korean conifers. J. Korean Geogr. Soc. 2004, 39, 528–543. [Google Scholar]
- Kong, W.S. Biogeography of native Korean Pinaceae. J. Korean Geogr. Soc. 2006, 41, 73–93. [Google Scholar]
- Kong, W.S.; Lee, S.G.; Park, H.N.; Lee, Y.M.; Oh, S.H. Time-spatial distribution of Pinus in the Korean Peninsula. Quat. Int. 2014, 344, 43–52. [Google Scholar] [CrossRef]
- Kim, K.H.; Zsuffa, L. Reforestation of South Korea: The history and analysis of a unique case in forest tree improvement and forestry. For. Chron. 1994, 70, 58–64. [Google Scholar] [CrossRef]
- Gullan, P.J.; Kosztarab, M.P. Adaptations in scale insects. Annu. Rev. Entomol. 1997, 42, 23–50. [Google Scholar] [CrossRef]
- Kondo, T.; Gullan, P.J.; Williams, D.J. Coccidology. The study of scale insects (Hemiptera: Sternorrhyncha: Coccoidea). Ciencia y Tecnología Agropecuaria 2008, 9, 55–61. [Google Scholar] [CrossRef]

| Species | Sex | Host | Locality | GPS | Date | Collector | GenBank Accession No. | ||
|---|---|---|---|---|---|---|---|---|---|
| North | East | 18S | 28S | ||||||
| Matsucoccus sp. | Female | Pinus thunbergii | Goheung, South Korea | 34.631599 | 127.380838 | Jan., 2017 | D. Cha | - | - |
| Matsucoccus sp. | Male | Pinus thunbergii | Goheung, South Korea | 34.631599 | 127.380838 | Jan., 2017 | D. Cha | MH574839 | MH574783 |
| Matsucoccus sp. | Male | Pinus thunbergii | Buan, South Korea | 35.596690 | 126.486645 | Jan., 2017 | D. Cha | MH574841 | MH574785 |
| Matsucoccus sp. | Male | Pinus thunbergii | Jeju, South Korea | 33.530302 | 126.718108 | Jan., 2017 | D. Cha | MH574845 | MH574789 |
| Matsucoccus sp. | Male | Pinus thunbergii | Pohang, South Korea | 36.055884 | 129.576762 | Jan., 2017 | D. Cha | MH574843 | MH574787 |
| Matsucoccus sp. | Male | Pinus thunbergii | Sacheon, South Korea | 34.948513 | 128.050597 | Jan., 2017 | D. Cha | MH574840 | MH574784 |
| Matsucoccus sp. | Male | Pinus densiflora | Seoul, South Korea | 37.598240 | 127.024651 | Jan., 2017 | D. Cha | MH574844 | MH574788 |
| Matsucoccus sp. | Male | Pinus thunbergii | Taean, South Korea | 36.781692 | 126.132603 | Jan., 2017 | D. Cha | MH574842 | MH574786 |
| Matsucoccus sp. | Female | Pinus thunbergii | Fukuoka, Japan | 33.580591 | 130.278336 | Jan., 2017 | D. Cha | - | - |
| Matsucoccus sp. | Male | Pinus thunbergii | Fukuoka, Japan | 33.580591 | 130.278336 | Jan., 2017 | D. Cha | MH574846 | MH574790 |
| Gene Regions | Direction | Primer Name | Sequences (5′–3′) | Annealing Temperature |
|---|---|---|---|---|
| 18S | Forward | Matsu_18S_F | CATGTCTAAGTGCAAGCCGG | 60 °C |
| Reverse | Matsu_18S_R | CCTCATAAGAGTCCCGTATCG | 60 °C | |
| 28S | Forward | Matsu_28S_F | AAACCACAGCCAAGGGAACG | 60 °C |
| Reverse | Matsu_28S_R | TTTTCTGACACCTCTCGCTG | 60 °C |
| No. | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Matsucoccus matsumurae from Fukuoka, Japan | ||||||||||
| 2 | Matsucoccus sp. from Buan, Korea | 0.6 | |||||||||
| 3 | Matsucoccus sp. from Goheung, Korea | 0.3 | 0.3 | ||||||||
| 4 | Matsucoccus sp. from Jeju, Korea | 0.3 | 0.3 | 0.0 | |||||||
| 5 | Matsucoccus sp. from Pohang, Korea | 0.3 | 0.3 | 0.0 | 0.0 | ||||||
| 6 | Matsucoccus sp. from Sacheon, Korea | 0.6 | 0.6 | 0.3 | 0.3 | 0.3 | |||||
| 7 | Matsucoccus sp. from Seoul, Korea | 0.3 | 0.3 | 0.0 | 0.0 | 0.0 | 0.3 | ||||
| 8 | Matsucoccus sp. from Taean, Korea | 0.3 | 0.3 | 0.0 | 0.0 | 0.0 | 0.3 | 0.0 | |||
| 9 | Matsucoccus macrocicatrices (KF053072) | 1.7 | 1.7 | 1.4 | 1.4 | 1.4 | 1.7 | 1.4 | 1.4 | ||
| 10 | Icerya purchasi (AY426078) | 11.2 | 11.2 | 10.8 | 10.8 | 10.8 | 11.2 | 10.8 | 10.8 | 10.2 |
| No. | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Matsucoccus matsumurae from Fukuoka, Japan | ||||||||||
| 2 | Matsucoccus sp. from Buan, Korea | 0.0 | |||||||||
| 3 | Matsucoccus sp. from Goheung, Korea | 0.0 | 0.0 | ||||||||
| 4 | Matsucoccus sp. from Jeju, Korea | 0.0 | 0.0 | 0.0 | |||||||
| 5 | Matsucoccus sp. from Pohang, Korea | 0.0 | 0.0 | 0.0 | 0.0 | ||||||
| 6 | Matsucoccus sp. from Sacheon, Korea | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | |||||
| 7 | Matsucoccus sp. from Seoul, Korea | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | ||||
| 8 | Matsucoccus sp. from Taean, Korea | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | |||
| 9 | Matsucoccus macrocicatrices (KF040573) | 1.5 | 1.5 | 1.5 | 1.6 | 1.5 | 1.5 | 1.5 | 1.5 | ||
| 10 | Icerya purchasi (KT199077) | 28.7 | 28.7 | 28.7 | 30.4 | 28.7 | 28.7 | 28.7 | 28.7 | 29.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, J.; Cha, D.; Kim, D.-S.; Lee, S. Review of Japanese Pine Bast Scale, Matsucoccus matsumurae (Kuwana) (Coccomorpha: Matsucoccidae), Occurring on Japanese Black Pine (Pinus thunbergii Parl.) and Japanese Red Pine (P. densiflora Siebold & Zucc.) from Korea. Forests 2019, 10, 639. https://doi.org/10.3390/f10080639
Choi J, Cha D, Kim D-S, Lee S. Review of Japanese Pine Bast Scale, Matsucoccus matsumurae (Kuwana) (Coccomorpha: Matsucoccidae), Occurring on Japanese Black Pine (Pinus thunbergii Parl.) and Japanese Red Pine (P. densiflora Siebold & Zucc.) from Korea. Forests. 2019; 10(8):639. https://doi.org/10.3390/f10080639
Chicago/Turabian StyleChoi, Jinyeong, Deokjea Cha, Dong-Soo Kim, and Seunghwan Lee. 2019. "Review of Japanese Pine Bast Scale, Matsucoccus matsumurae (Kuwana) (Coccomorpha: Matsucoccidae), Occurring on Japanese Black Pine (Pinus thunbergii Parl.) and Japanese Red Pine (P. densiflora Siebold & Zucc.) from Korea" Forests 10, no. 8: 639. https://doi.org/10.3390/f10080639
APA StyleChoi, J., Cha, D., Kim, D.-S., & Lee, S. (2019). Review of Japanese Pine Bast Scale, Matsucoccus matsumurae (Kuwana) (Coccomorpha: Matsucoccidae), Occurring on Japanese Black Pine (Pinus thunbergii Parl.) and Japanese Red Pine (P. densiflora Siebold & Zucc.) from Korea. Forests, 10(8), 639. https://doi.org/10.3390/f10080639




