Effect of Woodchips Biochar on Sensitivity to Temperature of Soil Greenhouse Gases Emissions
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Set Up and Soil and Biochar Characteristics
2.2. Measurement of Soil GHG Fluxes
2.3. Data Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cowie, A.; Barton, C.; Singh, B.; Ximenes, F.; Stone, C. Climate Change Impacts and Research Priorities for the Forestry Sector. In DPI Priority Actions for Climate Change Workshop; NSW Department of Primary Industries: Orange, NSW, Australia, 2007. [Google Scholar]
- Lehmann, J. A Handful of Carbon. Nature 2007, 447, 143–144. [Google Scholar] [CrossRef] [PubMed]
- Bridgwater, A.V. The Production of Biofuels and Renewable Chemicals by Fast Pyrolysis of Biomass. Int. J. Glob. Energy Issues 2007, 27, 160–203. [Google Scholar] [CrossRef]
- Lehmann, J. Bio-Energy in the Black. Front. Ecol. Environ. 2007, 5, 381–387. [Google Scholar] [CrossRef]
- Downie, A.; Munroe, P. Characteristics of Biochar—Physical and Structural Properties. In Biochar for Environmental Management: Science, Technology and Implementation; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2009; pp. 13–29. [Google Scholar]
- Lehmann, J.; Abiven, S.; Kleber, M.; Pan, G.; Singh, B.P.; Sohi, S.P.; Zimmerman, A.R. Persistence of Biochar in Soil. In Biochar for Environmental Management: Science, Technology and Implementation; Lehmann, J., Joseph, S., Eds.; Routledge: New York, NY, USA, 2015; pp. 235–282. [Google Scholar]
- Criscuoli, I.; Alberti, G.; Baronti, S.; Favilli, F.; Martinez, C.; Calzolari, C.; Pusceddu, E.; Rumpel, C.; Viola, R.; Miglietta, F. Carbon Sequestration and Fertility after Centennial Time Scale Incorporation of Charcoal into Soil. PLoS ONE 2014, 9, e91114. [Google Scholar] [CrossRef] [PubMed]
- Gurwick, N.P.; Moore, L.A.; Kelly, C.; Elias, P. A Systematic Review of Biochar Research, with a Focus on Its Stability in Situ and Its Promise as a Climate Mitigation Strategy. PLoS ONE 2013, 8, e75932. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.C.; Gale, N. Biochar and Forest Restoration: A Review and Meta-Analysis of Tree Growth Responses. New For. 2015, 46, 931–946. [Google Scholar] [CrossRef]
- Biederman, L.; Harpole, W.S. Biochar and Its Effects on Plant Productivity and Nutrient Cycling: A Meta-Analysis. GCB Bioenergy 2012, 5, 202–214. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, A.; Ji, C.; Joseph, S.; Bian, R.; Li, L.; Pan, G.; Paz-Ferreiro, J. Biochar’s Effect on Crop Productivity and the Dependence on Experimental Conditions—A Meta-Analysis of Literature Data. Plant Soil 2013, 373, 583–594. [Google Scholar] [CrossRef]
- Criscuoli, I.; Baronti, S.; Alberti, G.; Rumpel, C.; Giordan, M.; Camin, F.; Ziller, L.; Martinez, C.; Pusceddu, E.; Miglietta, F. Anthropogenic Charcoal-Rich Soils of the XIX Century Reveal That Biochar Leads to Enhanced Fertility and Fodder Quality of Alpine Grasslands. Plant Soil 2017, 411, 499–516. [Google Scholar] [CrossRef]
- Li, Y.; Hu, S.; Chen, J.; Mueller, K.; Li, Y.; Fu, W.; Lin, Z.; Wang, H. Effects of Biochar Application in Forest Ecosystems on Soil Properties and Greenhouse Gas Emissions: A Review. J. Soils Sediments 2018, 18, 546–563. [Google Scholar] [CrossRef]
- Ventura, M.; Sorrenti, G.; Panzacchi, P.; George, E.; Tonon, G. Biochar Reduces Short-Term Nitrate Leaching from a Horizon in an Apple Orchard. J. Environ. Qual. 2013, 42, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Major, J.; Steiner, C.; Downie, A.; Lehmann, J. Biochar Effects on Nutrient Leaching. In Biochar for Environmental Management: Science, Technology and Implementation; Lehmann, J., Joseph, S., Eds.; Routledge, Earthscam: New York, NY, USA, 2015; pp. 271–287. [Google Scholar]
- Sorrenti, G.; Ventura, M.; Toselli, M. Effect of Biochar on Nutrient Retention and Nectarine Tree Performance: A Three-Year Field Trial. J. Plant Nutr. Soil Sci. 2016, 179, 1–11. [Google Scholar] [CrossRef]
- Johnson, M.S.; Webster, C.; Jassal, R.S.; Hawthorne, I.; Black, T.A. Biochar Influences on Soil CO2 and CH4 Fluxes in Response to Wetting and Drying Cycles for a Forest Soil. Sci. Rep. 2017, 7, 6480. [Google Scholar] [CrossRef] [PubMed]
- Stavi, I. Biochar Use in Forestry and Tree-Based Agro-Ecosystems for Increasing Climate Change Mitigation and Adaptation. Int. J. Sustain. Dev. World Ecol. 2013, 20, 166–181. [Google Scholar] [CrossRef]
- Solomon, S.; Qin, D.; Manning, M.; Chen, Z.; Marquis, M.; Averyt, K.B.; Tignor, M.M.B.; Miller, H.L. Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2007. [Google Scholar]
- Oertel, C.; Matschullat, J.; Zurba, K.; Zimmermann, F.; Erasmi, S. Greenhouse Gas Emissions from Soils—A Review. Chemie der Erde—Geochemistry 2016, 76, 327–352. [Google Scholar] [CrossRef]
- Case, S.D.; McNamara, N.P.; Reay, D.S.; Whitaker, J. Can Biochar Reduce Soil Greenhouse Gas Emissions from a Miscanthus Bioenergy Crop? GCB Bioenergy 2014, 6, 76–89. [Google Scholar] [CrossRef]
- Novak, J.M.; Busscher, W.J.; Watts, D.W.; Laird, D.A.; Ahmedna, M.A.; Niandou, M.A.S. Short-Term CO2 Mineralization after Additions of Biochar and Switchgrass to a Typic Kandiudult. Geoderma 2010, 154, 281–288. [Google Scholar] [CrossRef]
- Ventura, M.; Alberti, G.; Panzacchi, P.; Delle Vedove, G.; Miglietta, F.; Tonon, G. Biochar Mineralization and Priming Effect on SOM Decomposition. Results from a Field Trial in a Short Rotation Coppice in Italy. In EGU General Assembly Conference Abstracts; EGU General Assembly: Vienna, Austria, 2016; Volume 18, p. 9109. [Google Scholar]
- Scheer, C.; Grace, P.R.; Rowlings, D.W.; Kimber, S.; van Zwieten, L. Effect of Biochar Amendment on the Soil-Atmosphere Exchange of Greenhouse Gases from an Intensive Subtropical Pasture in Northern New South Wales, Australia. Plant Soil 2011, 345, 47–58. [Google Scholar] [CrossRef]
- Wang, Z.; Li, Y.; Chang, S.X.; Zhang, J.; Jiang, P.; Zhou, G.; Shen, Z. Contrasting Effects of Bamboo Leaf and Its Biochar on Soil CO2 Efflux and Labile Organic Carbon in an Intensively Managed Chinese Chestnut Plantation. Biol. Fertil. Soils 2014, 50, 1109–1119. [Google Scholar] [CrossRef]
- Ventura, M.; Zhang, C.; Baldi, E.; Fornasier, F.; Sorrenti, G.; Panzacchi, P.; Tonon, G. Effect of Biochar Addition on Soil Respiration Partitioning and Root Dynamics in an Apple Orchard. Eur. J. Soil Sci. 2014, 65, 186–195. [Google Scholar] [CrossRef]
- Jeffery, S.; Verheijen, F.G.A.; Kammann, C.; Abalos, D. Biochar Effects on Methane Emissions from Soils: A Meta-Analysis. Soil Biol. Biochem. 2016, 101, 251–258. [Google Scholar] [CrossRef]
- Cayuela, M.L.; van Zwieten, L.; Singh, B.P.; Jeffery, S.; Roig, A.; Sánchez-Monedero, M.A. Biochar’s Role in Mitigating Soil Nitrous Oxide Emissions: A Review and Meta-Analysis. Agric. Ecosyst. Environ. 2014, 191, 5–16. [Google Scholar] [CrossRef]
- Reichstein, M.; Beer, C. Soil Respiration across Scales: The Importance of a Model-Data Integration Framework for Data Interpretation. J. Plant Nutr. Soil Sci. 2008, 171, 344–354. [Google Scholar] [CrossRef]
- Shen, Y.; Zhu, L.; Cheng, H.; Yue, S.; Li, S. Effects of Biochar Application on CO2 Emissions from a Cultivated Soil under Semiarid Climate Conditions in Northwest China. Sustainability 2017, 9, 1482. [Google Scholar] [CrossRef]
- Nguyen, B.T.; Lehmann, J.; Hockaday, W.C.; Joseph, S.; Masiello, C.A. Temperature Sensitivity of Black Carbon Decomposition and Oxidation. Environ. Sci. Technol. 2010, 44, 3324–3331. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, J.; Taylor, A. On the Temperature Dependence of Soil Respiration. Funct. Ecol. 1994, 8, 315–323. [Google Scholar] [CrossRef]
- Provincia Autonoma di Bolzano. Valori Medi Delle Temperature Massime e Minime—Merano/Quarazze cod. 23200MS. Available online: http://www.provincia.bz.it/meteo/download/23200MS-TS-MeranoQuarazze-MeranGratsch.pdf (accessed on 8 July 2019).
- Ihaka, R.; Gentleman, R. R: A Language for Data Analysis and Graphics. J. Comput. Graph. Stat. 1996, 5, 299–314. [Google Scholar]
- Luo, Y.; Wan, S.; Hui, D.; Wallace, L.L. Acclimatization of Soil Respiration to Warming in a Tall Grass Prairie. Nature 2001, 413, 622–625. [Google Scholar] [CrossRef]
- Zhou, T.; Shi, P.; Hui, D.; Luo, Y. Global Pattern of Temperature Sensitivity of Soil Heterotrophic Respiration (Q10) and Its Implications for Carbon-Climate Feedback. J. Geophys. Res. 2009, 114, 1–9. [Google Scholar] [CrossRef]
- Lu, X.; Li, Y.; Wang, H.; Singh, B.P.; Hu, S.; Luo, Y.; Li, J.; Xiao, Y.; Cai, X.; Li, Y. Responses of Soil Greenhouse Gas Emissions to Different Application Rates of Biochar in a Subtropical Chinese Chestnut Plantation. Agric. For. Meteorol. 2019, 271, 168–179. [Google Scholar] [CrossRef]
- Li, Y.; Li, Y.; Chang, S.X.; Yang, Y.; Fu, S.; Jiang, P.; Luo, Y.; Yang, M.; Chen, Z.; Hu, S.; et al. Biochar Reduces Soil Heterotrophic Respiration in a Subtropical Plantation through Increasing Soil Organic Carbon Recalcitrancy and Decreasing Carbon-Degrading Microbial Activity. Soil Biol. Biochem. 2018, 122, 173–185. [Google Scholar] [CrossRef]
- Bamminger, C.; Poll, C.; Marhan, S. Offsetting Global Warming-Induced Elevated Greenhouse Gas Emissions from an Arable Soil by Biochar Application. Glob. Chang. Biol. 2018, 24, 318–334. [Google Scholar] [CrossRef] [PubMed]
- Pei, J.; Zhuang, S.; Cui, J.; Li, J.; Li, B.; Wu, J.; Fang, C. Biochar Decreased the Temperature Sensitivity of Soil Carbon Decomposition in a Paddy Field. Agric. Ecosyst. Environ. 2017, 249, 156–164. [Google Scholar] [CrossRef]
- Chen, J.; Sun, X.; Zheng, J.; Zhang, X.; Liu, X.; Bian, R.; Li, L.; Chneg, K.; Zheng, J.; Pan, G. Biochar Amendment Changes Temperature Sensitivity of Soil Respiration and Composition of Microbial Communities 3 Years after Incorporation in an Organic Carbon-Poor Dry Cropland Soil. Biol. Fertil. Soils 2018, 54, 175–188. [Google Scholar] [CrossRef]
- He, X.; Du, Z.; Wang, Y.; Lu, N.; Zhang, Q. Sensitivity of Soil Respiration to Soil Temperature Decreased under Deep Biochar Amended Soils in Temperate Croplands. Appl. Soil Ecol. 2016, 108, 204–210. [Google Scholar] [CrossRef]
- Zhou, G.; Zhou, X.; Zhang, T.; Du, Z.; He, Y.; Wang, X.; Shao, J.; Cao, Y.; Xue, S.; Wang, H.; et al. Biochar Increased Soil Respiration in Temperate Forests but Had No Effects in Subtropical Forests. For. Ecol. Manage. 2017, 405, 339–349. [Google Scholar] [CrossRef]
- Conant, R.; Steinweg, J.; Haddix, M.; Paul, E.; Plante, A.; Six, J. Experimental Warming Shows That Decomposition Temperature Sensitivity Increases with Soil Organic Matter Recalcitrance. Ecology 2008, 89, 2384–2391. [Google Scholar] [CrossRef]
- Fang, Y.; Singh, B.P.; Matta, P.; Cowie, A.L.; Van Zwieten, L. Temperature Sensitivity and Priming of Organic Matter with Different Stabilities in a Vertisol with Aged Biochar. Soil Biol. Biochem. 2017, 115, 346–356. [Google Scholar] [CrossRef]
- Fang, Y.; Singh, B.P.; Singh, B. Temperature Sensitivity of Biochar and Native Carbon Mineralisation in Biochar-Amended Soils. Agric. Ecosyst. Environ. 2014, 191, 158–167. [Google Scholar] [CrossRef]
- Liao, N.; Li, Q.; Zhang, W.; Zhou, G.; Ma, L.; Min, W.; Ye, J.; Hou, Z. Effects of Biochar on Soil Microbial Community Composition and Activity in Drip-Irrigated Desert Soil. Eur. J. Soil Biol. 2016, 72, 27–34. [Google Scholar] [CrossRef]
- Mierzwa-Hersztek, M.; Klimkowicz-Pawlas, A.; Gondek, K. Influence of Poultry Litter and Poultry Litter Biochar on Soil Microbial Respiration and Nitrifying Bacteria Activity. Waste Biomass Valorization 2018, 9, 379–389. [Google Scholar] [CrossRef]
- Kolb, S.E.; Fermanich, K.J.; Dornbush, M.E. Effect of Charcoal Quantity on Microbial Biomass and Activity in Temperate Soils. Soil Sci. Soc. Am. J. 2009, 73, 1173–1181. [Google Scholar] [CrossRef]
- Zimmerman, A.R. Abiotic and Microbial Oxidation of Laboratory-Produced Black Carbon (Biochar). Environ. Sci. Technol. 2010, 44, 1295–1301. [Google Scholar] [CrossRef] [PubMed]
- Kuzyakov, Y.; Bogomolova, I.; Glaser, B. Biochar Stability in Soil: Decomposition during Eight Years and Transformation as Assessed by Compound-Specific 14C Analysis. Soil Biol. Biochem. 2014, 70, 229–236. [Google Scholar] [CrossRef]
- Wang, J.; Xiong, Z.; Kuzyakov, Y. Biochar Stability in Soil: Meta-Analysis of Decomposition and Priming Effects. GCB Bioenergy 2016, 8, 512–523. [Google Scholar] [CrossRef]
- Ventura, M.; Alberti, G.; Panzacchi, P.; Delle Vedove, G.; Miglietta, F.; Tonon, G. Biochar Mineralization and Priming Effect in a Poplar Short Rotation Coppice from a 3-Year Field Experiment. Biol. Fertil. Soils 2019, 55, 67–78. [Google Scholar] [CrossRef]
- Anderson, C.R.; Condron, L.M.; Clough, T.J.; Fiers, M.; Stewart, A.; Hill, R.A.; Sherlock, R.R. Biochar Induced Soil Microbial Community Change: Implications for Biogeochemical Cycling of Carbon, Nitrogen and Phosphorus. Pedobiologia 2011, 54, 309–320. [Google Scholar] [CrossRef]
- Zimmerman, A.R.; Gao, B.; Ahn, M.Y. Positive and Negative Carbon Mineralization Priming Effects among a Variety of Biochar-Amended Soils. Soil Biol. Biochem. 2011, 43, 1169–1179. [Google Scholar] [CrossRef]
- Maestrini, B.; Nannipieri, P.; Abiven, S. A Meta-Analysis on Pyrogenic Organic Matter Induced Priming Effect. GCB Bioenergy 2015, 7, 577–590. [Google Scholar] [CrossRef]
- Fidel, R.B.; Laird, D.A.; Parkin, T.B. Impact of Six Lignocellulosic Biochars on C and N Dynamics of Two Contrasting Soils. GCB Bioenergy 2017, 9, 1279–1291. [Google Scholar] [CrossRef]
- Zhou, Z.; Guo, C.; Meng, H. Temperature Sensitivity and Basal Rate of Soil Respiration and Their Determinants in Temperate Forests of North China. PLoS ONE 2013, 8, e81793. [Google Scholar] [CrossRef] [PubMed]
- Laird, D.A.; Fleming, P.; Davis, D.D.; Horton, R.; Wang, B.; Karlen, D.L. Impact of Biochar Amendments on the Quality of a Typical Midwestern Agricultural Soil. Geoderma 2010, 158, 443–449. [Google Scholar] [CrossRef]
- Novak, J.M.; Busscher, W.J.; Watts, D.W.; Amonette, J.E.; Ippolito, J.A.; Lima, I.M.; Gaskin, J.; Das, K.C.; Steiner, C.; Ahmedna, M.; et al. Biochars Impact on Soil-Moisture Storage in an Ultisol and Two Aridisols. Soil Sci. 2012, 177, 310–320. [Google Scholar] [CrossRef]
- Sinegani, A.A.S.; Maghsoudi, J. The Effects of Water Potential on Some Microbial Populations and Decrease Kinetic of Organic Carbon in Soil Treated with Cow Manure under Laboratory Conditions. J. Appl. Sci. Environ. Manag. 2011, 15, 179–188. [Google Scholar] [CrossRef][Green Version]
- Suwanwaree, P.; Robertson, G.P. Methane Oxidation in Forest, Successional, and No-till Agricultural Ecosystems: Effects of Nitrogen and Soil Disturbance. Soil Sci. Soc. Am. J. 2005, 69, 1722–1729. [Google Scholar] [CrossRef]
- Luo, G.J.; Kiese, R.; Wolf, B.; Butterbach-Bahl, K. Effects of Soil Temperature and Moisture on Methane Uptake and Nitrous Oxide Emissions across Three Different Ecosystem Types. Biogeosciences 2013, 10, 3205–3219. [Google Scholar] [CrossRef]
- Powlson, D.S.; Goulding, K.W.T.; Willison, T.W.; Webster, C.P.; Hütsch, B.W. The Effect of Agriculture on Methane Oxidation in Soil. Nutr. Cycl. Agroeco. 1997, 49, 59–70. [Google Scholar] [CrossRef]
- Karhu, K.; Mattila, T.; Bergström, I.; Regina, K. Biochar Addition to Agricultural Soil Increased CH4 Uptake and Water Holding Capacity—Results from a Short-Term Pilot Field Study. Agric. Ecosyst. Environ. 2011, 140, 309–313. [Google Scholar] [CrossRef]
- Feng, Y.; Xu, Y.; Yu, Y.; Xie, Z.; Lin, X. Mechanisms of Biochar Decreasing Methane Emission from Chinese Paddy Soils. Soil Biol. Biochem. 2012, 46, 80–88. [Google Scholar] [CrossRef]
- Spokas, K.A.; Baker, J.M.; Reicosky, D.C. Ethylene: Potential Key for Biochar Amendment Impacts. Plant Soil 2010, 333, 443–452. [Google Scholar] [CrossRef]
- Deem, L.M.; Yu, J.; Crow, S.E.; Deenik, J.; Penton, C.R. Biochar Increases Temperature Sensitivity of Soil Respiration and N2O Flux. 2016. Available online: https://biochar-us.org/presentation/biochar-increases-temperature-sensitivity-soil-respiration-and-n2o-flux (accessed on 8 July 2019).
- Chang, J.; Clay, D.E.; Clay, S.A.; Chintala, R.; Miller, J.M.; Schumacher, T. Biochar Reduced Nitrous Oxide and Carbon Dioxide Emissions from Soil with Different Water and Temperature Cycles. Agron. Soils Environ. Qual. 2016, 108, 2214–2221. [Google Scholar] [CrossRef]
- Curtin, D.; Campbell, C.A.; Jalil, A. Effects of Acidity on Mineralization: PH-Dependence of Organic Matter Mineralization in Weakly Acidic Soils. Soil Biol. Biochem. 1998, 30, 57–64. [Google Scholar] [CrossRef]
- Maliszewska-Kordybach, B.; Klimkowicz-Pawlas, A.; Smreczak, B.; Janusauskaite, D. Ecotoxic Effect of Phenanthrene on Nitrifying Bacteria in Soils of Different Properties. J. Environ. Qual. 2007, 36, 1635–1645. [Google Scholar] [CrossRef] [PubMed]
- Guo, G.X.; Deng, H.; Qiao, M.; Yao, H.Y.; Zhu, Y.G. Effect of Long-Term Wastewater Irrigation on Potential Denitrification and Denitrifying Communities in Soils at the Watershed Scale. Environ. Sci. Technol. 2013, 47, 3105–3113. [Google Scholar] [CrossRef]
- Rogovska, N.; Laird, D.; Cruse, R.; Fleming, P.; Parkin, T.; Meek, D. Impact of Biochar on Manure Carbon Stabilization and Greenhouse Gas Emissions. Soil Sci. Soc. Am. J. 2011, 75, 871–879. [Google Scholar] [CrossRef]
- Clough, T.J.; Condron, L.M.; Kammann, C.; Müller, C. A Review of Biochar and Soil Nitrogen Dynamics. Agronomy 2013, 3, 275–293. [Google Scholar] [CrossRef]
- Singh, B.P.; Hatton, B.; Balwant, S.; Cowie, A.L.; Kathuria, A. Influence of Biochars on Nitrous Oxide Emission and Nitrogen Leaching from Two Contrasting Soils. J. Environ. Qual. 2010, 39, 1224–1235. [Google Scholar] [CrossRef]
- Fidel, R.; Laird, D.; Parkin, T. Effect of Biochar on Soil Greenhouse Gas Emissions at the Laboratory and Field Scales. Soil Syst. 2019, 3, 8. [Google Scholar] [CrossRef]
- Spokas, K.A. Impact of Biochar Field Aging on Laboratory Greenhouse Gas Production Potentials. GCB Bioenergy 2013, 5, 165–176. [Google Scholar] [CrossRef]

| Parameter | Unit | Value | Uncertainty |
|---|---|---|---|
| pH | - | 12.4 | ± 0.5 |
| Sieve fraction <5 mm | % | 100 | ± 10 |
| Sieve fraction <2 mm | % | 97 | ± 10 |
| Sieve fraction <0.5 mm | % | 70 | ± 7 |
| Max. water retention | % w/w | 86 | ± 7 |
| Ash (550 °C) | % | 31 | ± 3 |
| Total C | % | 58.9 | - |
| C from CaCO3 | % | 1.1 | - |
| Organic C | % | 57 | ± 5 |
| H:C molar ratio | - | 0.10 | ± 0.01 |
| Total N | % | 0.39 | ± 0.04 |
| Total P | % | 0.64 | |
| Total K | % | 3.5 | ± 0.5 |
| PAHs 1 | mg/kg | <1 |
| Model Parameters | ||||
|---|---|---|---|---|
| b | R10 | |||
| CO2 | ||||
| N | 0.0686 | a | 1.0101 | a |
| B1 | 0.0862 | a | 1.3532 | ab |
| B2 | 0.0923 | a | 2.1456 | b |
| CH4 | ||||
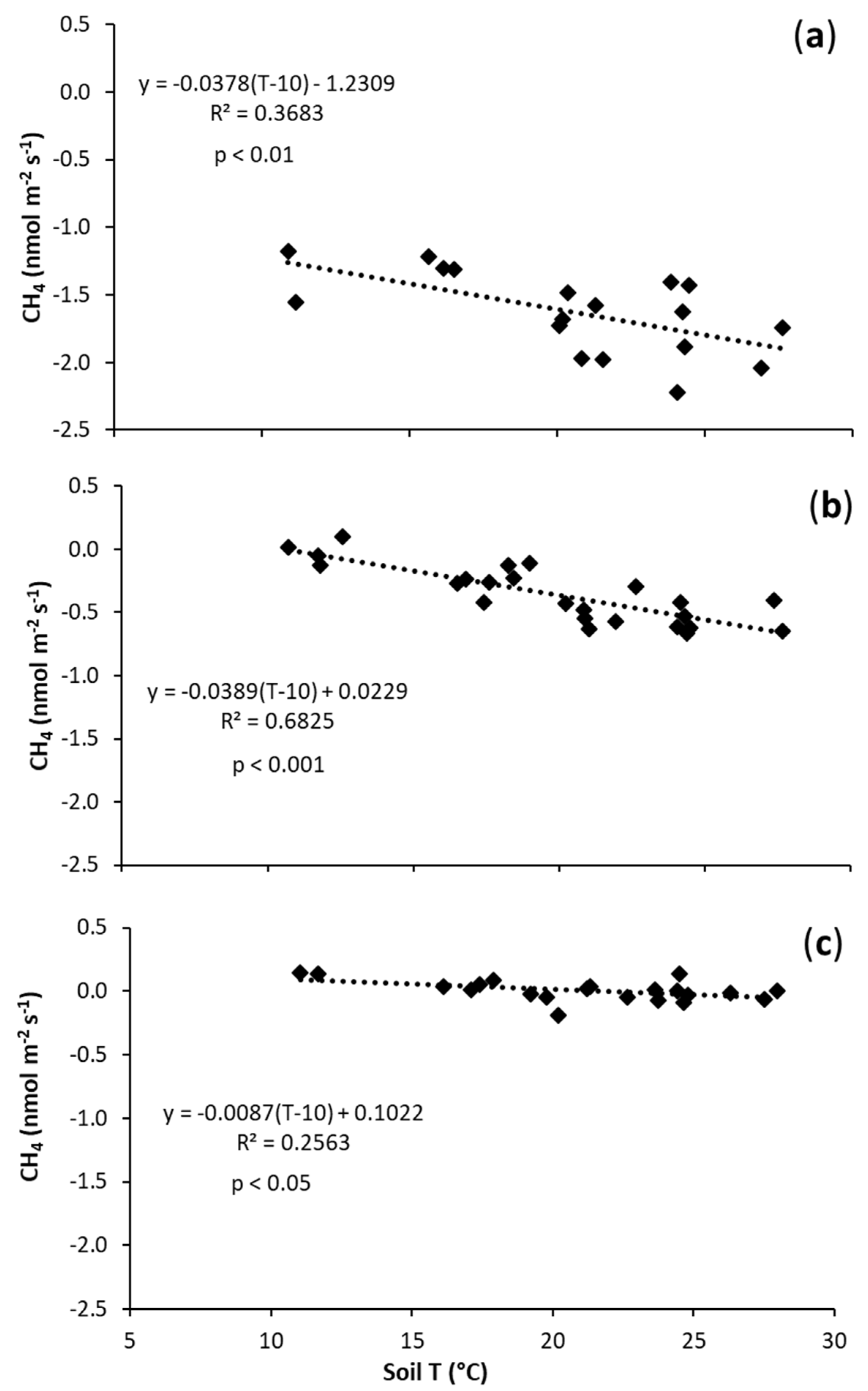
| N | −0.0378 | a | −1.2309 | a |
| B1 | −0.0389 | b | 0.0229 | a |
| B2 | −0.0087 | c | 0.1022 | a |
| N2O | ||||
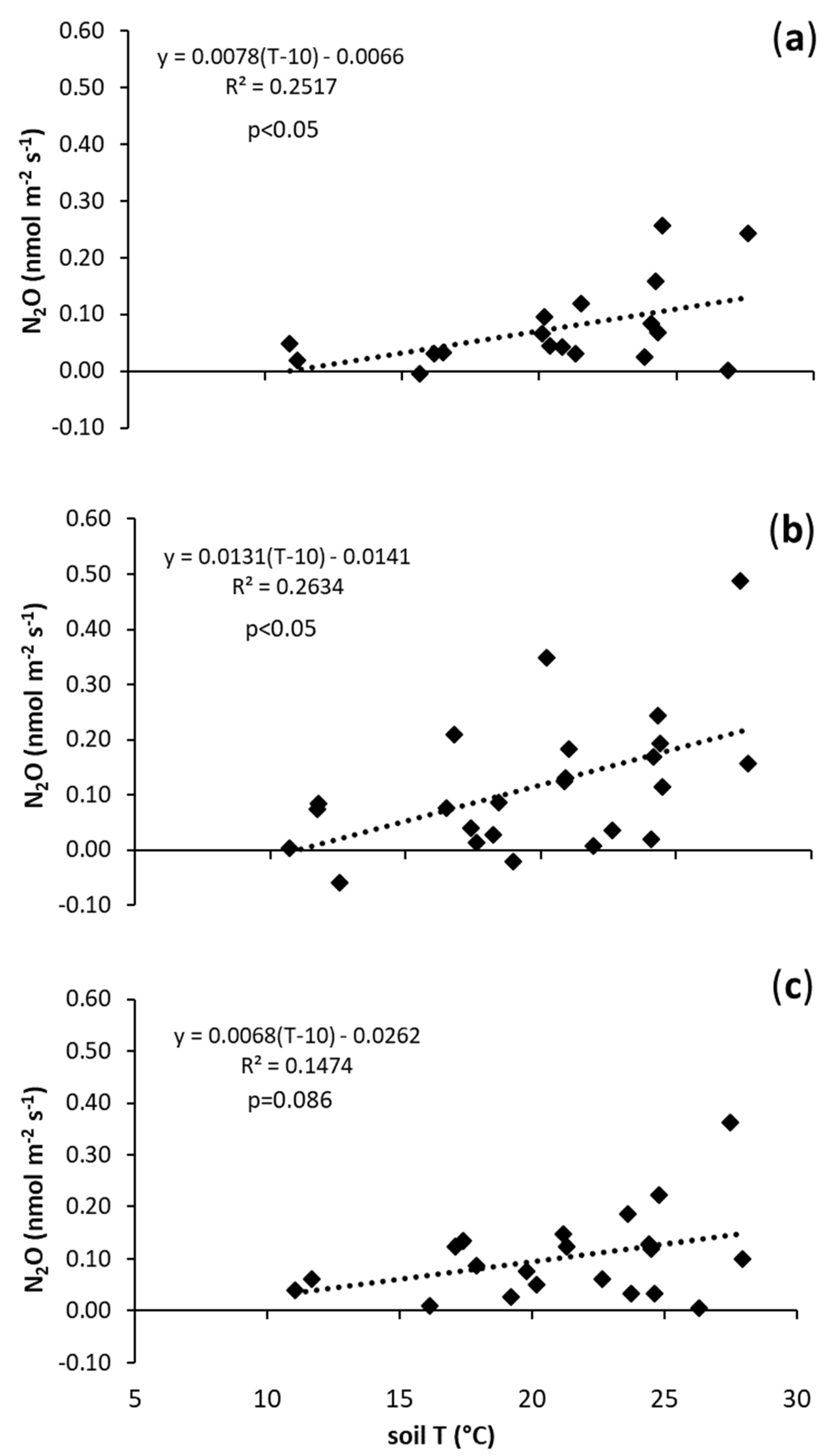
| N | 0.0078 | a | −0.0066 | a |
| B1 | 0.0131 | a | −0.0141 | a |
| B2 | 0.0068 | a | 0.0262 | a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Criscuoli, I.; Ventura, M.; Sperotto, A.; Panzacchi, P.; Tonon, G. Effect of Woodchips Biochar on Sensitivity to Temperature of Soil Greenhouse Gases Emissions. Forests 2019, 10, 594. https://doi.org/10.3390/f10070594
Criscuoli I, Ventura M, Sperotto A, Panzacchi P, Tonon G. Effect of Woodchips Biochar on Sensitivity to Temperature of Soil Greenhouse Gases Emissions. Forests. 2019; 10(7):594. https://doi.org/10.3390/f10070594
Chicago/Turabian StyleCriscuoli, Irene, Maurizio Ventura, Andrea Sperotto, Pietro Panzacchi, and Giustino Tonon. 2019. "Effect of Woodchips Biochar on Sensitivity to Temperature of Soil Greenhouse Gases Emissions" Forests 10, no. 7: 594. https://doi.org/10.3390/f10070594
APA StyleCriscuoli, I., Ventura, M., Sperotto, A., Panzacchi, P., & Tonon, G. (2019). Effect of Woodchips Biochar on Sensitivity to Temperature of Soil Greenhouse Gases Emissions. Forests, 10(7), 594. https://doi.org/10.3390/f10070594





