Abstract
Aquilaria sinensis (Lour.) Gilg is an economically important tree species that produce the highly prized agarwood. In recent years, agarwood production has been seriously threatened by the outbreak of Heortia vitessoides Moore, a leaf-eating pest that shows gregariousness during the larval stage. However, little attention has been paid to the aggregation behavior of H. vitessoides larvae. In the present study, we collected 102 cohorts of H. vitessoides larvae (13,173 individuals in total) in the wild; 54 cohorts were comprised of the same-instar larvae, and 48 cohorts were comprised of larvae with different developmental stages (instars). In general, young larvae (<third instar) tended to form large aggregations, whereas older-instar larvae were either solitary or formed small aggregations. Laboratory studies showed a strong aggregation tendency in the newly hatched and second-instar larvae of H. vitessoides, whenever the individuals originated from the same or different sibling cohorts. In addition, all newly hatched larvae died within two days after they were isolated. When newly hatched larvae were initially assigned in 10-larvae cohorts (containing sibling individuals) or 20-larvae cohorts (either containing individuals originating from the same or different sibling cohorts), their larval survivorship, duration of larval stage, and adult emergence were not significantly different. Interestingly, combining avermectin-treated larvae (donors) with untreated ones (receptors) significantly decreased larval survivorship and adult emergence of receptors, indicating a horizontal transfer of avermectin among H. vitessoides larvae. This study enhances our understanding of the population ecology of H. vitessoides, and may bring novel insights into the management strategies against this pest.
1. Introduction
Aquilaria sinensis (Lour.) Gilg is a tree species distributed in southern China and Southeast Asia. This tree is one of the main sources of the fragrant product “agarwood”, which has been widely used in traditional medicines, fragrance industries, and religious ceremonies. As a result of the scarcity and high demand of agarwood, the natural sources of A. sinensis trees have been seriously damaged by illegal logging and trade. Nowadays, A. sinensis and all other species in the genus Aquilaria are classified as “Endangered” in Appendix II of the Convention on International Trade in Endangered Species of Fauna and Flora [1], and the international trade of wild-originated agarwood is strictly monitored and limited. To increase agarwood supply, A. sinensis trees have been planted on large scales in many provinces (e.g., Guangdong, Guangxi, Hainan, and Fujian) in southern China. Meanwhile, many efforts have been made to develop artificial agarwood-producing methods based on physical wounding of the trunks or injecting agarwood-induced chemicals into the xylem [2,3,4,5].
The cultivation of A. sinensis trees and modern agarwood-inducing techniques not only conserve wild A. sinensis resources but also increase the income of farmers [6]. However, the outbreak of a lepidopteran pest, Heortia vitessoides Moore (Lepidoptera: Crambidae), has been reported in many A. sinensis plantations, usually causing complete defoliation of the trees (Figure 1). Although the majority of A. sinensis trees can survive after H. vitessoides damage, they will be too weak for farmers to carry out agarwood-inducing operations [7]. In recent years, increasing attention has been paid on the biology of H. vitessoides [8,9,10,11,12,13,14,15,16]. One interesting behavior of H. vitessoides larvae is that they often form large aggregations to cooperatively feed on the leaves [10]. However, many aspects of the aggregation behavior of H. vitessoides larvae remain unknown. In the present study, we conducted field and laboratory studies to investigate: (1) the aggregation patterns of H. vitessoides larvae under field and laboratory conditions; (2) the factors that affect the aggregation behavior of H. vitessoides larvae; and (3) the biological significance of aggregation behavior on the survival and development of H. vitessoides larvae.

Figure 1.
The outbreak of Heortia vitessoides Moore usually causes the complete defoliation of Aquilaria sinensis (Lour.) Gilg trees. The picture was taken in an Aquilaria sinensis plantation in Zhaoqin, Guangdong, China, on 13 November 2018 by Liping Tang.
Many chemical pesticides such as avermectin, spinosad, and trichlorfon have been applied to control H. vitessoides in the field [17], which may cause pesticide residue in the agarwood. Therefore, it is important to reduce the use of pesticides. The horizontal transfer of chemical pesticides is a mode of action that requires a limited amount of pesticides, which can be transferred from pesticide-exposed (treated) individuals to unexposed (untreated) individuals. This method has been widely applied to control the gregarious insects such as termites [18,19], ants [20,21], and bed bugs [22]. Here we hypothesized that the chemical pesticide (i.e., avermectin) can be horizontally transferred within the aggregations of H. vitessoides larvae, and avermectin-transfer bioassays were conducted to verify this under laboratory conditions.
2. Materials and Methods
2.1. Field Study
The egg clusters and larval cohorts of H. vitessoides were collected from Tianlu Lake Park (113°8′–113°11′ E, 23°12′–23°14′ N), Guangzhou, China, from 15 October to 3 December, 2018. The damaged branches of A. sinensis trees were searched and cut using hand-operated sheers or clippers (an averruncator). A cohort of H. vitessoides larvae was defined as >3 larvae (commonly with close body contact) found on the same leaf (Figure 2A), or on several adjacent leaves (Figure 2B). In the latter case, large aggregations with a few sub-cohorts on different leaves can be observed (Figure 2B). Each larval cohort was placed into a 300 mL plastic container (upper side: 8.6 cm in diameter; bottom side: 6.6 cm in diameter; height: 6.2 cm). These larvae were brought to the laboratory within 3 h, and then preserved in 75% alcohol. The developmental stages (instars) of larvae in each cohort were determined using the methods provided by Qiao et al. [23], and counted. Egg clusters were also brought to the laboratory, and a high-resolution picture was taken of each cluster for egg counting.

Figure 2.
The aggregation behaviors of Heortia vitessoides larvae were investigated under field and laboratory conditions. In the field, a cohort of Heortia vitessoides larvae was defined as >3 larvae (commonly with close body contact) found on the same leaf (A), or on the several adjacent leaves (B). To investigate the aggregation tendency of Heortia vitessoides larvae, 20 Aquilaria sinensis leaves were pasted onto the bottom of a container using the double-faced adhesive tape, and a newly hatched or second-instar larva was placed onto each leaf (C). To evaluate the horizontal transfer of avermectin among gregarious larvae of Heortia vitessoides, 10 untreated larvae (receptors) were released onto one side of the container with six fresh Aquilaria sinensis leaves, and 10 donor larvae (previously released onto the avermectin-treated surfaces for 30 min) were then placed on the other side of the container (D). The first two pictures were taken by Zhijia Huang and the second two by Shiping Liang.
2.2. Laboratory Study
2.2.1. Insects
Each egg cluster collected in the field (as mentioned earlier) was placed in a Petri dish (9 cm in diameter) with fresh A. sinensis leaves and sealed to prevent dehydration. The hatched larvae in each Petri dish (considered as a sibling cohort because all individuals originated from the same egg cluster) were then transferred to a 300 mL plastic container. These larvae (newly hatched to second-instar) were used to set up laboratory experiments 1 and 2 (see below). Because H. vitessoides larvae from non-sibling cohorts showed a strong aggregation tendency (see results), we combined and reared third-instar larvae from different sibling cohorts together in 750 mL plastic containers (upper side: 14.1 cm in diameter; bottom side: 11.3 cm in diameter; height: 6.5 cm) to set up laboratory experiment 3. Fresh A. sinensis leaves were added to the containers each day, and any feces and leaf debris were removed regularly. Our previous studies indicated that A. sinensis leaves collected from different trees may affect the feeding preference of H. vitessoides larvae [10]. As a result, the larvae used in each experiment were fed on leaves collected from a single A. sinensis tree. All eggs and larvae were maintained in an environmental chamber setting at 12:12 light: dark schedule and 25 ± 1 °C.
2.2.2. Experiment 1: Aggregation Tendency of H. vitessoides Larvae
Methods provided by Boulay et al. [24] were modified to investigate the aggregation tendency of H. vitessoides larvae that originated from the same or different sibling cohorts. The bioassay arenas were 2000 mL plastic containers (upper side: 24.8 cm in diameter; bottom side: 20.8 cm in diameter; height: 6.3 cm). Twenty A. sinensis leaves were pasted onto the bottom of each container using double-faced adhesive tape (Deli®, Ningbo, China), with each leaf contacted with the adjacent ones (Figure 2C). Newly hatched or second-instar larvae from six sibling cohorts were used in this experiment. Two treatments were set for the larvae of each instar: (1) 20 larvae from each sibling cohort were released into the container (one larva was randomly placed on the center of each leaf); and (2) 10 larvae from one sibling cohort and 10 from another (two cohorts were randomly selected from the 6 sibling cohorts) were randomly released onto leaves. Each treatment was repeated 6 times. The bioassay arenas were kept at room temperature (24–26 °C), and the number of larvae aggregated on each leaf was recorded at 1, 3, 5, 24, and 48 h after release. The aggregation index (I) was calculated using the formula provided by Boulay et al. [24] as follows:
Aggregation index (I) = (SD)2/mean number of larvae in each leaf
2.2.3. Experiment 2: Effects of Aggregation on H. vitessoides Survivorship and Development
The survivorship and development of H. vitessoides larvae were detected throughout the larval and early adult stages when they were assigned to different group sizes (1, 10, 20 larvae) and sibling status (originating from the same or different sibling cohorts) just after hatching. Eight sibling cohorts of newly hatched larvae were used in this experiment. The bioassays included 4 treatments: (1) single larva from each sibling cohort was transferred to a 300 mL plastic container with fresh A. sinensis leaves; (2) 10 larvae were randomly selected from each sibling cohort and transferred to a container; (3) 20 larvae were randomly selected from each sibling cohort and then released into a container; and (4) 10 larvae from one sibling cohort and 10 from another (two cohorts were randomly selected from the 8 sibling cohorts) were released into a container. Each treatment was repeated 8 times. Fresh A. sinensis leaves were added to the containers, and any feces, dead larvae, and leaf debris were removed each day. On day 11, larvae in each container were transferred to a bigger container (750 mL) because more space and food were needed as a result of the growth of the larvae. On day 18, wet sand (12% moisture) was added at the bottom of each 750 mL plastic container to a depth of 3 cm for the pupation of mature larvae. The bioassays were maintained in an environmental chamber (12:12 light–dark schedule and 25 ± 1 °C) throughout the experiment. The survivorship and developmental stages (instars) of larvae in each container were recorded daily until some individuals began to burrow into the sand for pupating after day 18. The duration of the larval stage (between hatching and burrowing into the sand for pupation) of each larva in each replication was recorded. We also recorded the number of emerged adults each day after pupating (until no more adult emerged for a 15-day period), and the emergence success was calculated as follows: emergence success (%) = (number of emerged adults/number of larvae initially released) × 100%.
2.2.4. Experiment 3: Horizontal Transfer of Avermectin among H. vitessoides Larvae
This experiment was conducted to investigate whether the chemical pesticide would transfer among gregarious larvae of H. vitessoides (transfer from treated larvae to untreated ones). Different concentrations of avermectin (Dugao®, Shijiazhuang, Hebei, China) solutions (0 (control), 100, or 200 ppm) were prepared. Each solution was sprayed once on the inner surfaces of a Petri dish and lid using a sprayer (Zhenxing Industrial Co., Ltd., Guangzhou, China)(0.92 ± 0.01 mL (mean ± SE, n = 10) solution was sprayed on the Petri dish or lid). After the solutions were air-dried (~40 min), 80 third-instar larvae were randomly selected and marked with a red ink spot on the dorsum, and then released onto the bottom of each Petri dish and covered. These larvae (donors) were allowed to move on the avermectin-treated surfaces for 30 min, and then transferred to an untreated Petri dish. The bioassay arenas were 1280 mL plastic containers (upper side: 19.1 cm in diameter; bottom side: 16.2 cm in diameter; height: 4.5 cm) with six fresh A. sinensis leaves placed on one side of the container (Figure 2D). Ten untreated larvae (receptors) were randomly selected and released onto leaves and allowed to acclimate for 1 h. Ten donors were then placed on the other side of the container (Figure 2D). Each of the three treatments (donor previously exposed to the Petri dish sprayed with 0, 100, or 200 ppm avermectin solutions) was repeated 7 times. The bioassays were prepared and maintained in an environmental chamber setting at a 12:12 light–dark schedule and 25 ± 1 °C.
After 12 h, donors and receptors in each container were separated and each group was transferred to a 450 mL plastic container (upper side: 11.2 cm in diameter; bottom side: 7.0 cm in diameter; height: 4.4 cm) with fresh A. sinensis leaves. Here we separated donors and receptors because the red ink spot on the donors would disappear within 1–2 days due to molting, and therefore we would not be able to distinguish donors and receptors. Larvae were reared as mentioned earlier. The survivorship of donors and receptors was recorded every 24 h after initial combination. On day 7, living donors and receptors were transferred to 750 mL plastic containers with fresh A. sinensis leaves and wet sand (12% moisture) on the bottom (depth = 3 cm). Because mature larvae began to burrow into the sand for pupating, we did not record larval survivorship after 7 days. The number of emerged donors and receptors were recorded until no new adults were observed for 15 days, and emergence success was calculated as mentioned above.
2.3. Data Analysis
For the field study, one-way ANOVA (Proc Mixed, SAS 9.4, Cary, NC, USA) was conducted to compare the number of individuals within egg clusters and the same-instar cohorts (comprised of newly hatched, first-, second-, or third–fifth instar larvae) as well as different-instar mixed cohorts. For experiment 1 (the laboratory study), the aggregation tendency was studied by comparing the aggregation index of newly hatched or second-instar larvae using the mixed ANOVA (IBM SPSS Statistics 24, Chicago, IL, USA), with time as the within-subjects factor and sibling status as the between-subjects factor. To investigate the change of aggregation index through time, we performed one-way repeated measures ANOVA (IBM SPSS Statistics 24) for each sibling status with time as the within-subjects factor. For experiment 2, because all isolated larva died within two days (see results), we only compared the survivorship of 10-sibling larvae, 20-sibling larvae, and 20-non-sibling larvae using mixed ANOVA, with time as the within-subjects factor and treatment as the between-subjects factor. For experiment 3, we compared survivorship of donors or receptors using the mixed ANOVA (IBM SPSS Statistics 24), with time as the within-subjects factor and treatment as the between-subjects factor. For both experiment 2 and 3, the one-way ANOVAs (IBM SPSS Statistics 24) were then used to compare larval survivorship among treatments within each time point. The duration of the larval stage (experiment 2) and emergence success of adults (experiment 2 and 3) were compared between treatments using the one-way ANOVAs (Proc Mixed, SAS 9.4). The significance level was determined at α = 0.05 for all tests.
3. Results
3.1. Field Study
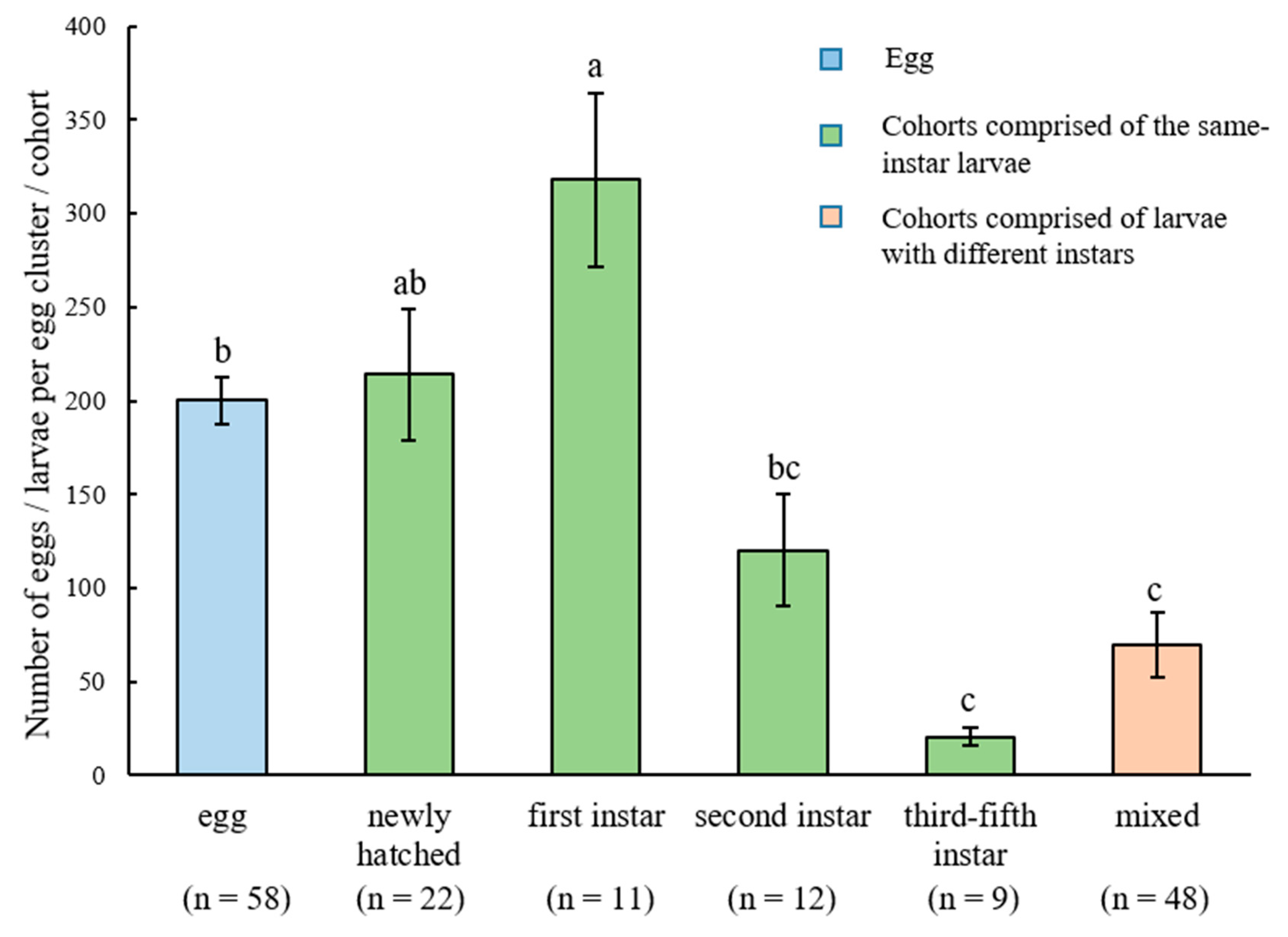
In total, 58 egg clusters and 102 larval cohorts were obtained from the field (Table 1). Fifty-four cohorts were composed of larvae with the same developmental stage (same-instar cohorts), while 48 cohorts were composed of larvae with different instars (different-instar mixed cohorts). The mean number of individuals in the same-instar cohorts containing the first-instar larvae was significantly higher than the mean number of eggs in the egg clusters, and both were not significantly different from the mean number of individuals in the same-instar cohorts containing newly hatched larvae (Figure 3). The number of individuals in the same-instar cohorts containing third-, fourth-, or fifth-instar larvae and different-instar mixed cohorts was similar, and all were significantly lower than that in the same-instar cohorts containing newly hatched or first-instar larvae (F = 14.84, df = 5, 154, p < 0.0001; Figure 3). The number of larvae in different-instar mixed cohorts was quite variable, ranging from several to hundreds of individuals (Table 2). Interestingly, 70.8%, 77.1%, and 52.1% of different-instar mixed cohorts contained second-, third-, and fourth-instar larvae, respectively, whereas only a few different-instar mixed cohorts contained newly hatched and first-instar larvae (Table 2).

Table 1.
Number of egg clusters and larval cohorts collected in each collection date.
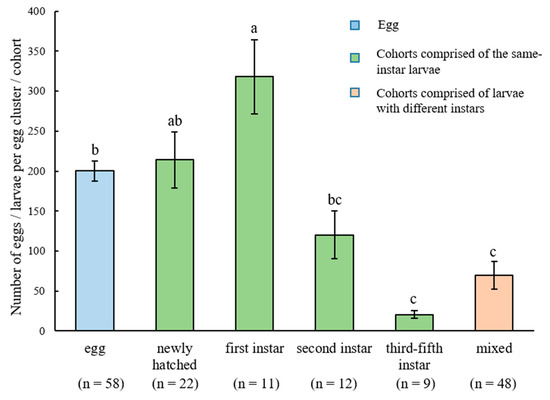
Figure 3.
The number of Heortia vitessoides individuals (mean ± SE) in egg clusters and in the cohorts containing the same-instar or different-instar larvae. Different letters indicate significant differences (p < 0.05).

Table 2.
The number of larvae in each different-instar mixed cohort.
3.2. Laboratory Study
3.2.1. Experiment 1: Aggregation Tendency of H. vitessoides Larvae
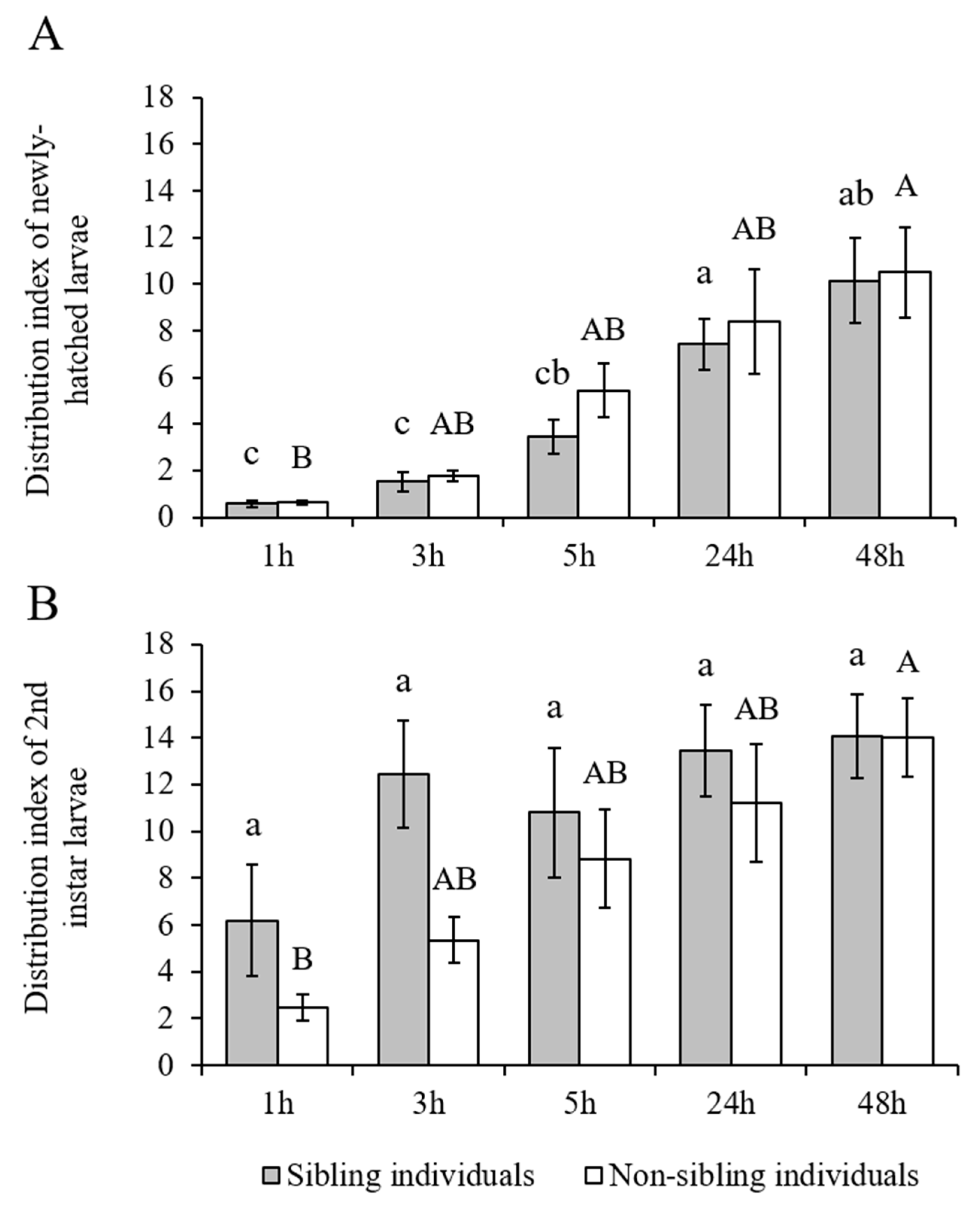
The H. vitessoides larvae showed a strong aggregation tendency, regardless of developmental stages (newly hatched or second-instar) and sibling status (originated from the same or different sibling cohorts) (Figure 4). The effect of sibling status (newly hatched larvae: F = 0.353, df = 1, 10, p = 0.566; second-instar larvae: F = 2.210, df = 1, 10, p = 0.168) and interaction effect between sibling status and time (newly hatched larvae: F = 0.312, df = 1.904, 19.039, p = 0.725; second-instar larvae: F = 1.372, df = 3.020, 30.201, p = 0.270) were not significant, but the aggregation index significantly increased with time for both newly hatched (F = 34.443, df = 1.904, 19.039, p < 0.001) and second-instar larvae (F = 10.975, df = 3.020, 30.201, p < 0.001; Figure 5; Supplementary Materials: Tables S1 and S2).
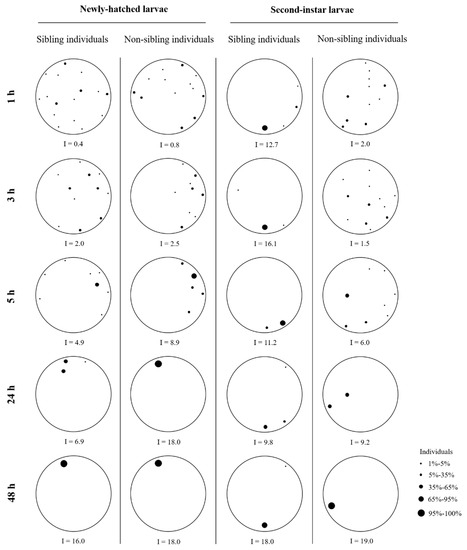
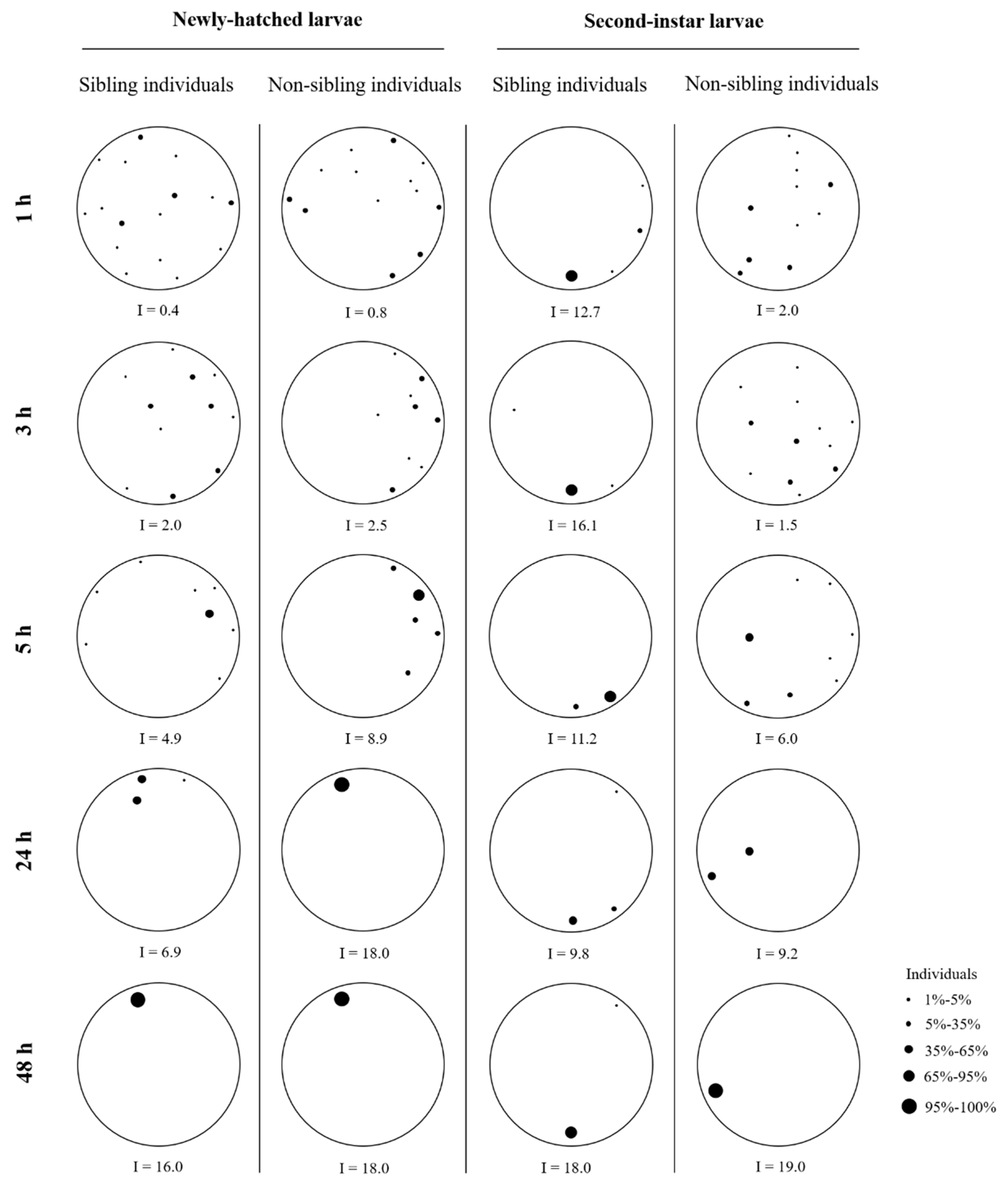
Figure 4.
Examples of aggregation behavior of newly hatched or second-instar larvae of Heortia vitessoides in the arena containing sibling individuals (20 larvae originated from the same sibling cohort), or non-sibling individuals (10 larvae originated from one sibling cohort and 10 from another sibling cohort).
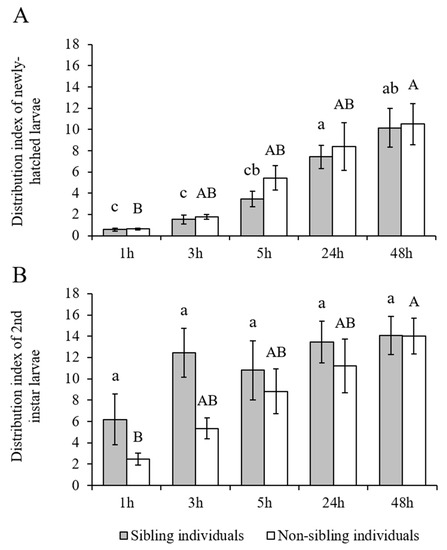
Figure 5.
(A) Aggregation index of newly hatched or (B) second-instar larvae of Heortia vitessoides in the arena containing sibling individuals (20 larvae originated from the same sibling cohort), or non-sibling individuals (10 larvae originated from one sibling cohort and 10 from another sibling cohort). Different lower-case letters indicate significant differences in aggregation index of sibling individuals compared among the five time points (p < 0.05). Different capital letters indicate significant differences in aggregation index of non-sibling individuals compared among the five time points (p < 0.05).
3.2.2. Experiment 2: Effects of Aggregation on H. vitessoides Survivorship and Development
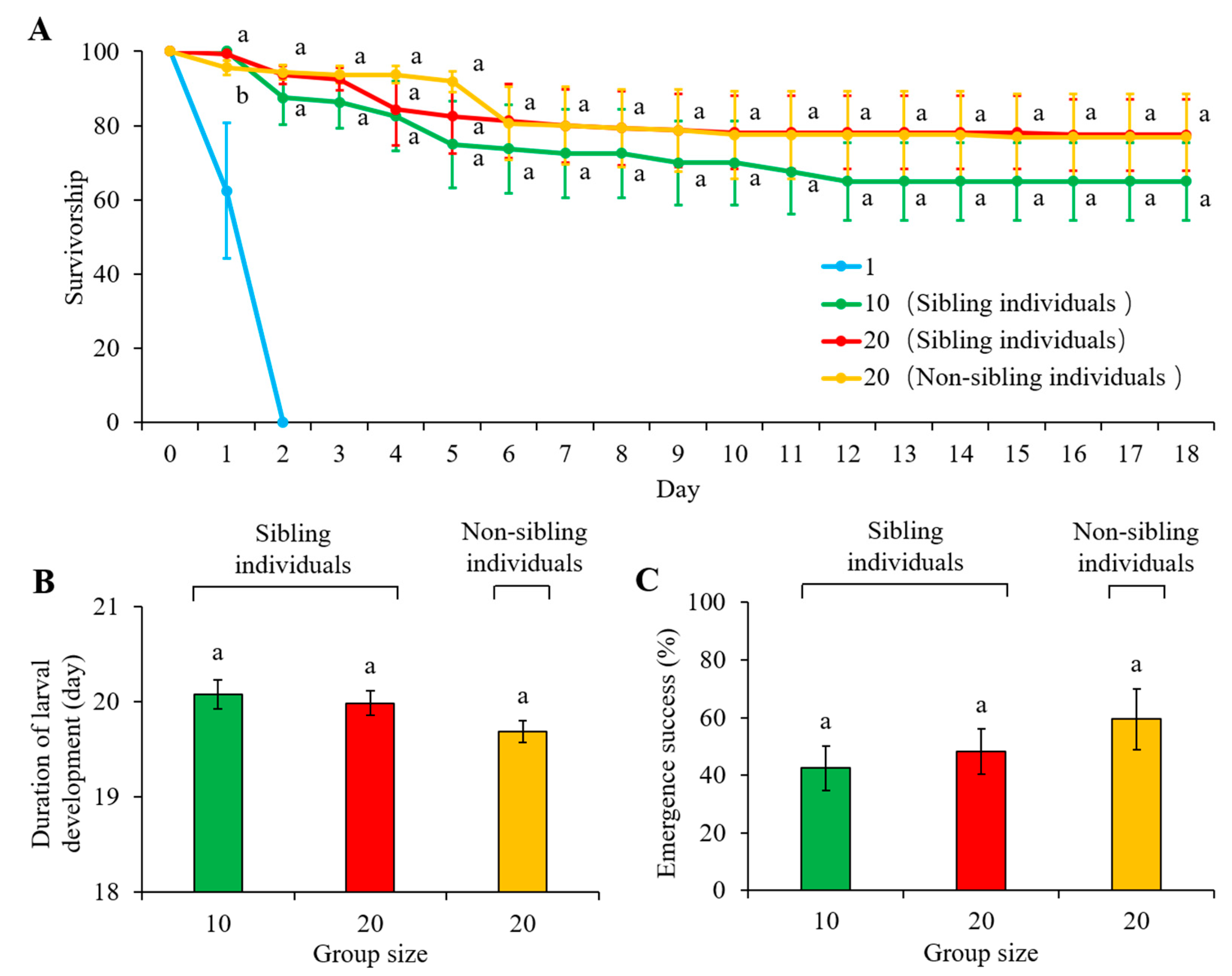
All newly hatched larvae died within two days after they were isolated (Figure 6A). No evidence of leaf consumption was observed before these larvae died. The effect of time (F = 19.272, df = 2.560, 46.080, p < 0.001) and the interaction effect of time and treatments (F = 2.647, df = 5.120, 46.080, p = 0.034) were significant, but there was no significant difference in larval survivorship between 10-larvae cohorts (containing sibling individuals) and 20-larvae cohorts (either containing individuals from the same or different sibling cohorts) (F = 1.312, df = 2, 18, p =0.294; Figure 6A; Supplementary Material: Table S3). In addition, the duration of the larval stage (F = 0.95; df =2, 21; p = 0.4031) and emergence success of adults (F = 2.39; df =2, 291; p = 0.0936) were similar when compared between 10-larvae cohorts and 20-larvae cohorts (either containing individuals from the same or different sibling cohorts) (Figure 6B,C). Interestingly, the development of larvae in some replications was asynchronous (Supplementary Materials: Table S4), even for the larvae originating from the same sibling cohorts (hatched from the same egg cluster at the same time) (Table 3).
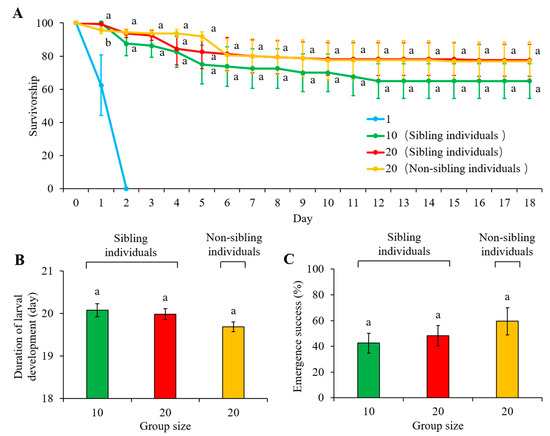
Figure 6.
(A) Larval survivorship, (B) duration of larval stage, and (C) emergence success of adults of Heortia vitessoides individuals in the treatments containing: (1) single larva originated from each sibling cohort; (2) 10 larvae originated from the same sibling cohort; (3) 20 larvae originated from the same sibling cohort; and (4) 10 larvae originated from one sibling cohort and 10 from another. Since all newly hatched larvae died within two days after they were isolated, the comparisons were conducted for the latter three treatments. Data were presented as mean ± SE, and different letters indicate significant differences (p < 0.05).

Table 3.
The developmental stages (I: first instar; II: second instar; III: third instar; IV: fourth instar; V: fifth instar; P: pupae) of Heortia vitessoides individuals during the whole larval stage after the newly hatched larvae were initially assigned into 20-larvae cohorts (containing sibling individuals). Note that the larvae of different developmental stages (instars) can be observed within the same sibling cohort at the same period.
3.2.3. Experiment 3: Horizontal Transfer of Avermectin among H. vitessoides Larvae
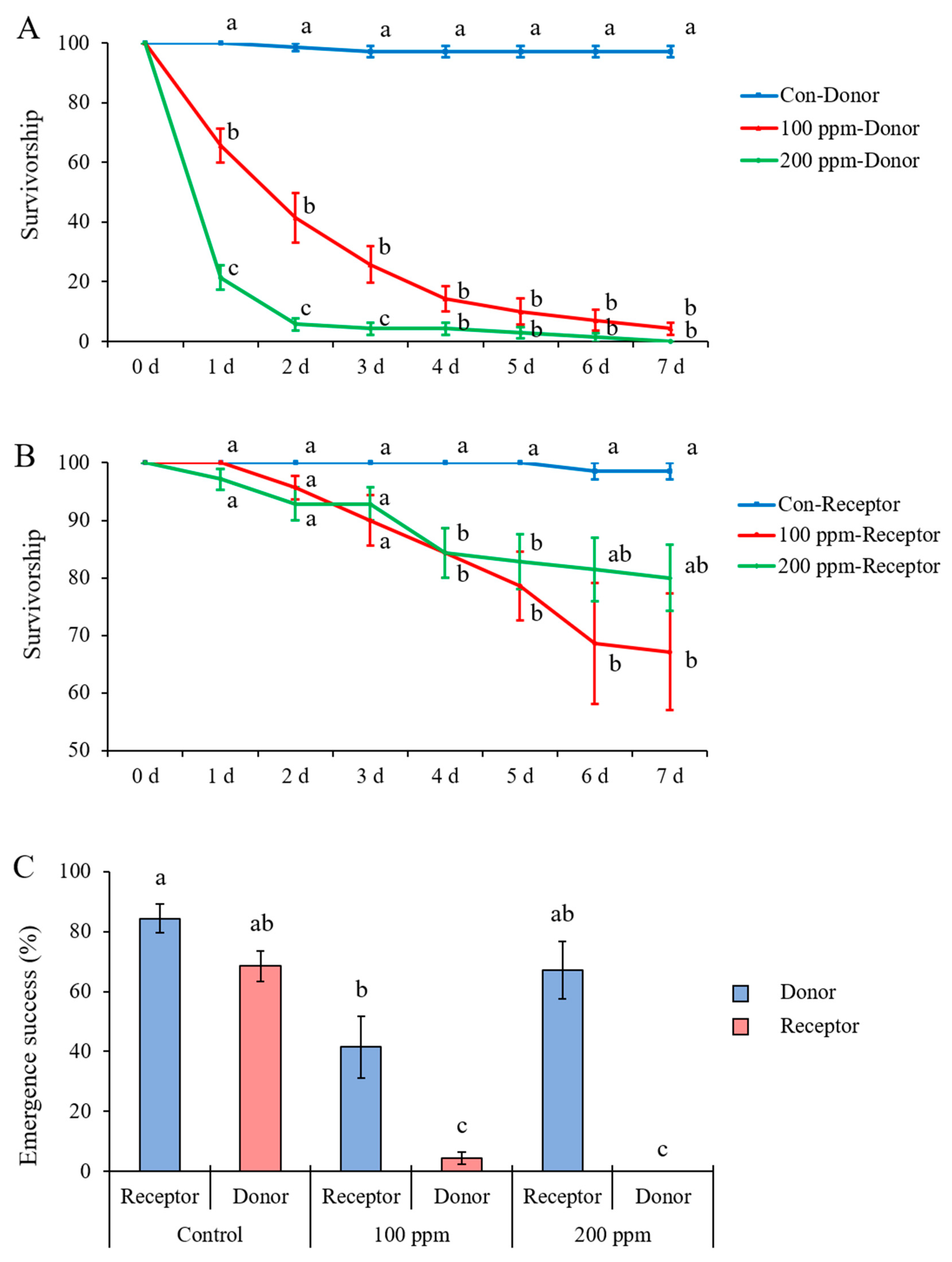
For larval survivorship of both donors and receptors, the effect of time (donors: F = 273.676, df = 3.258, 58.652, p < 0.001; receptors: F = 16.452, df = 1.353, 24.349, p < 0.001) and treatment (donors: F = 332.512, df = 2, 18, p < 0.001; receptors: F = 5.664, df = 2, 18, p = 0.012) and the interaction between time and treatment (donors: F = 73.615, df = 6.517, 58.652, p < 0.001; receptors: F = 4.650, df = 2.705, 24.349, p = 0.012) were significant. Comparisons within each time point showed that the donors previously exposed to the Petri dish sprayed with 100 or 200 ppm avermectin solution had significantly lower survivorships compared with control donors (previously exposed to the Petri dish sprayed with distilled water) (Figure 7A, Supplementary Materials: Table S5). The receptors combined with donors previously exposed to the Petri dish sprayed with 100 ppm avermectin solution had significantly lower survivorship compared with control receptors after day 4 (Figure 7B, statistic results are shown in Supplementary Materials: Table S6). While larval survivorship of receptors combined with donors previously exposed to the Petri dish sprayed with 200 ppm avermectin solution was lower than the control on days 4 and 5; it did not significantly differ from either the control or 100 ppm avermectin treatments on days 6 and 7 (Figure 7B). The emergence success of receptors combined with donors previously exposed to the Petri dish sprayed with 100 ppm avermectin solution was significantly lower than that of the control receptors (F = 29.71; df =5, 36; p < 0.0001) (Figure 7C).
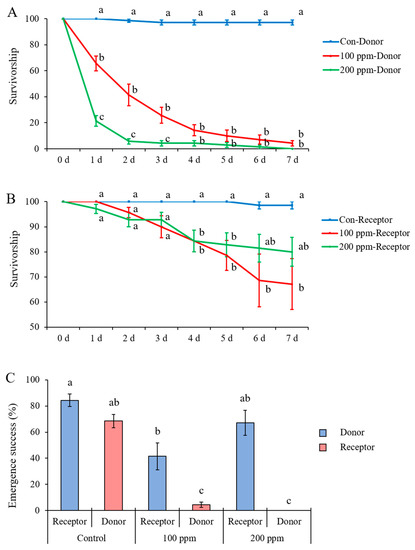
Figure 7.
(A) Larval survivorship of donors (B) and receptors and (C) emergence success of adults of Heortia vitessoides individuals in each treatment which donors were previously exposed to the Petri dish sprayed with 0, 100, or 200 ppm avermectin solution. Data were presented as mean ± SE, and different letters indicate significant differences (p < 0.05).
4. Discussion
Compared to adults, animals in larval stages are vulnerable because of their limited mobility and foraging abilities. Social aggregation is one of the strategies that provide protective and facilitation effects aiding the survival and development of larvae [25], and has arisen independently in diverse animal taxa [26,27,28,29,30]. The majority of insects have immature stages, and the aggregation behaviors of larvae have been reported in many lineages including true flies [24,31,32,33], sawflies [34,35,36,37], and beetles [38,39,40].
Although many lepidopteran adults lay single eggs and their larvae are solitary [41], some species lay eggs in clusters and the larvae show gregarious behavior. For example, Stamp [42] estimated that 5%–15% of butterfly species have gregarious larval stages. Larvae of some serious agricultural and forestry lepidopteran pests, such as Spodoptera litura (Fabr.) (Lepidoptera: Noctuidae), Malacosoma americanum (Fabr.) (Lepidoptera: Lasiocampidae) and Hemileuca Lucina Hy. Edw. (Lepidoptera: Saturniidae), are also gregarious [43,44,45]. Most of these species show gregariousness during first third or fourth instars with the last one or two instars being solitary [46], and the number of individuals in each aggregation was usually low [47]. Our field study also showed that H. vitessoides larvae are gregarious during early instars, and later instar larvae are largely solitary, though some of them still formed small aggregations. Some H. vitessoides aggregations can be very large in the wild, usually containing hundreds of individuals (Figure 3), which may be a result of cohort merging because: (1) the mean number of individuals in the first-instar cohorts was significantly higher than that of the eggs in the clusters; and (2) larvae of different developmental stages were usually observed within the same cohort (though this also can be caused by the asynchronous development of larvae within the same cohorts, as shown in Table 3). We believe that the aggregation behaviors of H. vitessoides larvae are an important reason accounting for the severe defoliation caused by this pest. Since the early instar larvae are highly concentrated, we suggest artificially removing H. vitessoides aggregations from A. sinensis trees before the dispersing of later instar larvae.
The group-size effects on the survival and development of gregarious larvae and the related potential mechanisms have been investigated in many lepidopteran species. For example, Clark and Faeth [48] reported that the larvae of Chlosyne lacinia (Geyer) (Lepidoptera: Nymphalidae) developed faster when the group size was bigger, because individuals in larger groups found it easier to overcome the physical toughness of host plant leaves. Santana et al. [28] reported that isolated larva of Ascia monuste (Godart) (Lepidoptera: Pieridae) showed lower permanency on the host plant than that of aggregations, and large aggregations had a lower predation rate by natural enemies compared with small ones. In addition, Klok and Chown [49] reported that group living of second- and third-instar larvae of Imbrasia belina (Westwood) (Lepidoptera: Saturniidae) helped to maintain the body temperature of individuals due to “the accumulation of higher heat loads by the larger masses of large aggregations”. Interestingly, Inouye and Johnson [41] found that the feeding rate of first-instar larvae of Chlosyne poecile (Felder) (Lepidoptera: Nymphalidae) significantly increased with group size, whereas the opposite trend was observed in second-instar larvae. Since large groups of second-instars larvae of C. poecile are commonly found in the field [41], benefits other than feeding simulation should exist. Similar to many previous studies, our study showed that isolated larva of H. vitessoides did not feed on leaves and eventually died within a short period, indicating that gregariousness may play a critical role in facilitating feeding for newly hatched larvae. However, we found that group size did not affect survivorship and development of H. vitessoides larvae under laboratory conditions (Figure 6). Future field studies are needed to investigate if the group size affects predation risks and thermoregulation of H. vitessoides larvae under natural conditions.
Many larval aggregations of lepidopteran species are composed of sibling individuals [48]. In a few species such as Euselasia chrysippe Bates (Lepidoptera: Riodinidae), non-sibling cohorts sometimes merge to form bigger aggregations, indicating that “benefits of living in large groups outweigh the costs of intra- or inter-specific competition in this species” [47]. Likewise, our studies showed that H. vitessoides larvae had a strong tendency to form the aggregations, whether they initially originated from the same or different sibling cohorts. This result probably showed that the larval gregariousness of H. vitessoides was not kin-selected. The developmental stage is another factor that might affect larval aggregation. For example, Cornell et al. [44] reported that the first-instar larvae of H. lucina barely aggregated, whereas older instar larvae can quickly form aggregations. Although our study did not compare the aggregation index between newly hatched and second-instar larvae of H. vitessoides, larvae in both stages successfully formed the aggregations during the experiment (Figure 4 and Figure 5).
Our study showed the horizontal transfer of avermectin among H. vitessoides larvae. To our best knowledge, this is the first report on pesticide horizontal transfer among gregarious larvae of a lepidopteran pest. To separate donors and receptors, previous studies on social insects, such as termites, commonly fed donors with dyes including Nile blue A and Sudan Red 7B [50,51,52]. However, marking the donors of H. vitessoides larvae would be challenging. Our preliminary studies showed that H. vitessoides larvae that fed on leaves treated with low concentrations of Nile blue A or Sudan Red 7B showed high mortality and limited mobility. Marking the body with inks did not affect moving and aggregation behaviors of H. vitessoides larvae; however, the ink spots easily faded after a short period. As a result, in the present study, donors were only allowed to combine with receptors for 12 h. The limited combining duration may to some extent reduce the effectiveness of avermectin transfer.
The horizontal transfer of pesticides among termites and ants can be caused by trophallaxis, grooming, and body contact among donors and receptors, as well as contact between receptors and pesticide-contaminated areas [53,54]. Since trophallaxis and grooming behaviors have not been observed in the gregarious larvae of H. vitessoides, contacting may account for the horizontal transfer of avermectin in this species. Here, donor larvae exposed to the Petri dish sprayed with the high concentration (200 ppm) of avermectin solution showed reduced movement and aggregation behaviors, which might reduce the chance of contact between donors and receptors. Therefore, donors treated with a high concentration of avermectin did not cause significantly lower larval survivorship and adult emergence of receptors as compared to the controls (Figure 7). This result showed that high concentrations of pesticide might negatively affect the effectiveness of horizontal transfer, and should be avoided in H. vitessoides management. It is important to note that this experiment is somewhat preliminary. Future studies are needed to investigate the factors (e.g., larval instars, donor–receptor ratio) that may influence horizontal transfer of various pesticides among gregarious larvae of H. vitessoides.
5. Conclusions
Our field study showed that H. vitessoides larvae are gregarious during early instars, and later instar larvae are largely solitary, though some of them still formed small aggregations. Some H. vitessoides aggregations can be very large in the wild, usually containing hundreds of individuals. In the laboratory, H. vitessoides larvae had a strong tendency to form the aggregations, whether they initially originated from the same or different sibling cohorts. When newly hatched larvae were isolated, they did not feed on leaves and eventually died within a short period. However, group size and sibling status did not affect survivorship and development of H. vitessoides larvae. In addition, our study is the first to show the horizontal transfer of avermectin among H. vitessoides larvae.
Supplementary Materials
The following are available online at https://www.mdpi.com/1999-4907/10/4/331/s1, Table S1: Statistics Analysis for Figure 5A, Table S2: Statistics Analysis for Figure 5B, Table S3: Statistics Analysis for Figure 6A, Table S4: The developmental stages (I: first instar; II: second instar; III: third instar; IV: fourth instar; V: fifth instar; P: pupae) of Heortia vitessoides individuals during the whole larval stage after the newly hatched larvae were initially assigned into 10 or 20-larvae cohorts, Table S5: Statistics Analysis for Figure 7A, Table S6: Statistics Analysis for Figure 7B.
Author Contributions
Formal Analysis, S.L., J.C., X.C. and C.W.; Investigation, S.L., Z.J., J.Z., Z.H., L.T., and C.W.; Resources, Z.S. and X.W.; Data Curation, S.L. and C.W.; Writing-Original Draft Preparation, S.L. and C.W.; Writing-Review & Editing, J.C., X.C., Z.J., J.Z., Z.H., L.T., Z.S. and X.W.; Visualization, S.L., J.C., X.C. and C.W.; Supervision, C.W.; Project Administration, C.W.; Funding Acquisition, C.W.
Funding
This research was funded by the Guangdong Natural Science Foundation (Grant No. 2016A030310445).
Acknowledgments
We sincerely thank Qinxi Xie, Hongpeng Xiong, and Wenquan Qin (College of Forestry and Landscape Architecture, South China Agricultural University) for valuable helps in H. vitessoides collection and rearing.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES). 2010. Available online: http://www.cites.org/ (accessed on 26 February 2019).
- Blanchette, R.; Van Beek, H.H. Cultivated Agarwood. Patent No. 6,848,211 B2, 1 February 2005. [Google Scholar]
- Wei, J.H.; Zhang, Z.; Yang, Y.; Meng, H.; Feng, J.D.; Gan, B.C. Production of Agarwood in Aquilaria sinensis Trees via Transfusion Technique. CN101755629B, 2010. [Google Scholar]
- Chen, H.Q.; Yang, Y.; Xue, J.; Wei, J.H.; Zhang, Z.; Chen, H.J. Comparison of compositions and antimicrobial activities of essential oils from chemically stimulated agarwood, wild agarwood and healthy Aquilaria sinensis (Lour.) gilg trees. Molecules 2011, 16, 4884–4896. [Google Scholar] [CrossRef]
- Liu, Y.Y.; Chen, H.Q.; Yang, Y.; Zhang, Z.; Wei, J.H.; Meng, H.; Chen, W.P.; Feng, J.D.; Gan, B.C.; Chen, X.Y.; et al. Whole-tree agarwood-inducing technique: An efficient novel technique for producing high-quality agarwood in cultivated Aquilaria sinensis trees. Molecules 2013, 18, 3086–3106. [Google Scholar] [CrossRef] [PubMed]
- Mamat, M.F.; Yacob, M.R.; Fui, L.H.; Rdam, A. Costs and benefits analysis of Aquilaria species on plantation for agarwood production in Malaysia. Int. J. Bus. Soc. Sci. 2010, 1, 162–174. [Google Scholar]
- Turjaman, M.; Hidayat, A.; Santoso, E. Development of agarwood induction technology using endophytic fungi. In Agarwood; Springer: Singapore, 2016; pp. 57–71. [Google Scholar]
- Qiao, H.L.; Lu, P.F.; Chen, J.; Ma, W.S.; Qin, R.M.; Li, X.M. Antennal and behavioural responses of Heortia vitessoides females to host plant volatiles of Aquilaria sinensis. Entomol. Exp. Appl. 2012, 143, 269–279. [Google Scholar] [CrossRef]
- Qiao, H.; Lu, P.F.; Liu, S.; Xu, C.Q.; Guo, K.; Xu, R.; Chen, J. Volatiles from Aquilaria sinensis damaged by Heortia vitessoides larvae deter the conspecific gravid adults and attract its predator Cantheconidea concinna. Sci. Rep. 2018, 8, 15067. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.F.; Ma, T.; Chang, M.S.; Wu, Y.J.; Liu, Z.T.; Sun, Z.H.; Shan, T.J.; Chen, X.Y.; Wen, X.J.; Wang, C. Aggregation and feeding preference of gregarious Heortia vitessoides (Lepidoptera: Crambidae) larvae to Aquilaria sinensis (Thymelaeaceae). J. Entomol. Sci. 2016, 51, 209–218. [Google Scholar] [CrossRef]
- Wen, Y.Z.; Jin, X.F.; Zhu, C.; Chen, X.; Ma, T.; Zhang, S.N.; Zhang, Y.; Zeng, S.C.; Chen, X.Y.; Sun, Z.H.; et al. Effect of substrate type and moisture on pupation and emergence of Heortia vitessoides (Lepidoptera: Crambidae): Choice and no-choice studies. J. Insect Behav. 2016, 29, 473–489. [Google Scholar] [CrossRef]
- Wen, Y.Z.; Qin, W.Q.; Chen, X.; Wen, X.J.; Ma, T.; Dong, X.; Lin, S.C.; Sun, Z.H.; Zeng, S.C.; Wang, C. Soil moisture effects on pupation behavior, physiology, and morphology of Heortia vitessoides (Lepidoptera: Crambidae). J. Entomol. Sci. 2017, 52, 229–238. [Google Scholar] [CrossRef]
- Cheng, J.; Chen, J.; Lin, T. De novo assembly and analysis of the Heortia vitessoides transcriptome via high-throughput Illumina sequencing. J. Asia-Pac. Entomol. 2017, 20, 1241–1248. [Google Scholar] [CrossRef]
- Cheng, J.; Wang, C.Y.; Lyu, Z.H.; Lin, T. Multiple Glutathione S-Transferase Genes in Heortia vitessoides (Lepidoptera: Crambidae): Identification and Expression Patterns. J. Insect Sci. 2018, 18, 23. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Wang, C.Y.; Lyu, Z.H.; Chen, J.X.; Tang, L.P.; Lin, T. Candidate olfactory genes identified in Heortia vitessoides (Lepidoptera: Crambidae) by antennal transcriptome analysis. Comp. Biochem. Phys. D 2019, 29, 117–130. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.X.; Lyu, Z.H.; Wang, C.Y.; Cheng, J.; Lin, T. RNA interference of a trehalose-6-phosphate synthase gene reveals its roles in the biosynthesis of chitin and lipids in Heortia vitessoides (Lepidoptera: Crambidae). Insect Sci. 2018, 1–12. [Google Scholar] [CrossRef]
- Chen, Z.Y.; Wang, L.; Li, D.W.; Li, Y.Z.; Huang, X.R.; Qin, C.S. A screening experiment on insecticides against Heortia vitessoides Moore. Chin. For. Sci. Technol. 2012, 26, 117–119. [Google Scholar] [CrossRef]
- Rust, M.K.; Saran, R.K. Toxicity, repellency, and transfer of chlorfenapyr against western subterranean termites (Isoptera: Rhinotermitidae). J. Econ. Entomol. 2006, 99, 864–872. [Google Scholar] [CrossRef]
- Bhatta, D.; Henderson, G. Horizontal Transfer of Spinosad in Coptotermes formosanus (Isoptera: Rhinotermitidae). J. Econ. Entomol. 2016, 109, 1813–1818. [Google Scholar] [CrossRef]
- Soeprono, A.M.; Rust, M.K. Effect of horizontal transfer of barrier insecticides to control Argentine ants (Hymenoptera: Formicidae). J. Econ. Entomol. 2004, 97, 1675–1681. [Google Scholar] [CrossRef]
- Buczkowski, G. Trap-treat-release: Horizontal transfer of fipronil in field colonies of black carpenter ants, Camponotus pennsylvanicus. Pest Manag. Sci. 2019. [Google Scholar] [CrossRef]
- Akhtar, Y.; Isman, M.B. Horizontal transfer of diatomaceous earth and botanical insecticides in the common bed bug, Cimex lectularius L.; Hemiptera: Cimicidae. PLoS ONE 2013, 8, e75626. [Google Scholar] [CrossRef] [PubMed]
- Qiao, H.L.; Lu, P.F.; Chen, J.; Xu, C.Q.; Ma, W.S.; Qin, R.M.; Li, X.M.; Cheng, H.Z. Biological characteristics and occurrence patterns of Heortia vitessoides. Chin. J. Appl. Entomol. 2013, 50, 1244–1252. [Google Scholar] [CrossRef]
- Boulay, J.; Devigne, C.; Gosset, D.; Charabidze, D. Evidence of active aggregation behaviour in Lucilia sericata larvae and possible implication of a conspecific mark. Anim. Behav. 2013, 85, 1191–1197. [Google Scholar] [CrossRef]
- Ruxton, G.D.; Sherratt, T.N. Aggregation, defence and warning signals: The evolutionary relationship. Proc. R. Soc. B Biol. Sci. 2006, 273, 2417–2424. [Google Scholar] [CrossRef]
- Ottmann, D.; Grorud-Colvert, K.; Sard, N.M.; Huntington, B.E.; Banks, M.A.; Sponaugle, S. Long-term aggregation of larval fish siblings during dispersal along an open coast. Proc. Natl. Acad. Sci. USA 2016, 113, 14067–14072. [Google Scholar] [CrossRef] [PubMed]
- Raven, C.; Shine, R.; Greenlees, M.; Schaerf, T.M.; Ward, A.J. The role of biotic and abiotic cues in stimulating aggregation by larval cane toads (Rhinella marina). Ethology 2017, 123, 724–735. [Google Scholar] [CrossRef]
- Santana, A.F.; Rodrigues, D.; Zucoloto, F.S. Larval aggregation in a Neotropical butterfly: Risky behaviors, per capita risk, and larval responses in Ascia monusteorseis. Behav. Ecol. Sociobiol. 2017, 71, 174. [Google Scholar] [CrossRef]
- Fouche, Q.; Hedouin, V.; Charabidze, D. Communication in necrophagous Diptera larvae: Interspecific effect of cues left behind by maggots and implications in their aggregation. Sci. Rep. 2018, 8, 2844. [Google Scholar] [CrossRef]
- Morimoto, J.; Nguyen, B.; Tabrizi, S.T.; Ponton, F.; Taylor, P. Social and nutritional factors shape larval aggregation, foraging, and body mass in a polyphagous fly. Sci. Rep. 2018, 8, 14750. [Google Scholar] [CrossRef] [PubMed]
- Heard, S.B. Resource patch density and larval aggregation in mushroom-breeding flies. Oikos 1998, 81, 187–195. [Google Scholar] [CrossRef]
- Slone, D.H.; Gruner, S.V. Thermoregulation in larval aggregations of carrion-feeding blow flies (Diptera: Calliphoridae). J. Med. Entomol. 2007, 44, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Mast, J.D.; De Moraes, C.M.; Alborn, H.T.; Lavis, L.D.; Stern, D.L. Evolved differences in larval social behavior mediated by novel pheromones. eLife 2014, 3, e04205. [Google Scholar] [CrossRef] [PubMed]
- Codella, S.G.; Raffa, K.F. Contributions of female oviposition patterns and larval behavior to group defense in conifer sawflies (Hymenoptera: Diprionidae). Oecologia 1995, 103, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Carita, L.; Johanna, M.; Jussi, P.; Martti, V. Effects of group size and pine defence chemicals on Diprionid sawfly survival against ant predation. Oecologia 2006, 150, 519–526. [Google Scholar] [CrossRef]
- Fletcher, L.E. Vibrational signals in a gregarious sawfly larva (Perga affinis): Group coordination or competitive signaling? Behav. Ecol. Sociobiol. 2007, 61, 1809–1821. [Google Scholar] [CrossRef]
- Costa, J.T.; Louque, R.W. Group foraging and trail following behavior of the red-headed pine sawfly Neodiprion lecontei (Fitch) (Hymenoptera: Symphyta: Diprionidae). Ann. Entomol. Soc. Am. 2014, 94, 480–489. [Google Scholar] [CrossRef]
- Grégoire, J.C. Larval gregariousness in the Chrysomelidae. In Biology of Chrysomelidae; Jolivet, P., Petitpierre, E., Hsiao, T.H., Eds.; Springer: Dordrecht, The Netherlands, 1988; Volume 42, pp. 253–260. [Google Scholar]
- Storer, A.; Wainhouse, D.; Speight, M. The effect of larval aggregation behaviour on larval growth of the spruce bark beetle Dendroctonus micans. Ecol. Entomol. 1997, 22, 109–115. [Google Scholar] [CrossRef]
- Weed, A.S. Benefits of larval group feeding by Chrysolina aurichalcea asclepiadis on Vincetoxicum: Improved host location or feeding facilitation? Entomol. Exp. Appl. 2010, 137, 220–228. [Google Scholar] [CrossRef]
- Inouye, B.D.; Johnson, D.M. Larval aggregation affects feeding rate in Chlosyne poecile (Lepidoptera: Nymphalidae). Flarida Entomol. 2005, 88, 247–252. [Google Scholar] [CrossRef]
- Stamp, N.E. Egg deposition in butterflies: Why do some species cluster their eggs rather than deposit them singly? Am. Nat. 1980, 115, 367–380. [Google Scholar] [CrossRef]
- Nakasuji, F.; Yamanaka, H.; Kiritani, K. The disturbing effect of Micryphantid spiders on the larval aggregation of the tobacco cutworm, Spodoptera litura (Lepidoptera: Noctuidae). Kontyu 1973, 41, 220–227. [Google Scholar]
- Cornell, J.C.; Stamp, N.E.; Bowers, M.D. Developmental change in aggregation, defense and escape behavior of buckmoth caterpillars, Hemileuca lucina (Saturniidae). Behav. Ecol. Sociobiol. 1987, 20, 383–388. [Google Scholar] [CrossRef]
- Casey, T.M.; Joos, B.; Fitzgerald, T.D.; Yurlina, M.E.; Young, P.A. Synchronized group foraging, thermoregulation, and growth of eastern tent caterpillars in relation to microclimate. Physiol. Biochem. Zool. 1988, 61, 372–377. [Google Scholar] [CrossRef]
- Denno, R.; Benrey, B. Aggregation facilitates larval growth in the neotropical nymphalid butterfly Chlosyne janais. Ecol. Entomol. 1997, 22, 133–141. [Google Scholar] [CrossRef]
- Allen, P.E. Group size effects on survivorship and adult development in the gregarious larvae of Euselasia chrysippe (Lepidoptera, Riodinidae). Insect Soc. 2010, 57, 199–204. [Google Scholar] [CrossRef]
- Clark, B.R.; Faeth, S.H. The consequences of larval aggregation in the butterfly Chlosyne lacinia. Ecol. Entomol. 2003, 22, 408–415. [Google Scholar] [CrossRef]
- Klok, C.J.; Chown, S.L. Assessing the benefits of aggregation: Thermal biology and water relations of anomalous Emperor Moth caterpillars. Funct. Ecol. 2002, 13, 417–427. [Google Scholar] [CrossRef]
- Su, N.Y.; Tamashiro, M.; Yates, J.R.; Lai, P.Y.; Haverty, M.I. A dye, Sudan Red 7B, as a marking material for foraging studies with the Formosan subterranean termite. Sociobiology 1983, 8, 91–97. [Google Scholar] [CrossRef]
- Grace, J.K.; Abdallay, A. Evaluation of the dye marker Sudan Red 7 B with Reticulitermes flavipes (Isoptera: Rhinotermitidae). Sociobiology 1989, 15, 71–78. [Google Scholar]
- Huang, Q.Y.; Lei, C.L.; Xue, D. Field evaluation of a fipronil bait against subterranean termite Odontotermes formosanus (Isoptera: Termitidae). J. Econ. Entomol. 2006, 99, 455–461. [Google Scholar] [CrossRef]
- Song, D.; Hu, X.P. Effects of dose, donor-recipient interaction time and ratio on fipronil transmission among the Formosan subterranean termite nestmates (Isoptera: Rhinotermitidae). Sociobiology 2006, 48, 237–246. [Google Scholar] [CrossRef]
- Wang, C.; Henderson, G.; Chen, X.; Gautam, B.K. Horizontal transfer of fipronil is enhanced with increased group size in Coptotermes formosanus (Isoptera: Rhinotermitidae). J. Econ. Entomol. 2013, 106, 2630–2632. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).