Spatial-Temporal Patterns of Spruce Budworm Defoliation within Plots in Québec
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Data Collection
2.2. Point Pattern Analyses
2.3. Spatial Autocorrelation Analyses
2.4. Tree Defoliation Regression Model
3. Results
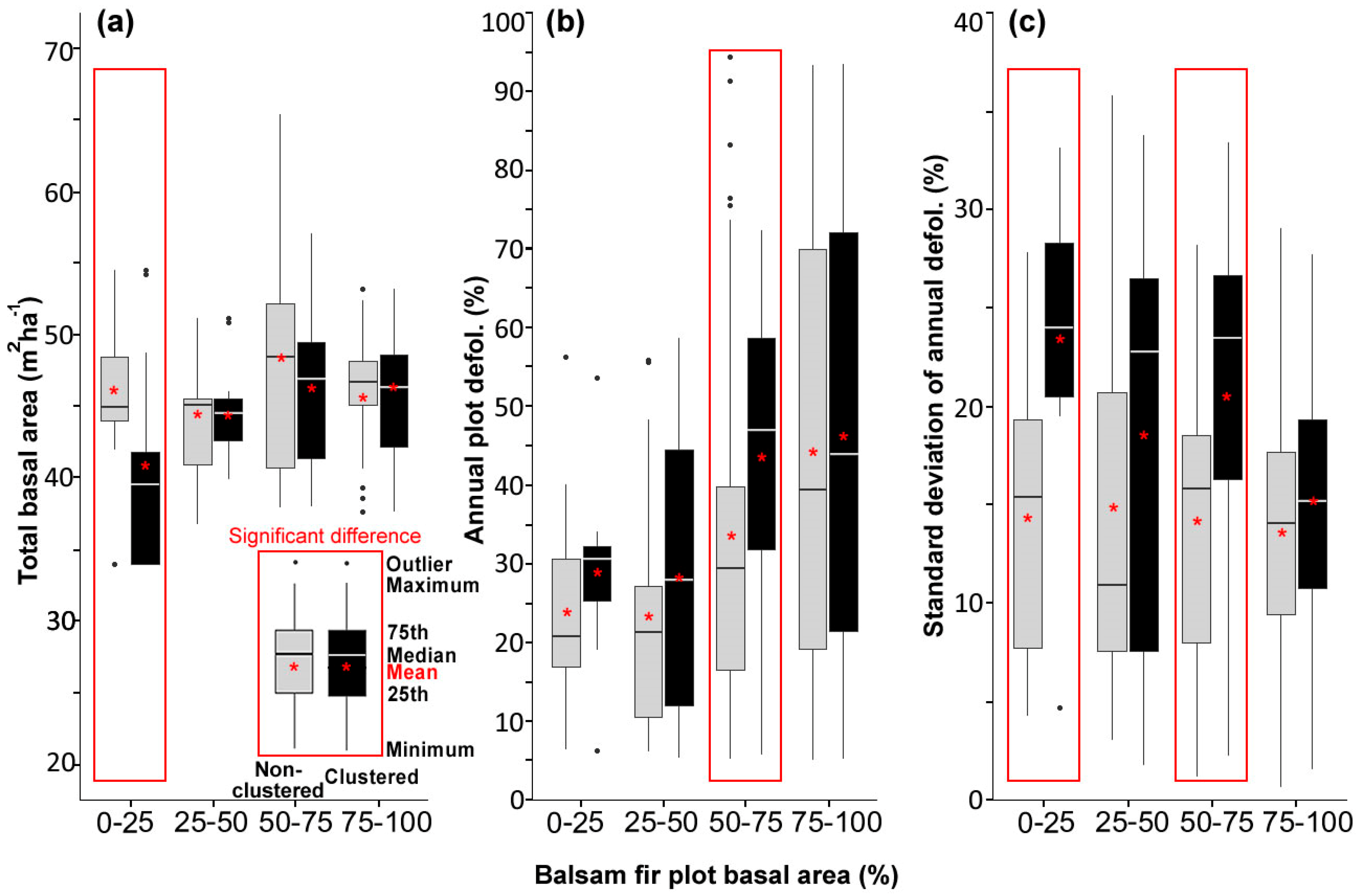
3.1. Stand and Plot Characteristics
3.2. Spatial Patterns of Tree Stems within Plots
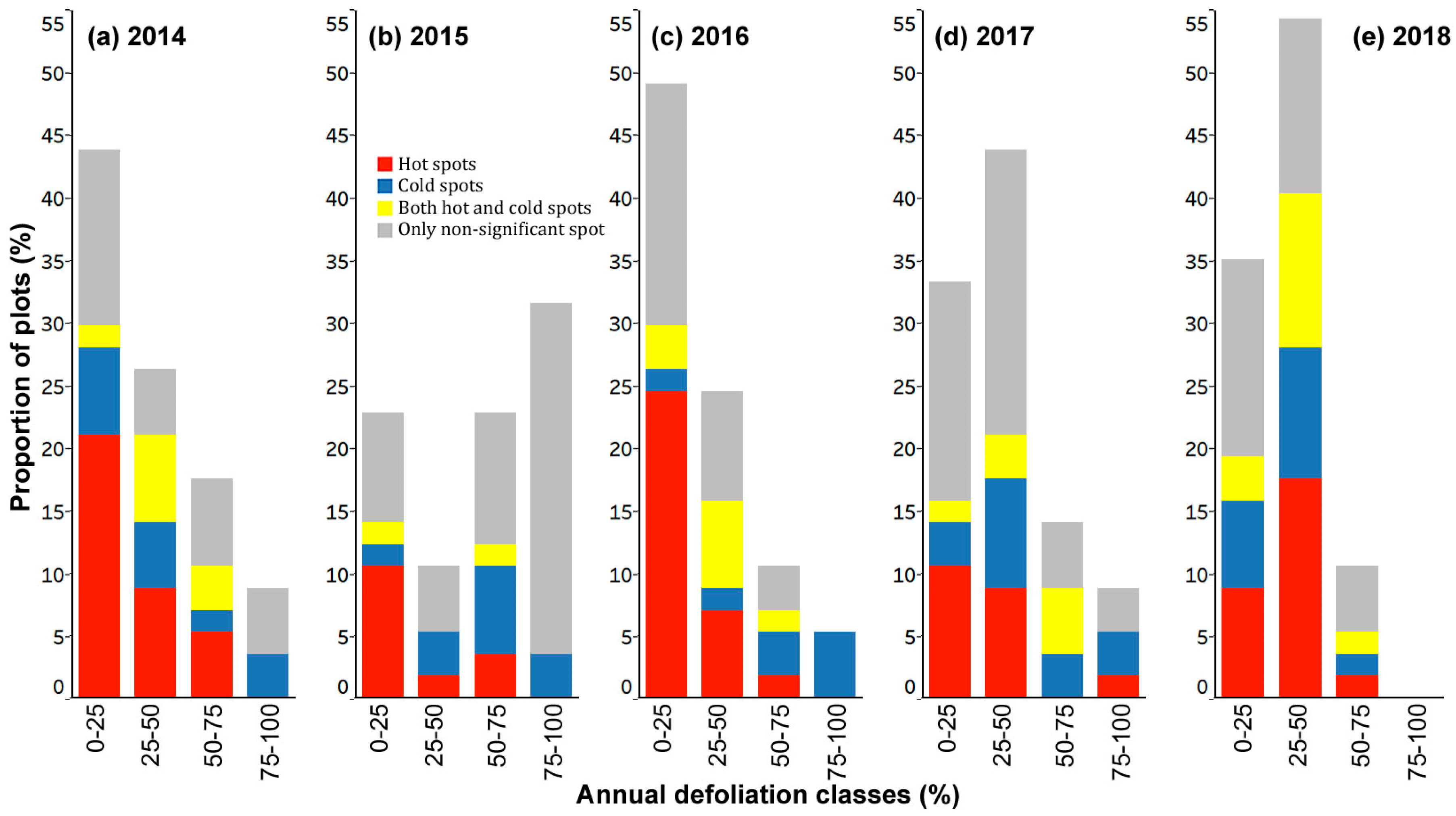
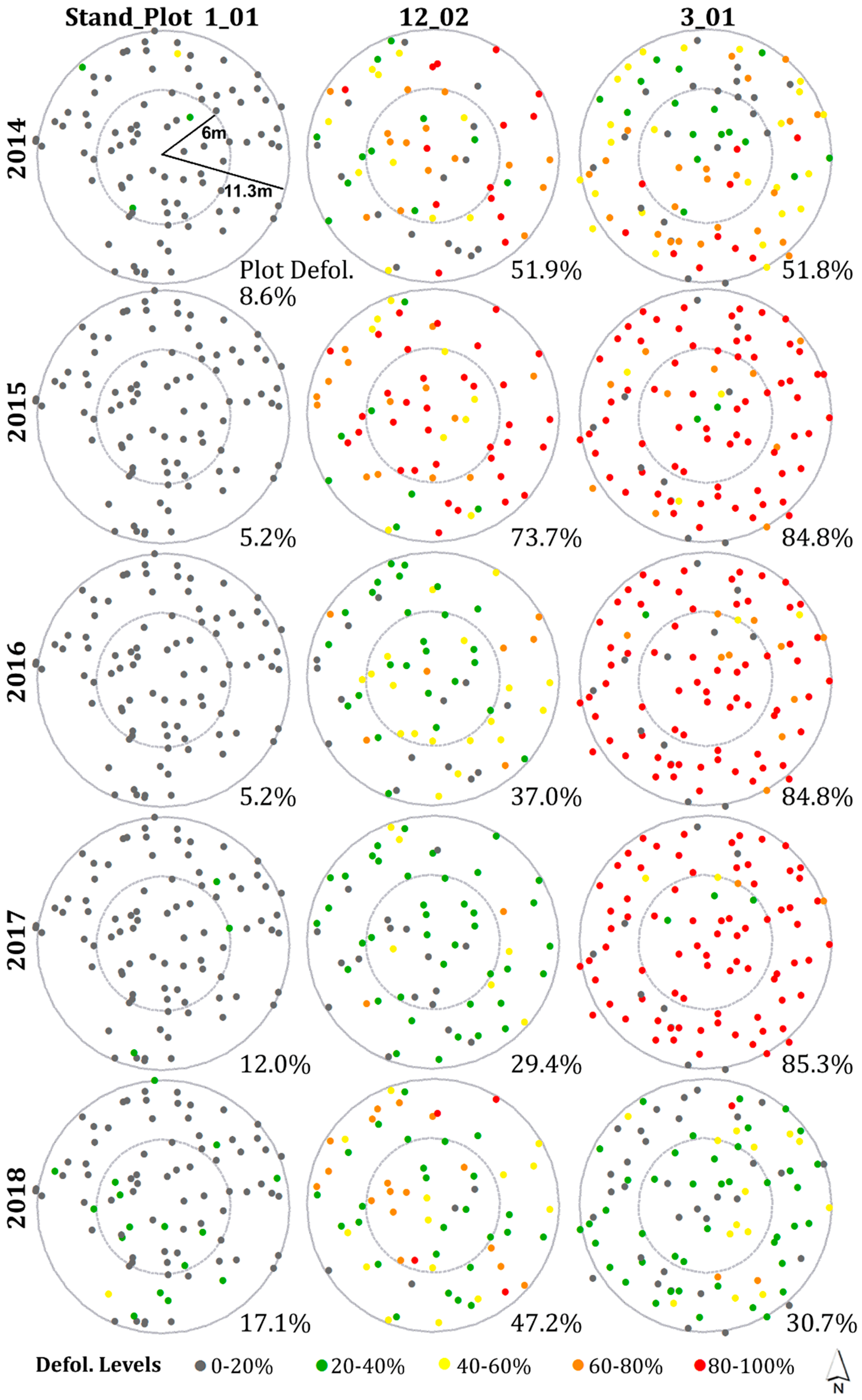
3.3. Spatial Patterns of Current Year Defoliation for Plots
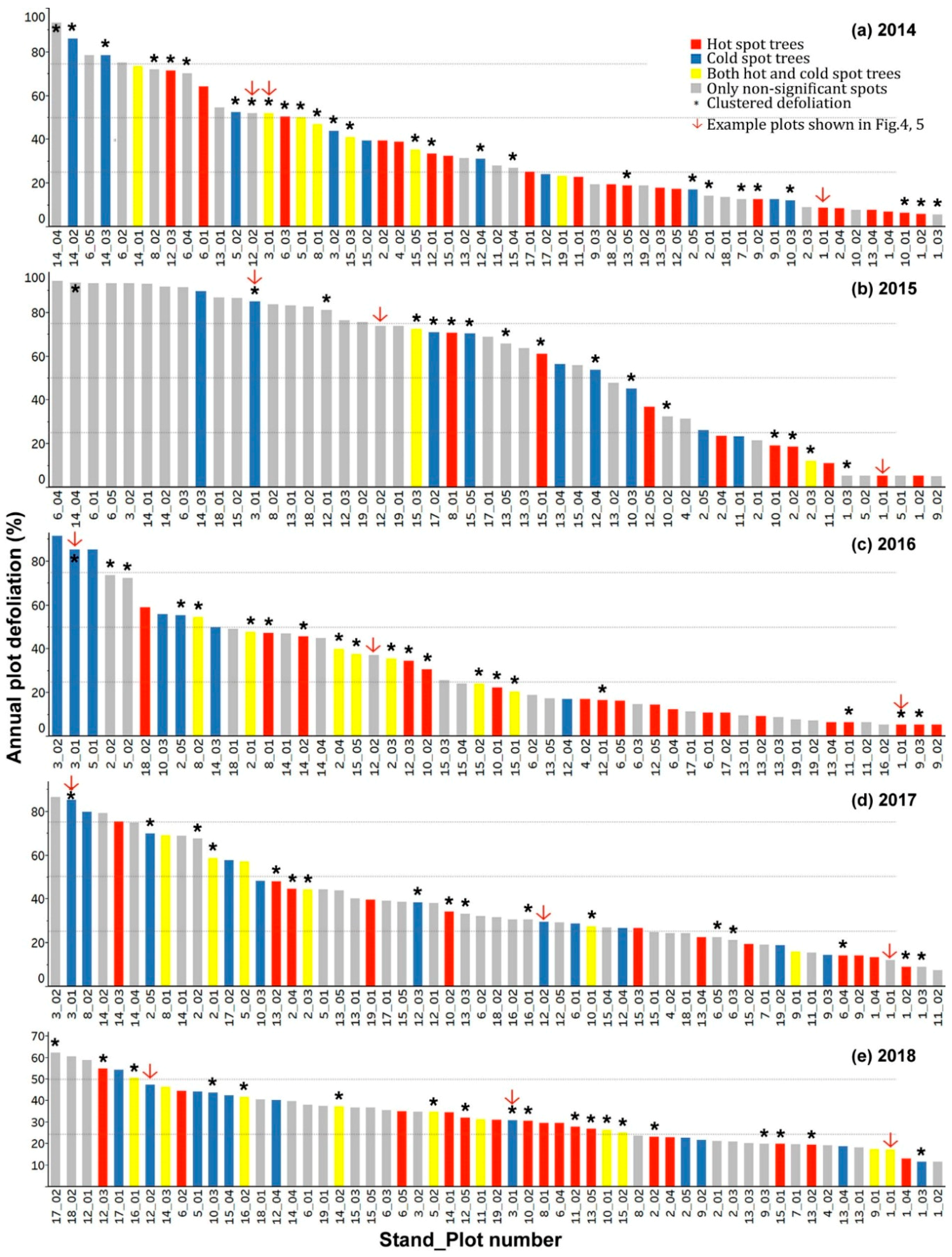
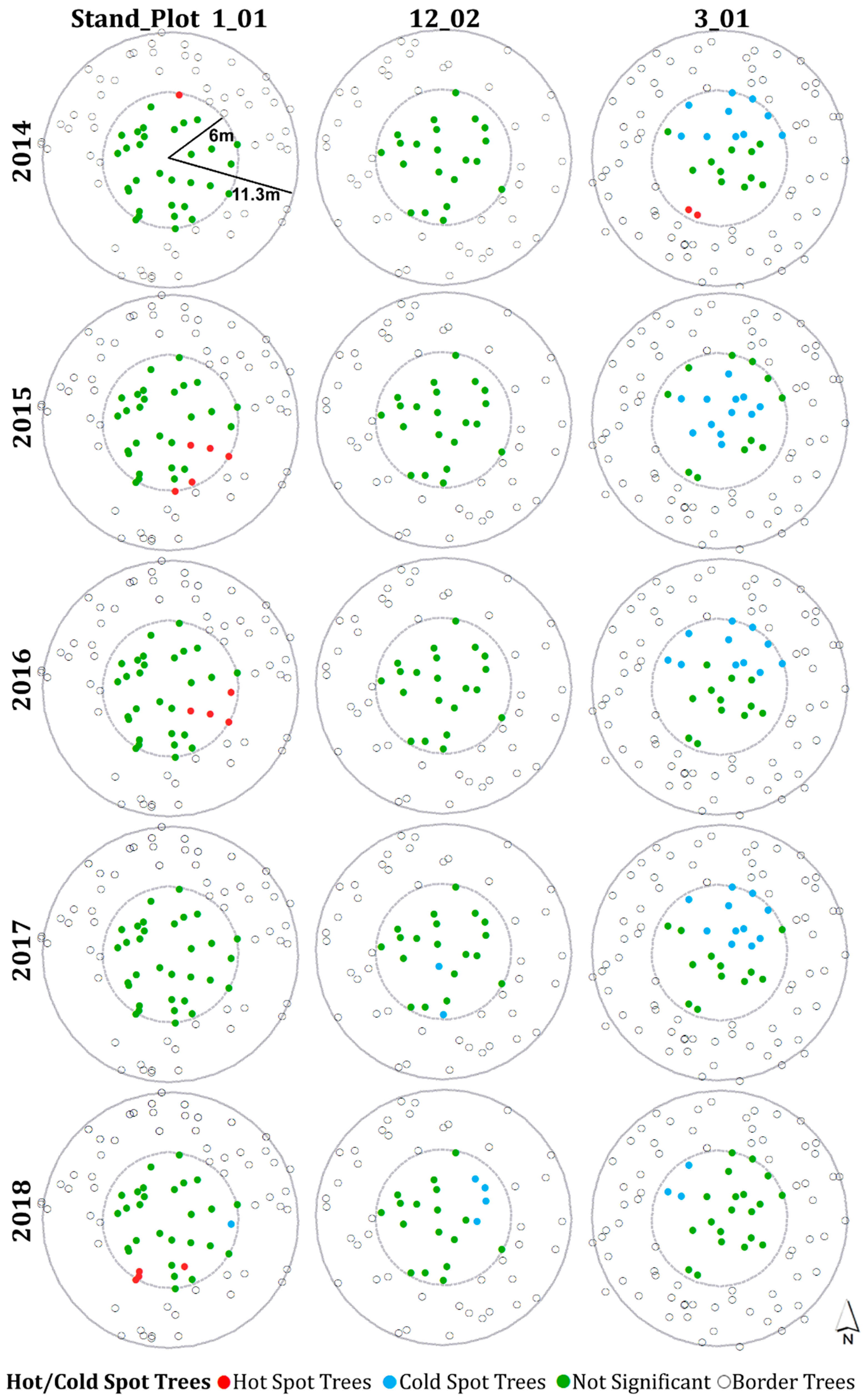
3.4. Hot Spot and Cold Spot Trees within Plots
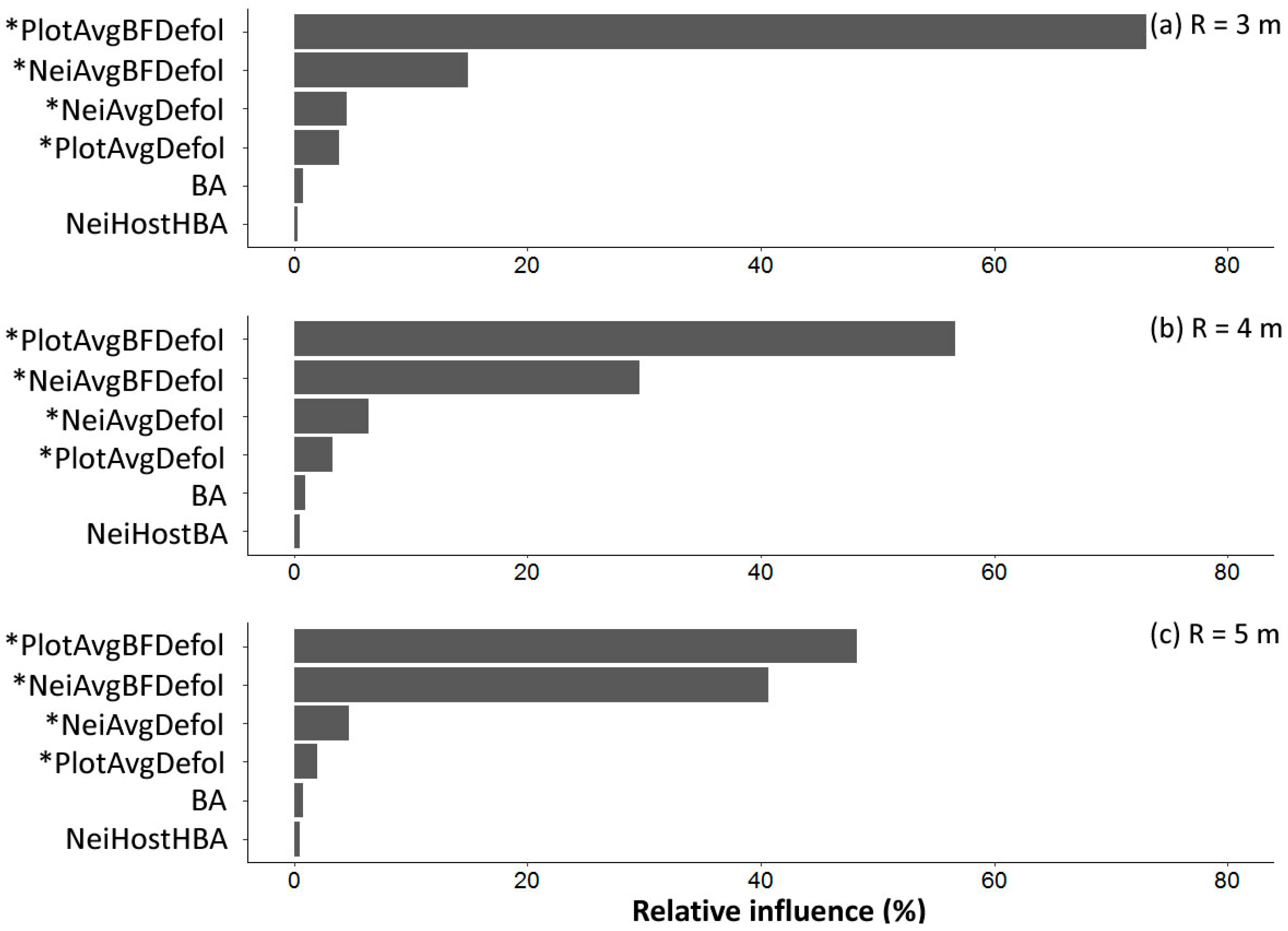
3.5. Prediction of Subject Tree Balsam Fir Defoliation Using Regression Models
4. Discussion
4.1. Is Defoliation of Individual Trees Clustered
4.2. Interpretation of Local Hot and Cold Spot Trees
4.3. Prediction of Subject Balsam Fir Defoliation
5. Conclusions
- Including all host species, 47%, 28%, 35%, 30%, and 33% of plots showed significantly clustered defoliation patterns from 2014 to 2018. Plots with clustered defoliation tended to have higher and less uniform defoliation among trees. Results suggested that spatial defoliation patterns resulted from uneven SBW pressure on trees, perhaps from oviposition site selection.
- Plots with severe defoliation generally tended to exhibit cold spot trees, and plots with light defoliation tended to have hot spot trees, because whether defoliation was high or low enough to be a hot or cold spot depended on the defoliation level of the entire plot.
- Plot-level average defoliation combined with subject tree basal area explained 80% of the variability of subject balsam fir defoliation, which was 2% to 5% higher than variability explained by the neighboring tree defoliation.
- Spatial variability of defoliation decreased with larger radius neighborhoods from 3 to 5 m, suggesting that a neighborhood search radius larger than 5 m (and thus plot sizes larger than 400 m2 (11.3 m radius) to deal with edge effects) may provide better predictions of subject balsam fir defoliation.
- For these primarily balsam fir plots, species composition at both plot and tree levels were not significant predictors of individual balsam fir defoliation.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dale, M.R.; Fortin, M.J. Spatial Analysis: A Guide for Ecologists; Cambridge University Press: Cambridge, UK, 2014. [Google Scholar]
- Sokal, R.R.; Oden, N.L. Spatial autocorrelation in biology 1. Methodology. Biol. J. Linn. Soc. 1977, 10, 199–228. [Google Scholar] [CrossRef]
- Cliff, A.D.; Ord, J.K. Spatial Processes: Models & Applications; Pion Ltd.: London, UK, 1981. [Google Scholar]
- Tobler, W.R. A computer movie simulating urban growth in the Detroit region. Geogr. Econ. 1970, 46, 234–240. [Google Scholar] [CrossRef]
- Augustin, N.H.; Mugglestone, M.A.; Buckland, S.T. The role of simulation in modelling spatially correlated data. Environmetrics 1998, 9, 175–796. [Google Scholar] [CrossRef]
- Knapp, R.A.; Matthews, K.R.; Preisler, H.K.; Jellison, R. Developing probabilistic models to predict amphibian site occupancy in a patchy landscape. Ecol. Appl. 2003, 13, 1069–1082. [Google Scholar] [CrossRef]
- Ryan, P.A.; Lyons, S.A.; Alsemgeest, D.; Thomas, P.; Kay, B.H. Spatial statistical analysis of adult mosquito (Diptera: Culicidae) counts: An example using light trap data, in Redland Shire, southeastern Queensland, Australia. J. Med. Entomol. 2004, 41, 1143–1156. [Google Scholar] [CrossRef] [PubMed]
- Foster, J.R.; Townsend, P.A.; Mladenoff, D.J. Spatial dynamics of a gypsy moth defoliation outbreak and dependence on habitat characteristics. Landsc. Ecol. 2013, 28, 1307–1320. [Google Scholar] [CrossRef]
- Hardy, Y.; Mainville, M.; Schmitt, D.M. Spruce Budworms Handbook: An Atlas of Spruce Budworm Defoliation in Eastern North America, 1938–1980; U.S. Department of Agriculture, Forest Service, Cooperative State Research Service: Washington, DC, USA, 1986.
- Hennigar, C.R.; MacLean, D.A.; Quiring, D.T.; Kershaw, J.A. Differences in spruce budworm defoliation among balsam fir and white, red, and black spruce. For. Sci. 2008, 54, 158–166. [Google Scholar]
- MacLean, D.A. Vulnerability of fir-spruce stands during uncontrolled spruce budworm outbreaks: A review and discussion. For. Chron. 1980, 56, 213–221. [Google Scholar] [CrossRef]
- MacLean, D.A. Effects of spruce budworm outbreaks on the productivity and stability of balsam fir forests. For. Chron. 1984, 60, 273–279. [Google Scholar] [CrossRef]
- Baskerville, G.L.; MacLean, D.A. Budworm-Caused Mortality and 20-Year Recovery in Immature Balsam fir Stands; Inf. Rep. M-X-102; Maritimes Forest Research Centre, Canadian Forestry Service: Fredericton, NB, Canada, 1979; p. 56.
- MacLean, D.A.; Ostaff, D.P. Patterns of balsam fir mortality caused by an uncontrolled spruce budworm outbreak. Can. J. For. Res. 1989, 19, 1087–1095. [Google Scholar] [CrossRef]
- Erdle, T.A.; MacLean, D.A. Stand growth model calibration for use in forest pest impact assessment. For. Chron. 1999, 75, 141–152. [Google Scholar] [CrossRef]
- Chen, C.; Weiskittel, A.; Bataineh, M.; MacLean, D.A. Even low levels of spruce budworm defoliation affect mortality and ingrowth but net growth is more driven by competition. Can. J. For. Res. 2017, 47, 1546–1556. [Google Scholar] [CrossRef]
- Lamb, S.M.; MacLean, D.A.; Hennigar, C.R.; Pitt, D.G. Forecasting forest inventory using imputed tree lists for LiDAR grid cells and a tree-list growth model. Forests 2018, 9, 167. [Google Scholar] [CrossRef]
- MacLean, D.A.; Lidstone, R.G. Defoliation by spruce budworm: Estimation by ocular and shoot-count methods and variability among branches, trees, and stands. Can. J. For. Res. 1982, 12, 582–594. [Google Scholar] [CrossRef]
- Hardy, Y.J.; Lafond, A.; Hamel, L. The epidemiology of the current spruce budworm outbreak in Quebec. For. Sci. 1983, 29, 715–725. [Google Scholar]
- Gray, D.R.; MacKinnon, W.E. Outbreak patterns of the spruce budworm and their impacts in Canada. For. Chron. 2006, 82, 550–561. [Google Scholar] [CrossRef]
- Zhao, K.; MacLean, D.A.; Hennigar, C.R. Spatial variability of spruce budworm defoliation at different scales. For. Ecol. Manag. 2014, 328, 10–19. [Google Scholar] [CrossRef]
- Ostaff, D.P.; MacLean, D.A. Spruce budworm populations, defoliation, and changes in stand condition during an uncontrolled spruce budworm outbreak on Cape Breton Island, Nova Scotia. Can. J. For. Res. 1989, 19, 1077–1086. [Google Scholar] [CrossRef]
- Alfaro, R.I.; Shore, T.L.; Wegwitz, E. Defoliation and mortality caused by western spruce budworm: Variability in a Douglas-fir stand. J. Entomol. Soc. 1984, 81, 33–38. [Google Scholar]
- Su, Q.; MacLean, D.A.; Needham, T.D. The influence of hardwood content on balsam fir defoliation by spruce budworm. Can. J. For. Res. 1996, 26, 1620–1628. [Google Scholar] [CrossRef]
- Zhang, B.; Maclean, D.A.; Johns, R.C.; Eveleigh, E.S. Effects of hardwood content on balsam fir defoliation during the building phase of a spruce budworm outbreak. Forests 2018, 9, 530. [Google Scholar] [CrossRef]
- Bognounou, F.; Grandpre, L.D.; Pureswaran, D.S.; Kneeshaw, D. Temporal variation in plant neighborhood effects on the defoliation of primary and secondary hosts by an insect pest. Ecosphere 2017, 8, 1–15. [Google Scholar] [CrossRef]
- Morris, R.F.; Mott, D.G. Dispersal and the spruce budworm. In The Dynamics of Epidemic Spruce Budworm Populations; Morris, R.F., Ed.; Memoirs of the Entomological Society of Canada: Ottawa, ON, Canada, 1963; Volume 95, pp. 180–189. [Google Scholar]
- Morris, R.F. The development of sampling techniques for forest insect defoliators, with particular reference to the spruce budworm. Can. J. Zool. 1955, 33, 107–223. [Google Scholar] [CrossRef]
- Beckwith, R.C.; Burnell, D.G. Spring larval dispersal of the western spruce budworm (Lepidoptera: Tortricidae) in north-central Washington. Environ. Entomol. 1982, 11, 828–832. [Google Scholar] [CrossRef]
- Miller, C.A. The measurement of spruce budworm populations and mortality during the first and second larval instars. Can. J. Zool. 1958, 36, 409–422. [Google Scholar] [CrossRef]
- Reay-Jones, F.P.F. Spatial distribution of the cereal leaf beetle (Coleoptera: Chrysomelidae) in wheat. Environ. Entomol. 2010, 39, 1943–1952. [Google Scholar] [CrossRef] [PubMed]
- Mercader, R.J.; Siegert, N.W.; Liebhold, A.M.; Mccullough, D.G. Dispersal of the emerald ash borer, Agrilus planipennis, in newly-colonized sites. Agric. For. Entomol. 2009, 11, 421–424. [Google Scholar] [CrossRef]
- Bélanger, L.; Bergeron, Y.; Camiré, C. Ecological Land Survey in Quebec. For. Chron. 1992, 68, 42–52. [Google Scholar] [CrossRef]
- QMRNF: Québec Ministère des Ressources Naturelles et de la Faune. Aires Infestées par la Tordeuse des Bourgeons de l’épinette au Québec en 2014. Available online: http://www.mffp.gouv.qc.ca/publications/forets/fimaq/insectes/tordeuse/TBE_2014_P.pdf (accessed on 20 October 2017).
- Donovan, S.D.; MacLean, D.A.; Kershaw, J.A.; Lavigne, M.B. Quantification of forest canopy changes caused by spruce budworm defoliation using digital hemispherical imagery. Agric. For. Meteorol. 2018, 262, 89–99. [Google Scholar] [CrossRef]
- MacLean, D.A.; MacKinnon, W.E. Sample sizes required to estimate defoliation of spruce and balsam fir caused by spruce budworm accurately. North. J. Appl. For. 1998, 15, 135–140. [Google Scholar]
- Ebdon, D. Statistics in Geography: A Practical Approach; Basil Blackwell Ltd.: New York, NY, USA, 1985. [Google Scholar]
- Liu, C. A comparison of five distance-based methods for spatial pattern analysis. J. Veg. Sci. 2001, 12, 411–416. [Google Scholar] [CrossRef]
- Pielou, E.C. The use of point-to-plant distances in the study of the pattern of plant populations. J. Ecol. 1959, 47, 607–613. [Google Scholar] [CrossRef]
- Clark, P.J.; Evans, F.C. Distance to nearest neighbor as a measure of spatial relationships in populations. Ecol. Soc. Am. 1954, 35, 445–453. [Google Scholar] [CrossRef]
- Moran, P.A.P. Notes on continuous stochastic phenomena. Biometrika 1950, 37, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Sokal, R.R.S.; Oden, N.L.; Thomson, B.A. Local spatial autocorrelation in biological variables. Biol. J. Linn. Soc. 1998, 65, 41–62. [Google Scholar] [CrossRef]
- Getis, A.; Ord, J.K. The analysis of spatial association by use of distance statistics. Geogr. Anal. 1992, 24, 189–206. [Google Scholar] [CrossRef]
- Ord, J.K.; Getis, A. Local spatial autocorrelation statistics: Distributional issues and an application. Geogr. Anal. 1995, 27, 286–305. [Google Scholar] [CrossRef]
- Monserud, R.A.; Ek, A.R. Plot edge bias in forest stand growth simulation models. Can. J. For. Res. 1974, 4, 419–423. [Google Scholar] [CrossRef]
- Sui, D.Z.; Hugill, P.J. A GIS-based spatial analysis on neighborhood effects and voter turn-out: A case study in College Station, Texas. Polit. Geogr. 2002, 21, 159–173. [Google Scholar] [CrossRef]
- Ripley, B.D. Tests of “randomness” for spatial point patterns. J. R. Stat. Soc. 1979, 41, 368–374. [Google Scholar] [CrossRef]
- Friedman, J.H. Greedy function approximation: A gradient boosting machine. Ann. Stat. 2001, 29, 1189–1232. [Google Scholar] [CrossRef]
- Ridgeway, G. Generalized boosted models: A guide to the GBM package. R Package Vignette. 2007. [Google Scholar]
- Kuhn, M. Building Predictive Models in R Using the caret Package. J. Stat. Softw. 2008, 28, 1–26. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Elith, J.; Leathwick, J.R.; Hastie, T. A working guide to boosted regression trees. J. Anim. Ecol. 2008, 77, 802–813. [Google Scholar] [CrossRef] [PubMed]
- Lindstrom, M.J.; Bates, D.M. Nonlinear mixed effects models for repeated measures data. Int. Biom. Soc. 1990, 46, 673–687. [Google Scholar] [CrossRef]
- Batzer, H.O. Hibernation site and dispersal of spruce budworm larvae as related to damage of sapling balsam fir. J. Environ. Entomol. 1968, 61, 216–220. [Google Scholar] [CrossRef]
- Kabos, S.; Csillag, F. The analysis of spatial association on a regular lattice by join-count statistics without the assumption of first-order homogeneity. Comput. Geosci. 2002, 28, 901–910. [Google Scholar] [CrossRef]
- Boots, B. Developing local measures of spatial association for categorical data. J. Geogr. Syst. 2003, 5, 139–160. [Google Scholar] [CrossRef]
- Getis, A. A history of the concept of spatial autocorrelation: A geographer’s perspective. Geogr. Anal. 2008, 40, 297–309. [Google Scholar] [CrossRef]
- Taylor, S.L.; MacLean, D.A. Spatiotemporal patterns of mortality in declining balsam fir and spruce stands. For. Ecol. Manag. 2007, 253, 188–201. [Google Scholar] [CrossRef]
- Taylor, S.L.; MacLean, D.A. Legacy of insect defoliators: Increased wind-related mortality two decades after a spruce budworm outbreak. For. Sci. 2009, 55, 256–267. [Google Scholar]
| Stand No. | No. Plots 1 | Density (stem ha−1) | DBH 2 (cm) | Height (m) | Basal Area (m2 ha−1) | Species Composition 3 (% Basal Area) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| σ | σ | σ | σ | BF | BS | WS | HW | OSW | ||||||
| 1 | 4 | 1919 | 289 | 15.0 | 1.1 | 13.7 | 0.9 | 38.8 | 3.8 | 54 | 30 | 4 | 6 | 7 |
| 2 | 5 | 1775 | 417 | 17.0 | 1.4 | 16.5 | 1.0 | 42.8 | 4.3 | 51 | 42 | 3 | 3 | |
| 3 | 2 | 1913 | 636 | 16.0 | 1.4 | 16.0 | 0.9 | 41.8 | 6.7 | 83 | 1 | 11 | 5 | |
| 4 | 1 | 1200 | 20.1 | 18.0 | 39.8 | 6 | 27 | 65 | 2 | |||||
| 5 | 2 | 1825 | 159 | 15.8 | 0.1 | 16.1 | 0.2 | 38.1 | 1.7 | 98 | 1 | 1 | ||
| 6 | 5 | 1600 | 329 | 16.5 | 2.2 | 14.8 | 2.0 | 38.7 | 4.0 | 77 | 7 | 4 | 7 | 7 |
| 7 | 1 | 1950 | 14.9 | 14.8 | 38.3 | 78 | 20 | 2 | ||||||
| 8 | 2 | 1800 | 159 | 15.8 | 0.4 | 15.5 | 0.6 | 38.1 | 1.7 | 70 | 20 | 5 | 6 | |
| 9 | 3 | 1808 | 426 | 16.7 | 2.6 | 16.2 | 1.3 | 44.2 | 4.2 | 86 | 2 | 11 | 1 | |
| 10 | 3 | 1475 | 275 | 17.2 | 0.9 | 15.9 | 0.5 | 36.3 | 2.7 | 22 | 70 | 7 | 1 | |
| 11 | 2 | 1238 | 106 | 18.1 | 1.0 | 16.5 | 0.7 | 34.9 | 1.8 | 54 | 41 | 2 | 2 | |
| 12 | 5 | 1775 | 191 | 17.0 | 0.8 | 17.3 | 1.0 | 43.5 | 5.7 | 50 | 38 | 9 | 2 | |
| 13 | 5 | 1250 | 317 | 20.0 | 1.7 | 18.4 | 1.1 | 42.2 | 5.5 | 57 | 41 | 2 | ||
| 14 | 4 | 1844 | 236 | 16.5 | 0.8 | 16.2 | 0.6 | 42.4 | 4.5 | 90 | 10 | |||
| 15 | 5 | 1730 | 224 | 16.7 | 0.4 | 15.2 | 0.7 | 40.3 | 5.3 | 69 | 28 | 2 | 1 | |
| 16 | 2 | 1113 | 177 | 22.2 | 1.1 | 19.2 | 0.2 | 47.7 | 2.7 | 55 | 43 | 2 | ||
| 17 | 2 | 1075 | 159 | 20.3 | 0.9 | 16.1 | 0.2 | 40.0 | 2.0 | 71 | 11 | 6 | 15 | |
| 18 | 2 | 1325 | 265 | 17.5 | 0.3 | 13.5 | 0.6 | 44.0 | 4.5 | 80 | 20 | |||
| 19 | 2 | 1763 | 636 | 17.3 | 2.9 | 14.2 | 3.0 | 51.3 | 8.5 | 59 | 11 | 14 | 18 | |
| Balsam Fir | Black Spruce | White Spruce | Hardwoods | All Host Species | |
|---|---|---|---|---|---|
| Clustered | 0 | 1 (2%) | 0 | 3 (5%) | 0 |
| Dispersed | 21 (37%) | 3 (5%) | 1 (2%) | 2 (4%) | 22 (39%) |
| Random | 35 (61%) | 20 (35%) | 12 (21%) | 25 (44%) | 35 (61%) |
| Year | Balsam Fir | Black Spruce | White Spruce | All Host Species |
|---|---|---|---|---|
| 2014 | 19 (33%) | 1 (2%) | 2 (4%) | 27 (47%) |
| 2015 | 11 (19%) | 3 (5%) | 0 | 16 (28%) |
| 2016 | 13 (23%) | 1 (2%) | 0 | 20 (35%) |
| 2017 | 6 (11%) | 2 (4%) | 0 | 17 (30%) |
| 2018 | 15 (26%) | 2 (4%) | 0 | 19 (33%) |
| Predictor Variables | Description |
|---|---|
| Plot level | |
| PlotAvgDefol | Average annual defoliation of all host species per plot (%) |
| PlotAvgBFDefol | Average annual defoliation of balsam fir per plot (%) |
| PlotBFBA | % basal area of balsam fir |
| PlotBSBA | % basal area of black spruce |
| PlotWSBA | % basal area of white spruce |
| PlotHWBA | % basal area of hardwoods |
| Spray | Dummy variable: whether the plot was sprayed by insecticide (1) in corresponding given year or not (0) |
| Tree level 1 | |
| BA | Basal area of the subject balsam fir (m2 ha−1) |
| PreYearDefol | Annual defoliation of subject balsam fir in previous year (%) |
| NeiAvgDefol | Average annual defoliation of neighboring1 host trees (%) |
| NeiAvgBFDefol | Average annual defoliation of neighboring balsam fir (%) |
| NeiAvgBSDefol | Average annual defoliation of neighboring black spruce (%) |
| NeiAvgWSDefol | Average annual defoliation of neighboring white spruce (%) |
| NeiHostBA | Total basal area of neighboring host trees (m2 ha−1) |
| NeiBFBA | Total basal area of neighboring balsam fir (m2 ha−1) |
| NeiSPBA | Total basal area of neighboring spruce trees (m2 ha−1) |
| NeiHWBA | Total basal area of neighboring hardwoods (m2 ha−1) |
| NeiHBA | Total basal area of all trees with basal area greater than the subject balsam fir in the neighborhood (m2 ha−1) |
| NeiHostHBA | Total basal area of host trees with basal area greater than the subject balsam fir in the neighborhood (m2 ha−1) |
| NeiSPHBA | Total basal area of spruce trees with basal area greater than the subject balsam fir in the neighborhood (m2 ha−1) |
| NeiBFHBA | Total basal area of balsam fir with basal area greater than the subject balsam fir in the neighborhood (m2 ha−1) |
| Candidate Models | Predictors | Fit Statistics 1 | ||
|---|---|---|---|---|
| Adjusted r2 | RMSE | Bias | ||
| Model 1 | PlotAvgBFDefol + BA | 0.8001 | 0.1411 | 0.0028 |
| Search radius = 3 m | ||||
| Model 2 | NeiAvgBFDefol + BA | 0.7539 | 0.1566 | 0.0017 |
| Model 3 | PlotAvgBFDefol + BA + NeiHostHBA | 0.8015 | 0.1406 | 0.0025 |
| Search radius = 4 m | ||||
| Model 4 | NeiAvgBFDefol + BA | 0.7823 | 0.1473 | 0.0019 |
| Model 5 | PlotAvgBFDefol + BA + NeiHostBA | 0.8007 | 0.1409 | 0.0027 |
| Search radius = 5 m | ||||
| Model 6 | NeiAvgBFDefol + BA | 0.7889 | 0.1450 | 0.0025 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, M.; MacLean, D.A.; Hennigar, C.R.; Ogilvie, J. Spatial-Temporal Patterns of Spruce Budworm Defoliation within Plots in Québec. Forests 2019, 10, 232. https://doi.org/10.3390/f10030232
Li M, MacLean DA, Hennigar CR, Ogilvie J. Spatial-Temporal Patterns of Spruce Budworm Defoliation within Plots in Québec. Forests. 2019; 10(3):232. https://doi.org/10.3390/f10030232
Chicago/Turabian StyleLi, Mingke, David A. MacLean, Chris R. Hennigar, and Jae Ogilvie. 2019. "Spatial-Temporal Patterns of Spruce Budworm Defoliation within Plots in Québec" Forests 10, no. 3: 232. https://doi.org/10.3390/f10030232
APA StyleLi, M., MacLean, D. A., Hennigar, C. R., & Ogilvie, J. (2019). Spatial-Temporal Patterns of Spruce Budworm Defoliation within Plots in Québec. Forests, 10(3), 232. https://doi.org/10.3390/f10030232





