Photocatalytic, Antimicrobial and Biocompatibility Features of Cotton Knit Coated with Fe-N-Doped Titanium Dioxide Nanoparticles
Abstract
:1. Introduction
2. Results
2.1. Photocatalyst Characterization
2.1.1. X-ray Diffraction Analysis
2.1.2. Mössbauer Spectroscopy
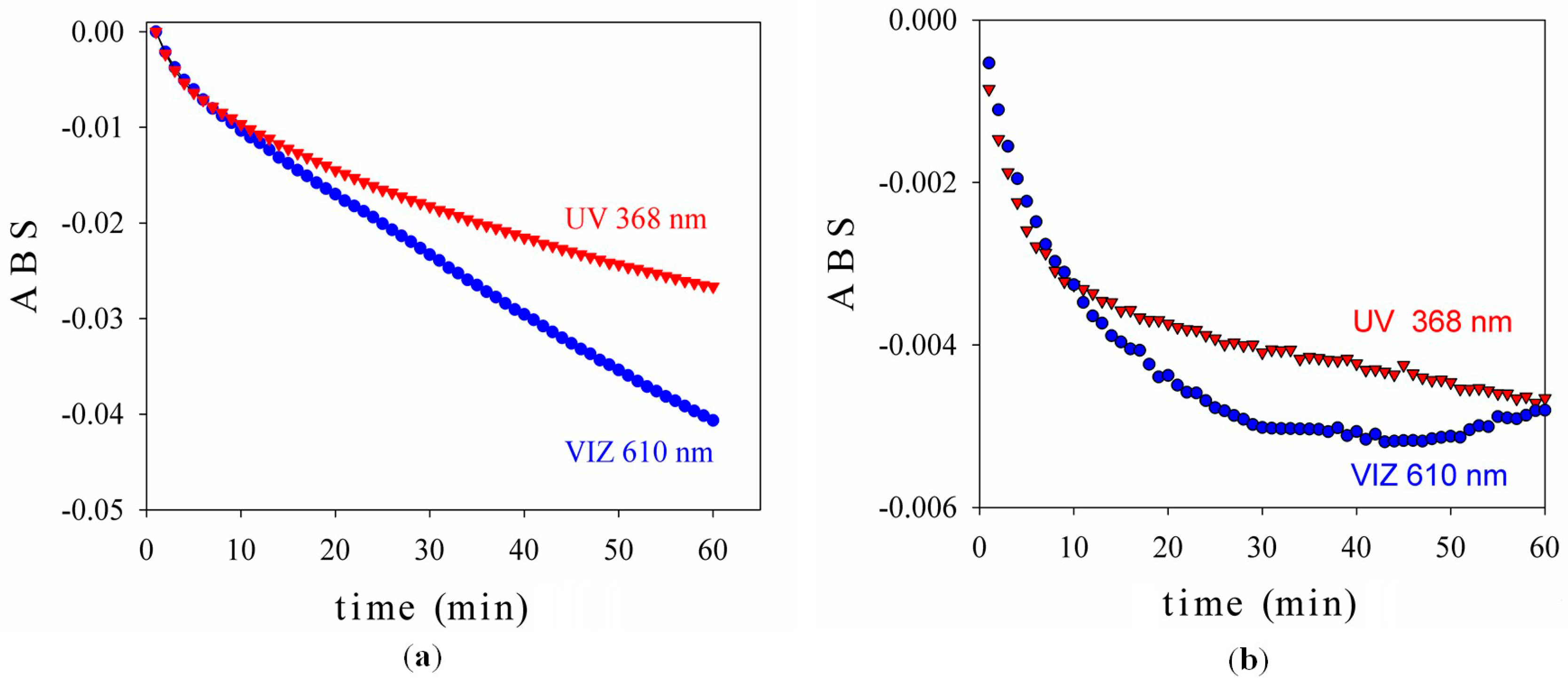
2.1.3. Photocatalytic Test
2.2. Characterization of Treated Textile Materials by SEM-EDX
2.3. Assessment of the Photocatalytic Effects of the Materials Treated with TiO2-1% Fe-N Samples
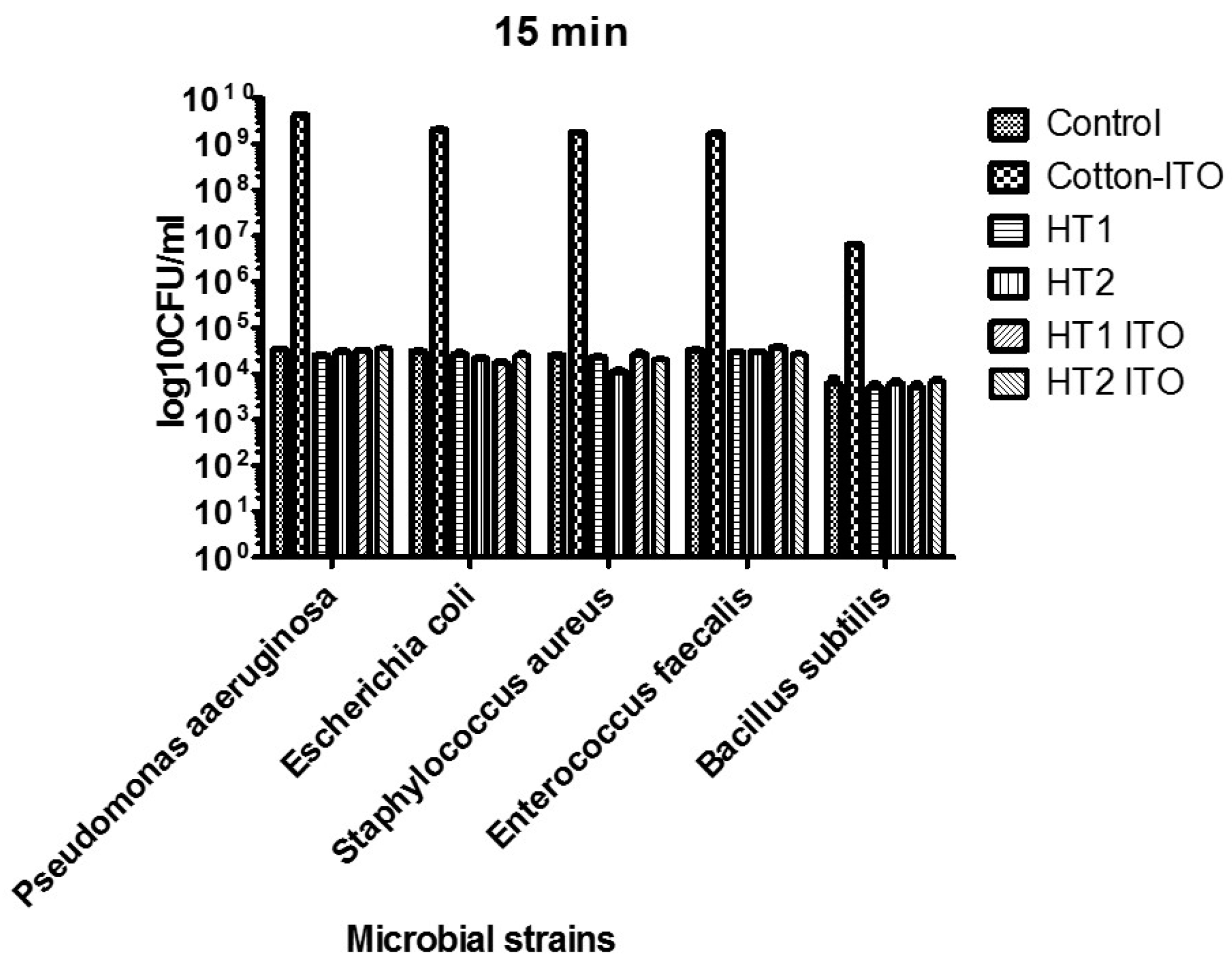
2.4. Antimicrobial Activity
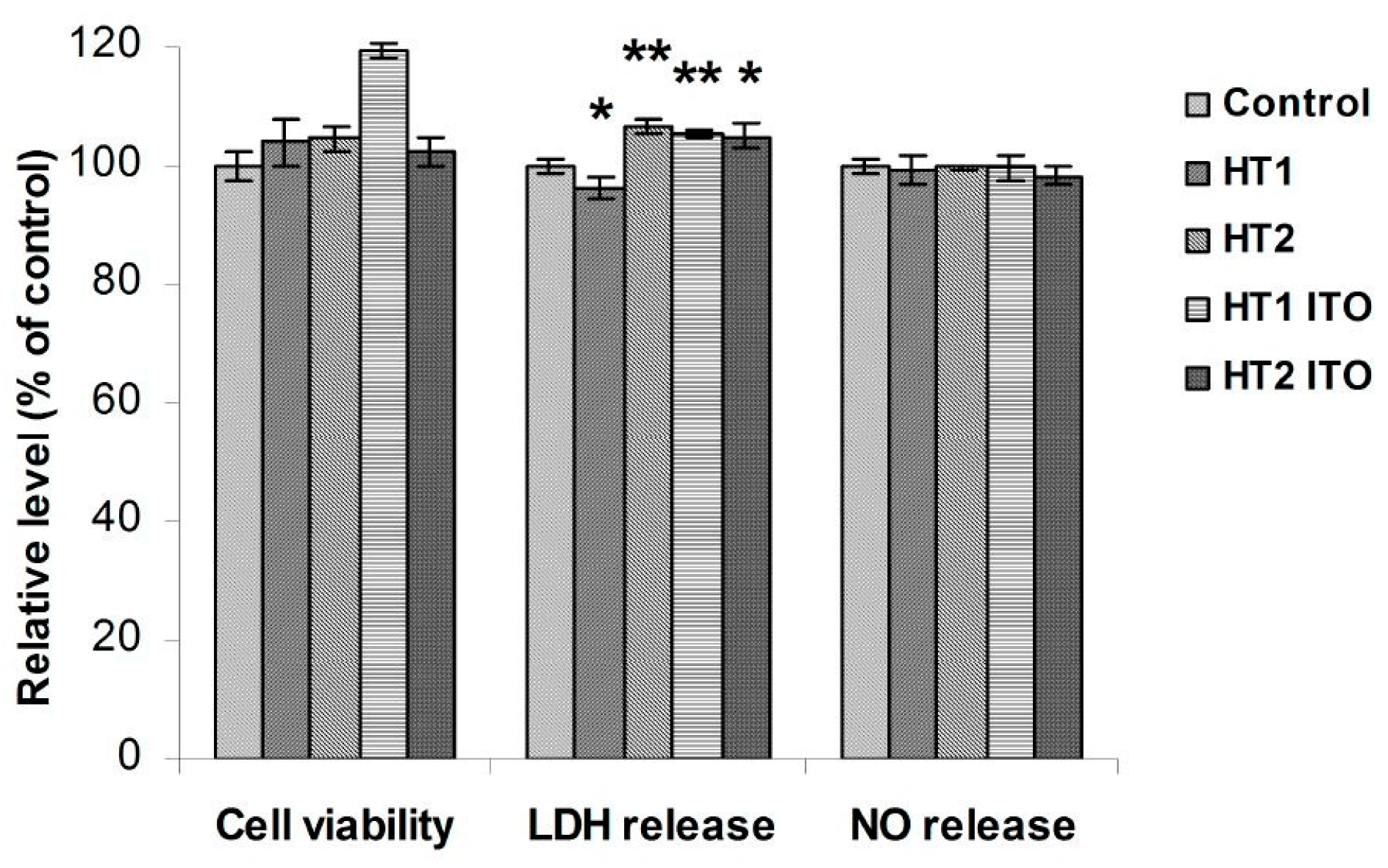
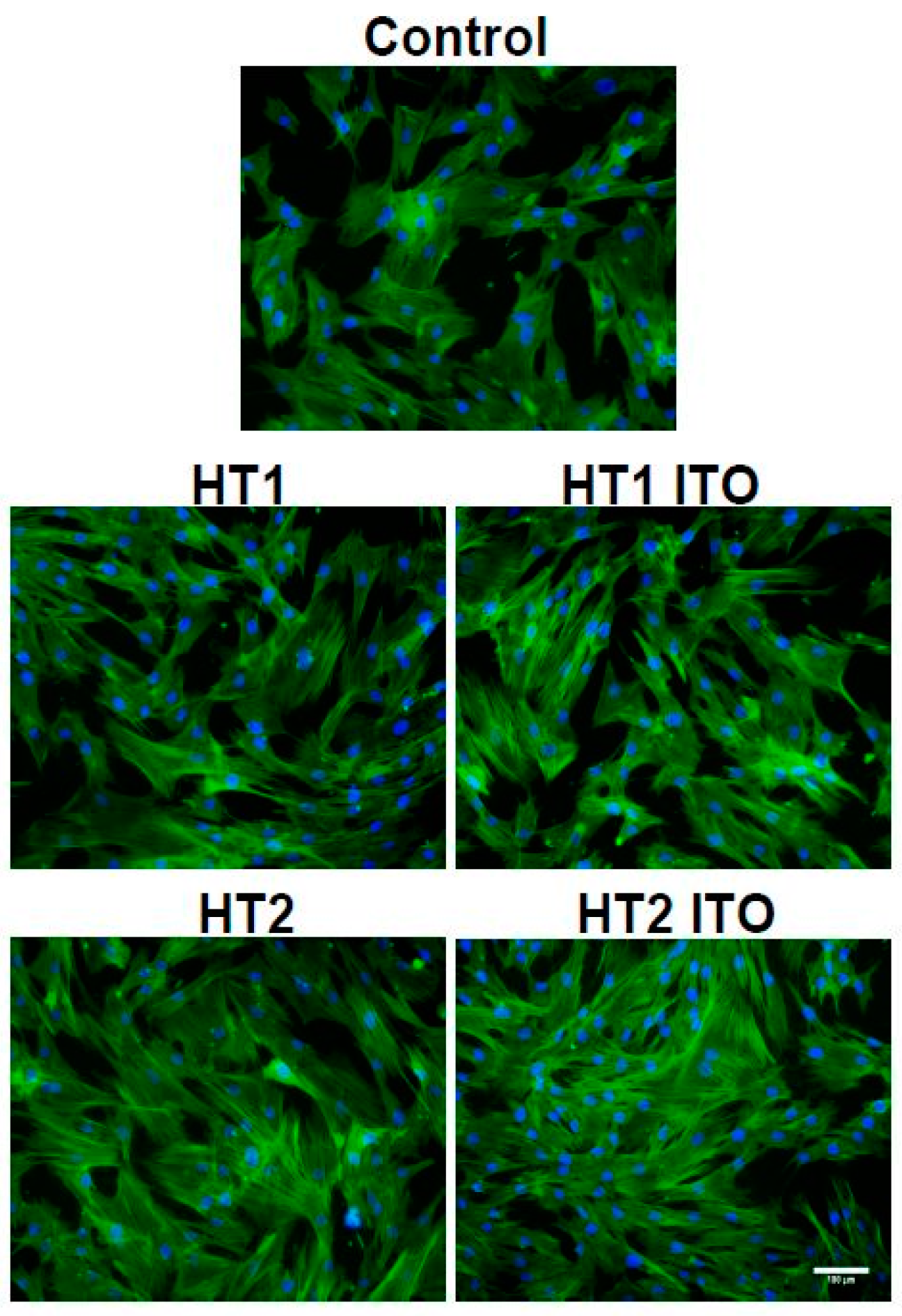
2.5. Biocompatibility of Cotton Knit Treated with TiO2-1% Fe-N Samples
3. Discussion
4. Materials and Methods
4.1. Synthesis of Photocatalysts
4.2. Preparation of the Photocatalysts for Deposition on Textiles
4.3. Cotton Knit Treatment
4.3.1. Method P1
4.3.2. Method P2
4.4. Photocatalyst Characterization
- L* represents lightness, a* signifies the red/green value and b* is the yellow/blue value;
- dL* = L*sample − L*control; if dL* is positive, the sample is lighter than the control and, if dL* is negative, the sample is darker than the control;
- da* = a*sample − a* control; if da* is positive, the sample is redder than the control. If da* is negative, the sample is greener than the control;
- db* = b*sample − b* control; if db* is positive, the sample is yellower than the control. If db* is negative, the sample is bluer than the control;
- dE* represents the total difference or distance on the CIELAB diagram as a single value for colour (da*, db*) and lightness (dL*) and, is calculated according to the following formula: dE* = .
4.5. Antimicrobial Activity Assay
4.6. In Vitro Biocompatibility Assessment
4.7. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ahmed, S.; Rasul, M.G.; Brown, R.; Hashib, M.A. Influence of parameters on the heterogeneous photocatalytic degradation of pesticides and phenolic contaminants in wastewater: A short review. J. Environ. Manag. 2010, 92, 311–330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abid, M.F.; Abdulrahman, A.A.; Hamza, N.H. Hydrodynamic and kinetic study of a hybrid detoxification process with zero liquid discharge system in an industrial wastewater treatment. J. Environ. Health Sci. Eng. 2014, 12, 145. [Google Scholar] [CrossRef] [PubMed]
- Ong, S.T.; Cheong, W.S.; Hung, Y.T. Photodegradation of commercial dye, methylene blue using immobilized TiO2. IPCBEE 2012, 43, 109–113. [Google Scholar]
- Kozlova, E.A.; Safatov, A.S.; Kiselev, S.A.; Marchenko, V.Y.; Sergeev, A.A.; Skarnovich, M.O.; Emelyanova, E.K.; Smetannikova, M.A.; Buryak, G.A.; Vorontsov, A.V. Inactivation and mineralization of aerosol deposited model pathogenic microorganisms over TiO2 and Pt/TiO2. Environ. Sci. Technol. 2010, 44, 5121–5126. [Google Scholar] [CrossRef] [PubMed]
- Westerhoff, P.; Song, G.; Hristovski, K.; Kiser, M.A. Occurrence and removal of titanium at full scale wastewater treatment plants: Implications for TiO2 nanomaterials. J. Environ. Monit. 2011, 13, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Kiser, M.A.; Westerhoff, P.; Benn, T.; Wang, Y.; Pérez-Rivera, J.; Hristovski, K. Titanium nanomaterial removal and release from wastewater treatment plants. Environ. Sci. Technol. 2009, 43, 6757–6763. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Ibáñez, P.; Blanco, J.; Malato, S.; de las Nieves, F.J. Application of the colloidal stability of TiO2 particles for recovery and reuse in solar photocatalysis. Water Res. 2003, 37, 3180–3188. [Google Scholar] [CrossRef]
- Patel, J.R.; Patel, M.H.; Shrivastav, P.S.; Sanyal, M. Synthesis and dyeing behavior of two remazol reactive dyes with sulfo vinyl sulfone functionality on cotton fabric and their degradation study. J. Phys. Chem. Sci. 2014, 1, 1–6. [Google Scholar]
- Jiang, P.; Ren, D.B.; He, D.; Fu, W.; Wang, J.; Gu, M. An easily sedimentable and effective TiO2 photocatalyst for removal of dyes in water. Sep. Purif. Technol. 2014, 122, 128–132. [Google Scholar] [CrossRef]
- Lee, S.Y.; Park, S.J. TiO2 photocatalyst for water treatment applications. J. Ind. Eng. Chem. 2013, 19, 1761–1769. [Google Scholar] [CrossRef]
- Patel Jigar, R.; Patel Mitesh, H.; ShrivastavPranav, S.; Sanyal, M. Synthesis of a modified reactive dye with improved dyeing performance on cotton fabric and its degradation study. Indian J. Sci. 2013, 3, 45–51. [Google Scholar]
- Malato, S.; Fernández-Ibáñez, P.; Maldonado, M.I.; Blanco, J.; Gernjak, W. Decontamination and disinfection of water by solar photocatalysis: Recent overview and trends. Catal. Today 2009, 147, 1–59. [Google Scholar] [CrossRef]
- Nahar, M.S.; Hasegawa, K.; Kagaya, S. Photocatalytic degradation of phenol by visible light-responsive iron-doped TiO2 and spontaneous sedimentation of the TiO2 particles. Chemosphere 2006, 65, 1976–1982. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.M.; Valente, J.P.S.; Jorge, S.M.A.; Padilha, P.M.; Saeki, M.J.; Castro, G.R.; Florentino, A.O. Coagulation-flocculation of TiO2 in suspension used in heterogeneous photocatalysis. Orbital Electron. J. Chem. 2013, 5, 233–241. [Google Scholar]
- Balasubramanian, G.; Dionysiou, D.D.; Suidan, M.T.; Baudin, I.; Laîné, J.M. Evaluating the activities of immobilized TiO2 powder films for the photocatalytic degradation of organic contaminants in water. Appl. Catal. B Environ. 2004, 47, 73–84. [Google Scholar] [CrossRef]
- Fan, J.W.; Liu, J.Y.; Hong, J.; Zhang, J. The synthesis of nanostructure TiO2 co-doped with N and Fe and their application for micro-polluted source water treatment. Environ. Technol. 2009, 30, 1447–1452. [Google Scholar] [CrossRef] [PubMed]
- He, R.L.; Wei, Y.; Cao, W.B. Preparation of (Fe, N)-doped TiO2 powders and their antibacterial activities under visible light irradiation. J. Nanosci. Nanotechnol. 2009, 9, 1094–1097. [Google Scholar] [CrossRef] [PubMed]
- Dumitrescu, I.; Iordache, O.; Popescu, A.; Varzaru, E.; Kim, S.; Basim, B.; Ükelge, G. The photocatalytic effects of textiles treated with TiO2 and Fe/TiO2. Ind. Text. 2015, 66, 297–305. [Google Scholar]
- Behzadnia, A.; Montazer, M.; Rashidi, A.; Rad, M.M. Sonosynthesis of nano TiO2 on wool using titanium isopropoxide or butoxide in acidic media producing multifunctional fabric. Ultrason. Sonochem. 2014, 21, 1815–1826. [Google Scholar] [CrossRef] [PubMed]
- Behzadnia, A.; Montazer, M.; Rashidi, A.; Rad, M.M. Rapid sonosynthesis of N-doped nano TiO2 on wool fabric at low temperature: Introducing self-cleaning, hydrophilicity, antibacterial/antifungal properties with low alkali solubility, yellowness and cytotoxicity. Photochem. Photobiol. 2014, 90, 1224–1233. [Google Scholar] [CrossRef] [PubMed]
- Klug, H.P.; Alexander, L.E. X-ray Diffraction Procedures for Polycrystalline and Amorphous Materials; Wiley: New York, NY, USA, 1966; p. 491. [Google Scholar]
- Diamandescu, L.; Feder, M.; Vasiliu, F.; Tanase, L.; Teodorescu, C.M.; Popescu, T.; Dumitrescu, I. Hydrothermal route to (Fe, N) codoped titania photocatalysts with increased visible light activity. Ind. Text. 2016, in press. [Google Scholar]
- International Organization for Standardization. Joint ISO/CIE Standard: Colorimetry—Part 4: CIE 1976 L*a*b* Colour Space; ISO 11664-4:2008(E)/CIE S 014-4/E:2007; ISO: Geneva, Switzerland, 2007. [Google Scholar]
- Prabhakar, P.K.; Raj, S.; Anuradha, P.R.; Sawant, S.N.; Doble, M. Biocompatibility studies on polyaniline and polyaniline–silver nanoparticle coated polyurethane composite. Colloids Surf. B Biointerfaces 2011, 86, 146–153. [Google Scholar] [CrossRef] [PubMed]
- International Organization for Standardization. Biological Evaluation of Medical Devices—Part 5: Tests for in Vitro Cytotoxicity; ISO 10993-5:2009; ISO: Geneva, Switzerland, 2009. [Google Scholar]
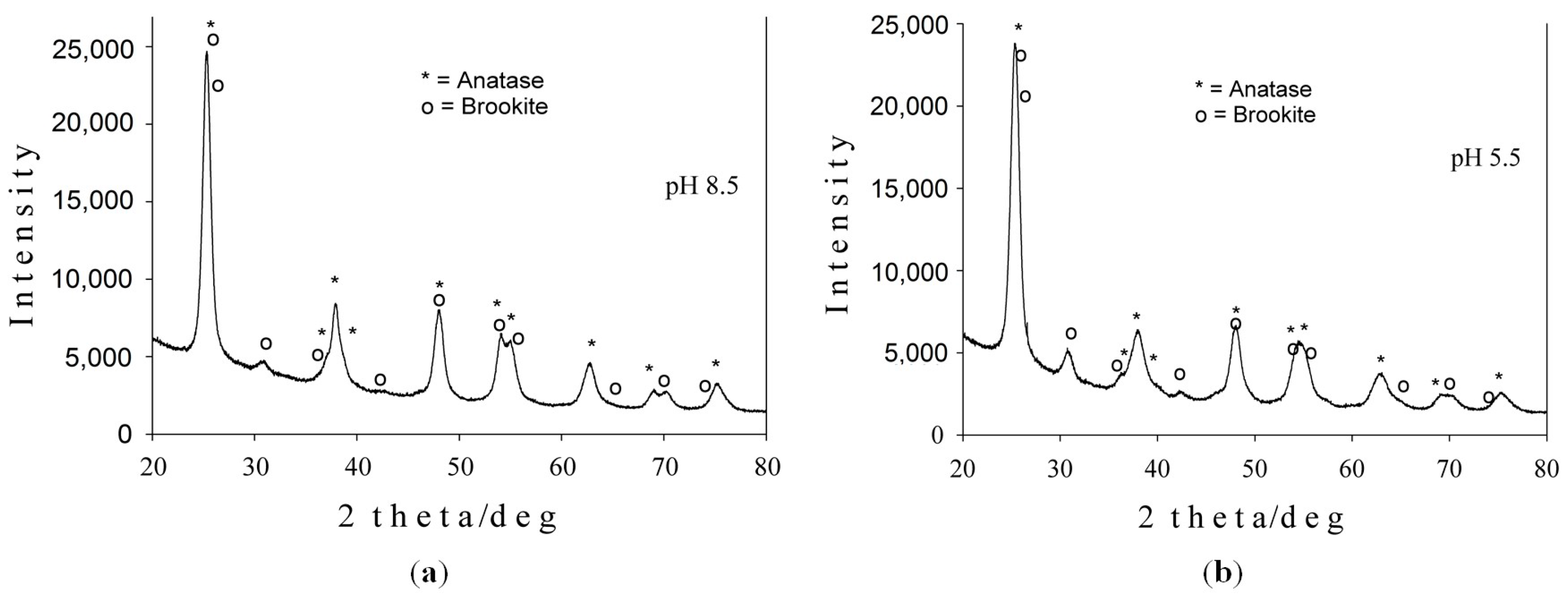
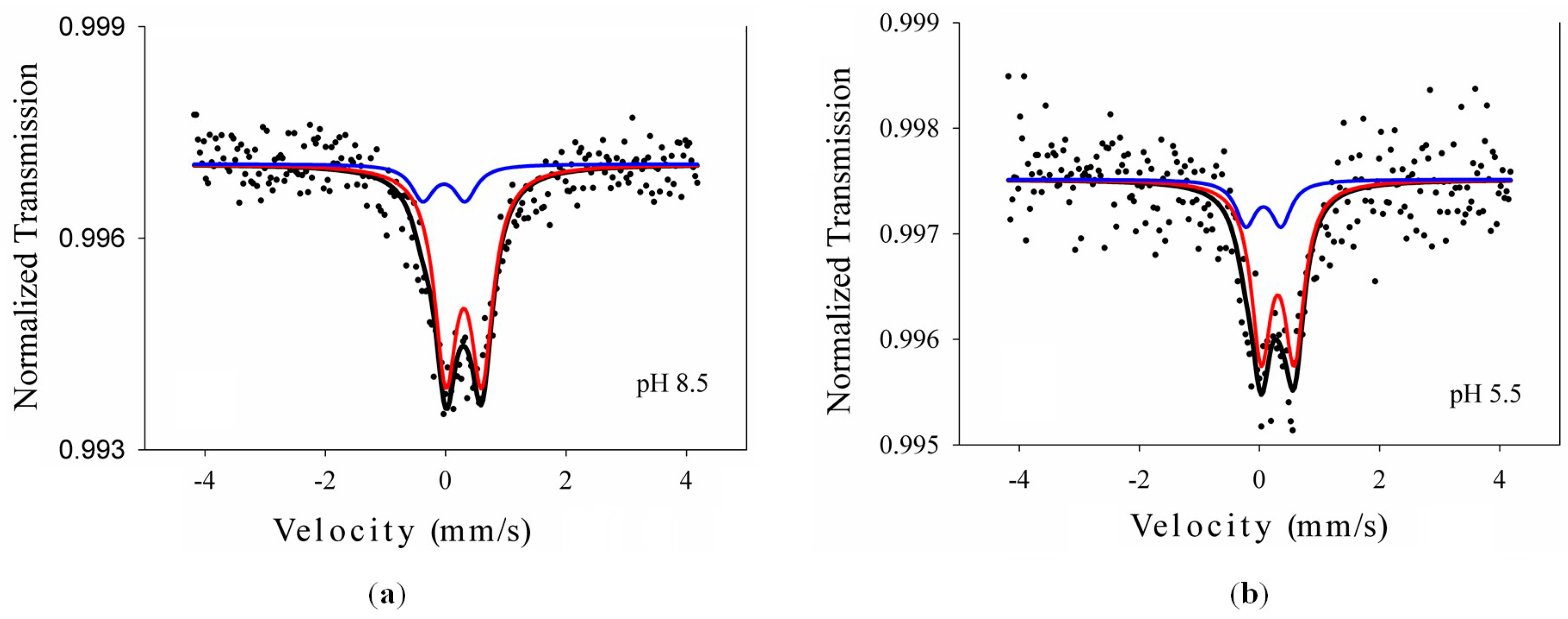


| Sample | Lattice Parameters (Å) | Crystallite Size (nm) | Phase Assignment/Abundance (wt %) | ||
|---|---|---|---|---|---|
| a | b | c | |||
| Sample 1 (pH = 8.5) | 3.7912 | - | 9.4909 | 12.3 | Anatase 85.3 |
| 9.1429 | 5.4215 | 5.2450 | 8.5 | Brookite 14.7 | |
| Sample 2 (pH = 5.5) | 3.7907 | - | 9.4743 | 10.4 | Anatase 79.4 |
| 9.1629 | 5.4437 | 5.1809 | 11.6 | Brookite 20.6 | |
| Errors | ±0.0005 | ±0.0005 | ±0.0005 | ±1.5 | ±1.4 |
| Sample | IS (mm/s) | ΔEQ (mm/s) | Γ (mm/s) | Areas (%) | Site/Phase Assignment |
|---|---|---|---|---|---|
| Sample 1 | 0.417 | 0.598 | 0.45 | 85.2 | Fe3+: Anatase |
| 0.096 | 0.703 | 0.46 | 14.8 | Fe3+: Brookite | |
| Sample 2 | 0.419 | 0.551 | 0.40 | 79.4 | Fe3+: Anatase |
| 0.182 | 0.587 | 0.41 | 20.6 | Fe3+: Brookite | |
| Errors | ±0.002 | ±0.004 | ±0.03 | ±0.4 | - |
| Element, wt % | HT1 | HT1 ITO | HT2 | HT2 ITO |
|---|---|---|---|---|
| C K | 46.19 | 45.99 | 49.20 | 68.01 |
| O K | 43.71 | 34.93 | 45.77 | 29.44 |
| Ti K | 10.09 | 19.07 | 5.04 | 2.55 |
| Total | 100 | 100 | 100 | 100 |
| Sample | L* | a* | b* | dL* | da* | db* | dE* | dC* | dH* | Grades |
|---|---|---|---|---|---|---|---|---|---|---|
| Control cotton knit stained with MB | 86.98 | −2.06 | −2.53 | 0.23 | 5.62 | 5.14 | 7.62 | −7.59 | 0.61 | 2 |
| Cotton knit ITO-MB | 82.32 | −3.83 | −7.02 | 2.46 | 1.04 | 1.23 | 2.94 | −1.58 | 0.30 | 3.50 |
| HT1-MB | 84.75 | −4.32 | −2.93 | 5.48 | 13.37 | 9.68 | 17.39 | −16.5 | −0.25 | 1 |
| HT1 ITO-MB | 90.35 | −2.01 | −0.34 | 13.31 | 14.66 | 13.27 | 23.84 | −19.48 | −3.39 | 1 |
| HT2-MB | 82.13 | −6.05 | −5.68 | 2.07 | 11.82 | 7.59 | 14.2 | −13.96 | 1.57 | 1 |
| HT2 ITO-MB | 89.68 | −3.07 | −1.47 | 5.92 | 14.92 | 10.49 | 2.2 | −18.2 | −1.2 | 1 |
| Control cotton knit stained with coffee | 80.26 | 4.96 | 19.72 | 3.46 | −0.68 | 0.45 | 3.56 | 0.26 | 0.77 | 3 |
| Cotton knit ITO-coffee | 71.95 | 6.86 | 22.17 | 1.82 | −0.72 | −2.42 | 3.12 | −2.53 | −0.03 | 3.50 |
| HT1-coffee | 78.38 | 5.2 | 21.92 | 5.06 | −0.91 | 0.6 | 5.18 | 22.18 | - | 2.5 |
| HT1 ITO-coffee | 82.25 | 4.99 | 22.61 | 4.88 | −1.33 | 0.68 | 5.1 | 0.34 | 1.46 | 2.5 |
| HT2-coffee | 81.29 | 5.25 | 23.69 | 4.05 | −1.16 | 0.6 | 4.25 | 0.3 | 1.27 | 2.5 |
| HT2 ITO-coffee | 80.98 | 5.56 | 25.26 | 2.9 | −0.67 | 1.54 | 3.35 | 1.34 | 1.01 | 3 |
| Sample | L* | a* | b* | dL* | da* | db* | dE* | dC* | dH* | Grades |
|---|---|---|---|---|---|---|---|---|---|---|
| Control cotton knit stained with MB | 87.38 | −5.06 | −2.61 | 3.2 | 5.68 | 6.58 | 9.26 | −8.44 | −2.07 | 1.5 |
| Cotton knit ITO-MB | 77.67 | −18.76 | −15.15 | −2.36 | −0.61 | −0.19 | 2.44 | 0.59 | −0.24 | 3.50 |
| HT1-MB | 81.7 | −8.01 | −3.02 | 1.7 | 7.09 | 8.03 | 10.84 | −10.15 | −3.42 | 1.5 |
| HT1 ITO-MB | −8.47 | −1.55 | 2.12 | 9.41 | 11.52 | 15.03 | 14.46 | −13.54 | −6.17 | 1 |
| HT2-MB | 82.13 | −6.05 | −5.68 | 2.07 | 11.82 | 7.59 | 14.2 | −13.96 | 1.57 | 1 |
| HT2 ITO-MB | 87.76 | −7.04 | 2.31 | 2.93 | 7.46 | 10.02 | 12.83 | −9.02 | −8.65 | 1 |
| Control cotton knit stained with coffee | 73.19 | 5.96 | 21.56 | −5.72 | 1.71 | 5.22 | 7.93 | 5.49 | −0.29 | 2 |
| Cotton knit ITO-coffee | 67.97 | 7.91 | 27.01 | 1.41 | −0.68 | −1.58 | 2.22 | −1.71 | 0.20 | 4.00 |
| HT1-coffee | 74.76 | 5.13 | 21.55 | 0.4 | −0.01 | 2.18 | 2.22 | 2.11 | 0.55 | 4 |
| HT1 ITO-coffee | 75.47 | 7.31 | 24.87 | 3.06 | −1.12 | −0.30 | 3.27 | −0.63 | 0.98 | 3 |
| HT2-coffee | 76.05 | 5.28 | 24.39 | 1.97 | −0.45 | 3.18 | 3.77 | 2.99 | 1.18 | 3 |
| HT2 ITO-coffee | 74.83 | 6.01 | 24.71 | 2.07 | −0.77 | 2.26 | 2.97 | 1.98 | 1.34 | 3.5 |
| Sample | L* | a* | b* | dL* | da* | db* | dE* | dC* | dH* | Grades |
|---|---|---|---|---|---|---|---|---|---|---|
| Control cotton knit stained with MB | 85.7 | −3.41 | −4.67 | 4.85 | 5.59 | 4.58 | 8.71 | −7.13 | 1.21 | 1.5 |
| Cotton knit ITO-MB | 78.79 | −10.54 | −11.62 | 0.08 | 9.36 | 4.93 | 10.58 | −10.20 | 2.83 | 1.50 |
| HT1-MB | 88.5 | −1.68 | −2.87 | 5.93 | 11.48 | 7.19 | 14.79 | −13.24 | 2.86 | 1 |
| HT2-MB | 85.36 | −3.02 | −2.4 | 1.58 | 7.77 | 6.53 | 10.27 | −10.15 | −0.14 | 1.5 |
| Control cotton knit stained with coffee | 69.48 | 8.07 | 26.77 | 0.23 | −1.08 | −3.57 | −3.73 | 0.01 | 1.01 | 3.5 |
| Cotton knit ITO-coffee | 68.44 | 7.21 | 24.32 | −1.43 | −0.64 | −3.35 | 3.70 | −3.40 | −0.32 | 3.00 |
| HT1-coffee | 78.17 | 4.64 | 21.04 | 2.54 | −0.59 | 1.25 | 2.9 | 1.08 | 0.87 | 3.5 |
| HT2-coffee | 80.26 | 3.71 | 17.58 | 5.8 | −2.16 | −3.38 | 7.05 | −3.8 | 1.29 | 2 |
| Microbial Strain | 15 min | 24 h | |
|---|---|---|---|
| Gram negative bacilli | P. aeruginosa | HT1, HT2, HT1 ITO | - |
| E. coli | HT1, HT2, HT1 ITO, HT2 ITO | HT1, HT2 ITO | |
| Gram positive bacilli | B. subtilis | HT1, HT1 ITO | - |
| Gram positive cocci | S. aureus | HT1, HT2, HT2 ITO | HT2, HT1 ITO, HT2 ITO |
| E. faecium | HT1, HT2, HT1 ITO | - | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stan, M.S.; Nica, I.C.; Dinischiotu, A.; Varzaru, E.; Iordache, O.G.; Dumitrescu, I.; Popa, M.; Chifiriuc, M.C.; Pircalabioru, G.G.; Lazar, V.; et al. Photocatalytic, Antimicrobial and Biocompatibility Features of Cotton Knit Coated with Fe-N-Doped Titanium Dioxide Nanoparticles. Materials 2016, 9, 789. https://doi.org/10.3390/ma9090789
Stan MS, Nica IC, Dinischiotu A, Varzaru E, Iordache OG, Dumitrescu I, Popa M, Chifiriuc MC, Pircalabioru GG, Lazar V, et al. Photocatalytic, Antimicrobial and Biocompatibility Features of Cotton Knit Coated with Fe-N-Doped Titanium Dioxide Nanoparticles. Materials. 2016; 9(9):789. https://doi.org/10.3390/ma9090789
Chicago/Turabian StyleStan, Miruna Silvia, Ionela Cristina Nica, Anca Dinischiotu, Elena Varzaru, Ovidiu George Iordache, Iuliana Dumitrescu, Marcela Popa, Mariana Carmen Chifiriuc, Gratiela G. Pircalabioru, Veronica Lazar, and et al. 2016. "Photocatalytic, Antimicrobial and Biocompatibility Features of Cotton Knit Coated with Fe-N-Doped Titanium Dioxide Nanoparticles" Materials 9, no. 9: 789. https://doi.org/10.3390/ma9090789
APA StyleStan, M. S., Nica, I. C., Dinischiotu, A., Varzaru, E., Iordache, O. G., Dumitrescu, I., Popa, M., Chifiriuc, M. C., Pircalabioru, G. G., Lazar, V., Bezirtzoglou, E., Feder, M., & Diamandescu, L. (2016). Photocatalytic, Antimicrobial and Biocompatibility Features of Cotton Knit Coated with Fe-N-Doped Titanium Dioxide Nanoparticles. Materials, 9(9), 789. https://doi.org/10.3390/ma9090789












