Novel Osteointegrative Sr-Substituted Apatitic Cements Enriched with Alginate
Abstract
:1. Introduction
2. Results
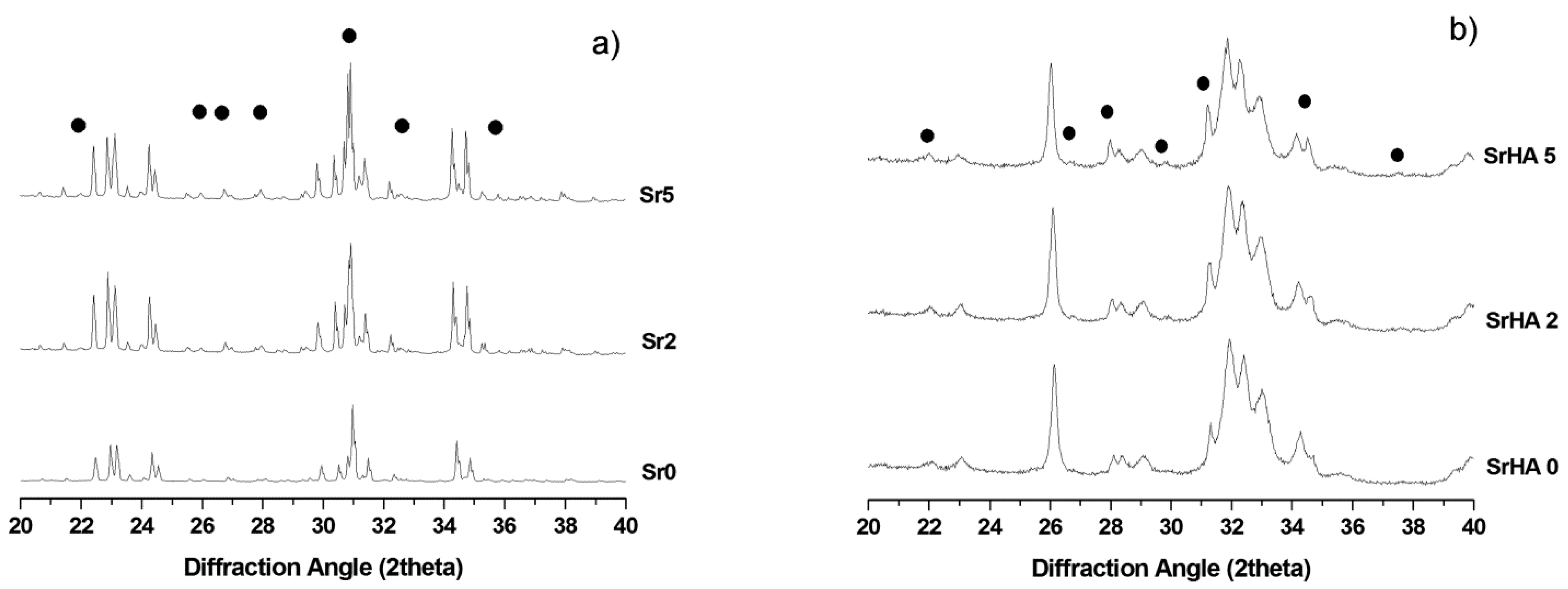
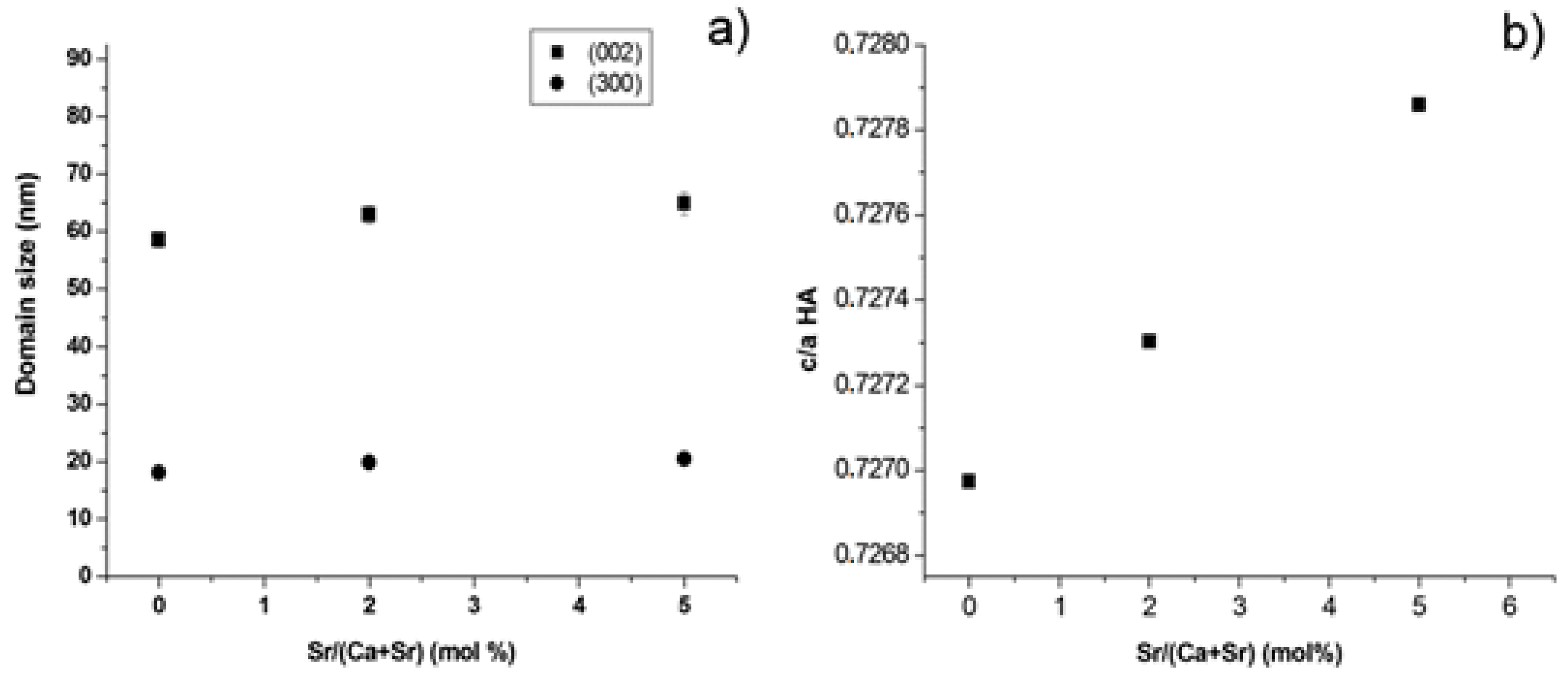
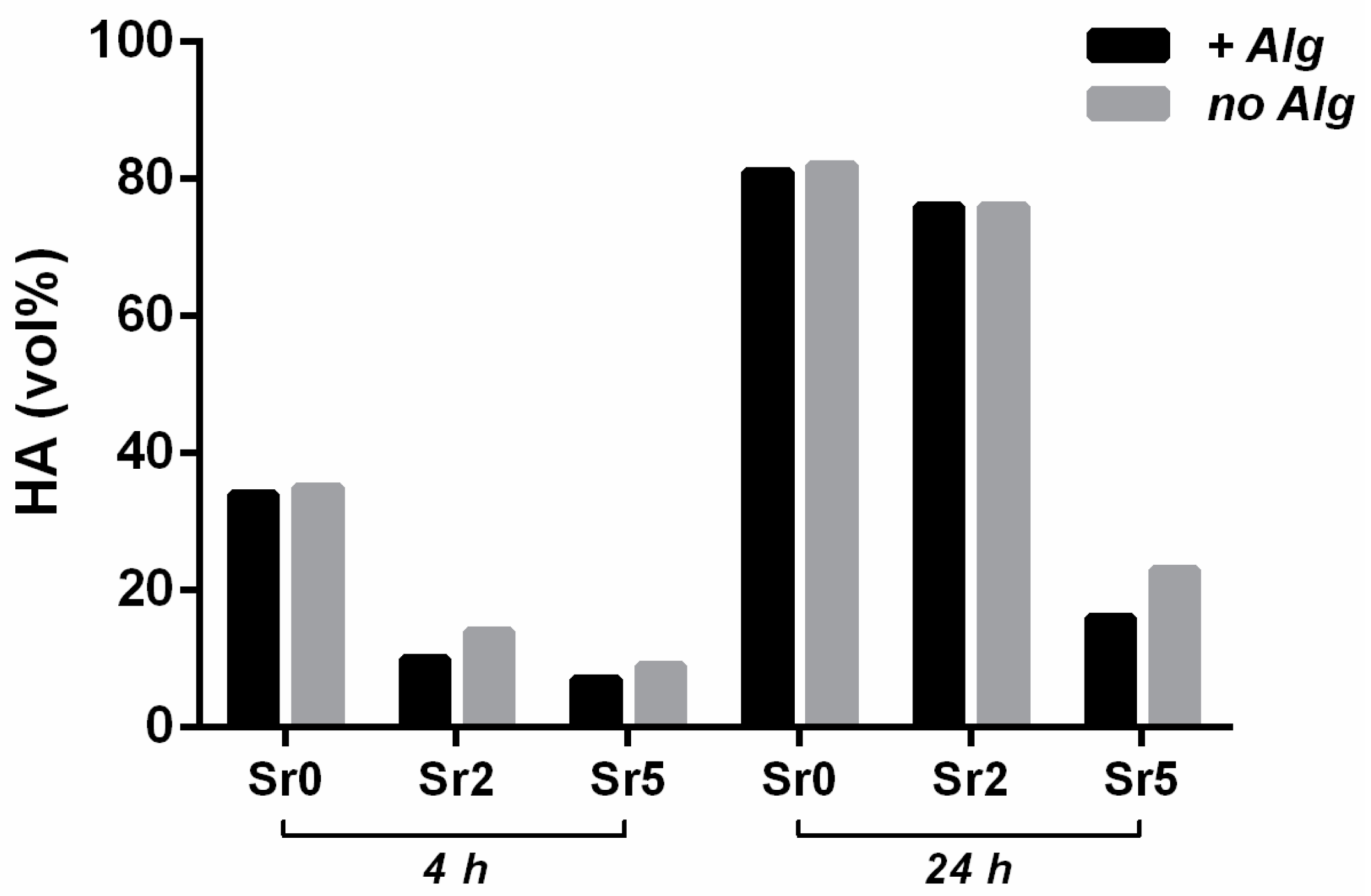
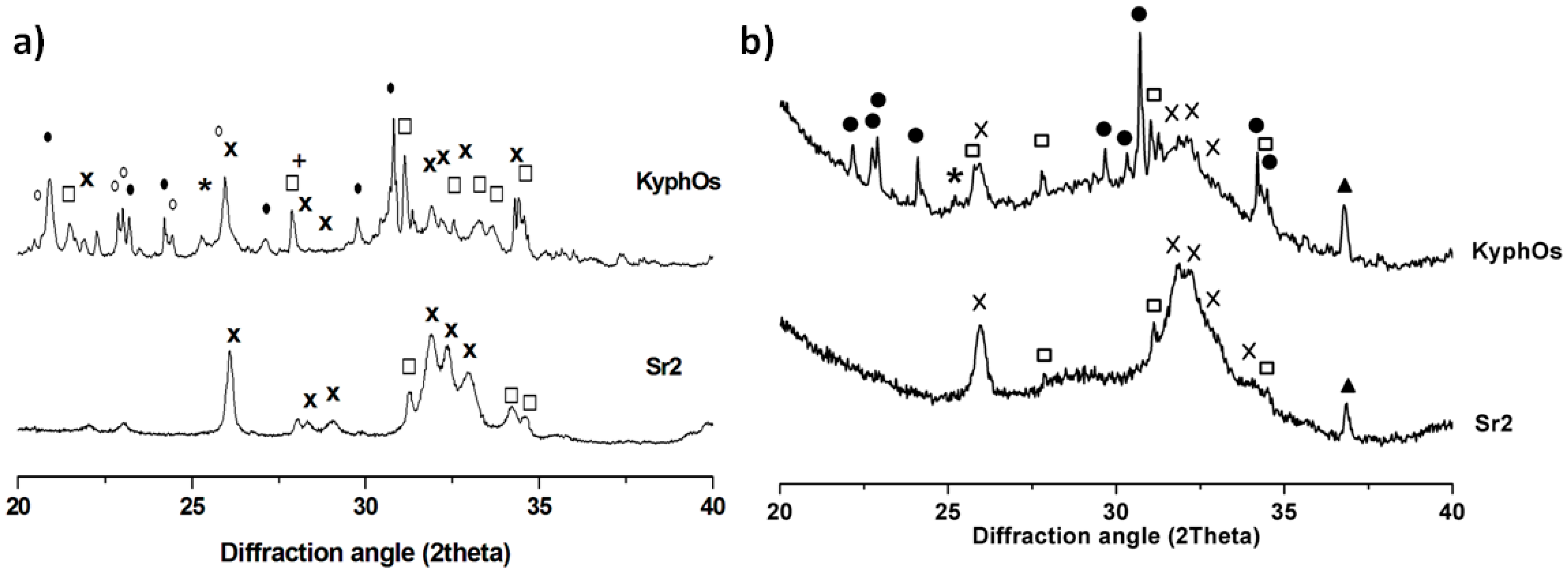
2.1. Physicochemical Properties and Setting Behavior
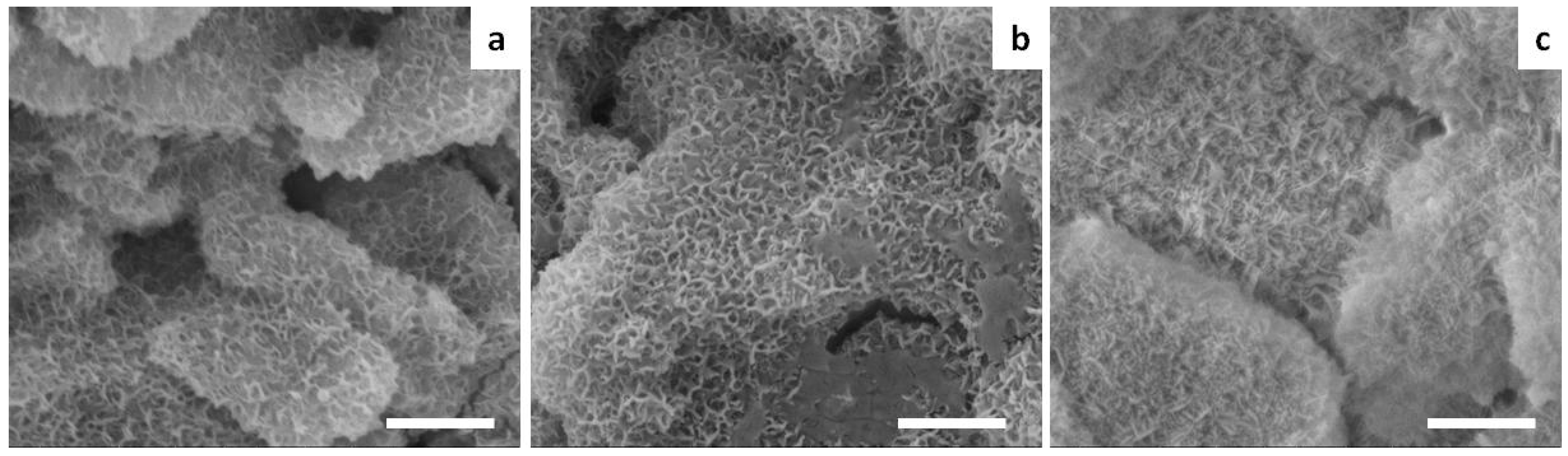
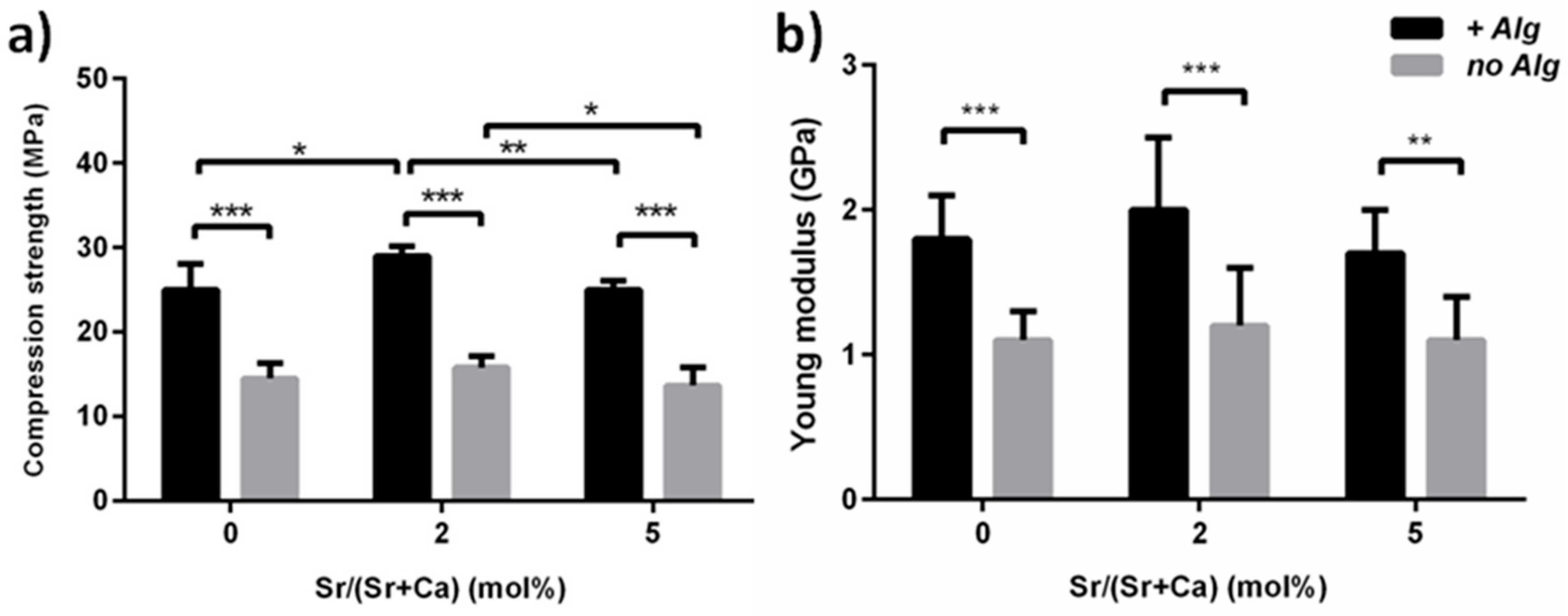
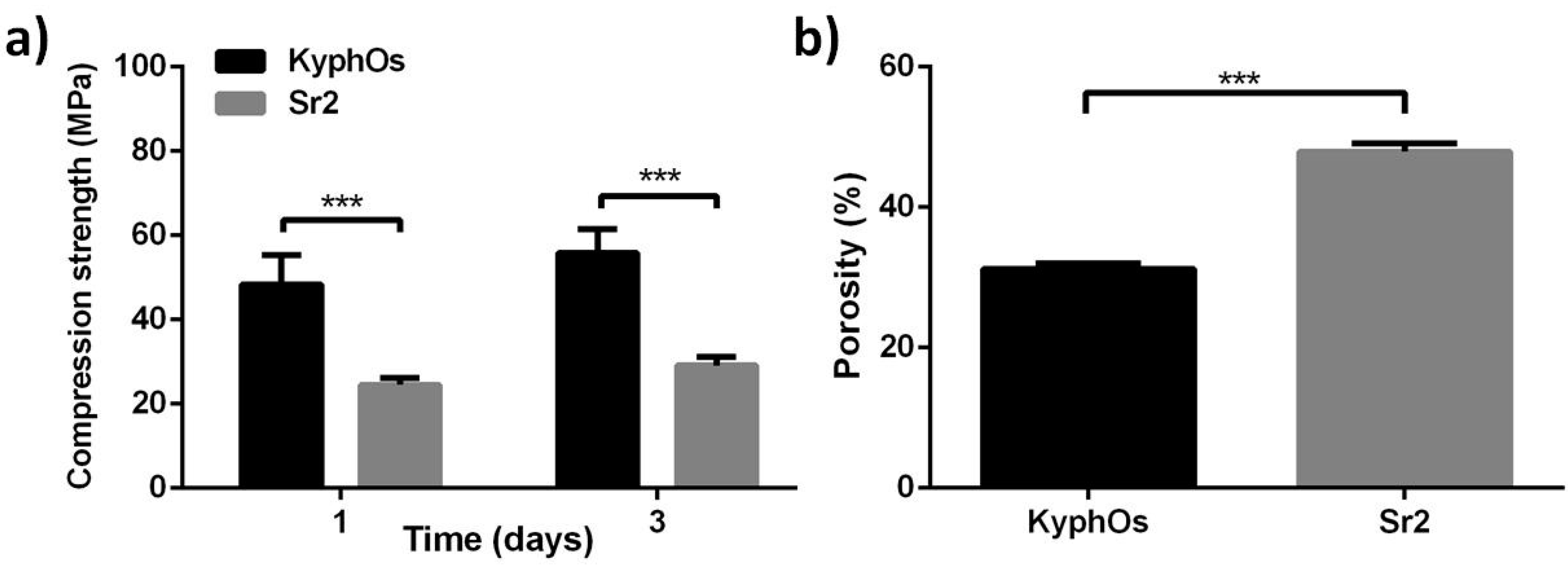
2.2. Morphological and Mechanical Aspects
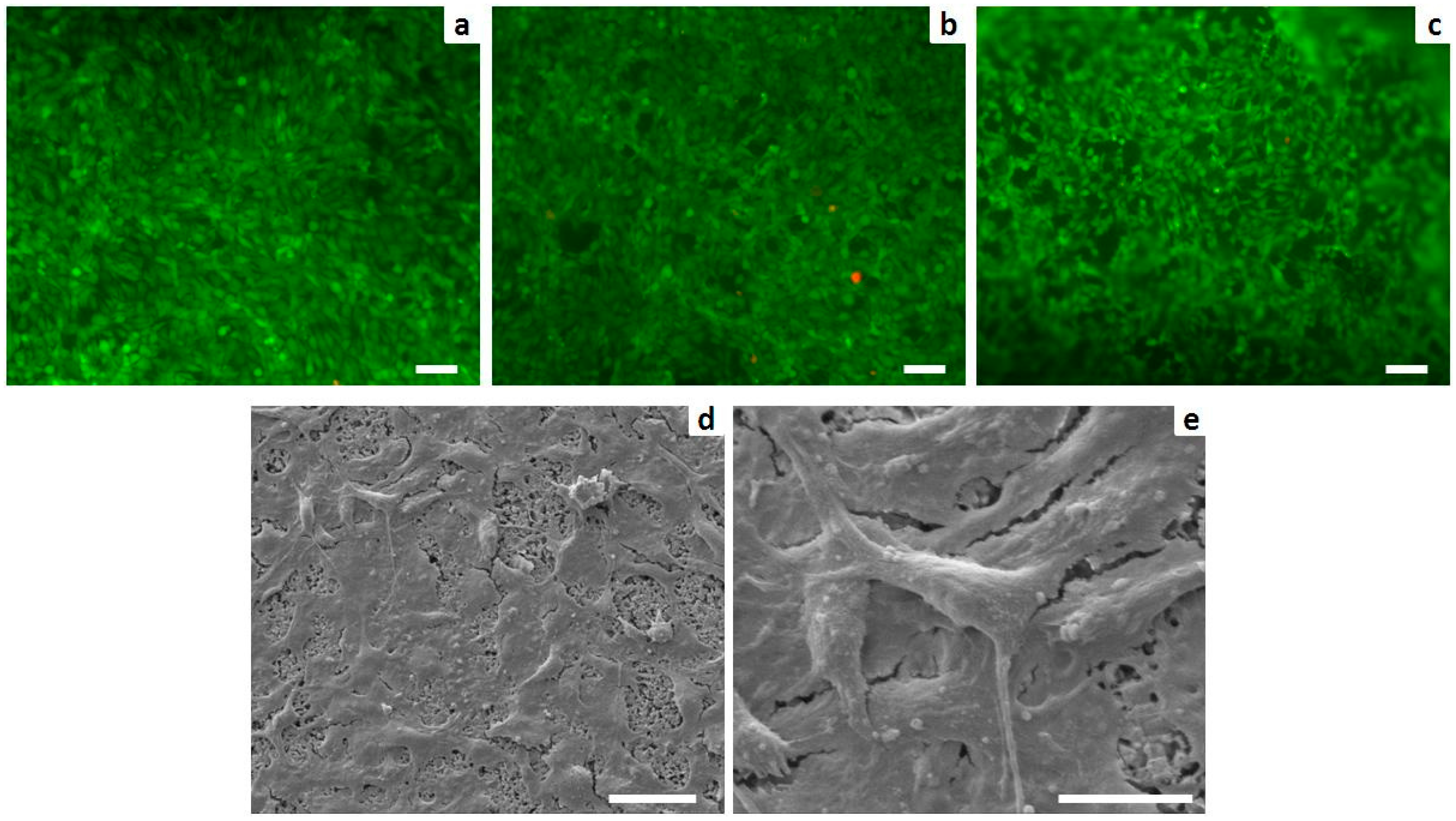
2.3. Biological Evaluation by in Vitro Cell Tests
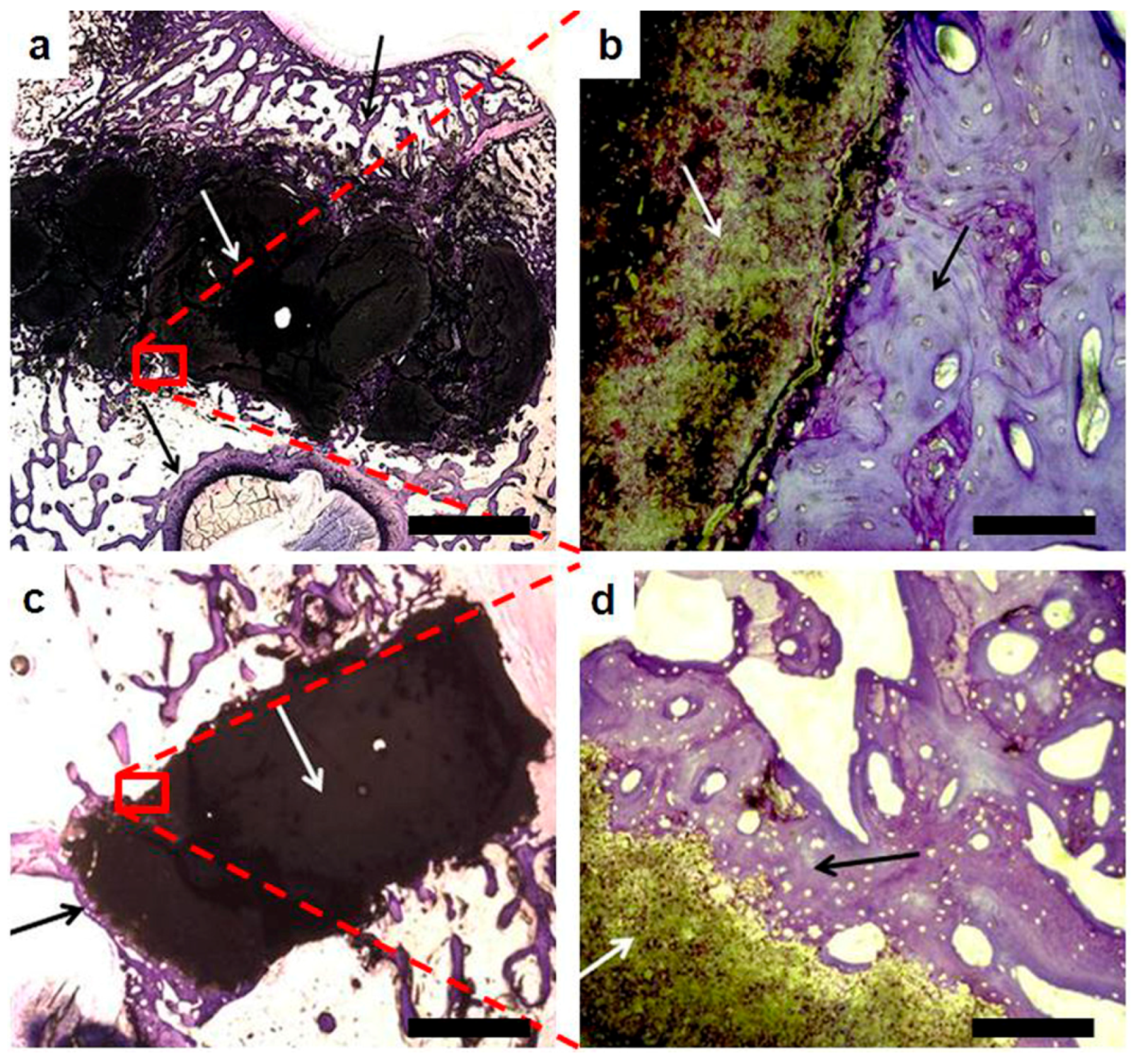
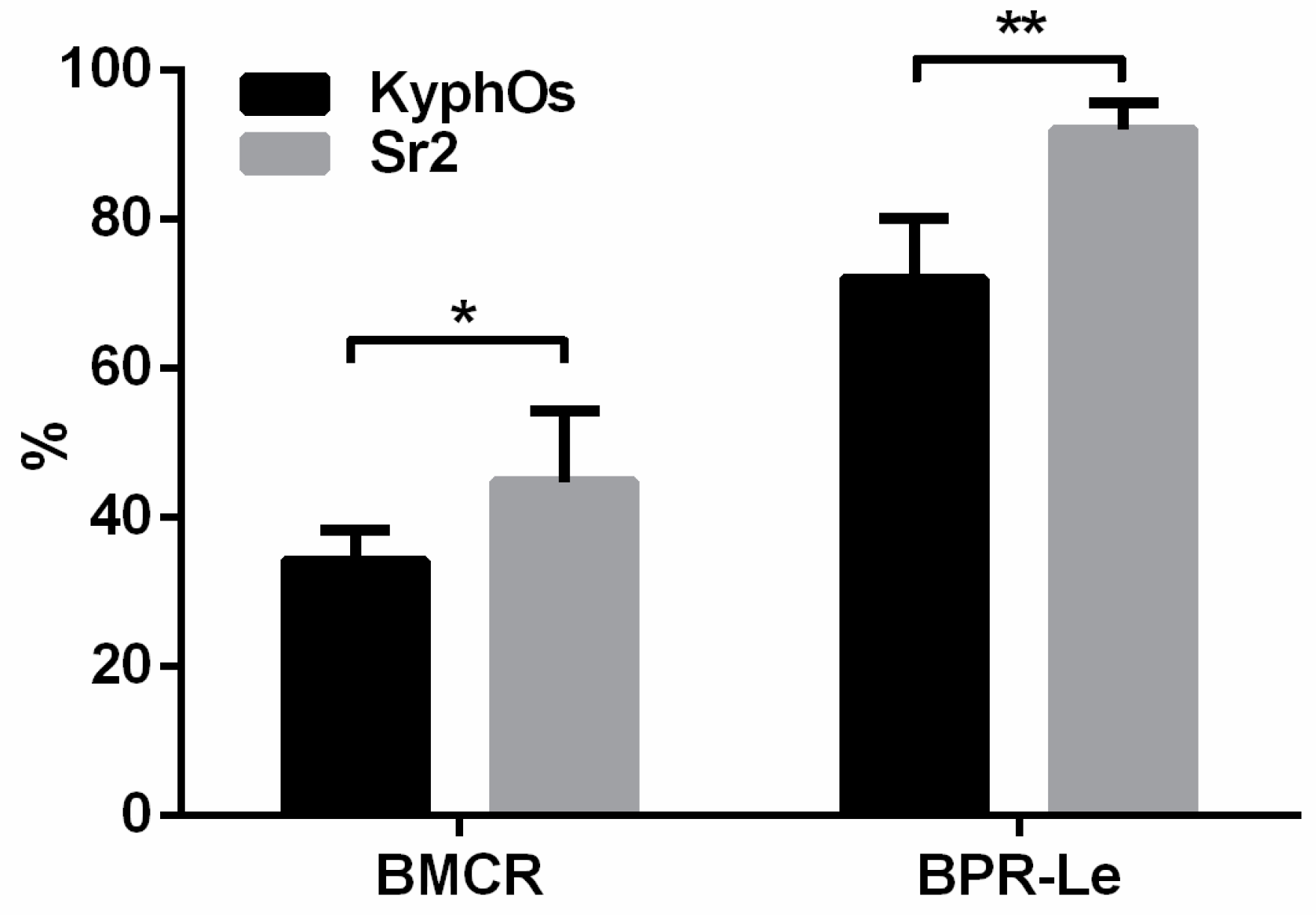
2.4. In Vivo Pilot Experiment and Histological Analysis
3. Discussion
4. Experimental Section
4.1. Preparation of the Inorganic Precursors
4.2. Characterization of the Pastes and Hardened Cements
4.2.1. Composition and Crystallographics
4.2.2. Injectability and Setting Times
4.2.3. Morphological Characterization
4.2.4. Mechanical Evaluation
4.2.5. Cell Culture
4.2.6. Cell Viability Assay
4.2.7. Cell Morphology Evaluation
4.2.8. In Vivo Pilot Experiment and Histological Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bohner, M. Calcium orthophosphates in medicine: From ceramics to calcium phosphate cements. Injury 2000, 31, 37–47. [Google Scholar] [CrossRef]
- Poitout, D.G. Biomechanics and Biomaterials in Orthopedics, 1st ed.; Springer: London, UK, 2004; p. 654. [Google Scholar]
- Saint-Jean, S.J.; Camire, C.L.; Nevsten, P.; Hansen, S.; Ginebra, M.P. Study of the reactivity and in vitro bioactivity of Sr-substituted alpha-TCP cements. J. Mater. Sci. Mater. Med. 2005, 16, 993–1001. [Google Scholar] [CrossRef] [PubMed]
- Watts, N.B.; Harris, S.T.; Genant, H.K. Treatment of painful osteoporotic vertebral fractures with percutaneous vertebroplasty or kyphoplasty. Osteoporos. Int. 2001, 12, 429–437. [Google Scholar] [CrossRef] [PubMed]
- Xue, W.; Dahlquist, K.; Banerjee, A.; Bandyopadhyay, A.; Bose, S. Synthesis and characterization of tricalcium phosphate with Zn and Mg based dopants. J. Mater. Sci. Mater. Med. 2008, 19, 2669–2677. [Google Scholar] [CrossRef] [PubMed]
- Kona, S.; Wadajkar, A.S.; Nguyen, K.T. Tissue engineering applications of injectable biomaterials. In Injectable Biomaterials; Vernon, B., Ed.; Woodhead Publishing: Cambridge, UK, 2011; pp. 142–182. [Google Scholar]
- Stankewich, C.J.; Swiontkowski, M.F.; Tencer, A.F.; Yetkinler, D.N.; Poser, R.D. Augmentation of femoral neck fracture fixation with an injectable calcium-phosphate bone mineral cement. J. Orthopaed. Res. 1996, 14, 786–793. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M. Design of ceramic-based cements and putties for bone graft substitution. Eur. Cells Mater. 2010, 20, 3–10. [Google Scholar]
- Canal, C.; Ginebra, M.P. Fibre-reinforced calcium phosphate cements: A review. J. Mech. Behav. Biomed. 2011, 4, 1658–1671. [Google Scholar] [CrossRef] [PubMed]
- Kruger, R.; Groll, J. Fiber reinforced calcium phosphate cements—On the way to degradable load bearing bone substitutes? Biomaterials 2012, 33, 5887–5900. [Google Scholar] [CrossRef] [PubMed]
- Becker, S.; Dabirrahmani, D.; Hogg, M.; Appleyard, R.; Baroud, G.; Gillies, M. Disadvantages of balloon kyphoplasty with PMMA—A clinical and biomechanical statement. J. Miner. Stoffwechs. 2011, 18, 9–12. [Google Scholar]
- Boner, V.; Kuhn, P.; Mendel, T.; Gisep, A. Temperature evaluation during PMMA screw augmentation in osteoporotic bone—An in vitro study about the risk of thermal necrosis in human femoral heads. J. Biomed. Mater. Res. B Appl. Biomater. 2009, 90, 842–848. [Google Scholar] [CrossRef] [PubMed]
- Deramond, H.; Wright, N.T.; Belkoff, S.M. Temperature elevation caused by bone cement polymerization during vertebroplasty. Bone 1999, 25, S17–S21. [Google Scholar] [CrossRef]
- Kolb, J.P.; Kueny, R.A.; Puschel, K.; Boger, A.; Rueger, J.M.; Morlock, M.M.; Huber, G.; Lehmann, W. Does the cement stiffness affect fatigue fracture strength of vertebrae after cement augmentation in osteoporotic patients? Eur. Spine J. 2013, 22, 1650–1656. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.T.; Liu, W.Z.; Schnitzler, V.; Tancret, F.; Bouler, J.M. Calcium phosphate cements for bone substitution: Chemistry, handling and mechanical properties. Acta Biomater. 2014, 10, 1035–1049. [Google Scholar] [CrossRef] [PubMed]
- Ormsby, R.; McNally, T.; Mitchell, C.; Dunne, N. Incorporation of multiwalled carbon nanotubes to acrylic based bone cements: Effects on mechanical and thermal properties. J. Mech. Behav. Biomed. Mater. 2010, 3, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Mavropoulos, E.; Rossi, A.M.; da Rocha, N.C.C.; Soares, G.A.; Moreira, J.C.; Moure, G.T. Dissolution of calcium-deficient hydroxyapatite synthesized at different conditions. Mater. Charact. 2003, 50, 203–207. [Google Scholar] [CrossRef]
- Landi, E.; Logroscino, G.; Proietti, L.; Tampieri, A.; Sandri, M.; Sprio, S. Biomimetic Mg-substituted hydroxyapatite: From synthesis to in vivo behaviour. J. Mater. Sci. Mater. Med. 2008, 19, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Landi, E.; Tampieri, A.; Celotti, G.; Sprio, S.; Sandri, M.; Logroscino, G. Sr-substituted hydroxyapatites for osteoporotic bone replacement. Acta Biomater. 2007, 3, 961–969. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.; Gelinsky, M. Strontium modified calcium phosphate cements—Approaches towards targeted stimulation of bone turnover. J. Mater. Chem. B 2015, 3, 4626–4640. [Google Scholar] [CrossRef]
- Sprio, S.; Tampieri, A.; Landi, E.; Sandri, M.; Martorana, S.; Celotti, G.; Logroscino, G. Physico-chemical properties and solubility behaviour of multi-substituted hydroxyapatite powders containing silicon. Mater. Sci. Eng. C 2008, 28, 179–187. [Google Scholar] [CrossRef]
- Dahl, S.G.; Allain, P.; Marie, P.J.; Mauras, Y.; Boivin, G.; Ammann, P.; Tsouderos, Y.; Delmas, P.D.; Christiansen, C. Incorporation and distribution of strontium in bone. Bone 2001, 28, 446–453. [Google Scholar] [CrossRef]
- Marie, P.J.; Ammann, P.; Boivin, G.; Rey, C. Mechanisms of action and therapeutic potential of strontium in bone. Calcif. Tissue Int. 2001, 69, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Grynpas, M.D.; Hamilton, E.; Cheung, R.; Tsouderos, Y.; Deloffre, P.; Hott, M.; Marie, P.J. Strontium increases vertebral bone volume in rats at a low dose that does not induce detectable mineralization defect. Bone 1996, 18, 253–259. [Google Scholar] [CrossRef]
- Andersen, O.Z.; Offermanns, V.; Sillassen, M.; Almtoft, K.P.; Andersen, I.H.; Sorensen, S.; Jeppesen, C.S.; Kraft, D.C.E.; Bottiger, J.; Rasse, M.; et al. Accelerated bone ingrowth by local delivery of strontium from surface functionalized titanium implants. Biomaterials 2013, 34, 5883–5890. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.F.; Li, Q.; Zhu, S.S.; Luo, E.; Li, J.H.; Feng, G.; Liao, Y.M.; Hu, J. The effect of strontium-substituted hydroxyapatite coating on implant fixation in ovariectomized rats. Biomaterials 2010, 31, 9006–9014. [Google Scholar] [CrossRef] [PubMed]
- Alkhraisat, M.H.; Moseke, C.; Blanco, L.; Barralet, J.E.; Lopez-Carbacos, E.; Gbureck, U. Strontium modified biocements with zero order release kinetics. Biomaterials 2008, 29, 4691–4697. [Google Scholar] [CrossRef] [PubMed]
- Baier, M.; Staudt, P.; Klein, R.; Sommer, U.; Wenz, R.; Grafe, I.; Meeder, P.J.; Nawroth, P.P.; Kasperk, C. Strontium enhances osseointegration of calcium phosphate cement: A histomorphometric pilot study in ovariectomized rats. J. Orthop. Surg. Res. 2013, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, M.; Henss, A.; Rohnke, M.; Gelinsky, M. A novel and easy-to-prepare strontium(II) modified calcium phosphate bone cement with enhanced mechanical properties. Acta Biomater. 2013, 9, 7536–7544. [Google Scholar] [CrossRef] [PubMed]
- Thormann, U.; Ray, S.; Sommer, U.; ElKhassawna, T.; Rehling, T.; Hundgeburth, M.; Henss, A.; Rohnke, M.; Janek, J.; Lips, K.S.; et al. Bone formation induced by strontium modified calcium phosphate cement in critical-size metaphyseal fracture defects in ovariectomized rats. Biomaterials 2013, 34, 8589–8598. [Google Scholar] [CrossRef] [PubMed]
- Khairoun, I.; Driessens, F.C.M.; Boltong, M.G.; Planell, J.A.; Wenz, R. Addition of cohesion promoters to calcium phosphate cements. Biomaterials 1999, 20, 393–398. [Google Scholar] [CrossRef]
- Ginebra, M.P.; Driessens, F.C.; Planell, J.A. Effect of the particle size on the micro and nanostructural features of a calcium phosphate cement: A kinetic analysis. Biomaterials 2004, 25, 3453–3462. [Google Scholar] [CrossRef] [PubMed]
- Montufar, E.B.; Maazouz, Y.; Ginebra, M.P. Relevance of the setting reaction to the injectability of tricalcium phosphate pastes. Acta Biomater. 2013, 9, 6188–6198. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.S.; Shao, H.F.; Chen, F.Y.; Zheng, H.Y. Effects of the granularity of raw materials on the hydration and hardening process of calcium phosphate cement. Biomaterials 2003, 24, 4103–4113. [Google Scholar] [CrossRef]
- Kuang, G.M.; Yau, W.P.; Lam, W.M.; Wu, J.; Chiu, K.Y.; Lu, W.W.; Pan, H.B. An effective approach by a chelate reaction in optimizing the setting process of strontium-incorporated calcium phosphate bone cement. J. Biomed. Mater. Res. B 2012, 100, 778–787. [Google Scholar] [CrossRef] [PubMed]
- Jack, V.; Buchanan, F.J.; Dunne, N.I. Particle attrition of alpha-tricalcium phosphate: Effect on mechanical, handling, and injectability properties of calcium phosphate cements. Proc. Inst. Mech. Eng. H 2008, 222, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.S.; Park, J.H.; Won, J.E.; Shin, U.S.; Kim, H.W. Alginate combined calcium phosphate cements: Mechanical properties and in vitro rat bone marrow stromal cell responses. J. Mater. Sci. Mater. Med. 2011, 22, 1257–1268. [Google Scholar] [CrossRef] [PubMed]
- Pina, S.; Torres, P.M.; Goetz-Neunhoeffer, F.; Neubauer, J.; Ferreira, J.M. Newly developed Sr-substituted alpha-TCP bone cements. Acta Biomater. 2010, 6, 928–935. [Google Scholar] [CrossRef] [PubMed]
- Bigi, A.; Foresti, E.; Gandolfi, M.; Gazzano, M.; Roveri, N. Isomorphous substitutions in β-tricalcium phosphate: The different effects of zinc and strontium. J. Inorg. Biochem. 1997, 66, 259–265. [Google Scholar] [CrossRef]
- Bigi, A.; Boanini, E.; Capuccini, C.; Gazzano, M. Strontium-substituted hydroxyapatite nanocrystals. Inorg. Chim. Acta 2007, 360, 1009–1016. [Google Scholar] [CrossRef]
- Donazzon, B.; Dechambre, G.; Lacout, J.L. Calcium-strontium hydroxyapatite: Hydrothermal preparation. Ann. Chim. Sci. Mater. 1998, 23, 53–56. [Google Scholar] [CrossRef]
- Bohner, M. Reactivity of calcium phosphate cements. J. Mater. Chem. 2007, 17, 3980–3986. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Calcium orthophosphate cements for biomedical application. J. Mater. Sci. 2008, 43, 3028–3057. [Google Scholar] [CrossRef]
- Perez, R.A.; Kim, H.W.; Ginebra, M.P. Polymeric additives to enhance the functional properties of calcium phosphate cements. J. Tissue Eng. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Boanini, E.; Panzavolta, S.; Rubini, K.; Gandolfi, M.; Bigi, A. Effect of strontium and gelatin on the reactivity of alpha-tricalcium phosphate. Acta Biomater. 2010, 6, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, L.A.; de Oliveria, L.C.; Rigo, E.C.; Carrodeguas, R.G.; Boschi, A.O.; de Arruda, A.C. Influence of polymeric additives on the mechanical properties of alpha-tricalcium phosphate cement. Bone 1999, 25, s99–s102. [Google Scholar] [CrossRef]
- Liu, W.Z.; Zhang, J.T.; Weiss, P.; Tancret, F.; Bouler, J.M. The influence of different cellulose ethers on both the handling and mechanical properties of calcium phosphate cements for bone substitution. Acta Biomater. 2013, 9, 5740–5750. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.G.; Xu, K.W.; Zhao, X.Y.; Han, Y. Development of a strontium-containing hydroxyapatite bone cement. Biomaterials 2005, 26, 4073–4083. [Google Scholar] [CrossRef] [PubMed]
- Oda, M.; Takeuchi, A.; Lin, X.; Matsuya, S.; Ishikawa, K. Effects of liquid phase on basic properties of alpha-tricalcium phosphate-based apatite cement. Dent. Mater. J. 2008, 27, 672–677. [Google Scholar] [CrossRef] [PubMed]
- TenHuisen, K.S.; Brown, P.W. Formation of calcium-deficient hydroxyapatite from alpha-tricalcium phosphate. Biomaterials 1998, 19, 2209–2217. [Google Scholar] [CrossRef]
- Sprio, S.; Fricia, M.; Maddalena, G.F.; Nataloni, A.; Tampieri, A. Osteointegration in cranial bone reconstruction: A goal to achieve. J. Appl. Biomater. Funct. Mater. 2016. [Google Scholar] [CrossRef] [PubMed]
- Stefini, R.; Esposito, G.; Zanotti, B.; Iaccarino, C.; Fontanella, M.M.; Servadei, F. Use of “custom made” porous hydroxyapatite implants for cranioplasty: Postoperative analysis of complications in 1549 patients. Surg. Neurol. Int. 2013, 4, 12. [Google Scholar] [CrossRef] [PubMed]
- Stefini, R.; Zanotti, B.; Nataloni, A.; Martinetti, R.; Scafuto, M.; Colasurdo, M.; Tampieri, A. The efficacy of custom-made porous hydroxyapatite prostheses for cranioplasty: Evaluation of postmarketing data on 2697 patients. J. Appl. Biomater. Funct. Mater. 2015, 13, 5000211. [Google Scholar] [CrossRef] [PubMed]
- Mathew, M.; Schroeder, L.W.; Dickens, B.; Brown, W.E. Crystal-structure of Alpha-Ca3(Po4)2. Acta Crystallogr. Sec. B Struct. Sci. 1977, 33, 1325–1333. [Google Scholar] [CrossRef]
- Yashima, M.; Sakai, A.; Kamiyama, T.; Hoshikawa, A. Crystal structure analysis of beta-tricalcium phosphate Ca-3(PO4)(2) by neutron powder diffraction. J. Solid State Chem. 2003, 175, 272–277. [Google Scholar] [CrossRef]
- Rodriguez-Lorenzo, L.M.; Hart, J.N.; Gross, K.A. Structural and chemical analysis of well-crystallized hydroxyfluorapatites. J. Phys. Chem. B 2003, 107, 8316–8320. [Google Scholar] [CrossRef]
- Oyane, A.; Kim, H.-M.; Furuya, T.; Kokubo, T.; Miyazaki, T.; Nakamura, T. Preparation and assessment of revised simulated body fluids. J. Biomed. Mater. Res. A 2003, 65, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, N.G.; Dedoussis, G.V.Z.; Spanakos, G.; Gritzapis, A.D.; Baxevanis, C.N.; Papamichail, M. An improved fluorescence assay for the determination of lymphocyte-mediated cytotoxicity using flow-cytometry. J. Immunol. Methods 1994, 177, 101–111. [Google Scholar] [CrossRef]
- Barkarmo, S.; Wennerberg, A.; Hoffman, M.; Kjellin, P.; Breding, K.; Handa, P.; Stenport, V. Nano-hydroxyapatite-coated PEEK implants: A pilot study in rabbit bone. J. Biomed. Mater. Res. A 2013, 101, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Landi, E.; Tampieri, A.; Celotti, G.; Belmonte, M.M.; Logroscino, G. Synthetic biomimetic nanostructured hydroxyapatite. Bioceramics 2005, 284, 949–952. [Google Scholar] [CrossRef]
- Dempster, D.W.; Compston, J.E.; Drezner, M.K.; Glorieux, F.H.; Kanis, J.A.; Malluche, H.; Meunier, P.J.; Ott, S.M.; Recker, R.R.; Parfitt, A.M. Standardized nomenclature, symbols, and units for bone histomorphometry: A 2012 update of the report of the ASBMR Histomorphometry Nomenclature Committee. J. Bone Miner. Res. 2013, 28, 2–17. [Google Scholar] [CrossRef] [PubMed]


| Nominal Sr Concentration | Actual Composition | ||
|---|---|---|---|
| Sr/(Ca + Sr) (mol %) | Ca/P (mol) | (Sr + Ca)/P (mol) | Sr/(Ca + Sr) (mol %) |
| 0 | 1.51 | 1.51 | 0 |
| 2 | 1.44 | 1.46 | 1.8 |
| 5 | 1.40 | 1.46 | 4.9 |
| αTCP | y0 | m | R2 |
| a | 12,849 Å | 0.0074 | 0.988 |
| b | 27,340 Å | 0.0151 | 0.988 |
| c | 15,217 Å | 0.0029 | 0.987 |
| Beta angle | 126.30° | 0.01616 | 0.963 |
| Cell volume | 4308 Å3 | 5.46 | 0.989 |
| Cell volume [38] | 4318 Å3 | 4.86 | – |
| βTCP | y0 | m | R2 |
| a | 10,414 Å | 0.0047 | 0.93 |
| c | 37,341 Å | 0.01281 | 0.993 |
| Cell volume | 3501 Å3 | 4.45 | 0.967 |
| Cell volume [39] | 3507 Å3 | 4.90 | – |
| HA | y0 | m | R2 |
| a | 9.45717 Å | 0.00274 | 0.997 |
| c | 6.87681 Å | 0.00324 | 0.998 |
| Cell volume | 532.8 Å3 | 0.56 | 0.998 |
| Cell volume [40] | 528.0 Å3 | 0.85 | – |
| Cell volume [41] | 527.0 Å3 | 0.71 | – |
| Setting Solution | Cement | Tin (min) | Tfin (min) |
|---|---|---|---|
| without Alginate | Sr0 | 10 ± 1 | 22 ± 3 |
| Sr2 | 13 ± 1 | 30 ± 3 | |
| Sr5 | 23 ± 3 | 47 ± 3 | |
| with Alginate | Sr0 | 9 ± 2 | 17 ± 2 |
| Sr2 | 11 ± 2 | 26 ± 3 | |
| Sr5 | 25 ± 3 | 51 ± 2 |
| Parameter | Abbreviation | Formula |
|---|---|---|
| Material Perimeter | M-Pm | – |
| Bone Material Contact Length | BMC-Le | – |
| Bone Pores Length | BPo-Le | – |
| Material Diameter | M-Dm | – |
| Bone Material Contact Rate | BMCR | (BMC-Le/M-Pm) × 100 |
| Bone Penetration Rate Length | BPR-Le | (BPo-Le/(M-Dm/2)) × 100 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sprio, S.; Dapporto, M.; Montesi, M.; Panseri, S.; Lattanzi, W.; Pola, E.; Logroscino, G.; Tampieri, A. Novel Osteointegrative Sr-Substituted Apatitic Cements Enriched with Alginate. Materials 2016, 9, 763. https://doi.org/10.3390/ma9090763
Sprio S, Dapporto M, Montesi M, Panseri S, Lattanzi W, Pola E, Logroscino G, Tampieri A. Novel Osteointegrative Sr-Substituted Apatitic Cements Enriched with Alginate. Materials. 2016; 9(9):763. https://doi.org/10.3390/ma9090763
Chicago/Turabian StyleSprio, Simone, Massimiliano Dapporto, Monica Montesi, Silvia Panseri, Wanda Lattanzi, Enrico Pola, Giandomenico Logroscino, and Anna Tampieri. 2016. "Novel Osteointegrative Sr-Substituted Apatitic Cements Enriched with Alginate" Materials 9, no. 9: 763. https://doi.org/10.3390/ma9090763
APA StyleSprio, S., Dapporto, M., Montesi, M., Panseri, S., Lattanzi, W., Pola, E., Logroscino, G., & Tampieri, A. (2016). Novel Osteointegrative Sr-Substituted Apatitic Cements Enriched with Alginate. Materials, 9(9), 763. https://doi.org/10.3390/ma9090763







