Revising the Subsystem Nurse’s A-Phase-Silicocarnotite within the System Ca3(PO4)2–Ca2SiO4
Abstract
:1. Introduction
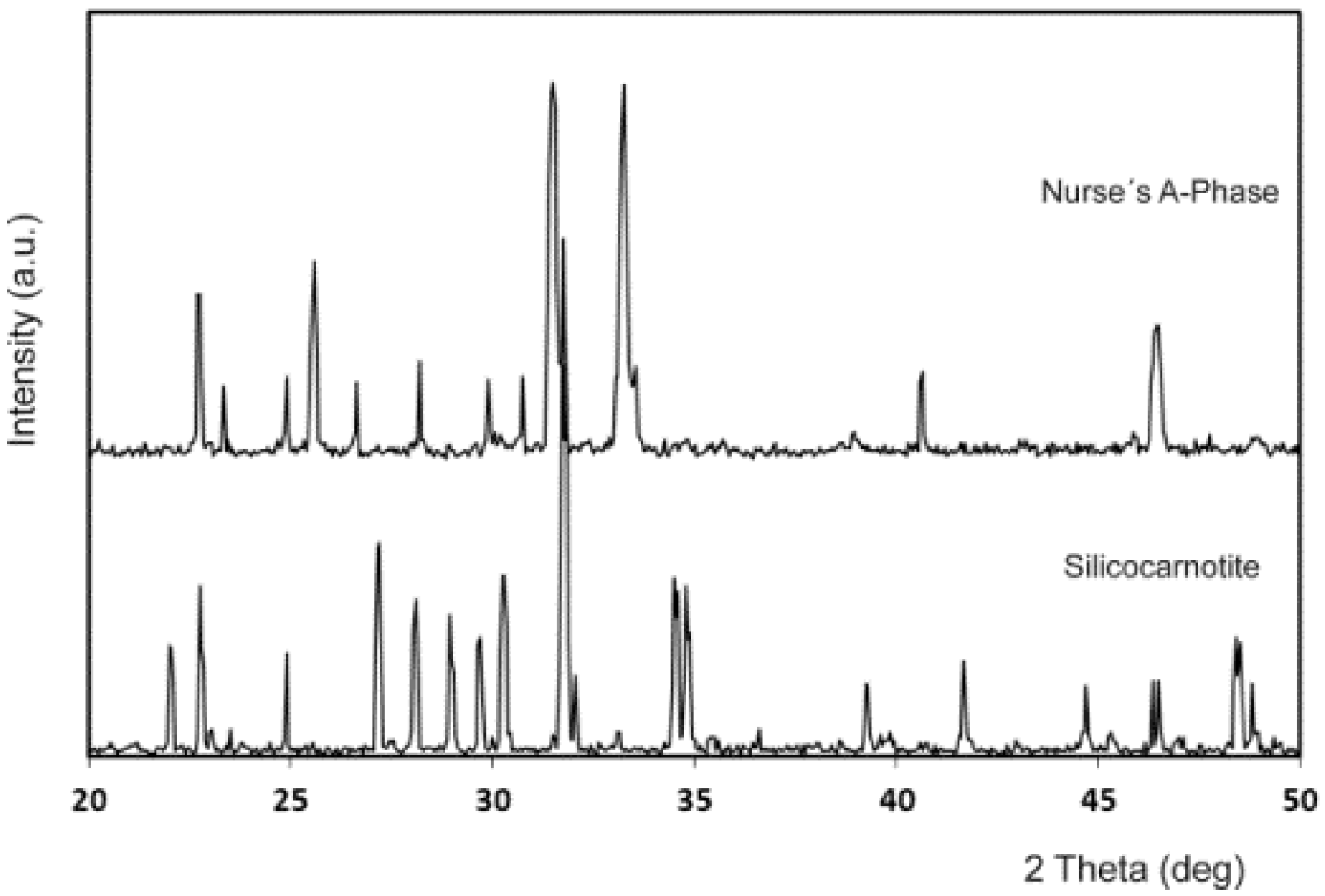
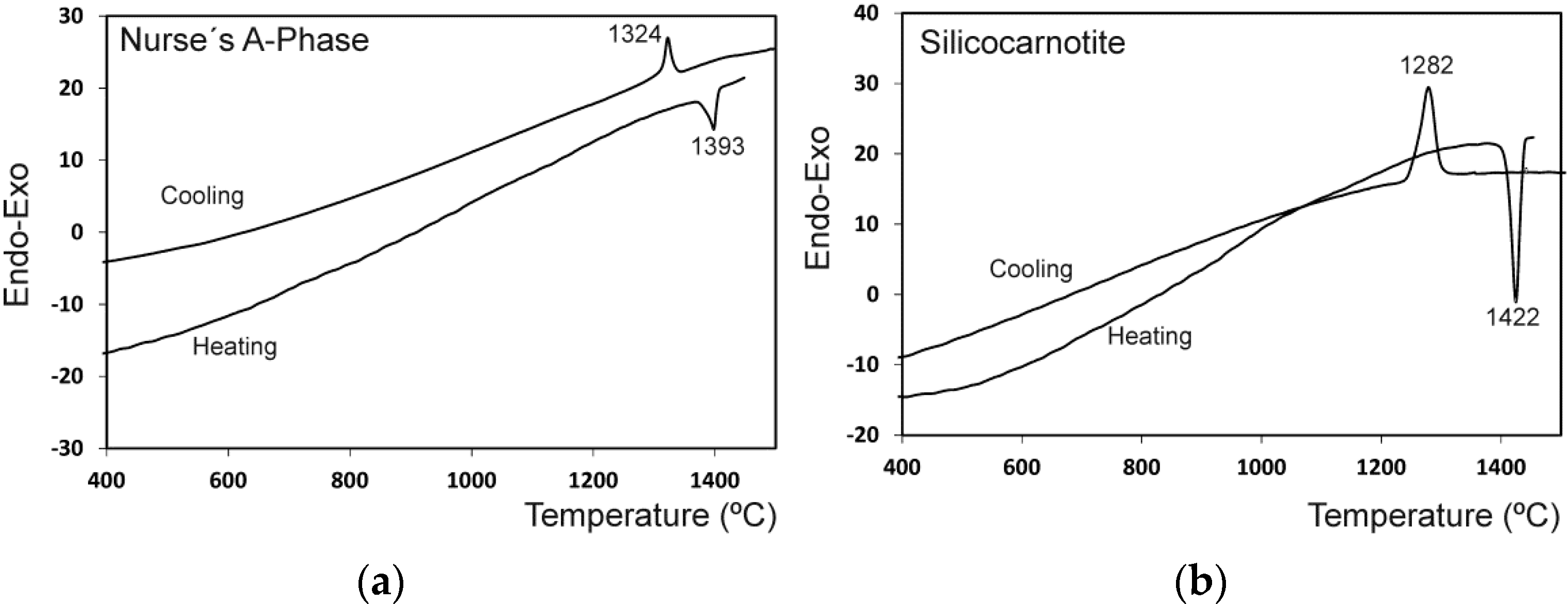
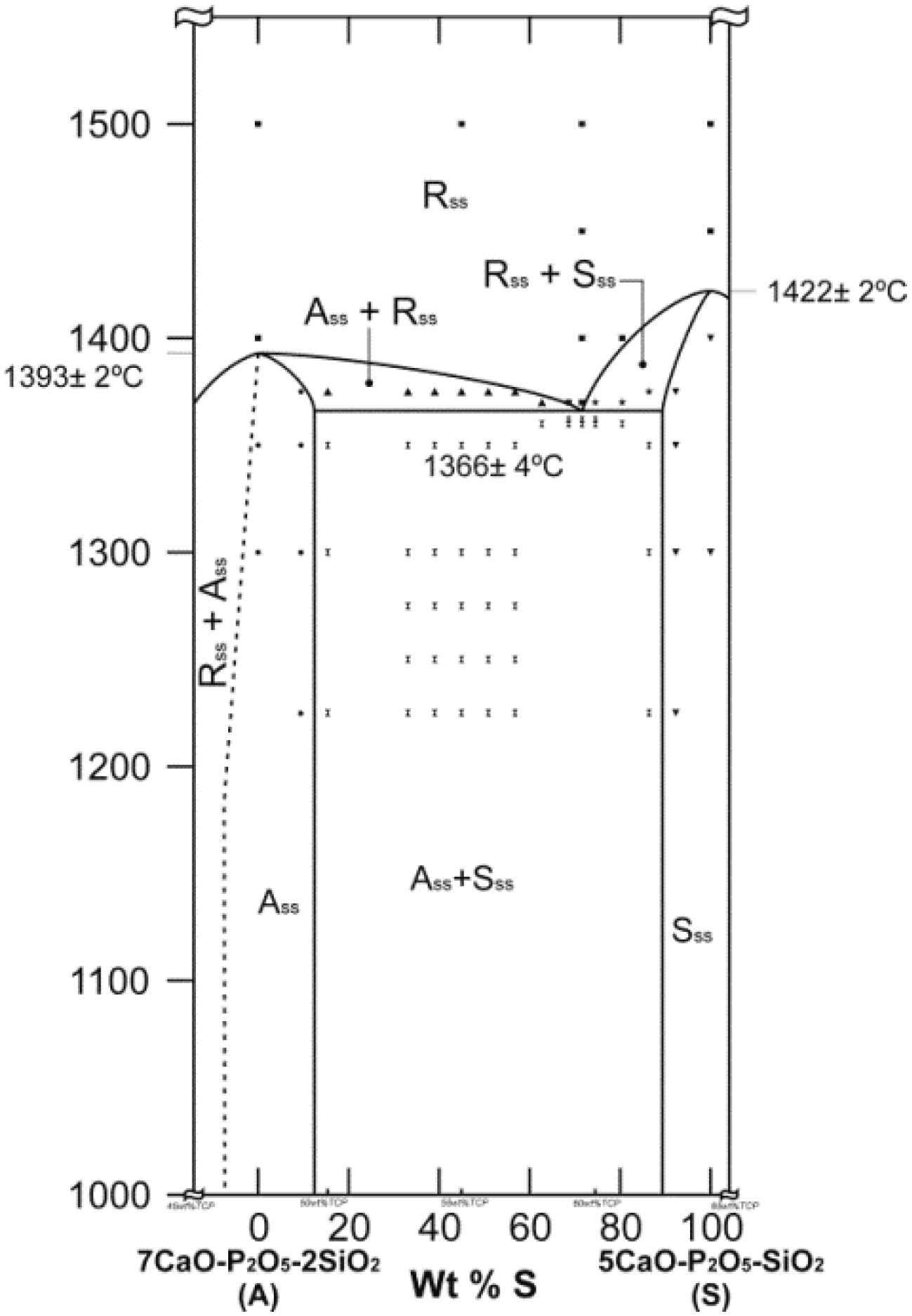
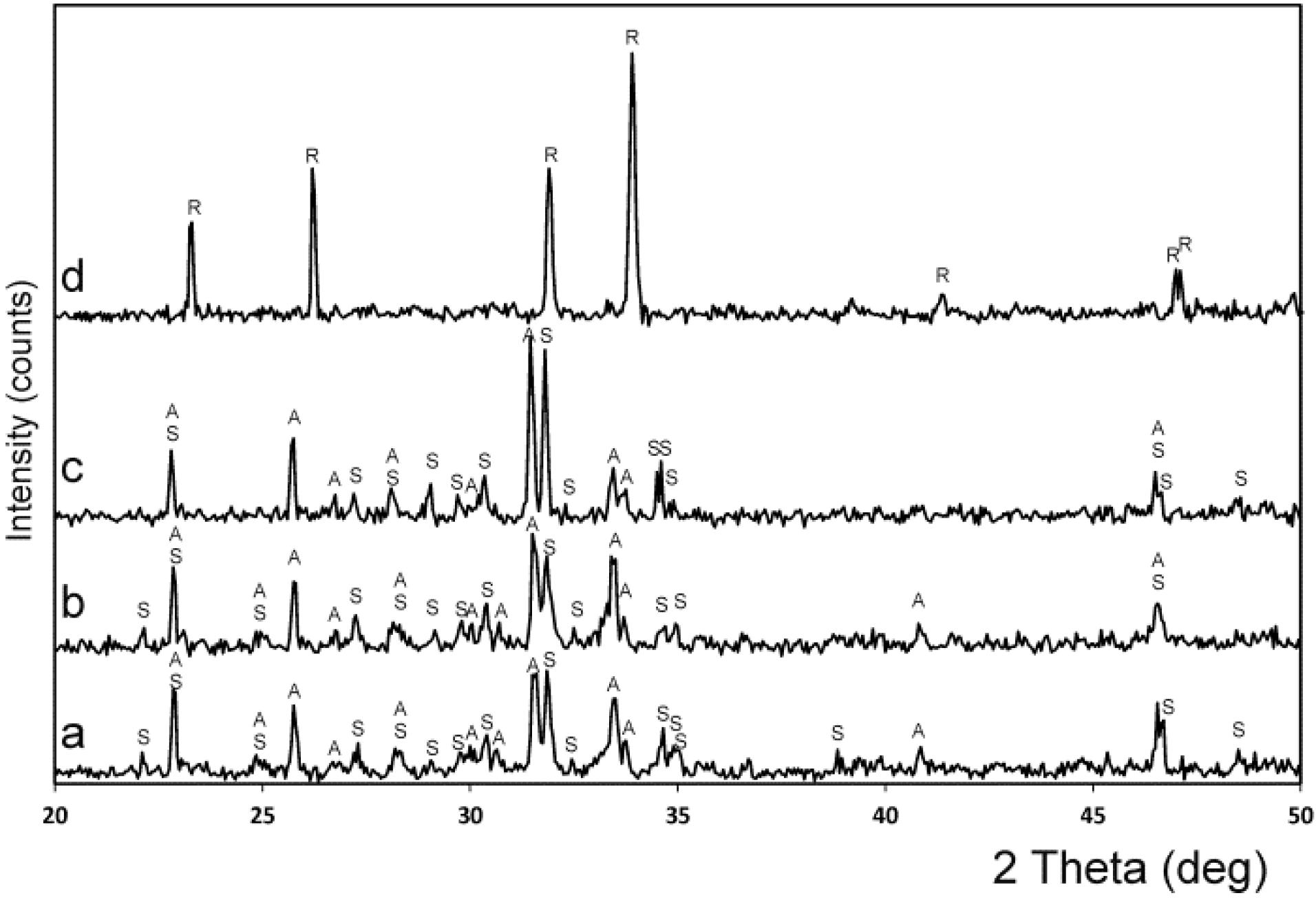
2. Results and Discussion
3. Materials and Methods
3.1. Chemical Analysis
3.2. Phase Diagram Studies
3.3. Thermal Analysis
3.4. XRD
3.5. Microstructure and Microanalysis
3.6. In Vitro Cell Tests
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chevalier, J.; Gremillard, L. Ceramics for medical applications: A picture for the next 20 years. J. Eur. Ceram. Soc. 2009, 29, 1245–1255. [Google Scholar] [CrossRef]
- Martinez, I.M.; Velasquez, P.A.; de Aza, P.N. Synthesis and stability of α-tricalcium phosphate doped with dicalcium silicate in the system Ca3(PO4)2–Ca2SiO4. Mater. Charact. 2010, 61, 761–767. [Google Scholar] [CrossRef]
- Nagelschmidt, G. A new calcium silicophosphate. J. Chem. Soc. 1937, 86, 5–7. [Google Scholar]
- Bredig, M.A. High-temperature crystal chemistry of AmBXn compounds with particular reference to calcium orthosilicate. J. Phys. Chem. 1945, 49, 537–553. [Google Scholar] [CrossRef]
- Bredig, M.A. Isomorphism and allotropy in compounds of the type A2XO4. J. Phys. Chem. 1942, 46, 747–764. [Google Scholar] [CrossRef]
- Franck, H.H.; Bredig, M.A.; Franck, R. Untersuchungenüber Kalk-Alkali Phosphate. Z. Anorg. Allgem. Chem. 1936, 230, 1–27. [Google Scholar] [CrossRef]
- Franck, H.H.; Bredig, M.A.; Kanert, E. Untersuchungenüber Kalk-Alkali Phosphate II. Z. Anorg. Allgem. Chem. 1938, 237, 49–78. [Google Scholar] [CrossRef]
- Lugo, G.J.; Mazón, P.; de Aza, P.N. Phase transitions in single phase Si–Ca–P-based ceramic under thermal treatment. J. Eur. Ceram. Soc. 2015, 35, 3693–3700. [Google Scholar] [CrossRef]
- Lugo, G.J.; Mazón, P.; Baudin, C.; de Aza, P.N. Nurse’s A phase: Synthesis and characterization in the binary system Ca2SiO4–Ca3(PO4)2. J. Am. Ceram. Soc. 2015, 98, 3042–3046. [Google Scholar] [CrossRef]
- Lugo, G.J.; Mazón, P.; de Aza, P.N. Material processing of a new calcium silicophosphate ceramic. Ceram. Int. 2016, 42, 673–680. [Google Scholar] [CrossRef]
- De Aza, P.N.; Mate-Sanchez de Val, J.E.; Baudin, C.; Perez Albacete-Martínez, C.; Armijo Salto, A.; Calvo-Guirado, J.L. Bone neoformation of a novel porous resorbable Si–Ca–P base ceramic with osteoconductive properties: Physical and mechanical characterization, histological and histomorphometric study. Clin. Oral Implant. Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Serena, S.; Sainz, M.A.; Caballero, A. Single-phase silicocarnotite synthesis in the subsystem Ca3(PO4)2–Ca2SiO4. Ceram. Int. 2014, 40, 8245–8252. [Google Scholar] [CrossRef]
- Radev, L.; Hristov, V.; Michailova, I.; Fernandes, M.H.V.; Salvado, I.M.M. Collagen/silicocarnotite composites, cross-linked with chondroitin sulphate: In vitro bioactivity. Process. Appl. Ceram. 2011, 5, 161–170. [Google Scholar] [CrossRef]
- Ruys, A.J. Silicon-Doped Hydroxyapatite. J. Austral. Ceram. Soc. 1993, 29, 71–80. [Google Scholar]
- Serena, S.; Caballero, A.; de Aza, P.N.; Sainz, M.A. New evaluation of the in vitro response of silicocarnotite monophasic material. Ceram. Int. 2015, 41, 9411–9419. [Google Scholar] [CrossRef]
- Stead, J.E.; Ridsdale, C.H. Crystals in basic converter slag. J. Chem. Soc. 1987, 51, 601–610. [Google Scholar] [CrossRef]
- Nurse, R.W.; Welch, J.H.; Gutt, W. High-Temperature Equilibria in the System Dicalcium Silicate-Tricalcium Phosphate. J. Chem. Soc. 1959, 220, 1077–1083. [Google Scholar] [CrossRef]
- Fix, W.; Heymann, H.; Heinke, R. Subsolidus relations in the system 2CaO·SiO2–3CaO·P2O5. J. Am. Ceram. Soc. 1969, 52, 346–347. [Google Scholar] [CrossRef]
- Rubio, V.; de la Casa-Lillo, M.A.; de Aza, S.; de Aza, P.N. The system Ca3(PO4)2–Ca2SiO4: The sub-system Ca2SiO4–7CaOP2O5·2SiO2. J. Am. Ceram.Soc. 2011, 94, 4459–4462. [Google Scholar] [CrossRef]
- Martinez, I.M.; Velasquez, P.A.; de Aza, P.N. The sub-system α-TCPss-Silicocarnotite within the binary system Ca3(PO4)2–Ca2SiO4. J. Am. Ceram. Soc. 2012, 95, 1112–1117. [Google Scholar] [CrossRef]
- Dickens, B.; Brown, W.E. The Crystal Structure of Ca5(PO4)·2SiO4 (Silicocarnotite). Tschermaks Min. Petr. Mitt. 1971, 16, 1–27. [Google Scholar] [CrossRef]
- Paredes, B.; Santana, A.; Arribas, M.I.; Vicente-Salar, N.; de Aza, P.N.; Roche, E.; Such, J.; Reig, J.A. Phenotypic differences during the osteogenic differentiation of single cell-derived clones isolated from human lipoaspirates. J. Tissue. Eng. Regen. Med. 2011, 5, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Meseguer-Olmo, L.; Aznar-Cervantes, S.; Mazón, P.; de Aza, P.N. In vitro behaviour of adult mesenchymal stem cells of human bone marrow origin seeded on a novel bioactive ceramics in the Ca2SiO4–Ca3(PO4)2 system. J. Mater. Sci. Mater. Med. 2012, 23, 3003–3014. [Google Scholar] [CrossRef] [PubMed]
- Martinez, I.M.; Velasquez, P.A.; Meseguer-Olmo, L.; de Aza, P.N. Production and study of in vitro behaviour of monolithic α-Tricalcium Phosphate based ceramics in the system Ca3(PO4)2–Ca2SiO4. Ceram. Int. 2011, 37, 2527–2535. [Google Scholar] [CrossRef]
- Scudiero, D.A.; Shoemaker, R.H.; Paul, K.D.; Monks, A.; Tiemey, S.; Nofziger, T.H.; Currens, M.J.; Seniff, D.; Boyd, M.R. Evaluation of a soluble tetrazolium/formazan assay for cell growth and drug sensitivity in culture using human and other tumor cell lines. Cancer Res. 1988, 48, 4827–4833. [Google Scholar] [PubMed]
- Martinez, I.M.; Velasquez, P.A.; Meseguer-Olmo, L.; Bernabeu-Esclapez, A.; de Aza, P.N. Preparation and characterization of novel bioactive α-Tricalcium Phosphate doped with Dicalcium Silicate ceramics. Mater. Sci. Eng. C. 2012, 32, 878–886. [Google Scholar] [CrossRef]
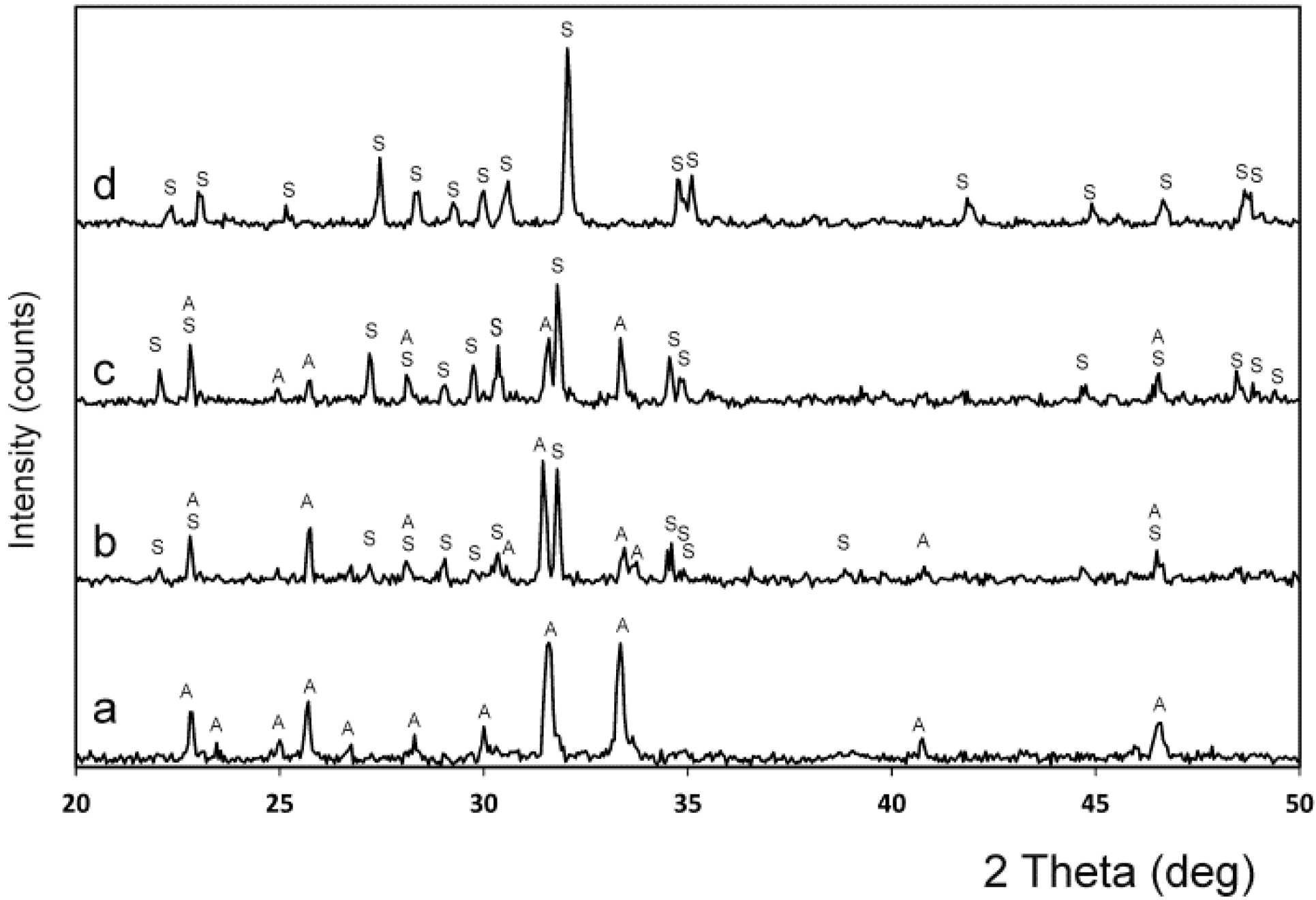
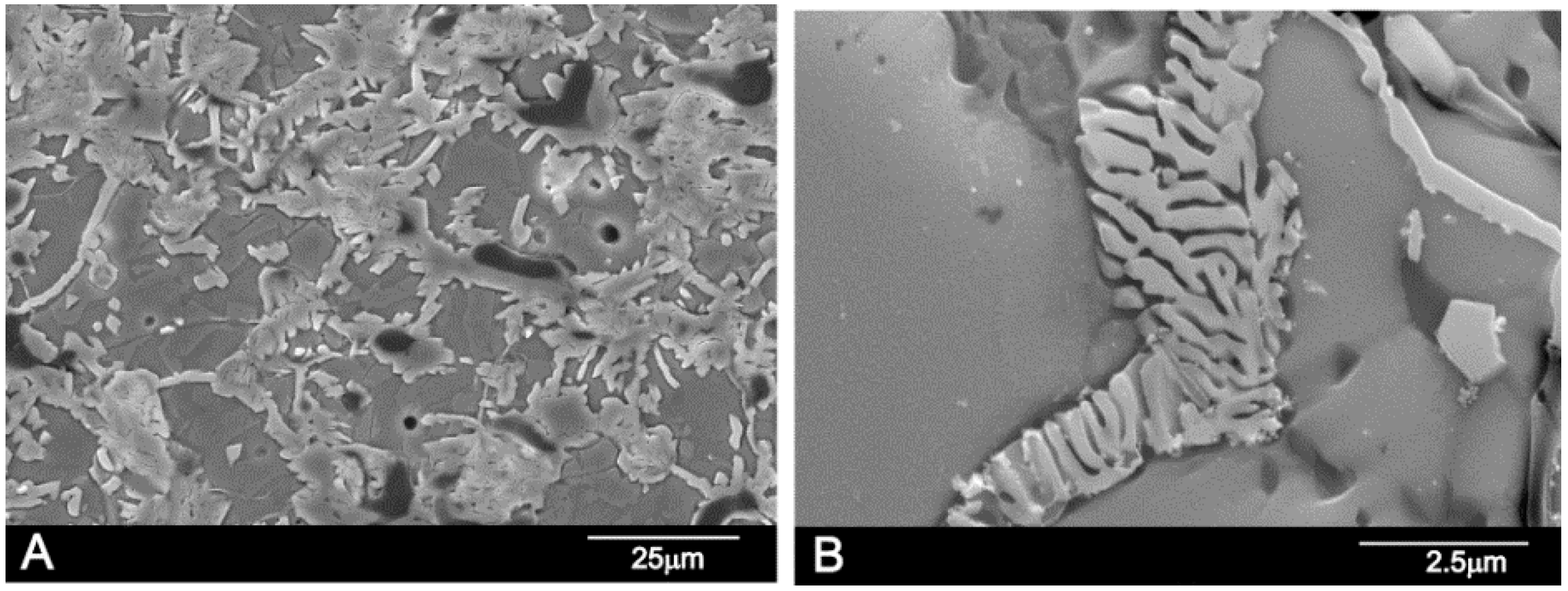
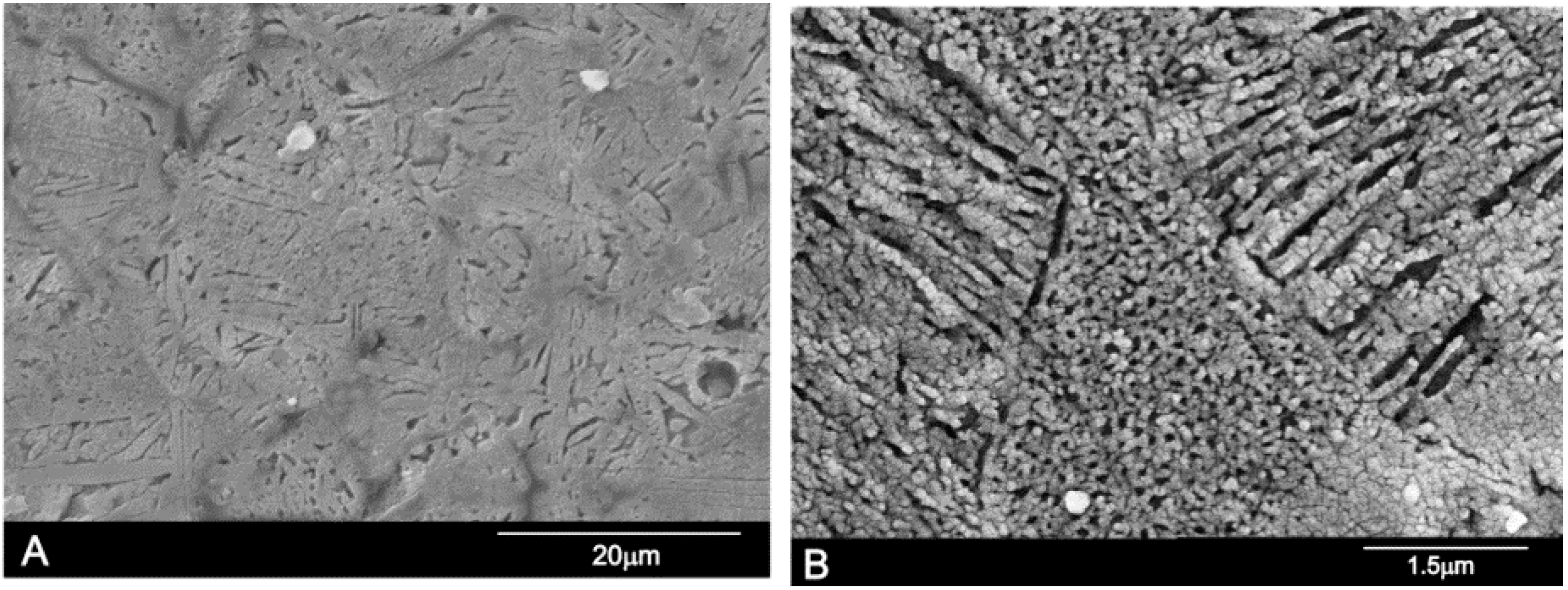


| Chemical Analysis (wt %) | Ass | Sss |
|---|---|---|
| CaO | 59.8 | 57.9 |
| SiO2 | 18.3 | 11.9 |
| P2O5 | 21.5 | 30 |
| MgO | 0.064 | 0.062 |
| Al2O3 | 0.084 | 0.066 |
| Na2O | 0.038 | 0.028 |
| K2O | 0.007 | 0.005 |
| Fe2O3 | 0.036 | 0.032 |
| TiO2 | 0.024 | 0.007 |
| MnO | – | – |
| Composition No. | Holding Conditions | Observed Phase | General Characteristics | |
|---|---|---|---|---|
| Temperature (°C) | Time (h) | |||
| 1 | 1300 | 144 | Ass | White solid |
| 1350 | 144 | Ass | White solid | |
| 1400 | 144 | Rss | White solid | |
| 1500 | 144 | Rss | White solid | |
| 2 | 1225 | 144 | Ass | White solid |
| 1300 | 144 | Ass | White solid | |
| 1350 | 144 | Ass | White solid | |
| 1375 | 144 | Ass | White solid | |
| 3 | 1225 | 144 | Ass + Sss | White solid |
| 1300 | 144 | Ass + Sss | White solid | |
| 1350 | 144 | Ass + Sss | White solid | |
| 1375 | 144 | Ass + Rss | White solid | |
| 4 | 1225 | 48 | Ass + Sss | White solid |
| 1250 | 48 | Ass + Sss | White solid | |
| 1275 | 48 | Ass + Sss | White solid | |
| 1300 | 48 | Ass + Sss | White solid | |
| 1350 | 144 | Ass + Sss | White solid | |
| 1375 | 144 | Ass + Rss | White solid | |
| 5 | 1225 | 48 | Ass + Sss | White solid |
| 1250 | 48 | Ass + Sss | White solid | |
| 1275 | 48 | Ass + Sss | White solid | |
| 1300 | 48 | Ass + Sss | White solid | |
| 1350 | 144 | Ass + Sss | White solid | |
| 1375 | 144 | Ass + Rss | White solid | |
| 6 * | 1225 | 48 | Ass + Sss | White solid |
| 1250 | 48 | Ass + Sss | White solid | |
| 1275 | 48 | Ass + Sss | White solid | |
| 1300 | 48 | Ass + Sss | White solid | |
| 1350 | 144 | Ass + Sss | White solid | |
| 1375 | 144 | Ass + Rss | White solid | |
| 1500 | 144 | Rss | White solid | |
| 7 | 1225 | 48 | Ass + Sss | White solid |
| 1250 | 48 | Ass + Sss | White solid | |
| 1275 | 48 | Ass + Sss | White solid | |
| 1300 | 48 | Ass + Sss | White solid | |
| 1350 | 144 | Ass + Sss | White solid | |
| 1375 | 144 | Ass + Rss | White solid | |
| 8 | 1225 | 48 | Ass + Sss | White solid |
| 1250 | 48 | Ass + Sss | White solid | |
| 1275 | 48 | Ass + Sss | White solid | |
| 1300 | 48 | Ass + Sss | White solid | |
| 1350 | 144 | Ass + Sss | White solid | |
| 1375 | 144 | Ass + Rss | White solid | |
| 9 | 1362 | 144 | Ass + Sss | White solid |
| 1370 | 144 | Ass + Rss | White solid | |
| 10 | 1362 | 144 | Ass + Sss | White solid |
| 1370 | 144 | Rss | White solid | |
| 11 | 1362 | 144 | Ass + Sss | White solid |
| 1370 | 144 | Rss | White solid | |
| 1400 | 144 | Rss | White solid | |
| 1450 | 144 | Rss | White solid | |
| 1500 | 144 | Rss | White solid | |
| 12 | 1362 | 144 | Rss + Sss | White solid |
| 1370 | 144 | Ass + Sss | White solid | |
| 13 | 1362 | 144 | Ass + Sss | White solid |
| 1370 | 144 | Ass + Sss | White solid | |
| 1400 | 144 | Sss | White solid | |
| 14 | 1225 | 48 | Ass + Sss | White solid |
| 1300 | 48 | Ass + Sss | White solid | |
| 1350 | 144 | Ass + Sss | White solid | |
| 1375 | 144 | Rss + Sss | White solid | |
| 15 | 1225 | 48 | Sss | White solid |
| 1300 | 48 | Sss | White solid | |
| 1350 | 144 | Sss | White solid | |
| 1375 | 144 | Sss | White solid | |
| 16 | 1300 | 144 | Sss | White solid |
| 1400 | 144 | Sss | White solid | |
| 1450 | 144 | Rss | White solid | |
| 1500 | 144 | Rss | White solid | |
| C2S | TCP | T (°C) | Composition No. | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| – | 1 | 6 | 11 | 16 | – | |||||||||
| d | I | d | I | d | I | d | I | d | I | d | I | |||
| 100 | 0 | 1525 | – | – | 3.99 | – | 3.99 | 30 | 4.00 | 30 | 3.98 | 40 | 3.98 | 41 |
| 52.61 | 47.39 | 1500 | 3.67 | 4 | – | – | 3.77 | 10 | 3.80 | 10 | 3.81 | 17 | 3.85 | 50 |
| 45 | 55 | 1500 | 2.92 | 61 | 3.75 | 74 | 2.95 | 80 | 2.94 | 100 | 2.94 | 100 | 2.96 | 100 |
| 40.50 | 59.50 | 1500 | 2.77 | 100 | 2.75 | 100 | 2.72 | 70 | 2.72 | 70 | 2.70 | 60 | 2.68 | 68 |
| 35.70 | 64.30 | 1500 | 2.28 | 9 | 2.19 | 22 | 2.20 | 16 | 2.21 | 8 | 2.22 | 22 | 2.22 | 17 |
| 0 | 100 | 1447 | 2.01 | 28 | 2.00 | – | 2.00 | 40 | 1.99 | 10 | 1.95 | 22 | 1.99 | 43 |
| Composition No. | Sample wt % TCP | Sample wt % C2S | Sample wt % S | Sample wt % A |
|---|---|---|---|---|
| 1 | 47.39 | 52.61 | 0.00 | 100.00 |
| 2 | 49.00 | 51.00 | 9.52 | 90.48 |
| 3 | 50.00 | 50.00 | 15.43 | 84.57 |
| 4 | 53.00 | 47.00 | 33.18 | 66.82 |
| 5 | 54.00 | 46.00 | 39.09 | 60.91 |
| 6 | 55.00 | 45.00 | 45.00 | 55.00 |
| 7 | 56.00 | 44.00 | 50.92 | 49.08 |
| 8 | 57.00 | 43.00 | 56.83 | 43.17 |
| 9 | 58.00 | 42.00 | 62.74 | 37.26 |
| 10 | 59.00 | 41.00 | 68.66 | 31.34 |
| 11 | 59.50 | 40.50 | 71.61 | 28.39 |
| 12 | 60.00 | 40.00 | 74.57 | 25.43 |
| 13 | 61.00 | 39.00 | 80.48 | 19.52 |
| 14 | 62.00 | 38.00 | 86.40 | 13.60 |
| 15 | 63.00 | 37.00 | 92.31 | 7.69 |
| 16 | 64.30 | 35.70 | 100.00 | 0.00 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ros-Tárraga, P.; Mazón, P.; Meseguer-Olmo, L.; De Aza, P.N. Revising the Subsystem Nurse’s A-Phase-Silicocarnotite within the System Ca3(PO4)2–Ca2SiO4. Materials 2016, 9, 322. https://doi.org/10.3390/ma9050322
Ros-Tárraga P, Mazón P, Meseguer-Olmo L, De Aza PN. Revising the Subsystem Nurse’s A-Phase-Silicocarnotite within the System Ca3(PO4)2–Ca2SiO4. Materials. 2016; 9(5):322. https://doi.org/10.3390/ma9050322
Chicago/Turabian StyleRos-Tárraga, Patricia, Patricia Mazón, Luis Meseguer-Olmo, and Piedad N. De Aza. 2016. "Revising the Subsystem Nurse’s A-Phase-Silicocarnotite within the System Ca3(PO4)2–Ca2SiO4" Materials 9, no. 5: 322. https://doi.org/10.3390/ma9050322
APA StyleRos-Tárraga, P., Mazón, P., Meseguer-Olmo, L., & De Aza, P. N. (2016). Revising the Subsystem Nurse’s A-Phase-Silicocarnotite within the System Ca3(PO4)2–Ca2SiO4. Materials, 9(5), 322. https://doi.org/10.3390/ma9050322








