Potential of Electrospun Nanofibers for Biomedical and Dental Applications
Abstract
:1. Introduction
| Years | Electrospinning | Oral/Dental Electrospinning | |
|---|---|---|---|
| Topic Search | Title Search | Topic Search | |
| 2005 | 296 | 114 | 1 |
| 2006 | 482 | 204 | 0 |
| 2007 | 623 | 259 | 0 |
| 2008 | 1047 | 373 | 1 |
| 2009 | 1183 | 442 | 2 |
| 2010 | 1431 | 507 | 4 |
| 2011 | 1845 | 579 | 3 |
| 2012 | 2102 | 627 | 12 |
| 2013 | 3377 | 639 | 8 |
| 2014 | 6117 | 793 | 10 |
| 2015 | 5233 | 600 | 6 |
2. Basic Principle and Technique

3. Factors Affecting Electrospinning
| Process Parameters | Systemic Parameters | Solution Parameters | Physical Parameters |
|---|---|---|---|
| Voltage | Polymer type | Viscosity | Humidity |
| Flow rate | Molecular weight | Concentration | Temperature |
| Collection plate | Polymer | Conductivity | Air velocity |
| Distance | Architecture | Dielectric constant | - |
| Angle | Solvent used | Surface tension | - |
| Motion | - | Charge of jet | - |
3.1. Solution Related Parameters
3.2. Polymer Concentration
3.3. Processing Conditions

3.4. Effect of Voltage
3.5. Volumetric Flow Rate
3.6. Distance of Collector
3.7. Effect of Conductivity
3.8. Effects of Solvent
4. Properties of Electrospun (ES) Materials
4.1. Physical Properties
4.2. Mechanical Properties
4.3. Biological Properties
4.4. Chemical Properties
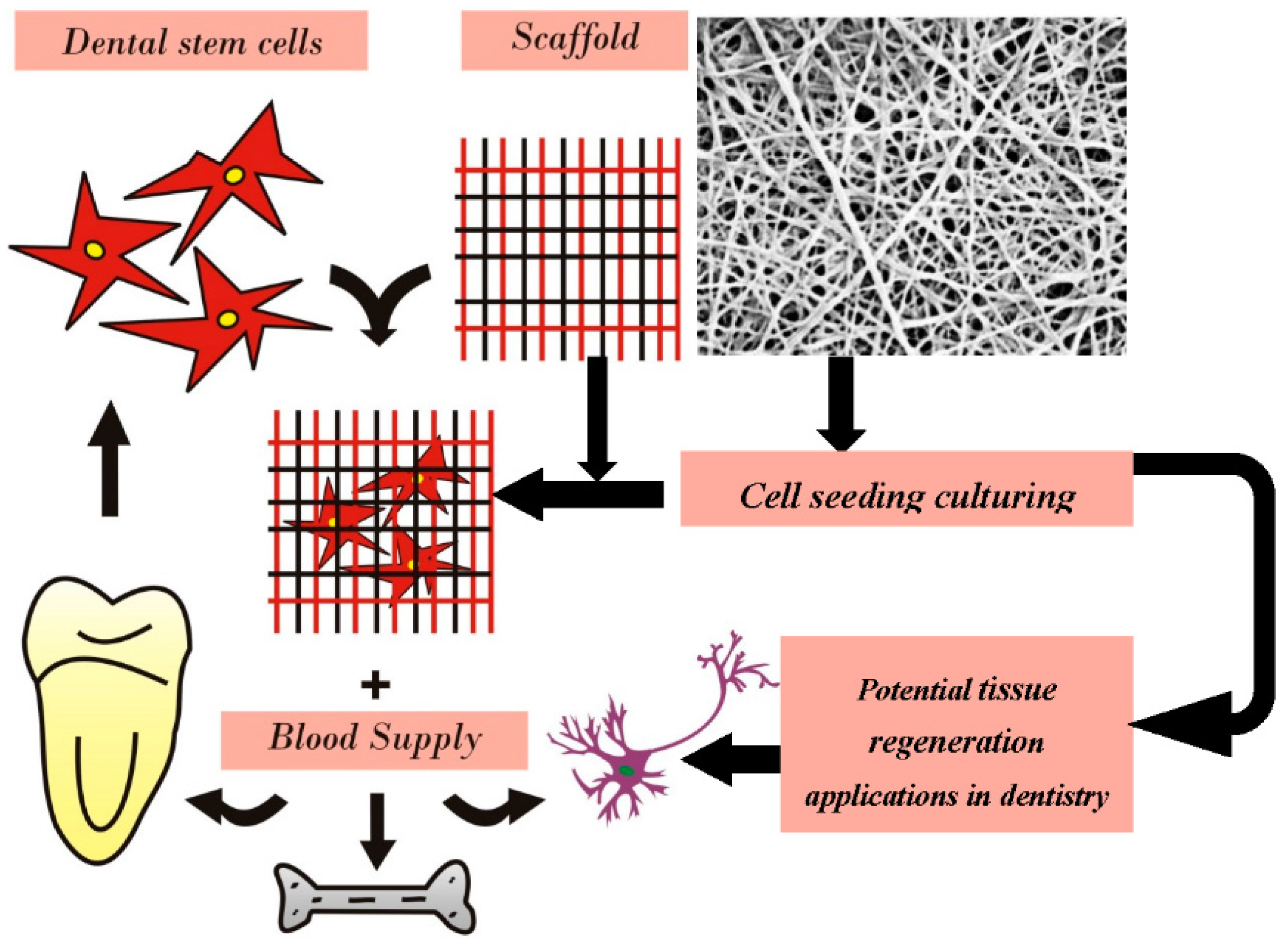
5. Electrospun Nanofibers for Dental Applications
| Applications in Dentistry | Material(s) Electrospun | References |
|---|---|---|
| Tooth regeneration | Polyvinyl alcohol (PVA) Polydiaxonone (PDS) | [80,81,82] |
| Guided tissue regeneration | Collagen, Poly (lactide-co-glycolide) (PLGA) Poly-L-Lactic Acid (PLLA) Poly carpolactone (PCL) Polyethylene oxide (PEO), Silk | [83,84,85,86,87,88] |
| Caries prevention | Chitosan | [89] |
| Reinforcement of resin composites | Polyvinyl alcohol (PVA), Polyacrylnitrile, Polystyrene, Nylon | [90,91,92,93,94] |
| Implant modification | PLGA, Collagen | [95] |
| Cartilage regeneration | PCL Polyethylene oxide (PEO), Chitosan | [96,97,98,99,100] |
| Drug delivery | Poly(ethylene-co-vinylacetate), Poly(lactic acid) (PLLA), Poly (lactide-co-glycolide) (PLGA) | [77,101,102,103,104,105,106] |
| Wound and mucosal repair | Poly-L-(lactic acid), Poly (lactide-co-glycolide) (PLGA), chitin, chitosan, silk fibroin, collagen | [107,108,109,110,111] |
5.1. Regeneration of Pulp Dentin Complex
5.2. Guided tissue Regeneration for Periodontium
5.3. Caries Prevention
5.4. Modification of Resin Composites
5.5. Implant Surface Modification
5.6. Cartilage Regeneration
5.7. Drug Delivery
5.8. Repair of Wounds and Oral Mucosa
6. Limitations of Electrospinning
7. Conclusions
Author Contributions
Conflicts of Interest
References
- Zhang, Y.; Lim, C.T.; Ramakrishna, S.; Huang, Z.M. Recent development of polymer nanofibers for biomedical and biotechnological applications. J. Mater. Sci. Mater. Med. 2005, 16, 933–946. [Google Scholar] [CrossRef] [PubMed]
- Bosworth, L.A.; Downes, S. Electrospinning for Tissue Regeneration; Bosworth, L., Downes, S., Eds.; Woodhead Publishing in Materials; Woodhead Publishing: Oxford, UK, 2011; p. 409. [Google Scholar]
- Stevens, M.M.; George, J.H. Exploring and Engineering the Cell Surface Interface. Science 2005, 310, 1135–1138. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Hsiao, B.S.; Chu, B. Functional electrospun nanofibrous scaffolds for biomedical applications. Adv. Drug Deliv. Rev. 2007, 59, 1392–1412. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.A.; Ma, P.X. Nano-fibrous scaffolds for tissue engineering. Colloids Surf. B Biointerfaces 2004, 39, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.X.; Zhang, R. Synthetic nano-scale fibrous extracellular matrix. J. Biomed. Mater. Res. 1999, 46, 60–72. [Google Scholar] [CrossRef]
- Yang, F.; Murugan, R.; Ramakrishna, S.; Wang, X.; Ma, Y.X.; Wang, S. Fabrication of nano-structured porous PLLA scaffold intended for nerve tissue engineering. Biomaterials 2004, 25, 1891–1900. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Goux, W.J.; Manohar, S.K. Synthesis of polyaniline nanofibers by “nanofiber seeding”. J. Am. Chem. Soc. 2004, 126, 4502–4503. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.R. Template synthesis of electronically conductive polymer nanostructures. Acc. Chem. Res. 1995, 28, 61–68. [Google Scholar] [CrossRef]
- Lakshmi, B.B.; Patrissi, C.J.; Martin, C.R. Sol-Gel Template Synthesis of Semiconductor Oxide Micro-and Nanostructures. Chem. Mater. 1997, 9, 2544–2550. [Google Scholar] [CrossRef]
- Hartgerink, J.D.; Beniash, E.; Stupp, S.I. Self-assembly and mineralization of peptide-amphiphile nanofibers. Science 2001, 294, 1684–1688. [Google Scholar] [CrossRef] [PubMed]
- Niece, K.L.; Hartgerink, J.D.; Donners, J.J.J.M.; Stupp, S.I. Self-assembly combining two bioactive peptide-amphiphile molecules into nanofibers by electrostatic attraction. J. Am. Chem. Soc. 2003, 125, 7146–7147. [Google Scholar] [CrossRef] [PubMed]
- Amiraliyan, N.; Nouri, M.; Kish, M.H. Electrospinning of silk nanofibers. I. An investigation of nanofiber morphology and process optimization using response surface methodology. Fibers Polym. 2009, 10, 167–176. [Google Scholar] [CrossRef]
- Wadbua, P.; Promdonkoy, B.; Maensiri, S.; Siri, S. Different properties of electrospun fibrous scaffolds of separated heavy-chain and light-chain fibroins of Bombyx mori. Int. J. Biol. Macromol. 2010, 46, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Chen, X.; Huang, L.; Shao, Z. Electrospinning of reconstituted silk fiber from aqueous silk fibroin solution. Mater. Sci. Eng. C 2009, 29, 2270–2274. [Google Scholar] [CrossRef]
- Kang, M.; Chen, P.; Jin, H. Preparation of multiwalled carbon nanotubes incorporated silk fibroin nanofibers by electrospinning. Curr. Appl. Phys. 2009, 9, S95–S97. [Google Scholar] [CrossRef]
- Zhang, X.; Reagan, M.R.; Kaplan, D.L. Electrospun silk biomaterial scaffolds for regenerative medicine. Adv. Drug Deliv. Rev. 2009, 61, 988–1006. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Cao, C.; Ma, X. A novel three-dimensional tubular scaffold prepared from silk fibroin by electrospinning. Int. J. Biol. Macromol. 2009, 45, 504–510. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.S.; Maniglio, D.; Motta, A.; Mano, J.F.; Reis, R.L.; Migliaresi, C. Genipin-modified silk-fibroin nanometric nets. Macromol. Biosci. 2008, 8, 766–774. [Google Scholar] [CrossRef] [PubMed]
- Greiner, A.; Wendorff, J.H. Electrospinning: A fascinating method for the preparation of ultrathin fibres. Angew. Chem. Int. Ed. 2007, 46, 5670–5703. [Google Scholar] [CrossRef] [PubMed]
- Jeong, L.; Lee, K.Y.; Park, W.H. Effect of solvent on the characteristics of electrospin regenerated silk fibroin nanofibers. Key Eng. Mater. 2007, 342–343, 813–816. [Google Scholar] [CrossRef]
- Bao, W.; Zhang, Y.; Yin, G.; Wu, J. The structure and property of the electrospinning silk fibroin/gelatin blend nanofibers. E Polym. 2008, 8, 1131–1139. [Google Scholar]
- Soffer, L.; Wang, X.; Mang, X.; Kluge, J.; Dorfmann, L.; Kaplan, D.L.; Leisk, G. Silk-based electrospun tubular scaffolds for tissue-engineered vascular grafts. J. Biomater. Sci. Polym. Ed. 2008, 19, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Meechaisue, C.; Wutticharoenmongkol, P.; Waraput, R.; Huangjing, T.; Ketbumrung, N.; Pavasant, P.; Supaphol, P. Preparation of electrospun silk fibroin fiber mats as bone scaffolds: A preliminary study. Biomed. Mater. 2007, 2, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Deitzel, J.M.; Kleinmeyer, J.; Harris, D.; Beck Tan, N.C. The effect of processing variables on the morphology of electrospun nanofibers and textiles. Polymer 2001, 42, 261–272. [Google Scholar] [CrossRef]
- Li, D.; Wang, Y.; Xia, Y. Electrospinning of polymeric and ceramic nanofibers as uniaxially aligned arrays. Nano Lett. 2003, 3, 1167–1171. [Google Scholar] [CrossRef]
- Subbiah, T.; Bhat, G.; Tock, R.; Parameswaran, S.; Ramkumar, S. Electrospinning of nanofibers. J. Appl. Polym. Sci. 2005, 96, 557–569. [Google Scholar] [CrossRef]
- Dersch, R.; Liu, T.; Schaper, A.; Greiner, A.; Wendorff, J. Electrospun nanofibers: Internal structure and intrinsic orientation. J. Polym. Sci. Part A Polym. Chem. 2003, 41, 545–553. [Google Scholar] [CrossRef]
- Ramakrishna, S.; Fujihara, K.; Teo, W.; Lim, T.; Ma, Z. Electrospinning process. In An Introduction to Electrospinning and Nanofibers; World Scientific Publishing: Singapore, Singapore, 2005; pp. 135–137. [Google Scholar]
- Ioannis, S.C. Novel nanocomposites and nanoceramics based on polymer nanofibers using electrospinning process—A review. J. Mater. Process. Technol. 2005, 167, 283–293. [Google Scholar]
- Fong, H.; Chun, I.; Reneker, D.H. Beaded nanofibers formed during electrospinning. Polymer 1999, 40, 4585–4592. [Google Scholar] [CrossRef]
- Moghe, A.K. Core-Sheath Differentially Biodegradable Nanofiber Structures for Tissue Engineering. Ph.D. Thesis, North Carolina State University, Raleigh, NC, USA, 2008. [Google Scholar]
- James, C.M. Assessment of Electrospinning as an in-House Fabrication Technique for Blood Vessel Mimic Cellular Scaffolding. Master's Thesis, California Polytechnic State University, San Luis Obispo, CA, USA, 2009. [Google Scholar]
- Tan, E.; Goh, C.; Sow, C.; Lim, C. Tensile test of a single nanofiber using an atomic force microscope tip. Appl. Phys. Lett. 2005, 86, 073115. [Google Scholar] [CrossRef]
- Taylor, G. Electrically Driven Jets. Proc. R. Soc. Lond. A Math. Phys. Sci. 1969, 313, 453–475. [Google Scholar] [CrossRef]
- Zeleny, J. The role of surface instability in electrical discharges from drops of alcohol and water in air at atmospheric pressure. J. Frankl. Inst. 1935, 219, 659–675. [Google Scholar] [CrossRef]
- Garg, K.; Bowlin, G.L. Electrospinning jets and nanofibrous structures. Biomicrofluidics 2011, 5, 013403. [Google Scholar] [CrossRef] [PubMed]
- Megelski, S.; Stephens, J.S.; Chase, D.B.; Rabolt, J.F. Micro-and nanostructured surface morphology on electrospun polymer fibers. Macromolecules 2002, 35, 8456–8466. [Google Scholar] [CrossRef]
- Pena, T.R. Preparation and Characterization of Electrospun Poly(D,L-lactide-co-glycolide) Scaffolds for Vascular Tissue Engineering and the Advancement of an In Vitro Blood Vessel Mimic. Master's Thesis, California Polytechnic State University, San Luis Obispo, CA, USA, 2009; p. 152. [Google Scholar]
- Matthews, J.A.; Wnek, G.E.; Simpson, D.G.; Bowlin, G.L. Electrospinning of collagen nanofibers. Biomacromolecules 2002, 3, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Tong, H.W.; Wang, M. Effects of Processing Parameters on the Morphology and Size of Electrospun PHBV Micro-and Nano-Fibers. Key Eng. Mater. 2007, 334–335, 1233–1236. [Google Scholar] [CrossRef]
- Jeun, J.; Kim, Y.; Lim, Y.; Choi, J.; Jung, C.; Kang, P.; Nho, Y. Electrospinning of Poly(L-lactide-co-D,L-lactide). J. Ind. Eng. Chem. 2007, 13, 592–596. [Google Scholar]
- Macossay, J.; Marruffo, A.; Rincon, R.; Eubanks, T.; Kuang, A. Effect of needle diameter on nanofiber diameter and thermal properties of electrospun poly(methyl methacrylate). Polym. Adv. Technol. 2007, 18, 180–183. [Google Scholar] [CrossRef]
- Baumgarten, P.K. Electrostatic spinning of acrylic microfibers. J. Colloid Interface Sci. 1971, 36, 71–79. [Google Scholar] [CrossRef]
- Zong, X.; Kim, K.; Fang, D.; Ran, S.; Hsiao, B.S.; Chu, B. Structure and process relationship of electrospun bioabsorbable nanofiber membranes. Polymer 2002, 43, 4403–4412. [Google Scholar] [CrossRef]
- Huang, L.; Nagapudi, K.; Apkarian, R.P.; Chaikof, E.L. Engineered collagen–PEO nanofibers and fabrics. J. Biomater. Sci. Polym. Ed. 2001, 12, 979–993. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.H.; Kim, H.Y.; Lee, D.R.; Park, S.Y.; Khil, M.S. Characterization of PVOH nonwoven mats prepared from surfactant-polymer system via electrospinning. Macromol. Res. 2005, 13, 385–390. [Google Scholar] [CrossRef]
- Lin, T.; Wang, H.; Wang, H.; Wang, X. The charge effect of cationic surfactants on the elimination of fibre beads in the electrospinning of polystyrene. Nanotechnology 2004, 15, 1375–1381. [Google Scholar] [CrossRef]
- Zeng, J.; Xu, X.; Chen, X.; Liang, Q.; Bian, X.; Yang, L.; Jing, X. Biodegradable electrospun fibers for drug delivery. J. Controlled Release 2003, 92, 227–231. [Google Scholar] [CrossRef]
- Sill, T.J.; von Recum, H.A. Electrospinning: Applications in drug delivery and tissue engineering. Biomaterials 2008, 29, 1989–2006. [Google Scholar] [CrossRef] [PubMed]
- Pillay, V.; Dott, C.; Choonara, Y.E.; Tyagi, C.; Tomar, L.; Kumar, P.; du Toit, L.C.; Ndesendo, V.M. A review of the effect of processing variables on the fabrication of electrospun nanofibers for drug delivery applications. J. Nanomater. 2013, 2013, 1–22. [Google Scholar] [CrossRef]
- Lannutti, J.; Reneker, D.; Ma, T.; Tomasko, D.; Farson, D. Electrospinning for tissue engineering scaffolds. Mater. Sci. Eng. C 2007, 27, 504–509. [Google Scholar] [CrossRef]
- McCann, J.T.; Li, D.; Xia, Y. Electrospinning of nanofibers with core-sheath, hollow, or porous structures. J. Mater. Chem. 2005, 15, 735–738. [Google Scholar] [CrossRef]
- Kwon, I.K.; Kidoaki, S.; Matsuda, T. Electrospun nano-to microfiber fabrics made of biodegradable copolyesters: Structural characteristics, mechanical properties and cell adhesion potential. Biomaterials 2005, 26, 3929–3939. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Q.; Lee, B.L.; Komvopoulos, K.; Yan, Z.; Li, S. Plasma surface chemical treatment of electrospun poly(L-lactide) microfibrous scaffolds for enhanced cell adhesion, growth, and infiltration. Tissue Eng. Part A 2013, 19, 1188–1198. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; He, X.; Jabbari, E. Osteogenic differentiation of marrow stromal cells on random and aligned electrospun poly(L-lactide) nanofibers. Ann. Biomed. Eng. 2011, 39, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Eichhorn, S.J.; Sampson, W.W. Statistical geometry of pores and statistics of porous nanofibrous assemblies. J. R. Soc. Interface 2005, 2, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Jong-Sang, K.; Reneker, D.H. Mechanical properties of composites using ultrafine electrospun fibers. Polym. Compos. 1999, 20, 124–131. [Google Scholar]
- Mahjour, S.B.; Fu, X.; Yang, X.; Fong, J.; Sefat, F.; Wang, H. Rapid creation of skin substitutes from human skin cells and biomimetic nanofibers for acute full-thickness wound repair. Burns 2015, 41, 1764–1774. [Google Scholar] [CrossRef] [PubMed]
- Sefat, F.; Denyer, M.; Youseffi, M. Imaging via widefield surface plasmon resonance microscope for studying bone cell interactions with micropatterned ECM proteins. J. Microsc. 2011, 241, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Miles, R.E. Random Polygons Determined by Random Lines in a Plane, II. Proc. Natl. Acad. Sci. USA 1964, 52, 1157–1160. [Google Scholar] [CrossRef] [PubMed]
- Sefat, F.; McKean, R.; Deshpande, P.; Ramachandran, C.; Hill, C.J.; Sangwan, V.S.; Ryan, A.J.; MacNeil, S. Production, sterilisation and storage of biodegradable electrospun PLGA membranes for delivery of limbal stem cells to the cornea. Procedia Eng. 2013, 59, 101–116. [Google Scholar] [CrossRef]
- Mauck, R.L.; Baker, B.M.; Nerurkar, N.L.; Burdick, J.A.; Li, W.; Tuan, R.S.; Elliott, D.M. Engineering on the straight and narrow: The mechanics of nanofibrous assemblies for fiber-reinforced tissue regeneration. Tissue Eng. Part B Rev. 2009, 15, 171–193. [Google Scholar] [CrossRef] [PubMed]
- McManus, M.C.; Boland, E.D.; Koo, H.P.; Barnes, C.P.; Pawlowski, K.J.; Wnek, G.E.; Simpson, D.G.; Bowlin, G.L. Mechanical properties of electrospun fibrinogen structures. Acta Biomater. 2006, 2, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Barnes, C.P.; Pemble, C.W.; Brand, D.D.; Simpson, D.G.; Bowlin, G.L. Cross-linking electrospun type II collagen tissue engineering scaffolds with carbodiimide in ethanol. Tissue Eng. 2007, 13, 1593–1605. [Google Scholar] [CrossRef] [PubMed]
- Panzavolta, S.; Gioffrè, M.; Focarete, M.L.; Gualandi, C.; Foroni, L.; Bigi, A. Electrospun gelatin nanofibers: Optimization of genipin cross-linking to preserve fiber morphology after exposure to water. Acta Biomater. 2011, 7, 1702–1709. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Jin, H.J.; Kaplan, D.L.; Rutledge, G.C. Mechanical properties of electrospun silk fibers. Macromolecules 2004, 37, 6856–6864. [Google Scholar] [CrossRef]
- Xue, Y.F.; He, C.L.; Mo, X.M. Electrospun spider nanofibers: The influence of ethanol on the structure and properties. In Proceedings of the 2007 International Forum on Biomedical Textile Materials, Shanghai, China, 30 May–2 June 2007; pp. 178–181.
- Min, B.M.; Jeong, L.; Lee, K.Y.; Park, W.H. Regenerated silk fibroin nanofibers: Water vapor-induced structural changes and their effects on the behavior of normal human cells. Macromol. Biosci. 2006, 6, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Mijovic, B.; Trcin, M.; Agic, A.; Zdraveva, E.; Bujic, M.; Spoljaric, I.; Kosec, V. Study on cell adhesion detection onto biodegradable electrospun PCL scaffolds. JFBI 2012, 5, 33–40. [Google Scholar] [CrossRef]
- Carlberg, B.; Axell, M.Z.; Nannmark, U.; Liu, J.; Kuhn, H.G. Electrospun polyurethane scaffolds for proliferation and neuronal differentiation of human embryonic stem cells. Biomed. Mater. 2009, 4, 045004. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, P.; Ramachandran, C.; Sefat, F.; Mariappan, I.; Johnson, C.; McKean, R.; Hannah, M.; Sangwan, V.S.; Claeyssens, F.; Ryan, A.J. Simplifying corneal surface regeneration using a biodegradable synthetic membrane and limbal tissue explants. Biomaterials 2013, 34, 5088–5106. [Google Scholar] [CrossRef] [PubMed]
- Lowery, J.L.; Datta, N.; Rutledge, G.C. Effect of fiber diameter, pore size and seeding method on growth of human dermal fibroblasts in electrospun poly (ɛ-caprolactone) fibrous mats. Biomaterials 2010, 31, 491–504. [Google Scholar] [CrossRef] [PubMed]
- Jayasinghe, S.N. Cell electrospinning: A novel tool for functionalising fibres, scaffolds and membranes with living cells and other advanced materials for regenerative biology and medicine. Analyst 2013, 138, 2215–2223. [Google Scholar] [CrossRef] [PubMed]
- Townsend-Nicholson, A.; Jayasinghe, S.N. Cell electrospinning: A unique biotechnique for encapsulating living organisms for generating active biological microthreads/scaffolds. Biomacromolecules 2006, 7, 3364–3369. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, Z.; Najeeb, S.; Khurshid, Z.; Verma, V.; Rashid, H.; Glogauer, M. Biodegradable Materials for Bone Repair and Tissue Engineering Applications. Materials 2015, 8, 5744–5794. [Google Scholar] [CrossRef]
- Kim, K.; Luu, Y.K.; Chang, C.; Fang, D.; Hsiao, B.S.; Chu, B.; Hadjiargyrou, M. Incorporation and controlled release of a hydrophilic antibiotic using poly(lactide-co-glycolide)-based electrospun nanofibrous scaffolds. J. Controlled Release 2004, 98, 47–56. [Google Scholar] [CrossRef] [PubMed]
- You, Y.; Min, B.; Lee, S.J.; Lee, T.S.; Park, W.H. In vitro degradation behavior of electrospun polyglycolide, polylactide, and poly(lactide-co-glycolide). J. Appl. Polym. Sci. 2005, 95, 193–200. [Google Scholar] [CrossRef]
- Bottino, M.C.; Kamocki, K.; Yassen, G.H.; Platt, J.A.; Vail, M.M.; Ehrlich, Y.; Spolnik, K.J.; Gregory, R.L. Bioactive nanofibrous scaffolds for regenerative endodontics. J. Dent. Res. 2013, 92, 963–969. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.; Asran, A.S.; Michler, G.H.; Simon, P.; Kim, J. Electrospun PVA/HAp nanocomposite nanofibers: Biomimetics of mineralized hard tissues at a lower level of complexity. Bioinspiration Biomim. 2008, 3, 046003. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.J.; Bae, W.J.; Kim, J.M.; Kim, J.J.; Lee, E.J.; Kim, H.W.; Kim, E.C. Mineralized polycaprolactone nanofibrous matrix for odontogenesis of human dental pulp cells. J. Biomater. Appl. 2014, 28, 1069–1078. [Google Scholar] [CrossRef] [PubMed]
- Bottino, M.C.; Yassen, G.H.; Platt, J.A.; Labban, N.; Windsor, L.J.; Spolnik, K.J.; Bressiani, A.H. A novel three-dimensional scaffold for regenerative endodontics: Materials and biological characterizations. J. Tissue Eng. Regen. Med. 2015, 9, E116–E123. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Nguyen, T.B.L.; Min, Y.; Lee, B. In vitro and in vivo studies of BMP-2-loaded PCL–gelatin–BCP electrospun scaffolds. Tissue Eng. Part A 2014, 20, 3279–3289. [Google Scholar] [CrossRef] [PubMed]
- Ranjbar-Mohammadi, M.; Zamani, M.; Prabhakaran, M.; Bahrami, S.H.; Ramakrishna, S. Electrospinning of PLGA/gum tragacanth nanofibers containing tetracycline hydrochloride for periodontal regeneration. Mater. Sci. Eng. C 2016, 58, 521–531. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ni, M.; Zhang, M.; Ratner, B. Calcium phosphate-chitosan composite scaffolds for bone tissue engineering. Tissue Eng. 2003, 9, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.; Seo, S.; Moon, H.; Yoo, M.; Park, I.; Kim, B.; Cho, C. Chitosan and its derivatives for tissue engineering applications. Biotechnol. Adv. 2008, 26, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Su, B.; Ramakrishna, S.; Lim, C. Chitosan nanofibers from an easily electrospinnable UHMWPEO-doped chitosan solution system. Biomacromolecules 2007, 9, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.J.; Fridrikh, S.; Rutledge, G.C.; Kaplan, D. Electrospinning bombyx mori silk with poly(ethylene oxide). Abstr. Pap. Am. Chem. Soc. 2002, 3, 1233–1239. [Google Scholar]
- Samprasit, W.; Kaomongkolgit, R.; Sukma, M.; Rojanarata, T.; Ngawhirunpat, T.; Opanasopit, P. Mucoadhesive electrospun chitosan-based nanofibre mats for dental caries prevention. Carbohydr. Polym. 2015, 117, 933–940. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Ciselli, P.; Kuznetsov, E.; Peijs, T.; Barber, A.H. Effective reinforcement in carbon nanotube-polymer composites. Philos. Trans. A. Math. Phys. Eng. Sci. 2008, 366, 1613–1626. [Google Scholar] [CrossRef] [PubMed]
- Behler, K.D.; Stravato, A.; Mochalin, V.; Korneva, G.; Yushin, G.; Gogotsi, Y. Nanodiamond-polymer composite fibers and coatings. ACS Nano 2009, 3, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Demir, M.M.; Horzum, N.; Tasdemirci, A.; Turan, K.; Güden, M. Mechanical Interlocking between Porous Electrospun Polystyrene Fibers and an Epoxy Matrix. ACS Appl. Mater. Interfaces 2014, 6, 21901–21905. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Borges, A.L.; Münchow, E.A.; de Oliveira, S.A.C.; Yoshida, T.; Vallittu, P.K.; Bottino, M.C. Effect of random/aligned nylon-6/MWCNT fibers on dental resin composite reinforcement. J. Mech. Behav. Biomed. Mater. 2015, 48, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.W.; An, S.; Jo, H.S.; Yoon, S.S.; Yarin, A.L. Self-healing Nanofiber-Reinforced Polymer Composites: Tensile Testing and Recovery of Mechanical Properties. ACS Appl. Mater. Interfaces 2015, 7, 19546–19554. [Google Scholar] [CrossRef] [PubMed]
- Ravichandran, R.; Ng, C.C.; Liao, S.; Pliszka, D.; Raghunath, M.; Ramakrishna, S.; Chan, C.K. Biomimetic surface modification of titanium surfaces for early cell capture by advanced electrospinning. Biomed. Mater. 2012, 7, 015001. [Google Scholar] [CrossRef] [PubMed]
- Ainola, M.; Tomaszewski, W.; Ostrowska, B.; Wesolowska, E.; Wagner, H.D.; Swieszkowski, W.; Sillat, T.; Peltola, E.; Konttinen, Y.T. A bioactive hybrid three-dimensional tissue-engineering construct for cartilage repair. J. Biomater. Appl. 2016, 30, 873–885. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, A.; Lin, H.Y.; Vu, D.; Larsen, G. Synthesis and evaluation of scaffolds prepared from chitosan fibers for potential use in cartilage tissue engineering. Biomed. Sci. Instrum. 2004, 40, 117–122. [Google Scholar] [PubMed]
- Levorson, E.J.; Sreerekha, P.R.; Chennazhi, K.P.; Kasper, F.K.; Nair, S.V.; Mikos, A.G. Fabrication and characterization of multiscale electrospun scaffolds for cartilage regeneration. Biomed. Mater. 2013, 8, 014103. [Google Scholar] [CrossRef] [PubMed]
- Thorvaldsson, A.; Stenhamre, H.; Gatenholm, P.; Walkenström, P. Electrospinning of highly porous scaffolds for cartilage regeneration. Biomacromolecules 2008, 9, 1044–1049. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Danielson, K.G.; Alexander, P.G.; Tuan, R.S. Biological response of chondrocytes cultured in three-dimensional nanofibrous poly (ϵ-caprolactone) scaffolds. J. Biomed. Mater. Res. Part A 2003, 67, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- Vacanti, N.M.; Cheng, H.; Hill, P.S.; Guerreiro, J.D.; Dang, T.T.; Ma, M.; Watson, S.; Hwang, N.S.; Langer, R.; Anderson, D.G. Localized delivery of dexamethasone from electrospun fibers reduces the foreign body response. Biomacromolecules 2012, 13, 3031–3038. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Yu, X.; Markel, D.C.; Shi, T.; Ren, W. Coaxial PCL/PVA electrospun nanofibers: Osseointegration enhancer and controlled drug release device. Biofabrication 2013, 5, 035006. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Li, F.; Wei, H.; Shi, J.F.; Rao, G.Z.; Li, A.; Gou, J.Z. Preliminary study of the dual release baicalin and rhBMP-2 system to improve periodontal tissue regeneration in minipigs. Shanghai Kou Qiang Yi Xue 2013, 22, 126–131. (In Chinese) [Google Scholar] [PubMed]
- Xue, J.; He, M.; Niu, Y.; Liu, H.; Crawford, A.; Coates, P.; Chen, D.; Shi, R.; Zhang, L. Preparation and in vivo efficient anti-infection property of GTR/GBR implant made by metronidazole loaded electrospun polycaprolactone nanofiber membrane. Int. J. Pharm. 2014, 475, 566–577. [Google Scholar] [CrossRef] [PubMed]
- Verreck, G.; Chun, I.; Peeters, J.; Rosenblatt, J.; Brewster, M.E. Preparation and characterization of nanofibers containing amorphous drug dispersions generated by electrostatic spinning. Pharm. Res. 2003, 20, 810–817. [Google Scholar] [CrossRef] [PubMed]
- Kenawy, E.; Bowlin, G.L.; Mansfield, K.; Layman, J.; Simpson, D.G.; Sanders, E.H.; Wnek, G.E. Release of tetracycline hydrochloride from electrospun poly(ethylene-co-vinylacetate), poly(lactic acid), and a blend. J. Controlled Release 2002, 81, 57–64. [Google Scholar] [CrossRef]
- Thakur, R.; Florek, C.; Kohn, J.; Michniak, B. Electrospun nanofibrous polymeric scaffold with targeted drug release profiles for potential application as wound dressing. Int. J. Pharm. 2008, 364, 87–93. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Wang, J.; Huang, P.; Zeng, B.; Li, H.; Cao, Q.; Zhang, S.; Luo, Z.; Deng, D.Y.; Zhang, H. Electrospinning polyvinylidene fluoride fibrous membranes containing anti-bacterial drugs used as wound dressing. Colloids Surf. B Biointerfaces 2015, 130, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Noh, H.K.; Lee, S.W.; Kim, J.; Oh, J.; Kim, K.; Chung, C.; Choi, S.; Park, W.H.; Min, B. Electrospinning of chitin nanofibers: Degradation behavior and cellular response to normal human keratinocytes and fibroblasts. Biomaterials 2006, 27, 3934–3944. [Google Scholar] [CrossRef] [PubMed]
- Blackwood, K.A.; McKean, R.; Canton, I.; Freeman, C.O.; Franklin, K.L.; Cole, D.; Brook, I.; Farthing, P.; Rimmer, S.; Haycock, J.W. Development of biodegradable electrospun scaffolds for dermal replacement. Biomaterials 2008, 29, 3091–3104. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Han, Y.; Zhang, F.; Ge, Z.; Liu, X.; Lu, Q. Buccal mucosa repair with electrospun silk fibroin matrix in a rat model. Int. J. Artif. Organs 2015, 38, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Damle, S.; Bhattal, H.; Loomba, A. Apexification of anterior teeth: A comparative evaluation of mineral trioxide aggregate and calcium hydroxide paste. J. Clin. Pediatr. Dent. 2012, 36, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Odabas, M.E.; Alacam, A.; Sillelioglu, H.; Deveci, C. Clinical and radiographic success rates of mineral trioxide aggregate and ferric sulphate pulpotomies performed by dental students. Eur. J. Paediatr. Dent. 2012, 13, 118–122. [Google Scholar] [PubMed]
- Rizvi, A.; Zafar, M.S.; Farid, W.M.; Gazal, G. Assessment of Antimicrobial Efficacy of MTAD, Sodium Hypochlorite, EDTA and Chlorhexidine for Endodontic Applications: An In vitro Study. Middle East J. Sci. Res. 2014, 21, 353–357. [Google Scholar]
- Pihlstrom, B.L.; Michalowicz, B.S.; Johnson, N.W. Periodontal diseases. Lancet 2005, 366, 1809–1820. [Google Scholar] [CrossRef]
- Bottino, M.C.; Thomas, V.; Schmidt, G.; Vohra, Y.K.; Chu, T.G.; Kowolik, M.J.; Janowski, G.M. Recent advances in the development of GTR/GBR membranes for periodontal regeneration—A materials perspective. Dent. Mater. 2012, 28, 703–721. [Google Scholar] [CrossRef] [PubMed]
- Gottlow, J.; Laurell, L.; Lundgren, D.; Mathisen, T.; Nyman, S.; Rylander, H.; Bogentoft, C. Periodontal tissue response to a new bioresorbable guided tissue regeneration device: A longitudinal study in monkeys. Int. J. Periodontics Restor. Dent. 1994, 14, 436–449. [Google Scholar]
- Al-Arrayed, F.; Adam, S.; Moran, J.; Dowell, P. Clinical trial of cross-linked human type I collagen as a barrier material in surgical periodontal treatment. J. Clin. Periodontol. 1995, 22, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Dowell, P.; Al-Arrayed, F.; Adam, S.; Moran, J. A comparative clinical study: The use of human type I collagen with and without the addition of metronidazole in the GTR method of treatment of periodontal disease. J. Clin. Periodontol. 1995, 22, 543–549. [Google Scholar] [CrossRef] [PubMed]
- Srouji, S.; Kizhner, T.; Suss-Tobi, E.; Livne, E.; Zussman, E. 3-D Nanofibrous electrospun multilayered construct is an alternative ECM mimicking scaffold. J. Mater. Sci. Mater. Med. 2008, 19, 1249–1255. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Gouma, P. Electrospun bioscaffolds that mimic the topology of extracellular matrix. Nanomed. Nanotechnol. Biol. Med. 2006, 2, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Prabhakaran, M.P.; Venugopal, J.; Ramakrishna, S. Electrospun nanostructured scaffolds for bone tissue engineering. Acta Biomater. 2009, 5, 2884–2893. [Google Scholar] [CrossRef] [PubMed]
- Bashur, C.A.; Dahlgren, L.A.; Goldstein, A.S. Effect of fiber diameter and orientation on fibroblast morphology and proliferation on electrospun poly(D,L-lactic-co-glycolic acid) meshes. Biomaterials 2006, 27, 5681–5688. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Laurencin, C.T.; Caterson, E.J.; Tuan, R.S.; Ko, F.K. Electrospun nanofibrous structure: A novel scaffold for tissue engineering. J. Biomed. Mater. Res. 2002, 60, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Shih, Y.V.; Chen, C.; Tsai, S.; Wang, Y.J.; Lee, O.K. Growth of mesenchymal stem cells on electrospun type I collagen nanofibers. Stem Cells 2006, 24, 2391–2397. [Google Scholar] [CrossRef] [PubMed]
- Ekaputra, A.K.; Zhou, Y.; Cool, S.M.; Hutmacher, D.W. Composite electrospun scaffolds for engineering tubular bone grafts. Tissue Eng. Part A 2009, 15, 3779–3788. [Google Scholar] [CrossRef] [PubMed]
- Yoshimoto, H.; Shin, Y.; Terai, H.; Vacanti, J. A biodegradable nanofiber scaffold by electrospinning and its potential for bone tissue engineering. Biomaterials 2003, 24, 2077–2082. [Google Scholar] [CrossRef]
- Ji, W.; Yang, F.; Ma, J.; Bouma, M.J.; Boerman, O.C.; Chen, Z.; van den Beucken, J.J.; Jansen, J.A. Incorporation of stromal cell-derived factor-1α in PCL/gelatin electrospun membranes for guided bone regeneration. Biomaterials 2013, 34, 735–745. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ouyang, H.; Lim, C.T.; Ramakrishna, S.; Huang, Z. Electrospinning of gelatin fibers and gelatin/PCL composite fibrous scaffolds. J. Biomed. Mater. Res. Part B Appl. Biomater. 2005, 72, 156–165. [Google Scholar] [CrossRef] [PubMed]
- Fang, R.; Zhang, E.; Xu, L.; Wei, S. Electrospun PCL/PLA/HA based nanofibers as scaffold for osteoblast-like cells. J. Nanosci. Nanotechnol. 2010, 10, 7747–7751. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, P.; Catledge, S.A.; Stanishevsky, A.; Thomas, V.; Vohra, Y. Nanomechanical properties of electrospun composite scaffolds based on polycaprolactone and hydroxyapatite. J. Nanosci. Nanotechnol. 2009, 9, 4839–4845. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, R.; Prabaharan, M.; Nair, S.; Tamura, H. Novel chitin and chitosan nanofibers in biomedical applications. Biotechnol. Adv. 2010, 28, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Zafar, M.S.; Belton, D.J.; Hanby, B.; Kaplan, D.L.; Perry, C.C. Functional Material Features of Bombyx Mori Silk Light vs. Heavy Chain Proteins. Biomacromolecules 2015, 16, 606–614. [Google Scholar] [CrossRef] [PubMed]
- Zafar, M.S.; Al-Samadani, K.H. Potential use of natural silk for bio-dental applications. J. Taibah Univ. Med. Sci. 2014, 9, 171–177. [Google Scholar] [CrossRef]
- Marelli, B.; Alessandrino, A.; Fare, S.; Freddi, G.; Mantovani, D.; Tanzi, M.C. Compliant electrospun silk fibroin tubes for small vessel bypass grafting. Acta Biomater. 2010, 6, 4019–4026. [Google Scholar] [CrossRef] [PubMed]
- Ki, C.S.; Kim, J.W.; Hyun, J.H.; Lee, K.H.; Hattori, M.; Rah, D.K.; Park, Y.H. Electrospun three-dimensional silk fibroin nanofibrous scaffold. J. Appl. Polym. Sci. 2007, 106, 3922–3928. [Google Scholar] [CrossRef]
- Qasim, S.B.; Delaine-Smith, R.M.; Fey, T.; Rawlinson, A.; Rehman, I.U. Freeze gelated porous membranes for periodontal tissue regeneration. Acta Biomater. 2015, 23, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Bottino, M.C.; Thomas, V.; Janowski, G.M. A novel spatially designed and functionally graded electrospun membrane for periodontal regeneration. Acta Biomater. 2011, 7, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Bottino, M.C.; Arthur, R.A.; Waeiss, R.A.; Kamocki, K.; Gregson, K.S.; Gregory, R.L. Biodegradable nanofibrous drug delivery systems: Effects of metronidazole and ciprofloxacin on periodontopathogens and commensal oral bacteria. Clin. Oral Investig. 2014, 18, 2151–2158. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, M.N.; Sowmya, S.; Deepthi, S.; Bumgardener, J.D.; Jayakumar, R. Bilayered construct for simultaneous regeneration of alveolar bone and periodontal ligament. J. Biomed. Mater. Res. Part B Appl. Biomater. 2015, 7. [Google Scholar] [CrossRef] [PubMed]
- Ullah, R.; Zafar, M.S. Oral and dental delivery of fluoride: A review. Fluoride 2015, 48, 195–204. [Google Scholar]
- Zafar, M.S.; Ahmed, N. Therapeutic roles of fluoride released from restorative dental materials. Fluoride 2015, 48, 184–194. [Google Scholar]
- Davies, G.M.; Bridgman, C.; Hough, D.; Davies, R. The application of fluoride varnish in the prevention and control of dental caries. Dent. Update 2009, 36, 410–412. [Google Scholar] [PubMed]
- Ismail, A.I.; Hasson, H. Fluoride supplements, dental caries and fluorosis: A systematic review. J. Am. Dent. Assoc. 2008, 139, 1457–1468. [Google Scholar] [CrossRef] [PubMed]
- Zafar, M.S. Effects of Surface Pre-Reacted Glass Particles on Fluoride Release of Dental Restorative Materials. World Appl. Sci. J. 2013, 28, 457–462. [Google Scholar]
- Dionysopoulos, D. The effect of fluoride-releasing restorative materials on inhibition of secondary caries formation. Fluoride 2014, 47, 258–265. [Google Scholar]
- Khurshid, Z.; Zafar, M.; Qasim, S.; Shahab, S.; Naseem, M.; AbuReqaiba, A. Advances in Nanotechnology for Restorative Dentistry. Materials 2015, 8, 717–731. [Google Scholar] [CrossRef]
- Najeeb, S.; Khurshid, Z.; Matinlinna, J.P.; Siddiqui, F.; Nassani, M.Z.; Baroudi, K. Nanomodified Peek Dental Implants: Bioactive Composites and Surface Modification—A Review. Int. J. Dent. 2015, 2015, 381759. [Google Scholar] [CrossRef] [PubMed]
- Najeeb, S.; Zafar, M.S.; Khurshid, Z.; Siddiqui, F. Applications of polyetheretherketone (PEEK) in oral implantology and prosthodontics. J. Prosthodont. Res. 2016, 60, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Albrektsson, T.; Brånemark, P.; Hansson, H.; Lindström, J. Osseointegrated titanium implants: Requirements for ensuring a long-lasting, direct bone-to-implant anchorage in man. Acta Orthop. 1981, 52, 155–170. [Google Scholar] [CrossRef]
- Brånemark, P.; Breine, U.; Adell, R.; Hansson, B.; Lindström, J.; Ohlsson, Å. Intra-osseous anchorage of dental prostheses: I. experimental studies. Scand. J. Plast. Reconstr. Surg. Hand Surg. 1969, 3, 81–100. [Google Scholar] [CrossRef]
- Cook, S.D.; Kay, J.F.; Thomas, K.A.; Jarcho, M. Interface mechanics and histology of titanium and hydroxylapatite-coated titanium for dental implant applications. Int. J. Oral Maxillofac. Implant. 1987, 2, 15–22. [Google Scholar]
- Jovanovic, S.A.; Spiekermann, H.; Richter, E.J. Bone regeneration around titanium dental implants in dehisced defect sites: A clinical study. Int. J. Oral Maxillofac. Implant. 1992, 7, 233–245. [Google Scholar]
- Berner, S.; Dard, M.; Gottlow, J.; Molenberg, A.; Wieland, M. Titanium-zirconium: A novel material for dental implants. Eur. Cells Mater. 2009, 17, 189–205. [Google Scholar]
- Gottlow, J.; Dard, M.; Kjellson, F.; Obrecht, M.; Sennerby, L. Evaluation of a new titanium-zirconium dental implant: A biomechanical and histological comparative study in the mini pig. Clin. Implant Dent. Relat. Res. 2012, 14, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Zembic, A.; Philipp, A.O.H.; Hämmerle, C.H.F.; Wohlwend, A.; Sailer, I. Eleven-Year Follow-Up of a Prospective Study of Zirconia Implant Abutments Supporting Single All-Ceramic Crowns in Anterior and Premolar Regions. Clin. Implant Dent. Relat. Res. 2015, 17, e417–e426. [Google Scholar] [CrossRef] [PubMed]
- Javed, F.; Vohra, F.; Zafar, S.; Almas, K. Significance of Osteogenic Surface Coatings on Implants to Enhance Osseointegration Under Osteoporotic-like Conditions. Implant Dent. 2014, 23, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Le Guéhennec, L.; Soueidan, A.; Layrolle, P.; Amouriq, Y. Surface treatments of titanium dental implants for rapid osseointegration. Dent. Mater. 2007, 23, 844–854. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, S.L. Eight-year clinical retrospective study of titanium plasma-sprayed and hydroxyapatite-coated cylinder implants. Int. J. Oral Maxillofac. Implant. 1996, 11, 340–350. [Google Scholar] [CrossRef]
- Sakka, S.; Coulthard, P. Implant failure: Etiology and complications. Med. Oral Patol. Oral Cir. Bucal 2011, 16, 42–44. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Mochalin, V.N.; Neitzel, I.; Knoke, I.Y.; Han, J.; Klug, C.A.; Zhou, J.G.; Lelkes, P.I.; Gogotsi, Y. Fluorescent PLLA-nanodiamond composites for bone tissue engineering. Biomaterials 2011, 32, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Mochalin, V.N.; Shenderova, O.; Ho, D.; Gogotsi, Y. The properties and applications of nanodiamonds. Nat. Nanotechnol. 2012, 7, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Passeri, D.; Rinaldi, F.; Ingallina, C.; Carafa, M.; Rossi, M.; Terranova, M.; Marianecci, C. Biomedical Applications of Nanodiamonds: An Overview. J. Nanosci. Nanotechnol. 2015, 15, 972–988. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, A.; Martins, A.; Pinho, E.D.; Faria, S.; Reis, R.L.; Neves, N.M. Solving cell infiltration limitations of electrospun nanofiber meshes for tissue engineering applications. Nanomedicine 2010, 5, 539–554. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.; Araújo, J.V.; Reis, R.L.; Neves, N.M. Electrospun nanostructured scaffolds for tissue engineering applications. Nanomedicine. 2007, 2, 929–942. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Liu, Y.; Xu, L.; Yu, J.; Sun, G. BioMimic fabrication of electrospun nanofibers with high-throughput. Chaos Solitons Fractals 2008, 37, 643–651. [Google Scholar] [CrossRef]
- Lee, O.J.; Ju, H.W.; Kim, J.H.; Lee, J.M.; Ki, C.S.; Kim, J.; Moon, B.M.; Park, H.J.; Sheikh, F.A.; Park, C.H. Development of artificial dermis using 3D electrospun silk fibroin nanofiber matrix. J. Biomed. Nanotechnol. 2014, 10, 1294–1303. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Cai, N.; Zhao, D.; Xu, J.; Dai, Q.; Xue, Y.; Luo, X.; Yang, Y.; Yu, F. Mechanical reinforcement of electrospun water-soluble polymer nanofibers using nanodiamonds. Polym. Compos. 2013, 34, 1735–1744. [Google Scholar] [CrossRef]
- Thompson, C.; Chase, G.; Yarin, A.; Reneker, D. Effects of parameters on nanofiber diameter determined from electrospinning model. Polymer 2007, 48, 6913–6922. [Google Scholar] [CrossRef]
- Tamimi, E.; Ardila, D.; Haskett, D.; Doetschman, T.; Slepian, M.; Kellar, R.; Geest, J.V. Biomechanical Comparison of Glutaraldehyde-Crosslinked Gelatin Fibrinogen Electrospun Scaffolds to Porcine Coronary Arteries. J. Biomech. Eng. 2016, 138, 011001. [Google Scholar] [CrossRef] [PubMed]
- Frohbergh, M.E.; Katsman, A.; Botta, G.P.; Lazarovici, P.; Schauer, C.L.; Wegst, U.G.; Lelkes, P.I. Electrospun hydroxyapatite-containing chitosan nanofibers crosslinked with genipin for bone tissue engineering. Biomaterials 2012, 33, 9167–9178. [Google Scholar] [CrossRef] [PubMed]
- De Vrieze, S.; Van Camp, T.; Nelvig, A.; Hagström, B.; Westbroek, P.; De Clerck, K. The effect of temperature and humidity on electrospinning. J. Mater. Sci. 2009, 44, 1357–1362. [Google Scholar] [CrossRef]
- Zafar, M.; Khurshid, Z.; Almas, K. Oral tissue engineering progress and challenges. Tissue Eng. Regen. Med. 2015, 12, 387–397. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zafar, M.; Najeeb, S.; Khurshid, Z.; Vazirzadeh, M.; Zohaib, S.; Najeeb, B.; Sefat, F. Potential of Electrospun Nanofibers for Biomedical and Dental Applications. Materials 2016, 9, 73. https://doi.org/10.3390/ma9020073
Zafar M, Najeeb S, Khurshid Z, Vazirzadeh M, Zohaib S, Najeeb B, Sefat F. Potential of Electrospun Nanofibers for Biomedical and Dental Applications. Materials. 2016; 9(2):73. https://doi.org/10.3390/ma9020073
Chicago/Turabian StyleZafar, Muhammad, Shariq Najeeb, Zohaib Khurshid, Masoud Vazirzadeh, Sana Zohaib, Bilal Najeeb, and Farshid Sefat. 2016. "Potential of Electrospun Nanofibers for Biomedical and Dental Applications" Materials 9, no. 2: 73. https://doi.org/10.3390/ma9020073
APA StyleZafar, M., Najeeb, S., Khurshid, Z., Vazirzadeh, M., Zohaib, S., Najeeb, B., & Sefat, F. (2016). Potential of Electrospun Nanofibers for Biomedical and Dental Applications. Materials, 9(2), 73. https://doi.org/10.3390/ma9020073









