Bottom-Up Synthesis and Sensor Applications of Biomimetic Nanostructures
Abstract
:1. Introduction
2. Design and Synthesis of Biomimetic Nanostructures
2.1. DNA-Based Nanostructures

2.2. Protein-Based Nanostructures
2.3. Peptide-Based Nanostructures
2.4. Virus-Based Nanostructures
2.5. Microorganism-Based Nanostructures
2.6. Synthesis Strategies of Biomimetic Hybrid Nanomaterials
| Bionanostructures | Hybrids | Strategy | Advantages | Ref. |
|---|---|---|---|---|
| 0D | protein-NP | chemical reduction of adsorbed metal ions on/in biomolecules, or biomimetic synthesis | simple, size-controllable, economic, soluble | [64,65,66,67,77,78] |
| peptide-NP | [93,94] | |||
| virus-NP | [33] | |||
| bacterium-NP | [127,128,129] | |||
| 1D | DNA-NP | chemical reduction of adsorbed ions or direct self-assembly of as-prepared NPs | nanowire synthesis, simple, size-controllable | [58,59,60,61] |
| protein-CNT-NP | [73] | |||
| nanofiber-NP | [88,89,90] | |||
| virus-NP | [117,118,119,120,121,122,125] | |||
| bacterium-NP | [34,129,130,131,132] | |||
| 2D | DNA grid-NP | chemical reduction of adsorbed ions or direct self-assembly of as-prepared NPs | 2D scaffold synthesis, green synthesis, size-controllable, function adjustable | [49,50] |
| DNA array-NP | [51] | |||
| DNA network-NP | [62,63] | |||
| protein-graphene-NP | [72,74] | |||
| nanofiber-graphene-NP | [32,113,114] | |||
| 3D | DNA-NP | biomimetic mineralization | green synthesis, controllable | [54,55,56] |
| nanofiber scaffold-NP | [91] | |||
| virus-NP | [33] |
3. Sensor Applications of Biomimetic Nanostructures
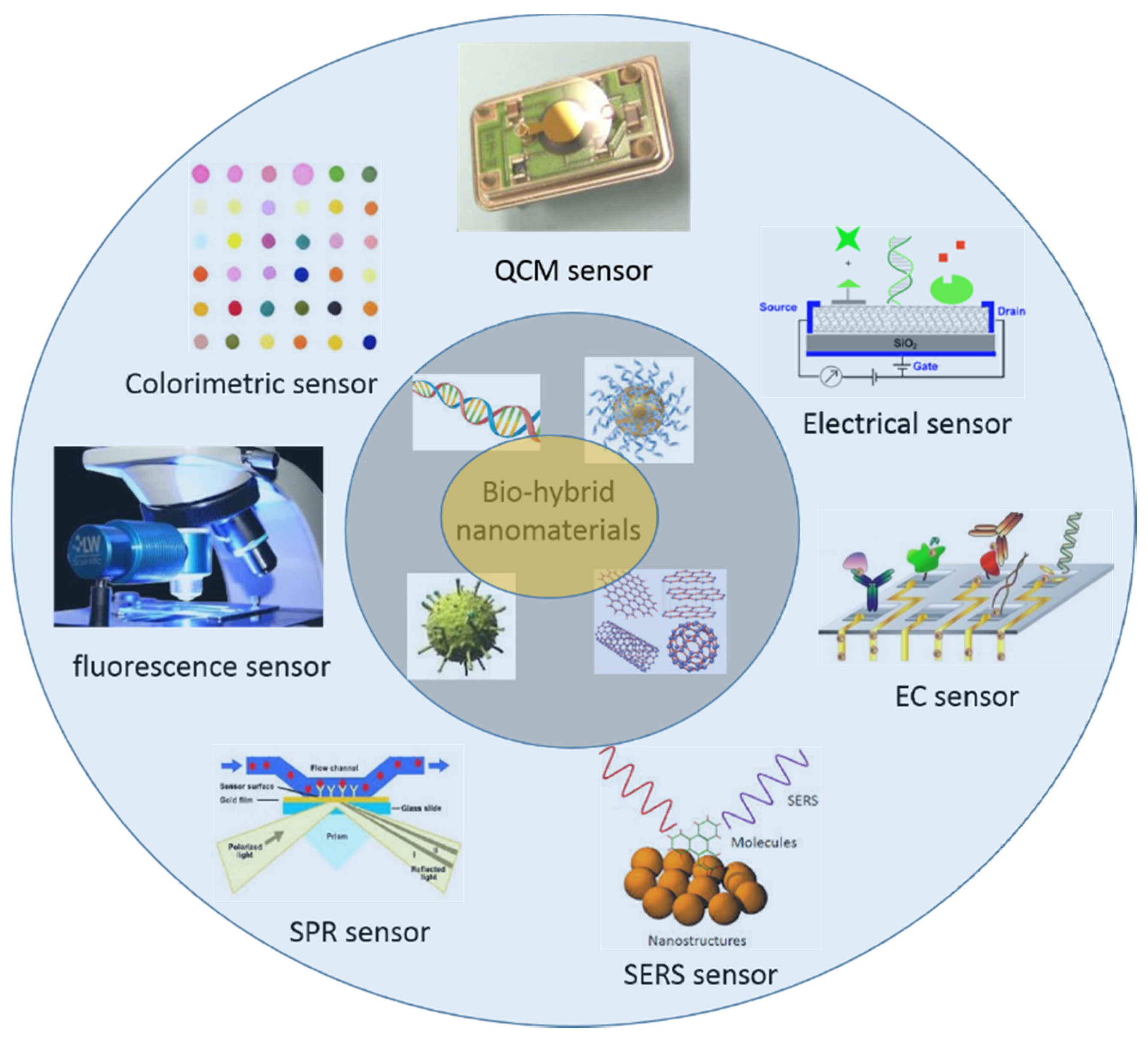
3.1. Colorimetric Sensing
3.2. Fluorescence Sensing

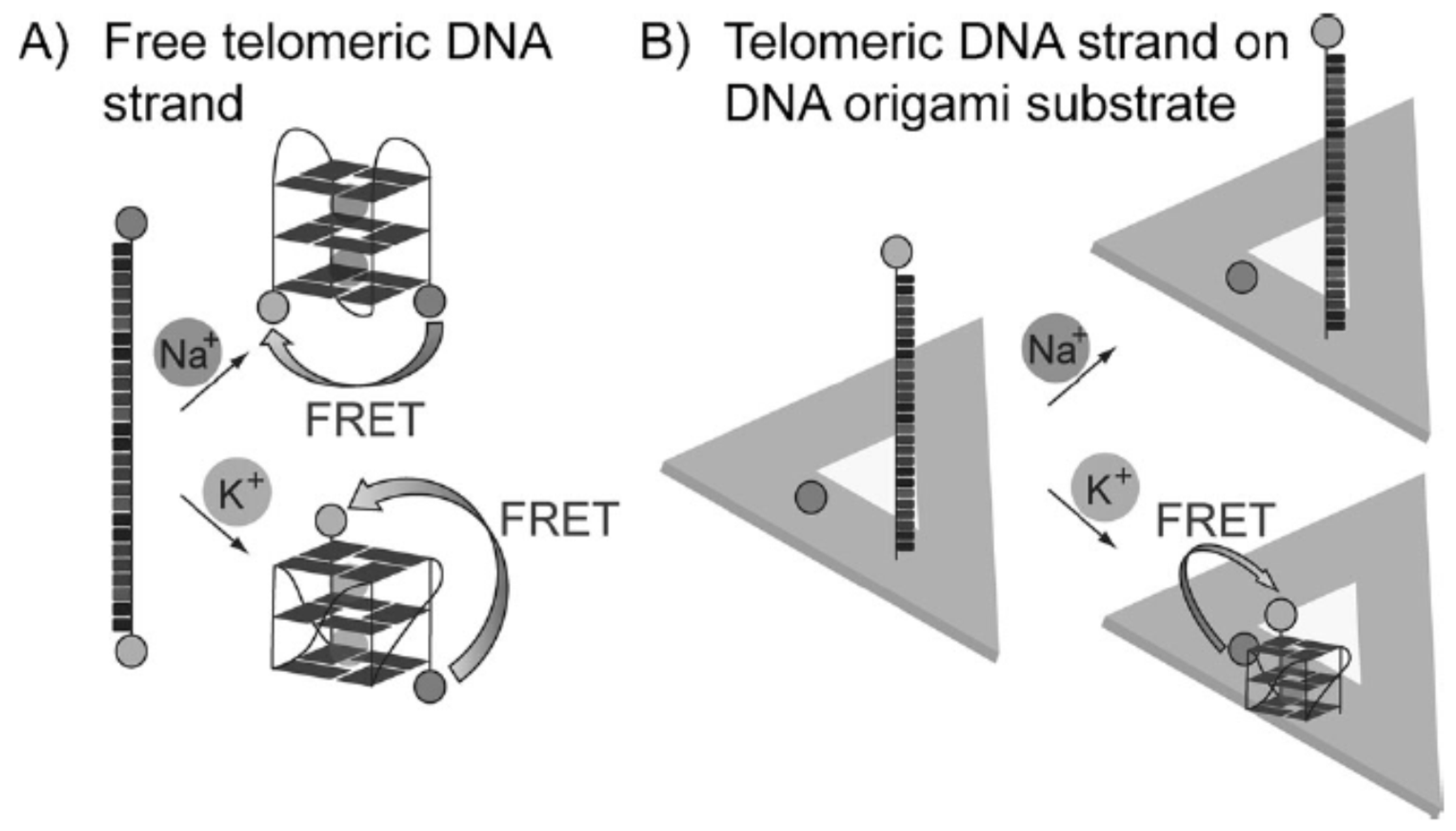
3.3. SPR Sensing
3.4. SERS Sensing
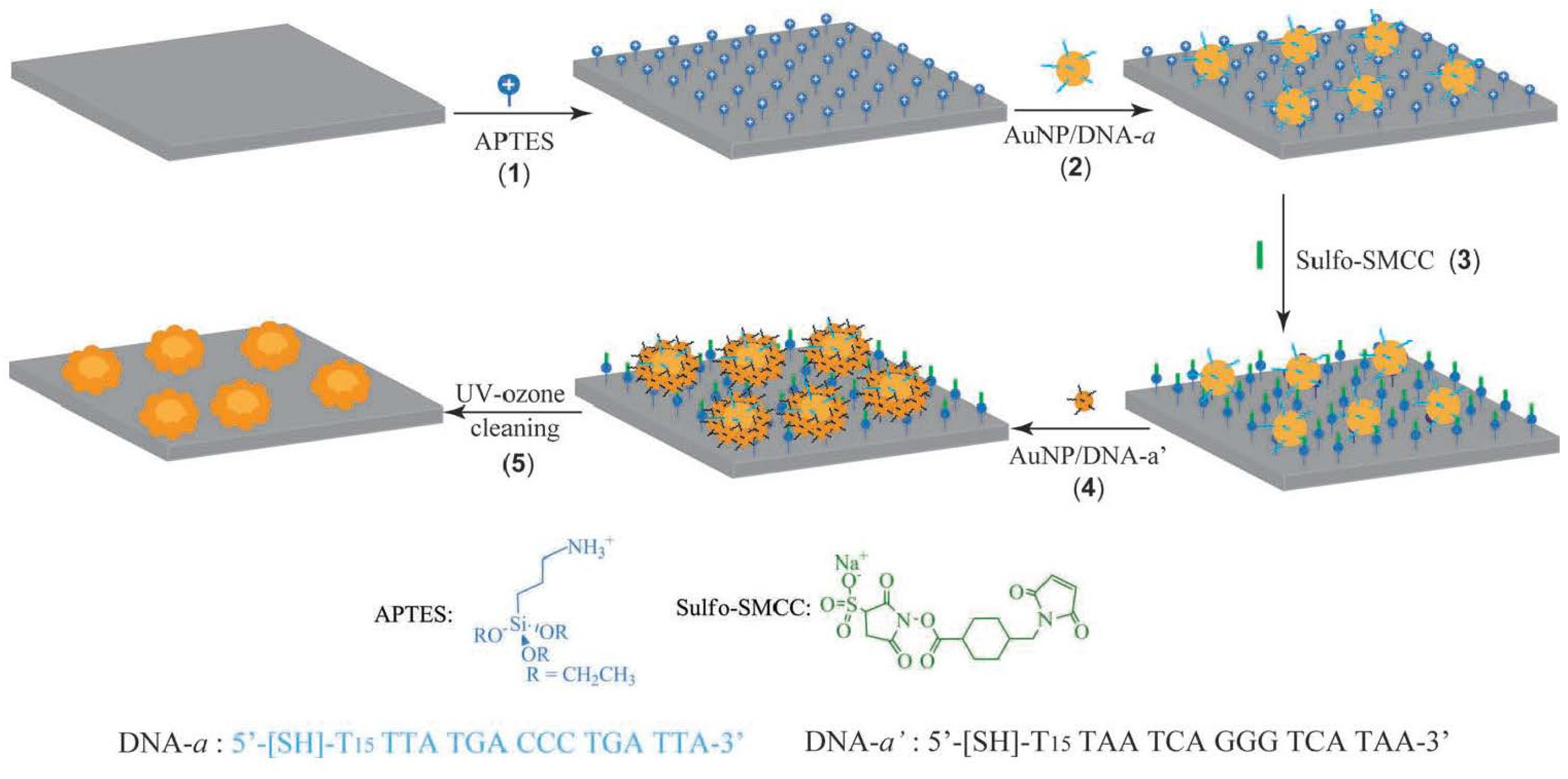
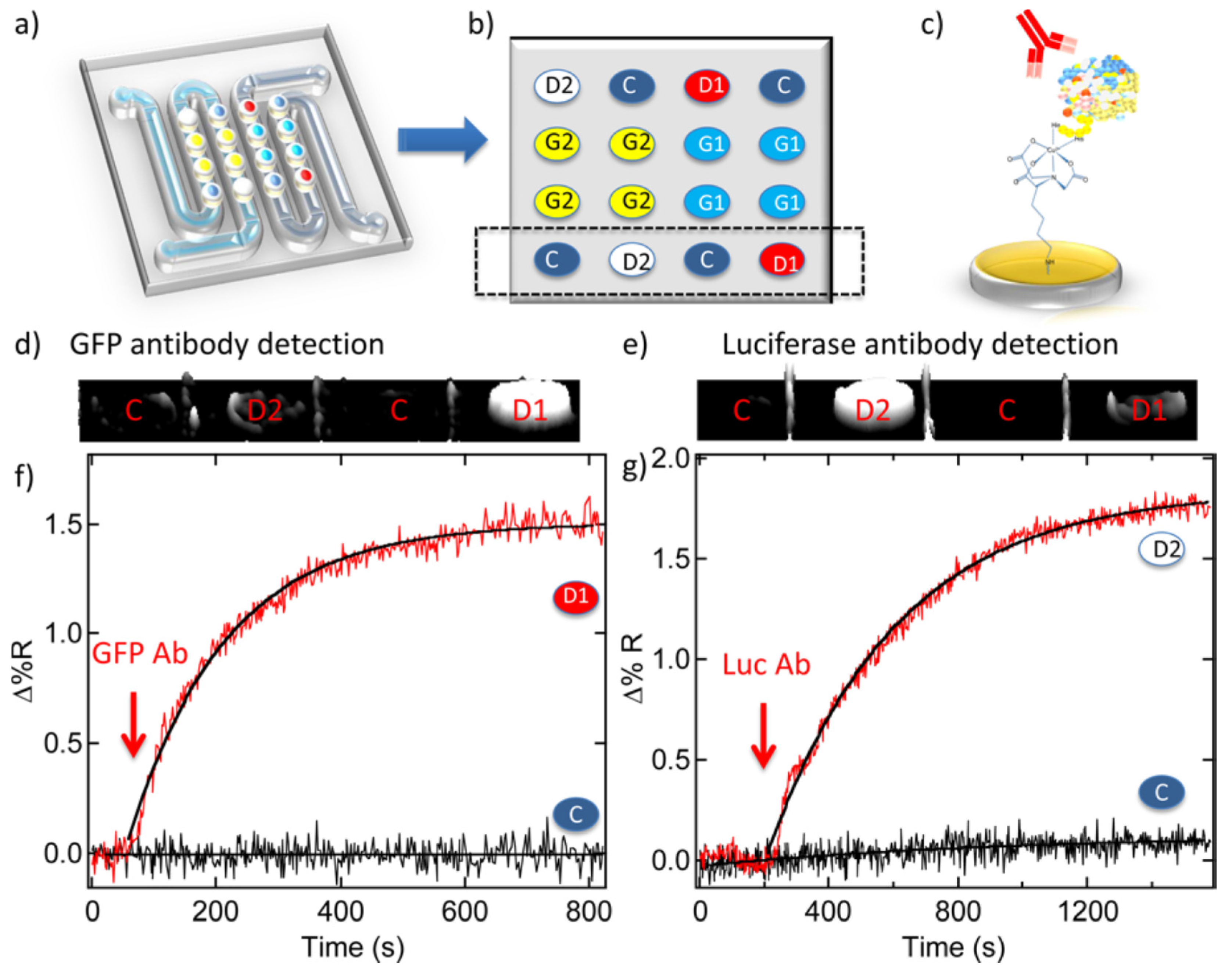
3.5. EC Sensing
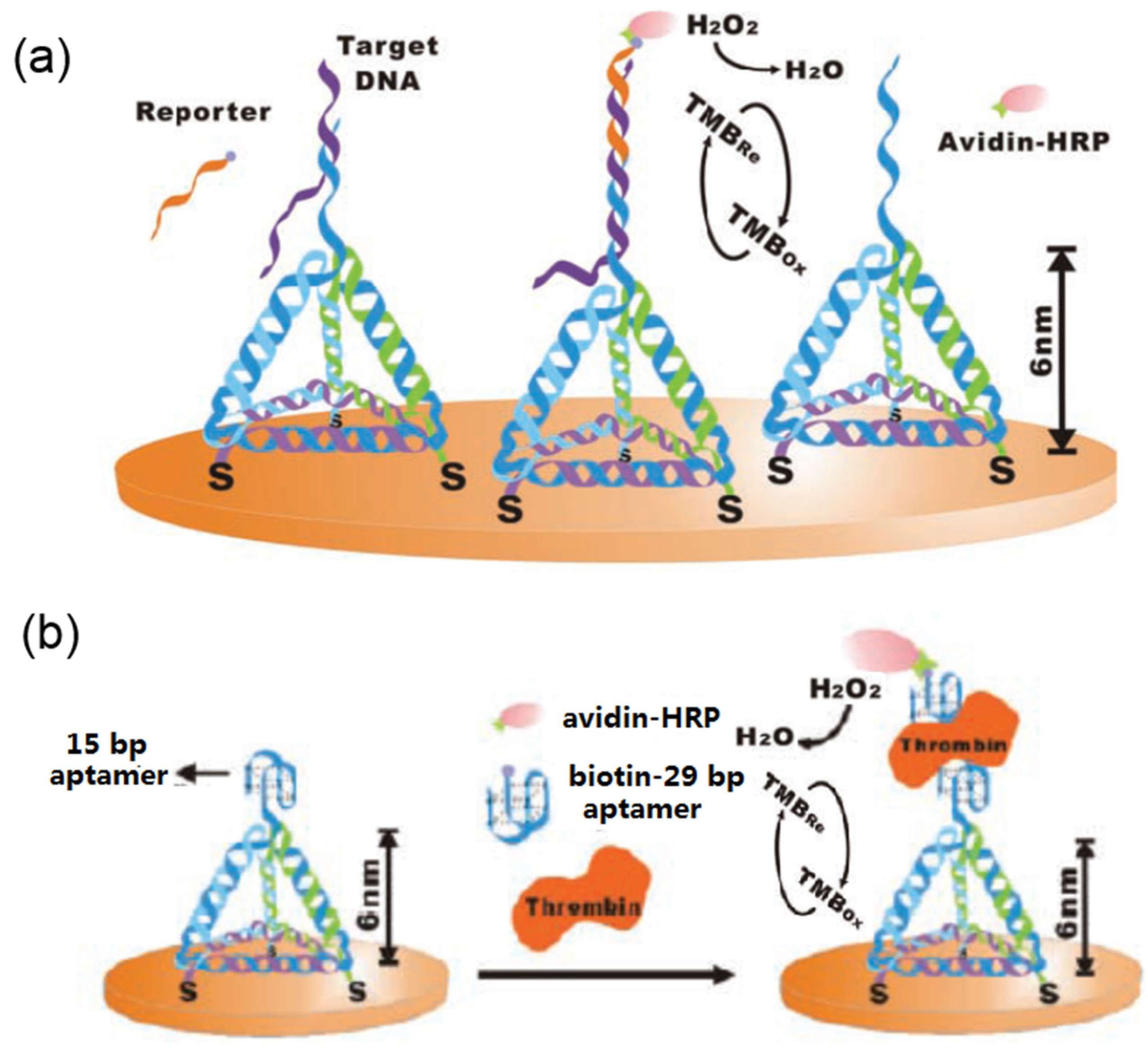
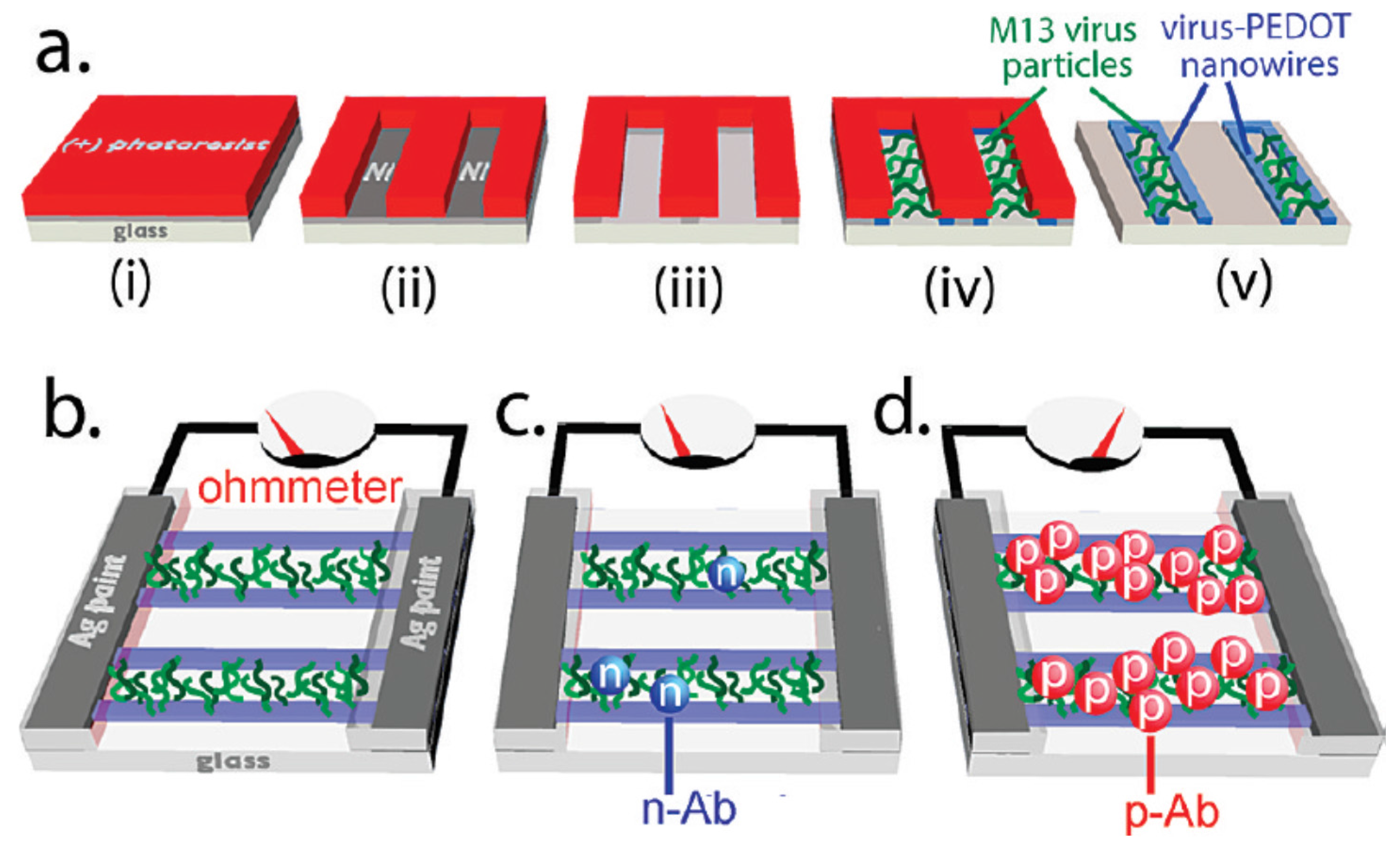
3.6. Electrical Sensing
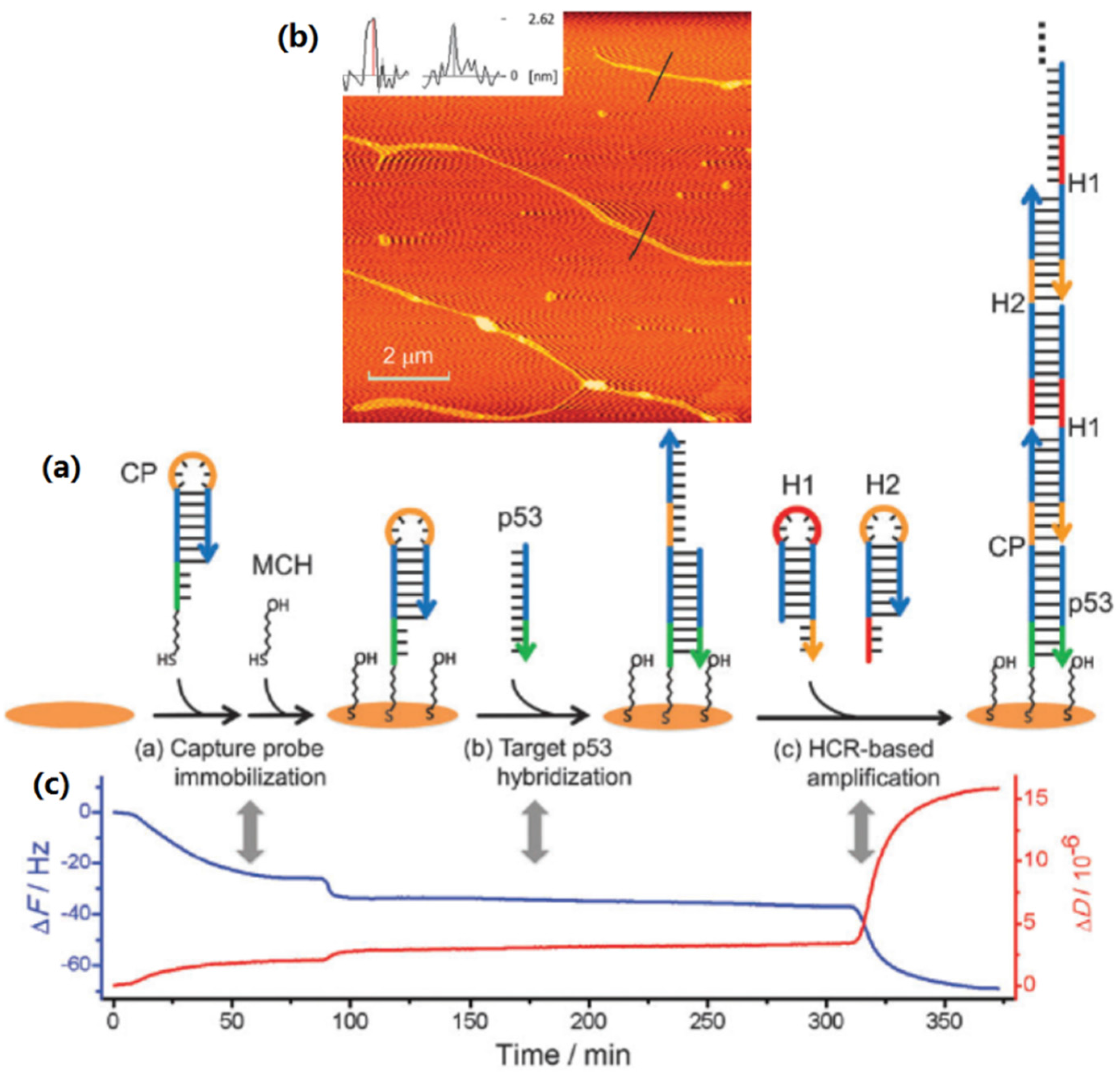
3.7. QCM Sensing
4. Conclusions and Outlooks
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Biswas, A.; Bayer, I.S.; Biris, A.S.; Wang, T.; Dervishi, E.; Faupel, F. Advances in top-down and bottom-up surface nanofabrication: Techniques, applications & future prospects. Adv. Colloid Interface Sci. 2012, 170, 2–27. [Google Scholar] [PubMed]
- Hobbs, R.G.; Petkov, N.; Holmes, J.D. Semiconductor nanowire fabrication by bottom-up and top-down paradigms. Chem. Mater. 2012, 24, 1975–1991. [Google Scholar] [CrossRef]
- Yu, H.-D.; Regulacio, M.D.; Ye, E.; Han, M.-Y. Chemical routes to top-down nanofabrication. Chem. Soc. Rev. 2013, 42, 6006–6018. [Google Scholar] [CrossRef] [PubMed]
- Gregorcayk, K.; Knez, M. Hybrid nanomaterials through molecular and atomic layer deposition: Top down, bottom up, and in-between approaches to new materials. Prog. Mater. Sci. 2016, 75, 1–37. [Google Scholar] [CrossRef]
- Piner, R.D.; Zhu, J.; Xu, F.; Hong, S.; Mirkin, C.A. Dip-Pen nanolithography. Science 1999, 283, 661–663. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-C.; Reinhoudt, D.N.; Otto, C.; Subramaniam, V.; Velders, A.H. Strategies for patterning biomolecules with dip-pen nanolithography. Small 2011, 7, 989–1002. [Google Scholar] [CrossRef] [PubMed]
- Wilbur, J.L.; Kumar, A.; Kim, E.; Whitesides, G.M. Microfabrication by microcontact printing of self-assembled monolayers. Adv. Mater. 1994, 6, 600–604. [Google Scholar] [CrossRef]
- Zankovych, Z.; Hoffmann, T.; Seekamp, J.; Bruch, J.-U.; Torres, C.M.S. Nanoimprint lithography: Challenges and prospects. Nanotechnology 2001, 12, 91–95. [Google Scholar] [CrossRef]
- Lu, W.; Lieber, C.M. Nanoelectronics from the bottom up. Nat. Mater. 2007, 6, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Freymann, G.; Kitaev, V.; Lotsch, B.V.; Ozin, G.A. Bottom-up assembly of photonic crystals. Chem. Soc. Rev. 2013, 42, 2528–2554. [Google Scholar] [CrossRef] [PubMed]
- Claridge, S.A.; Liao, W.-S.; Thomas, J.C.; Zhao, Y.; Cao, H.H.; Cheunkar, S.; Serino, A.C.; Andrews, A.M.; Weiss, P.S. From the bottom up: Dimensional control and characterization in molecular monolayers. Chem. Soc. Rev. 2013, 42, 2727–2745. [Google Scholar] [CrossRef] [PubMed]
- Seeman, N.C.; Belcher, A.M. Emulating biology: Building nanostructures from the bottom up. Proc. Natl. Acad. Sci. USA 2002, 99, 6451–6455. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Hosokawa, K.; Maeda, M. Rapid aggregation of gold nanoparticles induced by non-cross-linking DNA hybridization. J. Am. Chem. Soc. 2003, 125, 8102–8103. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.; Gao, F.; Komarneni, S. Biomolecule-assisted synthesis of highly ordered snowflakelike structure of bismuth sulfide nanorods. J. Am. Chem. Soc. 2004, 126, 54–55. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S. Fabrication of novel biomaterials through molecular self-assembly. Nat. Biotechnol. 2003, 21, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Yin, P.; Choi, H.M.T.; Calvert, C.R.; Pierce, N.A. Programming biomolecular self-assembly pathways. Nature 2008, 451, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Patil, A.J.; Muthusamy, E.; Mann, S. Synthesis and self-assembly of organoclay-wrapped biomolecules. Angew. Chem. 2004, 116, 5036–5041. [Google Scholar] [CrossRef]
- Kim, S.; Kim, J.H.; Lee, J.S.; Park, C.B. Beta-sheet-forming, self-assembled peptide nanomaterials towards optical, energy, and healthcare applications. Small 2015, 11, 3623–3640. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.; Kim, S.-W.; Kang, K.; Park, C.B. Mineralization of self-assembled peptide nanofibers for rechargeable lithium ion batteries. Adv. Mater. 2010, 22, 5537–5541. [Google Scholar] [CrossRef] [PubMed]
- Ulijn, R.V.; Smith, A.M. Designing peptide based nanomaterials. Chem. Soc. Rev. 2008, 37, 664–675. [Google Scholar] [CrossRef] [PubMed]
- Sotiropoulou, S.; Sierra-Sastre, Y.; Mark, S.S.; Batt, C.A. Biotemplated nanostructured materials. Chem. Mater. 2008, 20, 821–834. [Google Scholar] [CrossRef]
- Willner, I.; Willner, B. Biomolecule-based nanomaterials and nanostructures. Nano Lett. 2010, 10, 3805–3815. [Google Scholar] [CrossRef] [PubMed]
- Storhoff, J.J.; Mirkin, C.A. Programmed materials synthesis with DNA. Chem. Rev. 1999, 99, 1849–1862. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.-J.; Montemagno, C.D. Recent progress in advanced nanobiological materials for energy and environmental applications. Materials 2013, 6, 5821–5856. [Google Scholar] [CrossRef]
- Seeman, N.C. Nucleic acid junctions and lattices. J. Theor. Biol. 1982, 99, 237–247. [Google Scholar] [CrossRef]
- Winfree, E.; Liu, F.; Wenzler, L.A.; Seeman, N.C. Design and self-assembly of two-dimensional DNA crystals. Nature 1998, 394, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Zhang, X.; Shen, Z.; Seeman, N.C. A robust DNA mechanical device controlled by hybridization topology. Nature 2002, 415, 62–65. [Google Scholar] [CrossRef] [PubMed]
- Seeman, N.C. DNA in a material world. Nature 2003, 421, 427–431. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Liu, H.; Ribbe, A.E.; Mao, C. Self-assembly of hexagonal DNA two-dimensional arrays. J. Am. Chem. Soc. 2005, 127, 12202–12203. [Google Scholar] [CrossRef] [PubMed]
- Rothemund, P.W.K. Folding DNA to create nanoscale shapes and patterns. Nature 2006, 440, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.; Dong, X.; Sana, B.; Peng, T.; Paramelle, D.; Chen, P.; Lim, S. Ferritin-templated synthesis and self-assembly of Pt nanoparticles on a monolithic porous graphene network for electrocatalysis in fuel cells. ACS Appl. Mater. Interfaces 2013, 5, 782–787. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.; Shen, H.; Wang, H.; Wang, J.; Li, J.; Nienhaus, G.U.; Shang, L.; Wei, G. Motif-Designed Peptide Nanofibers Decorated with Graphene Quantum Dots for Simultaneous Targeting and Imaging of Tumor Cells. Adv. Funct. Mater. 2015, 25, 5472–5478. [Google Scholar] [CrossRef]
- Fontana, J.; Dressick, W.J.; Phelps, J.; Johnson, J.E.; Rendell, R.W.; Sampson, T.; Ratna, B.R.; Soto, C.M. Virus-templated plasmonic nanoclusters with icosahedral symmetry via directed self-assembly. Small 2014, 10, 3058–3063. [Google Scholar] [CrossRef] [PubMed]
- Berry, V.; Saraf, R.F. Self-assembly of nanoparticles on live bacterium: An avenue to fabricate electronic devices. Angew. Chem. 2005, 117, 6826–6831. [Google Scholar] [CrossRef]
- Han, D.; Pal, S.; Nangreave, J.; Deng, Z.; Liu, Y.; Yan, H. DNA origami with complex curvatures in three-dimensional space. Science 2011, 332, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Yang, Y.; Jiang, S.; Yan, H.; Liu, Y. Controlled nucleation and growth of DNA tile arrays within prescribed DNA origami frames and their dynamics. J. Am. Chem. Soc. 2014, 136, 3724–3727. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Jiang, S.; Samanta, A.; Liu, Y.; Yan, H. Unidirectional scaffold-strand arrangement in DNA origami. Angew. Chem. Int. Ed. 2013, 52, 9031–9034. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Pei, H.; Zhu, B.; Liang, L.; Wei, M.; He, H.; Chen, N.; Li, D.; Huang, Q.; Fan, C. Self-assembled multivalent DNA nanostructures for noninvasive intracellular delivery of immunostimulatory CpG oligonucleotides. ACS Nano 2011, 5, 8783–8789. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Zeng, D.; Chao, J.; Jin, Y.; Zhang, Z.; Liu, H.; Li, D.; Ma, H.; Huang, Q.; Gothelf, K.V.; Fan, C. Single-step rapid assembly of DNA origami nanostructures for addressable nanoscale bioreactors. J. Am. Chem. Soc. 2013, 135, 696–702. [Google Scholar] [CrossRef] [PubMed]
- Pei, H.; Zuo, X.; Zhu, D.; Huang, Q.; Fan, C. Functional DNA nanostructures for theranostic applications. Acc. Chem. Res. 2014, 47, 550–559. [Google Scholar] [CrossRef] [PubMed]
- Douglas, S.M.; Dietz, H.; Liedl, T.; Högberg, B.; Graf, F.; Shih, W.M. Self-assembly of DNA into nanoscale three-dimensional shapes. Nature 2009, 459, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Ke, Y.; Ong, L.L.; Shih, W.M.; Yin, P. Three-dimensional structures self-assembled from DNA bricks. Science 2012, 338, 1177–1183. [Google Scholar] [CrossRef] [PubMed]
- Yurke, B.; Turberfield, A.J.; Mills, A.P.; Simmel, F.C.; Neumann, J.L. A DNA-fuelled molecular machine made of DNA. Nature 2000, 406, 605–608. [Google Scholar] [PubMed]
- McLaughlin, C.K.; Hamblin, G.D.; Sleiman, H.F. Supramolecular DNA assembly. Chem. Soc. Rev. 2011, 40, 5647–5656. [Google Scholar] [CrossRef] [PubMed]
- Willner, O.I.; Willner, I. Functionalized DNA nanostructures. Chem. Rev. 2012, 112, 2528–2556. [Google Scholar] [CrossRef] [PubMed]
- Linko, V.; Dietz, H. The enabled state of DNA nanotechnology. Curr. Opin. Biotechnol. 2013, 24, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Nangreave, J.; Liu, Y.; Yan, H. Structural DNA nanotechnology: State of the art and future perspective. J. Am. Chem. Soc. 2014, 136, 11198–11211. [Google Scholar] [CrossRef] [PubMed]
- Geary, C.; Rothemund, P.W.K.; Andersen, E.S. A single-stranded architecture for cotranscriptional folding of RNA nanostructures. Science 2014, 345, 799–804. [Google Scholar] [CrossRef] [PubMed]
- Pinto, Y.Y.; Le, J.D.; Seeman, N.C.; Musier-Forsyth, K.; Taton, T.A.; Kiehl, R.A. Sequence-encoded self-assembly of multiple-nanocomponent arrays by 2D DNA scaffolding. Nano Lett. 2005, 5, 2399–2402. [Google Scholar] [CrossRef] [PubMed]
- Aldaye, F.A.; Sleiman, H.F. Sequential self-assembly of a DNA hexagon as a template for the organization of gold nanoparticles. Angew. Chem. 2006, 45, 2204–2209. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Park, S.H.; Finkelstein, G.; Reif, J.H.; LaBean, T.H. DNA-templated self-assembly of protein arrays and highly conductive nanowires. Science 2003, 301, 1882–1884. [Google Scholar] [CrossRef] [PubMed]
- Qi, H.; Ghodousi, M.; Du, Y.; Grun, C.; Bae, H.; Yin, P.; Khademhosseini, A. DNA-directed self-assembly of shape-controlled hydrogels. Nat. Commun. 2012, 4, 2275. [Google Scholar] [CrossRef] [PubMed]
- Kuzyk, A.; Schreiber, R.; Fan, Z.; Pardatscher, G.; Roller, E.M.; Högele, A.; Simmel, F.C.; Govorov, A.O.; Liedl, T. DNA-based self-assembly of chiral plasmonic nanostructures with tailored optical response. Nature 2012, 483, 311–314. [Google Scholar] [CrossRef] [PubMed]
- Helmi, S.; Ziegler, C.; Kauert, D.J.; Seidel, R. Shape-controlled synthesis of gold nanostructures using DNA origami molds. Nano Lett. 2014, 14, 6693–6698. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Boulais, E.; Hakobyan, Y.; Wang, W.L.; Guan, A.; Bathe, M.; Yin, P. Casting inorganic structures with DNA molds. Science 2014, 346, 1258361. [Google Scholar] [CrossRef] [PubMed]
- Shen, B.; Linko, V.; Tapio, K.; Kostiainen, M.A.; Toppari, J.J. Custom-shaped metal nanostructures based on DNA origami silhouettes. Nanoscale 2015, 7, 11267–11272. [Google Scholar] [CrossRef] [PubMed]
- Nakao, H.; Shigi, H.; Yamamoto, Y.; Tokonami, S.; Nagaoka, T.; Sugiyama, S.; Ohtani, T. Highly ordered assemblies of Au nanoparticles organized on DNA. Nano Lett. 2003, 3, 1391–1394. [Google Scholar] [CrossRef]
- Song, Y.; Li, Z.; Liu, Z.; Wei, G.; Wang, L.; Sun, L.; Guo, C.; Sun, Y.; Yang, T. A novel strategy to construct a flat-lying DNA monolayer on a mica surface. J. Phys. Chem. B 2006, 110, 10792–10798. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.; Wang, L.; Zhou, H.; Liu, Z.; Song, Y.; Li, Z. Electrostatic assembly of CTAB-capped silver nanoparticles along predefined λ-DNA template. Appl. Surf. Sci. 2005, 252, 1189–1196. [Google Scholar] [CrossRef]
- Wang, L.; Wei, G.; Qi, B.; Zhou, H.; Li, Z. Electrostatic assembly of Cu2O nanoparticles on DNA templates. Appl. Surf. Sci. 2006, 252, 2711–2716. [Google Scholar] [CrossRef]
- Sun, L.; Wei, G.; Song, Y.; Liu, Z.; Wang, L.; Li, Z. Fabrication of silver nanoparticles ring templated by plasmid DNA. Appl. Surf. Sci. 2006, 252, 4969–4974. [Google Scholar] [CrossRef]
- Wei, G.; Wang, L.; Liu, Z.; Song, Y.; Sun, L.; Yang, T.; Li, Z. DNA-Network-templated self-assembly of silver nanoparticles and their application in surface-enhanced Raman scattering. J. Phys. Chem. B 2005, 109, 23941–23947. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.; Zhou, H.; Liu, Z.; Song, Y.; Wang, L.; Sun, L.; Li, Z. One-step synthesis of silver nanoparticles, nanorods, and nanowires on the surface of DNA network. J. Phys. Chem. B 2005, 109, 8738–8743. [Google Scholar] [CrossRef] [PubMed]
- Meziani, M.J.; Sun, Y.P. Protein-conjugated nanoparticles from rapid expansion of supercritical fluid solution into aqueous. J. Am. Chem. Soc. 2003, 125, 8015–8018. [Google Scholar] [CrossRef] [PubMed]
- Burt, J.L.; Gutierrez-Wing, C.; Miki-Yoshida, M.; Jose-Yacaman, M. Noble-metal nanoparticles directly conjugated to globular proteins. Langmuir 2004, 20, 11778–11783. [Google Scholar] [CrossRef] [PubMed]
- Rangnekar, A.; Sarma, T.K.; Singh, A.K.; Deka, J.; Ramesh, A.; Chattopadhyay, A. Retention of enzymatic activity of α-amylase in the reductive synthesis of gold nanoparticles. Langmuir 2007, 23, 5700–5706. [Google Scholar] [CrossRef] [PubMed]
- Willner, I.; Baron, R.; Willner, B. Growing metal nanoparticles by enzymes. Adv. Mater. 2006, 18, 1109–1120. [Google Scholar] [CrossRef]
- Yang, T.; Li, Z.; Wang, L.; Guo, C.; Sun, Y. Synthesis, characterization, and self-assembly of protein lysozyme monolayer-stabilized gold nanoparticles. Langmuir 2007, 23, 10533–10538. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Zhang, Y.; Li, Z. Formation of gold nanoparticles decorated lysozyme microtubes. Biomacromolecules 2011, 12, 2027–2031. [Google Scholar] [CrossRef] [PubMed]
- Petrucci, O.D.; Buck, D.C.; Farrer, J.K.; Watt, R.K. A ferritin mediated photochemical method to synthesize biocompatible catalytically active gold nanoparticles: Size control synthesis for small, medium or large nanoparticles. RSC Adv. 2014, 4, 3472–3481. [Google Scholar] [CrossRef]
- Iwahori, K.; Yamane, M.; Fujita, S.; Yamashita, I. Synthesizing CdSe nanoparticles by using a low concentration of cadmium ions and the apoferritin protein cage of marine pennate diatoms. Mater. Lett. 2015, 160, 154–157. [Google Scholar] [CrossRef]
- Matsumoto, Y.; Chen, R.; Anikeeva, P.; Jasanoff, A. Engineering intracellular biomineralization and biosensing by a magnetic protein. Nat. Commun. 2015, 6, 8721. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, J.; Li, Z. Biomimetic synthesis of FePt nanoparticles on multi-walled carbon nanotubes for functional nanomaterials. Funct. Mater. Lett. 2013, 6, 1350006. [Google Scholar] [CrossRef]
- Wei, G.; Zhang, Y.; Steckbeck, S.; Su, Z.; Li, Z. Biomimetic graphene-FePt nanohybrids with high solubility, ferromagnetism, fluorescence, and enhanced electrocatalytic activity. J. Mater. Chem. 2012, 22, 17190–17195. [Google Scholar] [CrossRef]
- Wei, G.; Pan, C.; Reicher, J.; Jandt, K.D. Controlled assembly of protein-protected gold nanoparticles on noncovalent functionalized carbon nanotubes. Carbon 2010, 48, 645–653. [Google Scholar] [CrossRef]
- Wei, G.; Zhang, J.; Xie, L.; Jandt, K.D. Biomimetic growth of hydroxyapatite on super water-soluble carbon nanotube-protein hybrid nanofibers. Carbon 2011, 49, 2216–2226. [Google Scholar] [CrossRef]
- Wei, G.; Wang, L.; Sun, L.; Sun, Y.; Li, Z. Type I Collagen-mediated synthesis and assembly of UV-photoreduced gold nanoparticles and their application in Surface-enhanced Raman Scattering (SERS). J. Phys. Chem. C 2007, 111, 1976–1982. [Google Scholar] [CrossRef]
- Sun, Y.; Wei, G.; Song, Y.; Wang, L.; Sun, L.; Guo, C.; Yang, T.; Li, Z. Type I collagen-templated assembly of silver nanoparticles and their application in surface-enhanced Raman scattering. Nanotechnology 2008, 19, 115604. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Liu, M.; Liu, Y.; Woodbury, N.W.; Yan, H. Interenzyme substrate diffusion for an enzyme cascade organized on spatially addressable DNA nanostructures. J. Am. Chem. Soc. 2012, 134, 5516–5519. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Yang, Y.R.; Johnson-Buck, A.; Liu, M.; Walter, N.G.; Woodbury, N.W.; Yan, H. Multi-enzyme complexes on DNA scaffolds capable of substrate channeling with an artificial swinging arm. Nat. Nanotechnol. 2014, 9, 531–536. [Google Scholar] [CrossRef] [PubMed]
- Linko, V.; Eerikäinen, M.; Kostiainen, M.A. A modular DNA origami-based enzyme cascade nanoreactor. Chem. Commun. 2015, 51, 5351–5354. [Google Scholar] [CrossRef] [PubMed]
- Krebs, M.R.H.; Wilkins, D.K.; Chung, E.W.; Pitkeathly, M.C.; Chamberlain, A.K.; Zurdo, J.; Robinson, C.V.; Dobson, C.M. Formation and seeding or amyloid fibrils from wild-type hen lysozyme and a peptide fragment from the β-domain. J. Mol. Biol. 2000, 300, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Arnaudov, L.N.; de Vries, R. Thermally induced fibrillar aggregation of hen egg White lysozyme. Biophys. J. 2005, 88, 515–526. [Google Scholar] [CrossRef] [PubMed]
- Akkermans, C.; Venema, P.; van der Goot, A.J.; Gruppen, H.; Bakx, E.J.; Boom, R.; van der Linden, E. Peptides are building blocks of heat-induced fibrillary protein aggregated of β-lactoglobulin formed at pH 2. Biomacromolecules 2008, 9, 1474–1479. [Google Scholar] [CrossRef] [PubMed]
- Goda, S.; Takano, K.; Yamagata, Y.; Nagata, R.; Akutsu, H.; Maki, S.; Namba, K.; Yutani, K. Amyloid protofilament formation of hen egg lysozyme in highly concentrated ethanol solution. Protein Sci. 2000, 9, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Prausnitz, J.M.; Blanch, H.W. Amyloid fibril formation by peptide LYS (11–36) in aqueous trifluoroethanol. Biomacromolecules 2004, 5, 1818–1823. [Google Scholar] [CrossRef] [PubMed]
- Moosavi-Movahedi, A.A.; Pirzadeh, P.; Hashemnia, S.; Ahmadian, S.; Hemmateenejad, B.; Amani, M.; Saboury, A.A.; Ahmad, F.; Shamsipur, M.; Hakimelahi, G.H.; et al. Fibril formation of lysozyme upon interaction with sodium dodecyl sulfate at pH 9.2. Colloids Surf. B Biointerfaces 2007, 60, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.; Reichert, J.; Jandt, K.D. Controlled self-assembly and templated metallization of fibrinogen nanofibrils. Chem. Commun. 2008, 44, 3903–3905. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.; Reichert, J.; Bossert, J.; Jandt, K.D. Novel biopolymeric template for the nucleation and growth of hydroxyapatite crystals based on self-assembled fibrinogen fibrils. Biomacromolecules 2008, 9, 3258–3267. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.; Keller, T.F.; Zhang, J.; Jandt, K.D. Novel 1-D biophotonic hybrids: Protein nanofibers meet quantum dots. Soft Matter 2011, 7, 2011–2018. [Google Scholar] [CrossRef]
- Wang, J.; Wang, H.; Wang, Y.; Li, J.; Su, Z.; Wei, G. Alternate layer-by-layer assembly of graphene oxide nanosheets and fibrinogen nanofibers on silicon substrate for biomimetic three-dimensional hydroxyapatite scaffold. J. Mater. Chem. B 2014, 2, 7360–7368. [Google Scholar] [CrossRef]
- De la Rica, R.; Matsui, H. Applications of peptide and protein-based materials in bionanotechnology. Chem. Soc. Rev. 2010, 39, 3499–3509. [Google Scholar] [CrossRef] [PubMed]
- Gazit, E. Self-assembled peptide nanostructures: The design of molecular building blocks and their technological utilization. Chem. Soc. Rev. 2007, 36, 1263–1269. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; He, Q.; Liu, J.; Chen, Y.; Ma, M.; Zhang, L.; Shi, J. Nuclear-targeted drug delivery of TAT peptide-conjugated monodisperse mesoporous silica nanoparticles. J. Am. Chem. Soc. 2012, 134, 5722–5725. [Google Scholar] [CrossRef] [PubMed]
- Naik, R.R.; Stringer, S.J.; Agarwal, G.; Jones, S.E.; Stone, M.O. Biomimetic synthesis and patterning of silver nanoparticles. Nat. Mater. 2002, 1, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Slocik, J.M.; Stone, M.O.; Naik, R.R. Synthesis of gold nanoparticles using multifunctional peptides. Small 2005, 1, 1048–1052. [Google Scholar] [CrossRef] [PubMed]
- Graf, P.; Mantion, A.; Foelske, A.; Shkilnyy, A.; Masic, A.; Thünemann, A.F.; Taubert, A. Peptide-coated silver nanoparticles: Synthesis, surface chemistry, and pH-triggered, reversible assembly into particle assemblies. Chem. Eur. J. 2009, 15, 5831–5844. [Google Scholar] [CrossRef] [PubMed]
- Si, S.; Mandal, T.K. Tryptophan-based peptides to synthesize gold and silver nanoparticles: A mechanistic and kinetic study. Chem. Eur. J. 2007, 13, 3160–3168. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.N.; Lee, J.Y.; Wang, D.I.C. Uncovering the design rules for peptide synthesis of metal nanoparticles. J. Am. Chem. Soc. 2010, 132, 5677–5686. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.; Das, A.K.; Drew, M.G.B.; Banerjee, A. A short water-soluble self-assembling peptide forms amyloid-like fibrils. Chem. Commun. 2006, 42, 4230–4232. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Holmes, T.; Lockshin, C.; Rich, A. Spontaneous assembly of a self-complementary oligopeptide to form a stable macroscopic membrane. Proc. Natl. Acad. Sci. USA 1993, 90, 3334–3338. [Google Scholar] [CrossRef] [PubMed]
- Yokoi, H.; Kinoshita, T.; Zhang, S. Dynamic reassembly of peptide RADA16 nanofiber scaffold. Proc. Natl. Acad. Sci. USA 2005, 102, 8414–8419. [Google Scholar] [CrossRef] [PubMed]
- Kisiday, J.; Jin, M.; Kurz, B.; Huang, H.; Semino, C.; Zhang, S.; Grodzinsky, A.J. Self-assembling peptide hydrogel fosters chondrocyte extracellular matrix production and cell division: Implications for cartilage tissue repair. Proc. Natl. Acad. Sci. USA 2002, 99, 9996–10001. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Khoe, U.; Wang, X.; Horii, A.; Yokoi, H.; Zhang, S. Designer self-assembling peptide nanomaterials. Nano Today 2009, 4, 193–210. [Google Scholar]
- Burkhard, P.; Stetefekd, J.; Strelkov, S.V. Coiled coils: A highly versatile protein folding motif. Trends Cell Biol. 2001, 11, 82–88. [Google Scholar] [CrossRef]
- Papapostolou, D.; Smith, A.M.; Atkins, E.D.T.; Oliver, S.J.; Ryadnov, M.G.; Serpell, L.C.; Woolfson, D.N. Engineering nanoscale order into a designed protein fiber. Proc. Natl. Acad. Sci. USA 2007, 104, 10853–10858. [Google Scholar] [CrossRef] [PubMed]
- Hartgerink, J.D.; Beniash, E.; Stupp, S.I. Self-assembly and mineralization of peptide-amphiphile nanofibers. Science 2001, 294, 1684–1688. [Google Scholar] [CrossRef] [PubMed]
- Hartgerink, J.D.; Beniash, E.; Stupp, S.I. Peptide-amphiphile nanofibers: A versatile scaffold for the preparation of self-assembling materials. Proc. Natl. Acad. Sci. USA 2002, 99, 5133–5138. [Google Scholar] [CrossRef] [PubMed]
- Niece, K.L.; Hartgerink, J.D.; Donners, J.; Stupp, S.I. Self-assembly combining two bioactive peptide-amphiphile molecules into nanofibers by electrostatic attraction. J. Am. Chem. Soc. 2003, 125, 7146–7147. [Google Scholar] [CrossRef] [PubMed]
- Reches, M.; Gazit, E. Casting metal nanowires within discrete self-assembled peptide nanotubes. Science 2003, 300, 625–627. [Google Scholar] [CrossRef] [PubMed]
- Tomizaki, K.Y.; Kubo, S.; Ahn, S.A.; Satake, M.; Imai, T. Biomimetic alignment of zinc oxide nanoparticles along a peptide nanofiber. Langmuir 2012, 28, 13459–13466. [Google Scholar] [CrossRef] [PubMed]
- Maity, I.; Manna, M.K.; Rasale, D.B.; Das, A.K. Peptide-nanofiber-supported palladium nanoparticles as an efficient catalyst for the removal of N-terminus protecting groups. ChemPlusChem 2014, 79, 413–420. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, X.; Li, J.; Kuang, X.; Fan, Y.; Wei, G.; Su, Z. Electrostatic Assembly of Peptide Nanofiber-Biomimetic Silver Nanowires onto Graphene for Electrochemical Sensor. ACS Macro Lett. 2014, 3, 529–533. [Google Scholar] [CrossRef]
- Wang, J.; Ouyang, Z.; Li, J.; Zhang, P.; Wei, G.; Su, Z. Self-assembled peptide nanofibers on graphene oxide as a novel nanohybrid for biomimetic mineralization of hydroxyapatite. Carbon 2015, 89, 20–30. [Google Scholar] [CrossRef]
- Slocik, J.M.; Naik, R.R.; Stone, M.O.; Wright, D.W. Viral templates for gold nanoparticle synthesis. J. Mater. Chem. 2005, 15, 749–753. [Google Scholar] [CrossRef]
- Bruckman, M.A.; Steinmetz, N.F. Chemical modification of the inner and outer surfaces of tobacco mosaic virus (TMV). Methods Mol. Biol. 2014, 1108, 173–185. [Google Scholar] [PubMed]
- Shenton, W.; Douglas, T.; Yong, M.; Stubbs, G.; Mann, S. Inorganic-organic nanotube composites from template mineralization of tobacco mosaic virus. Adv. Mater. 1999, 11, 253–256. [Google Scholar] [CrossRef]
- Dujardin, E.; Peet, C.; Stubbs, G.; Culver, J.N.; Mann, S. Organization of metallic nanoparticles using tobacco mosaic virus templates. Nano Lett. 2003, 3, 413–417. [Google Scholar] [CrossRef]
- Knez, M.; Sumser, M.; Bittner, A.M.; Wege, C.; Jeske, H.; Martin, T.P.; Kern, K. Spatially selective nucleation of metal clusters on the tobacco mosaic virus. Adv. Funct. Mater. 2004, 14, 116–124. [Google Scholar]
- Knez, M.; Bittner, A.M.; Boes, F.; Wege, C.; Jeske, H.; Maiβ, E.; Kern, K. Biotemplate synthesis of 3-nm nickel and cobalt nanowires. Nano Lett. 2003, 3, 1079–1082. [Google Scholar] [CrossRef]
- Atanasova, P.; Rothenstein, D.; Schneider, J.J.; Hoffmann, R.C.; Dilfer, S.; Eiben, S.; Wege, C.; Jeske, H.; Bill, J. Virus-templated synthesis of ZnO nanostructures and formation of field-effect transistors. Adv. Mater. 2011, 23, 4918–4922. [Google Scholar] [CrossRef] [PubMed]
- Kostiainen, M.A.; Hiekkataipale, P.; de la Torre, J.A.; Nolte, R.J.M.; Cornelissen, J.L.M. Electrostatic self-assembly of virus-polymer complexes. J. Mater. Chem. 2011, 21, 2112–2117. [Google Scholar] [CrossRef]
- Douglas, T.; Yong, M. Host-guest encapsulation of materials by assembled virus protein cages. Nature 1998, 393, 152–155. [Google Scholar] [CrossRef]
- Mikkilä, J.; Eskelinen, A.P.; Niemelä, E.H.; Linko, V.; Frilander, M.J.; Törmä, P.; Konstiainen, M.A. Virus-encapsulated DNA origami nanostructures for cellular delivery. Nano Lett. 2014, 14, 2196–2200. [Google Scholar] [CrossRef] [PubMed]
- Mao, C.; Wang, F.; Cao, B. Controlling nanostructures of mesoporous silica fibers by supramolecular assembly of genetically modificable bacteriophages. Angew. Chem. Int. Ed. 2012, 124, 6517–6521. [Google Scholar] [CrossRef]
- Lee, S.Y.; Lim, J.S.; Harris, M.T. Synthesis and application of virus-based hybrid nanomaterials. Biotechnol. Bioeng. 2011, 109, 16–30. [Google Scholar] [CrossRef] [PubMed]
- Davis, S.A.; Burkett, S.L.; Mendelson, N.H.; Mann, S. Bacterial templating of ordered macrostructures in silica and silica-surfactant mesophases. Nature 1997, 385, 420–423. [Google Scholar] [CrossRef]
- Klaus, T.; Joerger, R.; Olsson, E.; Granqvist, C.G. Silver-based crystalline nanoparticles, microbially fabricated. Proc. Natl. Acad. Sci. USA 1999, 96, 13611–13614. [Google Scholar] [CrossRef] [PubMed]
- Shankar, S.S.; Rai, A.; Ahmad, A.; Sastry, M. Rapid synthesis of Au, Ag, and bimetallic Au core-Ag shell nanoparticles using Neem (Azadirachta indica) leaf broth. J. Colloid. Interface Sci. 2004, 275, 496–502. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.; Ahmad, A.; Mandal, D.; Senapati, S.; Sainkar, S.R.; Khan, M.I.; Parishcha, R.; Ajaykumar, P.V.; Alam, M.; Kumar, R.; et al. Fungus-mediated synthesis of silver nanoparticles and their immobilization in the mycelial matrix: A novel biological approach to nanoparticle synthesis. Nano Lett. 2001, 1, 515–519. [Google Scholar] [CrossRef]
- Shahverdi, A.R.; Minaeian, S.; Shahverdi, H.R.; Jamalifar, H.; Nohi, A.A. Rapid synthesis of silver nanoparticles using culture supernatants of Enterobacteria: A novel biological approach. Proc. Biochem. 2007, 42, 919–923. [Google Scholar] [CrossRef]
- Ayano, H.; Miyake, M.; Terasawa, K.; Kuroda, M.; Soda, S.; Sakaguchi, T.; Ike, M. Isolation of a selenite-reducing and cadmium-resistant bacterium Pseudomonas sp. strain RB for microbial synthesis of CdSe nanoparticles. J. Biosci. Bioeng. 2014, 117, 576–581. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Lin, L.; Sun, D.; Chen, H.; Yang, D.; Li, Q. Bio-inspired synthesis of metal nanomaterials and applications. Chem. Soc. Rev. 2015, 44, 6330–6374. [Google Scholar] [CrossRef] [PubMed]
- Elghanian, R.; Storhoff, J.J.; Mucic, R.C.; Letsinger, R.L.; Mirkin, C.A. Selective colorimetric detection of polynucleotides based on the distance-dependent optical properties of gold nanoparticles. Science 1997, 277, 1078–1081. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Han, M.S.; Mirkin, C.A. Colorimetric detection of mercuric ion (Hg2+) in aqueous media using DNA-functionalized gold nanoparticles. Angew. Chem. 2007, 119, 4171–4174. [Google Scholar] [CrossRef]
- Lee, J.S.; Ulmann, P.A.; Han, M.S.; Mirkin, C.A. A DNA-gold nanoparticle-based colorimetric competition assay for the detection of cysteine. Nano Lett. 2008, 8, 529–533. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lu, Y. A colorimetric lead biosensor using DNAzyme-directed assembly of gold nanoparticles. J. Am. Chem. Soc. 2003, 125, 6642–6643. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lu, Y. Rational design of “turn-on” allosteric DNAzyme catalytic beacons for aqueous mercury ions with ultrahigh sensitivity and selectivity. Angew. Chem. 2007, 119, 7731–7734. [Google Scholar] [CrossRef]
- Liu, J.; Lu, Y. Adenosine-dependent assembly of aptazyme-functionalized gold nanoparticles and its application as a colorimetric biosensor. Anal. Chem. 2004, 76, 1627–1632. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lu, Y. Fast colorimetric sensing of adenosine and cocaine based on a general sensor design involving aptamers and nanoparticles. Angew. Chem. 2005, 118, 96–100. [Google Scholar] [CrossRef]
- Jiang, X.; Sun, C.; Guo, Y.; Nie, G.; Xu, L. Peroxidase-like activity of apoferritin paired gold clusters for glucose detection. Biosens. Bioelectron. 2015, 64, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Chen, B.; Zhang, H.; Sun, Y.; Wang, J.; Zhang, J.; Fu, Y. BSA-stabilized Pt nanozyme for peroxidase mimetics and its application on colorimetric detection of mercury(II) ions. Biosens. Bioelectron. 2015, 66, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.-W.; Chung, W.-J.; Heo, K.; Jin, H.-E.; Lee, B.Y.; Wang, E.; Zueger, C.; Wong, W.; Meyer, J.; Kim, C.; et al. Biomimetic virus-based colorimetric sensors. Nat. Commun. 2014, 5, 3043. [Google Scholar] [CrossRef] [PubMed]
- Sacca, B.; Niemeyer, C.M. DNA origami: The art of folding DNA. Angew. Chem. Int. Ed. 2012, 51, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Ke, Y.; Lindsay, S.; Chang, Y.; Liu, Y.; Yan, H. Self-assembled water-soluble nucleic acid probe tiles for label-free RNA hybridization assays. Science 2008, 319, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Olejko, L.; Cywinski, P.J.; Bald, I. Ion-selective formation of a guanine quadruplex on DNA origami structures. Angew. Chem. Int. Ed. 2015, 54, 673–677. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.H.; Yang, H.H.; Zhu, C.L.; Chen, X.; Chen, G.N. A graphene platform for sensing biomoelcules. Angew. Chem. Int. Ed. 2009, 48, 4785–4787. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Tang, L.; Wang, Y.; Jiang, J.; Li, J. Graphene fluorescence resonance energy transfer aptasensor for the thrombin detection. Anal. Chem. 2010, 82, 2341–2346. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Zhao, X.; Ji, Y.; Ouyang, Z.; Wen, X.; Li, J.; Su, Z.; Wei, G. Electrospinning graphene quantum dots into nanofibrous polymer membrane for dual-purpose fluorescent and electrochemical biosensors. J. Mater. Chem. B 2015, 3, 2487–2496. [Google Scholar] [CrossRef]
- Xie, J.; Zheng, Y.; Ying, J.Y. Highly selective and ultrasensitive detection of Hg2+ based on fluorescence quenching of Au nanoclusters by Hg2+–Au+ interactions. Chem. Commun. 2010, 46, 961–963. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Irudayaraj, J. Fluorescent Ag clusters via a protein-directed approach as a Hg(II) ion sensor. Anal. Chem. 2011, 83, 2883–2889. [Google Scholar] [CrossRef] [PubMed]
- Patching, S.G. Surface plasmon resonance spectroscopy for characterisation of membrane protein–ligand interactions and its potential for drug discovery. Biochim. Biophys. Acta 2014, 1838, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Homola, J.; Yee, S.S.; Gauglitz, G. Surface plasmon resonance sensors: Review. Sens. Actuators B Chem. 1999, 54, 3–15. [Google Scholar] [CrossRef]
- Pelossof, G.; Tel-Vered, R.T.; Liu, X.Q.; Willner, I. Amplified surface plasmon resonance based DNA biosensors, aptasensors, and Hg2+ sensors using hemin/G-quadruplexes and Au nanoparticles. Chem. Eur. J. 2011, 17, 8904–8912. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Feng, F.; Zhao, L.; Wang, C.; Wang, H.; Tian, M.; Qin, J.; Duan, Y.; He, X. Aptamer/thrombin/aptamer-AuNPs sandwich enhanced surface plasmon resonance sensor for the detection of subnanomolar thrombin. Biosens. Bioelectron. 2013, 47, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.R.; Hau, B.Y.H.; Endo, T.; Kerman, K. Au nanoparticle-modified DNA sensor based on simultaneous electrochemical impedance spectroscopy and localized surface plasmon resonance. Biosens. Bioelectron. 2014, 53, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Seefeld, T.H.; Halpern, A.R.; Corn, R.M. On-chip synthesis of protein microarrays from DNA microarrays via coupled in vitro transcription and translation for surface plasmon resonance imaging biosensor applications. J. Am. Chem. Soc. 2012, 134, 12358–12361. [Google Scholar] [CrossRef] [PubMed]
- Peng, C.; Song, Y.; Wei, G.; Zhang, W.; Li, Z.; Dong, W. Self-assembly of λ-DNA/Ag nanoparticles: Hybrid architecture and active-SERS substrate. J. Colloid Interface Sci. 2008, 317, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Thai, T.; Reineck, P.; Qiu, L.; Guo, Y.; Bach, U. DNA-directed self-assembly of core-satellite plasmonic nanostructures: A highly sensitive and reproducible near-IR SERS sensor. Adv. Funct. Mater. 2013, 23, 1519–1526. [Google Scholar] [CrossRef]
- Sun, Y.; Xu, F.; Zhang, Y.; Shi, Y.; Wen, Z.; Li, Z. Metallic nanostructures assembled by DNA and related applications in surface-enhancement Raman scattering (SERS) detection. J. Mater. Chem. 2011, 21, 16675–16685. [Google Scholar] [CrossRef]
- Wang, L.; Sun, Y.; Cui, Y.; Wang, J.; Li, Z. Synthesis of silver nanoplates with fibronectin nanofibril template and their SERS application. Bull. Korean Chem. Soc. 2013, 34, 443–446. [Google Scholar] [CrossRef]
- Yang, D.P.; Chen, S.; Huang, P.; Wang, X.; Jiang, W.; Pandoli, O.; Cui, D. Bacteria-template synthesized silver microspheres with hollow and porous structures as excellent SERS substrate. Green Chem. 2010, 12, 2038–2042. [Google Scholar] [CrossRef]
- Sun, D.; Zhang, G.; Jiang, X.; Huang, J.; Jiang, X.; Zheng, Y.; He, J.; Li, Q. Biogenic flower-shaped Au-Pd nanoparticles: Synthesis, SERS detection and catalysis towards benzyl alcohol oxidation. J. Mater. Chem. A 2014, 2, 1767–1773. [Google Scholar] [CrossRef]
- Sun, D.; Zhang, G.; Huang, J.; Wang, H.; Li, Q. Plant-mediated fabrication and surface enhanced Raman property of flower-like Au@Pd nanoparticles. Materials 2014, 7, 1360–1369. [Google Scholar] [CrossRef]
- Pei, H.; Lu, N.; Wen, Y.; Song, S.; Liu, Y.; Yan, H.; Fan, C. A DNA nanostructure-based biomolecular probe carrier platform for electrochemical biosensing. Adv. Mater. 2010, 22, 4754–4758. [Google Scholar] [CrossRef] [PubMed]
- Wen, Y.; Pei, H.; Wan, Y.; Su, Y.; Huang, Q.; Song, S.; Fan, C. DNA nanostructure-decorated surfaces for enhanced aptamer-target binding and electrochemical cocaine sensors. Anal. Chem. 2011, 83, 7418–7423. [Google Scholar] [CrossRef] [PubMed]
- Pei, H.; Wan, Y.; Li, J.; Hu, H.; Su, Y.; Huang, Q.; Fan, C. Regenerable electrochemical immunological sensing at DNA nanostructure-decorated gold surfaces. Chem. Commun. 2011, 47, 6454–6456. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Wen, Y.; Li, L.; Pei, H.; Liu, G.; Song, H.; Zou, X.; Fan, C.; Huang, Q. Targe-responsive, DNA nanostructure-based E-DNA sensor for microRNA analysis. Anal. Chem. 2014, 86, 2285–2288. [Google Scholar] [CrossRef] [PubMed]
- Ge, Z.; Lin, M.; Wang, P.; Pei, H.; Yan, J.; Shi, J.; Huang, Q.; He, D.; Fan, C.; Zuo, X. Hybridization chain reaction amplification of microRNA detection with a tetrahedral DNA nanostructure-based electrochemical biosensor. Anal. Chem. 2014, 86, 2124–2130. [Google Scholar] [CrossRef] [PubMed]
- Mor-Piperberg, G.; Tel-Vered, R.; Elbaz, J.; Willner, I. Nanoengineered electrically contacted enzymes on DNA scaffolds: Functional assemblies for the selective analysis of Hg2+ ions. J. Am. Chem. Soc. 2010, 132, 6878–6879. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Yang, D.P.; Xu, K.; Cao, H.; Wu, B.; Cui, D.; Jia, N. Ag@BSA core/shell microspheres as an electrochemical interface for sensitive detection of urinary retinal-binding protein. Anal. Chem. 2012, 84, 10324–10331. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Yang, D.P.; Wang, Z.; Yu, L.; Zhang, J.; Jia, N. Improved EIS performance of an electrochemical cytosensor using three-dimensional architecture Au@BSA as sensing layer. Anal. Chem. 2013, 85, 5200–5206. [Google Scholar] [CrossRef] [PubMed]
- Wei, G.; Xu, F.; Li, Z.; Jandt, K.D. Protein-promoted synthesis of Pt nanoparticles on carbon nanotubes for electrocatalytic nanohybrids with enhanced glucose sensing. J. Phys. Chem. C 2011, 115, 11453–11460. [Google Scholar] [CrossRef]
- Wang, L.; Ni, P.; Wei, G.; Wang, J.; Li, Z. Collagen nanofiber-templated silver nanowires on graphene nanosheets for nonenzymatic amperometric biosensor of hydrogen peroxide. Chem. Lett. 2014, 43, 544–546. [Google Scholar] [CrossRef]
- Park, S.J.; Taton, T.A.; Mirkin, C.A. Array-based electrical detection of DNA with nanoparticle probes. Science 2002, 295, 1503–1506. [Google Scholar] [PubMed]
- Porath, D.; Bezryadin, A.; de Vries, S.; Dekker, C. Direct measurement of electrical transport through DNA molecules. Nature 2000, 403, 635–638. [Google Scholar] [CrossRef] [PubMed]
- Linko, V.; Paasonen, S.T.; Kuzyk, A.; Torma, P.; Toppari, J.J. Characterization of the conductance mechanisms of DNA origami by AC impedance spectroscopy. Small 2009, 5, 2382–2386. [Google Scholar] [CrossRef] [PubMed]
- Plesa, C.; Ananth, A.N.; Linko, V.; Gulcher, C.; Katan, A.J.; Dietz, H.; Dekker, C. Ionic permeability and mechanical properties of DNA origami nanoplates on solid-state nanopores. ACS Nano 2014, 8, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Livshits, G.I.; Stern, A.; Rotem, D.; Borovok, N.; Eidelshtein, G.; Migliore, A.; Penzo, E.; Wind, S.J.; Felice, R.D.; Skourtis, S.S.; et al. Long-range charge transport in single G-quadruplex DNA molecules. Nat. Nanotechnol. 2014, 9, 1040–1046. [Google Scholar] [CrossRef] [PubMed]
- Bell, N.A.W.; Engst, C.R.; Ablay, M.; Divitini, G.; Ducati, C.; Liedl, T.; Keyser, U.F. DNA origami nanopores. Nano Lett. 2012, 12, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Wei, R.; Martin, T.G.; Rant, U.; Dietz, H. DNA origami gatekeepers for solid-state nanopores. Angew. Chem. Int. Ed. 2012, 51, 4864–4867. [Google Scholar] [CrossRef] [PubMed]
- Arter, J.A.; Taggart, D.K.; McIntire, T.M.; Penner, R.M.; Weiss, G.A. Virus-PEDOT nanowires for biosensing. Nano Lett. 2010, 10, 4858–4862. [Google Scholar] [CrossRef] [PubMed]
- Moon, C.H.; Zhang, M.; Myung, N.V.; Haberer, E.D. Highly sensitive hydrogen sulfide (H2S) gas sensors from viral-templated nanocrystalline gold nanowires. Nanotechnology 2014, 25, 135205. [Google Scholar] [CrossRef] [PubMed]
- Moon, C.H.; Yan, Y.; Zhang, M.; Myung, N.V.; Haberer, E.D. Viral-templated nanocrystalline Pd nanowires for chemiresistive hydrogen (H2) sensors. SPIE NanoSci. Eng. 2014, 9171, 91710U. [Google Scholar]
- Keren, K.; Berman, R.S.; Buchstab, E.; Sivan, U.; Braun, E. DNA-templated carbon nanotube field-effect transistor. Science 2003, 302, 1380–1382. [Google Scholar] [CrossRef] [PubMed]
- Maune, H.T.; Han, S.P.; Barish, R.D.; Bockrath, M.; Goddard III, W.A.; Rothemund, P.W.K.; Winfree, E. Self-assembly of carbon naotubes into two-dimensional geometries using DNA origami templates. Nat. Nanotechnol. 2009, 8, 61–66. [Google Scholar]
- Prakrankamanant, P.; Leelayuwat, C.; Promptmas, C.; Limpaiboon, T.; Wanram, S.; Prasongdee, P.; Pientong, C.; Daduang, J.; Jearanalkoon, P. The development of DNA-based quartz crystal microbalance integrated with isothermal DNA amplification system for human papillomavirus type 58 detection. Biosens. Bioelectron. 2013, 40, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.I.; Chang, Y.P.; Chu, Y.H. Biomolecular interactions and tools for their recognition: Focus on the quartz crystal microbalance and its diverse surface chemistries and applications. Chem. Soc. Rev. 2012, 41, 1947–1971. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Wang, D.; Xu, Y.; Li, N.; Liu, F. A self-assembled DNA nanostructure-amplified quartz crystal microbalance with dissipation biosensing platform for nucleic acids. Chem. Commun. 2012, 48, 6678–6680. [Google Scholar] [CrossRef] [PubMed]
- Altintas, Z.; Tothill, I.E. DNA-based biosensor platforms for the detection of TP53 mutation. Sens. Actuators B Chem. 2012, 169, 188–194. [Google Scholar] [CrossRef]
- Hao, R.Z.; Song, H.B.; Zuo, G.M.; Yang, R.F.; Wei, H.P.; Wang, D.B.; Cui, Z.Q.; Cheng, Z.X.; Zhang, X.E. DNA probe functionalized QCM biosensor based on gold nanoparticle amplification for bacillus anthracis detection. Biosens. Bioelectron. 2011, 26, 3398–3404. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xiao, S.; Yao, D.; Lam, M.H.W.; Liang, H. A smart DNA-gold nanoparticle probe for detecting single-base changes on the platform of a quartz crystal microbalance. Chem. Commun. 2015, 51, 4670–4673. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wang, H.; Tang, W.; Hu, S.; Liu, F. An in situ assembly of a DNA-streptavidin dendrimer nanostructure: A new amplified quartz crystal microbalance platform for nucleic acid sensing. Chem. Commun. 2015, 51, 10660–10663. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Sun, Y.; Li, Z.; Wu, A.; Wei, G. Bottom-Up Synthesis and Sensor Applications of Biomimetic Nanostructures. Materials 2016, 9, 53. https://doi.org/10.3390/ma9010053
Wang L, Sun Y, Li Z, Wu A, Wei G. Bottom-Up Synthesis and Sensor Applications of Biomimetic Nanostructures. Materials. 2016; 9(1):53. https://doi.org/10.3390/ma9010053
Chicago/Turabian StyleWang, Li, Yujing Sun, Zhuang Li, Aiguo Wu, and Gang Wei. 2016. "Bottom-Up Synthesis and Sensor Applications of Biomimetic Nanostructures" Materials 9, no. 1: 53. https://doi.org/10.3390/ma9010053
APA StyleWang, L., Sun, Y., Li, Z., Wu, A., & Wei, G. (2016). Bottom-Up Synthesis and Sensor Applications of Biomimetic Nanostructures. Materials, 9(1), 53. https://doi.org/10.3390/ma9010053








