Conductive Polymer Porous Film with Tunable Wettability and Adhesion
Abstract
:1. Introduction
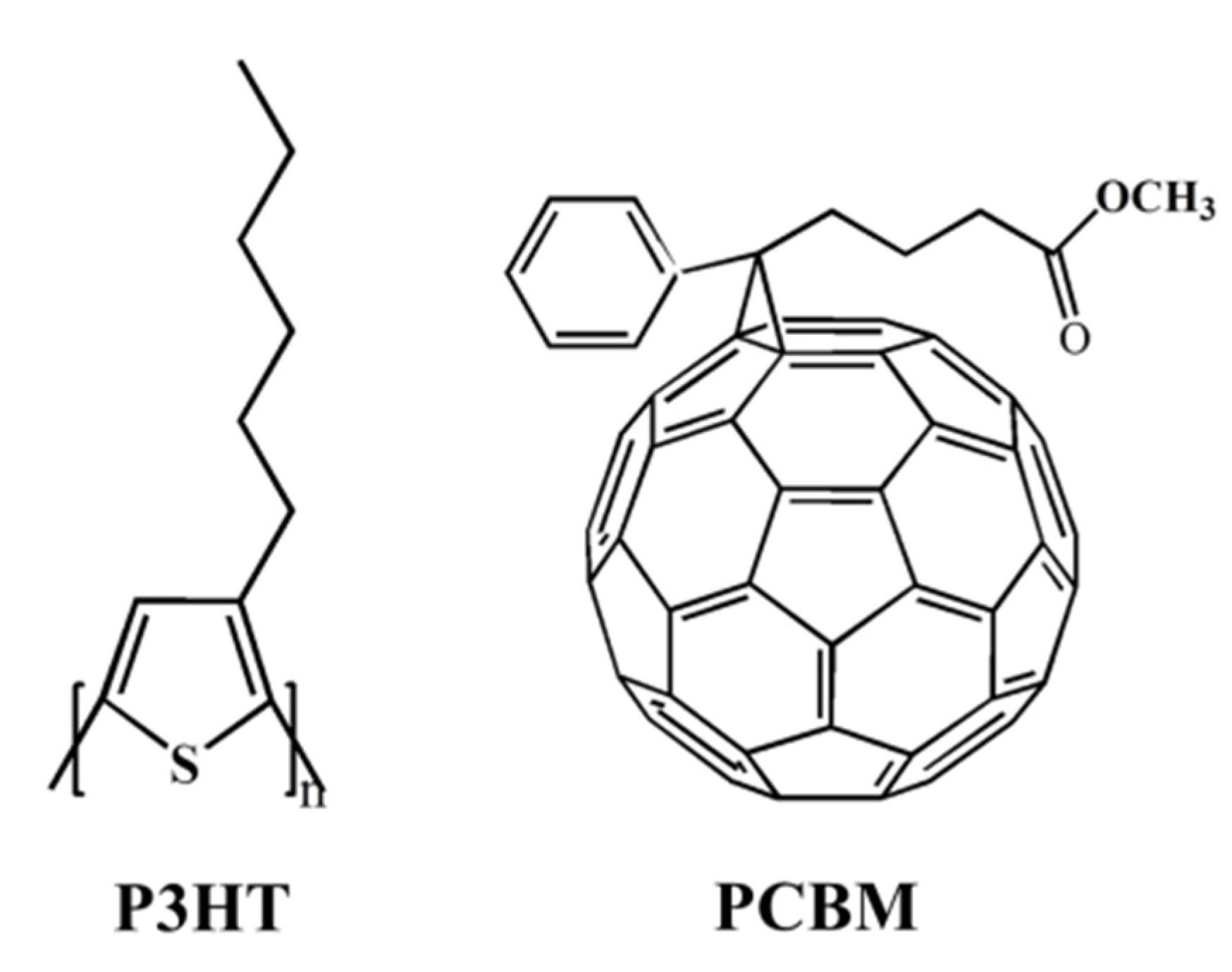
2. Experimental Section
2.1. Materials and Characterization
2.2. Preparation of Conductive Polymer Porous Film

3. Results and Discussion
3.1. Preparation and Morphology of Conductive Polymer Porous Film


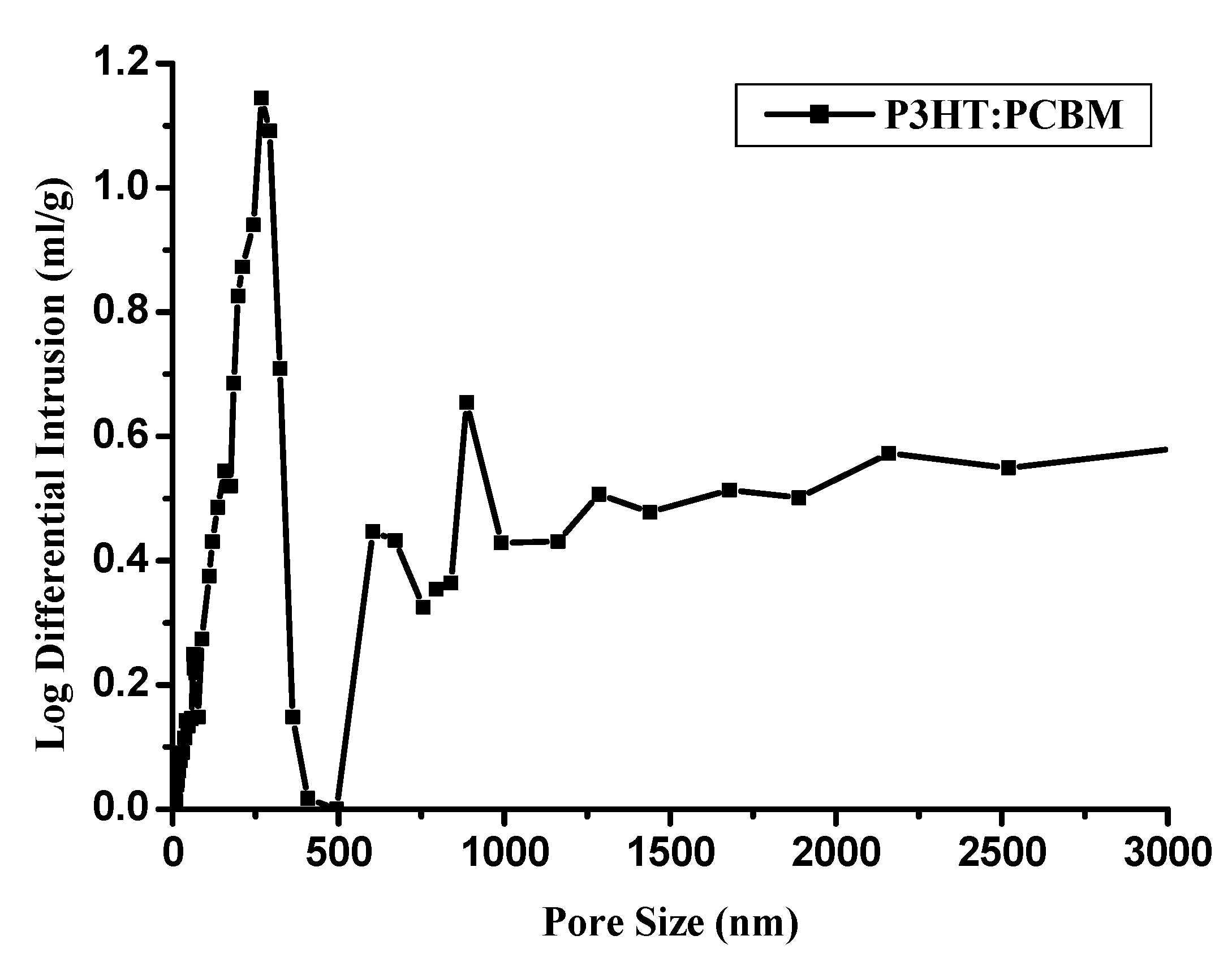
3.2. The Porosity and the Pore Size Distribution
3.3. The Electrical Conductivity
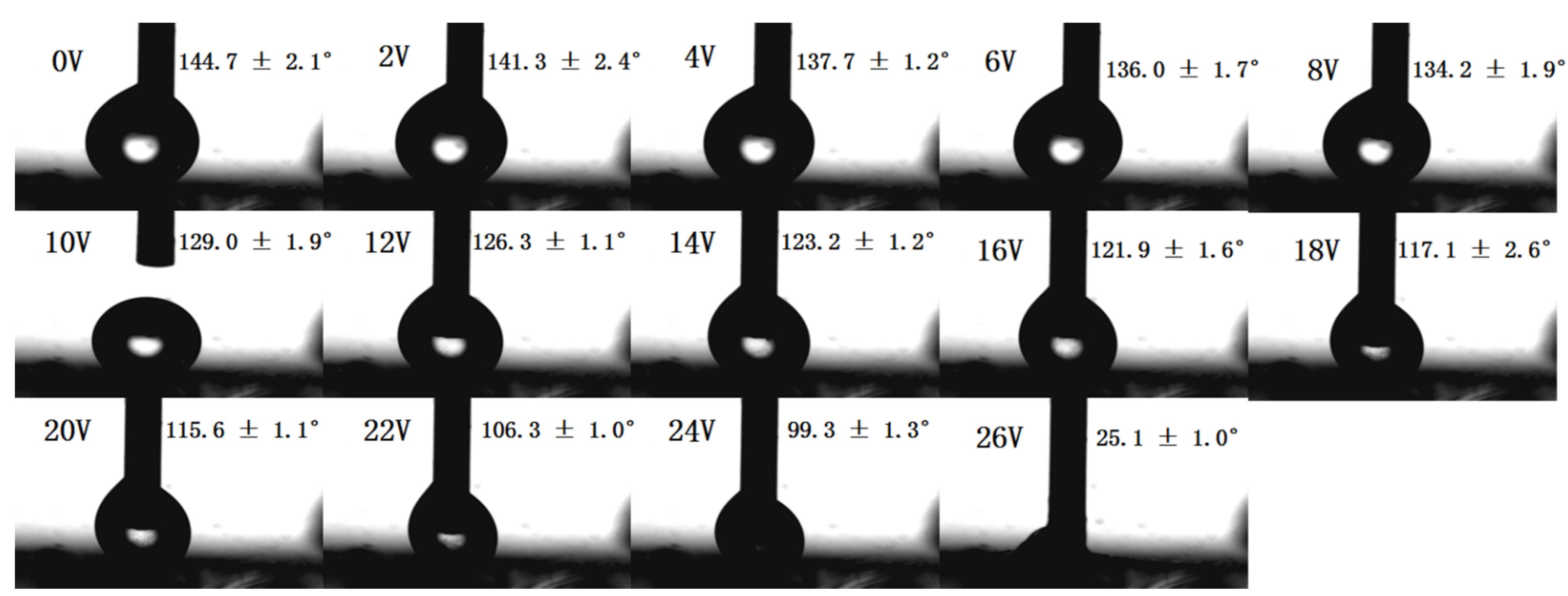
3.4. The Electro-Responsive Wettability


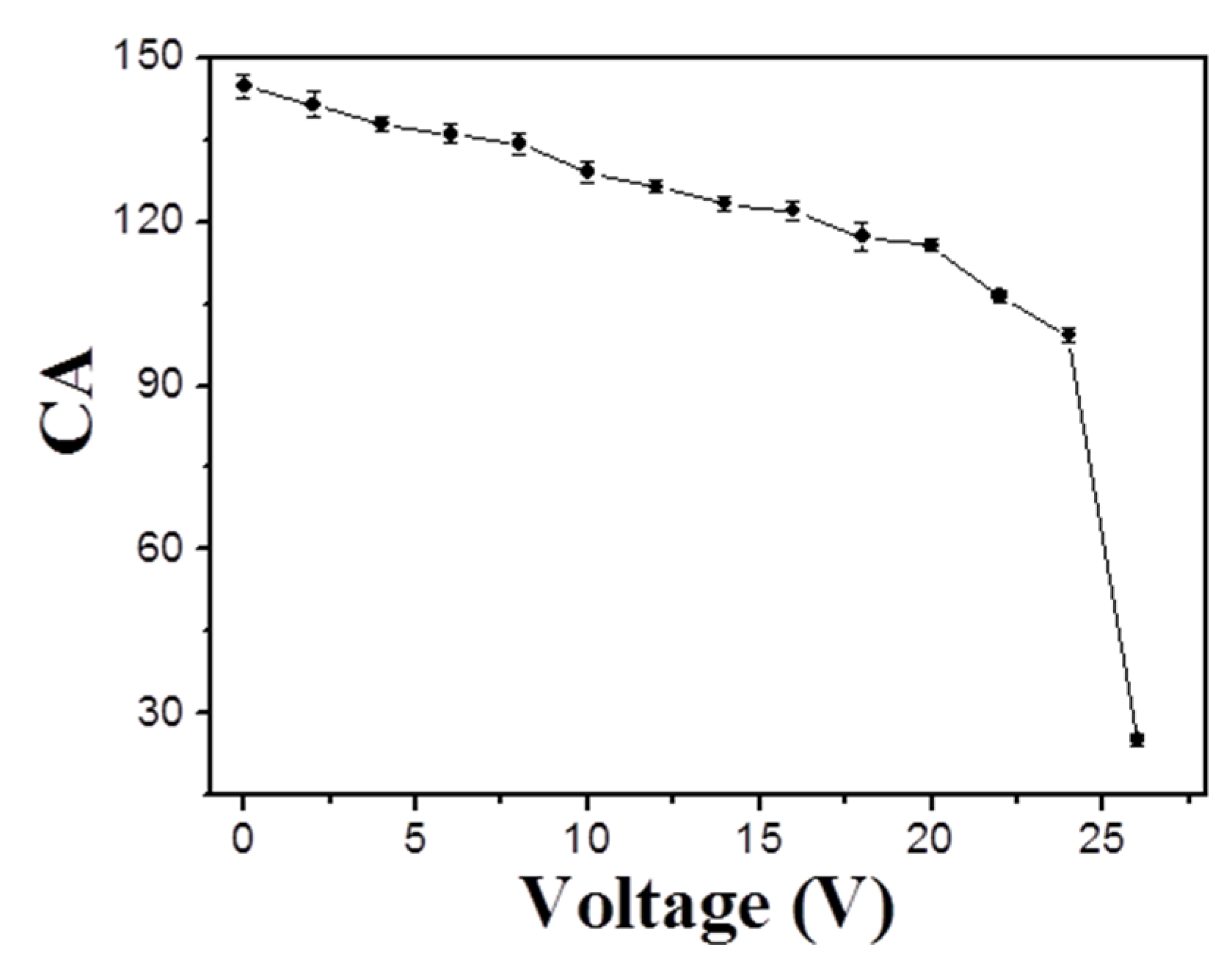
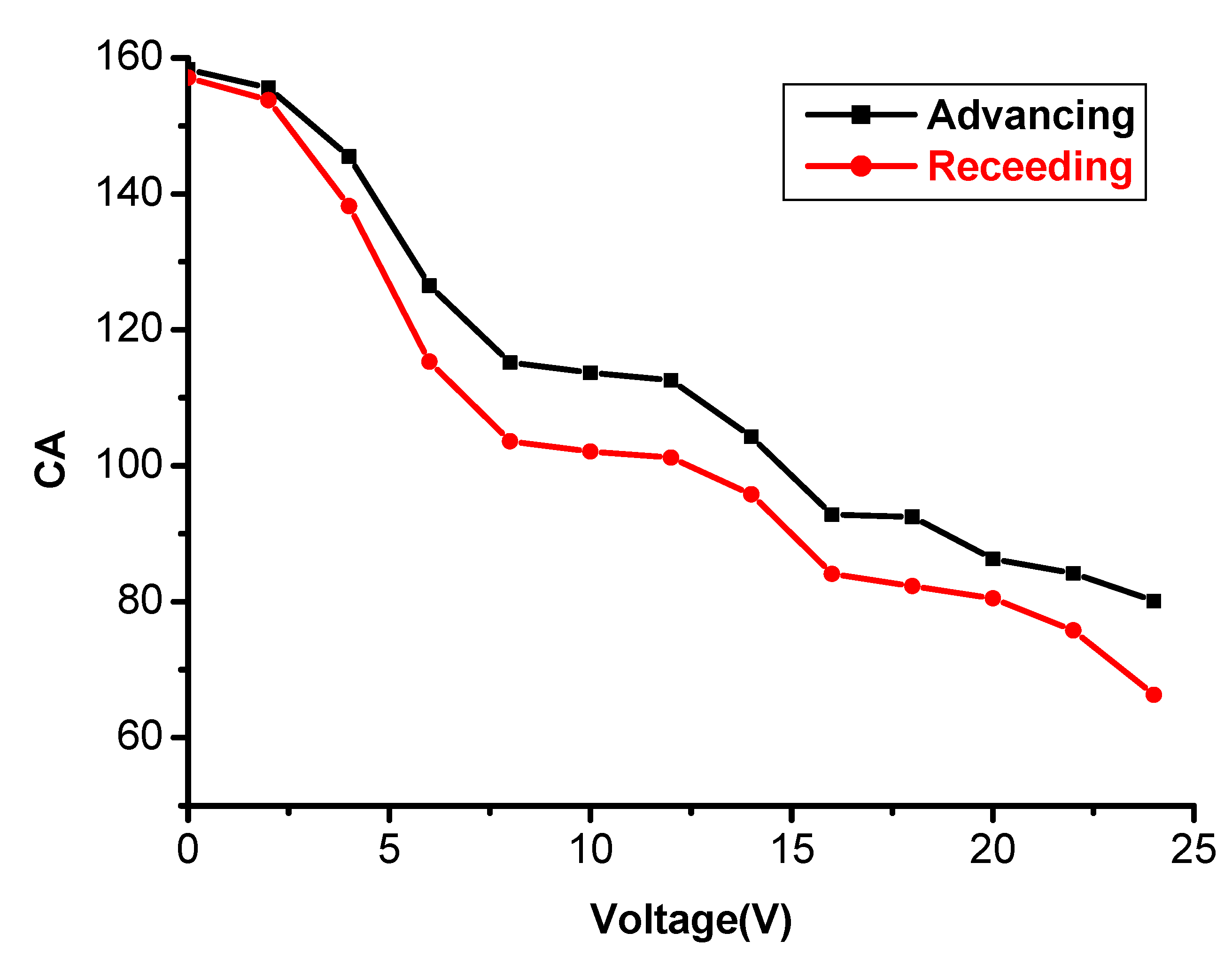
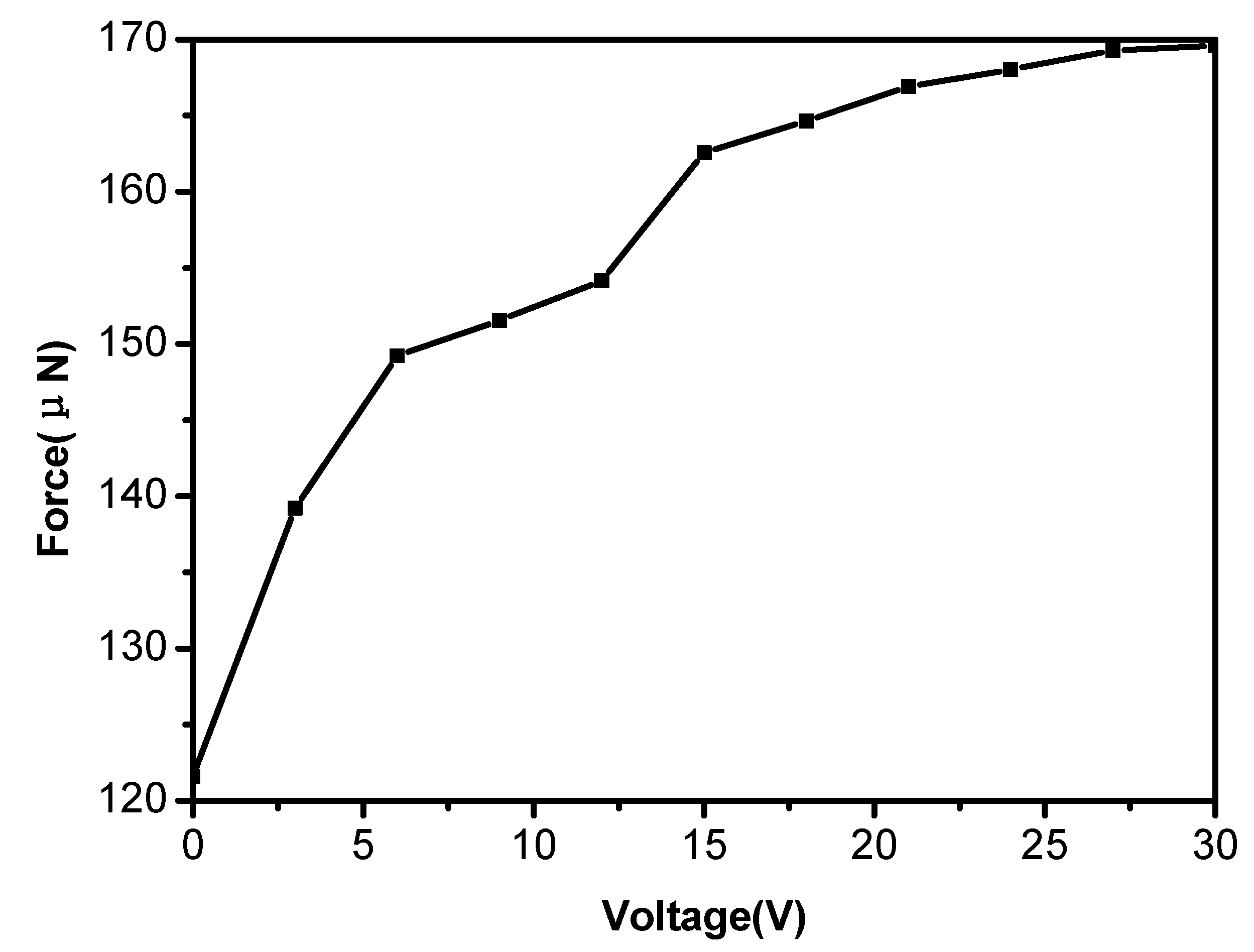
3.5. Tunable Water Adhesion Properties of the Polymer Porous Film
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dai, Z.; Lee, C.-S.; Kim, B.-Y.; Kwak, C.-H.; Yoon, J.-W.; Jeong, H.-M.; Lee, J.-H. Honeycomb-like periodic porous LaFeO3 thin film chemiresistors with enhanced gas-sensing performances. ACS Appl. Mater. Interfaces 2014, 6, 16217–16226. [Google Scholar] [CrossRef] [PubMed]
- Tuller, M.; Or, D.; Dudley, L.M. Adsorption and capillary condensation in porous media: Liquid retention and interfacial configurations in angular pores. Water Resour. Res. 1999, 35, 1949–1964. [Google Scholar] [CrossRef]
- Mondal, K.; Kumar, J.; Sharma, A. Self-organized macroporous thin carbon films for supported metal catalysis. Colloids Surf. A 2013, 427, 83–94. [Google Scholar] [CrossRef]
- Zhang, L.L.; Zhao, X.; Stoller, M.D.; Zhu, Y.; Ji, H.; Murali, S.; Wu, Y.; Perales, S.; Clevenger, B.; Ruoff, R.S. Highly conductive and porous activated reduced graphene oxide films for high-power supercapacitors. Nano Lett. 2012, 12, 1806–1812. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Pasta, M.; Mantia, F.L.; Cui, L.; Jeong, S.; Deshazer, H.D.; Choi, J.W.; Han, S.M.; Cui, Y. Stretchable, porous, and conductive energy textiles. Nano Lett. 2010, 10, 708–714. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, F.J.; Harley, B.A.; Yannas, I.V.; Gibson, L. Influence of freezing rate on pore structure in freeze-dried collagen-GAG scaffolds. Biomaterials 2004, 25, 1077–1086. [Google Scholar] [CrossRef] [PubMed]
- Mikos, A.G.; Sarakinos, G.; Leite, S.M.; Vacant, J.P.; Langer, R. Laminated three-dimensional biodegradable foams for use in tissue engineering. Biomaterials 1993, 14, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Nam, Y.S.; Park, T.G. Porous biodegradable polymeric scaffolds prepared by thermally induced phase separation. J. Biomed. Mater. Res. 1999, 47, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Schugens, Ch.; Maquet, V.; Grandfils, C.; Jérôme, R.; Teyssié, Ph. Biodegradable and macroporous polylactide implants for cell transplantation: 1. Preparation of macroporous polylactide supports by solid-liquid phase separation. Polymer 1996, 37, 1027–1038. [Google Scholar] [CrossRef]
- Schugens, Ch.; Maquet, V.; Grandfils, C.; Jérôme, R.; Teyssié, Ph. Polylactide macroporous biodegradable implants for cell transplantation. II. Preparation of polylactide foams by liquid-liquid phase separation. J. Biomed. Mater. Res. 1996, 30, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Imhof, A.; Pine, D.J. Ordered macroporous materials by emulsion templatin. Nature 1997, 389, 948–951. [Google Scholar] [CrossRef]
- Cameron, N.R. High internal phase emulsion templating as a route to well-defined porous polymers. Polymer 2005, 46, 1439–1449. [Google Scholar] [CrossRef]
- Barg, S.; Soltmann, C.; Andrade, M.; Koch, D.; Grathwohl, G. Cellular ceramics by direct foaming of emulsified ceramic powder suspensions. J. Am. Ceram. Soc. 2008, 91, 2823–2829. [Google Scholar] [CrossRef]
- Fu, Q.; Rahaman, M.N.; Bal, B.S.; Brown, R.F.; Day, D.E. Mechanical and in vitro performance of 13-93 bioactive glass scaffolds prepared by a polymer foam replication technique. Acta Biomater. 2008, 4, 1854–1864. [Google Scholar] [CrossRef] [PubMed]
- Fu, H.; Fu, Q.; Zhou, N.L.; Huang, W.; Rahamana, M.N.; Wang, D.; Liu, X. In vitro evaluation of borate-based bioactive glass scaffolds prepared by a polymer foam replication method. Mater. Sci. Eng. C 2009, 29, 2275–2281. [Google Scholar] [CrossRef]
- Heng, L.; Wang, B.; Li, M.; Zhang, Y.; Jiang, L. Advances in fabrication materials of honeycomb structure films by the breath-figure method. Materials 2013, 6, 460–482. [Google Scholar] [CrossRef]
- Heng, L.; Li, J.; Li, M.; Tian, D.; Fan, L.-Z.; Jiang, L.; Tang, B.Z. Ordered honeycomb structure surface generated by breath figures for liquid reprography. Adv. Funct. Mater. 2014. [Google Scholar] [CrossRef]
- Zhai, S.; Ye, J.-R.; Wang, N.; Jiang, L.-H.; Shen, Q. Fabrication of porous film with controlled pore size and wettability by electric breath figure method. J. Mater. Chem. C 2014, 2, 7168–7172. [Google Scholar] [CrossRef]
- Lee, J.T.Y.; Chow, K.L. SEM sample preparation for cells on 3D scaffolds by freeze-drying and HMDS. Scanning 2011, 33, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.; Grijpma, D.W.; Feijen, J. Preparation of interconnected highly porous polymeric structures by a replication and freeze drying process. J. Biomed. Mater. Res. B 2003, 67B, 732–740. [Google Scholar] [CrossRef]
- Aranaz, I.; Gutiérrez, M.C.; Ferrer, M.L.; del Monte, F. Preparation of chitosan nanocompositeswith a macroporous structure by unidirectional freezing andsubsequent freeze-drying. Mar. Drugs 2014, 12, 5619–5642. [Google Scholar] [CrossRef] [PubMed]
- Qian, L.; Zhang, H. Controlled freezing and freeze drying: A versatile route for porous and micro-/nano-structured materials. J. Chem. Technol. Biotechnol. 2011, 86, 172–184. [Google Scholar] [CrossRef]
- Munch, E.; Saiz, E.; Tomsia, A.P. Architectural control of freeze-cast ceramics through additives and templating. J. Am. Ceram. Soc. 2009, 92, 1534–1539. [Google Scholar] [CrossRef]
- Deville, S.; Saiz, E.; Tomsia, A.P. Ice-templated porous alumina structures. Acta Mater. 2007, 55, 1965–1974. [Google Scholar] [CrossRef]
- Madihally, S.V.; Matthew, H.W.T. Porous chitosan scaffolds for tissue engineering. Biomaterials 1999, 20, 1133–1142. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.H.; Eliason, J.F.; Matthew, H.W.T. Application of porous glycosaminoglycan-based scaffolds for expansion of human cord blood stem cells in perfusion culture. J. Biomed. Mater. Res. 2008, 86A, 98–107. [Google Scholar] [CrossRef]
- Zhang, Z.; Sèbe, G.; Rentsch, D.; Zimmermann, T.; Tingaut, P. Ultralightweight and flexible silylated nanocellulose sponges for the selective removal of oil from water. Chem. Mater. 2014, 26, 2659–2668. [Google Scholar] [CrossRef]
- Wen, L.; Hou, X.; Tian, Y.; Nie, F.-Q.; Song, Y.; Zhai, J.; Jiang, L. Bioinspired smart gating of nanochannels toward photoelectric-conversion systems. Adv. Mater. 2010, 22, 1021–1024. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Jin, M.; Liu, Z.; Tryk, D.A.; Nishimoto, S.; Murakami, T.; Fujishima, A. Superhydrophobic TiO2 surfaces: Preparation, photocatalytic wettability conversion, and superhydrophobic–superhydrophilic patterning. J. Phys. Chem. C 2007, 111, 14521–14529. [Google Scholar] [CrossRef]
- Faustini, M.; Nicole, L.; Boissière, C.; Innocenzi, P.; Sanchez, C.; Grosso, D. Hydrophobic, antireflective, self-Cleaning, and antifogging sol–gel coatings: An example of multifunctional nanostructured materials for photovoltaic cells. Chem. Mater. 2010, 22, 4406–4413. [Google Scholar] [CrossRef]
- O’Brien, F.J.; Harley, B.A.; Yannas, I.V.; Gibson, L.J. The effect of pore size on cell adhesion in collagen-GAG scaffolds. Biomaterials 2005, 26, 433–441. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.; Dai, L.; Stone, M.; Xia, Z.; Wang, Z.L. Carbon nanotube arrays with strong shear binding-on and easy normal lifting-off. Science 2008, 322, 238–242. [Google Scholar] [CrossRef] [PubMed]
- Cho, W.K.; Choi, I.S. Fabrication of hairy polymeric films inspired by geckos: Wetting and high adhesion properties. Adv. Funct. Mater. 2008, 18, 1089–1096. [Google Scholar] [CrossRef]
- Feng, L.; Zhang, Y.; Xi, J.; Zhu, Y.; Wang, N.; Xia, F.; Jiang, L. Petal effect: A superhydrophobic state with high adhesive force. Langmuir 2008, 24, 4114–4119. [Google Scholar] [CrossRef] [PubMed]
- Heng, L.; Meng, X.; Wang, B.; Jiang, L. Bioinspired design of honeycomb structure interfaces with controllable water adhesion. Langmuir 2013, 29, 9491–9498. [Google Scholar] [CrossRef] [PubMed]
- Treat, N.D.; Brady, M.A.; Smith, G.; Toney, M.F.; Kramer, E.J.; Hawker, C.J.; Chabinyc, M.L. Interdiffusion of PCBM and P3HT reveals miscibility in a photovoltaically active blend. Adv. Energy Mater. 2011, 1, 82–89. [Google Scholar] [CrossRef]
- Chen, D.; Nakahara, A.; Wei, D.; Nordlund, D.; Russell, T.P. P3HT/PCBM bulk heterojunction organic photovoltaics: Correlating efficiency and morphology. Nano Lett. 2011, 11, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Stenzel-Rosenbaum, M.H.; Davis, T.P.; Fane, A.G.; Chen, V. Porous polymer films and honeycomb structures made by the self-organization of well-defined macromolecular structures created by living radical polymerization techniques. Angew. Chem. Int. Ed. 2001, 40, 3428–3432. [Google Scholar] [CrossRef]
- Mugele, F.; Baret, J.-C. Electrowetting: From basics to applications. J. Phys.: Condens. Matter 2005, 17, R705–R774. [Google Scholar]
- Wixforth, A.; Strobl, C.; Gauer, C.; Toegl, A.; Scriba, J.; von Guttenberg, Z. Acoustic manipulation of small droplets. Anal. Bioanal. Chem. 2004, 379, 982–991. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teng, Y.; Zhang, Y.; Heng, L.; Meng, X.; Yang, Q.; Jiang, L. Conductive Polymer Porous Film with Tunable Wettability and Adhesion. Materials 2015, 8, 1817-1830. https://doi.org/10.3390/ma8041817
Teng Y, Zhang Y, Heng L, Meng X, Yang Q, Jiang L. Conductive Polymer Porous Film with Tunable Wettability and Adhesion. Materials. 2015; 8(4):1817-1830. https://doi.org/10.3390/ma8041817
Chicago/Turabian StyleTeng, Yuqi, Yuqi Zhang, Liping Heng, Xiangfu Meng, Qiaowen Yang, and Lei Jiang. 2015. "Conductive Polymer Porous Film with Tunable Wettability and Adhesion" Materials 8, no. 4: 1817-1830. https://doi.org/10.3390/ma8041817
APA StyleTeng, Y., Zhang, Y., Heng, L., Meng, X., Yang, Q., & Jiang, L. (2015). Conductive Polymer Porous Film with Tunable Wettability and Adhesion. Materials, 8(4), 1817-1830. https://doi.org/10.3390/ma8041817





