Spherical V-Fe-MCM-48: The Synthesis, Characterization and Hydrothermal Stability
Abstract
:1. Introduction
2. Results and Discussion
2.1. Mesoporous Structure of V-Fe-MCM-48 Sample
2.1.1. XRD
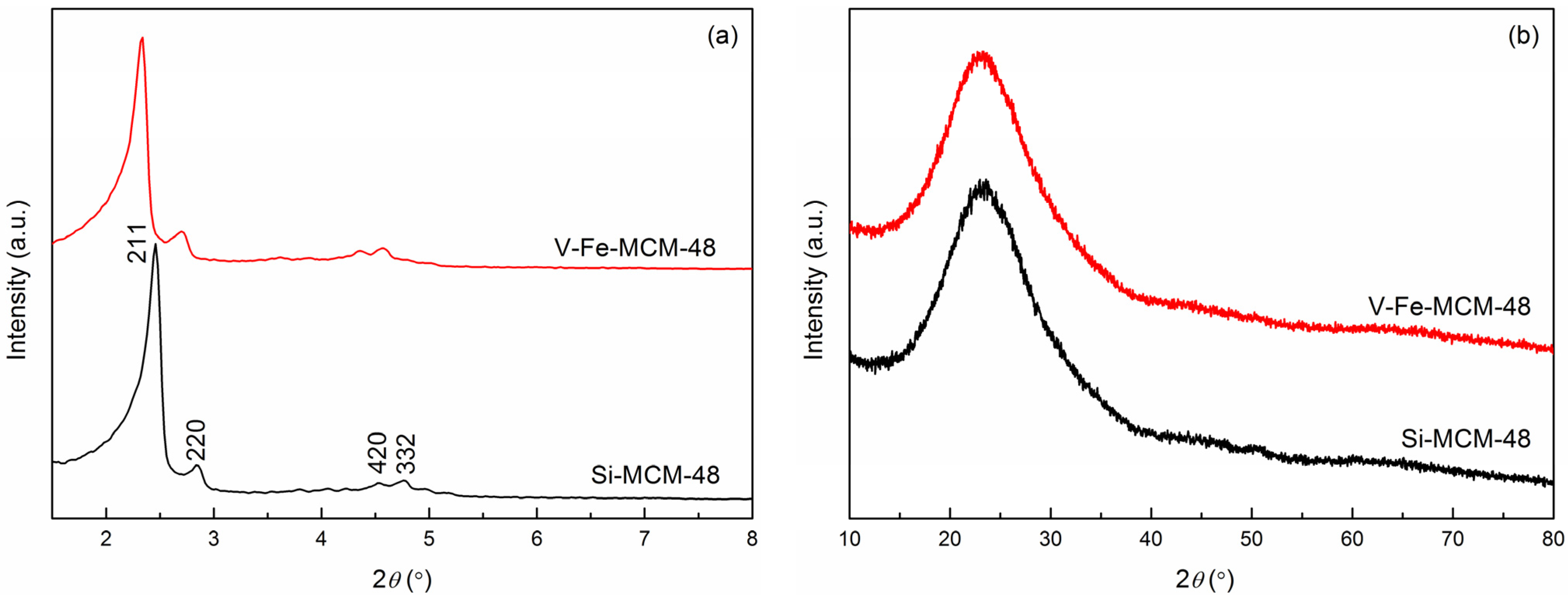
2.1.2. Nitrogen Adsorption-Desorption
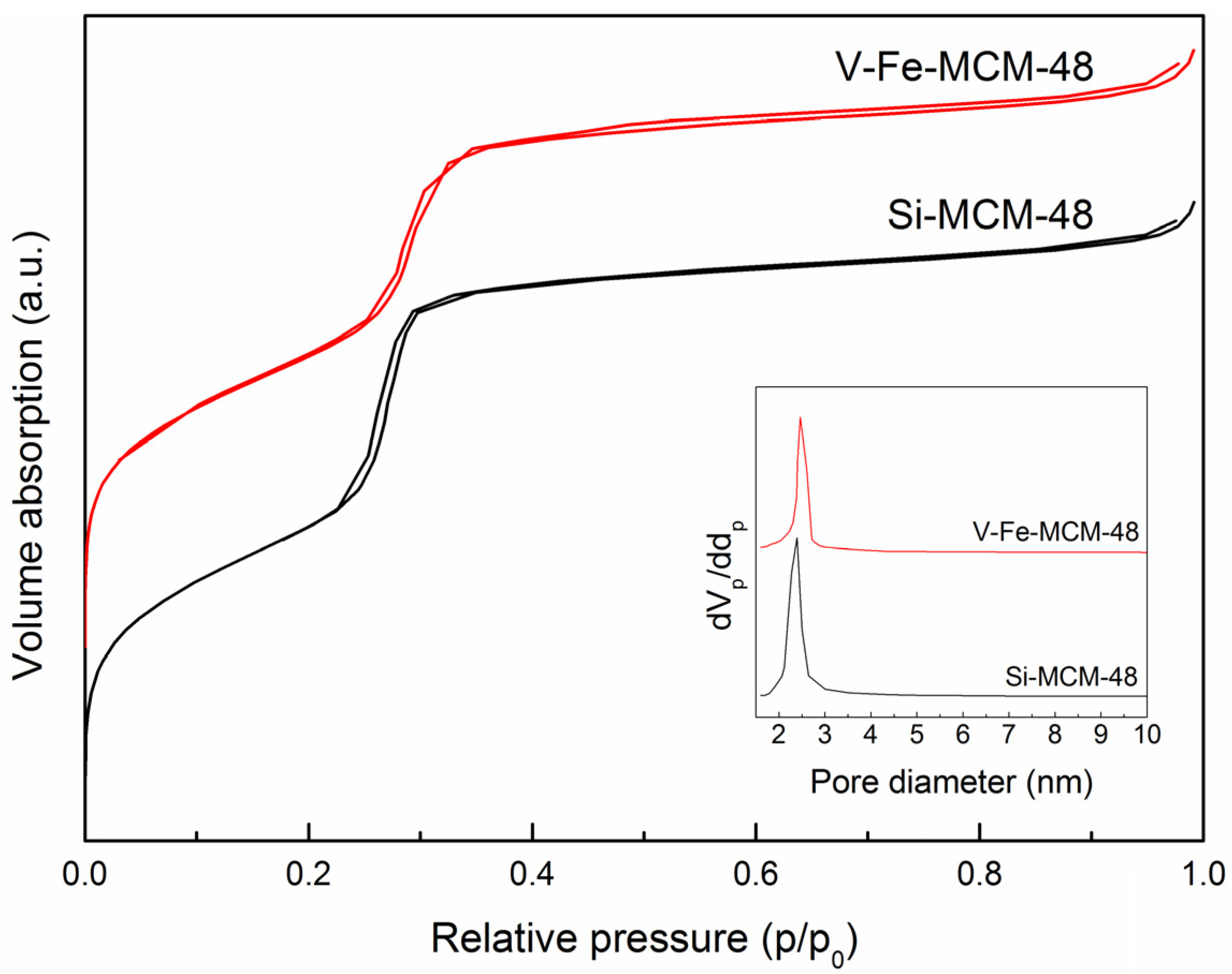
| Sample | V/Si a | V/Si b | Fe/Si a | Fe/Si b | d211 | a0 c | dp d | as, BET | tw e | Vp |
|---|---|---|---|---|---|---|---|---|---|---|
| (Molar Ratio) | (Molar Ratio) | (Molar Ratio) | (Molar Ratio) | (nm) | (nm) | (nm) | (m2/g) | (nm) | (cm3/g) | |
| Si-MCM-48 | — | — | — | — | 3.59 | 8.80 | 2.39 | 1369.4 | 1.65 | 0.832 |
| V-Fe-MCM-48 | 0.0296 | 0.0066 | 0.0062 | 0.0021 | 3.81 | 9.33 | 2.46 | 1213.2 | 1.79 | 0.777 |
2.1.3. SEM and TEM

2.2. Statues of Heteroatoms
2.2.1. UV-Vis

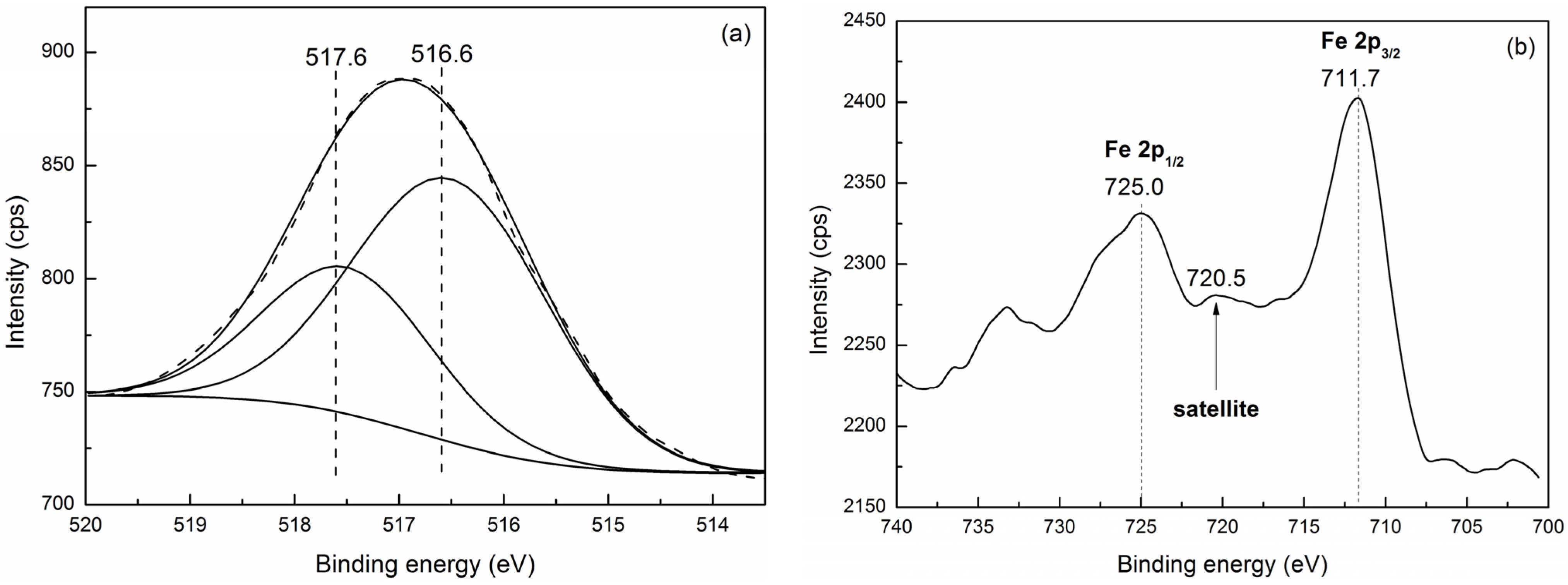
2.2.2. XPS
2.3. Hydrothermal Stability Test
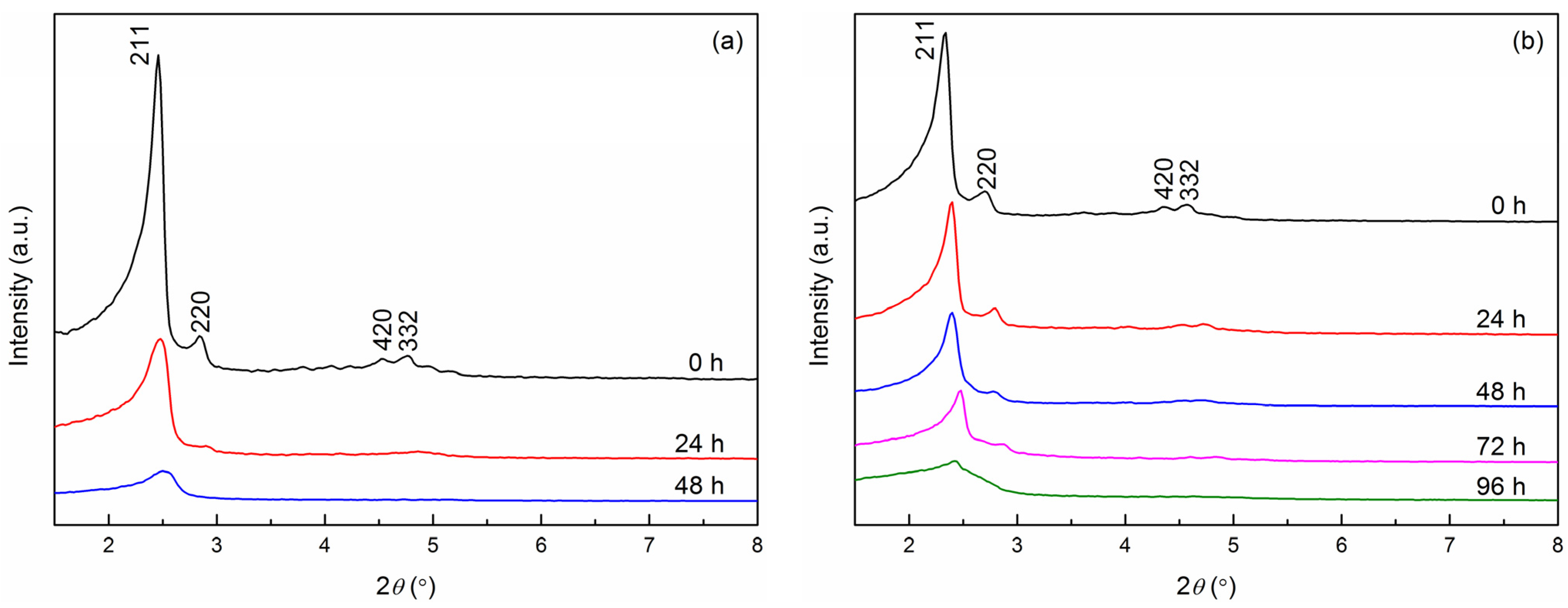

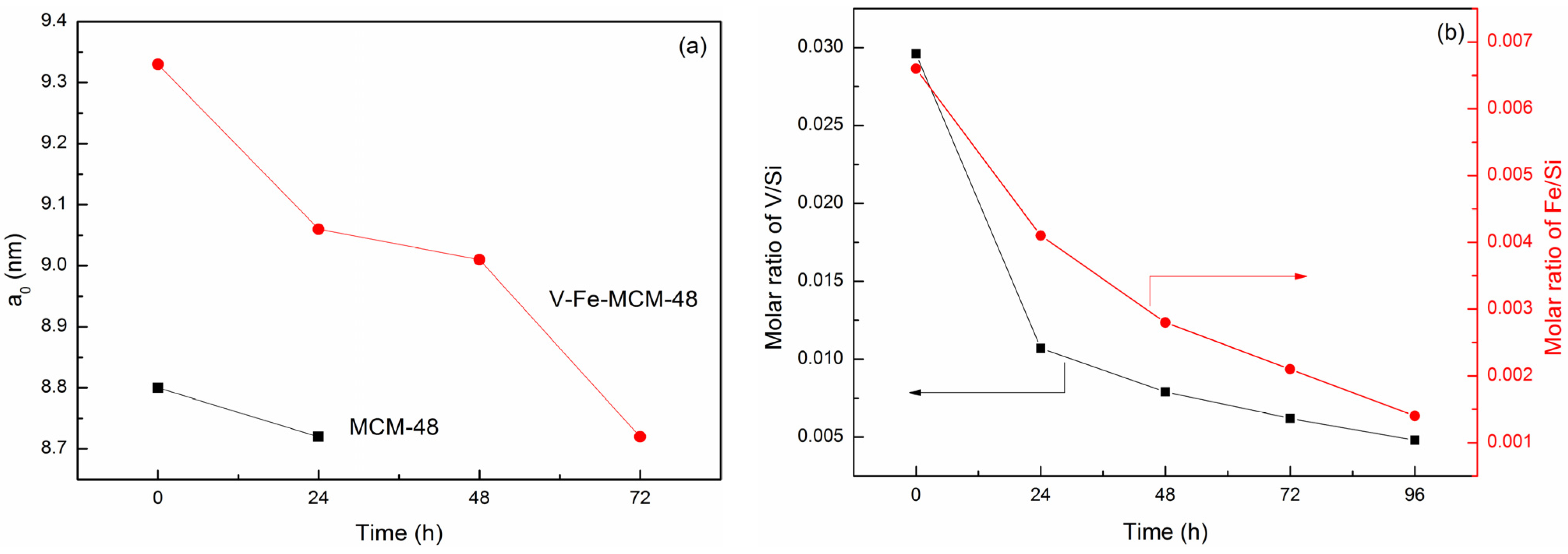
| Time (h) | MCM-48 | V-Fe-MCM-48 | ||||
|---|---|---|---|---|---|---|
| d211 (nm) | a0 a (nm) | V/Si a (Molar Ratio) | Fe/Si b (Molar Ratio) | d211 (nm) | a0 a (nm) | |
| 0 | 3.59 | 8.80 | 0.0296 | 0.0066 | 3.81 | 9.33 |
| 24 | 3.56 | 8.72 | 0.0107 | 0.0041 | 3.70 | 9.06 |
| 48 | – | – | 0.0079 | 0.0028 | 3.68 | 9.01 |
| 72 | – | – | 0.0062 | 0.0021 | 3.56 | 8.72 |
| 96 | – | – | 0.0048 | 0.0014 | – | – |
3. Experimental Section
3.1. Materials
3.2. Synthesis of the Samples
3.3. Characterization
3.4. Hydrothermal Stability Test
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kresge, C.T.; Leonowicz, M.E.; Roth, W.J.; Vartuli, J.C.; Beck, J.S. Ordered mesoporous molecular-sieves synthesized by a liquid-crystal template mechanism. Nature 1992, 359, 710–712. [Google Scholar] [CrossRef]
- Egi, M.; Sugiyama, K.; Saneto, M.; Hanada, R.; Kato, K.; Akai, S. A mesoporous-silica-immobilized oxovanadium cocatalyst for the lipase-catalyzed dynamic kinetic resolution of racemic alcohols. Angew. Chem. Int. Ed. 2013, 52, 3654–3658. [Google Scholar] [CrossRef]
- Zhou, Y.; Lin, W.G.; Wan, M.M.; Yang, J.; Zhu, J.H. Novel selective adsorbent derived from hierarchical rockery-like MCM-41 monolith. J. Mater. Chem. 2012, 22, 23633–23641. [Google Scholar] [CrossRef]
- Wang, H.; Qian, W.; Chen, J.; Wu, Y.; Xu, X.; Wang, J.; Kong, Y. Spherical V-MCM-48: The synthesis, characterization and catalytic performance in styrene oxidation. RSC Adv. 2014, 4, 50832–50839. [Google Scholar] [CrossRef]
- Kim, T.W.; Kleitz, F.; Paul, B.; Ryoo, R. MCM-48-like large mesoporous silicas with tailored pore structure: Facile synthesis domain in a ternary triblock copolymer-butanol-water system. J. Am. Chem. Soc. 2005, 127, 7601–7610. [Google Scholar] [CrossRef] [PubMed]
- Gomez, S.; Garces, L.J.; Villegas, J.; Ghosh, R.; Giraldo, O.; Suib, S.L. Synthesis and characterization of TM-MCM-48 (TM = Mn, V, Cr) and their catalytic activity in the oxidation of styrene. J. Catal. 2005, 233, 60–67. [Google Scholar] [CrossRef]
- Suteewong, T.; Sai, H.; Cohen, R.; Wang, S.; Bradbury, M.; Baird, B.; Gruner, S.M.; Wiesner, U. Highly aminated mesoporous silica nanoparticles with cubic pore structure. J. Am. Chem. Soc. 2010, 133, 172–175. [Google Scholar] [PubMed]
- Alfredsson, V.; Anderson, M.W. Structure of MCM-48 revealed by transmission electron microscopy. Chem. Mater. 1996, 8, 1141–1146. [Google Scholar] [CrossRef]
- Zhao, W.; Qin, M.; Wang, L.N.; Chu, J.L.; Qu, J.K.; Li, S.H.; Li, Q.Z.; Qi, T. Synthesis of submicron spherical Fe-MCM-48 with actual gyroid like structure. J. Colloid Interface Sci. 2012, 384, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.W.; Chung, P.W.; Lin, V.S.Y. Facile synthesis of monodisperse spherical MCM-48 mesoporous silica nanoparticles with controlled particle size. Chem. Mater. 2010, 22, 5093–5104. [Google Scholar] [CrossRef]
- Hu, J.; Zhou, L.H.; Han, X.; Liu, H.L.; Hu, Y. The complex morphologies of Ti-MCM-41 templated by gemini surfactant. Mater. Res. Bull. 2007, 42, 102–112. [Google Scholar] [CrossRef]
- Chen, Y.; Shi, X.; Han, B.; Qin, H.; Li, Z.; Lu, Y.; Wang, J.; Kong, Y. The complete control for the nanosize of spherical MCM-41. J. Nanosci. Nanotechnol. 2012, 12, 7239–7249. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.C.; Wang, X.M.; Lee, S.; Pfefferle, L.D.; Haller, G.L. Surfactant chain length effect on the hexagonal-to-cubic phase transition in mesoporous silica synthesis. Microporous Mesoporous Mater. 2012, 147, 242–251. [Google Scholar] [CrossRef]
- Selvam, P.; Dapurkar, S.E. Catalytic activity of highly ordered mesoporous VMCM-48. Appl. Catal. A 2004, 276, 257–265. [Google Scholar] [CrossRef]
- Han, B.; Wang, H.; Kong, Y.; Wang, J. Improvement on the mesostructural ordering and catalytic activity of Co-MCM-41 with ascorbic acid as auxiliary. Mater. Lett. 2013, 100, 159–162. [Google Scholar] [CrossRef]
- Ma, H.; Kong, Y.; Hou, W.; Yan, Q. Synthesis of ordered hexagonal porous tin-doped zirconium oxides with a high surface area. Microporous Mesoporous Mater. 2005, 77, 241–243. [Google Scholar] [CrossRef]
- Kong, Y.; Zhu, H.Y.; Yang, G.; Guo, X.F.; Hou, W.H.; Yan, Q.J.; Gu, M.; Hu, C. Investigation of the structure of MCM-41 samples with a high copper content. Adv. Funct. Mater. 2004, 14, 816–820. [Google Scholar] [CrossRef]
- Betiha, M.A.; Hassan, H.M.A.; Al-Sabagh, A.M.; Khder, A.E.R.S.; Ahmed, E.A. Direct synthesis and the morphological control of highly ordered mesoporous AlSBA-15 using urea-tetrachloroaluminate as a novel aluminum source. J. Mater. Chem. 2012, 22, 17551–17559. [Google Scholar]
- Piumetti, M.; Armandi, M.; Garrone, E.; Bonelli, B. An IR spectroscopy assessment of the surface acidity of mesoporous VOx-SiO2 catalysts. Microporous Mesoporous Mater. 2012, 164, 111–119. [Google Scholar] [CrossRef]
- Samanta, S.; Das, S.; Samanta, P.K.; Dutta, S.; Biswas, P. A mononuclear copper(II) complex immobilized in mesoporous silica: An efficient heterogeneous catalyst for the aerobic oxidation of benzylic alcohols. Rsc Adv. 2013, 3, 19455–19466. [Google Scholar] [CrossRef]
- Zhang, G.; Long, J.; Wang, X.; Zhang, Z.; Dai, W.; Liu, P.; Li, Z.; Wu, L.; Fu, X. Catalytic role of Cu sites of Cu/MCM-41 in phenol hydroxylation. Langmuir 2010, 26, 1362–1371. [Google Scholar] [CrossRef] [PubMed]
- Lou, L.L.; Jiang, S.; Yu, K.; Gu, Z.; Ji, R.; Dong, Y.; Liu, S. Mesoporous silicas functionalized with aminopropyl via co-condensation: Effective supports for chiral Mn(III) salen complex. Microporous Mesoporous Mater. 2011, 142, 214–220. [Google Scholar] [CrossRef]
- Xiong, H.; Pham, H.N.; Datye, A.K. Hydrothermally stable heterogeneous catalysts for conversion of biorenewables. Green Chem. 2014, 16, 4627–4643. [Google Scholar]
- Wang, J.; Fan, D.; Yu, T.; Wang, J.; Hao, T.; Hu, X.; Shen, M.; Li, W. Improvement of low-temperature hydrothermal stability of Cu/SAPO-34 catalysts by Cu2+ species. J. Catal. 2015, 322, 84–90. [Google Scholar] [CrossRef]
- Xia, Y.; Mokaya, R. Enhanced hydrothermal stability of Al-grafted MCM-48 prepared via various alumination routes. Microporous Mesoporous Mater. 2004, 74, 179–188. [Google Scholar] [CrossRef]
- Li, Q.; Wu, Z.; Tu, B.; Park, S.S.; Ha, C.-S.; Zhao, D. Highly hydrothermal stability of ordered mesoporous aluminosilicates Al-SBA-15 with high Si/Al ratio. Microporous Mesoporous Mater. 2010, 135, 95–104. [Google Scholar] [CrossRef]
- Pan, D.; Yuan, P.; Zhao, L.; Liu, N.; Zhou, L.; Wei, G.; Zhang, J.; Ling, Y.; Fan, Y.; Wei, B.; et al. New understanding and simple approach to synthesize highly hydrothermally stable and ordered mesoporous materials. Chem. Mater. 2009, 21, 5413–5425. [Google Scholar] [CrossRef]
- Wang, J.; Ge, H.; Bao, W. Synthesis and characteristics of SBA-15 with thick pore wall and high hydrothermal stability. Mater. Lett. 2015, 145, 312–315. [Google Scholar] [CrossRef]
- Zhang, Z.; Han, Y.; Zhu, L.; Wang, R.; Yu, Y.; Qiu, S.; Zhao, D.; Xiao, F.S. Strongly acidic and high-temperature hydrothermally stable mesoporous aluminosilicates with ordered hexagonal structure. Angew. Chem. Int. Ed. 2001, 40, 1258–1262. [Google Scholar] [CrossRef]
- Jun, S.; Kim, J.M.; Ryoo, R.; Ahn, Y.S.; Han, M.H. Hydrothermal stability of MCM-48 improved by post-synthesis restructuring in salt solution. Microporous Mesoporous Mater. 2000, 41, 119–127. [Google Scholar] [CrossRef]
- Cao, L.; Zhao, X.; Jin, J.; Xu, C.; Gao, X.; Lan, L.; Yuan, X.; Liu, H.; Liu, H. Crystal-seeds-based strategy for the synthesis of hydrothermally stable mesoporous aluminosilicates with a largely decreased H2O amount. Ind. Eng. Chem. Res. 2014, 53, 17286–17293. [Google Scholar] [CrossRef]
- Xu, X.Y.; Kong, Y.; Chen, Y.; Zhao, N.; Shi, X.B.; Wang, J. Facile synthesis of spherical V-MCM-48. Chin. J. Inorg. Chem. 2012, 28, 2478–2484. [Google Scholar]
- Kong, Y.; Zhang, R.; Xu, X.; Wang, Z.; Wang, J. Synthesis and characterization of V-MCM41 with high vanadium content. Chin. J. Inorg. Chem. 2008, 24, 1124–1127. [Google Scholar]
- Wu, Y.; Zhang, Y.; Cheng, J.; Li, Z.; Wang, H.; Sun, Q.; Han, B.; Kong, Y. Synthesis, characterization and catalytic activity of binary metallic titanium and iron containing mesoporous silica. Microporous Mesoporous Mater. 2012, 162, 51–59. [Google Scholar] [CrossRef]
- Zhang, Y.; Gao, F.; Wan, H.; Wu, C.; Kong, Y.; Wu, X.; Zhao, B.; Dong, L.; Chen, Y. Synthesis, characterization of bimetallic Ce-Fe-SBA-15 and its catalytic performance in the phenol hydroxylation. Microporous Mesoporous Mater. 2008, 113, 393–401. [Google Scholar] [CrossRef]
- Jiang, S.; Kong, Y.; Wang, J.; Ren, X.; Yan, Q. Synthesis, characterization of bimetallic Sn-Zn-MCM41 and its catalytic performance in the hydroxylation of phenol. J. Porous Mat. 2006, 13, 341–346. [Google Scholar] [CrossRef]
- Kong, Y.; Jiang, S.; Wang, J.; Wang, S.; Yan, Q.; Lu, Y. Synthesis and characterization of Cu-Ti-MCM41. Microporous Mesoporous Mater. 2005, 86, 191–197. [Google Scholar] [CrossRef]
- He, N.; Lu, Z.; Yuan, C.; Hong, J.; Yang, C.; Bao, S.; Xu, Q. Effect of trivalent elements on the thermal and hydrothermal stability of MCM-41 mesoporous molecular materials. Supramol. Sci. 1998, 5, 553–558. [Google Scholar] [CrossRef]
- Kumar, D.; Pillai, K.T.; Sudersanan, V.; Dey, G.K.; Gupta, N.M. Hydrothermal synthesis and characterization of uranium-containing MCM-48 samples. Chem. Mater. 2003, 15, 3859–3865. [Google Scholar] [CrossRef]
- Brunauer, S.; Deming, L.S.; Deming, W.E.; Teller, E. On a theory of the van der waals adsorption of gases. J. Am. Chem. Soc. 1940, 62, 1723–1732. [Google Scholar] [CrossRef]
- Greenwood, N.N.; Earnshaw, A. Chemistry of the Elements, 2nd ed.; Butterworth-Heinemann: Oxford, UK, 1997; p. 984. [Google Scholar]
- Vekilov, P.G. Nucleation. Cryst. Growth Des. 2010, 10, 5007–5019. [Google Scholar] [CrossRef] [PubMed]
- Kashchiev, D.; van Rosmalen, G.M. Review: Nucleation in solutions revisited. Cryst. Res. Technol. 2003, 38, 555–574. [Google Scholar] [CrossRef]
- Maneesuwan, H.; Chaisuwan, T.; Wongkasemjit, S. Synthesis of Fe-Ti-MCM-48 from silatrane precursor via sol-gel process and its hydrothermal stability. Mater. Chem. Phys. 2014, 146, 374–379. [Google Scholar] [CrossRef]
- Maneesuwan, H.; Longloilert, R.; Chaisuwan, T.; Wongkasemjit, S. Synthesis and characterization of Fe-Ce-MCM-48 from silatrane precursor via sol-gel process. Mater. Lett. 2013, 94, 65–68. [Google Scholar] [CrossRef]
- Hu, L.; Yue, B.; Wang, C.; Chen, X.; He, H. Enhanced catalytic activity over vanadium-containing silylated SBA-15 catalysts for styrene epoxidation and benzene hydroxylation. Appl. Catal. A 2014, 477, 141–146. [Google Scholar] [CrossRef]
- Gao, F.; Zhang, Y.H.; Wan, H.Q.; Kong, Y.; Wu, X.C.; Dong, L.; Li, B.Q.; Chen, Y. The states of vanadium species in V-SBA-15 synthesized under different pH values. Microporous Mesoporous Mater. 2008, 110, 508–516. [Google Scholar] [CrossRef]
- Zhao, W.; Luo, Y.F.; Deng, P.; Li, Q.Z. Synthesis of Fe-MCM-48 and its catalytic performance in phenol hydroxylation. Catal. Lett. 2001, 73, 199–202. [Google Scholar] [CrossRef]
- Zhang, H.; Tang, C.; Sun, C.; Qi, L.; Gao, F.; Dong, L.; Chen, Y. Direct synthesis, characterization and catalytic performance of bimetallic Fe-Mo-SBA-15 materials in selective catalytic reduction of NO with NH3. Microporous Mesoporous Mater. 2012, 151, 44–55. [Google Scholar] [CrossRef]
- Hess, C.; Wild, U.; Schlögl, R. The mechanism for the controlled synthesis of highly dispersed vanadia supported on silica SBA-15. Microporous Mesoporous Mater. 2006, 95, 339–349. [Google Scholar]
- De Stefanis, A.; Kaciulis, S.; Pandolfi, L. Preparation and characterization of Fe-MCM-41 catalysts employed in the degradation of plastic materials. Microporous Mesoporous Mater. 2007, 99, 140–148. [Google Scholar]
- Yamashita, T.; Hayes, P. Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials. Appl. Surf. Sci. 2008, 254, 2441–2449. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qian, W.; Wang, H.; Chen, J.; Kong, Y. Spherical V-Fe-MCM-48: The Synthesis, Characterization and Hydrothermal Stability. Materials 2015, 8, 1752-1765. https://doi.org/10.3390/ma8041752
Qian W, Wang H, Chen J, Kong Y. Spherical V-Fe-MCM-48: The Synthesis, Characterization and Hydrothermal Stability. Materials. 2015; 8(4):1752-1765. https://doi.org/10.3390/ma8041752
Chicago/Turabian StyleQian, Wang, Haiqing Wang, Jin Chen, and Yan Kong. 2015. "Spherical V-Fe-MCM-48: The Synthesis, Characterization and Hydrothermal Stability" Materials 8, no. 4: 1752-1765. https://doi.org/10.3390/ma8041752
APA StyleQian, W., Wang, H., Chen, J., & Kong, Y. (2015). Spherical V-Fe-MCM-48: The Synthesis, Characterization and Hydrothermal Stability. Materials, 8(4), 1752-1765. https://doi.org/10.3390/ma8041752





