A Critical Review of Dental Implant Materials with an Emphasis on Titanium versus Zirconia
Abstract
:1. Introduction
| Implant Material | Common Name or Abbreviation |
|---|---|
| I. Metals | |
| Titanium | CpTi |
| Titanium Alloys | Ti-6A1-4V extra low interstitial (ELI) |
| Ti-6A1-4V | |
| Ti-6Al-7Nb | |
| Ti-5Al-2.5Fe | |
| Ti-15 Zr-4Nb-2Ta-0.2Pd | |
| Ti-29Nb-13Ta-4.6Zr | |
| Roxolid (83%–87%Ti-13%–17%Zr) | |
| Stainless Steel | SS, 316 LSS |
| Cobalt Chromium Alloy | Vitallium, Co-Cr-Mo |
| Gold Alloys | Au Alloys |
| Tantalum | Ta |
| II. Ceramics | |
| Alumina | Al2O3, polycrystalline alumina or single-crystal sapphire |
| Hydroxyapatite | HA, Ca10(PO4)10, (OH)2 |
| Beta-Tricalcium phosphate | β-TCP, Ca3(PO4)2 |
| Carbon | C |
| vitreous, | |
| low-temperature isotropic (LTI), | |
| ultra-low-temperature isotropic (ULTI) | |
| Carbon-Silicon | C-Si |
| Bioglass | SiO2/CaO/Na2O/P2O5 |
| Zirconia | ZrO2 |
| Zirconia-toughened alumina | ZTA |
| III. Polymers | |
| Polymethylmethacrylate | PMMA |
| Polytetrafluoroethylene | PTFE |
| Polyethylene | PE |
| Polysulfone | PSF |
| Polyurethane | PU |
| Polyether ether ketone | PEEK |
2. Titanium and Its Alloys
2.1. Physical and Mechanical Properties of Titanium and Its Alloys
| Material | Modulus (GPa) | Ultimate Tensile Strength (MPa) | Yield Strength (MPa) | Elongation (%) | Density (g/cc) | Type of Alloy |
|---|---|---|---|---|---|---|
| Cp Ti grade I | 102 | 240 | 170 | 24 | 4.5 | α |
| Cp Ti grade II | 102 | 345 | 275 | 20 | 4.5 | α |
| Cp Ti grade III | 102 | 450 | 380 | 18 | 4.5 | α |
| Cp Ti grade IV | 104 | 550 | 483 | 15 | 4.5 | α |
| Ti-6Al-4V- ELI | 113 | 860 | 795 | 10 | 4.4 | α + β |
| Ti-6Al-4V | 113 | 930 | 860 | 10 | 4.4 | α + β |
| Ti-6Al-7Nb | 114 | 900–1050 | 880–950 | 8–15 | 4.4 | α + β |
| Ti-5Al-2.5Fe | 112 | 1020 | 895 | 15 | 4.4 | α + β |
| Ti-15Zr-4Nb-2Ta-0.2Pd | 94–99 | 715–919 | 693–806 | 18–28 | 4.4 | α + β |
| Ti-29Nb-13Ta-4.6Zr | 80 | 911 | 864 | 13.2 | 4.4 | β |
2.2. Titanium Sensitivity Associated with Dental Implants
2.3. Failure Mode of Titanium

3. Ceramics
3.1. Ceramics as Dental Implant Coatings
| Material | Chemical Composition |
|---|---|
| Hydroxylapatite (HA) | Ca10(PO4)6(OH)2 |
| Tricalcium phosphate (TCP) | α, β,Ca3(PO4)2 |
| Fluorapatite (FA) | Ca10(PO4)6F2 |
| Tetracalcium phosphate | Ca4P2O9 |
| Calcium pyrophosphate | Ca4P2O7 |
| Brushite | CaHPO4, CaHPO4·2H2O |
| Bioglasses | SiO2-CaO-Na2O-P205-MgO, etc. |
| Aluminium oxide | Al2O3 |
| Zirconium oxide | ZrO2 |
3.2. Ceramics as Dental Implant Materials
3.2.1. Mechanical Properties of Zirconia
3.2.2. Low Temperature Degradation
3.2.3. Failure Mode of Zirconia



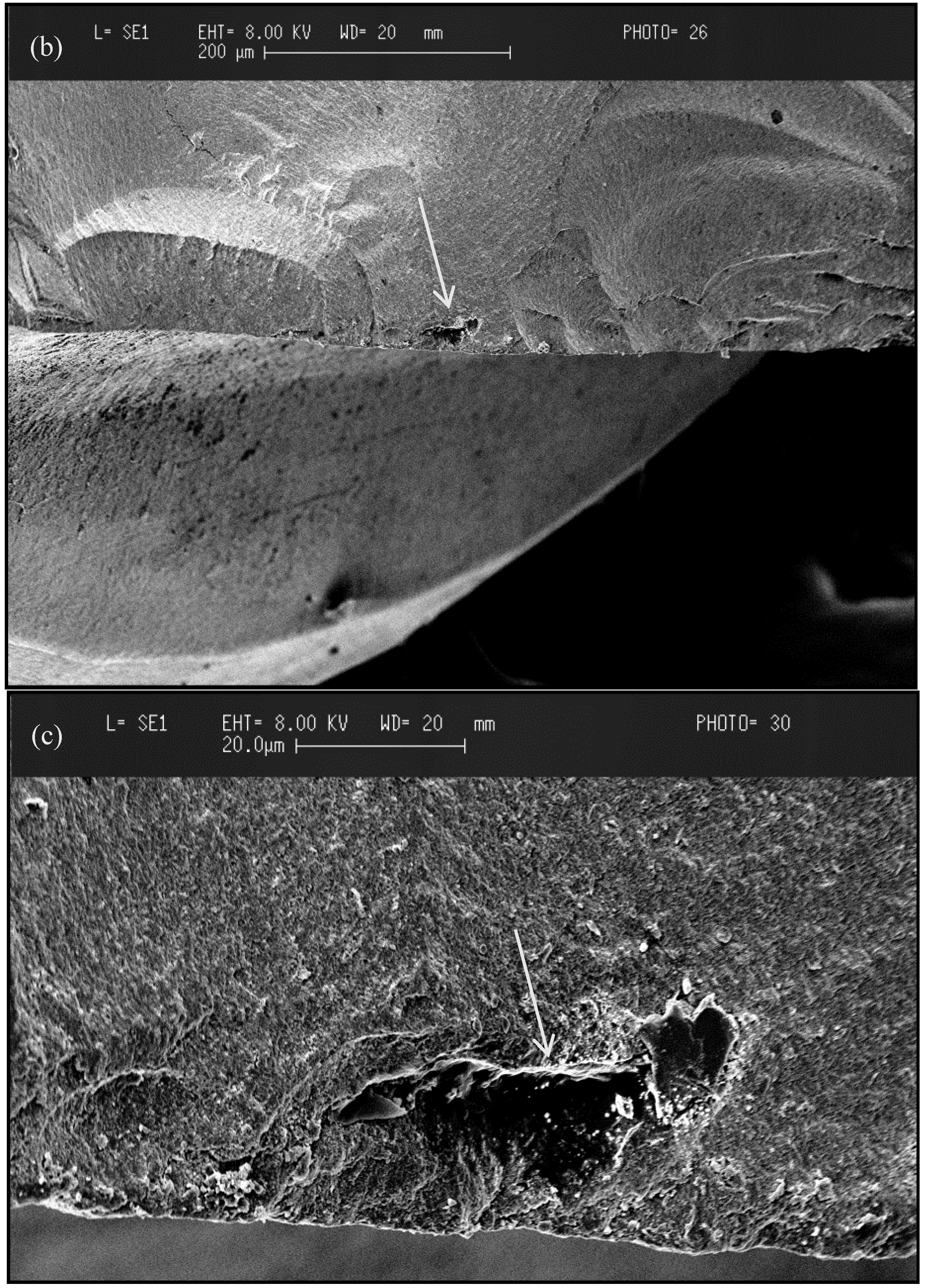
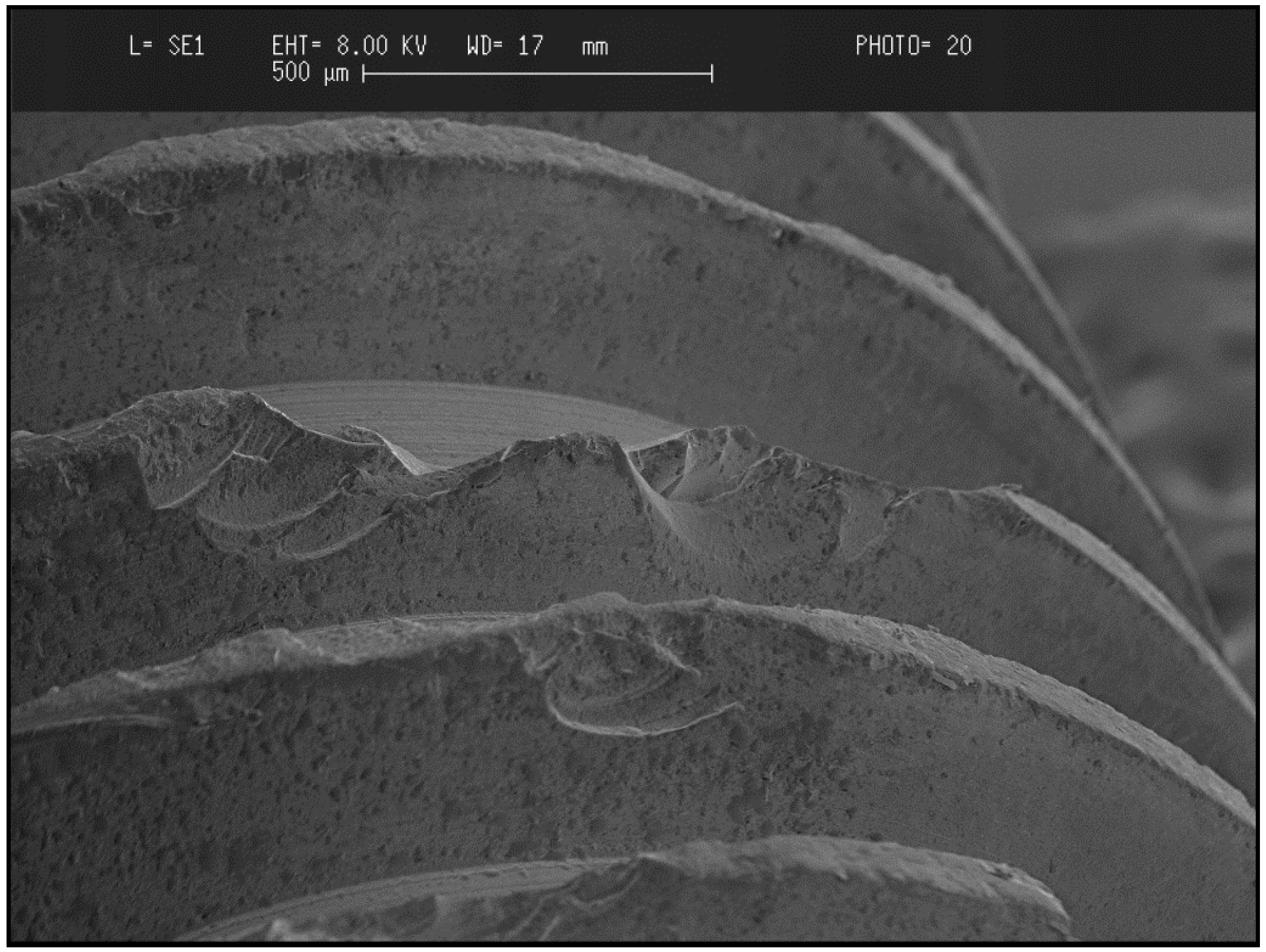
3.2.4. Types of Zirconia Used in Dentistry
(i) Yttrium-Stabilized Tetragonal Zirconia Polycrystals (3Y-TZP)
(ii) Glass-Infiltrated Zirconia-Toughened Alumina (ZTA)
(iii) Alumina Toughened Zirconia (ATZ)
4. Osseointegration of Y-TZP versus Titanium Dental Implants
5. Peri-Implant Soft Tissues around Zirconia and Titanium Implants
6. Clinical Studies, Case Reports and Case Series on Zirconia Implants
7. Discussion
Acknowledgments
Conflicts of Interest
References
- Brånemark, P.I.; Hansson, B.O.; Adell, R.; Breine, U.; Lindström, J.; Hallén, O.; Ohman, A. Osseointegrated implants in the treatment of the edentulous jaw. Experience from a 10-year period. Scand. J. Plast. Reconstr. Surg. 1977, 16, 1–132. [Google Scholar]
- Smith, D.C. Dental implants: Materials and design considerations. Int. J. Prosthodont. 1993, 6, 106–117. [Google Scholar] [PubMed]
- Parr, G.R.; Gardner, L.K.; Toth, R.W. Titanium: The mystery metal of implant dentistry. Dental materials aspect. J. Prosthet. Dent. 1985, 54, 410–414. [Google Scholar] [CrossRef] [PubMed]
- Sykaras, N.; Lacopino, A.M.; Marker, V.A.; Triplett, R.G.; Woody, R.D. Implant materials, designs, and surface topographies: Their effect on osseointegration. A literature review. Int. J. Oral Maxillofac. Implants 2000, 15, 675–690. [Google Scholar] [PubMed]
- Williams, D.E. Implants in dental and maxillofacial surgery. Biomaterials 1981, 2, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Lemons, J.E. Dental implants biomaterials. J. Am. Dent. Assoc. 1990, 121, 716–719. [Google Scholar] [CrossRef] [PubMed]
- Craig, R.G. Restorative Dental Materials, 9th ed.; C.V. Mosby: St. Louis, MO, USA, 1993; p. 169. [Google Scholar]
- Sagomonyants, K.B.; Jarman-Smith, M.L.; Devine, J.N.; Aronow, M.S.; Gronowicz, G.A. The in vitro response of human osteoblasts to polyetheretherketone (PEEK) substrates compared to commercially pure titanium. Biomaterials 2007, 24, 3115–3123. [Google Scholar]
- Berner, S.; Dard, M.; Gottlow, J.; Molenberg, A.; Wieland, M. Titanium-zirconium: A novel material for dental implants. Eur. Cells Mater. 2009, 17, 16. [Google Scholar]
- Adell, R.; Eriksson, B.; Lekholm, U.; Brånemark, P.I.; Jemt, T. A long-term follow-up study of osseointegrated implants in the treatment of totally edentulous jaws. Int. J. Oral Maxillofac. Implants 1990, 5, 347–359. [Google Scholar]
- Jemt, T.; Chai, J.; Harnett, J.; Heath, M.R.; Hutton, J.E.; Johns, R.B.; McKenna, S.; McNamara, D.C.; van Steenberghe, D.; Taylor, R.; et al. A 5-year prospective multicenter follow-up report on overdentures supported by osseointegrated implants. Int. J. Oral Maxillofac. Implants 1996, 11, 291–298. [Google Scholar] [PubMed]
- Niinomi, M. Mechanical properties of biomedical titanium alloy. Mat. Sci. Eng. A 1998, 243, 231–236. [Google Scholar] [CrossRef]
- Wataha, J.C. Materials for endosseous dental implants. J. Oral Rehabil. 1996, 23, 79–90. [Google Scholar] [CrossRef] [PubMed]
- McCracken, M. Dental implant materials: Commercially pure titanium and titanium alloys. J. Prosthodont. 1999, 8, 40–43. [Google Scholar] [CrossRef] [PubMed]
- González, J.E.G.; Mirza-Rosca, J.C. Study of the corrosion behavior of titanium and some of its alloys for biomedical and dental implant applications. J. Electroanal Chem. 1999, 471, 109–115. [Google Scholar] [CrossRef]
- Bidez, M.W.; Misch, C.F. Force transfer in implant dentistry: Basic concepts and principles. J. Oral Implantol. 1992, 18, 264–274. [Google Scholar] [PubMed]
- Odin, G.; Savoldelli, C.; Bouchard, P.O.; Tillier, Y. Determination of Young’s modulus of mandibular bone using inverse analysis. Med. Eng. Phys. 2010, 32, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Grandin, H.M.; Berner, S.; Dart, M. A review of titanium zirconium (TiZr) alloys for use in endosseous dental implants. Materials 2015, 5, 1348–1360. [Google Scholar] [CrossRef]
- Karl, M.; Krafft, T.; Kelly, J.R. Fracture of a narrow-diameter Roxolid implant: Clinical and fractographic considerations. Int. J. Oral Maxillofac. Implants 2014, 29, 1193–1196. [Google Scholar] [CrossRef] [PubMed]
- Olmedo, D.G.; Paparella, M.L.; Brandizzi, D.; Cabrini, R.L. Reactive lesions of peri-implant mucosa associated with titanium dental implants: A report of 2 cases. Int. J. Oral Maxillofac. Surg. 2010, 39, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Egusa, H.; Ko, N.; Shimazu, T.; Yatani, H. Suspected association of an allergic reaction with titanium dental implants: A clinical report. J. Prosthet. Dent. 2008, 100, 344–347. [Google Scholar] [CrossRef] [PubMed]
- Flatebø, R.S.; Johannessen, A.C.; Grønningsæter, A.G.; Bøe, O.E.; Gjerdet, N.R.; Grung, B.; Leknes, K.N. Host response to titanium dental implant placement evaluated in a human oral model. J. Periodontol. 2006, 77, 1201–1210. [Google Scholar] [CrossRef]
- Sicilia, A.; Cuesta, S.; Coma, G.; Arregui, I.; Guisasola, C.; Ruiz, E.; Maestro, A. Titanium allergy in dental implant patients: A clinical study on 1500 consecutive patients. Clin. Oral Implants Res. 2008, 19, 823–835. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, T.P. An overview of the corrosion aspect of dental implants (titanium and its alloys). Indian J. Dent. Res. 2009, 20, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, A.; Payne, A.G.T.; de Silva, R.K.; Duncan, W.J. Titanium allergy: Could it affect dental implant integration? Clin. Oral Implants Res. 2011, 22, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Javed, F.; Al-Hezaimi, K.; Almas, K.; Romanos, G.E. Is titanium sensitivity associated with allergic reactions in patients with dental implants? A systematic review. Clin. Implant Dent. Relat. Res. 2013, 15, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Hallab, N.; Merritt, K.; Jacobs, J.J. Metal sensitivity in patients with orthopaedic implants. J. Bone Joint Surg. Am. 2001, 83, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Frisken, K.W.; Dandie, G.W.; Lugowski, S.; Jordan, G. A study of titanium release into body organs following the insertion of single threaded screw implants into the mandibles of sheep. Aust. Dent. J. 2002, 47, 214–217. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, J.J.; Skipor, A.K.; Black, J.; Urban, R.; Galante, J.O. Release and excretion of metal in patients who have a total hip replacement component made of titanium- base alloy. J. Bone Joint Surg. Am. 1991, 73, 1475–1486. [Google Scholar] [PubMed]
- Witt, J.D.; Swann, M. Metal wear and tissue response in failed titanium alloy total hip replacements. J. Bone Joint Surg. Br. 1991, 73, 559–563. [Google Scholar] [PubMed]
- Tamai, K.; Mitsumori, M.; Fujishiro, S.; Kokubo, M.; Ooya, N.; Nagata, Y.; Sasai, K.; Hiraoka, M.; Inamoto, T. A case of allergic reaction to surgical metal clips inserted for postoperative boost irradiation in a patient undergoing breast conserving therapy. Breast Cancer 2001, 8, 90–92. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.; Bandl, W.D.; Maier, S.; Summer, B.; Przybilla, B. Hypersensitivity to titanium osteosynthesis with impaired fracture healing, eczema, and T-cell hyper responsiveness in vitro: Case report and review of the literature. Contact Dermat. 2006, 55, 199–202. [Google Scholar] [CrossRef]
- Haug, R.H. Retention of asymptomatic bone plates used for orthognathic surgery and facial fractures. J. Oral Maxillofac. Surg. 1996, 54, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Schramm, M.; Pitto, R.P. Clinical relevance of allergological tests in total hip joint replacement. In Bioceramics in Hip Joint Replacement; Willmann, G., Zweymüller, K., Eds.; Thieme: New York, NY, USA, 2000; pp. 101–106. [Google Scholar]
- Bass, J.K.; Fine, H.; Cisneros, G.J. Nickel hypersensitivity in the orthodontic patient. Am. J. Orthod. Dentofac. Orthop. 1993, 103, 280–285. [Google Scholar] [CrossRef]
- Mitchell, D.L.; Synnott, S.A.; van Dercreek, J.A. Tissue reaction involving an intraoral skin graft and CP titanium abutments: A clinical report. Int. J. Oral Maxillofac. Implants 1990, 5, 79–84. [Google Scholar] [PubMed]
- Du Preez, L.A.; Bütow, K.W.; Swart, T.J. Implant failure due to titanium hypersensitivity/allergy?—Report of a case. SADJ 2007, 62, 22–25. [Google Scholar]
- Müller, K.; Valentine-Thon, E. Hypersensitivity to titanium: Clinical and laboratory evidence. Neuro. Endocrinol. Lett. 2006, 27, 31–35. [Google Scholar]
- Chaturvedi, T.P. Allergy related to dental implant and its clinical significance. Clin. Cosmet. Investig. Dent. 2013, 5, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Balshi, T.H. An analysis and management of fractured implants: A clinical report. Int. J. Oral Maxillofac. Implants 1996, 11, 660–666. [Google Scholar] [PubMed]
- Tolman, D.E.; Laney, W.R. Tissue integrated prosthesis complications. Int. J. Oral Maxillofac. Implants 1992, 7, 477–484. [Google Scholar] [PubMed]
- Jemt, T.; Lekholm, U. Oral implant treatment in posterior partially edentulous jaws: A 5-year follow-up report. Int. J. Oral Maxillofac. Implants 1993, 8, 635–640. [Google Scholar] [PubMed]
- Zarb, G.; Schmitt, A. The longitudinal clinical effectiveness of osseointegrated dental implants: The Toronto study. Part III: Problems and complications encountered. J. Prosthet. Dent. 1990, 64, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Piattelli, A.; Scarano, A.; Piattelli, M.; Vaia, E.; Matarasso, S. Hollow implants retreived for fracture: A light and scanning electron microscope analysis of 4 cases. J. Periodontol. 1998, 69, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Piattelli, A.; Piattelli, M.; Scarano, M.; Montesani, L. Light and scanning electron microscopic report of four fractured implants. Int. J. Oral Maxillofac. Implants 1998, 13, 561–564. [Google Scholar] [PubMed]
- Eckert, S.E.; Meraw, S.J.; Cal, E.; Ow, R.K. Analysis of incidence and associated factors with fractured implants: A retrospective study. Int. J. Oral Maxillofac. Implants 2000, 15, 662–667. [Google Scholar] [PubMed]
- Green, N.T.; Machtei, E.E.; Horwitz, J.; Peled, M. Fracture of dental implants: Literature review and report of a case. Implant Dent. 2002, 11, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Patterson, E.A.; Johns, R.B. Theoretical analysis of the fatigue life of fixture screws in oseeointegrated dental implants. Int. J. Oral Maxillofac. Implants 1992, 7, 26–34. [Google Scholar] [PubMed]
- Morgan, M.J.; James, D.F.; Pilliar, R.M. Fractures of fixture component of an oseeointegrated implant. Int. J. Oral Maxillofac. Implants 1993, 8, 409–414. [Google Scholar] [PubMed]
- Virdee, P.; Bishop, K. A review of the aetiology and management of fractured dental implants and a case report. Br. Dent. J. 2007, 203, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, K.; Ichikawa, T.; Murakami, H.; Miyamoto, Y.; Asaoka, K. Fracture mechanisms of retrieved titanium screw thread in dental implant. Biomaterials 2002, 23, 2459–2465. [Google Scholar] [CrossRef] [PubMed]
- Lacefield, W.R. Current status of ceramic coatings for dental implants. Implant Dent. 1998, 7, 315–322. [Google Scholar] [CrossRef] [PubMed]
- De Groot, K.; Wolke, J.C.G.; Jansen, J.A. State of the art: Hydroxylapatite coatings for dental implants. J. Oral Implant. 1994, 20, 232–234. [Google Scholar]
- Morris, H.F.; Ochi, S.; Spray, J.R.; Olson, J.W. Periodontal-type measurements associated with hydroxyapatite-coated and non-HA-coated implants: Uncovering to 36 months. Ann. Periodontol. 2000, 5, 56–67. [Google Scholar] [CrossRef] [PubMed]
- Barrere, F.; van der Valk, C.M.; Meijer, G.; Dalmeijer, R.A.; de Groot, K.; Layrolle, P. Osteointegration of biomimetic apatite coating applied onto dense and porous metal implants in femurs of goats. J. Biomed. Mater. Res. 2003, 67, 655–665. [Google Scholar] [CrossRef]
- Le Guéhennec, L.; Soueidan, A.; Layrolle, P.; Amouriq, Y. Surface treatments of titanium dental implants for rapid osseointegration. J. Dent. Mater. 2007, 23, 844–854. [Google Scholar] [CrossRef]
- Wheeler, S. Eight-year clinical retrospective study of titanium plasma-sprayed and hydroxyapatite-coated cylinder implants. Int. J. Oral Maxillofac. Implants 1996, 11, 340–350. [Google Scholar] [PubMed]
- Chang, Y.-L.; Lew, D.; Park, J.B.; Keller, J.C. Biomechanical and morphometric analysis of hydroxyapatite-coated implants with varying crystallinity. J. Oral Maxillofac. Surg. 1999, 57, 1096–1108. [Google Scholar] [CrossRef] [PubMed]
- Tinsley, D.; Watson, C.; Russell, J. A comparison of hydroxyapatite coated implant retained fixed and removable mandibular prostheses over 4 to 6 years. Clin. Oral Implants Res. 2001, 12, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Kohal, R.J.; Att, W.; Bächle, M.; Butz, F. Ceramic abutments and ceramic oral implants. An update. Periodontol. 2000 2008, 47, 224–243. [Google Scholar] [CrossRef] [PubMed]
- Black, J.; Hastings, G. Oxide bioceramics: Inert ceramic materials in medicine and dentistry. In Handbook of Biomaterial Properties, 1st ed.; Chapman & Hall: London, UK, 1998; p. 340. [Google Scholar]
- Denry, I.; Kelly, J.R. State of the art of zirconia for dental applications. Dent. Mater. 2008, 24, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Piconi, C.; Maccauro, G. Zirconia as a ceramic biomaterial. Biomaterials 1999, 20, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Kohal, R.J.; Klaus, G.; Strub, J.R. Zirconia-implant supported all-ceramic crowns withstand long-term load: A pilot investigation. Clin. Oral Implants Res. 2006, 17, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Kohal, R.J.; Wolkewitz, M.; Tsakona, A. The effects of cyclic loading and preparation on the fracture strength of zirconium-dioxide implants: An in vitro investigation. Clin. Oral Implants Res. 2011, 22, 808–814. [Google Scholar] [CrossRef] [PubMed]
- Silva, N.; Coelho, P.G.; Fernandes, C.; Navarro, J.M.; Dias, R.A.; Thompson, V.P. Reliability of one-piece ceramic implant. Biomed. Mater. Res. B App. Biomater. 2009, 88, 419–426. [Google Scholar] [CrossRef]
- Kohal, R.J.; Finke, H.C.; Klaus, G. Stability of prototype two piece zirconia and titanium implants after artificial aging: An in vitro pilot study. Clin. Implant Dent. Relat. Res. 2009, 11, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Garvie, R.; Hannink, R.; Pascoe, R. Ceramic steel? Nature 1975, 258, 703–704. [Google Scholar] [CrossRef]
- Chevalier, J.; Loh, J.; Gremillard, L.; Meille, S.; Adolfson, E. Low-temperature degradation in zirconia with a porous surface. Acta Biomater. 2011, 7, 2986–2993. [Google Scholar] [CrossRef] [PubMed]
- Lawson, S. Environmental degradation of zirconia ceramics. J. Eur. Ceram. Soc. 1995, 15, 485–502. [Google Scholar] [CrossRef]
- Chevalier, J. What future for zirconia as a biomaterial? Biomaterials 2006, 27, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Iio, S.; Fukuura, I. Ageing behaviour of Y-TZP. In Science and Technology of Zirconia II, Advances in Ceramics; Claussen, N., Ruhle, M., Heuer, A.H., Eds.; The American Ceramic Society, Inc.: Columbus, OH, USA, 1984; Volume 12, pp. 391–398. [Google Scholar]
- Att, W.; Grigoriadou, M.; Strub, J.R. ZrO2 three-unit fixed partial dentures: Comparison of failure load before and after exposure to a mastication simulator. J. Oral Rehabil. 2007, 34, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Alghazzawi, T.F.; Lemons, J.; Liu, P.R.; Essig, M.E.; Bartolucci, A.A.; Janowski, G.M. Influence of low temperature environmental exposure on the mechanical properties and structural stability of dental zirconia. J. Prosthodont. 2012, 21, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Samodurova, A.; Andraž, K.; Swain, M.V.; Tomaž, K. The combined effect of alumina and silica co-doping on the ageing resistance of 3Y-TZP bioceramics. Acta Biomater. 2014, 11, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Piconi, C.; Burger, W.; Richter, H.G.; Cittadini, A.; Maccauro, G.; Covacci, V.; Bruzzese, N.; Ricci, G.A.; Marmo, E. Y-TZP for artificial joint replacements. Biomaterials 1998, 19, 1489–1494. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.K.; Tandon, R.; Ready, M.J.; Lawn, B.R. Scratch damage on zirconia ceramics. J. Am. Cream. Soc. 2000, 83, 1428–1432. [Google Scholar]
- Osman, R.B.; Ma, S.; Duncan, W.; de Silva, R.K.; Siddiqi, A.; Swain, M.V. Fractured Zirconia implants and related implant designs: Scanning electron microscopy analysis. Clin. Oral Implants Res. 2013, 24, 592–597. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sailer, I.; Lawn, B.R. Fatigue of dental ceramics. J. Dent. 2013, 41, 1135–1147. [Google Scholar] [CrossRef] [PubMed]
- Gahlert, M.; Burtscher, D.; Grunert, I.; Kniha, H.; Steinhauser, E. Failure analysis of fractured dental zirconia implants. Clin. Oral Implants Res. 2012, 23, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Sanon, C.; Chevalier, J.; Douillard, T.; Cattani-Lorente, M.; Scherrer, S.S.; Gremillard, L. A new testing protocol for zirconia dental implants. Dent. Mater. 2015, 31, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, L.; Readey, M.J. Effect of heat-treatment on grain size, phase assemblage, and mechanical properties of 3 mol % Y-TZP. J. Am. Ceram. Soc. 1996, 79, 2331–2340. [Google Scholar] [CrossRef]
- Heuer, A.H.; Claussen, N.; Kriven, W.M.; Ruhle, M. Stability of tetragonal ZrO2 particles in ceramic matrices. J. Am. Ceram. Soc. 1982, 65, 642–650. [Google Scholar] [CrossRef]
- Cottom, B.A.; Mayo, M.J. Fracture toughness of nanocrystalline ZrO2–3 mol % Y2O3 determined by Vickers indentation. Scr. Mater. 1996, 34, 809–814. [Google Scholar] [CrossRef]
- Filser, F.; Kocher, P.; Gauckler, L.J. Net-shaping of ceramic components by direct ceramic machining. Assembly Autom. 2003, 23, 382–390. [Google Scholar] [CrossRef]
- Ross, I.M.; Rainforth, W.M.; McComb, D.W.; Scott, A.J.; Brydson, R. The role of trace additions of alumina to yttria-tetragonal zirconia polycrystal (Y-TZP). Scr. Mater. 2001, 45, 653–660. [Google Scholar] [CrossRef]
- Li, L.F.; Watanabe, R. Influence of a small amount of Al2O3 addition on the transformation of Y2O3-partially stabilized ZrO2 during annealing. J. Mater. Sci. 1997, 32, 1149–1153. [Google Scholar] [CrossRef]
- Tsubakino, H.; Nozato, R.; Hamamoto, M. Effect of alumina addition on the tetragonal-to-monoclinic phase transformation in zirconia–3 mol % yttria. J. Am. Ceram. Soc. 1991, 74, 440–443. [Google Scholar] [CrossRef]
- Guazzato, M.; Albakry, M.; Quach, L.; Swain, M.V. Influence of grinding, sandblasting, polishing and heat treatment on the flexural strength of a glass-infiltrated alumina-reinforced dental ceramic. Biomaterials 2004, 25, 2153–2160. [Google Scholar] [CrossRef] [PubMed]
- Guazzato, M.; Albakry, M.; Swain, M.V.; Ringer, S.P. Microstructure of alumina-and alumina/zirconia-glass infiltrated dental ceramics. Bioceramics 2003, 15, 879–882. [Google Scholar]
- Guazzato, M.; Albakry, M.; Ringer, S.P.; Swain, M.V. Strength, fracture toughness and microstructure of a selection of all-ceramic materials. Part II. Zirconia-based dental ceramics. Dent. Mater. 2004, 20, 449–456. [Google Scholar] [CrossRef] [PubMed]
- Pabst, W.; Havrda, E.; Gregorová, E.; Krčmová, B. Alumina toughened zirconia made by room temperature extrusion of ceramic pastes. J. Am. Ceram. Soc. 2000, 44, 41–47. [Google Scholar]
- Akagawa, Y.; Ichikawa, Y.; Nikai, H.; Tsuru, H. Interface histology of unloaded and early loaded partially stabilized zirconia endosseous implant in initial bone healing. J. Prosthet. Dent. 1993, 69, 599–604. [Google Scholar] [CrossRef] [PubMed]
- Akagawa, Y.; Hosok, R.; Sato, Y.; Kamayama, K. Comparison between freestanding and tooth-connected partially stabilized zirconia implants after two years function in monkeys: A clinical and histologic study. J. Prosthet. Dent. 1998, 80, 551–558. [Google Scholar] [CrossRef] [PubMed]
- Kohal, R.J.; Weng, D.; Bӓchle, M.; Strub, J.R. Loaded custom-made zirconia and titanium implants show similar osseointegration: An animal experiment. J. Periodontol. 2004, 75, 1262–1268. [Google Scholar] [CrossRef] [PubMed]
- Dubruille, J.H.; Viguier, E.; Le Naour, G.; Dubruille, M.T.; Auriol, M.; Le Charpentier, Y. Evaluation of combinations of titanium, zirconia, and alumina implants with 2 bone fillers in the dog. Int. J. Oral Maxillofac. Implants 1999, 14, 271–277. [Google Scholar] [PubMed]
- Scarano, A.; Di Carlo, F.; Quaranta, M.; Piattelli, A. Bone response to zirconia ceramic implants: An experimental study in rabbits. J. Oral Implantol. 2003, 29, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Depprich, R.; Zipprich, H.; Ommerborn, M.; Mahn, E.; Lammers, L.; Handschel, J.; Naujoks, C.; Wiesmann, H.P.; Kübler, N.R.; Meyer, U. Osseointegration of zirconia implants: An SEM observation of the bone-implant interface. Head Face Med. 2008, 6, 4–25. [Google Scholar]
- Hoffmann, O.; Angelov, N.; Gallez, F.; Jung, R.E.; Weber, F.E. The zirconia implant-bone interface: A preliminary histologic evaluation in rabbits. Int. J. Oral Maxillofac. Implants 2008, 23, 691–695. [Google Scholar] [PubMed]
- Gahlert, M.; Roehling, S.; Sprecher, C.M.; Kniha, H.; Milz, S.; Bormann, K. In vivo performance of zirconia and titanium implants: A histomorphometric study in mini pig maxillae. Clin. Oral Implants Res. 2012, 23, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Schultze-Mosgau, S.; Schliephake, H.; Radespiel-Troger, M.; Neukam, F.W. Osseointegration of endodontic endosseous cones: Zirconium oxide vs titanium. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2000, 89, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Sennerby, L.; Dasmah, A.; Larsson, B.; Iverhed, M. Bone tissue response to surface modified zirconia implants: A histomorphometric and removal torque study in the rabbit. Clin. Implant Dent. Relat. Res. 2005, 7, S13–S20. [Google Scholar] [CrossRef] [PubMed]
- Rocchietta, I.; Fontana, F.; Addis, A.; Schupbach, P.; Simon, M. Surface-modified zirconia implants: Tissue response in rabbits. Clin. Oral Implants Res. 2009, 20, 844–850. [Google Scholar] [CrossRef] [PubMed]
- Gahlert, M.; Gudehus, T.; Eichhorn, S.; Steinhauser, E.; Kniha, H.; Erhardt, W. Biomechanical and histomorphometric comparison between zirconia implants with varying surface textures and a titanium implant in the maxilla of miniature pigs. Clin. Oral Implants Res. 2007, 18, 662–668. [Google Scholar] [CrossRef] [PubMed]
- Schliephake, H.; Hefti, T.; Schlottig, F.; Gédet, P.; Staedt, H. Mechanical anchorage and peri-implant bone formation of surface-modified zirconia in minipigs. J. Clin. Periodontol. 2010, 37, 818–828. [Google Scholar] [CrossRef] [PubMed]
- Bächle, M.; Butz, F.; Hübner, U.; Bakalinis, E.; Kohal, R.J. Behavior of CAL72 osteoblast-like cells cultured on zirconia ceramics with different surface topographies. Clin. Oral Implants Res. 2007, 18, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Hempel, U.; Hefti, T.; Kalbacova, M.; Wolf-Brandstetter, C.; Dieter, P.; Schlottig, F. Response of osteoblast-like SAOS-2 cells to zirconia ceramics with different surface topographies. Clin. Oral Implants Res. 2010, 21, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Hao, L.; Lawrence, J.; Chian, K.S. Osteoblast cell adhesion on a laser modified zirconia based bioceramic. J. Mater. Sci. Mater. Med. 2005, 16, 719–726. [Google Scholar] [CrossRef] [PubMed]
- Tuna, T.; Wein, M.; Swain, M.; Fischer, J.; Att, W. Influence of ultraviolet photofunctionalization on the surface characteristics of zirconia-based dental implant materials. Dent. Mater. 2015, 31, e14–e24. [Google Scholar] [CrossRef] [PubMed]
- Aldini, N.N.; Fini, M.; Giavaresi, G.; Martini, L.; Dubini, B.; Ponzi-Bossi, M.G.; Rustichelli, F.; Krajewski, A.; Ravaglioli, A.; Mazzocchi, M.; et al. Osteointegration of bioactive glass coated and uncoated zirconia in osteopenic bone: An in vivo experimental study. J. Biomed. Mater. Res. 2004, 68A, 264–272. [Google Scholar] [CrossRef]
- Ferguson, S.J.; Langhoff, J.D.; Voelter, K.; von Rechenberg, B.; Scharnweber, D.; Bierbaum, S.; Schnabelrauch, M.; Kautz, A.R.; Frauchiger, V.M.; Mueller, T.L.; et al. Biomechanical comparison of different surface modifications for dental implants. Int. J. Oral Maxillofac. Implants 2008, 23, 1037–1046. [Google Scholar] [PubMed]
- Payer, M.; Lorenzoni, M.; Jakse, N.; Kirmeier, R.; Dohr, G.; Stopper, M.; Pert, C. Cell growth on different zirconia and titanium surface textures: A morphologic in vitro study. Z. Zahnӓrztl. Implants 2010, 26, 20–33. [Google Scholar]
- Zinelis, S.; Thomas, A.; Syres, K.; Silikas, N.; Eliades, G. Surface characterization of zirconia dental implants. Dent. Mater. 2010, 26, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.S.; Chung, S.H.; Shon, W.J. Peri-implant bone formation and surface characteristics of rough surface zirconia implants manufactured by powder injection molding technique in rabbit tibiae. Clin. Oral Implants Res. 2013, 24, 586–591. [Google Scholar] [CrossRef] [PubMed]
- Manzano, G.; Herrero, L.R.; Montero, J. Comparison of clinical performance of zirconia implants and titanium implants in animal models: A systematic review. Int. J. Oral Maxillofac. Implants 2014, 29, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Mellinghoff, J. Quality of the peri-implant soft tissue attachment of zirconia implants-abutments. Z. Zahnärztl. Implants 2010, 26, 62–71. [Google Scholar]
- Pae, A.; Lee, H.; Kim, H.S.; Kwon, Y.D.; Woo, Y.H. Attachment and growth behavior of human gingival fibroblasts on titanium and zirconia ceramic surfaces. Biomed. Mater. 2009, 2, 025005–025012. [Google Scholar] [CrossRef]
- Rimondini, L.; Cerroni, L.; Carrassi, A.; Torricelli, P. Bacterial colonization of zirconia ceramic surfaces: An in vitro and in vivo Study. Int. J. Oral Maxillofac. Implants 2002, 17, 793–798. [Google Scholar] [PubMed]
- Scarano, A.; Piattelli, M.; Caputi, S.; Favero, G.A.; Piattelli, A. Bacterial adhesion on commercially pure titanium and zirconium oxide discs: An in vivo human study. J. Periodontol. 2004, 75, 292–296. [Google Scholar] [CrossRef] [PubMed]
- Degidi, M.; Artese, L.; Scarano, A.; Perrotti, V.; Gehrke, P.; Piattelli, A. Inflammatory infiltrate, microvessel density, nitric oxide synthase expression, vascular endothelial growth factor expression, and proliferative activity in peri-implant soft tissues around titanium and zirconium oxide healing caps. J. Periodontol. 2006, 77, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Größner-Schreiber, B.; Herzog, M.; Hedderich, J.; Dück, A.; Hannig, M.; Griepentrog, M. Focal adhesion contact formation by fibroblasts cultured on surface-modified dental implants: An in vitro study. Clin. Oral Implants Res. 2006, 17, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Welander, M.; Abrahamsson, I.; Berglundh, T. The mucosal barrier at implant abutments of different materials. Clin. Oral Implants Res. 2008, 19, 635–641. [Google Scholar] [PubMed]
- Lima, E.M.C.X.; Koo, H.; Vacca-Smith, A.M.; Rosalen, P.L.; Del Bel Cury, A.A. Adsorption of salivary and serum proteins, and bacterial adherence on titanium and zirconia ceramic surfaces. Clin. Oral Implants Res. 2008, 19, 780–785. [Google Scholar] [CrossRef] [PubMed]
- Al-Ahmad, A.; Wiedmann-Al-Ahmad, M.; Faust, J.; Bӓchle, M.; Follo, M.; Wolkewitz, M.; Hannig, C.; Hellwig, E.; Carvalho, C.; Kohal, R. Biofilm formation and composition on different implant materials in vivo. J. Biomed. Mater. Res. B Appl. Biomater. 2010, 95B, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Depprich, R.; Naujoks, C.; Ommerborn, M.; Schwarz, F.; Kübler, N.R.; Handschel, J. Current findings regarding zirconia implants. Clin. Implant Dent. Relat. Res. 2014, 16, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Nevins, M.; Camelo, M.; Nevins, M.L.; Schupbach, P.; Kim, D.M. Pilot clinical and histologic evaluations of a two-piece zirconia implant. Int. J. Periodontics Restor. 2011, 31, 157–163. [Google Scholar]
- Kohal, R.J.; Klaus, G. A zirconia implant-crown system: A case report. Int. J. Periodontics Restor. 2004, 24, 147–153. [Google Scholar]
- Oliva, J.; Oliva, X.; Oliva, J.D. Ovoid zirconia implants: Anatomic design for premolar replacement. Int. J. Periodontics Restor. 2008, 28, 609–615. [Google Scholar]
- Oliva, J.; Oliva, X.; Oliva, J.D. Zirconia implants and all ceramic restorations for esthetic replacement of the maxillary central incisors. Eur. J. Esthet. Dent. 2008, 3, 174–185. [Google Scholar] [PubMed]
- Oliva, X.; Oliva, J.; Oliva, J.D. Replacement of congenitally missing maxillary permanent canine with a zirconium oxide dental implant and crown. A case report from an ongoing clinical study. Oral Surg. 2008, 1, 140–144. [Google Scholar] [CrossRef]
- Pirker, W.; Kocher, A. Immediate, non-submerged, root-analogue zirconia implant in single tooth replacement. Int. J. Oral Maxillofac. Surg. 2008, 37, 293–295. [Google Scholar] [CrossRef] [PubMed]
- Pirker, W.; Kocher, A. Root analog zirconia implants: True anatomical design for molar replacement-A case report. Int. J. Periodontics Restor. 2011, 31, 663–668. [Google Scholar]
- Aydin, C.; Yilmaz, H.; Ata, S.O. Single-tooth zirconia implant located in anterior maxilla. N. Y. State Dent. J. 2010, 76, 30–33. [Google Scholar] [PubMed]
- Pirker, W.; Wiedemann, D.; Lidauer, A.; Kocher, A. Immediate, single stage, truly anatomic zirconia implant in lower molar replacement: A case report with 2.5 years follow-up. Int. J. Oral Maxillofac. Surg. 2011, 40, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Borgonovo, A.E.; Corrocher, G.; Dolci, M.; Censi, R.; Vavassori, V.; Maiorana, C. Clinical evaluation of zirconium dental implants placed in esthetic areas: A case series study. Eur. J. Esthet. Dent. 2013, 8, 532–545. [Google Scholar] [PubMed]
- Payer, M.; Arnetzl, V.; Kirmeier, R.; Koller, M.; Arnetzl, G.; Jakse, N. Immediate provisional restoration of single-piece zirconia implants: A prospective case series—Results after 24 months of clinical function. Clin. Oral Implants Res. 2013, 24, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Pirker, W.; Kocher, A. Immediate, non-submerged, root-analogue zirconia implants placed into single-rooted extraction sockets: 2-year follow-up of a clinical study. Int. J. Oral Maxillofac. Surg. 2009, 3, 1127–1132. [Google Scholar] [CrossRef]
- Oliva, J.; Oliva, X.; Oliva, J. Five year success rate of 831 consecutively placed zirconia dental implants in humans: A comparison of three different rough surfaces. Int. J. Oral Maxillofac. Implants 2010, 25, 336–344. [Google Scholar] [PubMed]
- Borgonovo, A.E.; Fabbri, A.; Vavassori, V.; Censi, R.; Maiorana, C. Multiple teeth replacement with endosseous one-piece yttrium-stabilized zirconia dental implants. Med. Oral Patol. Oral Cir. Bucal 2012, 17, e981–e987. [Google Scholar] [CrossRef] [PubMed]
- Borgonovo, A.E.; Censi, R.; Vavassori, V.; Dolci, M.; Josè Luis, C.-G.; Ruiz, R.A.D.; Maiorana, C. Evaluation of the success criteria for zirconia dental implants: A four-year clinical and radiological study. Int. J. Dent. 2013, 2013. [Google Scholar] [CrossRef]
- Brüll, F.; van Winkelhoff, A.; Cune, M.S. Zirconia dental implants: A clinical, radiographic, and microbiologic evaluation up to 3 years. Int. J. Oral Maxillofac. Implants 2014, 29, 914–920. [Google Scholar] [CrossRef] [PubMed]
- Kohal, R.J.; Knauf, M.; Larsson, B.; Sahlin, H.; Butz, F. One-piece zirconia oral implants: One-year results from a prospective cohort study. 1. Single tooth replacement. J. Clin. Periodontol. 2012, 39, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Kohal, R.-J.; Patzelt, S.B.M.; Butz, F.; Sahlin, H. One-piece zirconia oral implants: One-year results from a prospective case series. 2. Three-unit fixed dental prosthesis (FDP) reconstruction. J. Clin. Periodontol. 2013, 40, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Cannizzaro, G.; Torchio, C.; Felice, P.; Leone, M.; Esposito, M. Immediate occlusal versus non-occlusal loading of single zirconia implants. A multicentre pragmatic randomized clinical trial. Eur. J. Oral Implantol. 2010, 3, 111–120. [Google Scholar] [PubMed]
- Cionca, N.; Müller, N.; Mombelli, A. Two-piece-zirconia implants supporting all-ceramic crowns. A prospective clinical study. Clin. Oral Implants Res. 2014. [Google Scholar] [CrossRef]
- Payer, M.; Heschl, A.; Koller, M.; Arnetzl, G.; Lorenzoni, M.; Jakse, N. All-ceramic restoration of zirconia two-piece implants—A randomized controlled clinical trial. Clin. Oral Implants Res. 2014. [Google Scholar] [CrossRef]
- Osman, R.B.; Morgaine, K.C.; Duncan, W.; Swain, M.V.; Ma, S. Patients’ perspectives on zirconia and titanium implants with a novel distribution supporting maxillary and mandibular overdentures: A qualitative study. Clin. Oral Implants Res. 2014, 25, 587–597. [Google Scholar] [CrossRef] [PubMed]
- Osman, R.B.; Swain, M.V.; Atieh, M.; Ma, S.; Duncan, W. Ceramic implants (Y-TZP): Are they a viable alternative to titanium implants for the support of overdentures?A randomized clinical trial. Clin. Oral Implants Res. 2014, 25, 1366–1377. [Google Scholar] [CrossRef]
- Silva, R.F.A.; Sailer, I.; Zhang, Y.; Coelho, P.G.; Guess, P.C.; Zembic, A.; Kohal, R.J. Performance of zirconia for dental healthcare. Materials 2010, 3, 863–896. [Google Scholar] [CrossRef]
- Abduo, J.; Lyons, K.; Bennamoun, M. Trends in computer-aided manufacturing in prosthodontics: A review of the available streams. Int. J. Dent. 2014, 2014, 1–15. [Google Scholar] [CrossRef]
- Wätjen, A.M.; Gingter, P.; Kramer, M.; Telle, R. Novel prospects and possibilities in additive manufacturing of ceramics by means of direct inkjet printing. Adv. Mech. Eng. 2014, 2014. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Osman, R.B.; Swain, M.V. A Critical Review of Dental Implant Materials with an Emphasis on Titanium versus Zirconia. Materials 2015, 8, 932-958. https://doi.org/10.3390/ma8030932
Osman RB, Swain MV. A Critical Review of Dental Implant Materials with an Emphasis on Titanium versus Zirconia. Materials. 2015; 8(3):932-958. https://doi.org/10.3390/ma8030932
Chicago/Turabian StyleOsman, Reham B., and Michael V. Swain. 2015. "A Critical Review of Dental Implant Materials with an Emphasis on Titanium versus Zirconia" Materials 8, no. 3: 932-958. https://doi.org/10.3390/ma8030932
APA StyleOsman, R. B., & Swain, M. V. (2015). A Critical Review of Dental Implant Materials with an Emphasis on Titanium versus Zirconia. Materials, 8(3), 932-958. https://doi.org/10.3390/ma8030932





